Submitted:
12 May 2024
Posted:
13 May 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Plant Material
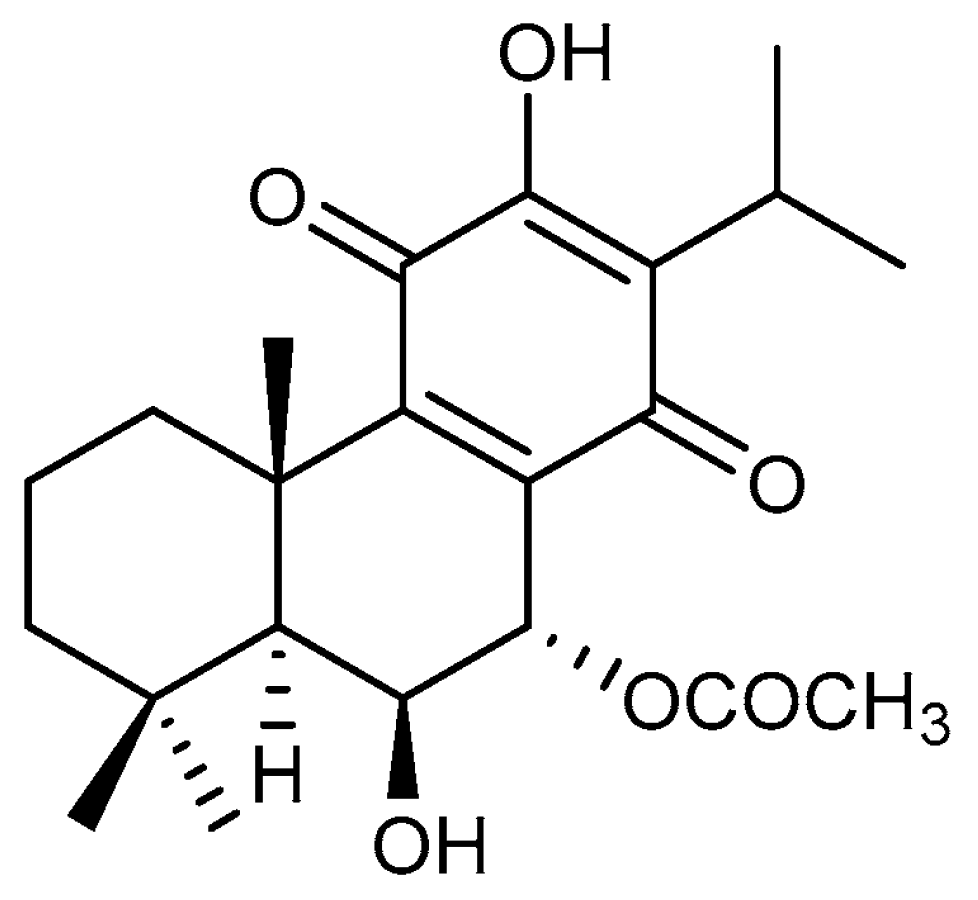
2.2. Isolation of Roy
2.3. Synthesis of Roy-BODIPY
2.4. Cell Culture
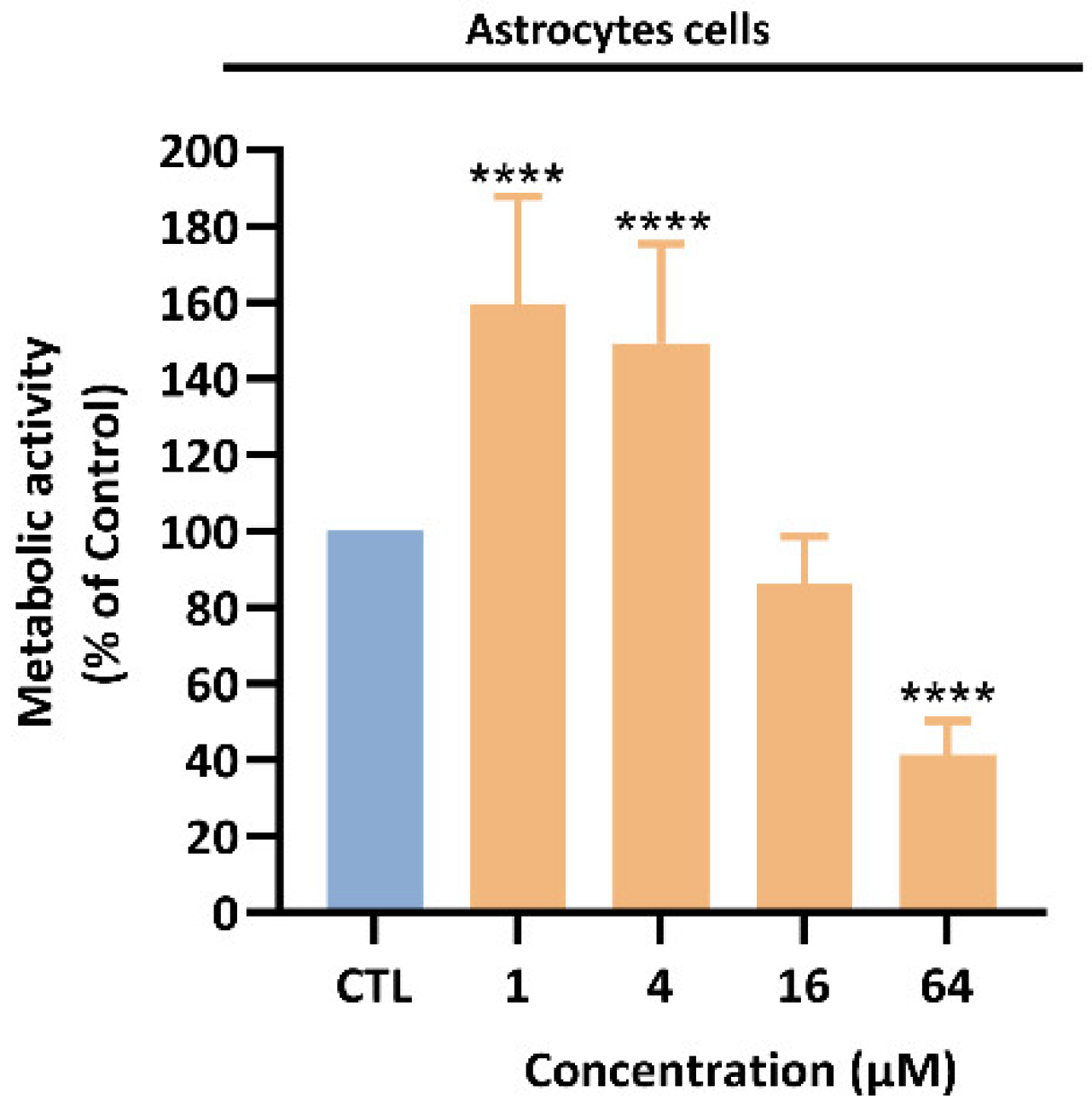
2.5. Primary Rat Culture of Astrocytes
2.6. 3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyl tetrazolium Bromide (MTT) Assay
2.7. Alamar Blue® Assay
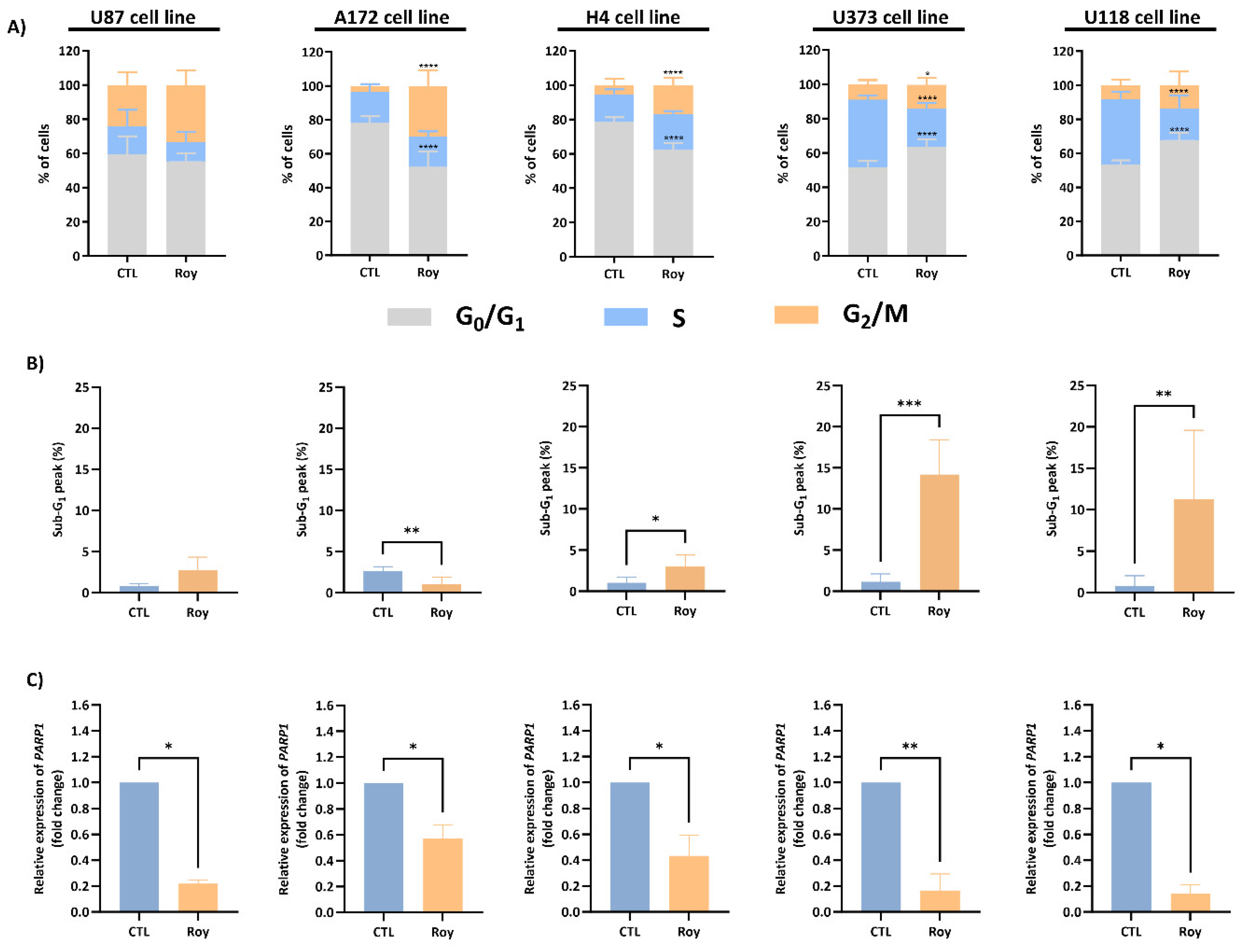
2.8. Cell Cycle
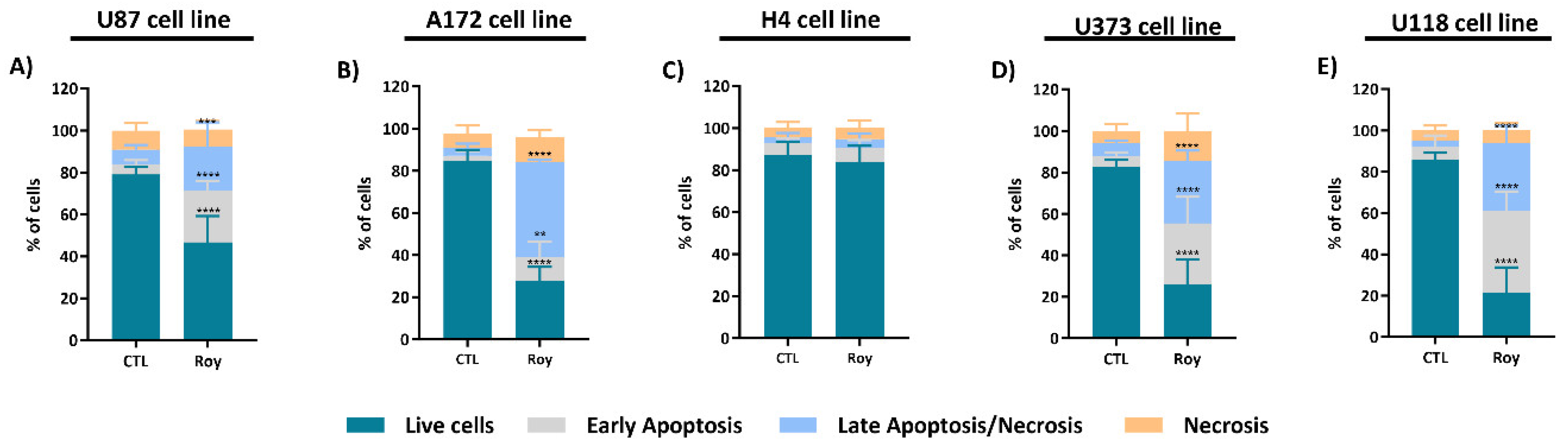
2.9. Cell Death Assay
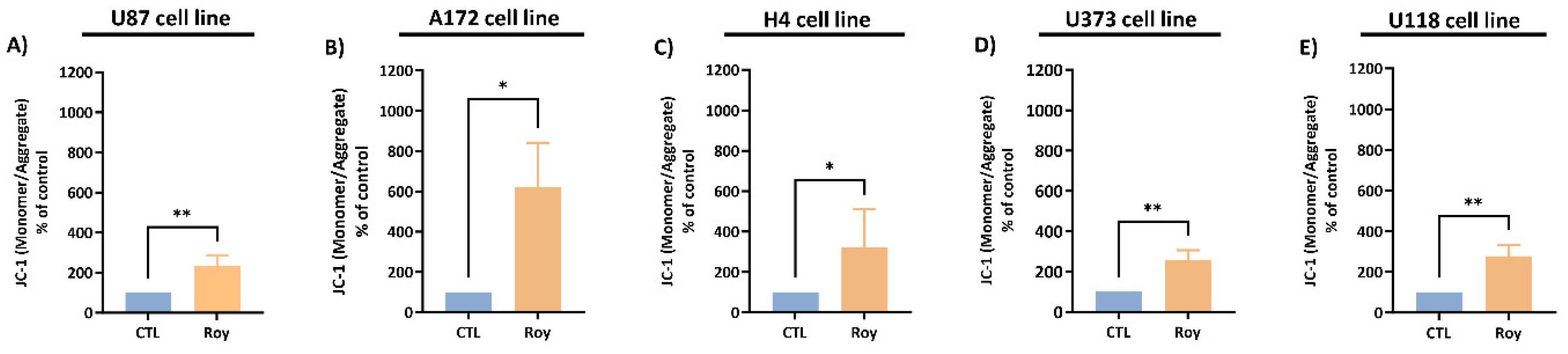
2.10. Mitochondrial Membrane Potential
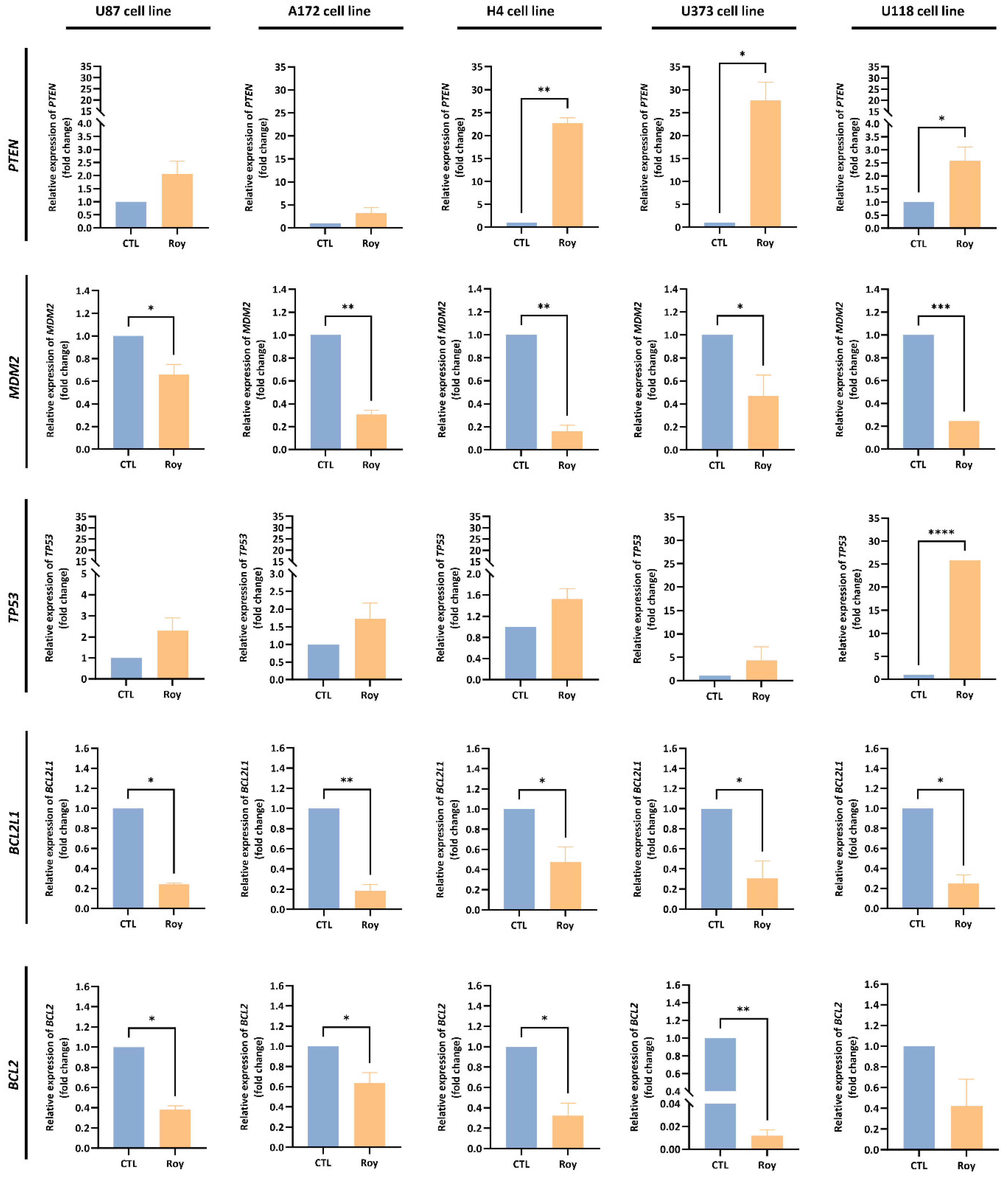
2.11. Quantitative Real-Time PCR
2.12. Western Blotting
2.13. Activated Caspase-3 Expression Analysis
2.14. Confocal Microscopy
2.15. Statistical Analysis
3. Results
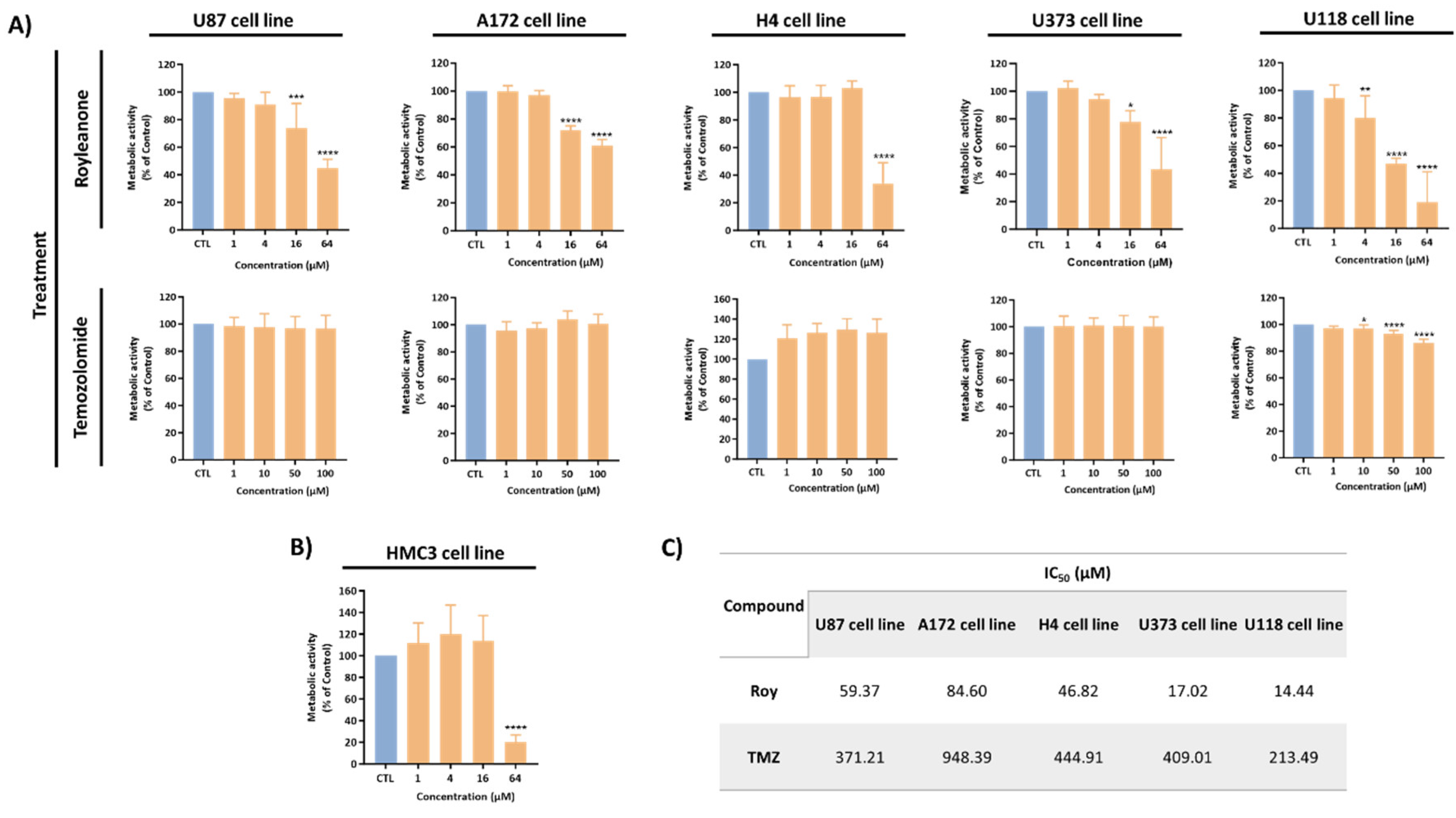
3.1. Roy Effect in the Metabolic Activity of GB Cells
3.2. Roy Effect in Cell Viability of GB Cells
3.3. Roy Effect in Cell Cycle Regulation of GB Cells
3.4. Effect of Roy on Mitochondrial Membrane Potential of GB Cells
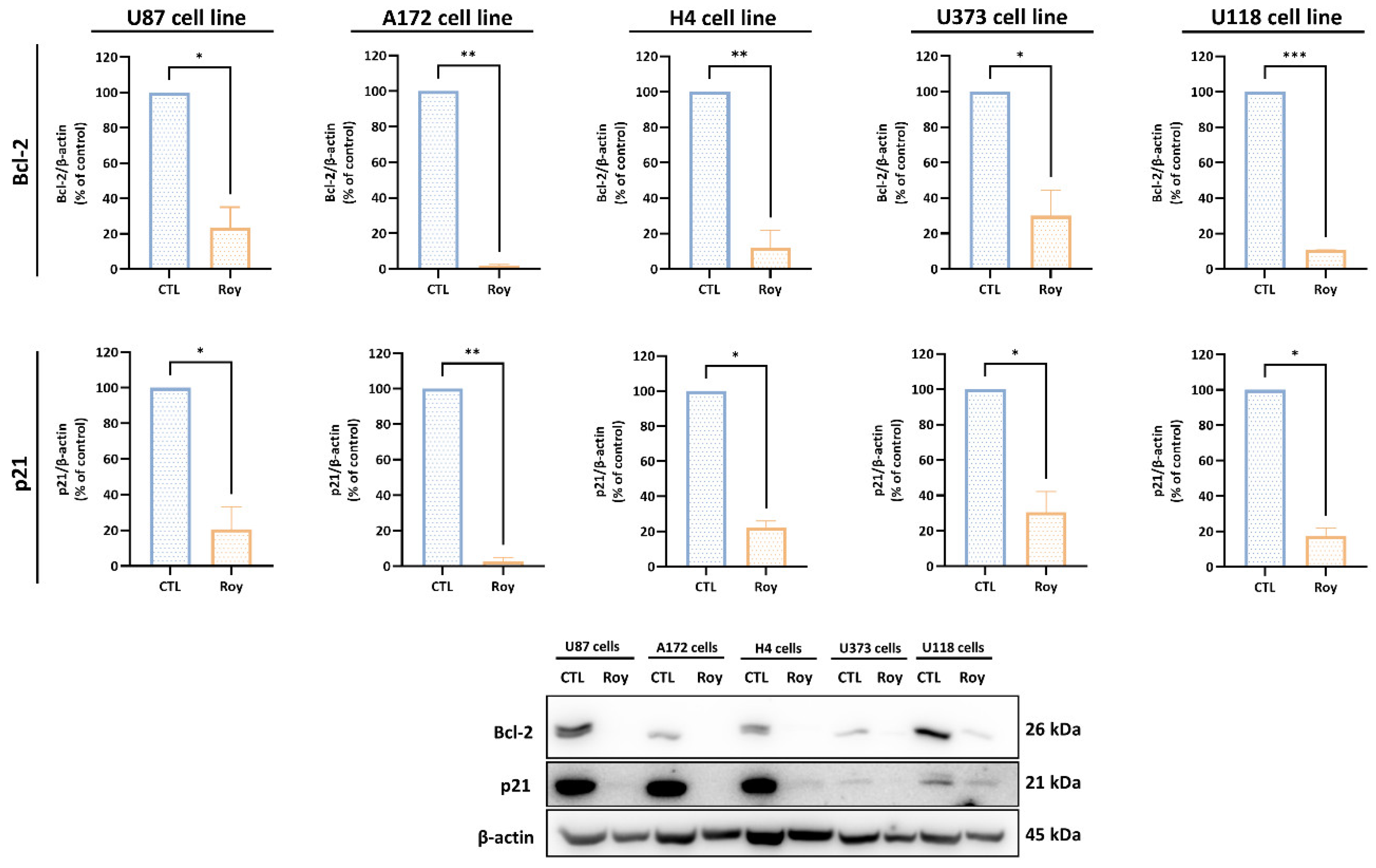
3.5. Roy Effect on Tumor Suppressor Genes mRNA Levels and Anti-Apoptotic Proteins Expression in GB Cells
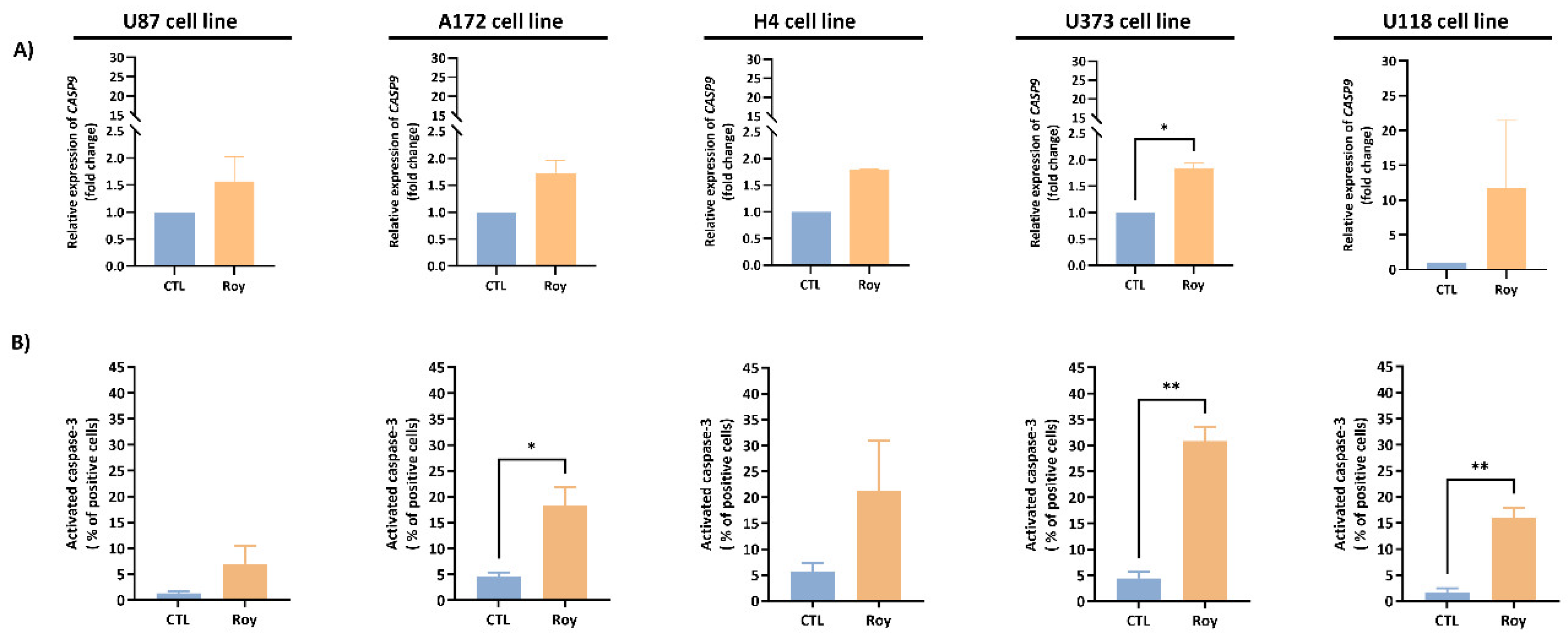
3.6. Roy Effect in Caspase Levels of GB Cells
3.7. Internalization of Fluorescently-Labeled Roy and Its Effect on the Mitochondrial Morphology in GB Cells
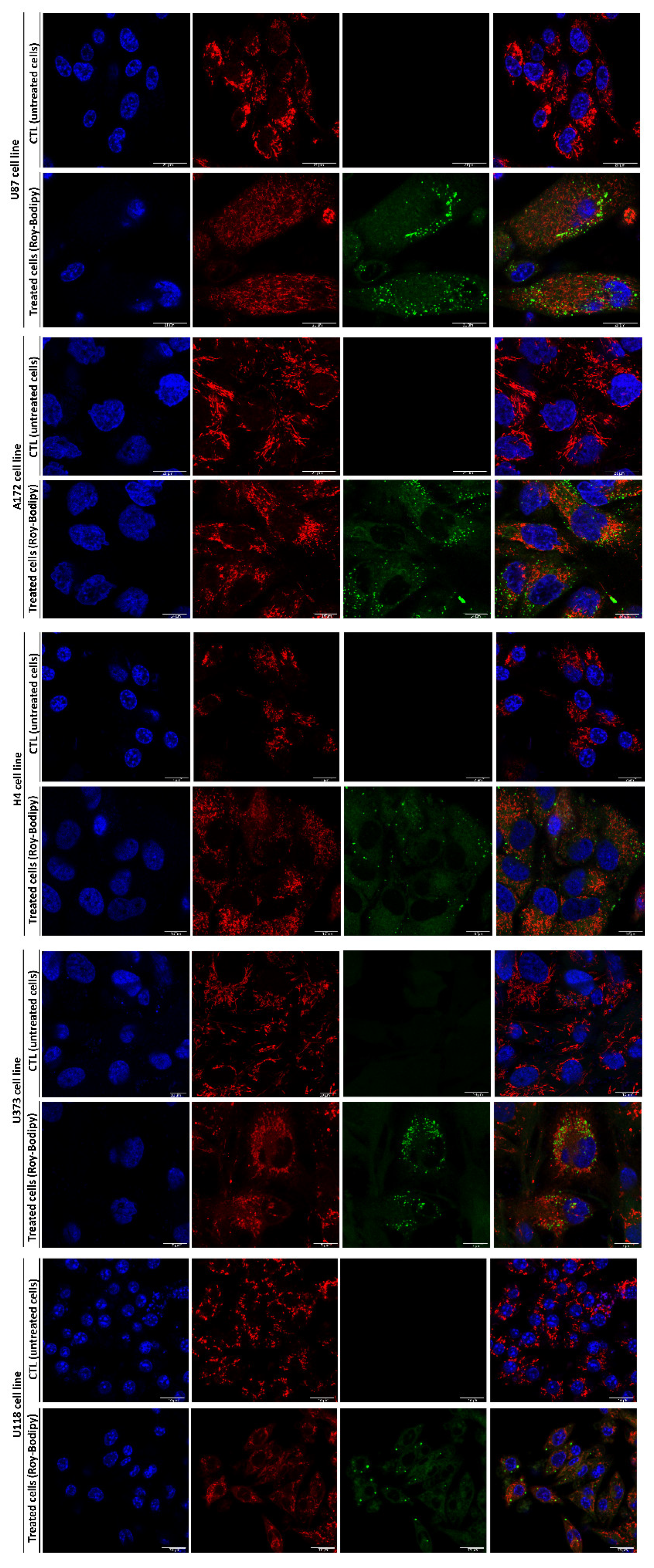
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Louis, D.N.; Perry, A.; Wesseling, P.; Brat, D.J.; Cree, I.A.; Figarella-Branger, D.; Hawkins, C.; Ng, H.K.; Pfister, S.M.; Reifenberger, G.; et al. The 2021 WHO Classification of Tumors of the Central Nervous System: A Summary. Neuro. Oncol. 2021, 23, 1231–1251. [Google Scholar] [CrossRef] [PubMed]
- Wen, P.Y.; Packer, R.J. The 2021 WHO Classification of Tumors of the Central Nervous System: Clinical Implications. Neuro. Oncol. 2021, 23, 1215–1217. [Google Scholar] [CrossRef] [PubMed]
- Hazaymeh, M.; Löber-Handwerker, R.; Döring, K.; Abboud, T.; Mielke, D.; Rohde, V.; Malinova, V. Prognostic Differences and Implications on Treatment Strategies between Butterfly Glioblastoma and Glioblastoma with Unilateral Corpus Callosum Infiltration. Sci. Rep. 2022, 12, 19208. [Google Scholar] [CrossRef] [PubMed]
- Kow, C.Y.; Kim, B.J.H.; Park, T.I.-H.; Chen, J.C.C.; Vong, C.K.; Kim, J.Y.; Shim, V.; Dragunow, M.; Heppner, P. Extent of Resection Affects Prognosis for Patients with Glioblastoma in Non-Eloquent Regions. J. Clin. Neurosci. 2020, 80, 242–249. [Google Scholar] [CrossRef] [PubMed]
- Tan, A.C.; Ashley, D.M.; López, G.Y.; Malinzak, M.; Friedman, H.S.; Khasraw, M. Management of Glioblastoma: State of the Art and Future Directions. CA. Cancer J. Clin. 2020, 70, 299–312. [Google Scholar] [CrossRef] [PubMed]
- Rončević, A.; Koruga, N.; Soldo Koruga, A.; Rončević, R.; Rotim, T.; Šimundić, T.; Kretić, D.; Perić, M.; Turk, T.; Štimac, D. Personalized Treatment of Glioblastoma: Current State and Future Perspective. Biomedicines 2023, 11, 1579. [Google Scholar] [CrossRef] [PubMed]
- Angom, R.S.; Nakka, N.M.; Bhattacharya, S. Advances in Glioblastoma Therapy: An Update on Current Approaches. Brain Sci. 2023, 13, 1536. [Google Scholar] [CrossRef] [PubMed]
- Chien, C.-H.; Hsueh, W.-T.; Chuang, J.-Y.; Chang, K.-Y. Dissecting the Mechanism of Temozolomide Resistance and Its Association with the Regulatory Roles of Intracellular Reactive Oxygen Species in Glioblastoma. J. Biomed. Sci. 2021, 28, 18. [Google Scholar] [CrossRef]
- Ortiz, R.; Perazzoli, G.; Cabeza, L.; Jiménez-Luna, C.; Luque, R.; Prados, J.; Melguizo, C. Temozolomide: An Updated Overview of Resistance Mechanisms, Nanotechnology Advances and Clinical Applications. Curr. Neuropharmacol. 2021, 19, 513–537. [Google Scholar] [CrossRef]
- Howes, M.J.R. The Evolution of Anticancer Drug Discovery from Plants. Lancet Oncol. 2018, 19, 293–294. [Google Scholar] [CrossRef]
- Rodrigues, T.; Reker, D.; Schneider, P.; Schneider, G. Counting on Natural Products for Drug Design. Nat. Chem. 2016, 8, 531–541. [Google Scholar] [CrossRef] [PubMed]
- Rayan, A.; Raiyn, J.; Falah, M. Nature Is the Best Source of Anticancer Drugs: Indexing Natural Products for Their Anticancer Bioactivity. PLoS One 2017, 12, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Domínguez-Martín, E.M.; Magalhães, M.; Efferth, T.; Díaz-Lanza, A.M.; Cabral, C.; Rijo, P. Chapter 12—Terpenes: A Hope for Glioblastoma Patients. In New Insights Into Glioblastoma: Diagnosis, Therapeutics and Theranostics; Vitorino, C., Balaña, C., Cabral, C.B.T., Eds.; Academic Press, 2023; pp. 227–269. ISBN 978-0-323-99873-4. [Google Scholar]
- Yang, W.; Chen, X.; Li, Y.; Guo, S.; Wang, Z.; Yu, X. Advances in Pharmacological Activities of Terpenoids. Nat. Prod. Commun. 2020, 15, 1934578x20903555. [Google Scholar] [CrossRef]
- Domínguez-Martín, E.M.; Magalhães, M.; Díaz-Lanza, A.M.; Marques, M.P.; Princiotto, S.; Gómez, A.M.; Efferth, T.; Cabral, C.; Rijo, P. Phytochemical Study and Antiglioblastoma Activity Assessment of Plectranthus Hadiensis (Forssk.) Schweinf. Ex Sprenger Var. Hadiensis Stems. Molecules 2022, 27, 3813. [Google Scholar] [CrossRef] [PubMed]
- Śliwiński, T.; Sitarek, P.; Skała, E.; M. S. Isca, V.; Synowiec, E.; Kowalczyk, T.; Bijak, M.; Rijo, P. Diterpenoids from Plectranthus Spp. as Potential Chemotherapeutic Agents via Apoptosis. Pharmaceuticals 2020, 13, 123. [Google Scholar] [CrossRef] [PubMed]
- Sitarek, P.; Toma, M.; Ntungwe, E.; Kowalczyk, T.; Skała, E.; Wieczfinska, J.; Śliwiński, T.; Rijo, P. Insight the Biological Activities of Selected Abietane Diterpenes Isolated from Plectranthus spp. Biomolecules 2020, 10, 194. [Google Scholar] [CrossRef] [PubMed]
- Ntungwe, E.; Domínguez-Martín, E.M.; Teodósio, C.; Teixidó-Trujillo, S.; Armas Capote, N.; Saraiva, L.; Díaz-Lanza, A.M.; Duarte, N.; Rijo, P. Preliminary Biological Activity Screening of Plectranthus Spp. Extracts for the Search of Anticancer Lead Molecules. Pharmaceuticals 2021, 14, 402. [Google Scholar] [CrossRef] [PubMed]
- Massenzio, F.; Peña-altamira, E.; Petralla, S.; Virgili, M.; Zuccheri, G.; Miti, A.; Polazzi, E.; Mengoni, I.; Piffaretti, D.; Monti, B. Microglial Overexpression of FALS-Linked Mutant SOD1 Induces SOD1 Processing Impairment, Activation and Neurotoxicity and Is Counteracted by the Autophagy Inducer Trehalose. BBA - Mol. Basis Dis. 2018, 1864, 3771–3785. [Google Scholar] [CrossRef]
- Polazzi, E.; Altamira, L.E.P.; Eleuteri, S.; Barbaro, R.; Casadio, C.; Contestabile, A.; Monti, B. Neuroprotection of Microglial Conditioned Medium on 6-Hydroxydopamine-Induced Neuronal Death: Role of Transforming Growth Factor Beta-2. J. Neurochem. 2009, 110, 545–556. [Google Scholar] [CrossRef]
- Magalhães, M.; Farinha, D.; de Lima, M.C.P.; Faneca, H. Increased Gene Delivery Efficiency and Specificity of a Lipid-Based Nanosystem Incorporating a Glycolipid. Int. J. Nanomedicine 2014, 9, 4979–4989. [Google Scholar] [CrossRef]
- Magalhães, M.; Domínguez-Martín, E.M.; Jorge, J.; Gonçalves, A.C.; Díaz-Lanza, A.M.; Manadas, B.; Efferth, T.; Rijo, P.; Cabral, C. Parvifloron D-Based Potential Therapy for Glioblastoma: Inducing Apoptosis via the Mitochondria Dependent Pathway. Front. Pharmacol. 2022, 13, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Alves, R.; Gonçalves, A.C.; Jorge, J.; Alves, J.; Alves da Silva, A.; Freitas-Tavares, P.; Nascimento Costa, J.M.; Almeida, A.M.; Sarmento-Ribeiro, A.B. Everolimus in Combination with Imatinib Overcomes Resistance in Chronic Myeloid Leukaemia. Med. Oncol. 2019, 36, 30. [Google Scholar] [CrossRef] [PubMed]
- Gonçalves, A.C.; Alves, V.; Silva, T.; Carvalho, C.; Oliveira, C.R. de; Sarmento-Ribeiro, A.B. Oxidative Stress Mediates Apoptotic Effects of Ascorbate and Dehydroascorbate in Human Myelodysplasia Cells in Vitro. Toxicol. Vitr. 2013, 27, 1542–1549. [Google Scholar] [CrossRef] [PubMed]
- Chaudhry, A.; Shi, R.; Luciani, D.S. A Pipeline for Multidimensional Confocal Analysis of Mitochondrial Morphology, Function, and Dynamics in Pancreatic β-Cells. Am. J. Physiol. Metab. 2019, 318, E87–E101. [Google Scholar] [CrossRef] [PubMed]
- Clinic, M. Https://Www.Mayoclinic.Org/Drugs-Supplements/Temozolomide-Oral-Route/Proper-Use/Drg-20066228.
- Miyazono, Y.; Hirashima, S.; Ishihara, N.; Kusukawa, J.; Nakamura, K.; Ohta, K. Uncoupled Mitochondria Quickly Shorten along Their Long Axis to Form Indented Spheroids, Instead of Rings, in a Fission-Independent Manner. Sci. Rep. 2018, 8, 350. [Google Scholar] [CrossRef] [PubMed]
- Magalhães, M.; Cabral, C.; Costa, B.M.; Manadas, B. Chapter 5—Secretome Analysis of Patient-Derived Glioblastoma Cells for Potential Biomarker Identification. In New Insights Into Glioblastoma: Diagnosis, Therapeutics and Theranostics; Vitorino, C., Balaña, C., Cabral, C.B.T., Eds.; Academic Press, 2023; pp. 81–97. ISBN 978-0-323-99873-4. [Google Scholar] [CrossRef]
- Verhaak, R.G.W.; Hoadley, K.A.; Purdom, E.; Wang, V.; Qi, Y.; Wilkerson, M.D.; Miller, C.R.; Ding, L.; Golub, T.; Mesirov, J.P.; et al. Integrated Genomic Analysis Identifies Clinically Relevant Subtypes of Glioblastoma Characterized by Abnormalities in PDGFRA, IDH1, EGFR, and NF1. Cancer Cell 2010, 17, 98–110. [Google Scholar] [CrossRef] [PubMed]
- Lane, R.; Simon, T.; Vintu, M.; Solkin, B.; Koch, B.; Stewart, N.; Benstead-Hume, G.; Pearl, F.M.G.; Critchley, G.; Stebbing, J.; et al. Cell-Derived Extracellular Vesicles Can Be Used as a Biomarker Reservoir for Glioblastoma Tumor Subtyping. Commun. Biol. 2019, 2, 315. [Google Scholar] [CrossRef] [PubMed]
- Plesca, D.; Mazumder, S.; Almasan, A.B.T.-M. in E. DNA Damage Response and Apoptosis. In Programmed Cell Death, The Biology and Therapeutic Implications of Cell Death, Part B; Academic Press, 2008; Volume 446, pp. 107–122. ISBN 0076-6879. [CrossRef]
- Pan, Z.; Zhang, X.; Yu, P.; Chen, X.; Lu, P.; Li, M.; Liu, X.; Li, Z.; Wei, F.; Wang, K.; et al. Cinobufagin Induces Cell Cycle Arrest at the G2/M Phase and Promotes Apoptosis in Malignant Melanoma Cells. Front. Oncol. 2019, 9. [Google Scholar] [CrossRef] [PubMed]
- Koul, D. PTEN Signaling Pathways in Glioblastoma. Cancer Biol. Ther. 2008, 7, 1321–1325. [Google Scholar] [CrossRef]
- Abraham, A.G.; O’Neill, E. PI3K/Akt-Mediated Regulation of P53 in Cancer. Biochem. Soc. Trans. 2014, 42, 798–803. [Google Scholar] [CrossRef]
- Pearson, J.R.D.; Regad, T. Targeting Cellular Pathways in Glioblastoma Multiforme. Signal Transduct. Target. Ther. 2017, 2, 17040. [Google Scholar] [CrossRef] [PubMed]
- Warren, C.F.A.; Wong-Brown, M.W.; Bowden, N.A. BCL-2 Family Isoforms in Apoptosis and Cancer. Cell Death Dis. 2019, 10, 177. [Google Scholar] [CrossRef] [PubMed]
- Abbas, T.; Dutta, A. P21 in Cancer: Intricate Networks and Multiple Activities. Nat. Rev. Cancer 2009, 9, 400–414. [Google Scholar] [CrossRef] [PubMed]
- Shamloo, B.; Usluer, S. P21 in Cancer Research. Cancers 2019, 11, 1178. [Google Scholar] [CrossRef] [PubMed]
- Yadav, P.; Yadav, R.; Jain, S.; Vaidya, A. Caspase-3: A Primary Target for Natural and Synthetic Compounds for Cancer Therapy. Chem. Biol. Drug Des. 2021, 98, 144–165. [Google Scholar] [CrossRef]
- Elmore, S. Apoptosis: A Review of Programmed Cell Death. Toxicol. Pathol. 2007, 35, 495–516. [Google Scholar] [CrossRef]
| Gene | Primers concentration (nM) | Forward (5′-3′) | Reverse (5′-3′) |
|---|---|---|---|
| BCL2 | 100 | GAGGATTGTGGCCTTCTTTGAG | AGCCTCCGTTATCCTGGATC |
| BCL2L1 | 150 | GCCACTTACCTGAATGACCACC | AACCAGCGGTTGAAGCGTTCCT |
| PARP1 | 300 | CGGAGTCTTCGGATAAGCTCT | TTTCCATCAAACATGGGCGAC |
| MDM2 | 150 | TGTTTGGCGTGCCAAGCTTCTC | CACAGATGTACCTGAGTCCGATG |
| CASP9 | 150 | GTTTGAGGACCTTCGACCAGCT | CAACGTACCAGGAGCCACTCTT |
| PTEN | 100 | TGAGTTCCCTCAGCCGTTACCT | GAGGTTTCCTCTGGTCCTGGTA |
| TP53 | 75 | CAGCACATGACGGAGGTTGT | TCATCCAAATACTCCACACGC |
| GAPDH | 150 | GTCTCCTCTGACTTCAACAGCG | ACCACCCTGTTGCTGTAGCCAA |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




