Submitted:
13 August 2024
Posted:
14 August 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
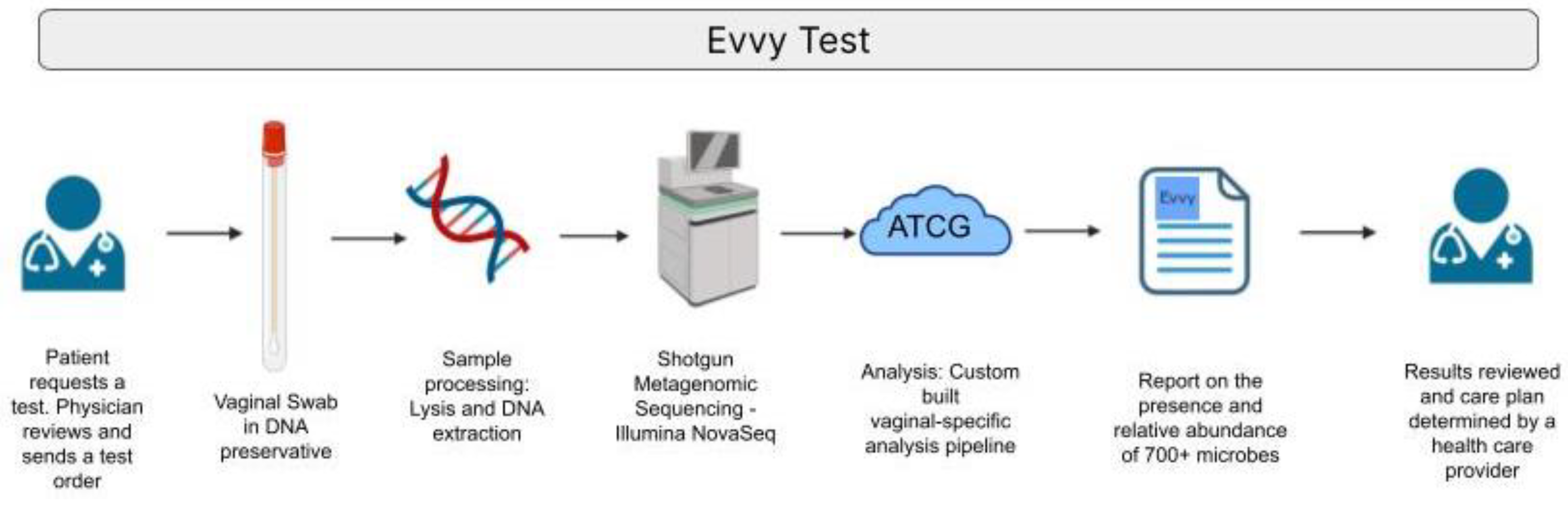
2.1. Evvy Test Workflow
2.2. Isolate Sequencing
2.3. Limit of Detection and Precision Studies
2.4. Mock Community Analysis
2.5. Study Participants, Ethics, and Sample Collection and Transportation
2.6. Shotgun Metagenomics Analysis of Vaginal Samples
3. Results
3.1. Sensitivity and Specificity
3.2. Limit of Detection and Precision
| Average Inter-assay CV | Average Intra-assay CV | |
|---|---|---|
| K. oxytoca | 1.57 | 4.30 |
| L. gasseri | 1.60 | 2.03 |
| C. albicans | 2.56 | 1.49 |
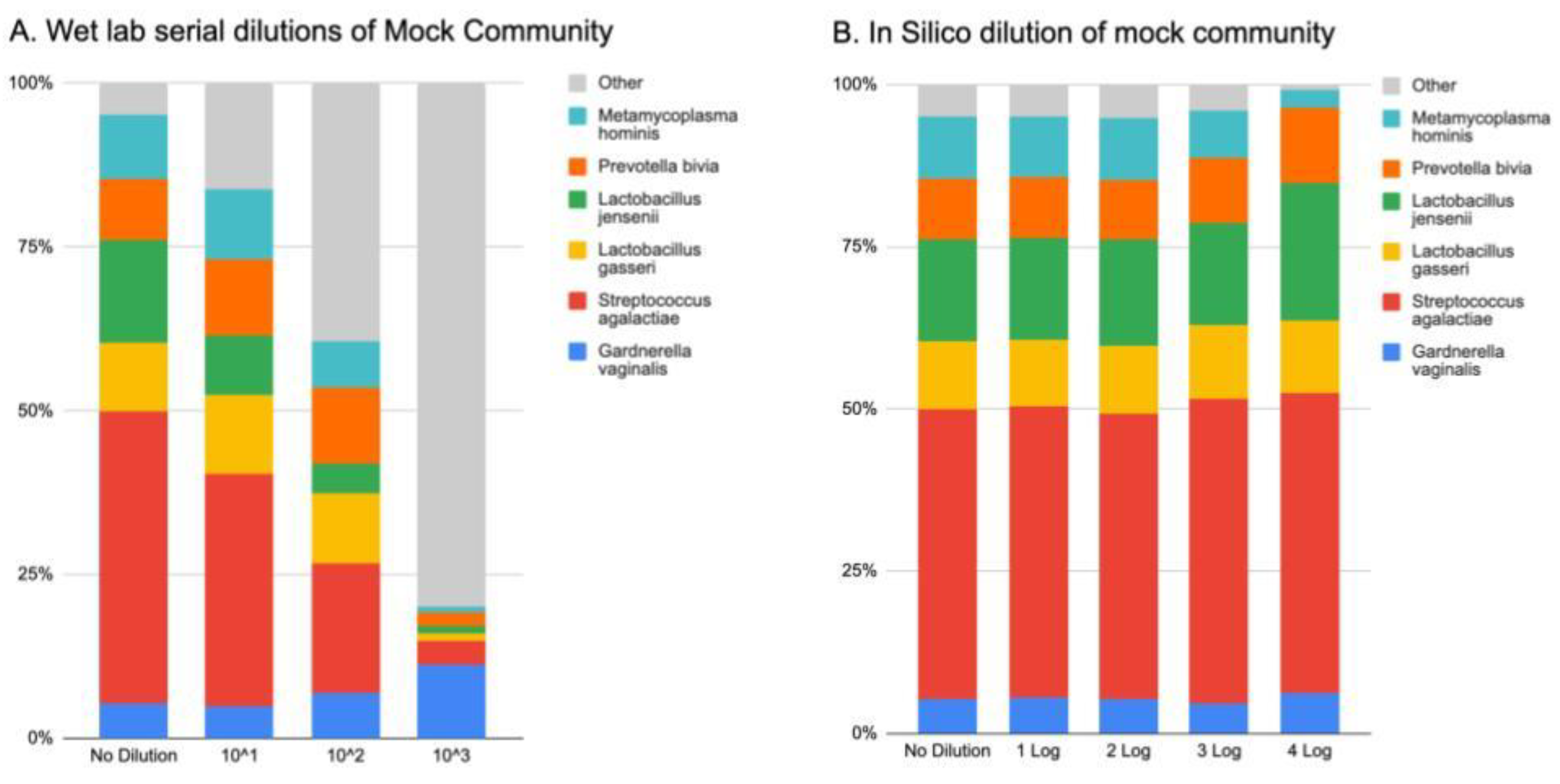
3.3. Mock Community Analysis
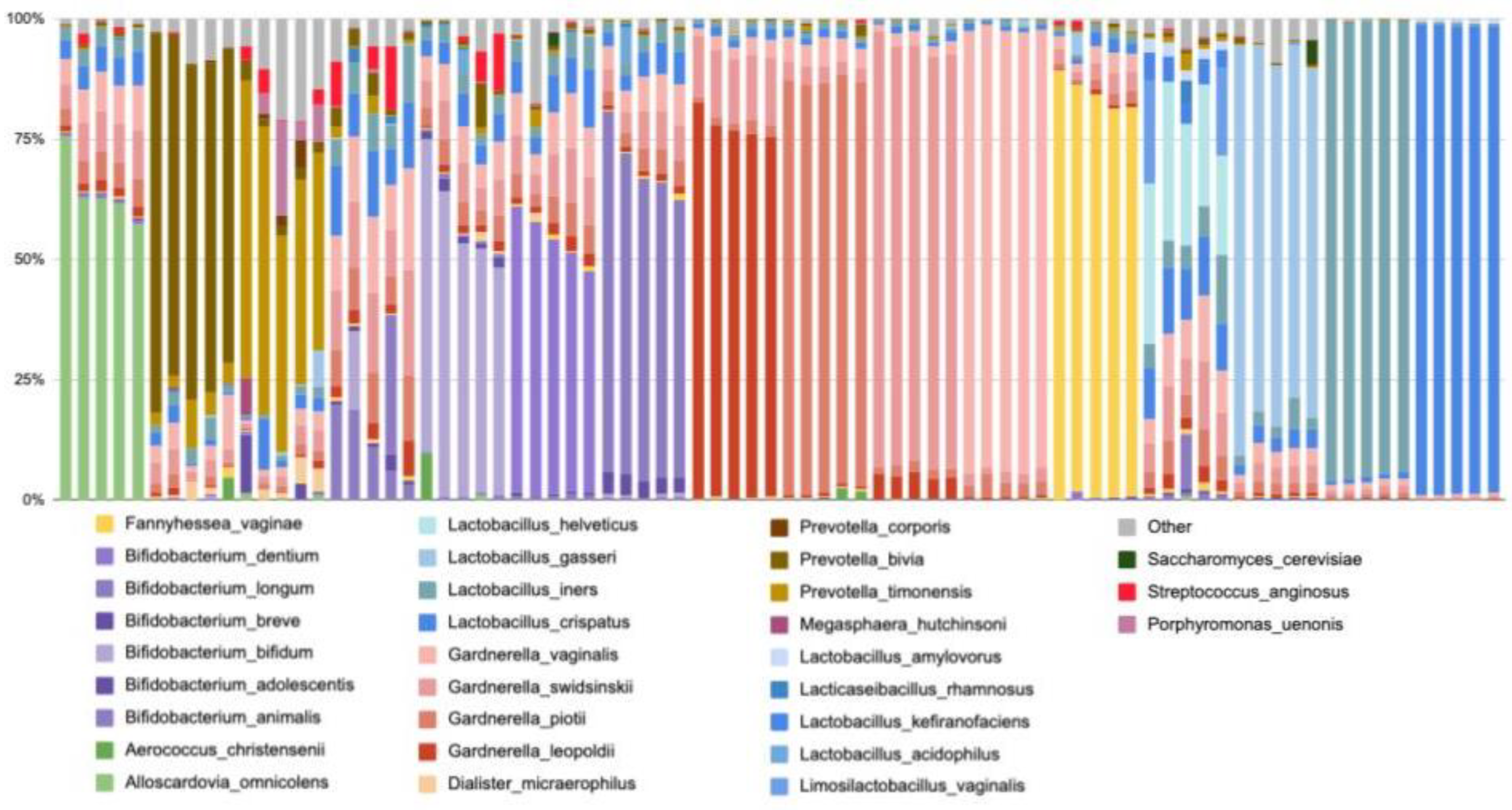
3.4.1. Findings from Evvy’s Vaginal Metagenomics Test
| Top 10 species by frequency of detection | Percentage of tests detected | Top 10 Species by relative abundance | Average Abundance | Percent of tests detected |
|---|---|---|---|---|
| Gardnerella vaginalis | 99% | Lactobacillus crispatus | 33% | 93% |
| Gardnerella swidinskii | 99% | Lactobacillus helveticus | 23% | 0.1% |
| Lactobacillus iners | 95% | Bifidobacterium dentium | 19% | 0.9% |
| Gardnerella piotii | 95% | Lactobacillus iners | 18% | 95% |
| Lactobacillus crispatus | 93% | Lactobacillus gasseri | 15% | 14% |
| Gardnerella leopoldii | 78% | Bifidobacterium longum | 13% | 2% |
| Prevotella bivia | 46% | Bifidobacterium bifidum | 13% | 0.7% |
| Fannyhessae vaginae | 33% | Gardnerella swidinskii | 12% | 99% |
| Bifidobacterium animalis | 31% | Alloscardovia omnicolens | 12% | 2% |
| Prevotella timonensis | 27% | Gardnerella vaginalis | 11% | 99% |
3.4.2. Co-occurrence of Pathogens Detected
3.4.3. Comparison to US Census Data
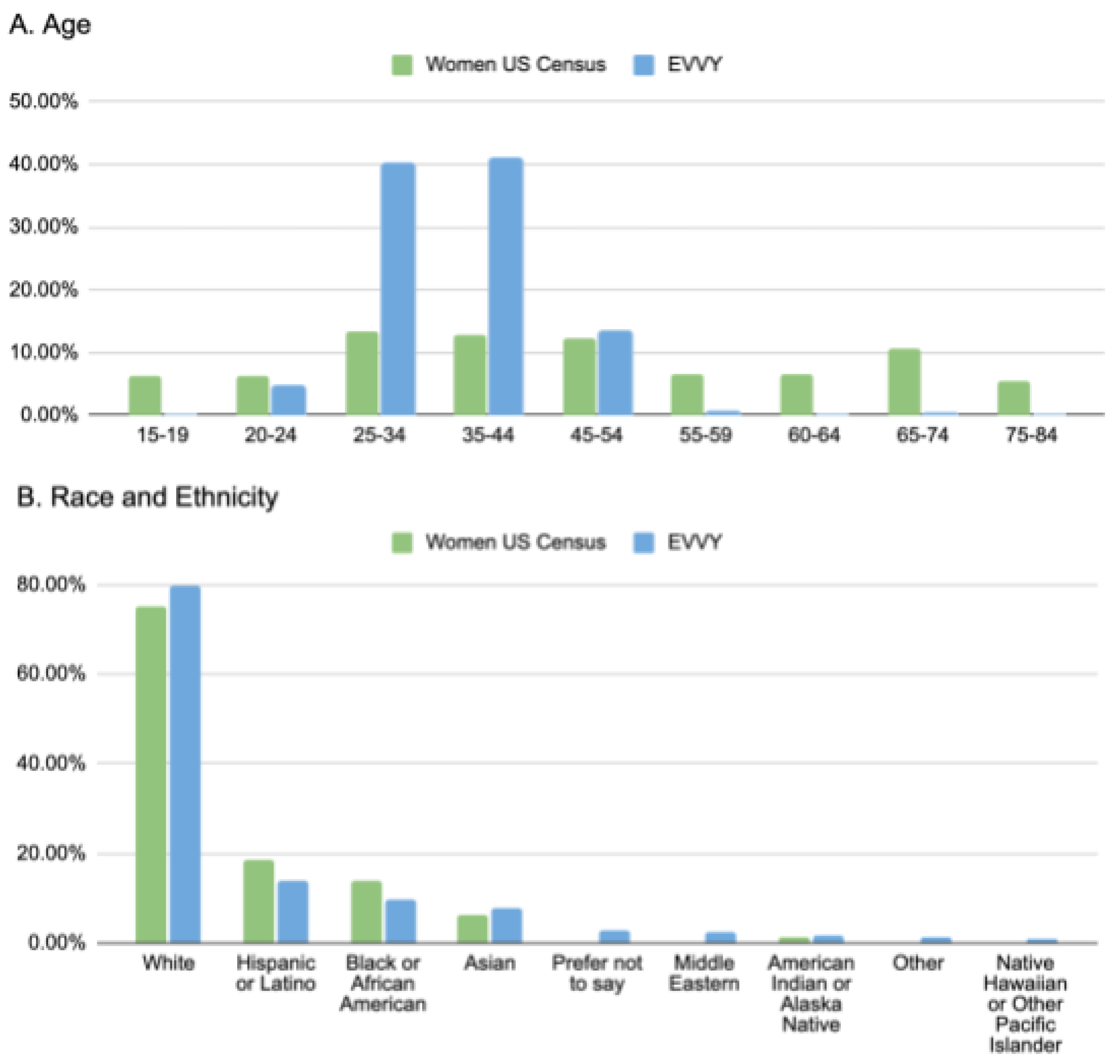
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- A Muzny, C.; Balkus, J.; Mitchell, C.; Sobel, J.D.; Workowski, K.; Marrazzo, J.; Schwebke, J.R. Diagnosis and Management of Bacterial Vaginosis: Summary of Evidence Reviewed for the 2021 Centers for Disease Control and Prevention Sexually Transmitted Infections Treatment Guidelines. Clin. Infect. Dis. 2022, 74, S144–S151. [CrossRef]
- Peebles, K.; Velloza, J.; Balkus, J.E.; McClelland, R.S.; Barnabas, R.V. High Global Burden and Costs of Bacterial Vaginosis: A Systematic Review and Meta-Analysis. Sex. Transm. Dis. 2019, 46, 304–311. [CrossRef]
- Foxman, B., et al., Prevalence of recurrent vulvovaginal candidiasis in 5 European countries and the United States: results from an internet panel survey. J Low Genit Tract Dis, 2013. 17(3): p. 340-5.
- Flagg, E.W.; Meites, E.; Phillips, C.B.; Papp, J.; Torrone, E.A. Prevalence of Trichomonas vaginalis Among Civilian, Noninstitutionalized Male and Female Population Aged 14 to 59 Years: United States, 2013 to 2016. Sex. Transm. Dis. 2019, 46, e93–e96. [CrossRef]
- Amabebe, E.; Anumba, D.O.C. The Vaginal Microenvironment: The Physiologic Role of Lactobacilli. Front. Med. 2018, 5, 181. [CrossRef]
- Sobel, J.D.; Subramanian, C.; Foxman, B.; Fairfax, M.; Gygax, S.E. Mixed Vaginitis—More Than Coinfection and With Therapeutic Implications. Curr. Infect. Dis. Rep. 2013, 15, 104–108. [CrossRef]
- Nugent, R.P.; A Krohn, M.; Hillier, S.L. Reliability of diagnosing bacterial vaginosis is improved by a standardized method of gram stain interpretation. J. Clin. Microbiol. 1991, 29, 297–301. [CrossRef]
- Amsel, R., et al., Nonspecific vaginitis. Diagnostic criteria and microbial and epidemiologic associations. Am J Med, 1983. 74(1): p. 14-22.
- Schwiertz, A., et al., Throwing the dice for the diagnosis of vaginal complaints? Ann Clin Microbiol Antimicrob, 2006. 5: p. 4.
- Muzny, C.A.; Cerca, N.; Elnaggar, J.H.; Taylor, C.M.; Sobel, J.D.; Van Der Pol, B. State of the Art for Diagnosis of Bacterial Vaginosis. J. Clin. Microbiol. 2023, 61, e0083722. [CrossRef]
- Lebeer, S.; Ahannach, S.; Gehrmann, T.; Wittouck, S.; Eilers, T.; Oerlemans, E.; Condori, S.; Dillen, J.; Spacova, I.; Donck, L.V.; et al. A citizen-science-enabled catalogue of the vaginal microbiome and associated factors. Nat. Microbiol. 2023, 8, 2183–2195. [CrossRef]
- Ravel, J.; Moreno, I.; Simón, C. Bacterial vaginosis and its association with infertility, endometritis, and pelvic inflammatory disease. Am. J. Obstet. Gynecol. 2021, 224, 251–257. [CrossRef]
- Langille, M.G., et al., Predictive functional profiling of microbial communities using 16S rRNA marker gene sequences. Nat Biotechnol, 2013. 31(9): p. 814-21.
- Trevethan, R. Sensitivity, Specificity, and Predictive Values: Foundations, Pliabilities, and Pitfalls in Research and Practice. Front. Public Heal. 2017, 5, 307–307. [CrossRef]
- Shi, Y.; Wang, G.; Lau, H.C.-H.; Yu, J. Metagenomic Sequencing for Microbial DNA in Human Samples: Emerging Technological Advances. Int. J. Mol. Sci. 2022, 23, 2181. [CrossRef]
- Bolger, A.M.; Lohse, M.; Usadel, B. Trimmomatic: A flexible trimmer for Illumina sequence data. Bioinformatics 2014, 30, 2114–2120. [CrossRef]
- Salter, S.J.; Cox, M.J.; Turek, E.M.; Calus, S.T.; Cookson, W.O.; Moffatt, M.F.; Turner, P.; Parkhill, J.; Loman, N.J.; Walker, A.W. Reagent and laboratory contamination can critically impact sequence-based microbiome analyses. BMC Biol. 2014, 12, 87. [CrossRef]
- Ravel, J.; Gajer, P.; Abdo, Z.; Schneider, G.M.; Koenig, S.S.K.; McCulle, S.L.; Karlebach, S.; Gorle, R.; Russell, J.; Tacket, C.O.; et al. Vaginal microbiome of reproductive-age women. Proc. Natl. Acad. Sci. USA 2011, 108 (Suppl. S1), 4680–4687. [CrossRef]
- Romero, R.; Theis, K.R.; Gomez-Lopez, N.; Winters, A.D.; Panzer, J.J.; Lin, H.; Galaz, J.; Greenberg, J.M.; Shaffer, Z.; Kracht, D.J.; et al. The Vaginal Microbiota of Pregnant Women Varies with Gestational Age, Maternal Age, and Parity. Microbiol. Spectr. 2023, 11, e0342922. [CrossRef]
- Paavonen, J.A.; Brunham, R.C. Vaginitis in Nonpregnant Patients: ACOG Practice Bulletin Number 215. Obstet. Gynecol. 2020, 135, 1229–1230. [CrossRef]
- Healthcare, A. 2022 Survey of Physician Appointment Wait Times and Medicare and Medicaid Acceptance Rates. 2022; Available from: https://www.wsha.org/wp-content/uploads/mha2022waittimesurveyfinal.pdf.
- Peterson, C.-L.; Alexander, D.; Chen, J.C.-Y.; Adam, H.; Walker, M.; Ali, J.; Forbes, J.; Taboada, E.; Barker, D.O.R.; Graham, M.; et al. Clinical Metagenomics Is Increasingly Accurate and Affordable to Detect Enteric Bacterial Pathogens in Stool. Microorganisms 2022, 10, 441. [CrossRef]
- Mu, S.; Hu, L.; Zhang, Y.; Liu, Y.; Cui, X.; Zou, X.; Wang, Y.; Lu, B.; Zhou, S.; Liang, X.; et al. Prospective Evaluation of a Rapid Clinical Metagenomics Test for Bacterial Pneumonia. Front. Cell. Infect. Microbiol. 2021, 11. [CrossRef]
- P, V.-B., S. CK, and S. JD, International Society for theStudy of Vulvovaginal Disease recommendations for the diagnosis andtreatment of vaginitis. Lisbon: Admedic, 2023.
- K, T.-W.; F, W.; P, N. Incidence and Symptom Profiling of Vaginitis Containing Aerobic and Anaerobic Pathogens. Am. J. Obstet. Gynecol. 2024, 230, S641–S642. [CrossRef]
- Skafte-Holm, A.; Humaidan, P.; Bernabeu, A.; Lledo, B.; Jensen, J.S.; Haahr, T. The Association between Vaginal Dysbiosis and Reproductive Outcomes in Sub-Fertile Women Undergoing IVF-Treatment: A Systematic PRISMA Review and Meta-Analysis. Pathogens 2021, 10, 295. [CrossRef]
- Zhou, Q.; Yu, Y.; Zhou, J.; Liu, J.; Gao, J. Relationship of Lactobacillus Vaginal Microbiota Changes and the Risk of Preterm Birth: A Systematic Review and Meta-Analysis. J. Women's Heal. 2024, 33, 228–238. [CrossRef]
- Kosti, I.; Lyalina, S.; Pollard, K.S.; Butte, A.J.; Sirota, M. Meta-Analysis of Vaginal Microbiome Data Provides New Insights Into Preterm Birth. Front. Microbiol. 2020, 11, 476. [CrossRef]
- Torcia, M.G. Interplay among Vaginal Microbiome, Immune Response and Sexually Transmitted Viral Infections. Int. J. Mol. Sci. 2019, 20, 266. [CrossRef]
- Łaniewski, P.; Herbst-Kralovetz, M.M. Connecting microbiome and menopause for healthy ageing. Nat. Microbiol. 2022, 7, 354–358. [CrossRef]
- Sharifian, K.; Shoja, Z.; Jalilvand, S. The interplay between human papillomavirus and vaginal microbiota in cervical cancer development. Virol. J. 2023, 20, 1–9. [CrossRef]
- Yang, X.; Da, M.; Zhang, W.; Qi, Q.; Zhang, C.; Han, S. Role of Lactobacillus in cervical cancer. Cancer Manag. Res. 2018, ume 10, 1219–1229. [CrossRef]
| Positive | Negative | Total | Predictive Value | ||
|---|---|---|---|---|---|
| NovaSeq | Positive | 95 | 11 | 106 | 89.6% (95/106) PPV |
| Negative | 7 | 99 | 106 | 93.4% (99/106) NPV | |
| Total | 102 | 110 | |||
| Sensitivity 93.1% (95/102) |
Specificity 90% (99/110) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




