Submitted:
03 April 2024
Posted:
03 April 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
1.1. Escherichia coli
1.2. Reservoirs
1.3. Zoonotic spillover
1.4. STEC Detection Methods
1.5. Gap Analysis
1.6. Aims of the Pilot Study
- to assess the viability of new molecular methodologies applied to raw milk filters enabling the identification of the presence of STEC serotypes in milk production, as a way to have an estimation of the prevalence of these pathogens in the herds;
- to apply the same methods to identify the presence of these pathogens in calf feces, as a way to identify a potential way of spreading, but also a critical point for a potential prevention of the spread.
2. Materials and Methods
2.1. Herds and Animals
2.2. Samples Collection
2.3. Samples Preparation
2.4. DNA Extraction
2.5. Real Time PCR Assay
2.5.1. Escherichia Coli O157:H7 and STEC Virulence Factors Identification
2.5.2. STEC Serotype Identification
2.6. Protocol Validation
2.7. Statistical Analysis
3. Results
3.1. Protocol Validation
3.2. Data Description
3.3. Serotypes Distribution
4. Discussion
4.1 STEC Prevalence in the Different Matrices
- Milk: 25 ml of raw milk are sampled from the bulk tank, capable of holding 150 to 10000 liters of milk at 4°C, which result in a poor detection level, particularly when the prevalence of STEC positive cows is very low and/or when milking practice are optimal.
- Milk Filters: with this type of sample is easier to find a positivity since the main task of the filter is to block and retain any type of fecal or litter debris coming from the milking routine, and all the milk pass through the filter; therefore, there is no dilution effect.
- Bovine feces: Since E. coli STEC is part of the intestinal microflora of bovines the higher prevalence of positive samples with this type of matrix is expected.
4.2. Distribution of Serotypes
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Gyles, C.L. Escherichia coli in Domestic Animals and Humans; Gyles, C.L. (Eds.) ; CABI, 1994; ISBN 0851989217.
- Gyles, C.L.; Prescott, J.F.; Songer, J.G.; Thoen, C.O. Pathogenesis of Bacterial Infections in Animals: Fourth Edition; Wiley-Blackwell, 2010; ISBN 9780813812373.
- Nataro, J.P.; Kaper, J.B. Diarrheagenic Escherichia coli. Clin. Microbiol. Rev. 1998, 11, 142–201. [Google Scholar] [CrossRef]
- Sora, V.M.; Meroni, G.; Martino, P.A.; Soggiu, A.; Bonizzi, L.; Zecconi, A. Extraintestinal pathogenic escherichia coli: Virulence factors and antibiotic resistance. Pathogens 2021, 10. [Google Scholar] [CrossRef]
- Gyles, C.L. Shiga toxin-producing Escherichia coli: an overview. J. Anim. Sci. 2007, 85. [Google Scholar] [CrossRef]
- Karmali, M.A.; Gannon, V.; Sargeant, J.M. Verocytotoxin-producing Escherichia coli (VTEC). Vet. Microbiol. 2010, 140, 360–370. [Google Scholar] [CrossRef]
- EFSA Panel on Biological Hazards Scientific Opinion on VTEC-seropathotype and scientific criteria regarding pathogenicity assessment. EFSA J. 2013, 11, 3138. [CrossRef]
- USDA Working Group usda. 2011.
- Costa, M.; Sucari, A.; Epszteyn, S.; Oteiza, J.; Gentiluomo, J.; Melamed, C.; Figueroa, Y.; Mingorance, S.; Grisaro, A.; Spioussas, S.; et al. Comparison of six commercial systems for the detection of non-O157 STEC in meat and vegetables. Food Microbiol. 2019, 84. [Google Scholar] [CrossRef]
- Li, B.; Liu, H.; Wang, W. Multiplex real-time PCR assay for detection of Escherichia coli O157:H7 and screening for non-O157 Shiga toxin-producing E. coli. BMC Microbiol. 2017, 17, 1–13. [Google Scholar] [CrossRef]
- Albonico, F.; Gusmara, C.; Gugliotta, T.; Loiacono, M.; Mortarino, M.; Zecconi, A. A new integrated approach to analyze bulk tank milk and raw milk filters for the presence of the E. coli serogroups frequently associated with VTEC status. Res. Vet. Sci. 2017, 115, 401–406. [Google Scholar] [CrossRef]
- Varcasia, B.; Tomassetti, F.; De Santis, L.; Di Giamberardino, F.; Lovari, S.; Bilei, S.; De Santis, P. Presence of Shiga Toxin-Producing Escherichia coli (STEC) in Fresh Beef Marketed in 13 Regions of ITALY (2017). Microorganisms 2018, 6, 126. [Google Scholar] [CrossRef]
- Vélez, M. V.; Colello, R.; Etcheverría, A.I.; Padola, N.L. Shiga toxin producing Escherichia coli: the challenge of adherence to survive. Rev. Argent. Microbiol. 2023, 55, 100–107. [Google Scholar] [CrossRef]
- Montero, D.A.; Canto, F. Del; Velasco, J.; Colello, R.; Padola, N.L.; Salazar, J.C.; Martin, C.S.; Oñate, A.; Blanco, J.; Rasko, D.A.; et al. Cumulative acquisition of pathogenicity islands has shaped virulence potential and contributed to the emergence of LEE-negative Shiga toxin-producing Escherichia coli strains. Emerg. Microbes Infect. 2019, 8, 486–502. [Google Scholar] [CrossRef]
- Caprioli, A.; Morabito, S.; Brugère, H.; Oswald, E. Enterohaemorrhagic Escherichia coli: Emerging issues on virulence and modes of transmission. Vet. Res. 2005, 36, 289–311. [Google Scholar] [CrossRef]
- Duffy, G.; Burgess, C.M.; Bolton, D.J. A review of factors that affect transmission and survival of verocytotoxigenic Escherichia coli in the European farm to fork beef chain. Meat Sci. 2014, 97, 375–383. [Google Scholar] [CrossRef]
- Peng, Z.; Liang, W.; Hu, Z.; Li, X.; Guo, R.; Hua, L.; Tang, X.; Tan, C.; Chen, H.; Wang, X.; et al. O-serogroups, virulence genes, antimicrobial susceptibility, and MLST genotypes of Shiga toxin-producing Escherichia coli from swine and cattle in Central China. BMC Vet. Res. 2019, 15, 1–13. [Google Scholar] [CrossRef]
- Scaife, H.R.; Cowan, D.; Finney, J.; Kinghorn-Perry, S.F.; Crook, B. Wild rabbits (Oryctolagus cuniculus) as potential carriers of verocytotoxin-producing Escherichia coli. Vet. Rec. 2006, 159, 175–178. [Google Scholar] [CrossRef]
- Szczerba-Turek, A.; Chierchia, F.; Socha, P.; Szweda, W. Shiga Toxin-Producing Escherichia coli in Faecal Samples from Wild Ruminants. animals 2023, 13. [Google Scholar] [CrossRef]
- Sanches, L.A.; Gomes, M. da S.; Teixeira, R.H.F.; Cunha, M.P.V.; Oliveira, M.G.X. de; Vieira, M.A.M.; Gomes, T.A.T.; Knobl, T. Captive wild birds as reservoirs of enteropathogenic E. coli (EPEC) and Shiga-toxin producing E. coli (STEC). Brazilian J. Microbiol. 2017, 48, 760–763. [Google Scholar] [CrossRef]
- Bentancor, A.; Rumi, M. V.; Carbonari, C.; Gerhardt, E.; Larzábal, M.; Vilte, D.A.; Pistone-Creydt, V.; Chinen, I.; Ibarra, C.; Cataldi, A.; et al. Profile of Shiga toxin-producing Escherichia coli strains isolated from dogs and cats and genetic relationships with isolates from cattle, meat and humans. Vet. Microbiol. 2012, 156, 336–342. [Google Scholar] [CrossRef]
- Karama, M.; Mainga, A.O.; Cenci-Goga, B.T.; Malahlela, M.; El-Ashram, S.; Kalake, A. Molecular profiling and antimicrobial resistance of Shiga toxin-producing Escherichia coli O26, O45, O103, O121, O145 and O157 isolates from cattle on cow-calf operations in South Africa. Sci. Rep. 2019, 9, 1–15. [Google Scholar] [CrossRef]
- Hussein, H.S.; Sakuma, T. Shiga toxin-producing Escherichia coli: Pre- and postharvest control measures to ensure safety of dairy cattle products. J. Food Prot. 2005, 68, 199–207. [Google Scholar] [CrossRef]
- Etcheverría, A.I.; Padola, N.L. Shiga toxin-producing Escherichia coli: Factors involved in virulence and cattle colonization. Virulence 2013, 4, 366–372. [Google Scholar] [CrossRef]
- Farrokh, C.; Jordan, K.; Auvray, F.; Glass, K.; Oppegaard, H.; Raynaud, S.; Thevenot, D.; Condron, R.; De Reu, K.; Govaris, A.; et al. Review of Shiga-toxin-producing Escherichia coli (STEC) and their significance in dairy production. Int. J. Food Microbiol. 2013, 162, 190–212. [Google Scholar] [CrossRef]
- Rastawicki, W.; Śmietańska, K.; Rokosz-Chudziak, N.; Wołkowicz, T. Antibody response to lipopolysaccharides and recombinant proteins of Shiga toxin (STX)-producing Escherichia coli (STEC) in children with haemolytic uraemic syndrome in Poland. Lett. Appl. Microbiol. 2020, 70, 440–446. [Google Scholar] [CrossRef]
- Dastmalchi Saei, H.; Ayremlou, N. Characterization of Shiga toxin-producing Escherichia coli (STEC) in feces of healthy and diarrheic calves in Urmia region, Iran. Iran. J. Microbiol. 2012, 4, 63–69. [Google Scholar]
- Renter, D.G.; Sargeant, J.M.; Hungerford, L.L. Distribution of Escherichia coli O157:H7 within and among cattle operations in pasture-based agricultural areas. Am. J. Vet. Res. 2004, 65, 1367–1376. [Google Scholar] [CrossRef]
- ISO/TS 13136:2012 Microbiology of food and animal feed Real-time polymerase chain reaction (PCR)-based method for the detection of food-borne pathogens Horizontal method for the detection of Shiga toxin-producing Escherichia coli (STEC) and the determination of O157, O111,. 2012.
- Parsons, B.D.; Zelyas, N.; Berenger, B.M.; Chui, L. Detection, characterization, and typing of shiga toxin-producing Escherichia coli. Front. Microbiol. 2016, 7, 1–12. [Google Scholar] [CrossRef]
- Wang, J.; Katani, R.; Li, L.; Hegde, N.; Roberts, E.L.; Kapur, V.; DebRoy, C. Rapid detection of Escherichia coli O157 and shiga toxins by lateral flow immunoassays. Toxins (Basel). 2016, 8. [Google Scholar] [CrossRef]
- Wijnsma, K.L.; van Bommel, S.A.M.; van der Velden, T.; Volokhina, E.; Schreuder, M.F.; van den Heuvel, L.P.; van de Kar, N.C.A.J. Fecal diagnostics in combination with serology: best test to establish STEC-HUS. Pediatr. Nephrol. 2016, 31, 2163–2170. [Google Scholar] [CrossRef]
- Beutin, L.; Fach, P. Detection of Shiga Toxin-Producing Escherichia coli from Nonhuman Sources and Strain Typing. Microbiol. Spectr. 2014, 2, 1–23. [Google Scholar] [CrossRef]
- World Health Organization Shiga toxin-producing Escherichia coli (STEC) and food: attribution, characterization, and monitoring MICROBIOLOGICAL RISK ASSESSMENT SERIES 31 REPORT Shiga toxin-producing Escherichia coli (STEC) and food: attribution, characterisation, and monitoring; 2019; ISBN 978-92-5-130682-6.
- DISCONTOOLS. Available online: https://www.discontools.eu/database.html?rid=9839&v=html (accessed on Dec 5, 2023).
- Murphy, B.P.; Murphy, M.; Buckley, J.F.; Gilroy, D.; Rowe, M.T.; McCleery, D.; Fanning, S. In-line milk filter analysis: Escherichia coli O157 surveillance of milk production holdings. Int. J. Hyg. Environ. Health 2005, 208, 407–413. [Google Scholar] [CrossRef]
- Giacometti, F.; Serraino, A.; Finazzi, G.; Daminelli, P.; Losio, M.N.; Bonilauri, P.; Arrigoni, N.; Garigliani, A.; Mattioli, R.; Alonso, S.; et al. Foodborne pathogens in in-line milk filters and associated on-farm risk factors in dairy farms authorized to produce and sell raw milk in northern Italy. J. Food Prot. 2012, 75, 1263–1269. [Google Scholar] [CrossRef]
- Van Kessel, J.A.S.; Karns, J.S.; Lombard, J.E.; Kopral, C.A. Prevalence of Salmonella enterica, Listeria monocytogenes, and Escherichia coli virulence factors in bulk tank milk and in-line filters from U.S. dairies. J. Food Prot. 2011, 74, 759–768. [Google Scholar] [CrossRef]
- Salaheen, S.; Kim, S.W.; Springer, H.R.; Hovingh, E.P.; Van Kessel, J.A.S.; Haley, B.J. Genomic diversity of antimicrobial-resistant and Shiga toxin gene-harboring non-O157 Escherichia coli from dairy calves. J. Glob. Antimicrob. Resist. 2023, 33, 164–170. [Google Scholar] [CrossRef]
- Fernández, M.; Casaux, M.L.; Fraga, M.; Vignoli, R.; Bado, I.; Zunino, P.; Umpiérrez, A. Shiga Toxin-Producing Escherichia coli (STEC) Associated with Calf Mortality in Uruguay. Microorganisms 2023, 11, 1–12. [Google Scholar] [CrossRef]
- Auvray, F.; Bièche-Terrier, C.; Um, M.M.; Dupouy, V.; Nzuzi, N.; David, L.; Allais, L.; Drouet, M.; Oswald, E.; Bibbal, D.; et al. Prevalence and characterization of the seven major serotypes of Shiga toxin-producing Escherichia coli (STEC) in veal calves slaughtered in France. Vet. Microbiol. 2023, 282. [Google Scholar] [CrossRef]
- Dell’Orco, F.; Gusmara, C.; Loiacono, M.; Gugliotta, T.; Albonico, F.; Mortarino, M.; Zecconi, A. Evaluation of virulence factors profiles and antimicrobials resistance of Escherichia coli isolated from bulk tank milk and raw milk filters. Res. Vet. Sci. 2019, 123, 77–83. [Google Scholar] [CrossRef]
- Geraghty, T.; Graham, D.A.; Mullowney, P.; More, S.J. A review of bovine Johne’s disease control activities in 6 endemically infected countries. Prev. Vet. Med. 2014, 116, 1–11. [Google Scholar] [CrossRef]
- Capps, K.M.; Ludwig, J.B.; Shridhar, P.B.; Shi, X.; Roberts, E.; DebRoy, C.; Cernicchiaro, N.; Phebus, R.K.; Bai, J.; Nagaraja, T.G. Identification, Shiga toxin subtypes and prevalence of minor serogroups of Shiga toxin-producing Escherichia coli in feedlot cattle feces. Sci. Rep. 2021, 11, 1–12. [Google Scholar] [CrossRef]
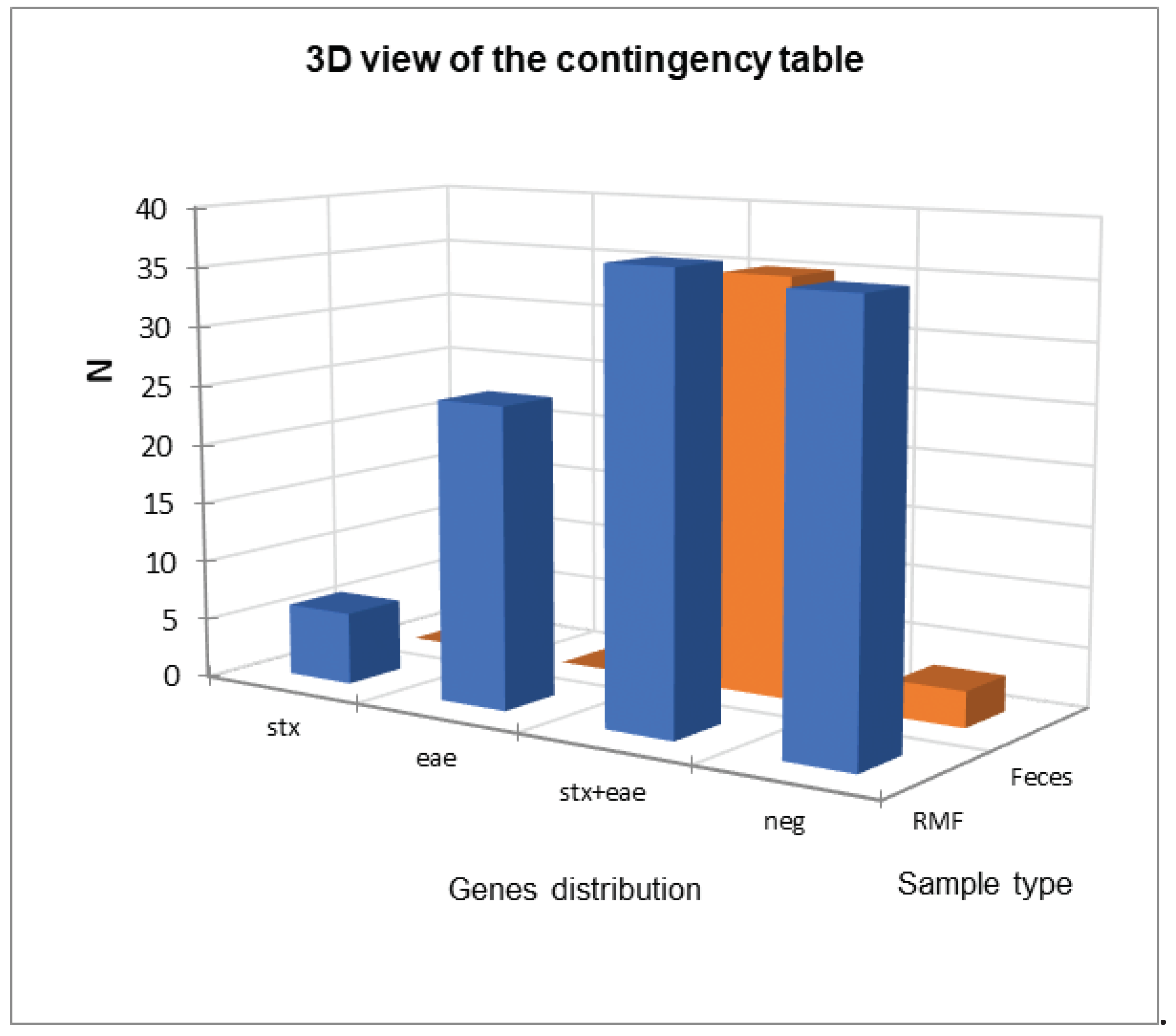
| Virulence gene | BTM samples N (%) | RMF samples N (%) |
| stx | 3 (3/88=3.4%) | 6 (6/104=5.8%) |
| eae | 10 (10/88=11.4%) | 25 (25/104=24%) |
| eae + stx | 0 (0/88=0%) | 37 (37/104=35.6%) |
| Negative | 75(75/88=85.2%) | 36 (36/104=34.6%) |
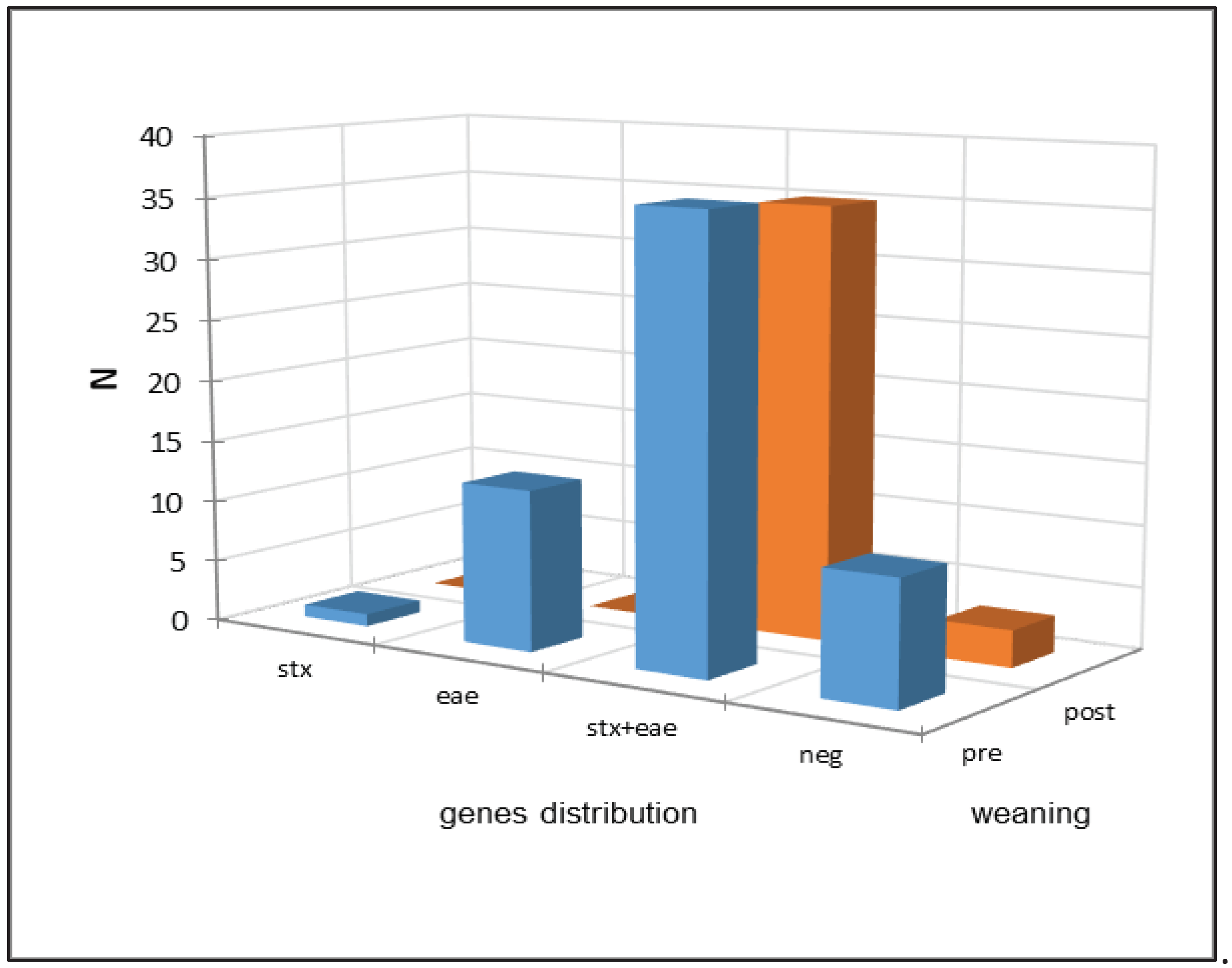
| Virulence gene | Calves’ feces N (%) | Pre-weaning N (%) | Post-weaning N (%) |
| stx | 1 (1/98=1%) | 1 (1/60=1.7%) | 0 (0/38=0%) |
| eae | 13 (13/98=13.3%) | 13 (13/60=21.6%) | 0 (0/38=0%) |
| eae + stx | 71 (71/98=72.4%) | 36 (36/60=60%) | 35 (35/38=92.1%) |
| Negative | 13 (13/98=13.3%) | 10 (10/60=16.7%) | 3 (3/38=7.9%) |
| Genes | RMF | Feces | ||
| Fisher's exact test (P) | Observed vs expected frequency |
Fisher's exact test (P) | Observed vs expected frequency | |
| stx | 0,192 | >1 | 0,192 | < |
| eae | 0,000 | > | 0,000 | < |
| stx+eae | <0,0001 | <2 | <0,0001 | > |
| negative | 0,001 | > | 0,001 | < |
| Genes | Pre-weaning | Post-weaning | ||
| Fisher's exact test (P) | Observed vs expected frequency |
Fisher's exact test (P) | Observed vs expected frequency | |
| stx | 1,000 | >1 | 1,000 | < |
| eae | 0,001 | > | 0,001 | < |
| stx+eae | 0,000 | <2 | 0,000 | > |
| negative | 0,360 | > | 0,360 | < |
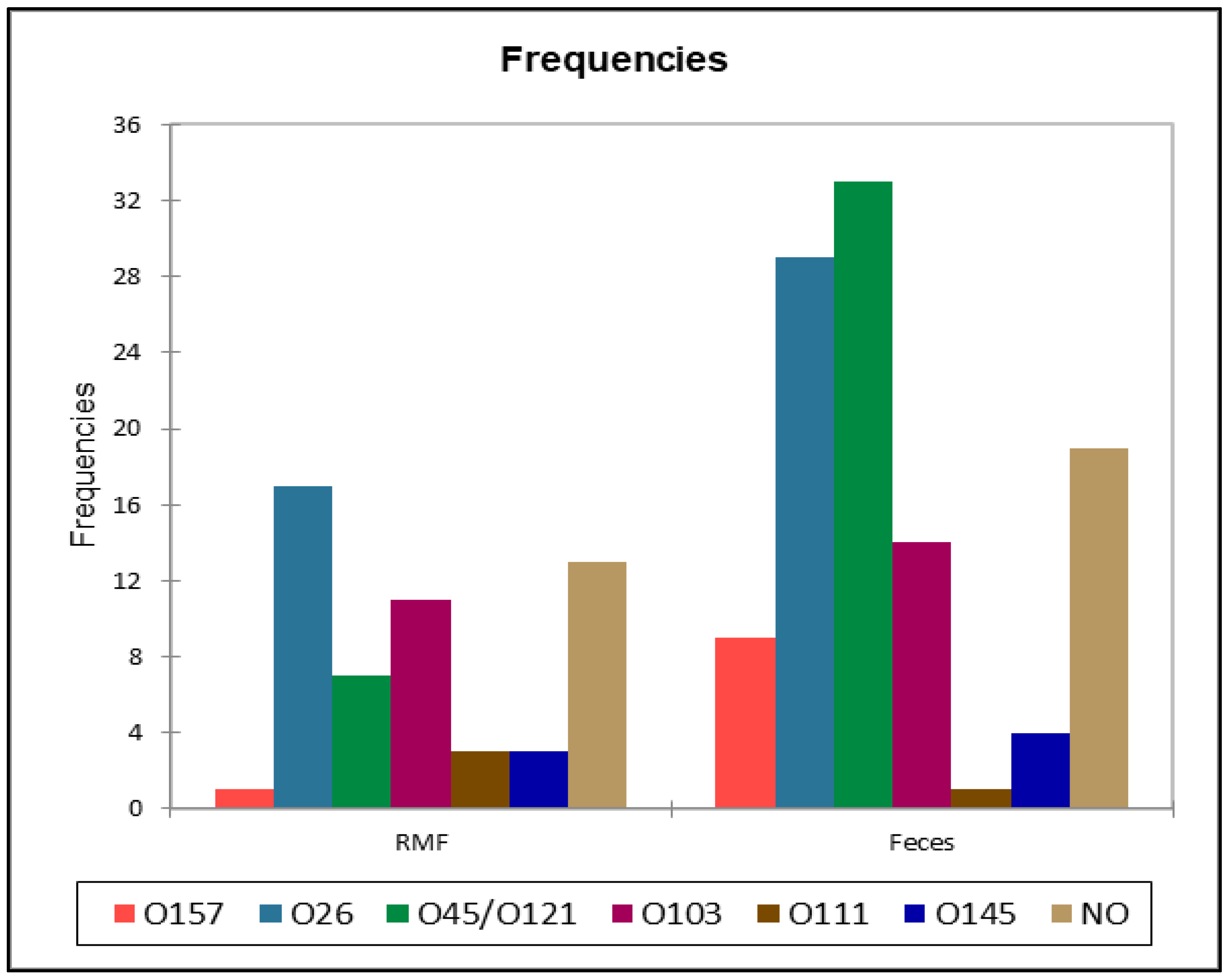
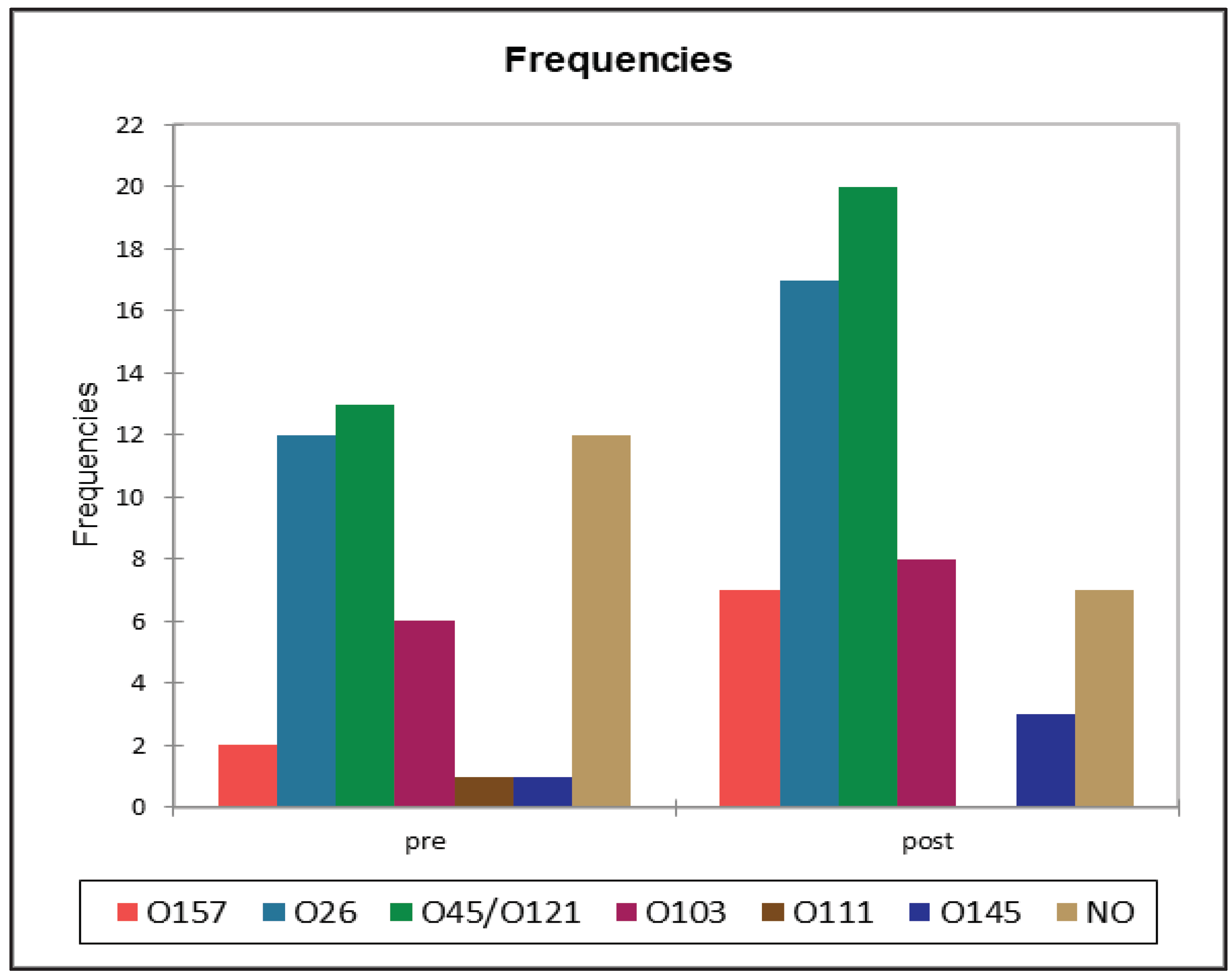
| Serotype | BTM samples (%) | RMF samples (%) | Feces pre-weaning (%) | Feces post-weaning (%) |
| O157 | 0 (0%) | 1 (0.9%) | 2 (3.3%) | 7 (18.4%) |
| O26 | 0 (0%) | 17 (16.3%) | 12 (20%) | 17 (44.7%) |
| O45/O121 | 0 (0%) | 7 (6.7%) | 13 (21.6%) | 20 (52.6%) |
| O103 | 0 (0%) | 11 (10.6%) | 6 (10%) | 8 (21.1%) |
| O111 | 0 (0%) | 3 (2.9%) | 1 (1.6%) | 0 (0%) |
| O145 | 0 (0%) | 3 (2.9%) | 1 (1.6%) | 3 (7.9%) |
| NO1 | 3 (3.4%) | 13 (12.5%) | 12 (20%) | 7 (18.4%) |
| Serotype | RMF | Feces | ||
| Fisher's exact test (P) | Observed vs expected frequency |
Fisher's exact test (P) | Observed vs expected frequency |
|
| O157 | 0,167 | < | 0,167 | > |
| O26 | 0,584 | > | 0,584 | < |
| O45/O121 | 0,013 | < | 0,013 | > |
| O103 | 0,254 | > | 0,254 | < |
| O111 | 0,110 | > | 0,110 | < |
| O145 | 0,688 | > | 0,688 | < |
| NO1 | 0,405 | > | 0,405 | < |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




