Submitted:
23 April 2024
Posted:
23 April 2024
Read the latest preprint version here
Abstract
Keywords:
1. Introduction
The Close Relationship between Autoimmunity and Aging
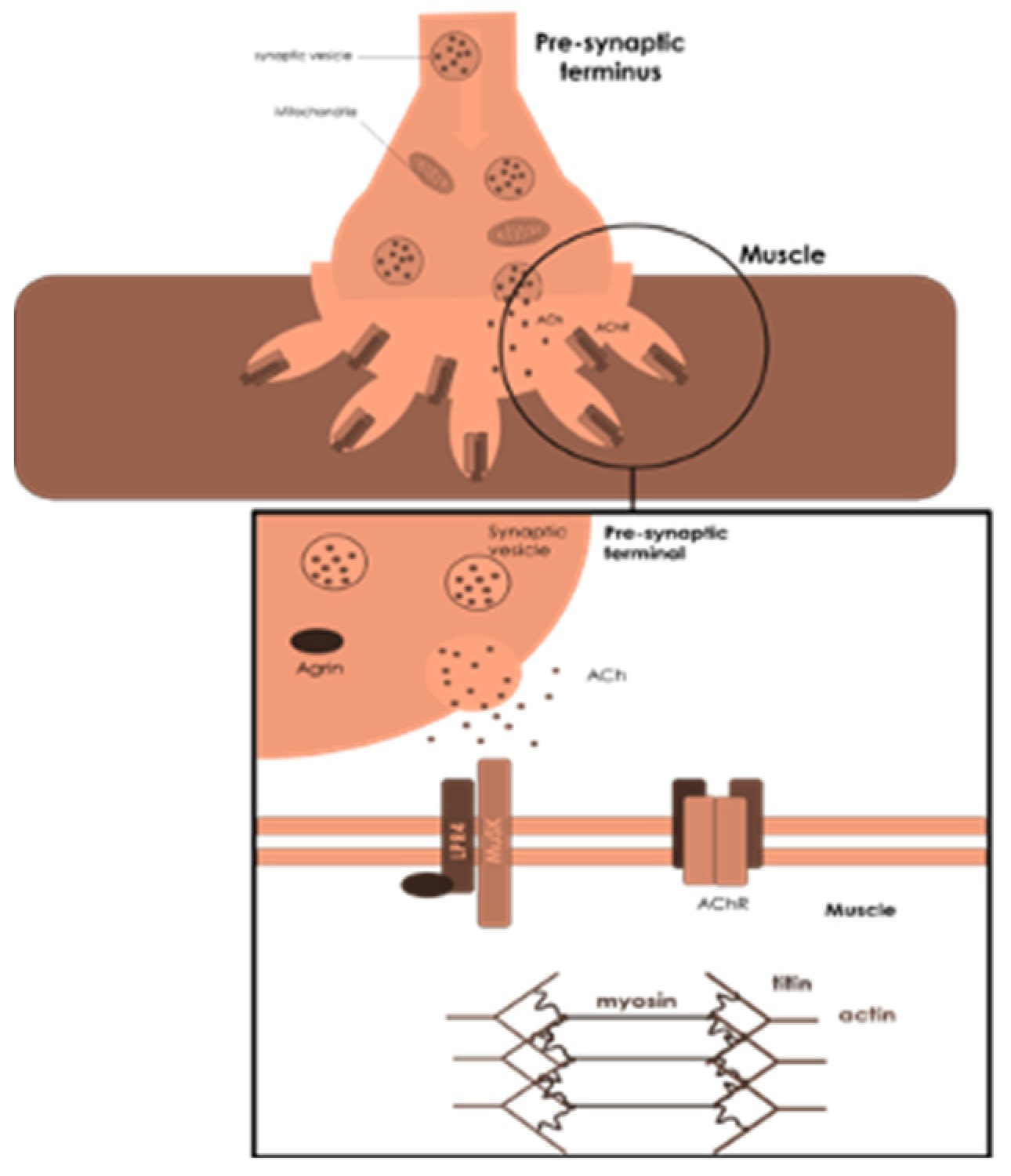
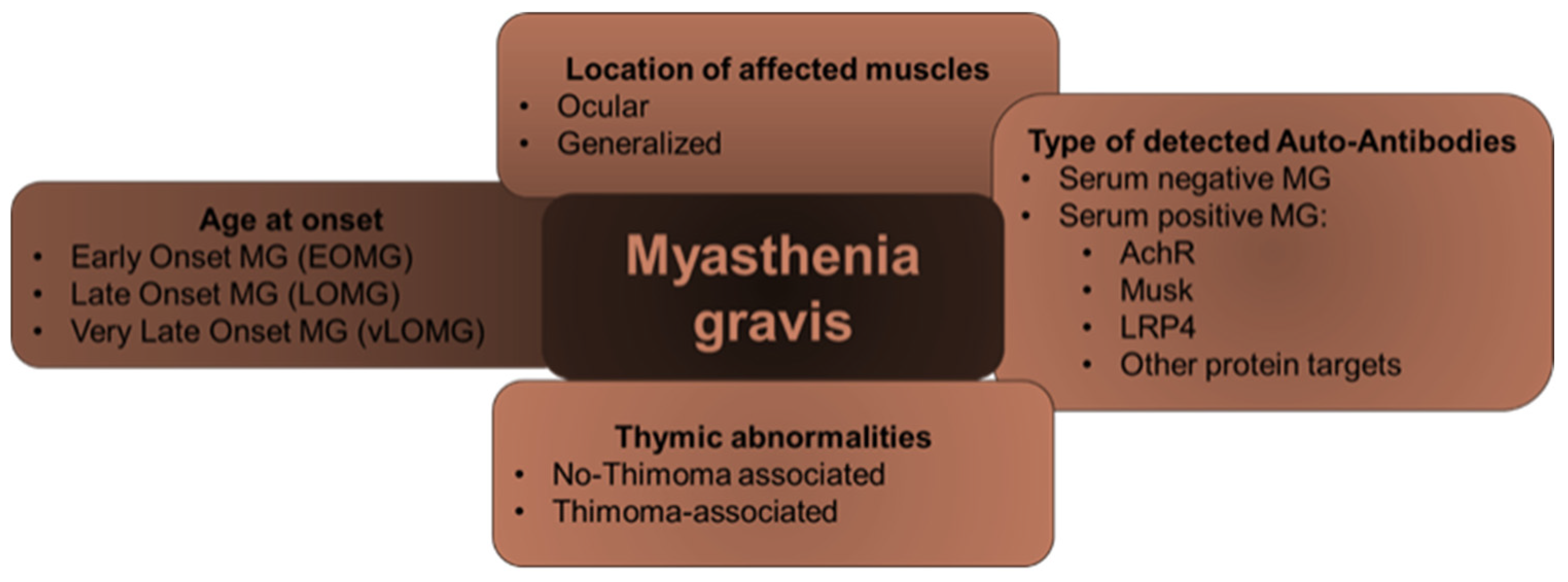
2. MG: Its Features
2.1. The Contribution of Age- and Sex-Related Factors in Etiopathogenesis of MG
2.2. Clinical Presentation of MG According to Age and Gender
3. Genetic Susceptibility for MG and Epigenetic Modifications
4. Clinical and Biological Heterogeneity in MG Associated with Typical Change in the Thymus Morphology According to Age and Sex
5. Distinctive Biomarkers in Early and Late-Onset MG, According to Sex/Gender and Age Differences
6. Typical MG Autoantibodies in Relationship to Age and Sex-Related Factors
7. Neuropsychology of MG Patients
8. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Balistreri, C. R., Candore, G., Accardi, G., Bova, M., Buffa, S., Bulati, M., Forte, G. I., Listì, F., Martorana, A., Palmeri, M., Pellicanò, M., Vaccarino, L., Scola, L., Lio, D., & Colonna-Romano, G. (2012). Genetics of longevity. data from the studies on Sicilian centenarians. Immunity & ageing: I & A, 9(1), 8. [CrossRef]
- Santoro, A., Bientinesi, E., & Monti, D. (2021). Immunosenescence and inflammaging in the aging process: Age-related diseases or longevity?. Ageing research reviews, 71, 101422. [CrossRef]
- Wang, Y., Dong, C., Han, Y., Gu, Z., & Sun, C. (2022). Immunosenescence, aging and successful aging. Frontiers in immunology, 13, 942796. [CrossRef]
- Hohman, L. S., & Osborne, L. C. (2022). A gut-centric view of aging: Do intestinal epithelial cells contribute to age-associated microbiota changes, inflammaging, and immunosenescence?. Aging cell, 21(9), e13700. [CrossRef]
- Bulut, O., Kilic, G., & Domínguez-Andrés, J. (2022). Immune Memory in Aging: A Wide Perspective Covering Microbiota, Brain, Metabolism, and Epigenetics. Clinical reviews in allergy & immunology, 63(3), 499–529. [CrossRef]
- Alrosan, A. Z., Heilat, G. B., Alrosan, K., Aleikish, A. A., Rabbaa, A. N., Shakhatreh, A. M., Alshalout, E. M., & Al Momany, E. M. A. (2024). Autonomic brain functioning and age-related health concerns. Current research in physiology, 7, 100123. [CrossRef]
- Balistreri, C. R., & Monastero, R. (2023). Neuroinflammation and Neurodegenerative Diseases: How Much Do We Still Not Know?. Brain sciences, 14(1), 19. [CrossRef]
- Schirò, G., Iacono, S., & Balistreri, C. R. (2023). The Role of Human Microbiota in Myasthenia Gravis: A Narrative Review. Neurology international, 15(1), 392–404. [CrossRef]
- Balistreri, C. R., Candore, G., Accardi, G., Colonna-Romano, G., & Lio, D. (2013). NF-κB pathway activators as potential ageing biomarkers: Targets for new therapeutic strategies. Immunity & ageing: I & A, 10(1), 24. [CrossRef]
- Jeejeebhoy K. N. (2012). Malnutrition, fatigue, frailty, vulnerability, sarcopenia and cachexia: Overlap of clinical features. Current opinion in clinical nutrition and metabolic care, 15(3), 213–219. [CrossRef]
- Bowirrat A. (2022). Immunosenescence and Aging: Neuroinflammation Is a Prominent Feature of Alzheimer's Disease and Is a Likely Contributor to Neurodegenerative Disease Pathogenesis. Journal of personalized medicine, 12(11), 1817. [CrossRef]
- Balistreri C. R. (2022). Promising Strategies for Preserving Adult Endothelium Health and Reversing Its Dysfunction: From Liquid Biopsy to New Omics Technologies and Noninvasive Circulating Biomarkers. International journal of molecular sciences, 23(14), 7548. [CrossRef]
- Vaiserman, A., De Falco, E., Koliada, A., Maslova, O., & Balistreri, C. R. (2019). Anti-ageing gene therapy: Not so far away?. Ageing research reviews, 56, 100977. [CrossRef]
- Balistreri C. R. (2018). Anti-Inflamm-Ageing and/or Anti-Age-Related Disease Emerging Treatments: A Historical Alchemy or Revolutionary Effective Procedures?. Mediators of inflammation, 2018, 3705389. [CrossRef]
- Lio, D., Scola, L., Giarratana, R. M., Candore, G., Colonna-Romano, G., Caruso, C., & Balistreri, C. R. (2021). SARS CoV2 infection _The longevity study perspectives. Ageing research reviews, 67, 101299. [CrossRef]
- Fulop, T. (2020) ‘Immunosenescence is both functional / adaptive and dysfunctional / maladaptive’, pp. 521–536.
- Ray, D. and Yung, R. (2018) ‘Immune senescence, epigenetics and autoimmunity’, Clinical Immunology, 196, pp. 59–63. [CrossRef]
- Nussinovitch, U. and Shoenfeld, Y. (2012) ‘The role of gender and organ specific autoimmunity’, Autoimmunity Reviews. [CrossRef]
- Wiendl, H., Abicht, A., Chan, A., Della Marina, A., Hagenacker, T., Hekmat, K., Hoffmann, S., Hoffmann, H. S., Jander, S., Keller, C., Marx, A., Melms, A., Melzer, N., Müller-Felber, W., Pawlitzki, M., Rückert, J. C., Schneider-Gold, C., Schoser, B., Schreiner, B., Schroeter, M., … Meisel, A. (2023). Guideline for the management of myasthenic syndromes. Therapeutic advances in neurological disorders, 16, 17562864231213240. [CrossRef]
- García Estévez, D. A., & Pardo Fernández, J. (2023). Myasthenia gravis. Update on diagnosis and therapy. Miastenia gravis. Actualización diagnóstica y terapéutica. Medicina clinica, 161(3), 119–127. [CrossRef]
- Iyer, S. R., Shah, S. B., & Lovering, R. M. (2021). The Neuromuscular Junction: Roles in Aging and Neuromuscular Disease. International journal of molecular sciences, 22(15), 8058. [CrossRef]
- Bubuioc, A. M., Kudebayeva, A., Turuspekova, S., Lisnic, V., & Leone, M. A. (2021). The epidemiology of myasthenia gravis. Journal of medicine and life, 14(1), 7–16. [CrossRef]
- Mishra, A. K., & Varma, A. (2023). Myasthenia Gravis: A Systematic Review. Cureus, 15(12), e50017. [CrossRef]
- Zhu, Y., Wang, B., Hao, Y., & Zhu, R. (2023). Clinical features of myasthenia gravis with neurological and systemic autoimmune diseases. Frontiers in immunology, 14, 1223322. [CrossRef]
- Deymeer F. (2020). Myasthenia gravis: MuSK MG, late-onset MG and ocular MG. Acta myologica: Myopathies and cardiomyopathies: Official journal of the Mediterranean Society of Myology, 39(4), 345–352. [CrossRef]
- Fan, L., Ma, S., Yang, Y., Yan, Z., Li, J., & Li, Z. (2019). Clinical differences of early and late-onset myasthenia gravis in 985 patients. Neurological research, 41(1), 45–51. [CrossRef]
- Cortés-Vicente, E., Álvarez-Velasco, R., Segovia, S., Paradas, C., Casasnovas, C., Guerrero-Sola, A., Pardo, J., Ramos-Fransi, A., Sevilla, T., López de Munain, A., Gómez, M. T., Jericó, I., Gutiérrez-Gutiérrez, G., Pelayo-Negro, A. L., Martín, M. A., Mendoza, M. D., Morís, G., Rojas-Garcia, R., Díaz-Manera, J., Querol, L., … Illa, I. (2020). Clinical and therapeutic features of myasthenia gravis in adults based on age at onset. Neurology, 94(11), e1171–e1180. [CrossRef]
- Renton, A. E., Pliner, H. A., Provenzano, C., Evoli, A., Ricciardi, R., Nalls, M. A., Marangi, G., Abramzon, Y., Arepalli, S., Chong, S., Hernandez, D. G., Johnson, J. O., Bartoccioni, E., Scuderi, F., Maestri, M., Gibbs, J. R., Errichiello, E., Chiò, A., Restagno, G., Sabatelli, M., … Traynor, B. J. (2015). A genome-wide association study of myasthenia gravis. JAMA neurology, 72(4), 396–404. [CrossRef]
- Li, J., Wang, F., Li, Z., Feng, J., Men, Y., Han, J., Xia, J., Zhang, C., Han, Y., Chen, T., Zhao, Y., Zhou, S., Da, Y., Chai, G., & Hao, J. (2024). Integrative multi-omics analysis identifies genetically supported druggable targets and immune cell specificity for myasthenia gravis. Journal of translational medicine, 22(1), 302. [CrossRef]
- Chia, R., Saez-Atienzar, S., Murphy, N., Chiò, A., Blauwendraat, C., International Myasthenia Gravis Genomics Consortium, Roda, R. H., Tienari, P. J., Kaminski, H. J., Ricciardi, R., Guida, M., De Rosa, A., Petrucci, L., Evoli, A., Provenzano, C., Drachman, D. B., & Traynor, B. J. (2022). Identification of genetic risk loci and prioritization of genes and pathways for myasthenia gravis: A genome-wide association study. Proceedings of the National Academy of Sciences of the United States of America, 119(5), e2108672119. [CrossRef]
- Zhu, Z., Chen, X., Wang, C., & Cheng, L. (2022). Novel genes/loci validate the small effect size of ERBB2 in patients with myasthenia gravis. Proceedings of the National Academy of Sciences of the United States of America, 119(36), e2207273119. [CrossRef]
- Spagni, G., Todi, L., Monte, G., Valentini, M., Di Sante, G., Damato, V., Marino, M., Evoli, A., Lantieri, F., & Provenzano, C. (2021). Human Leukocyte Antigen Class II associations in late-onset Myasthenia Gravis. Annals of clinical and translational neurology, 8(3), 656–665. [CrossRef]
- Hong, Y., Li, H. F., Romi, F., Skeie, G. O., & Gilhus, N. E. (2018). HLA and MuSK-positive myasthenia gravis: A systemic review and meta-analysis. Acta neurologica Scandinavica, 138(3), 219–226. [CrossRef]
- Panhuber, A., Lamorte, G., Bruno, V., Cetin, H., Bauer, W., Höftberger, R., Erber, A. C., Frommlet, F., & Koneczny, I. (2022). A systematic review and meta-analysis of HLA class II associations in patients with IgG4 autoimmunity. Scientific reports, 12(1), 9229. [CrossRef]
- Zhong, H., Zhao, C., & Luo, S. (2019). HLA in myasthenia gravis: From superficial correlation to underlying mechanism. Autoimmunity reviews, 18(9), 102349. [CrossRef]
- Salvado, M., Caro, J. L., Garcia, C., Rudilla, F., Zalba-Jadraque, L., Lopez, E., Sanjuan, E., Gamez, J., & Vidal-Taboada, J. M. (2022). HLA-DQB1*05:02, *05:03, and *03:01 alleles as risk factors for myasthenia gravis in a Spanish cohort. Neurological sciences: Official journal of the Italian Neurological Society and of the Italian Society of Clinical Neurophysiology, 43(8), 5057–5065. [CrossRef]
- Nicolì, V., Tabano, S. M., Colapietro, P., Maestri, M., Ricciardi, R., Stoccoro, A., Fontana, L., Guida, M., Miozzo, M., Coppedè, F., & Migliore, L. (2022). Preferential X Chromosome Inactivation as a Mechanism to Explain Female Preponderance in Myasthenia Gravis. Genes, 13(4), 696. [CrossRef]
- Asmail, A., Kesler, A., Kolb, H., Drory, V. E., & Karni, A. (2019). A tri-modal distribution of age-of-onset in female patients with myasthenia gravis is associated with the gender-related clinical differences. The International journal of neuroscience, 129(4), 313–319. [CrossRef]
- Marx, A., Pfister, F., Schalke, B., Saruhan-Direskeneli, G., Melms, A., & Ströbel, P. (2013). The different roles of the thymus in the pathogenesis of the various myasthenia gravis subtypes. Autoimmunity reviews, 12(9), 875–884. [CrossRef]
- Gradolatto, A., Nazzal, D., Foti, M., Bismuth, J., Truffault, F., Le Panse, R., & Berrih-Aknin, S. (2012). Defects of immunoregulatory mechanisms in myasthenia gravis: Role of IL-17. Annals of the New York Academy of Sciences, 1274, 40–47. [CrossRef]
- Molin, C.J., Westerberg, E. and Punga, A.R. (2016) ‘Profile of upregulated inflammatory proteins in sera of Myasthenia Gravis patients OPEN’. [CrossRef]
- Romi, F.R., Gilhus, N.E. and Luckman, S.P. (2008) ‘Serum matrix metalloproteinase-3 levels are elevated in myasthenia gravis’, Journal of Neuroimmunology, 195(1–2), 96–99. [CrossRef]
- Molin, C.J., Westerberg, E. and Punga, A.R. (2016) ‘Profile of upregulated inflammatory proteins in sera of Myasthenia Gravis patients OPEN’. [CrossRef]
- Berzi, A. et al. (2008) ‘BDNF and its receptors in human myasthenic thymus: Implications for cell fate in thymic pathology’, Journal of Neuroimmunology, 197(2), pp. 128–139. [CrossRef]
- Schirò, G., Iacono, S., Ragonese, P., Aridon, P., Salemi, G., & Balistreri, C. R. (2022). A Brief Overview on BDNF-Trk Pathway in the Nervous System: A Potential Biomarker or Possible Target in Treatment of Multiple Sclerosis?. Frontiers in neurology, 13, 917527. [CrossRef]
- Sabre, L., Evoli, A. and Punga, A.R. (2019) ‘Cognitive dysfunction in mice with passively induced MuSK antibody seropositive myasthenia gravis’, Journal of the neurological sciences, 399, 15–21. [CrossRef]
- Cortés-Vicente, E. et al. (2020a) ‘Clinical and therapeutic features of myasthenia gravis in adults based on age at onset’, Neurology, 94(11), e1171. [CrossRef]
- Lazaridis, K., & Tzartos, S. J. (2020). Myasthenia Gravis: Autoantibody Specificities and Their Role in MG Management. Frontiers in neurology, 11, 596981. [CrossRef]
- Živković, S.A., Clemens, P.R. and Lacomis, D. (2012) ‘Characteristics of late-onset myasthenia gravis’, Journal of Neurology, 259(10), 2167–2171. [CrossRef]
- Szczudlik, P., Szyluk, B., Lipowska, M., Ryniewicz, B., Kubiszewska, J., Dutkiewicz, M., Gilhus, N. E., & Kostera-Pruszczyk, A. (2014). Antititin antibody in early- and late-onset myasthenia gravis. Acta neurologica Scandinavica, 130(4), 229–233. [CrossRef]
- Vitturi, B. K., Kim, A. I. H., Mitre, L. P., Pellegrinelli, A., & Valerio, B. C. O. (2021). Social, professional and neuropsychiatric outcomes in patients with myasthenia gravis. Neurological sciences: Official journal of the Italian Neurological Society and of the Italian Society of Clinical Neurophysiology, 42(1), 167–173. [CrossRef]
- Kaltsatou, A., Fotiou, D., Tsiptsios, D., & Orologas, A. (2015). Cognitive impairment as a central cholinergic deficit in patients with Myasthenia Gravis. BBA clinical, 3, 299–303. [CrossRef]
- Zhou, X., Cao, S., Hou, J., Gui, T., Zhu, F., & Xue, Q. (2023). Association between myasthenia gravis and cognitive disorders: A PRISMA-compliant meta-analysis. The International journal of neuroscience, 133(9), 987–998. [CrossRef]
- Law, C., Flaherty, C. V., & Bandyopadhyay, S. (2020). A Review of Psychiatric Comorbidity in Myasthenia Gravis. Cureus, 12(7), e9184. [CrossRef]
- Tarawneh, R., D'Angelo, G., Crimmins, D., Herries, E., Griest, T., Fagan, A. M., Zipfel, G. J., Ladenson, J. H., Morris, J. C., & Holtzman, D. M. (2016). Diagnostic and Prognostic Utility of the Synaptic Marker Neurogranin in Alzheimer Disease. JAMA neurology, 73(5), 561–571. [CrossRef]
- Dong, D., Chong, M. K., Wu, Y., Kaminski, H., Cutter, G., Xu, X., Li, H., Zhao, C., Yin, J., Yu, S., & Zhu, J. (2020). Gender differences in quality of life among patients with myasthenia gravis in China. Health and quality of life outcomes, 18(1), 296. [CrossRef]
- Wang, J., Yan, C., Zhao, Z., Chen, H., Shi, Z., Du, Q., Zhang, Y., Qiu, Y., Lang, Y., Kong, L., Cai, L., & Zhou, H. (2021). Sexual dysfunction in patients with myasthenia gravis. Journal of neuroimmunology, 358, 577669. [CrossRef]
- Tannemaat, M. R., Huijbers, M. G., & Verschuuren, J. J. G. M. (2024). Myasthenia gravis-Pathophysiology, diagnosis, and treatment. Handbook of clinical neurology, 200, 283–305. [CrossRef]
- Crisafulli, S., Boccanegra, B., Carollo, M., Bottani, E., Mantuano, P., Trifirò, G., & De Luca, A. (2024). Myasthenia Gravis Treatment: From Old Drugs to Innovative Therapies with a Glimpse into the Future. CNS drugs, 38(1), 15–32. [CrossRef]
- Matic, A., Alfaidi, N., & Bril, V. (2023). An evaluation of rozanolixizumab-noli for the treatment of anti-AChR and anti-MuSK antibody-positive generalized myasthenia gravis. Expert opinion on biological therapy, 23(12), 1163–1171. [CrossRef]
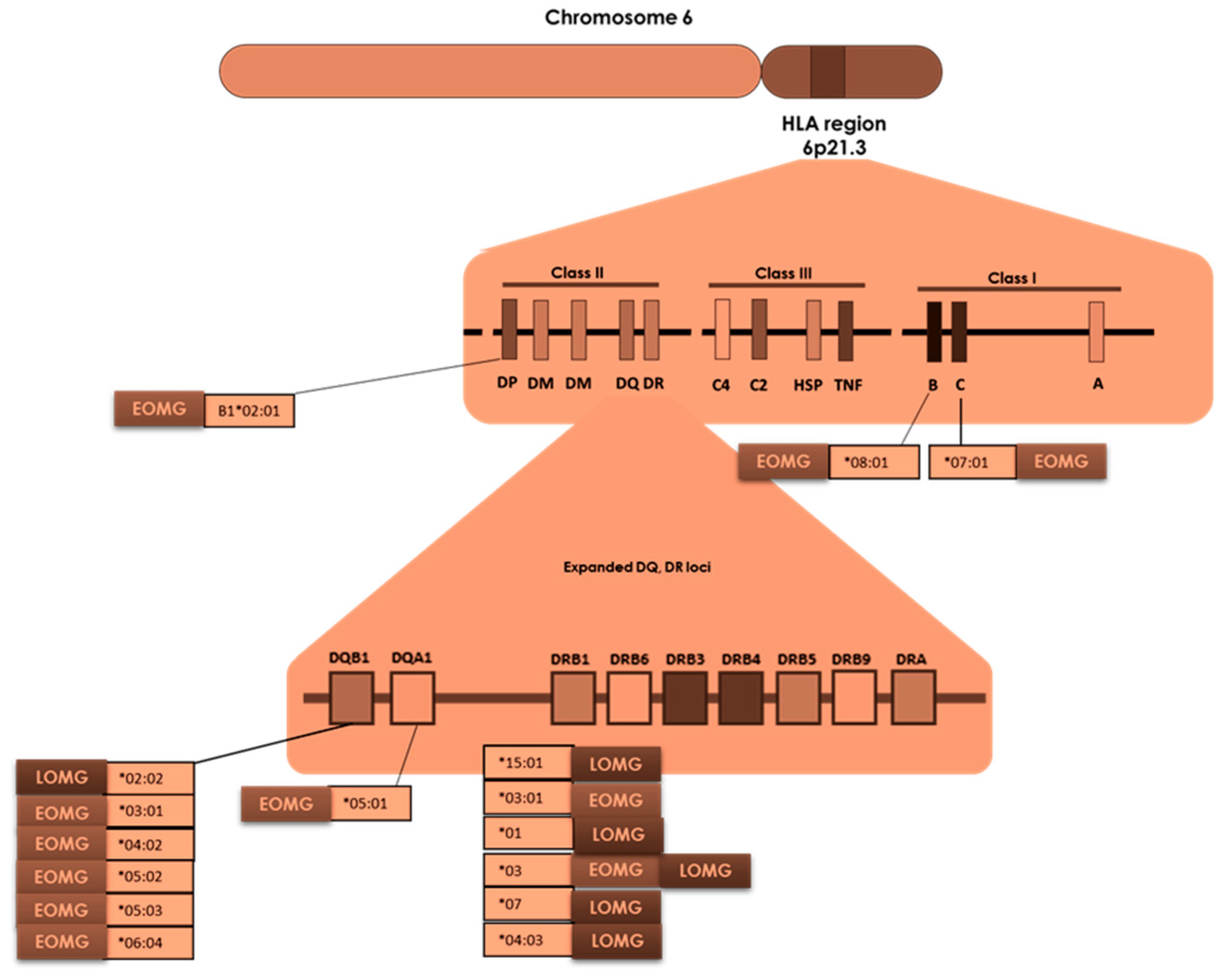
| Alleles/Haplotypes | Effects (risk or protection) | Population |
|---|---|---|
| HLA-B*08 | EOMG | Norwegian population |
| HLA-B*08:01 | EOMG | Swedish population |
| HLA-B*08:01 | EOMG | Saudi population |
| HLA-B*08:01 | EOMG |
Turkey population |
| HLA-C*07:01 | ||
|
HLA-DRB*15:01 |
LOMG | Norwegian, Italian and Swedish populations |
| HLA-DRB1*15:01 | LOMG | Norwegian population |
| HLA-DRB1*03:01 | EOMG | Multicenter study (Sweden Norway; England; France; Netherlands; Germany; Poland) |
| HLA-DRB1*03 | Increased risk for both LOMG and EOMG, more with EOMG | North of Portugal |
| HLA-DRB1*01 | LOMG | North of Portugal |
| HLA-DRB1*07-DQB1*02 | LOMG | Italian population |
| HLA DRB1*07 | LOMG |
Multicentric study (data from a metanalysis) |
|
HLA-DRB1 0403 | ||
| HLA-DRB1*04:04 | EOMG | Norwegian population |
| HLA-DRw53 | EOMG | Japan population |
| HLA-DPB1*0201 | EOMG | Japan population |
| HLA-DQA1*05:01 | EOMG | Multicenter study (patients from Sweden, Norway, England, France, Netherlands, Germany and Poland) |
| HLA-DQB1*03 | EOMG | Japan population |
| HLA-DQB1*02:02 | EOMG | Spanish population |
| HLA-DQB1*03:01 | ||
| HLA-DQB1*04:02 | EOMG |
Patients from Southeast region of Texas |
| HLA-DQB1 *05:02 | ||
| HLA-DQB1*05:03 | ||
| HLA-DQB1*06:04 | ||
| HLA-DQB1∗05:02-DRB1∗16 | LOMG | Italian population |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




