Submitted:
27 April 2024
Posted:
29 April 2024
You are already at the latest version
Abstract
Keywords:
Introduction
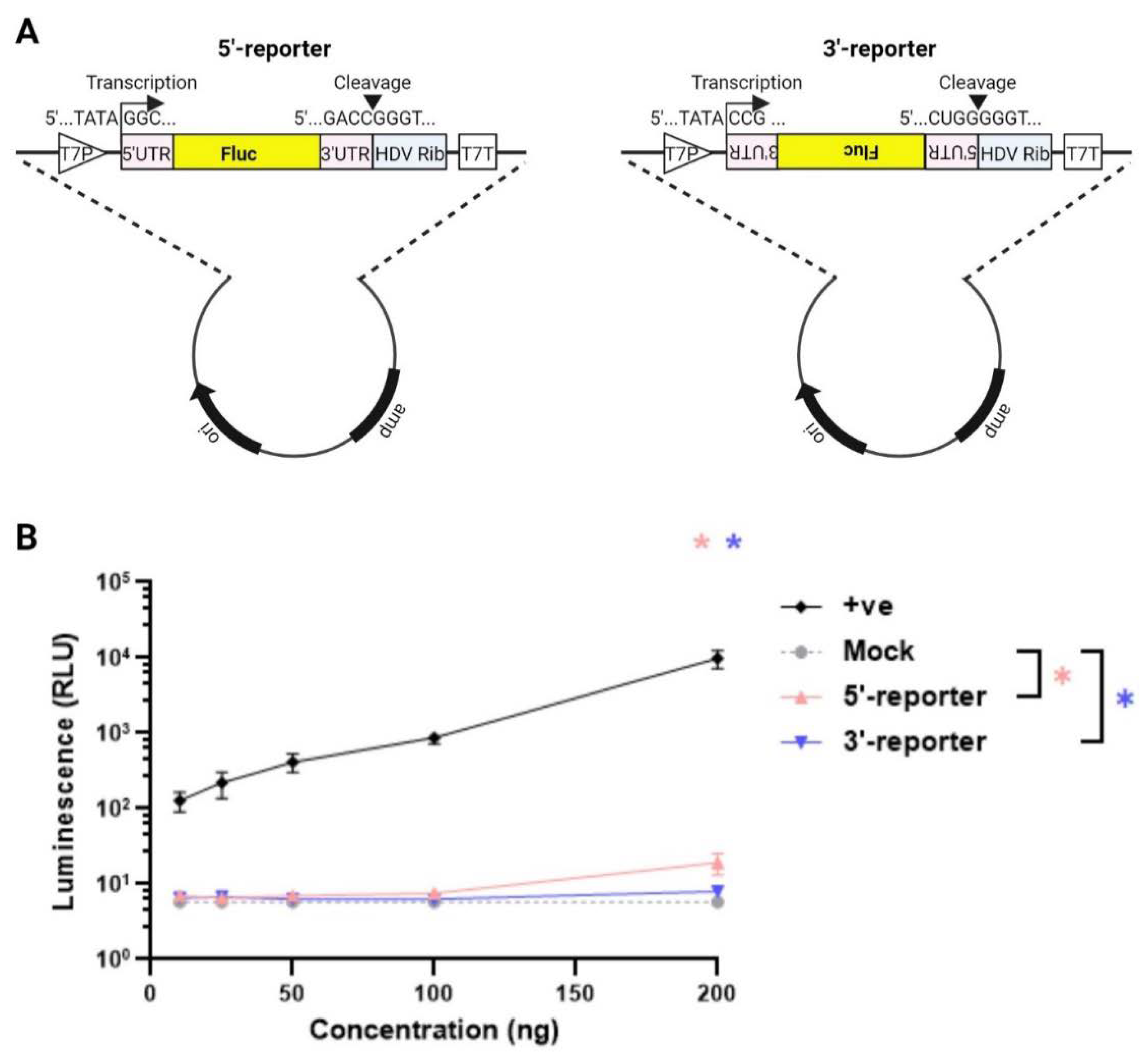
Materials and Methods
| Target gene | Sequence (5' to 3') | Use |
|---|---|---|
| Fluc in pMA plasmid | TAATACGACTCACTATAGGG TCGTCCACTCGGATGGCTA |
Sequence 5’- and 3’- plasmids containing Fluc gene |
| VP1 plasmid | GGAAGGAGAGATGTACCAGGA | Sequence mutations of GDD motif in VP1 plasmid |
Results
Discussion
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Troeger, C.; Khalil, I.A.; Rao, P.C.; Cao, S.; Blacker, B.F.; Ahmed, T.; Armah, G.; Bines, J.E.; Brewer, T.G.; Colombara, D.V.; et al. Rotavirus Vaccination and the Global Burden of Rotavirus Diarrhea Among Children Younger Than 5 Years. JAMA Pediatrics 2018, 172, 958–965. [Google Scholar] [CrossRef]
- Bányai, K.; Kemenesi, G.; Budinski, I.; Földes, F.; Zana, B.; Marton, S.; Varga-Kugler, R.; Oldal, M.; Kurucz, K.; Jakab, F. Candidate new rotavirus species in Schreiber's bats, Serbia. Infection, Genetics and Evolution 2017, 48, 19–26. [Google Scholar] [CrossRef]
- Johne, R.; Schilling-Loeffler, K.; Ulrich, R.G.; Tausch, S.H. Whole Genome Sequence Analysis of a Prototype Strain of the Novel Putative Rotavirus Species L. Viruses 2022, 14. [Google Scholar] [CrossRef] [PubMed]
- Johne, R.; Tausch, S.H.; Ulrich, R.G.; Schilling-Loeffler, K. Genome analysis of the novel putative rotavirus species K. Virus Res 2023, 334, 199171. [Google Scholar] [CrossRef] [PubMed]
- Matthijnssens, J.; Otto, P.H.; Ciarlet, M.; Desselberger, U.; Van Ranst, M.; Johne, R. VP6-sequence-based cutoff values as a criterion for rotavirus species demarcation. Archives of Virology 2012, 157, 1177–1182. [Google Scholar] [CrossRef] [PubMed]
- Mihalov-Kovács, E.; Gellért, Á.; Marton, S.; Farkas, S.L.; Fehér, E.; Oldal, M.; Jakab, F.; Martella, V.; Bányai, K. Candidate new rotavirus species in sheltered dogs, Hungary. Emerg Infect Dis 2015, 21, 660–663. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Baker, M.L.; Jiang, W.; Estes, M.K.; Prasad, B.V. Rotavirus architecture at subnanometer resolution. J Virol 2009, 83, 1754–1766. [Google Scholar] [CrossRef] [PubMed]
- Settembre, E.C.; Chen, J.Z.; Dormitzer, P.R.; Grigorieff, N.; Harrison, S.C. Atomic model of an infectious rotavirus particle. Embo j 2011, 30, 408–416. [Google Scholar] [CrossRef] [PubMed]
- McClain, B.; Settembre, E.; Temple, B.R.; Bellamy, A.R.; Harrison, S.C. X-ray crystal structure of the rotavirus inner capsid particle at 3.8 A resolution. J Mol Biol 2010, 397, 587–599. [Google Scholar] [CrossRef]
- Estrozi, L.F.; Settembre, E.C.; Goret, G.; McClain, B.; Zhang, X.; Chen, J.Z.; Grigorieff, N.; Harrison, S.C. Location of the dsRNA-dependent polymerase, VP1, in rotavirus particles. J Mol Biol 2013, 425, 124–132. [Google Scholar] [CrossRef]
- Prasad, B.V.V.; Rothnagel, R.; Zeng, C.Q.Y.; Jakana, J.; Lawton, J.A.; Chiu, W.; Estes, M.K. Visualization of ordered genomic RNA and localization of transcriptional complexes in rotavirus. Nature 1996, 382, 471–473. [Google Scholar] [CrossRef] [PubMed]
- Guglielmi, KM.; McDonald, SM.; Patton, JT. Mechanism of intraparticle synthesis of the rotavirus double-stranded RNA genome. The Journal of biological chemistry 2010, 285, 18123–18128. [Google Scholar] [CrossRef] [PubMed]
- Jenni, S.; Salgado, E.N.; Herrmann, T.; Li, Z.; Grant, T.; Grigorieff, N.; Trapani, S.; Estrozi, L.F.; Harrison, S.C. In situ Structure of Rotavirus VP1 RNA-Dependent RNA Polymerase. J Mol Biol 2019, 431, 3124–3138. [Google Scholar] [CrossRef] [PubMed]
- Crawford S.E.; Ding S.D.; Greenberg H.B.; M.K., E. Rotaviruses; Howley PM., Ed. 2023.
- Periz, J.; Celma, C.; Jing, B.; Pinkney, J.N.; Roy, P.; Kapanidis, A.N. Rotavirus mRNAS are released by transcript-specific channels in the double-layered viral capsid. Proc Natl Acad Sci U S A 2013, 110, 12042–12047. [Google Scholar] [CrossRef] [PubMed]
- Tortorici, M.A.; Broering, T.J.; Nibert, M.L.; Patton, J.T. Template recognition and formation of initiation complexes by the replicase of a segmented double-stranded RNA virus. The Journal of biological chemistry 2003, 278, 32673–32682. [Google Scholar] [CrossRef] [PubMed]
- Tortorici M., A.; Shapiro B., A.; Patton J., T. A base-specific recognition signal in the 5' consensus sequence of rotavirus plus-strand RNAs promotes replication of the double-stranded RNA genome segments. RNA (New York, N.Y.) 2006, 12, 133–146. [Google Scholar] [CrossRef] [PubMed]
- Ding, K.; Celma, C.C.; Zhang, X.; Chang, T.; Shen, W.; Atanasov, I.; Roy, P.; Zhou, Z.H. In situ structures of rotavirus polymerase in action and mechanism of mRNA transcription and release. Nature Communications 2019, 10, 2216. [Google Scholar] [CrossRef]
- Lu, X.; McDonald, S.M.; Tortorici, M.A.; Tao, Y.J.; Vasquez-Del Carpio, R.; Nibert, M.L.; Patton, J.T.; Harrison, S.C. Mechanism for coordinated RNA packaging and genome replication by rotavirus polymerase VP1. Structure (London, England : 1993) 2008, 16, 1678–1688. [Google Scholar] [CrossRef]
- Barro, M.; Mandiola, P.; Chen, D.; Patton, J.T.; Spencer, E. Identification of sequences in rotavirus mRNAs important for minus strand synthesis using antisense oligonucleotides. Virology 2001, 288, 71–80. [Google Scholar] [CrossRef]
- Chen, D.; Barros, M.; Spencer, E.; Patton, J.T. Features of the 3'-consensus sequence of rotavirus mRNAs critical to minus strand synthesis. Virology 2001, 282, 221–229. [Google Scholar] [CrossRef]
- Navarro, A.; Trask S., D.; Patton J., T. Generation of genetically stable recombinant rotaviruses containing novel genome rearrangements and heterologous sequences by reverse genetics. J Virol 2013, 87, 6211–6220. [Google Scholar] [CrossRef] [PubMed]
- Patton J., T.; Chnaiderman, J.; Spencer, E. Open reading frame in rotavirus mRNA specifically promotes synthesis of double-stranded RNA: template size also affects replication efficiency. Virology 1999, 264, 167–180. [Google Scholar] [CrossRef] [PubMed]
- Chen, D.; Patton J., T. Rotavirus RNA replication requires a single-stranded 3' end for efficient minus-strand synthesis. J Virol 1998, 72, 7387–7396. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Manktelow, E.; von Kirchbach, J.C.; Gog, J.R.; Desselberger, U.; Lever, A.M. Genomic analysis of codon, sequence and structural conservation with selective biochemical-structure mapping reveals highly conserved and dynamic structures in rotavirus RNAs with potential cis-acting functions. Nucleic Acids Res 2010, 38, 7718–7735. [Google Scholar] [CrossRef] [PubMed]
- Ogden, K.M.; Ramanathan, H.N.; Patton, J.T. Mutational analysis of residues involved in nucleotide and divalent cation stabilization in the rotavirus RNA-dependent RNA polymerase catalytic pocket. Virology 2012, 431, 12–20. [Google Scholar] [CrossRef] [PubMed]
- Steger, C.; Brown, M.; Sullivan, O.; Boudreaux, C.; Cohen, C.; LaConte, L.; McDonald, S. In Vitro Double-Stranded RNA Synthesis by Rotavirus Polymerase Mutants with Lesions at Core Shell Contact Sites. Journal of Virology 2019, 93. [Google Scholar] [CrossRef]
- Tao, Y.; Farsetta, D.L.; Nibert, M.L.; Harrison, S.C. RNA synthesis in a cage--structural studies of reovirus polymerase lambda3. Cell 2002, 111, 733–745. [Google Scholar] [CrossRef]
- Zeng, C.Q.; Estes, M.K.; Charpilienne, A.; Cohen, J. The N terminus of rotavirus VP2 is necessary for encapsidation of VP1 and VP3. J Virol 1998, 72, 201–208. [Google Scholar] [CrossRef]
- Gridley C., L.; Patton J., T. Regulation of rotavirus polymerase activity by inner capsid proteins. Current opinion in virology 2014, 9, 31–38. [Google Scholar] [CrossRef]
- Patton, J.T.; Jones, M.T.; Kalbach, A.N.; He, Y.W.; Xiaobo, J. Rotavirus RNA polymerase requires the core shell protein to synthesize the double-stranded RNA genome. J Virol 1997, 71, 9618–9626. [Google Scholar] [CrossRef]
- Diebold, O.; Gonzalez, V.; Venditti, L.; Sharp, C.; Blake, R.A.; Tan, W.S.; Stevens, J.; Caddy, S.; Digard, P.; Borodavka, A.; et al. Using Species a Rotavirus Reverse Genetics to Engineer Chimeric Viruses Expressing SARS-CoV-2 Spike Epitopes. J Virol 2022, 96, e0048822. [Google Scholar] [CrossRef] [PubMed]
- Kanai, Y.; Komoto, S.; Kawagishi, T.; Nouda, R.; Nagasawa, N.; Onishi, M.; Matsuura, Y.; Taniguchi, K.; Kobayashi, T. Entirely plasmid-based reverse genetics system for rotaviruses. Proceedings of the National Academy of Sciences 2017, 114, 2349. [Google Scholar] [CrossRef] [PubMed]
- Komoto, S.; Fukuda, S.; Ide, T.; Ito, N.; Sugiyama, M.; Yoshikawa, T.; Murata, T.; Taniguchi, K. Generation of Recombinant Rotaviruses Expressing Fluorescent Proteins by Using an Optimized Reverse Genetics System. Journal of Virology 2018, 92, e00588-00518. [Google Scholar] [CrossRef] [PubMed]
- Komoto, S.; Kanai, Y.; Fukuda, S.; Kugita, M.; Kawagishi, T.; Ito, N.; Sugiyama, M.; Matsuura, Y.; Kobayashi, T.; Taniguchi, K. Reverse Genetics System Demonstrates that Rotavirus Nonstructural Protein NSP6 Is Not Essential for Viral Replication in Cell Culture. Journal of Virology 2017, 91, e00695-00617. [Google Scholar] [CrossRef] [PubMed]
- Percy, N.; Barclay, W.S.; Sullivan, M.; Almond, J.W. A poliovirus replicon containing the chloramphenicol acetyltransferase gene can be used to study the replication and encapsidation of poliovirus RNA. Journal of Virology 1992, 66, 5040–5046. [Google Scholar] [CrossRef] [PubMed]
- Sidhu, M.S.; et al. Rescue of Synthetic Measles Virus Minireplicons: Measles Genomic Termini Direct Efficient Expression and Propagation of a Reporter Gene. Virology 1995, 208, 800–807. [Google Scholar] [CrossRef] [PubMed]
- Groseth, A.; et al. RNA Polymerase I-Driven Minigenome System for Ebola Viruses. Journal of Virology 2005, 79, 4425–4433. [Google Scholar] [CrossRef]
- Lutz, A.; et al. Virus-inducible reporter genes as a tool for detecting and quantifying influenza A virus replication. Journal of Virological Methods 2005, 126, 13–20. [Google Scholar] [CrossRef] [PubMed]
- Simmonds, P. SSE: a nucleotide and amino acid sequence analysis platform. BMC Research Notes 2012, 5, 50. [Google Scholar] [CrossRef]
- Matrosovich, M.; Matrosovich, T.; Garten, W.; Klenk, H.-D. New low-viscosity overlay medium for viral plaque assays. Virology Journal 2006, 3, 63. [Google Scholar] [CrossRef]
- Arnold M.; Patton J.T.; McDonald S.M. Culturing, storage, and quantification of rotaviruses; 2009; Vol. Chapter 15.
- Wise, HM.; Foeglein, A.; Sun, J.; Dalton Rosa, M.; Patel, S.; Howard, W.; Anderson E., C.; Barclay W., S.; Digard, P. A Complicated Message: Identification of a Novel PB1-Related Protein Translated from Influenza A Virus Segment 2 mRNA. Journal of Virology 2009, 83, 8021–8031. [Google Scholar] [CrossRef] [PubMed]
- Roner M., R.; Joklik W., K. Reovirus reverse genetics: Incorporation of the CAT gene into the reovirus genome. Proc Natl Acad Sci U S A 2001, 98, 8036–8041. [Google Scholar] [CrossRef]
- Desselberger, U. Rotaviruses. Virus Res 2014, 190, 75–96. [Google Scholar] [CrossRef] [PubMed]
- Patton, J.T.; Jones, M.T.; Kalbach, A.N.; He, Y.W.; Xiaobo, J. Rotavirus RNA polymerase requires the core shell protein to synthesize the double-stranded RNA genome. 1997, 71, 9618–9626. [Google Scholar] [CrossRef] [PubMed]
- te Velthuis A; Long J; Barclay W. Assays to Measure the Activity of Influenza Virus Polymerase. 2018; Vol. 1836, pp. 343–374.
- Noton, S.L.; Cowton, V.M.; Zack, C.R.; McGivern, D.R.; Fearns, R. Evidence that the polymerase of respiratory syncytial virus initiates RNA replication in a nontemplated fashion. Proc Natl Acad Sci U S A 2010, 107, 10226–10231. [Google Scholar] [CrossRef] [PubMed]
- Paul, A.V.; Rieder, E.; Kim, D.W.; Boom, J.H.v.; Wimmer, E. Identification of an RNA Hairpin in Poliovirus RNA That Serves as the Primary Template in the In Vitro Uridylylation of VPg. 2000, 74, 10359–10370. [Google Scholar] [CrossRef] [PubMed]
- Wehrfritz, J.M.; Boyce, M.; Mirza, S.; Roy, P. Reconstitution of bluetongue virus polymerase activity from isolated domains based on a three-dimensional structural model. 2007, 86, 83–94. [Google Scholar] [CrossRef] [PubMed]
- Vende, P.; Piron, M.; Castagné, N.; Poncet, D. Efficient Translation of Rotavirus mRNA Requires Simultaneous Interaction of NSP3 with the Eukaryotic Translation Initiation Factor eIF4G and the mRNA 3′ End. Journal of Virology 2000, 74, 7064–7071. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Tacuba, L.; Feng, N.; Meade N., J.; Mellits K., H.; Jaïs P., H.; Yasukawa L., L.; Resch T., K.; Jiang, B.; López, S.; Ding, S.; et al. An Optimized Reverse Genetics System Suitable for Efficient Recovery of Simian, Human, and Murine-Like Rotaviruses. J Virol 2020, 94. [Google Scholar] [CrossRef]
- Zheng, H.; Palese, P.; García-Sastre, A. Nonconserved nucleotides at the 3' and 5' ends of an influenza A virus RNA play an important role in viral RNA replication. Virology 1996, 217, 242–251. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




