Submitted:
15 April 2024
Posted:
16 April 2024
You are already at the latest version
Abstract
Keywords:
1. Background
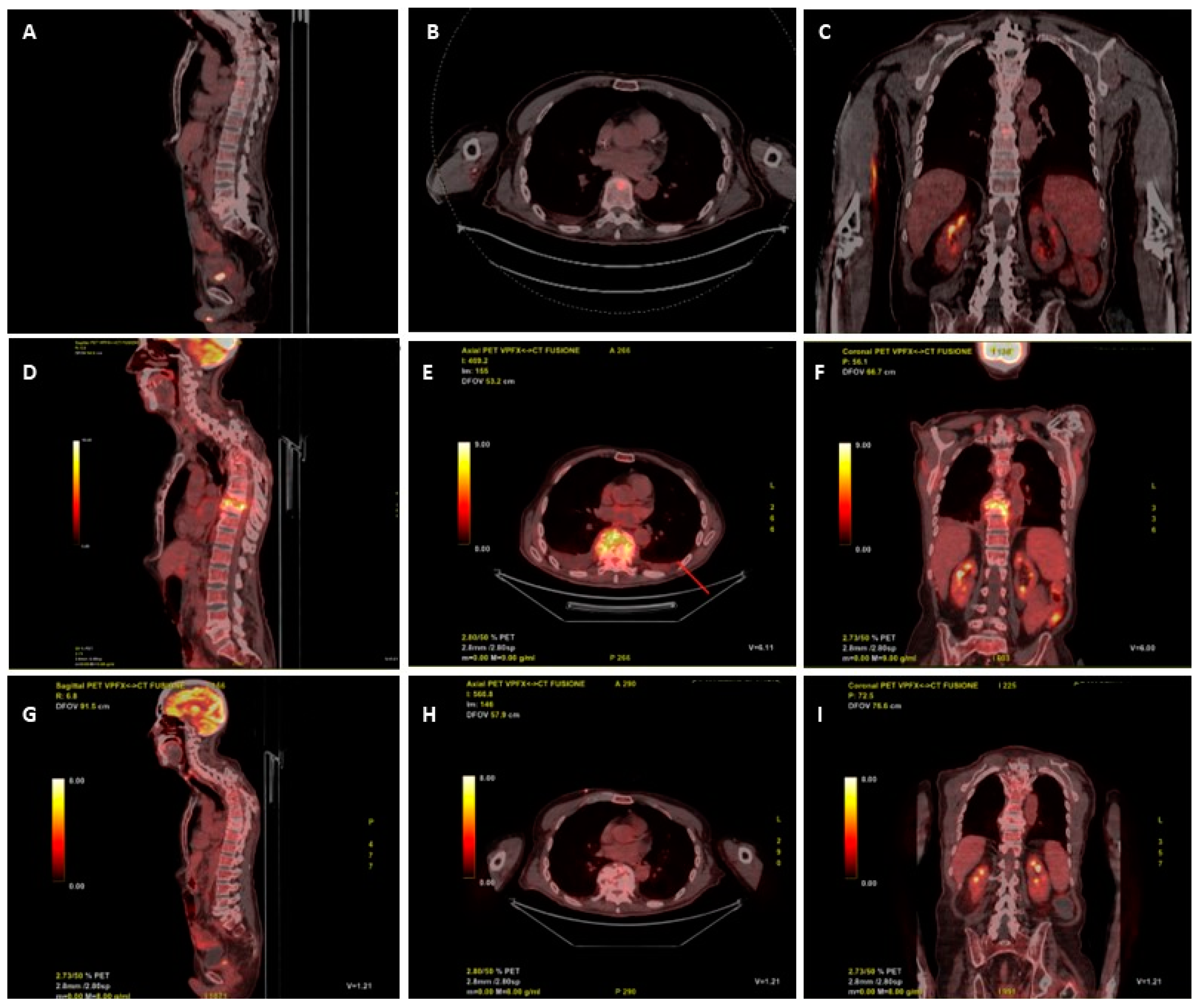
2. Case Report
3. Discussion
4. Conclusion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data availability
Aknowledgement
Transparency declaration
References
- Gouliouris T, Aliyu SH, Brown NM. Spondylodiscitis: update on diagnosis and management. Journal of Antimicrobial Chemotherapy. 2010, 65, iii11–24. [Google Scholar]
- Soriano A, Honore PM, Puerta-Alcalde P, Garcia-Vidal C, Pagotto A, Gonçalves-Bradley DC, et al. Invasive candidiasis: current clinical challenges and unmet needs in adult populations. Journal of Antimicrobial Chemotherapy. 2023, 78, 1569–1585. [Google Scholar] [CrossRef] [PubMed]
- Adelhoefer SJ, Gonzalez MR, Bedi A, Kienzle A, Bäcker HC, Andronic O, et al. Candida spondylodiscitis: a systematic review and meta-analysis of seventy two studies. Int Orthop. 2024, 48, 5–20. [Google Scholar] [CrossRef] [PubMed]
- Branco J, Miranda IM, Rodrigues AG. Candida parapsilosis Virulence and Antifungal Resistance Mechanisms: A Comprehensive Review of Key Determinants. Journal of Fungi. 2023, 9, 80. [Google Scholar] [CrossRef] [PubMed]
- Arendrup MC, Arikan-Akdagli S, Jørgensen KM, Barac A, Steinmann J, Toscano C, et al. European candidaemia is characterised by notable differential epidemiology and susceptibility pattern: Results from the ECMM Candida III study. Journal of Infection. 2023, 87, 428–437. [Google Scholar] [CrossRef] [PubMed]
- Pappas PG, Kauffman CA, Andes DR, Clancy CJ, Marr KA, Ostrosky-Zeichner L, et al. Clinical Practice Guideline for the Management of Candidiasis: 2016 Update by the Infectious Diseases Society of America. Clinical Infectious Diseases. 2016, 62, e1–50. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Effron, G. Rezafungin—Mechanisms of Action, Susceptibility and Resistance: Similarities and Differences with the Other Echinocandins. Journal of Fungi. 2020, 6, 262. [Google Scholar] [CrossRef] [PubMed]
- Arendrup MC, Meletiadis J, Zaragoza O, Jørgensen KM, Marcos-Zambrano LJ, Kanioura L, et al. Multicentre determination of rezafungin (CD101) susceptibility of Candida species by the EUCAST method. Clinical Microbiology and Infection. 2018, 24, 1200–1204. [Google Scholar] [CrossRef] [PubMed]
- Pfaller MA, Carvalhaes C, Messer SA, Rhomberg PR, Castanheira M. Activity of a Long-Acting Echinocandin, Rezafungin, and Comparator Antifungal Agents Tested against Contemporary Invasive Fungal Isolates (SENTRY Program, 2016 to 2018). Antimicrob Agents Chemother. 2020, 64. [Google Scholar]
- Thompson GR, Soriano A, Skoutelis A, Vazquez JA, Honore PM, Horcajada JP, et al. Rezafungin Versus Caspofungin in a Phase 2, Randomized, Double-blind Study for the Treatment of Candidemia and Invasive Candidiasis: The STRIVE Trial. Clinical Infectious Diseases. 2021, 73, e3647–55. [Google Scholar] [CrossRef] [PubMed]
- Thompson GR, Soriano A, Cornely OA, Kullberg BJ, Kollef M, Vazquez J, et al. Rezafungin versus caspofungin for treatment of candidaemia and invasive candidiasis (ReSTORE): a multicentre, double-blind, double-dummy, randomised phase 3 trial. The Lancet. 2023, 401, 49–59. [Google Scholar] [CrossRef] [PubMed]
- Pechacek J, Yakubu I, Vissichelli NC, Bruno D, Morales MK. Successful expanded access use of rezafungin, a novel echinocandin, to eradicate refractory invasive candidiasis in a liver transplant recipient. Journal of Antimicrobial Chemotherapy. 2022, 77, 2571–2573. [Google Scholar] [CrossRef] [PubMed]
- Melenotte C, Ratiney R, Hermine O, Bougnoux ME, Lanternier F. Successful Rezafungin Treatment of an Azole-Resistant Chronic Mucocutaneous Candidiasis in a STAT-1 Gain-of-Function Patient. J Clin Immunol. 2023, 43, 1182–1184. [Google Scholar] [CrossRef] [PubMed]
- 14. Adeel A, Qu MD, Siddiqui E, Levitz SM, Ellison RT. Expanded access use of rezafungin for salvage therapy of invasive Candida glabrata infection: A case report. Open Forum Infect Dis.
- Ripp SL, Aram JA, Bowman CJ, Chmielewski G, Conte U, Cross DM, et al. Tissue Distribution of Anidulafungin in Neonatal Rats. Birth Defects Res B Dev Reprod Toxicol. 2012, 95, 89–94. [Google Scholar] [CrossRef] [PubMed]
- Felton T, Troke PF, Hope WW. Tissue Penetration of Antifungal Agents. Clin Microbiol Rev. 2014, 27, 68–88. [Google Scholar] [CrossRef] [PubMed]
- Roepcke S, Passarell J, Walker H, Flanagan S. Population pharmacokinetic modeling and target attainment analyses of rezafungin for the treatment of candidemia and invasive candidiasis. Antimicrob Agents Chemother. 2023, 67. [Google Scholar]
- Vazquez JA, Flanagan S, Pappas P, Thompson GR, Sandison T, Honore PM. 637. Outcomes by Body Mass Index (BMI) in the STRIVE Phase 2 Trial of Once-Weekly Rezafungin for Treatment of Candidemia and Invasive Candidiasis Compared with Caspofungin. Open Forum Infect Dis. 2020, 7, S378–9. [Google Scholar] [CrossRef]
- Flanagan S, Walker H, Ong V, Sandison T. Absence of Clinically Meaningful Drug-Drug Interactions with Rezafungin: Outcome of Investigations. Microbiol Spectr. 2023, 11. [Google Scholar]
| Blood cultures | Bone | |||
|---|---|---|---|---|
| Candida parapsilosis | Candida parapsilosis | |||
| DRUG | MIC (mg/L) | interpretation | MIC (mg/L) | Interpretation |
| Caspofungin | 1 | S | 0.5 | S |
| Fluconazole | 128 | R | 32 | R |
| Voriconazole | 1 | R | 0.5 | I |
| Anidulafungin | 1 | S | 1 | S |
| Micafungin | 2 | S | 2 | S |
| 5-flucytosine | 0.12 | S | 0.06 | S |
| Posaconazole | 1 | R | 0.06 | S |
| Itraconazole | 0.25 | I | 0.12 | S |
| Amphotericin B | 0.5 | S | 1 | S |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




