Submitted:
08 April 2024
Posted:
09 April 2024
You are already at the latest version
Abstract
Keywords:
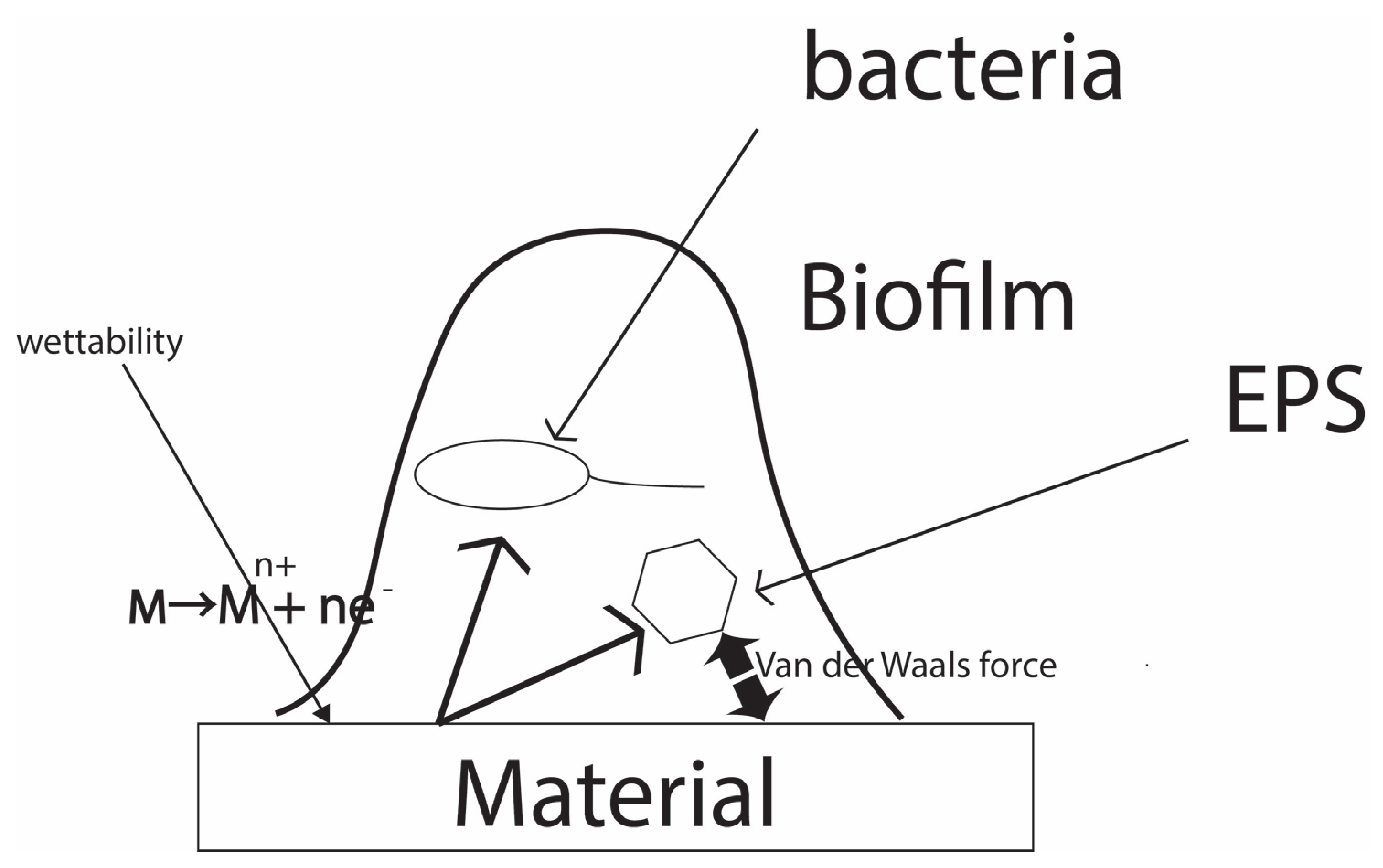
1. Introduction
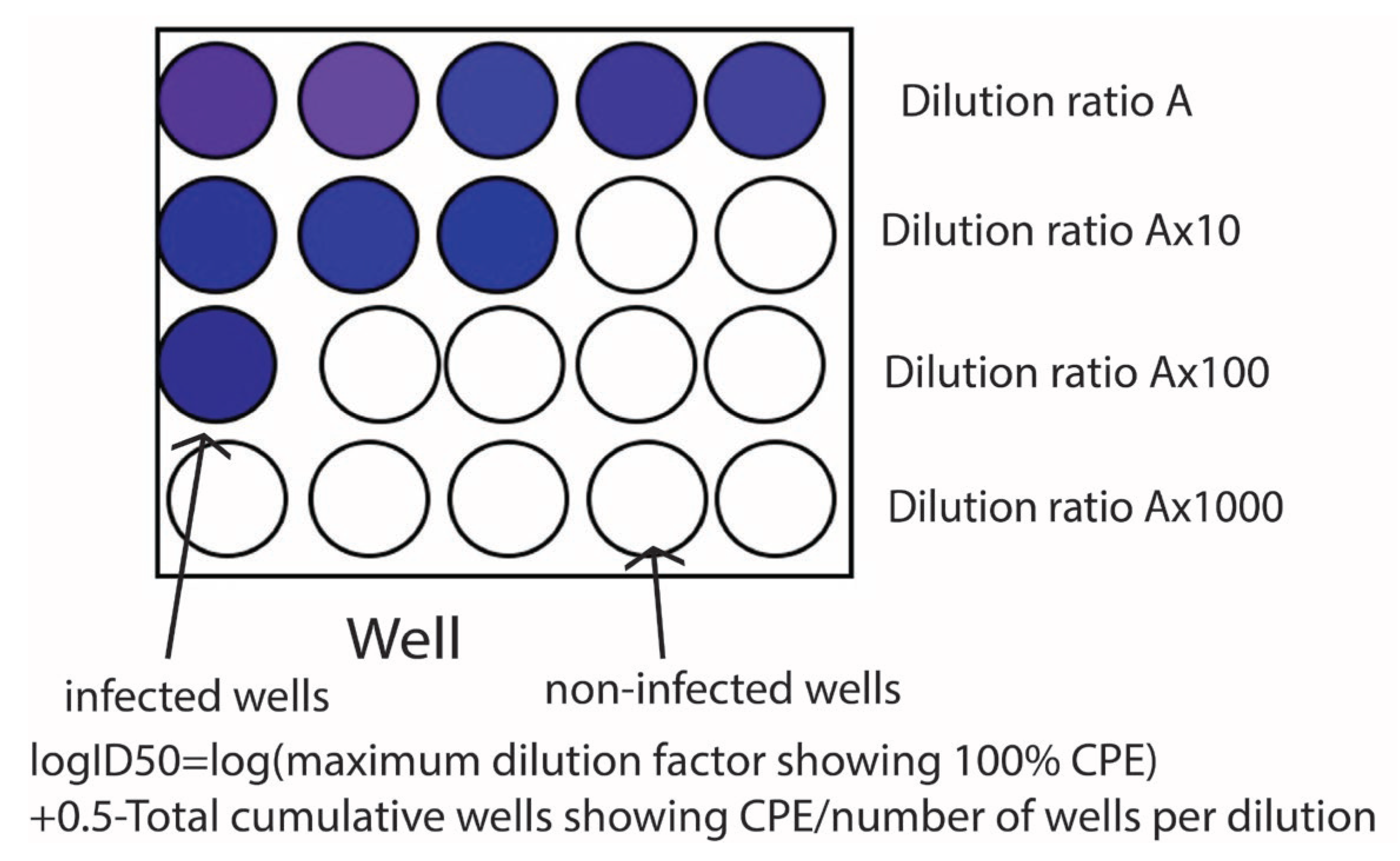
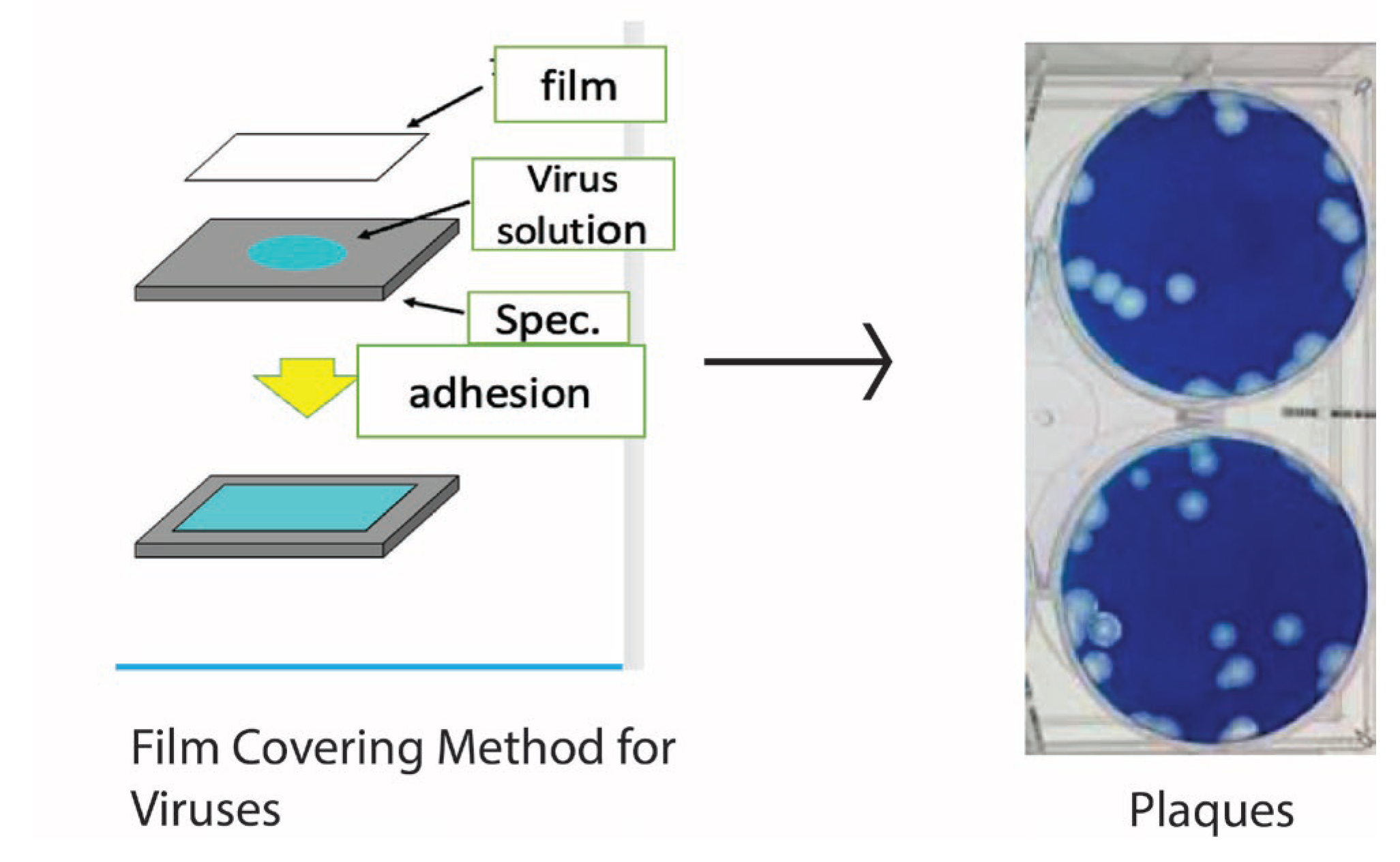
2. Antimicrobial Resistance Tests and the ISO Standard
- Ci: Number of viable bacteria (CFU) immediately after bacterial inoculation when using the material as a control.
- C0: Number of viable bacteria (CFU) after a certain time with the target material.
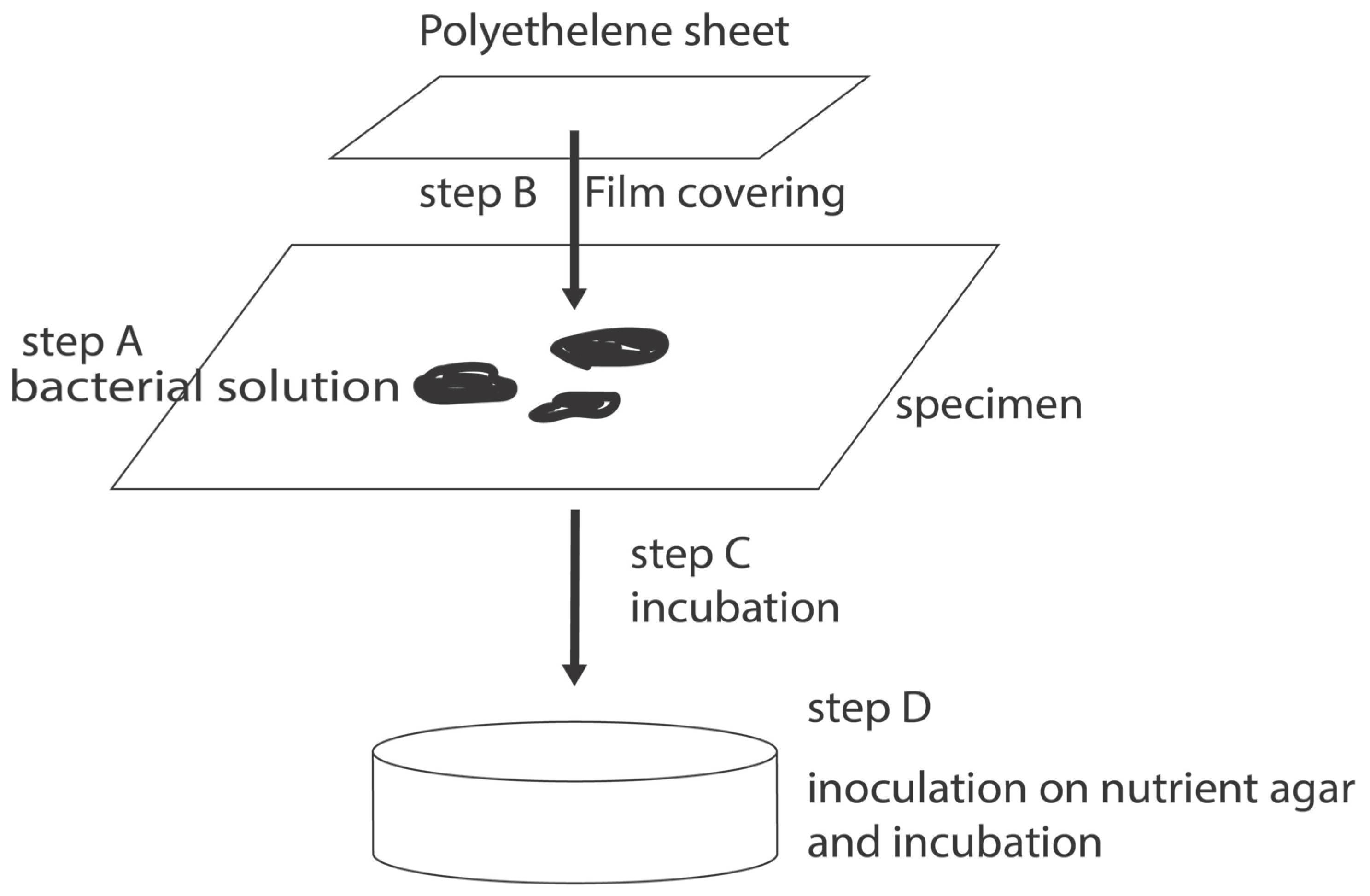
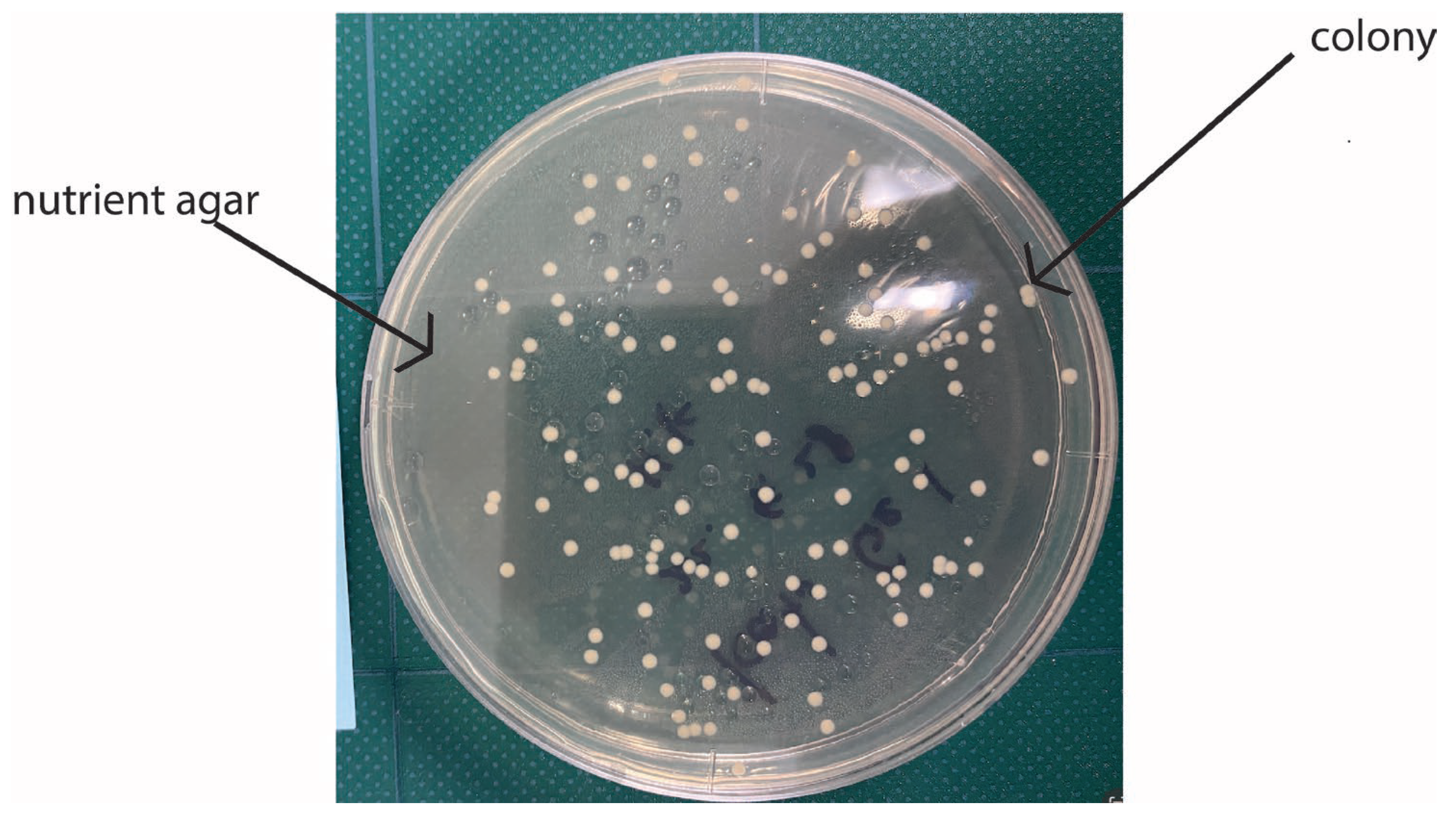
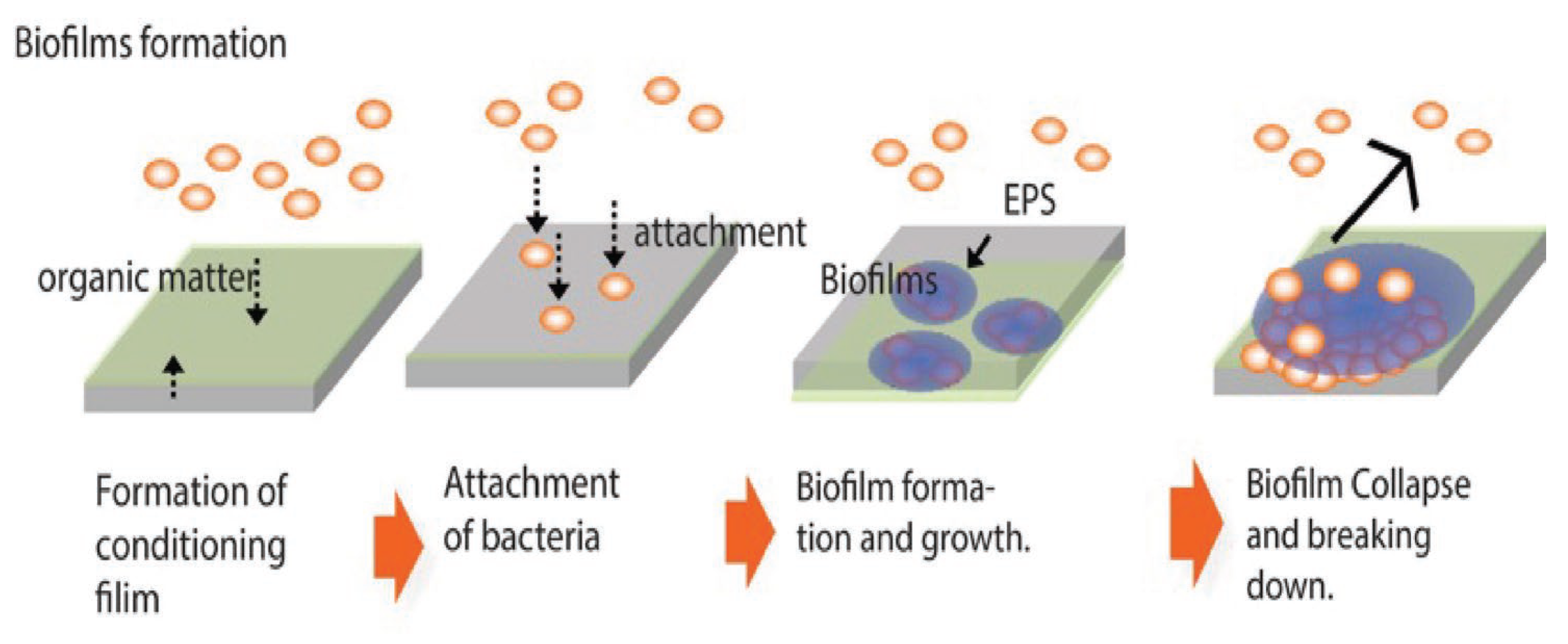
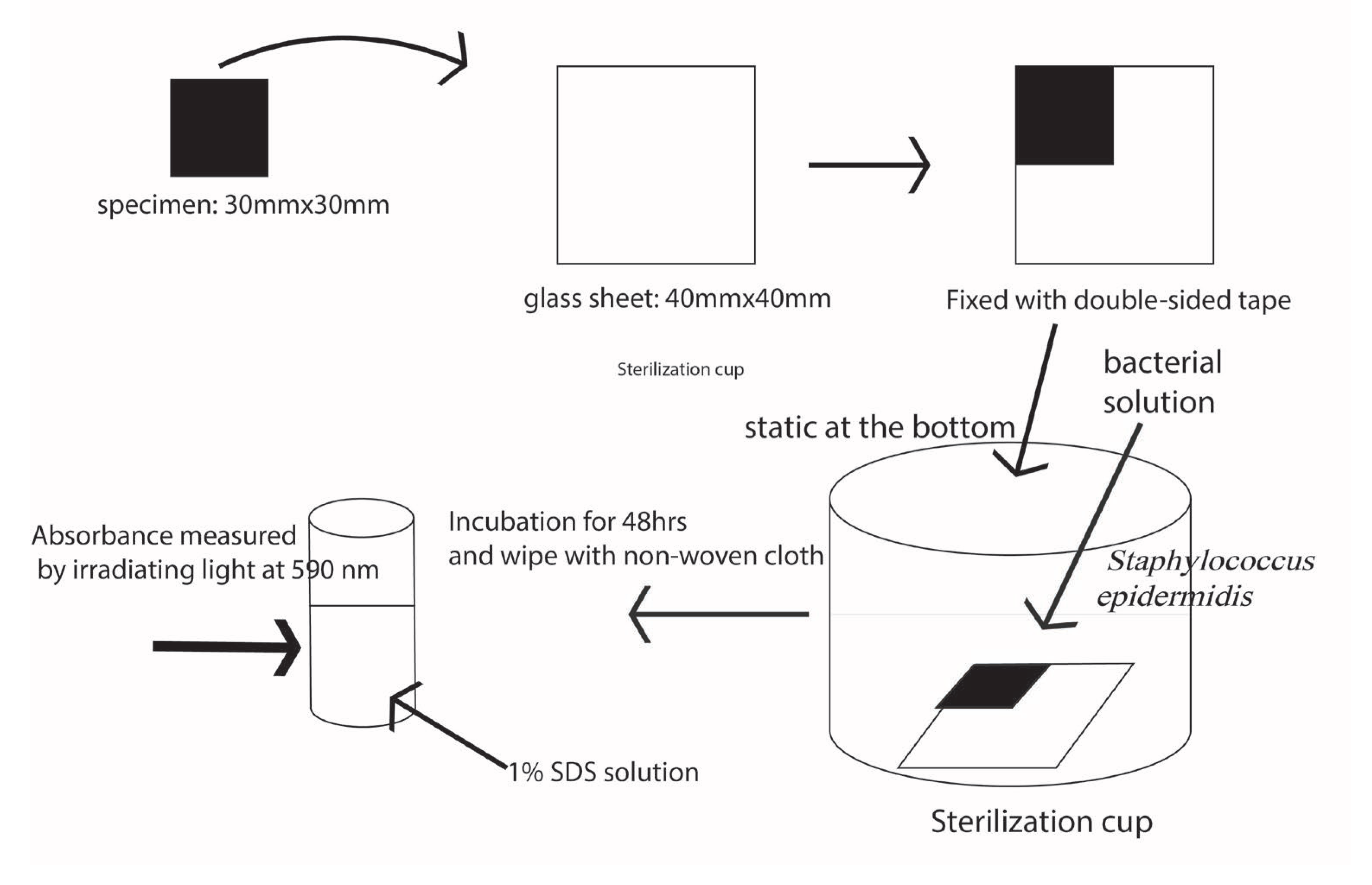
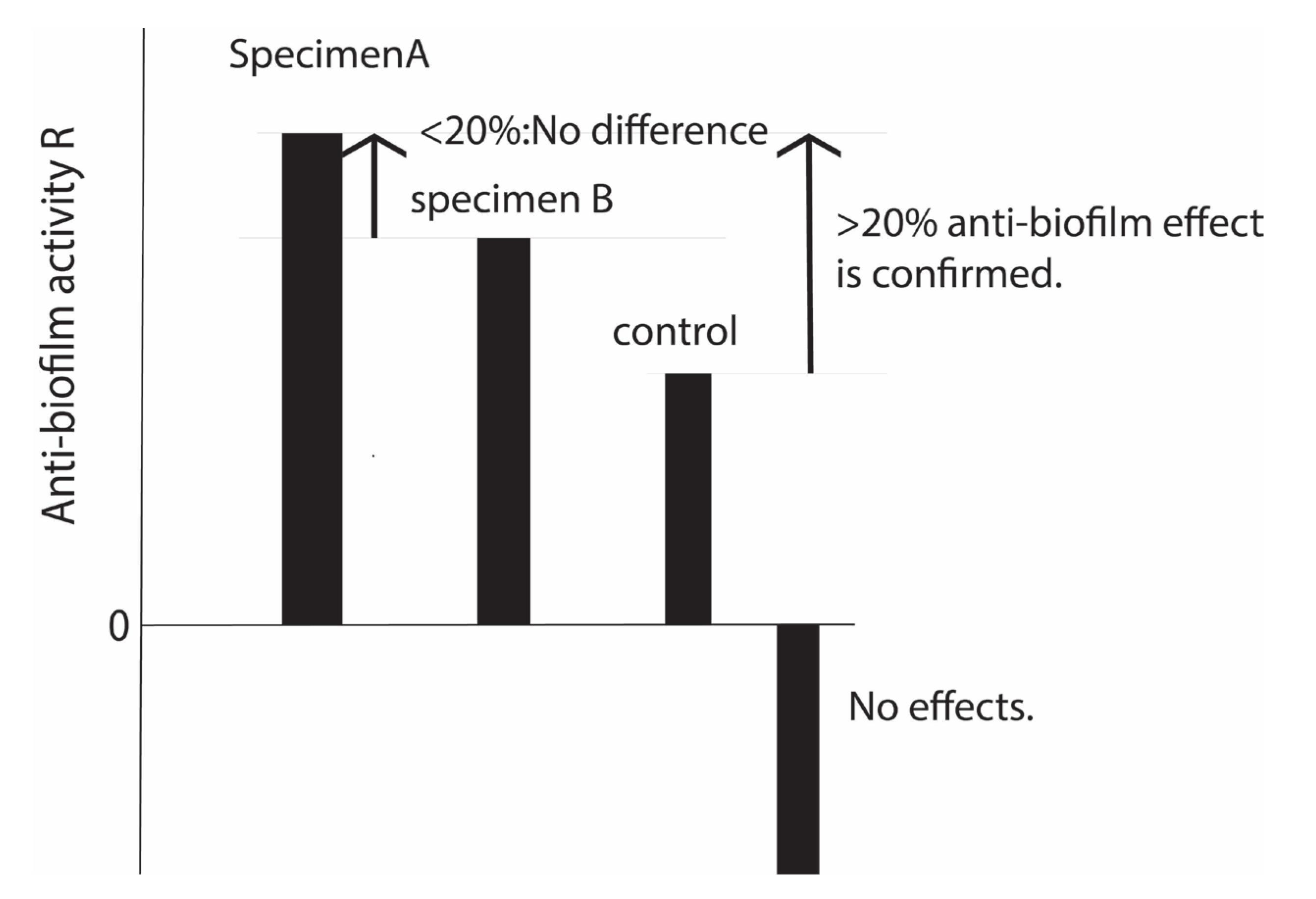
4. Antibiofilm Resistance Test and Its Standard
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kanematsu, H.; Barry, D.M. Biofilm and Materials Science; Springer: New York, the USA, 2015; p. 196. [Google Scholar]
- Kanematsu, H.; Barry, D.M. Formation and Control of Biofilm in Various Environments; Springer Nature: Singapore, 2020. [Google Scholar]
- SIAA. SIAA home page. Available online: https://www.kohkin.net/en_index.html.
- International Standard Organization ISO 4768:2023 Measurement method of anti-biofilm activity on plastic and other non-porous surfaces. 2023, ISO 4768:2023, 1-12.
- International Standard Organization, ISO 22196:2011 Measurement of antibacterial activity on plastics and other non-porous surfaces. ISO/TC 61/SC 6 ICS: 83.080.01 2011, SO 22196:2011, 1-15.
- International Standard Organization, Measurement of antiviral activity on plastics and other non-porous surfaces. 2019, ISO 21702:2019, 1-20.
- Kanematsu, H.; Barry, D.M.; Ikegai, H.; Mizunoe, Y. Biofilm control on metallic materials in medical fields from the viewpoint of materials science–from the fundamental aspects to evaluation. International Materials Reviews 2022, 68, 247–271. [Google Scholar] [CrossRef]
- Vimbela, G.V.; Ngo, S.M.; Fraze, C.; Yang, L.; Stout, D.A. Antibacterial properties and toxicity from metallic nanomaterials. International journal of nanomedicine 2017, 12, 3941–3965. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Popovich, J.; Zhang, W.; Ganser, C.; Haydel, S.E.; Seo, D.K. Superior ion release properties and antibacterial efficacy of nanostructured zeolites ion-exchanged with zinc, copper, and iron. RSC advances 2018, 8, 37949–37957. [Google Scholar] [CrossRef] [PubMed]
- Feng, A.; Cao, J.; Wei, J.; Chang, F.; Yang, Y.; Xiao, Z. Facile Synthesis of Silver Nanoparticle with High Antibacterial Activity. 2018.
- Nielsen, C.K.; Subbiahdoss, G.; Zeng, G.; Salmi, Z.; Kjems, J.; Mygind, T.; Snabe, T.; Meyer, R.L. Antibacterial isoeugenol coating on stainless steel and polyethylene surfaces prevents biofilm growth. J Appl Microbiol 2018, 124, 179–187. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, C.K.; Subbiahdoss, G.; Zeng, G.; Salmi, Z.; Kjems, J.; Mygind, T.; Snabe, T.; Meyer, R.L. Antibacterial isoeugenol coating on stainless steel and polyethylene surfaces prevents biofilm growth. J Appl Microbiol 2018, 124, 179–187. [Google Scholar] [CrossRef] [PubMed]
- Cunliffe, A.J.; Askew, P.D.; Stephan, I.; Iredale, G.; Cosemans, P.; Simmons, L.M.; Verran, J.; Redfern, J. How do we determine the efficacy of an antibacterial surface? A review of standardised antibacterial material testing methods. Antibiotics 2021, 10, 1069. [Google Scholar] [CrossRef] [PubMed]
- Tobias, R. Antibacterial properties of dental restorative materials: a review. International endodontic journal 1988, 21, 155–160. [Google Scholar] [CrossRef] [PubMed]
- Olmos, D.; González-Benito, J. Polymeric materials with antibacterial activity: A review. Polymers 2021, 13, 613. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, H. Celluloses as support materials for antibacterial agents: a review. Cellulose 2021, 28, 2715–2761. [Google Scholar] [CrossRef]
- Karwowska, E. Antibacterial potential of nanocomposite-based materials–a short review. Nanotechnology Reviews 2017, 6, 243–254. [Google Scholar] [CrossRef]
- Makvandi, P.; Jamaledin, R.; Jabbari, M.; Nikfarjam, N.; Borzacchiello, A. Antibacterial quaternary ammonium compounds in dental materials: A systematic review. Dental Materials 2018, 34, 851–867. [Google Scholar] [CrossRef] [PubMed]
- Loh, X.J. Latest advances in antibacterial materials. Journal of Molecular and Engineering Materials 2017, 5, 1740001. [Google Scholar] [CrossRef]
- Ferrando-Magraner, E.; Bellot-Arcís, C.; Paredes-Gallardo, V.; Almerich-Silla, J.M.; García-Sanz, V.; Fernández-Alonso, M.; Montiel-Company, J.M. Antibacterial properties of nanoparticles in dental restorative materials. A systematic review and meta-analysis. Medicina 2020, 56, 55. [Google Scholar] [CrossRef] [PubMed]
- Lei, C.; Yang, J.; Hu, J.; Sun, X. On the calculation of TCID50 for quantitation of virus infectivity. Virologica Sinica 2021, 36, 141–144. [Google Scholar] [CrossRef] [PubMed]
- LaBarre, D.D.; Lowy, R.J. Improvements in methods for calculating virus titer estimates from TCID50 and plaque assays. Journal of virological methods 2001, 96, 107–126. [Google Scholar] [CrossRef] [PubMed]
- Malenovska, H. Virus quantitation by transmission electron microscopy, TCID50, and the role of timing virus harvesting: A case study of three animal viruses. Journal of virological methods 2013, 191, 136–140. [Google Scholar] [CrossRef] [PubMed]
- Sachs, L.A.; Schnurr, D.; Yagi, S.; Lachowicz-Scroggins, M.E.; Widdicombe, J.H. Quantitative real-time PCR for rhinovirus, and its use in determining the relationship between TCID50 and the number of viral particles. Journal of virological methods 2011, 171, 212–218. [Google Scholar] [CrossRef] [PubMed]
- Lin, H.-T.; Tsai, H.-Y.; Liu, C.-P.; Yuan, T.T.-T. Comparability of bovine virus titers obtained by TCID50/ml and FAID50/ml. Journal of virological methods 2010, 165, 121–124. [Google Scholar] [CrossRef] [PubMed]
- Tyrrell, D.; Parsons, R. Some virus isolations from common colds. III. Cytopathic effects in tissue cultures. Lancet 1960, 239–242. [Google Scholar]
- Cotarelo, M.; Catalán, P.; Sánchez-Carrillo, C.; Menasalvas, A.; Cercenado, E.; Tenorio, A.; Bouza, E. Cytopathic effect inhibition assay for determining the in-vitro susceptibility of herpes simplex virus to antiviral agents. Journal of Antimicrobial Chemotherapy 1999, 44, 705–708. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.; Ma, C.; Wang, J. Cytopathic effect assay and plaque assay to evaluate in vitro activity of antiviral compounds against human coronaviruses 229E, OC43, and NL63. Bio-protocol 2022, 12, e4314–e4314. [Google Scholar] [CrossRef] [PubMed]
- Mehrbod, P.; Motamed, N.; Tabatabaeian, M.; SOLEYMANI, E.R.; Amini, E.; Shahidi, M.; Kheyri, M. In Vitro Antiviral Effect Of “Nanosilver” on Influenza Virus. DARU 2009, 17, 88–93. [Google Scholar]
- Wang, Y.-Z.; Cui, X.-L.; Gao, Y.-J.; Guo, S.-S.; Wang, X.-K.; Huang, Y.; Zhao, Y.; Gong, W.-F. Antivirus effects of extract from gardenia. Zhongguo Zhong yao za zhi= Zhongguo zhongyao zazhi= China journal of Chinese materia medica 2006, 31, 1176–1178. [Google Scholar] [PubMed]
- Nelson, D.L.; Cox, M.M. Lehninger Principles of Biochemistry, 6th Edition ed.; W.H.Freeman and Company: New York, the USA, 2013. [Google Scholar]
- Lee, G.; Bishop, P. Microbiology - and infection control for health professional, 6th Edition ed.; 2015.
- Lostroh, P. Molecular and cellular biology of viruses; Garland Science: 2019.
- Wulff, N.H.; Tzatzaris, M.; Young, P.J. Monte carlo simulation of the spearman-kaerber TCID50. Journal of clinical bioinformatics 2012, 2, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Wilrich, C.; Wilrich, P.-T. Estimation of the POD function and the LOD of a qualitative microbiological measurement method. Journal of AOAC International 2009, 92, 1763–1772. [Google Scholar] [CrossRef] [PubMed]
- Darling, A.J.; Boose, J.A.; Spaltro, J. Virus assay methods: accuracy and validation. Biologicals 1998, 26, 105–110. [Google Scholar] [CrossRef] [PubMed]
- Characklis, W.G. Fouling biofilm development: a process analysis. Biotechnology and bioengineering 1981, 23, 1923–1960. [Google Scholar] [CrossRef]
- Characklis, W.G.; Cooksey, K.E. Biofilms and microbial fouling. In Advances in applied microbiology; Elsevier: 1983; Volume 29, pp. 93-138.
- J.W.Costerton; K.J.Cheng; Geesey, G.G.; I.Timothy; J.Ladd; Nckel, C.; Dasgupta, M.; Marrie, T.J. Bacterial biofilms in nature and disease. Annual reviews in Microbiology 1987, 41, 435-464.
- Lappin-Scott, H.M.; Costerton, J.W. Bacterial biofilms and surface fouling. Biofouling 1989, 1, 323–342. [Google Scholar] [CrossRef]
- Flemming, H.-C. Biofilme, Biofouling und mikrobielle Schädigung von Werkstoffen; Oldenbourg, München, Germany: München, 1994; pp.xvi, 277.
- Costerton, J.W.; Lewandowski, Z. The biofilm lifestyle. Advances in Dental Research 1997, 11, 192–195. [Google Scholar] [CrossRef]
- Flemming, H.-C.; Szewzyk, U.; Griebe, T. Biofilms - Investigative Methods & Applications; CRC Press: Boca Raton, FL, the USA, 2000. [Google Scholar]
- Lewandowski, Z.; Beyenal, H. Fundamentals of Biofilm Research, Second Edition ed.; CRC Press: Boca Raton, London, New York, 2014. [Google Scholar]
- Merritt, J.H.; Kadouri, D.E.; O’Toole, G.A. Growing and analyzing static biofilms. Current protocols in microbiology 2011, 22, 1B. 1.1–1B 118. [Google Scholar] [CrossRef]
- Abbanat, D.; Shang, W.; Amsler, K.; Santoro, C.; Baum, E.; Crespo-Carbone, S.; Lynch, A.S. Evaluation of the in vitro activities of ceftobiprole and comparators in staphylococcal colony or microtitre plate biofilm assays. International journal of antimicrobial agents 2014, 43, 32–39. [Google Scholar] [CrossRef] [PubMed]
- Eigentler, L.; Davidson, F.A.; Stanley-Wall, N.R. Mechanisms driving spatial distribution of residents in colony biofilms: an interdisciplinary perspective. Open Biology 2022, 12, 220194. [Google Scholar] [CrossRef] [PubMed]
- Stepanović, S.; Vuković, D.; Hola, V.; Bonaventura, G.D.; Djukić, S.; Ćirković, I.; Ruzicka, F. Quantification of biofilm in microtiter plates: overview of testing conditions and practical recommendations for assessment of biofilm production by staphylococci. Apmis 2007, 115, 891–899. [Google Scholar] [CrossRef] [PubMed]
- O’Toole, G.A.Microtiter dish biofilm formation assay. JoVE (Journal of Visualized Experiments) 2011, e2437.
- Djordjevic, D.; Wiedmann, M.; McLandsborough, L. Microtiter plate assay for assessment of Listeria monocytogenes biofilm formation. Applied and environmental microbiology 2002, 68, 2950–2958. [Google Scholar] [CrossRef] [PubMed]
- Haney, E.F.; Trimble, M.J.; Hancock, R.E. Microtiter plate assays to assess antibiofilm activity against bacteria. Nature protocols 2021, 16, 2615–2632. [Google Scholar] [CrossRef]
- Elkhatib, W.F.; Khairalla, A.S.; Ashour, H.M. Evaluation of different microtiter plate-based methods for the quantitative assessment of Staphylococcus aureus biofilms. Future Microbiology 2014, 9, 725–735. [Google Scholar] [CrossRef] [PubMed]
- Boltz, J.P.; Smets, B.F.; Rittmann, B.E.; Van Loosdrecht, M.C.; Morgenroth, E.; Daigger, G.T. From biofilm ecology to reactors: a focused review. Water Science and Technology 2017, 75, 1753–1760. [Google Scholar] [CrossRef] [PubMed]
- Germec, M.; Demirci, A.; Turhan, I. Biofilm reactors for value-added products production: an in-depth review. Biocatalysis and agricultural biotechnology 2020, 27, 101662. [Google Scholar] [CrossRef]
- Saravanan, V.; Sreekrishnan, T. Modelling anaerobic biofilm reactors—A review. Journal of environmental management 2006, 81, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Ercan, D.; Demirci, A. Current and future trends for biofilm reactors for fermentation processes. Critical reviews in biotechnology 2015, 35, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Shahot, K.; Idris, A.; Omar, R.; Yusoff, H.M. Review on biofilm processes for wastewater treatment. Life Sci J 2014, 11, 1–13. [Google Scholar]
- Gjaltema, A.; Arts, P.; Van Loosdrecht, M.; Kuenen, J.G.; Heijnen, J. Heterogeneity of biofilms in rotating annular reactors: occurrence, structure, and consequences. Biotechnology and bioengineering 1994, 44, 194–204. [Google Scholar] [CrossRef] [PubMed]
- Hoh, D.; Watson, S.; Kan, E. Algal biofilm reactors for integrated wastewater treatment and biofuel production: a review. Chemical Engineering Journal 2016, 287, 466–473. [Google Scholar] [CrossRef]
- di Biase, A.; Kowalski, M.S.; Devlin, T.R.; Oleszkiewicz, J.A. Moving bed biofilm reactor technology in municipal wastewater treatment: a review. Journal of environmental management 2019, 247, 849–866. [Google Scholar] [CrossRef] [PubMed]
- Kuehn, M.; Hausner, M.; Bungartz, H.-J.; Wagner, M.; Wilderer, P.A.; Wuertz, S. Automated confocal laser scanning microscopy and semiautomated image processing for analysis of biofilms. Applied and environmental microbiology 1998, 64, 4115–4127. [Google Scholar] [CrossRef] [PubMed]
- Lawrence¹, J.R.; Wolfaardt, G.M.; Neu, T.R. The study of biofilms using confocal laser scanning microscopy. Digital Image Analysis of Microbes: Imaging, Morphometry, Fluorometry and Motility Techniques and Applications 1998, 431.
- Neu, T.R.; Lawrence, J.R. Investigation of microbial biofilm structure by laser scanning microscopy. Productive Biofilms 2014, 1–51. [Google Scholar]
- Schlafer, S.; Meyer, R.L. Confocal microscopy imaging of the biofilm matrix. Journal of Microbiological methods 2017, 138, 50–59. [Google Scholar] [CrossRef] [PubMed]
- Reichhardt, C.; Parsek, M.R. Confocal laser scanning microscopy for analysis of Pseudomonas aeruginosa biofilm architecture and matrix localization. Frontiers in microbiology 2019, 10, 677. [Google Scholar] [CrossRef] [PubMed]
- Little, B., Wagner, P., Ray, R., Pope, R., & Scheetz, R.(1991). Biofilms: an ESEM evaluation of artifacts introduced during SEM preparation. Journal of industrial microbiology, 8(4), 213–221.
- Araujo, J. C., Téran, F. C., Oliveira, R. A., Nour, E. A., Montenegro, M. A., Campos, J. R., & Vazoller, R. F. (2003). Comparison of hexamethyldisilazane and critical point drying treatments for SEM analysis of anaerobic biofilms and granular sludge. Journal of electron microscopy, 52(4), 429–433.
- Schaudinn, C., Carr, G., Gorur, A., Jaramillo, D.,Costerton, J. W., & Webster, P. (2009). Imaging of endodontic biofilms by combined microscopy (FISH/cLSM SEM). Journal of microscopy, 235(2), 124–127.
- Hoa, M., Tomovic, S., Nistico, L., Hall-Stoodley, L.,Stoodley, P., Sachdeva, L., … & Coticchia, J. M. (2009). Identification of adenoid biofilms with middle ear pathogens in otitis-prone children utilizing SEM and FISH. International journal of pediatric otorhinolaryngology, 73(9), 1242–1248.
- Reese, S., & Guggenheim, B. (2007). A novel TEM contrasting technique for extracellular polysaccharides in in vitro biofilms. Microscopy research and technique, 70(9), 816–822.
- Amorena, B., Gracia, E., Monzón, M., Leiva, J., Oteiza, C., Pérez, M.,…& Hernández-Yago, J. (1999). Antibiotic susceptibility assay for Staphylococcus aureus in biofilms developed in vitro. Journal of Antimicrobial Chemotherapy, 44 (1), 43–55.
- Surman, S. B., Walker, J. T., Goddard, D. T., Morton, L. H. G., Keevil, C. W., Weaver, W.,…& Kurtz, J. (1996). Comparison of microscope techniques for the examination of biofilms. Journal of Microbiological Methods, 25(1), 57–70.
- Sanclement, J. A., Webster, P., Thomas, J., & Ramadan, H. H. (2005). Bacterial biofilms in surgical specimens of patients with chronic rhinosinusitis. The Laryngoscope, 115(4), 578–582.
- Buret, A., Ward, K. H., Olson, M. E., & Costerton, J. W. (1991). An in vivo model to study the pathobiology of infectious biofilms on biomaterial surfaces. Journal of biomedical materials research, 25(7), 865–874.
- Beech, I. B., Cheung, C. S., Johnson, D. B., & Smith, J. R. (1996). Comparative studies of bacterial biofilms on steel surfaces using atomic force microscopy and environmental scanning electron microscopy.Biofouling, 10(1-3), 65–77.
- Fang, H. H., Chan, K.-Y., & Xu, L.-C. (2000). Quantification of bacterial adhesion forces using atomic force microscopy (AFM). Journal of microbiological methods, 40(1), 89–97.
- Auerbach, I. D., Sorensen, C., Hansma, H. G., & Holden, P. A. (2000). Physical morphology and surface properties of unsaturated Pseudomonas putida biofilms. Journal of bacteriology, 182(13), 3809–3815.
- Beech, I. B., Smith, J. R., Steele, A. A., Penegar, I., & Campbell, S. A. (2002). The use of atomic force microscopy for studying interactions of bacterial biofilms with surfaces. Colloids and Surfaces B: Biointerfaces, 23(2-3), 231–247.
- Yuan, S. J., & Pehkonen, S. O. (2007). Microbiologically influenced corrosion of 304 stainless steel by aerobic Pseudomonas NCIMB 2021 bacteria: AFM and XPS study. Colloids Surf B Biointerfaces, 59(1), 87–99. [CrossRef]
- Wright, C. J., Shah, M. K., Powell, L. C., & Armstrong, I. (2010). Application of AFM from microbial cell to biofilm. Scanning, 32(3), 134–149.
- Díaz, C., Schilardi, P., Salvarezza, R., & de Mele, M. F. L. (2011). Have flagella a preferred orientation during early stages of biofilm formation?: AFM study using patterned substrates. Colloids and Surfaces B: Biointerfaces, 82(2), 536–542.
- Harimawan, A., Zhong, S., Lim, C. T., & Ting, Y. P. (2013). Adhesion of B. subtilis spores and vegetative cells onto stainless steel--DLVO theories and AFM spectroscopy. J Colloid Interface Sci, 405, 233–241. [CrossRef]
- Beech, I. B. (1996). The potential use of atomic force microscopy for studying corrosion of metals in the presence of bacterial biofilms—an overview. International biodeterioration & biodegradation, 37 (3-4), 141–149.
- Beech, I. B., Cheung, C. S., Johnson, D. B., & Smith, J.R. (1996). Comparative studies of bacterial biofilms on steel surfaces using atomic force microscopy and environmental scanning electron microscopy.Biofouling, 10(1-3), 65–77.
- Arnold, J.,&Bailey, G. (2000). Surfacefinishes on stainless steel reduce bacterial attachment and early biofilm formation: scanning electron and atomic force microscopy study. Poultry science, 79(12), 1839–1845.
- Neu, T. R., & Lawrence, J. R. (1999). In situ characterization of extracellular polymeric substances (EPS) in biofilm systems. In Microbial Extracellular Polymeric Substances (pp. 21–47): Springer.
- Beech, I. B., Tapper, R. C., & Gubner, R. J. (2000). Microscopy methods for studying biofilms. Biofilms: Recent advances in their study and control, 51–70.
- Malvankar, N. S., Tuominen, M. T., & Lovley, D. R. (2012). Lack of cytochrome involvement in longrange electron transport through conductive biofilms and nanowires of Geobacter sulfurreducens. Energy & Environmental Science, 5(9), 8651–8659.
- Neu, T. R., Manz, B., Volke, F., Dynes, J. J., Hitchcock, A. P., & Lawrence, J. R. (2010). Advanced imaging techniques for assessment of structure, composition and function in biofilm systems. FEMS microbiology ecology, 72(1), 1–21.
- Schechter, M., Schechter, A., Rozenfeld, S., Efrat, E., & Cahan, R. (2014). Anode biofilm. In Technology and Application of Microbial Fuel Cells: IntechOpen.
- Millo, D., Harnisch, F., Patil, S. A., Ly, H. K., Schröder, U., & Hildebrandt, P. (2011). In situ spectroelectrochemical investigation of electrocatalytic microbial biofilms by surface-enhanced resonance Raman spectroscopy. Angewandte Chemie International Edition, 50(11), 2625–2627.
- Schmid, T., Messmer, A., Yeo, B. S., Zhang, W., & Zenobi, R. (2008). Towards chemical analysis of nanostructures in biofilms II: tip-enhanced Raman spectroscopy of alginates. Analytical and bioanalytical chemistry, 391(5), 1907–1916.
- Virdis, B., Harnisch, F., Batstone, D. J., Rabaey, K., & Donose, B. C. (2012). Non-invasive characterization of electrochemically active microbial biofilms using confocal Raman microscopy. Energy & Environmental Science, 5(5), 7017–7024.
- Sandt, C., Smith-Palmer, T., Pink, J., Brennan, L., & Pink, D. (2007). Confocal Raman microspectroscopy as a tool for studying the chemical heterogeneities of biofilms in situ. Journal of applied microbiology, 103 (5), 1808–1820.
- Schmitt, J., & Flemming, H. C. (1998). FTIR-spectroscopy in microbial and material analysis. International Biodeterioration & Biodegradation, 41 (1), 1–11.
- Schmitt, J., Nivens, D., White, D. C., & Flemming, H. C. (1995). Changes of biofilm properties in response to sorbed substances-an FTIR-ATR study. Water Science and Technology, 32(8), 149–155.
- Holman, H. Y. N., Miles, R., Hao, Z., Wozei, E., Anderson, L. M., & Yang, H. (2009). Real-time chemical imaging of bacterial activity in biofilms using open channel microfluidics and synchrotron FTIR spectromicroscopy. Analytical chemistry, 81 (20), 8564–8570.
- Quilès, F., Humbert, F., & Delille, A. (2010). Analysis of changes in attenuated total reflection FTIR fingerprints of Pseudomonas fluorescens from planktonic state to nascent biofilm state. Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy, 75(2), 610–616.
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



