Submitted:
22 April 2024
Posted:
23 April 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
- SOW: dross from pure aluminium (expected approx. 80% aluminium);
- HDC: dross from 7% aluminium alloys (expected approx. 40% aluminium and approx. impurity content: Si 7%, Mg 2%, Ti 0,5% Sr 1%);
- RM: dross from 1xxx series aluminium (approx. 40% aluminium), (approx. other impurity content: B 1%, Ti 2% V 5%).
3. Results
3.1. Thermogravimetric Analysis
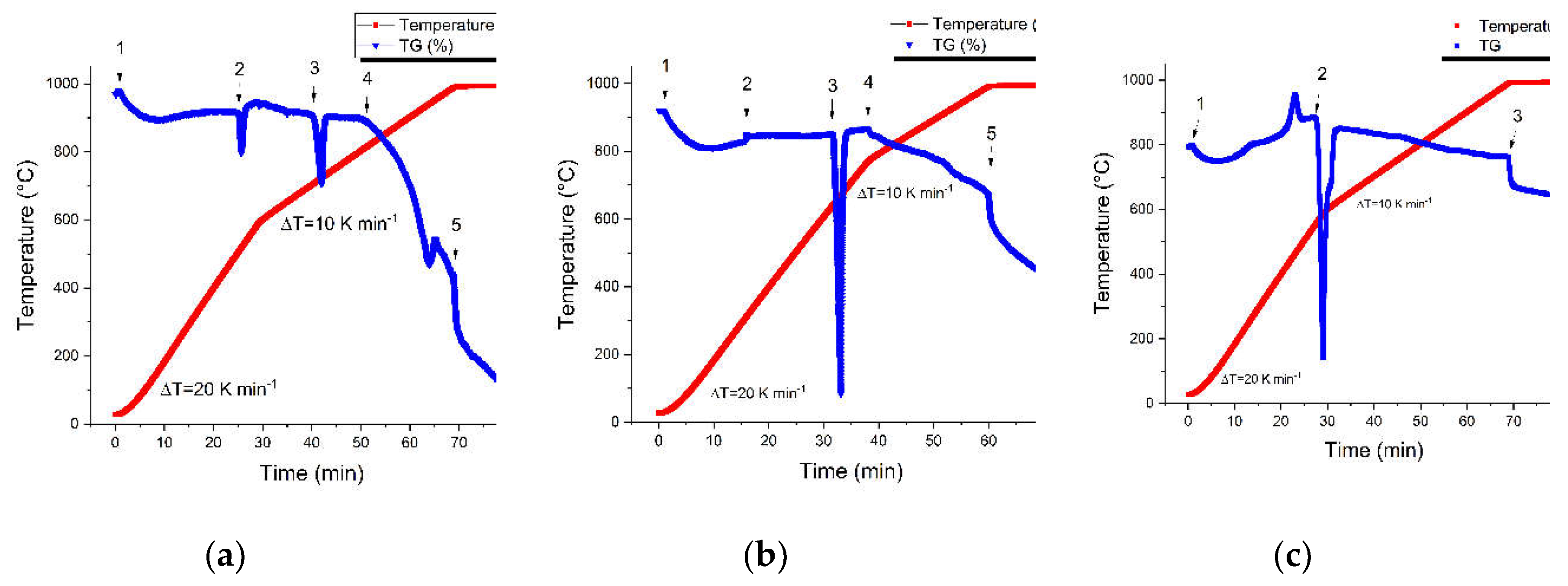
3.2. Elemental and Structural Analysis
3.2.1. SOW Sample
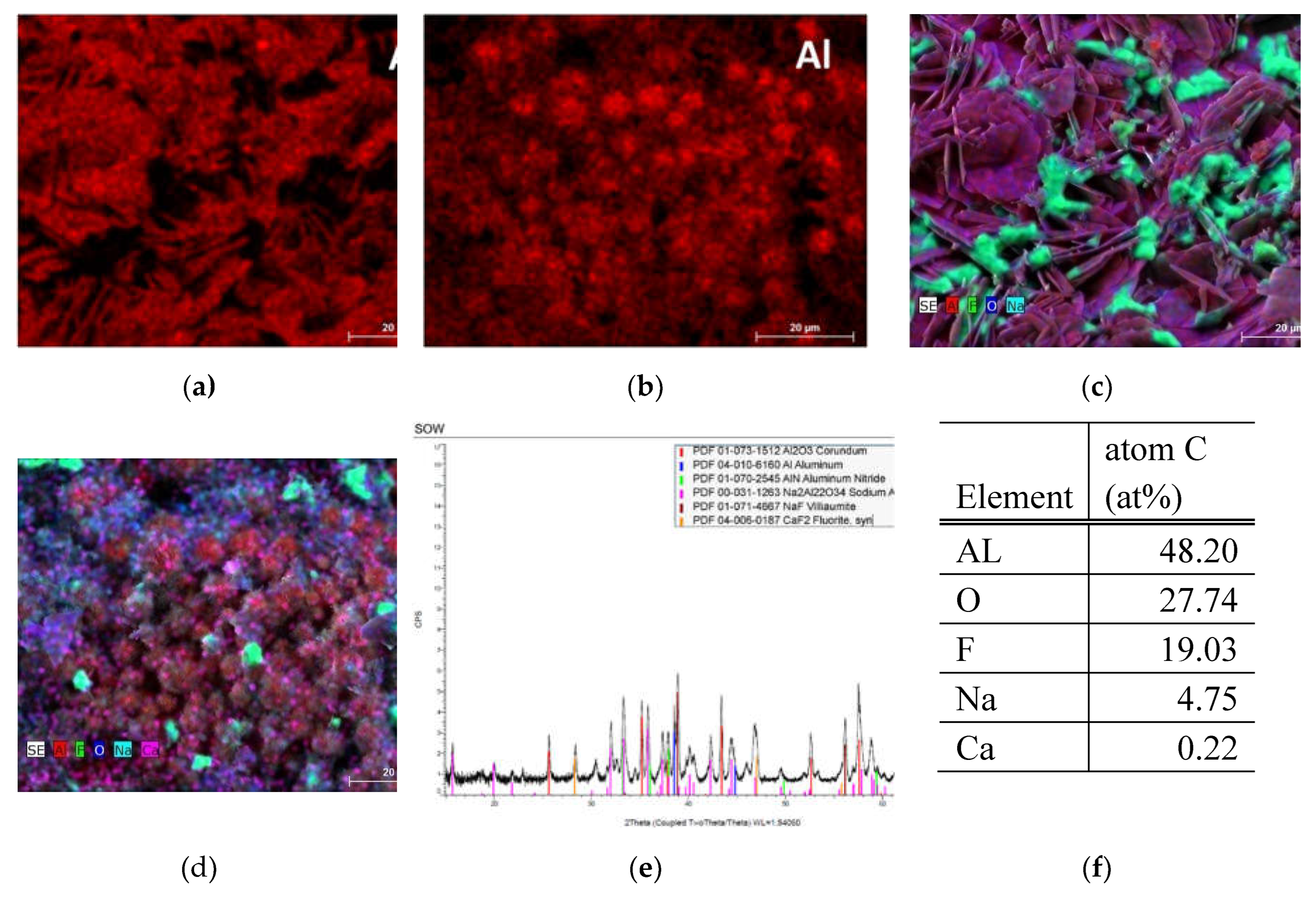
3.2.1. RM Sample

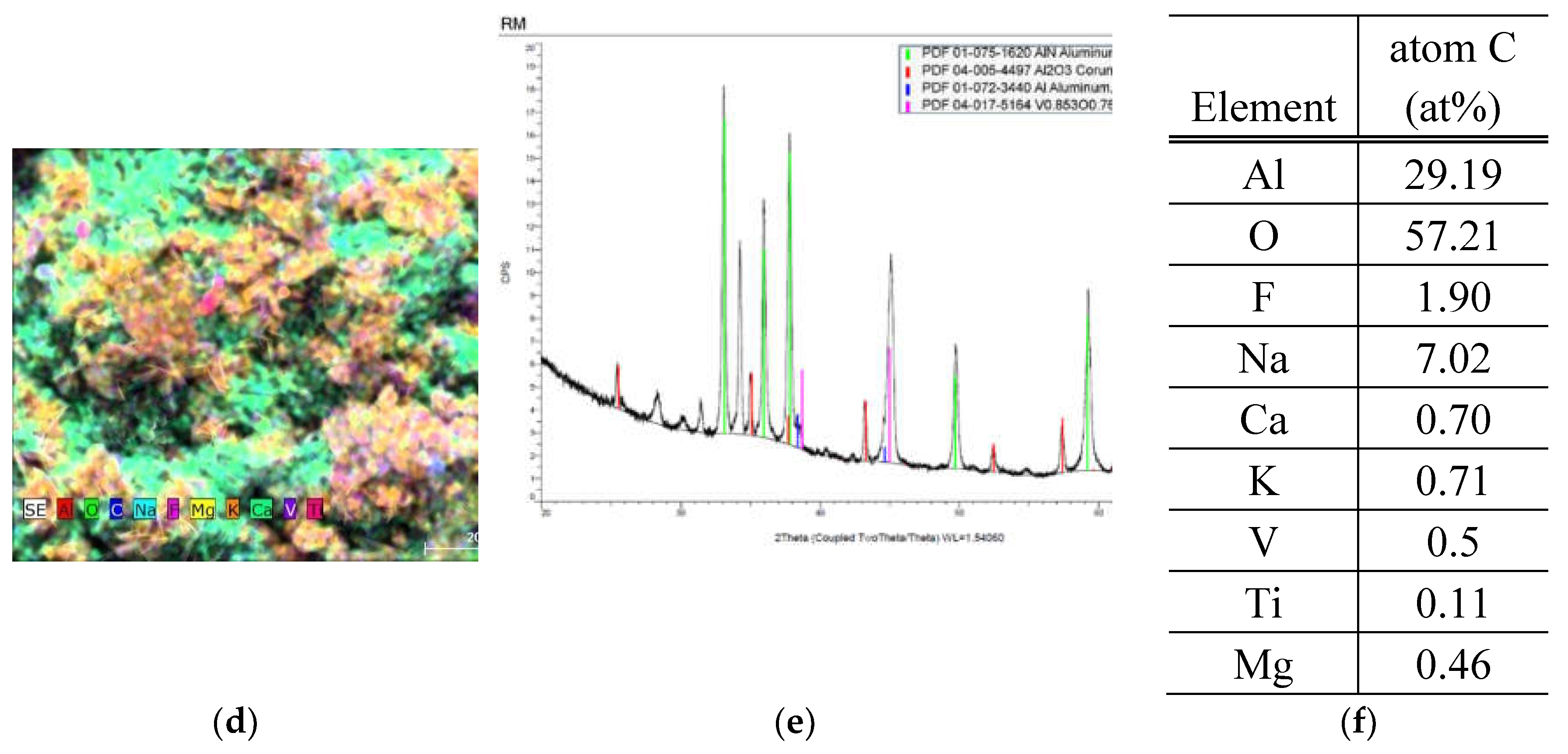
3.2.2. HDC Sample
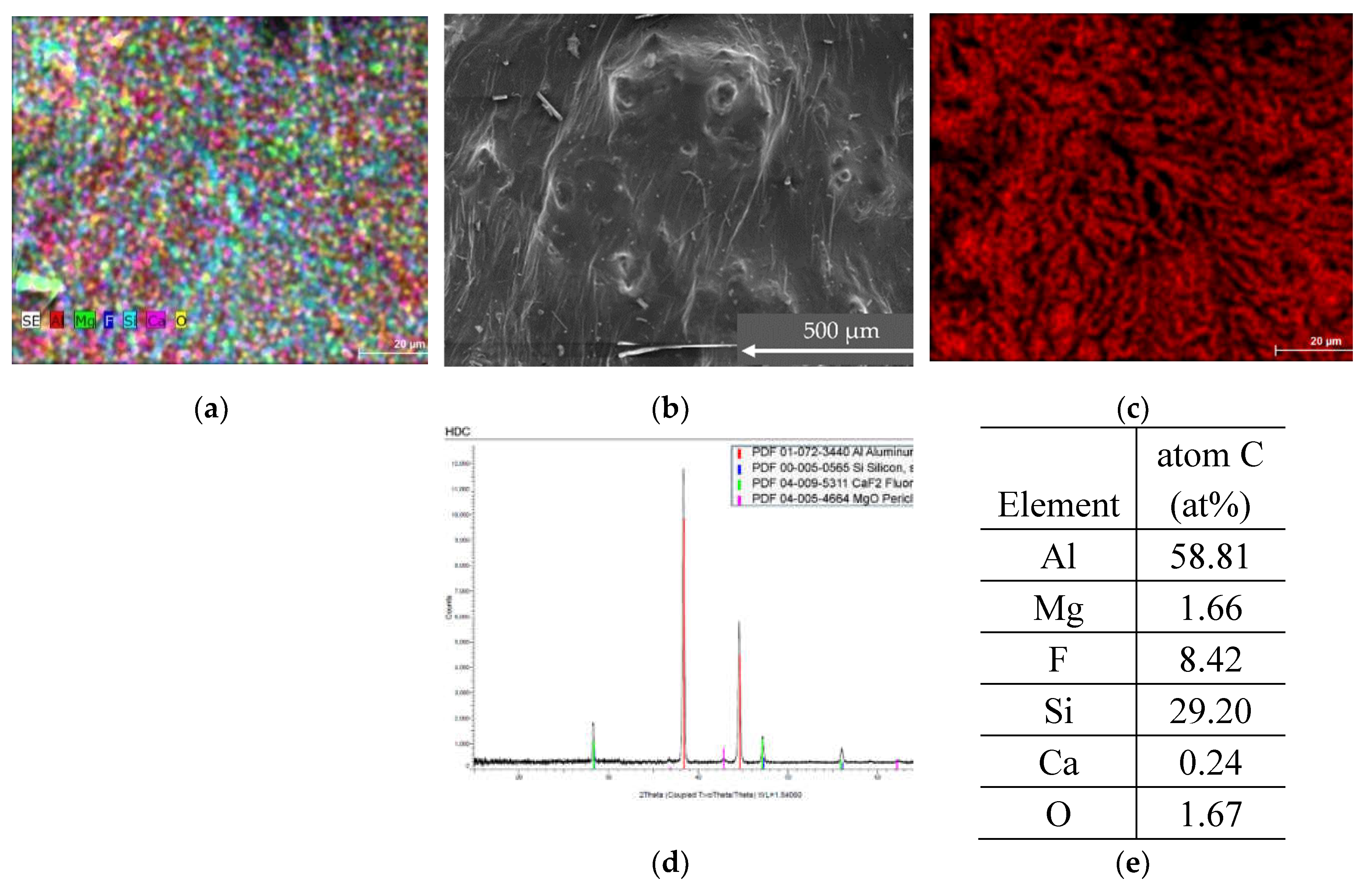
3.3. Hydrogen Production from Samples
3.3.1. SOW Sample
3.3.2. RM Sample
3.3.3. HDC Sample
3.3.4. Comparison of Hydrogen Generation and Reaction Efficiency
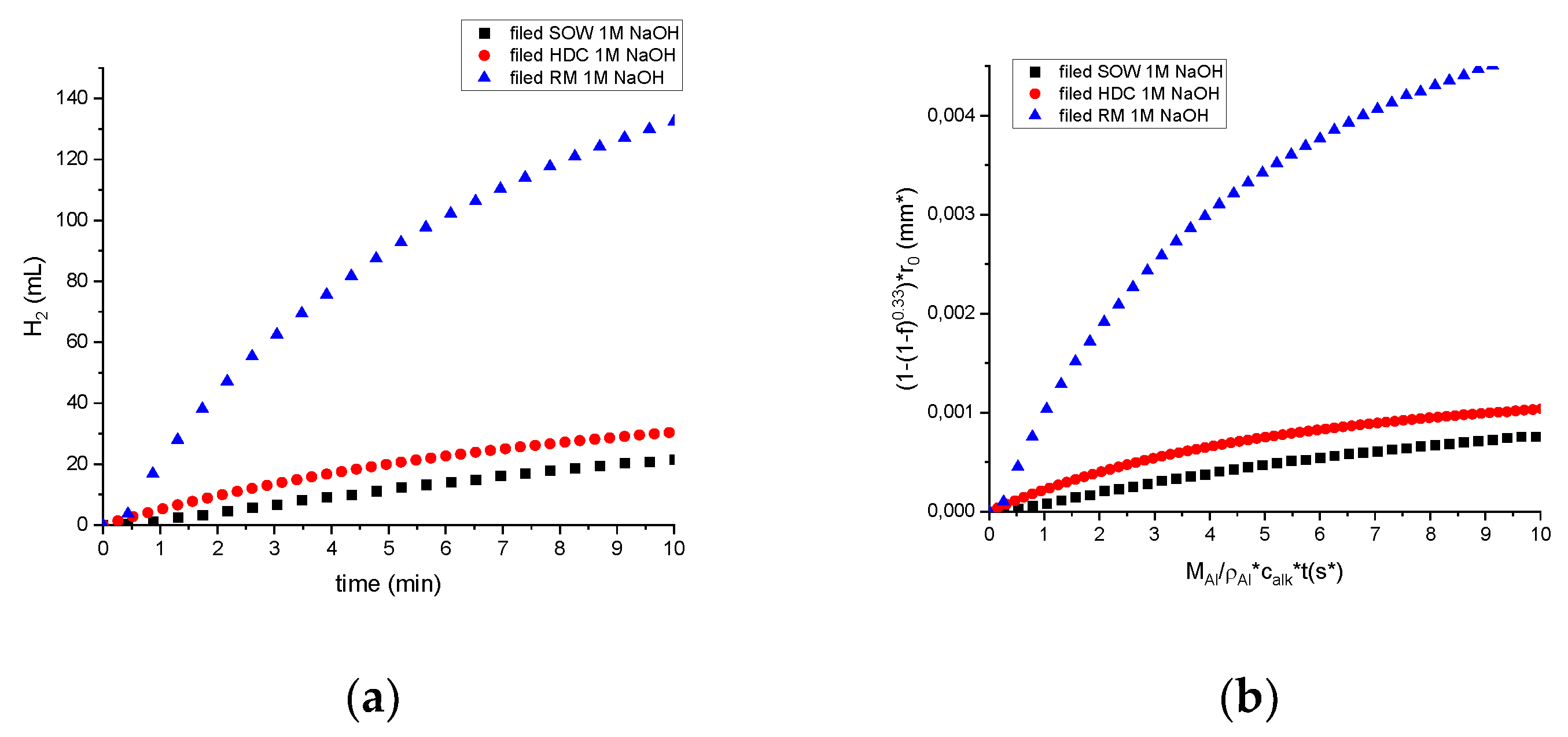
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Supporting S, Resource M U.S. Geological Survey Mineral Resources Program— 2007,. Program.
- Capuzzi S, Timelli G Preparation and melting of scrap in aluminum recycling: A review 2018,. Metals (Basel). 8:249.
- Blomberg J, Söderholm P The economics of secondary aluminium supply: An econometric analysis based on European data 2009,. Resour Conserv Recycl Volume 53, pp. 455–463. [CrossRef]
- John A. S. Green Aluminum Recycling and Processing for Energy Conservation and Sustainability 2007,.
- Padamata SK, Yasinskiy A, Polyakov P A Review of Secondary Aluminum Production and Its Byproducts 2021,. JOM 73:2603–2614.
- Bell S, Davis B, Javaid A, Essadiqi E Final Report on Refining Technologies of Aluminum Enhanced Recycling, Action Plan 2000 on Climate Change, Minerals and Metals Program-The Government of Canada Action Plan 2000 on Climate Change Minerals and Metals Program, managed by the Minerals and Meta 2003.
- Smith YR, Nagel JR, Rajamani RK Eddy current separation for recovery of non-ferrous metallic particles: A comprehensive review 2019,. Miner. Eng. 133:149–159.
- Schlesinger ME Aluminum Recycling 2006,. CRC Press.
- Mesina MB, De Jong TPR, Dalmijn WL Improvements in separation of non-ferrous scrap metals using an electromagnetic sensor 2003,. Phys Sep Sci Eng Volume 12, pp. 87–101. [CrossRef]
- Coates G, Rahimifard S Modelling of post-fragmentation waste stream processing within UK shredder facilities 2009,. Waste Manag Volume 29, pp. 44–53. [CrossRef]
- Venkoba Rao B, Kapur PC, Konnur R Modeling the size-density partition surface of dense-medium separators 2003,. Int J Miner Process Volume 72, pp. 443–453. [CrossRef]
- Hiraki T, Takeuchi M, Hisa M, Akiyama T Hydrogen production from waste aluminum at different temperatures, with LCA 2005,. Mater Trans Volume 46, pp. 1052–1057. 1057. [CrossRef]
- Mezulis A, Richter C, Lesnicenoks P, et al. Studies on Water–Aluminum Scrap Reaction Kinetics in Two Steps and the Efficiency of Green Hydrogen Production 2023,. Energies Volume 16, pp. 5554. [CrossRef]
- Ingason HT, Sigfusson TI Processing of Aluminum Dross: The Birth of a Closed Industrial Process 2014,. Jom Volume 66, pp. 2235–2242. [CrossRef]
- Cheng X, Shi Z, Glass N, et al. A review of PEM hydrogen fuel cell contamination: Impacts, mechanisms, and mitigation 2007,. J. Power Sources 165:739–756.
- Hiraki T, Takeuchi M, Hisa M, Akiyama T Hydrogen production from waste aluminum at different temperatures, with LCA 2005,. Mater Trans Volume 46, pp. 1052–1057. [CrossRef]
- Urbonavicius M, Varnagiris S, Mezulis A, et al. Hydrogen from industrial aluminium scraps: Hydrolysis under various conditions, modelling of pH behaviour and analysis of reaction by-product 2024,. Int J Hydrogen Energy Volume 50, pp. 431–446. [CrossRef]
- Hong JP, Wang J, Chen HY, et al. Process of aluminum dross recycling and life cycle assessment for Al-Si alloys and brown fused alumina 2010,. Trans Nonferrous Met Soc China (English Ed Volume 20, pp. 2155–2161. [CrossRef]
- David E, Kopac J Hydrolysis of aluminum dross material to achieve zero hazardous waste 2012,. J Hazard Mater Volume 209–210, pp. 501–509. [CrossRef]
- Olivares-Ramirez JM, de Jesus AM, Jimenez-Sandoval O, Pless RC Hydrogen Generation by Treatment of Aluminium Metal with Aqueous Solutions: Procedures and Uses 2012,. In: Hydrogen Energy - Challenges and Perspectives. IntechOpen.
- Zhao Z, Chen X, Hao M Hydrogen generation by splitting water with Al-Ca alloy 2011,. Energy Volume 36, pp. 2782–2787. [CrossRef]
- Küp Aylikci N, Mert SO, Aylikci V, et al. Microhydrogen production with water splitting from daily used waste aluminum 2021,. Int J Hydrogen Energy Volume 46, pp. 28912–28924. [CrossRef]
- Varnagiris S, Urbonavicius M Hydrogen generation kinetics via hydrolysis of Mg2Ni and Mg2NiH4 powders 2021,. Int J Hydrogen Energy Volume 46, pp. 36323–36335. [CrossRef]
- Alviani VN, Hirano N, Watanabe N, et al. Local initiative hydrogen production by utilization of aluminum waste materials and natural acidic hot-spring water 2021,. Appl Energy Volume 293, pp. 116909. [CrossRef]
- Elsarrag E, Elhoweris A, Alhorr Y The production of hydrogen as an alternative energy carrier from aluminium waste 2017,. Energy Sustain Soc Volume 7, pp. 1–14. [CrossRef]
- Raabe D, Ponge D, Uggowitzer P, et al. Making sustainable aluminum by recycling scrap: The science of “dirty” alloys 2022,. Prog Mater Sci Volume 128, pp. 100947. [CrossRef]
- Dangtungee R, Vatcharakajon P, Techawinyutham L Aluminium dross neutralization and its application as plant fertilizer 2021,. In: Materials Today: Proceedings. Elsevier, pp 2420–2426.
| SOW gas analysis | Beginning of reaction | End of reaction |
|---|---|---|
| Argon | 0.0 | 0.0 |
| Carbon Dioxide | 0.1 | 0.0 |
| Ethane | 0.0 | 0.0 |
| Hydrogen | 97.4 | 99.6 |
| Methane | 0.2 | 0.2 |
| Nitrogen | 1.2 | 0.0 |
| Oxygen | 0.3 | 0.0 |
| Water | 0.7 | 0.1 |
| RM gas analysis | Beginning of reaction | End of reaction |
|---|---|---|
| Argon | 0.0 | 0.0 |
| Carbon Dioxide | 0.2 | 0.2 |
| Ethane | 0.0 | 0.0 |
| Hydrogen | 98.3 | 97.2 |
| Methane | 0.0 | 0.0 |
| Nitrogen | 0.4 | 0.5 |
| Oxygen | 0.1 | 0.2 |
| Water | 1.9 | 1.9 |
| HDC gas analysis | Beginning of reaction | End of reaction |
|---|---|---|
| Argon | 0.0 | 0.0 |
| Carbon Dioxide | 0.2 | 0.2 |
| Ethane | 0.0 | 0.1 |
| Hydrogen | 98.3 | 92.5 |
| Methane | 0.0 | 0.4 |
| Nitrogen | 0.4 | 5.2 |
| Oxygen | 0.1 | 1.0 |
| Water | 0.9 | 0.5 |
| Sample and measurement | Oxygen | Nitrogen |
|---|---|---|
| RM | ||
| 1 | 5.31 | 0.009 |
| 2 | 6.87 | 2.595 |
| 3 | 0.58 | 0.013 |
| SOW | ||
| 1 | 0.330 | 0.007 |
| 2 | 0.067 | 0.004 |
| 3 | 4.86 | 0.530 |
| HDC | ||
| 1 | 1.329 | 0.044 |
| 2 | 2.714 | 0.070 |
| Sample | H2 generation (mL) | Yield of the theoretical value (%) | Reaction rate constant ks (mm/s) |
|---|---|---|---|
| RM | 224 | 65.7 | 0.001 |
| SOW | 61 | 24.9 | 0.000107 |
| HDC | 56 | 12.7 | 0.000229 |
| Source | Al | Cr | Fe | Ca | Si | Na | K | Cu | Zn | Ni | Mg | Ti | Pb | Sn | Mn | B | C | F | O |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| [19] | 43.3 | 0.088 | 4.32 | 0.45 | 10.9 | 0.8 | 0.21 | 1.17 | 0.9 | 0.87 | 1.85 | 0.27 | 0.053 | - | 0.2 | - | |||
| [18] | 73.05 | - | 0.91 | 2.79 | 7.13 | 5.78 | 9.26 | 1.08 | |||||||||||
| [24] | 56 | 12.7 | 0.000229 | ||||||||||||||||
| [25] | 11.65 | 0.18 | 1.51 | 0.08 | 17.96 | 0.05 | - | - | - | 0.02 | |||||||||
| [25] | 40.02 | 0.08 | 0.34 | 0.07 | 0.48 | 0.02 | - | - | - | 0.86 | |||||||||
| Al_w1 [17] | 94.3 | - | - | 0.6 | 5.0 | 0.1 | |||||||||||||
| RM | 29.19 - 32.46 | - | - | - | - | - | Trace - 0.71 | Trace - 0.1 | - | - | - | Trace - 0.28 | - | - | - | - | Trace - 5.70 | Trace - 1.98 | 13.19 - 57.76 |
| SOW | 19.11-48.20 | - | Trace - 0.29 | Trace - 0.29 | - | 4.75 - 18.96 | - | - | - | - | - | - | - | - | 19.03 - 53.76 | 7.58 – 27.74 | |||
| HDC | 58.81 | trace | 0.24 | 29.20 | trace | 1.66 | trace | - | - | - | Trace - 28.32 | 8.42 - 16.60 | 1.67 - 4.23 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




