Submitted:
10 May 2024
Posted:
13 May 2024
You are already at the latest version
Abstract
Keywords:
Introduction
Methods
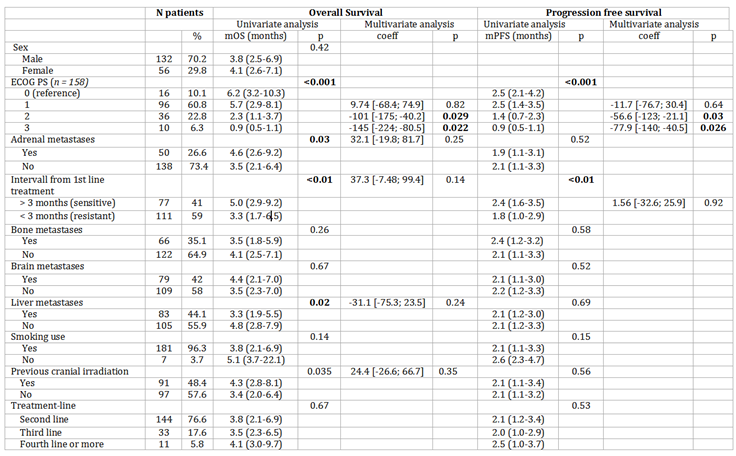
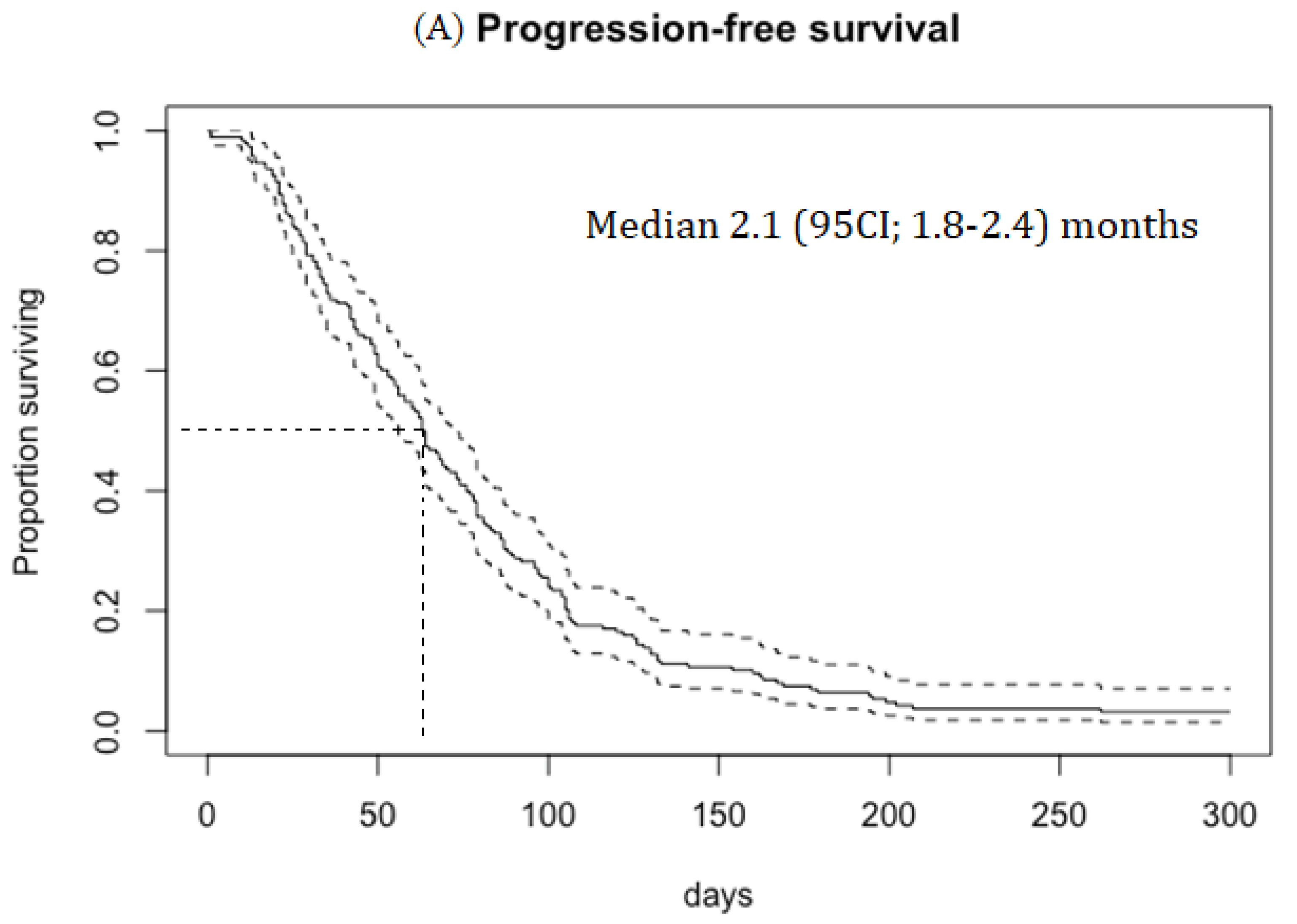
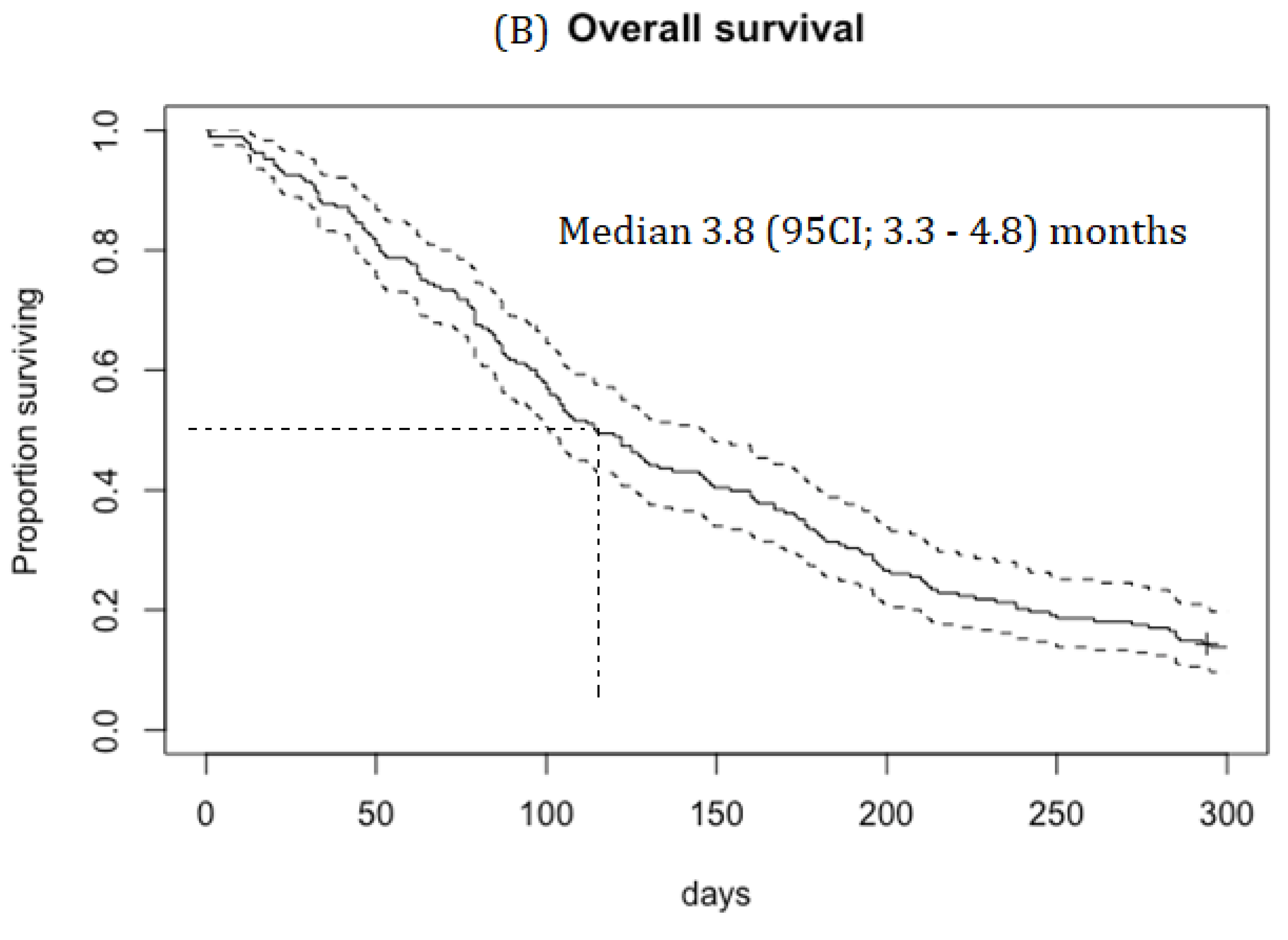
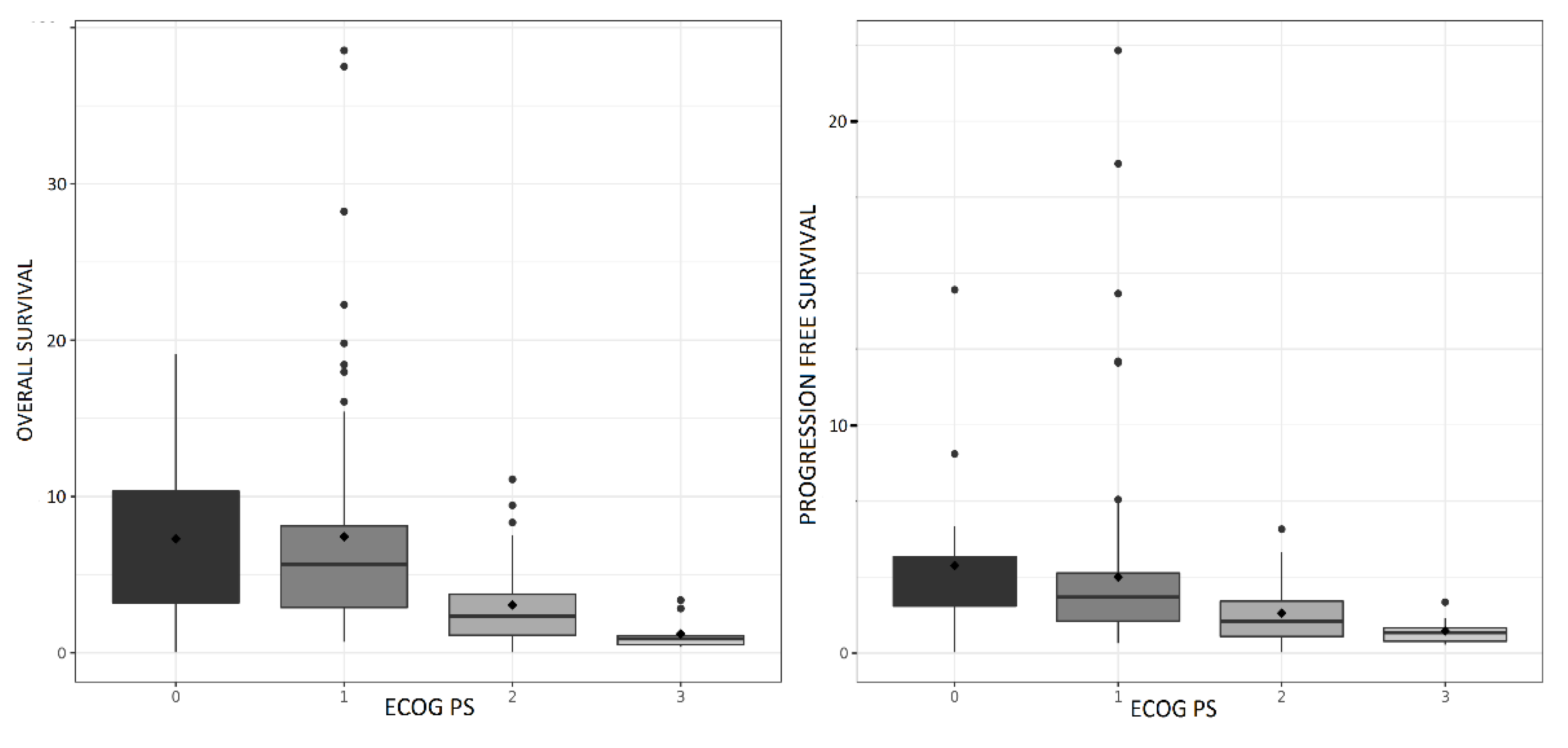
Results
Patients Characteristics
Efficacy
Safety
Discussion
Conclusions
Ethics
Funding
Data availability Statement
Conflict of Interests
Abbreviations
References
- Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018;68(6):394–424. [CrossRef]
- Bernhardt EB, Jalal SI. Small Cell Lung Cancer. Cancer Treat Res. 2016;170:301–22. [CrossRef]
- Pinsky PF, Church TR, Izmirlian G, Kramer BS. The National Lung Screening Trial: Results stratified by demographics, smoking history, and lung cancer histology. Cancer. 2013 Nov;119(22):3976–83. [CrossRef]
- Roth BJ, Johnson DH, Einhorn LH, Schacter LP, Cherng NC, Cohen HJ; et al. Randomized study of cyclophosphamide, doxorubicin, and vincristine versus etoposide and cisplatin versus alternation of these two regimens in extensive small-cell lung cancer: A phase III trial of the Southeastern Cancer Study Group. J Clin Oncol Off J Am Soc Clin Oncol. 1992 Feb;10(2):282–91. [CrossRef]
- Paz-Ares L, Dvorkin M, Chen Y, Reinmuth N, Hotta K, Trukhin D; et al. Durvalumab plus platinum-etoposide versus platinum-etoposide in first-line treatment of extensive-stage small-cell lung cancer (CASPIAN): A randomised, controlled, open-label, phase 3 trial. Lancet Lond Engl. 2019 Nov;394(10212):1929–39. [CrossRef]
- Horn L, Mansfield AS, Szczęsna A, Havel L, Krzakowski M, Hochmair MJ; et al. First-Line Atezolizumab plus Chemotherapy in Extensive-Stage Small-Cell Lung Cancer. N Engl J Med. 2018 Dec;379(23):2220–9. [CrossRef]
- Simon GR, Turrisi A, American College of Chest Physicians. Management of small cell lung cancer: ACCP evidence-based clinical practice guidelines (2nd edition). Chest. 2007 Sep;132(3 Suppl):324S-339S. [CrossRef]
- Owonikoko TK, Behera M, Chen Z, Bhimani C, Curran WJ, Khuri FR; et al. A systematic analysis of efficacy of second-line chemotherapy in sensitive and refractory small-cell lung cancer. J Thorac Oncol Off Publ Int Assoc Study Lung Cancer. 2012 May;7(5):866–72. [CrossRef]
- O’Brien MER, Ciuleanu T-E, Tsekov H, Shparyk Y, Cuceviá B, Juhasz G; et al. Phase III trial comparing supportive care alone with supportive care with oral topotecan in patients with relapsed small-cell lung cancer. J Clin Oncol Off J Am Soc Clin Oncol. 2006 Dec;24(34):5441–7. [CrossRef]
- von Pawel J, Schiller JH, Shepherd FA, Fields SZ, Kleisbauer JP, Chrysson NG; et al. Topotecan versus cyclophosphamide, doxorubicin, and vincristine for the treatment of recurrent small-cell lung cancer. J Clin Oncol Off J Am Soc Clin Oncol. 1999 Feb;17(2):658–67. [CrossRef]
- Baize N, Monnet I, Greillier L, Geier M, Lena H, Janicot H; et al. Carboplatin plus etoposide versus topotecan as second-line treatment for patients with sensitive relapsed small-cell lung cancer: An open-label, multicentre, randomised, phase 3 trial. Lancet Oncol. 2020 Sep;21(9):1224–33. [CrossRef]
- Ahn MJ, Cho BC, Felip E, Korantzis I, Ohashi K, Majem M; et al. Tarlatamab for Patients with Previously Treated Small-Cell Lung Cancer. N Engl J Med. 2023;389(22):2063-75. [CrossRef]
- Früh M, Panje CM, Reck M, Blackhall F, Califano R, Cappuzzo F; et al. Choice of second-line systemic therapy in stage IV small cell lung cancer (SCLC) - A decision-making analysis amongst European lung cancer experts. Lung Cancer Amst Neth. 2020 Aug;146:6–11. [CrossRef]
- Dongiovanni V, Buffoni L, Berruti A, Dongiovanni D, Grillo R, Barone C; et al. Second-line chemotherapy with weekly paclitaxel and gemcitabine in patients with small-cell lung cancer pretreated with platinum and etoposide: A single institution phase II trial. Cancer Chemother Pharmacol. 2006 Aug;58(2):203–9. [CrossRef]
- Dazzi C, Cariello A, Casanova C, Verlicchi A, Montanari M, Papiani G; et al. Gemcitabine and paclitaxel combination as second-line chemotherapy in patients with small-cell lung cancer: A phase II study. Clin Lung Cancer. 2013 Jan;14(1):28–33. [CrossRef]
- Yun T, Kim HT, Han J-Y, Yoon SJ, Kim HY, Nam B-H; et al. A Phase II Study of Weekly Paclitaxel Plus Gemcitabine as a Second-Line Therapy in Patients with Metastatic or Recurrent Small Cell Lung Cancer. Cancer Res Treat. 2016 Apr;48(2):465–72. [CrossRef]
- Spigel DR, Vicente D, Ciuleanu TE, Gettinger S, Peters S, Horn L; et al. Second-line nivolumab in relapsed small-cell lung cancer: CheckMate 331☆. Ann Oncol Off J Eur Soc Med Oncol. 2021 May;32(5):631–41. [CrossRef]
- Schmid S, Mauti LA, Friedlaender A, Blum V, Rothschild SI, Bouchaab H; et al. Outcomes with immune checkpoint inhibitors for relapsed small-cell lung cancer in a Swiss cohort. Cancer Immunol Immunother CII. 2020 Aug;69(8):1605–13. [CrossRef]
- Trigo J, Subbiah V, Besse B, Moreno V, López R, Sala MA; et al. Lurbinectedin as second-line treatment for patients with small-cell lung cancer: A single-arm, open-label, phase 2 basket trial. Lancet Oncol. 2020 May;21(5):645–54. [CrossRef]
- Aix SP, Ciuleanu TE, Navarro A, Cousin S, Bonanno L, Smit EF; et al. Combination lurbinectedin and doxorubicin versus physician's choice of chemotherapy in patients with relapsed small-cell lung cancer (ATLANTIS): A multicentre, randomised, open-label, phase 3 trial. Lancet Respir Med. 2023 Jan;11(1):74-86. [CrossRef]
| Characteristic | Values (n =188) |
| Age, years | 62.0 (57.0; 68.0) |
| Male gender | 132 (70.0%) |
| ECOG PS | |
| 0 or 1 | 112 (59.5%) |
|
≥2 Missing data |
46 (24.5%) 30 (16.0%) |
| Current or former smoker | 181 (96.2%) |
| No neuro-endocrine marker Mean Ki67 Index (%) |
3 (1.6%) 90 (+/-13.5) |
| Limited stage at diagnosis | 56 (29.8%) |
| Number of previous treatment line(s) One Two Three or more |
144 (76.6%) 33 (17.6%) 11 (5.8%) |
| Platinum-Sensitive disease Platinum-Resistant disease |
77 (41.0%) 111 (59%) |
| Metastases Liver metastases Bone metastases Adrenal metastases Brain metastases No extra-thoracic metastases |
83 (44.1%) 66 (35.1%) 50 (26.6%) 79 (42.0%) 35 (18.6%) |
| Previous thoracic radiotherapy | 69 (36.7%) |
| Cranial irradiation PCI WBRT |
21 (11.1%) 41 (21.2%) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




