Submitted:
22 April 2024
Posted:
23 April 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
2.1. Characteristics of the Patients
2.2. Identification of the Different NK Cell Subpopulations in Patients with ALL and IFN-γ, CD107a, NKp44, and NKG2D expression.
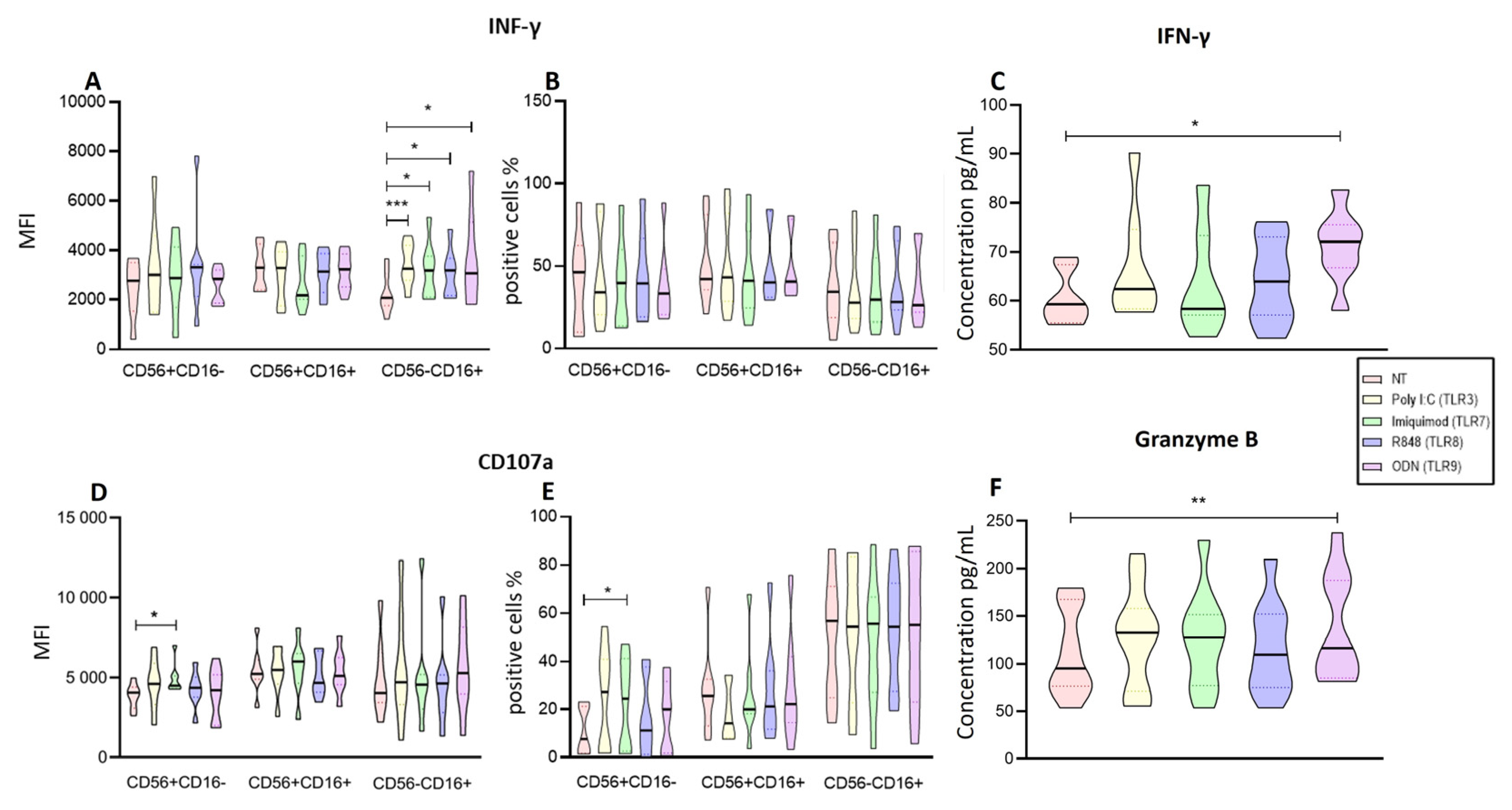
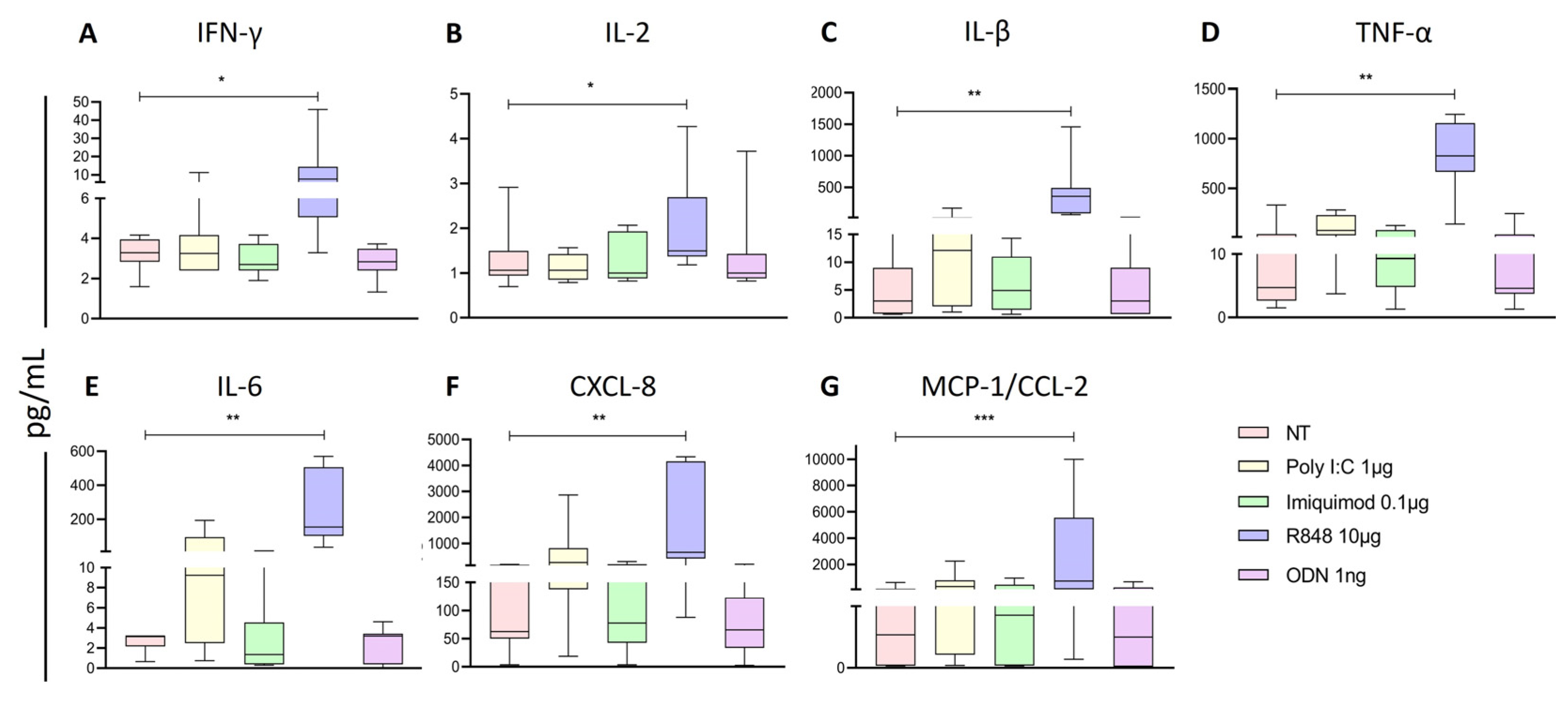
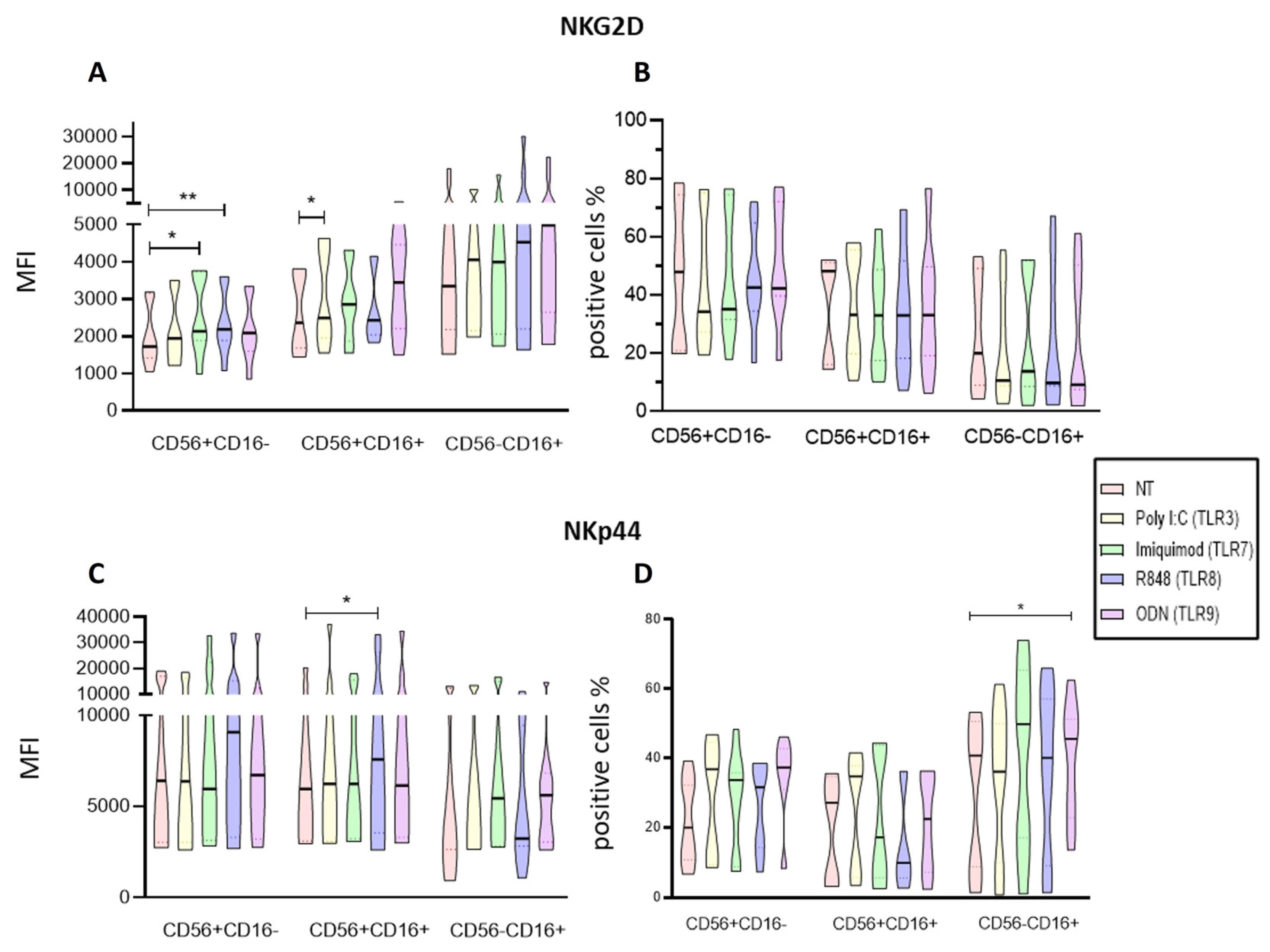
2.3. Poly I:C and R848 Increase the Activity of NK Subpopulations within the PBMC.
2.4. Imiquimod and ODN2006 Increase the Function of Enriched NK Cells Subpopulations
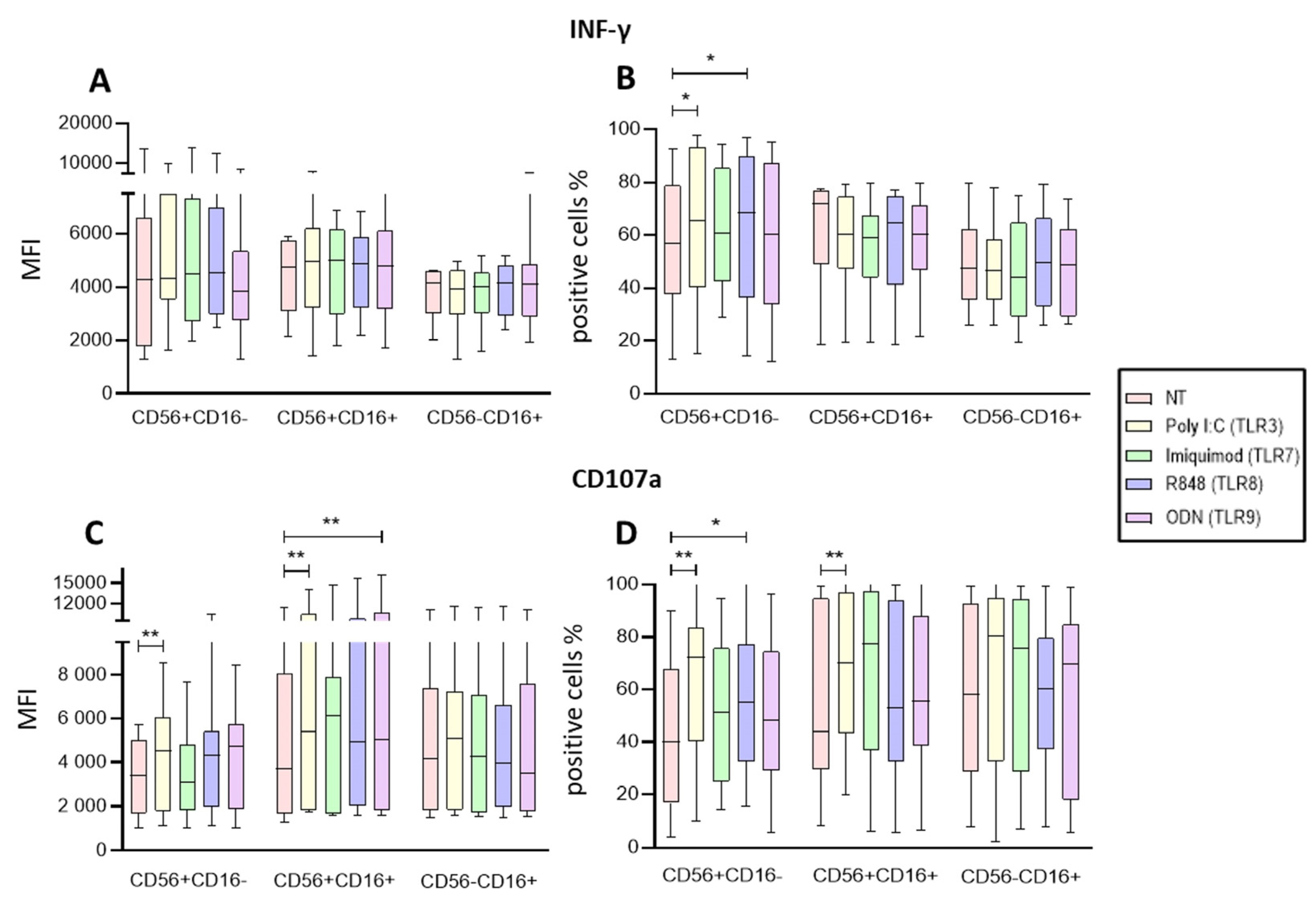
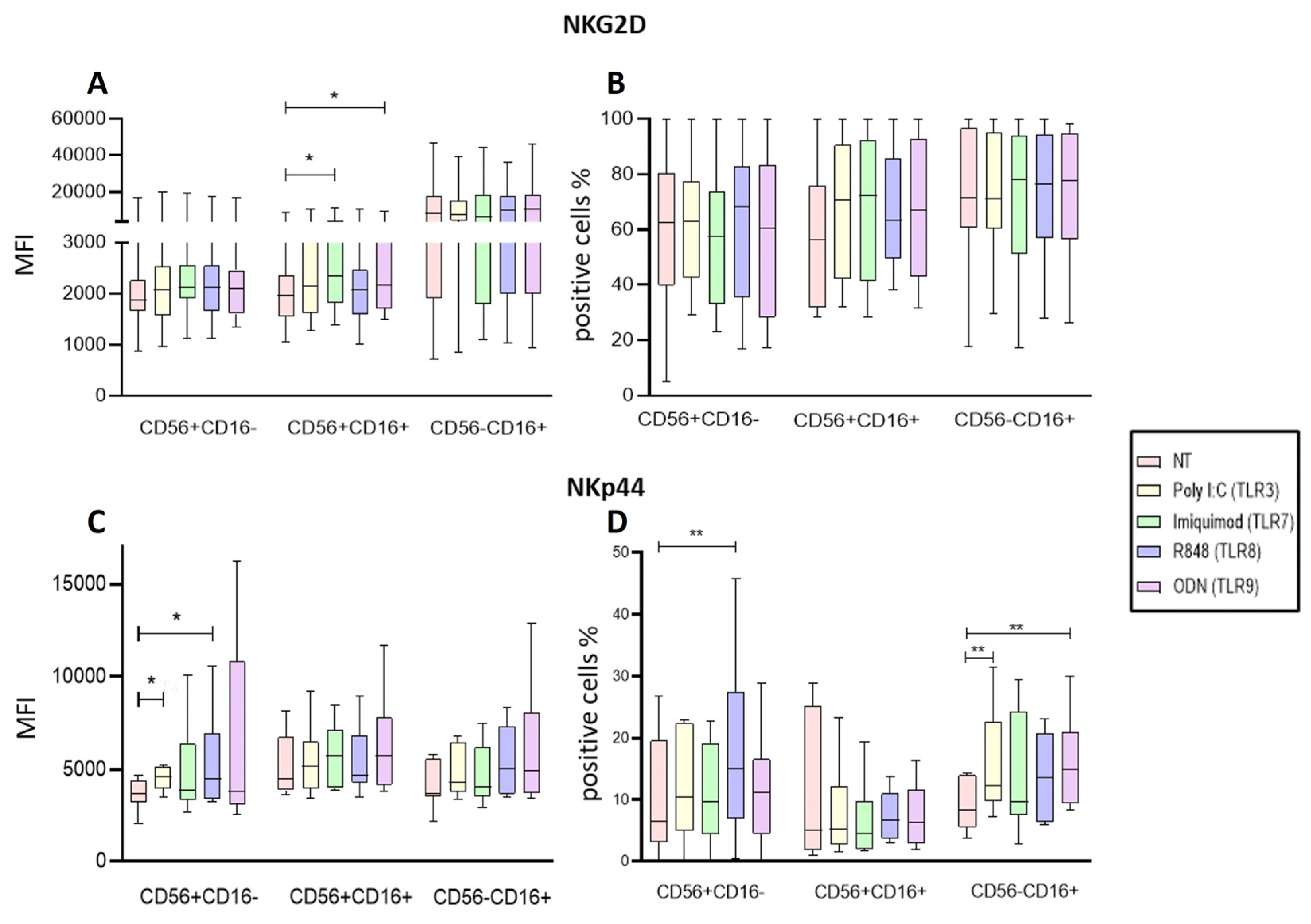
2.5. Endosomal TLRs Increase the Activation of NK Subpopulations within the PBMC.
2.6. Endosomal TLRs Ligands Increase the Activation of Enriched NK Cell Subpopulations
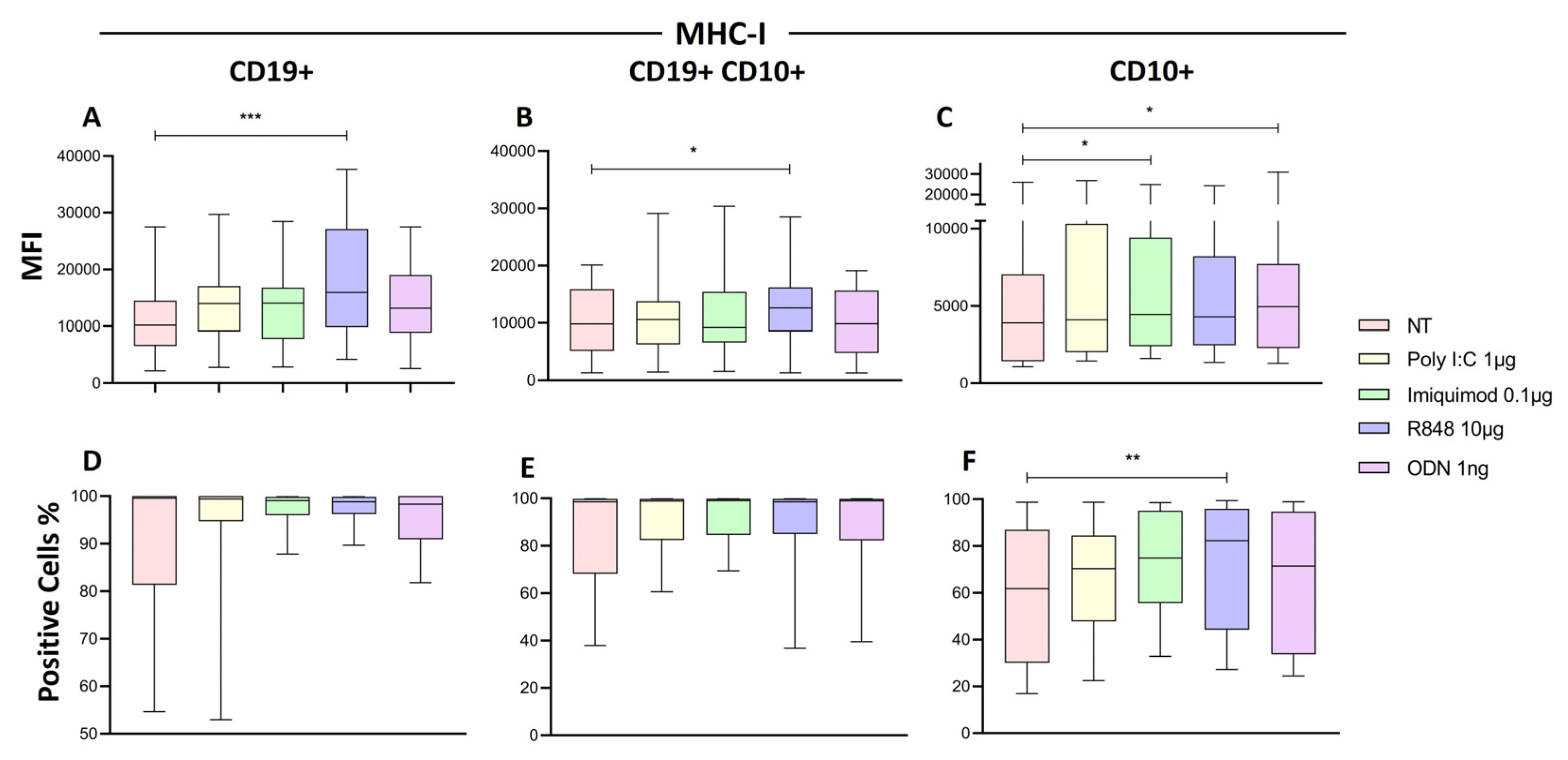
2.7. R848 Increase HLA-I Expression in Immature B Cells of Patients with ALL
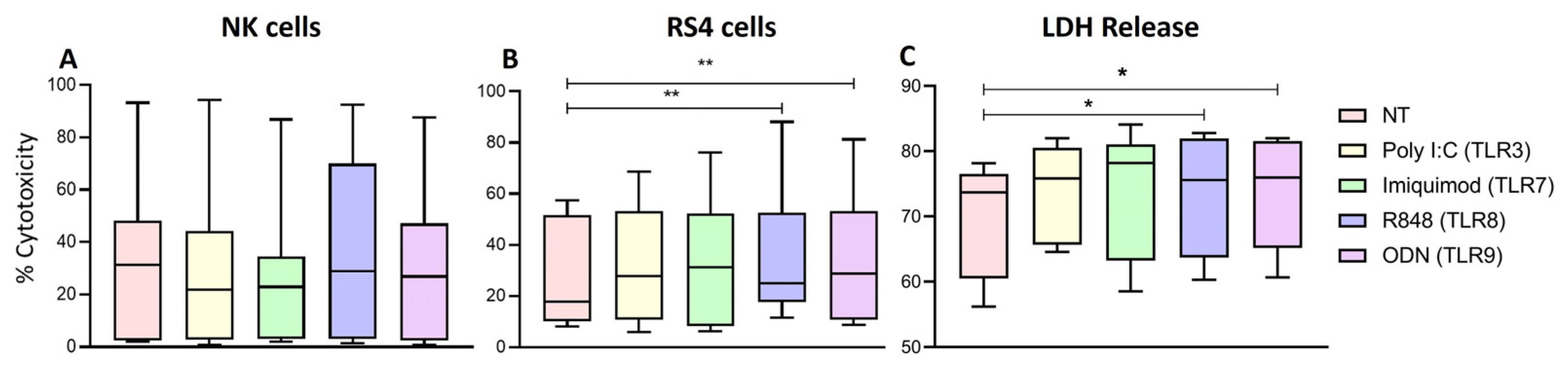
2.8. TLR8 and TLR9 Ligands Increase the Cytotoxic Activity of NK Cells
3. Discussion
4. Materials and Methods
4.1. Patients
4.2. Obtaining Mononuclear Cells, Isolating NK Cells, Culture and Activation with TLR Ligands
4.3. Characterization of NK Cells and Determination of Activation
4.4. Granzyme B Detection
4.5. Cytokines Determination
4.6. Interferon Gamma Determination
4.7. Cytotoxicity Assays
4.7.1. Zombie NIR Assay
4.7.2. LDH Release Assay
4.8. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Maldonado-Bernal C, Sánchez-Herrera D. Toll-Like Receptors and Natural Killer Cells. Toll-like Receptors, IntechOpen; 2020. [CrossRef]
- Cooper MA, Fehniger TA, Caligiuri MA. The biology of human natural killer-cell subsets. Trends Immunol 2001;22:633–40. [CrossRef]
- Huntington ND, Vosshenrich CAJ, Di Santo JP. Developmental pathways that generate natural-killer-cell diversity in mice and humans. Nat Rev Immunol 2007;7:703–14. [CrossRef]
- Tang D, Kang R, Coyne CB, Zeh HJ, Lotze MT. PAMPs and DAMPs: Signal 0s that spur autophagy and immunity. Immunol Rev 2012;249:158–75. [CrossRef]
- Jiménez-Morales S, Hidalgo-Miranda A, Ramírez-Bello J. Acute lymphoblastic leukemia: A genomic perspective. Bol Med Hosp Infant Mex 2017;74:13–26. [CrossRef]
- Ceppi F, Cazzaniga G, Colombini A, Biondi A, Conter V. Risk factors for relapse in childhood acute lymphoblastic leukemia: Prediction and prevention. Expert Rev Hematol 2015;8:57–70. [CrossRef]
- Greaves M. A causal mechanism for childhood acute lymphoblastic leukaemia. Nat Rev Cancer 2018;18:471–84. [CrossRef]
- Chiossone L, Dumas P-Y, Vienne M, Vivier E. Natural killer cells and other innate lymphoid cells in cancer. Nat Rev Immunol 2018;18:671–88. [CrossRef]
- Valenzuela-Vazquez L, Núñez-Enríquez JC, Sánchez-Herrera J, Jiménez-Hernández E, Martín-Trejo JA, Espinoza-Hernández LE; et al. Functional characterization of NK cells in Mexican pediatric patients with acute lymphoblastic leukemia: Report from the Mexican Interinstitutional Group for the Identification of the Causes of Childhood Leukemia. PLoS ONE 2020;15:1–15. [CrossRef]
- Guillerey C, Huntington ND, Smyth MJ. Targeting natural killer cells in cancer immunotherapy. Nat Immunol 2016;17:1025–36. [CrossRef]
- Rouce RH, Shaim H, Sekine T, Weber G, Ballard B, Ku S; et al. The TGF-β/SMAD pathway is an important mechanism for NK cell immune evasion in childhood B-acute lymphoblastic leukemia. Leukemia 2016;30:800–11. [CrossRef]
- Valenzuela-Vázquez L, Nuñez-Enriquez JC, Sánchez-Herrera J, Medina-Sanson A, Pérez-Saldivar ML, Jiménez-Hernández E; et al. NK cells with decreased expression of multiple activating receptors is a dominant phenotype in pediatric patients with acute lymphoblastic leukemia. Front Oncol 2022;12. [CrossRef]
- Kawasaki T, Kawai T. Toll-Like Receptor Signaling Pathways. Front Immunol 2014;0:461. [CrossRef]
- Guo Q, Zhang C. Critical role of Toll-like receptor signaling in NK cell activation. Chinese Science Bulletin 2012;57:3192–202. [CrossRef]
- Vacchelli E, Eggermont A, Sautè;s-Fridman C, Galon J, Zitvogel L, Kroemer G; et al. Trial watch toll-like receptor agonists for cancer therapy. Oncoimmunology 2013;2:1–14. [CrossRef]
- Barbalat R, Ewald SE, Mouchess ML, Barton GM. Nucleic Acid Recognition by the Innate Immune System. Annu Rev Immunol 2011;29:185–214. [CrossRef]
- Blasius AL, Beutler B. Intracellular Toll-like Receptors. Immunity 2010;32:305–15. [CrossRef]
- Roach JC, Glusman G, Rowen L, Kaur A, Purcell MK, Smith KD; et al. The evolution of vertebrate Toll-like receptors. Proceedings of the National Academy of Sciences 2005;102:9577–82. [CrossRef]
- Takeda K, Akira S. TLR signaling pathways. Semin Immunol 2004;16:3–9. [CrossRef]
- Akira S, Takeda K, Kaisho T. Toll-like receptors: Critical proteins linking innate and acquired immunity. Nature Immunology 2001 2:8 2001;2:675–80. [CrossRef]
- Rakoff-Nahoum S, Medzhitov R. Toll-like receptors and cancer. Nat Rev Cancer 2009;9:57–63. [CrossRef]
- Sánchez-Herrera D, Contreras-Ramos A, Jiménez-Hernández E, Medina-Sansón A, Giono-Cerezo S, Maldonado-Bernal C. NK Cell Subpopulation Is Altered and the Expression of TLR1 and TLR9 Is Decreased in Patients with Acute Lymphoblastic Leukemia. J Oncol 2021;2021:1–12. [CrossRef]
- Della Chiesa M, De Maria A, Muccio L, Bozzano F, Sivori S, Moretta L. Human NK Cells and Herpesviruses: Mechanisms of Recognition, Response and Adaptation. Front Microbiol 2019;10. [CrossRef]
- Cooper MA, Fehniger TA, Fuchs A, Colonna M, Caligiuri MA. NK cell and DC interactions. Trends Immunol 2004;25:47–52. [CrossRef]
- Guillerey C, Chow MT, Miles K, Olver S, Sceneay J, Takeda K; et al. Toll-like receptor 3 regulates NK cell responses to cytokines and controls experimental metastasis. Oncoimmunology 2015;4:1–11. [CrossRef]
- Gruijs M, Ganzevles SH, Stigter-van Walsum M, van der Mast R, van Ostaijen-ten Dam MM, Tuk CW; et al. NK Cell-Dependent Antibody-Mediated Immunotherapy Is Improved In Vitro and In Vivo When Combined with Agonists for Toll-like Receptor 2 in Head and Neck Cancer Models. Int J Mol Sci 2021;22:11057. [CrossRef]
- Li T, Yang Y, Song H, Li H, Cui A, Liu Y; et al. Activated NK cells kill hepatic stellate cells via p38/PI3K signaling in a TRAIL-involved degranulation manner. J Leukoc Biol 2019;105:695–704. [CrossRef]
- Samudio I, Rezvani K, Shaim H, Hofs E, Ngom M, Bu L; et al. UV-inactivated HSV-1 potently activates NK cell killing of leukemic cells. Blood, The Journal of the American Society of Hematology 2016;127:2575–86. [CrossRef]
- Gorski KS, Waller EL, Bjornton-Severson J, Hanten JA, Riter CL, Kieper WC; et al. Distinct indirect pathways govern human NK-cell activation by TLR-7 and TLR-8 agonists. Int Immunol 2006;18:1115–26. [CrossRef]
- Lauzon NM, Mian F, MacKenzie R, Ashkar AA. The direct effects of Toll-like receptor ligands on human NK cell cytokine production and cytotoxicity. Cell Immunol 2006;241:102–12. [CrossRef]
- Ronsley R, Kariminia A, Ng B, Mostafavi S, Reid G, Subrt P; et al. The TLR9 agonist (GNKG168) induces a unique immune activation pattern in vivo in children with minimal residual disease positive acute leukemia: Results of the TACL T2009-008 phase I study. Pediatr Hematol Oncol 2019;36:468–81. [CrossRef]
- Sivori S, Falco M, Della Chiesa M, Carlomagno S, Vitale M, Moretta L; et al. CpG and double-stranded RNA trigger human NK cells by toll-like receptors: Induction of cytokine release and cytotoxicity against tumors dendritic cells. Proc Natl Acad Sci U S A 2004;101:10116–21. [CrossRef]
- Khanna V, Kim H, Zhang W, Larson P, Shah M, Griffith TS; et al. Novel TLR 7/8 agonists for improving NK cell mediated antibody-dependent cellular cytotoxicity (ADCC). Sci Rep 2021;11:3346. [CrossRef]
- Portale F, Di Mitri D. NK Cells in Cancer: Mechanisms of Dysfunction and Therapeutic Potential. Int J Mol Sci 2023;24:9521. [CrossRef]
- Veneziani I, Alicata C, Moretta L, Maggi E. The Latest Approach of Immunotherapy with Endosomal TLR Agonists Improving NK Cell Function: An Overview. Biomedicines 2022;11:64. [CrossRef]
- Cooper MA, Fehniger TA, Turner SC, Chen KS, Ghaheri BA, Carson WE; et al. Human natural killer cells: A unique innate immunoregulatory role for the CD56bright subset. Blood 2000;96:3146–51. [CrossRef]
- Poli A, Michel T, Thérésine M, Andrès E, Hentges F, Zimmer J. CD56bright natural killer (NK) cells: An important NK cell subset. Immunology 2009;126:458–65. [CrossRef]
- Prager I, Liesche C, van Ooijen H, Urlaub D, Verron Q, Sandström N; et al. NK cells switch from granzyme B to death receptor–mediated cytotoxicity during serial killing. Journal of Experimental Medicine 2019;216:2113–27. [CrossRef]
- Khanna V, Kim H, Zhang W, Larson P, Shah M, Griffith TS; et al. Novel TLR 7/8 agonists for improving NK cell mediated antibody-dependent cellular cytotoxicity (ADCC). Sci Rep 2021;11:3346. [CrossRef]
- Cooper MA, Fehniger TA, Turner SC, Chen KS, Ghaheri BA, Ghayur T; et al. Human natural killer cells: A unique innate immunoregulatory role for the CD56bright subset. Blood 2001;97:3146–51. [CrossRef]
- Veneziani I, Alicata C, Pelosi A, Landolina N, Ricci B, D’Oria V; et al. Toll-like receptor 8 agonists improve NK-cell function primarily targeting CD56brightCD16- subset. J Immunother Cancer 2022;10. [CrossRef]
- Wang R, Jaw JJ, Stutzman NC, Zou Z, Sun PD. Natural killer cell-produced IFN-γ and TNF-α induce target cell cytolysis through up-regulation of ICAM-J Leukoc Biol 2012;91:299. [CrossRef]
- Montaldo E, Vitale C, Cottalasso F, Conte R, Glatzer T, Ambrosini P; et al. Human NK cells at early stages of differentiation produce CXCL8 and express CD161 molecule that functions as an activating receptor. Blood 2012;119:3987–96. [CrossRef]
- Girart M V, Fuertes MB, Domaica CI, Rossi LE, Zwirner NW. Engagement of TLR3, TLR7, and NKG2D regulate IFN-γ secretion but not NKG2D-mediated cytotoxicity by human NK cells stimulated with suboptimal doses of IL-The Journal of Immunology 2007;179:3472–9. [CrossRef]
- Kasahara Y, Shin C, Kubo N, Mihara K, Iwabuchi H, Takachi T; et al. Development and characterisation of NKp44-based chimeric antigen receptors that confer T cells with NK cell-like specificity. Clin Transl Immunology 2020;9. [CrossRef]
- Müller-Durovic B, Grählert J, Devine OP, Akbar AN, Hess C. CD56-negative NK cells with impaired effector function expand in CMV and EBV co-infected healthy donors with age. Aging 2019;11:724–40. [CrossRef]
- Forconi CS, Cosgrove CP, Saikumar-Lakshmi P, Nixon CE, Foley J, Ong’echa JM; et al. Poorly cytotoxic terminally differentiated CD56negCD16pos NK cells accumulate in Kenyan children with Burkitt lymphomas. Blood Adv 2018;2:1101–14. [CrossRef]
- Chretien A-S, Devillier R, Granjeaud S, Cordier C, Demerle C, Salem N; et al. High-dimensional mass cytometry analysis of NK cell alterations in AML identifies a subgroup with adverse clinical outcome. Proceedings of the National Academy of Sciences 2021;118. [CrossRef]
- Voigt J, Malone DFG, Dias J, Leeansyah E, Björkström NK, Ljunggren H; et al. Proteome analysis of human CD56negNK cells reveals a homogeneous phenotype surprisingly similar to CD56dimNK cells. Eur J Immunol 2018;48:1456–69. [CrossRef]
- Reiners KS, Topolar D, Henke A, Simhadri VR, Kessler J, Sauer M; et al. Soluble ligands for NK cell receptors promote evasion of chronic lymphocytic leukemia cells from NK cell anti-tumor activity. Blood 2013;121:3658–65. [CrossRef]
- Clinicaltrials.gov. Poly-ICLC (Hiltonol) and Anti-PD1 or Anti-PD-L1 (NCT03721679). Bethesda (MD): National Library of Medicine (US) 2000 Feb 29 2018. https://clinicaltrials.gov/study/NCT03721679?cond=Cancer&term=Polyinosinic-polycytidylic%20acid&rank=1 (accessed January 15, 2024).
- Clinicaltrials.gov. Study of PolyIC and PD-1 mAb in Subjects With Unresectable Hepatocellular Carcinoma (NCT03732547). Bethesda (MD): National Library of Medicine (US) 2000 Feb 29 2018. https://clinicaltrials.gov/study/NCT03732547?cond=Cancer&term=poly-IC&intr=Therapy&page=2&rank=11 (accessed January 15, 2024).
- ClinicalTrials.gov. Combination Therapy of Topical Imiquimod Plus Multipeptide Vaccination for Cutaneous Metastases of Melanoma (MEL53) (NCT01264731). Bethesda (MD): National Library of Medicine (US) 2000 Feb 29 2011. https://clinicaltrials.gov/study/NCT01264731?cond=Cancer&term=imiquimod&intr=Therapy&page=3&rank=25 (accessed January 13, 2024).
- Clinicaltrials.Gov. Predicting Response In Cervical Intraepithelial Neoplasia to Topical Imiquimod Treatment (PRedICT-TOPIC) (NCT05405270). Bethesda (MD): National Library of Medicine (US) 2000 Feb 29 2022. https://clinicaltrials.gov/study/NCT05405270?cond=Cancer&term=imiquimod&intr=Therapy&page=1&rank=6 (accessed January 13, 2024).
- Rook AH, Gelfand JM, Wysocka M, Troxel AB, Benoit B, Surber C; et al. Topical resiquimod can induce disease regression and enhance T-cell effector functions in cutaneous T-cell lymphoma. Blood 2015;126:1452–61. [CrossRef]
- clinicaltrials.gov. Vaccine Therapy and Resiquimod in Treating Patients With Stage II, Stage III, or Stage IV Melanoma That Has Been Completely Removed by Surgery (NCT00470379). Bethesda (MD): National Library of Medicine (US) 2000 Feb 29 2006. https://clinicaltrials.gov/study/NCT00470379?cond=Cancer&term=R848&intr=Therapy&page=1&rank=2 (accessed January 15, 2024).
- Clinicaltrials.gov. Intratumor CpG-ODN Injection Boosters Immune Killing Against in Situ Tumor Antigen Release for Advanced Solid Tumors (NCT04952272). Bethesda (MD): National Library of Medicine (US) 2000 Feb 29 2021. https://clinicaltrials.gov/study/NCT04952272?cond=Cancer&term=ODN%20&intr=Therapy&rank=1 (accessed January 15, 2024).

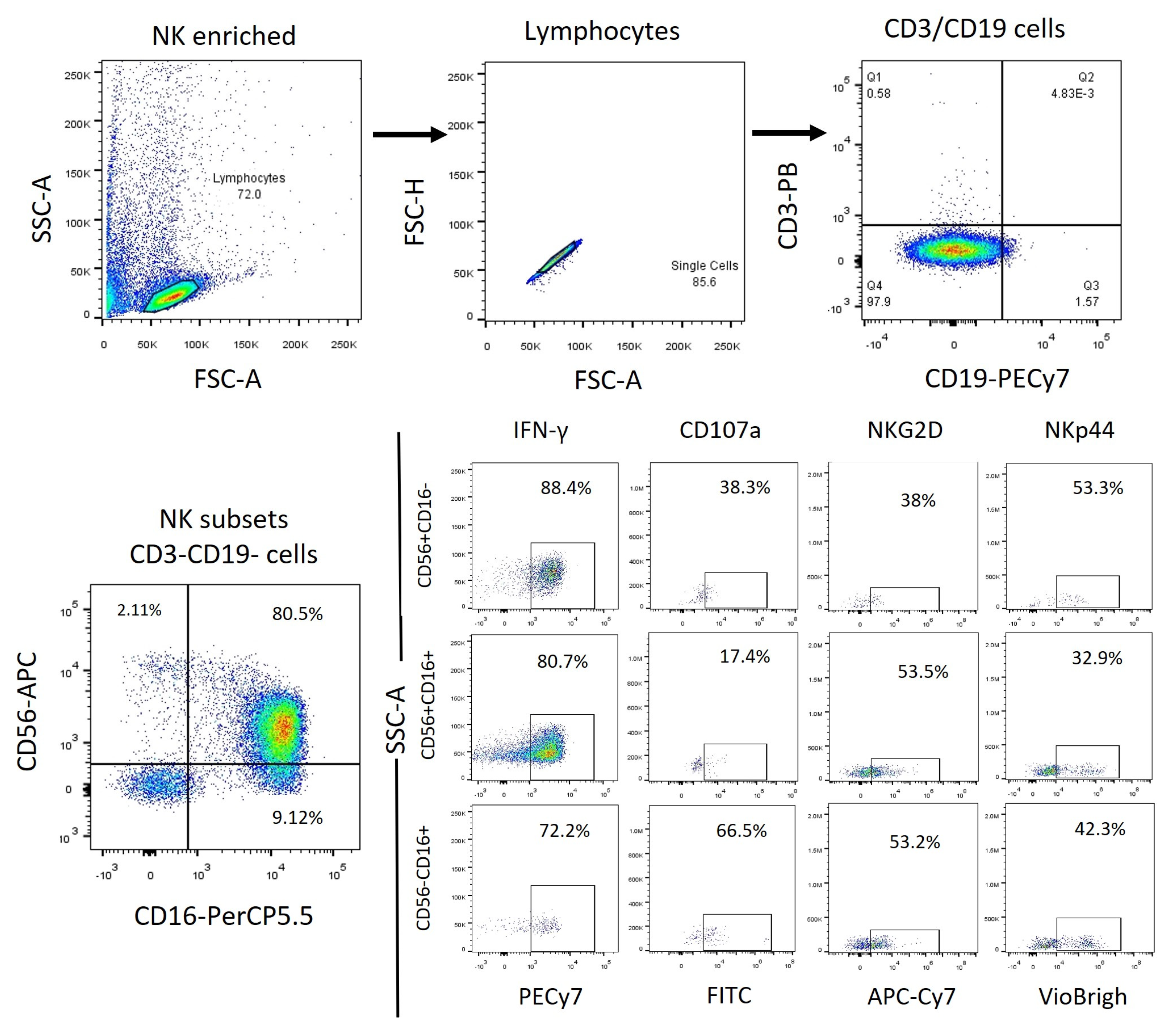
| Patients with acute lymphoblastic leukemia | |
|---|---|
| Characteristics | Peripheral blood |
| N° of cases | 24 |
| Sex (M:F) | 14:10 |
| Age media (range) | 7 (1-17) years |
| Immunophenotype | |
| Pre-B | 22 |
| not defined | 2 |
| % NK Cells (range) | 1.87 (0.4-5.97) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




