Submitted:
30 March 2024
Posted:
01 April 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
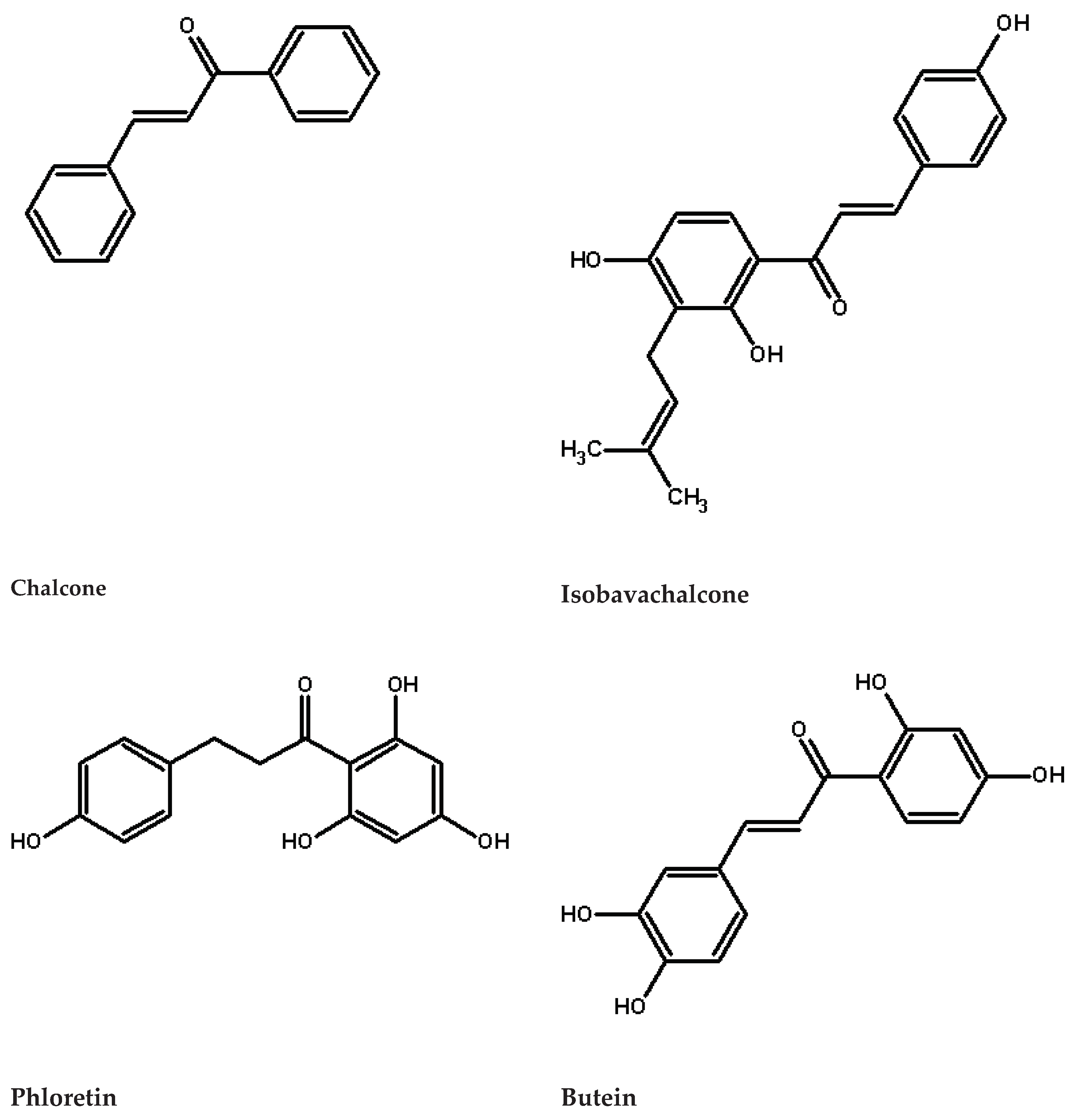
2. Characteristic
3. Chalkones Identification
Sample Preparation
Liquid Chromatography Coupled with Absorbance Detectors
Two Dimensional High Performance Liquid Chromatography (2D-HPLC)
Infrared Spectroscopy, FTIR and HNMR
Liquid Chromatography Coupled with Mass Spectrometry
MALDI Technique
4. Properties
Chalcones Biological Activities and Their Applications in Agriculture
Herbicides and Plant Growth Regulators
Fungicides
Nematicides
Insecticides
5. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Jash, S.K. Chemistry and Role of Flavonoids in Agriculture: A Recent Update. In Flavonoid Metabolism-Recent Advances and Applications in Crop Breeding. IntechOpen. [CrossRef]
- Panche, A.N.; Diwan, A.D.; Chandra, S.R. R. Flavonoids: an overview. J Nutr Sci. 2016, 5, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Tomás-Barberán, F.A.; Clifford, M.N. Flavanones, chalcones and dihydrochalcones - Nature, occurrence and dietary burden. Journal of the Science of Food and Agriculture 2000, 80, 1073–1080. [Google Scholar] [CrossRef]
- Chen, S.; Zhang, M.; Feng, S.; Gong, C.; Zhou, Y.; Xing Li He, B.; Wub, Y.; Xue, W. Design, synthesis and biological activity of chalcone derivatives containing pyridazine. Arabian Journal of Chemistry 2023, 16, 104852. [Google Scholar] [CrossRef]
- Rozmer, Z.; Perjési, P. Naturally occurring chalcones and their biological activities. Phytochemistry Reviews 2014, 15, 87–120. [Google Scholar] [CrossRef]
- Díaz-Tielas, C.; Graña, E.; Reigosa, M.J.; Sánchez-Moreiras, A.M. Biological Activities And Novel Applications Of Chalcones. Planta Daninha, Viçosa-MG 2016, 34, 607–616. [Google Scholar] [CrossRef]
- Zhou, B.; Xing, C. Diverse molecular targets for chalcones with varied bioactivities. Med Chem. 2015, 5, 388–404. [Google Scholar] [CrossRef] [PubMed]
- Kumar, R.; Sharma, P.; Shard, A.; Tewary, D.K.; Nadda, G.; Sinha, A.K. K. Chalcones as promising pesticidal agents against diamondback moth (Plutella xylostella): Microwave assisted synthesis and structure-activity relationship. Medicinal Chemistry Research 2012, 21, 922–931. [Google Scholar] [CrossRef]
- Diaz-Tielas, C.; Sotelo, T.; Grana, E.; Reigosa, M.J.; Sanchez-Moreiras. Phytotoxic potential of trans-chalcone on crop plants and model species. J. Plant Growth Regul. 2014, 33, 181–194. [Google Scholar] [CrossRef]
- Chotsaeng, N.; Laosinwattana, C.; Charoenying, P. Herbicidal Activity of Flavokawains and Related trans-Chalcones against Amaranthus tricolor L. and Echinochloa crus-galli (L.) Beauv. ACS Omega 2019, 4, 20748–20755. [Google Scholar] [CrossRef]
- Batovska, D.I.; Todorova, I.T. Trends in utilization of the pharmacological potential of chalcones. Curr Clin Pharmacol. 2010, 5, 1–29. [Google Scholar] [CrossRef]
- Kuete, V.; Sandjo, L.P. P. Isobavachalcone: An Overview. Chin J Integr Med 2012, 18, 543–547. [Google Scholar] [CrossRef] [PubMed]
- Aljamali, N.M.; Hamzah Daylee, S.; Jaber Kadhium, A. Review On Chemical-Biological Fields Of Chalcone Compounds. Forefront Journal of Engineering & Technology Volume 2020, 2, 33–44. [Google Scholar]
- Jasim, H.A.; Nahar, L.; Jasim, M.A.; Moore, S.A.; Ritchie, K.J.; Sarker, S.D. Chalcones: Synthetic Chemistry Follows Where Nature Leads. Biomolecules 2021, 11, 1203. [Google Scholar] [CrossRef] [PubMed]
- Dao, T.T.; Linthorst, H.J.; Verpoorte, R. Chalcone synthase and its functions in plant resistance. Phytochem Rev. 2011, 10, 397–412. [Google Scholar] [CrossRef] [PubMed]
- Jiwrajka, M.; Phillips, A.; Butler, M.; Rossi, M.; Pocock, J.M. The plant-derived chalcone 2, 2′, 5′-trihydroxychalcone provides neuroprotection against toll-like receptor 4 triggered inflammation in microglia. Oxidative Medicine and Cellular Longevity, 2016.
- Zhuang CZhang, W.; Sheng CZhang, W.; Xing, C. Chalcone: A privileged structure in medicinal chemistry. Chem. Rev. 2017, 117, 7762–7810. [Google Scholar] [CrossRef] [PubMed]
- Roychoudhury, A. 2023. Biology and Biotechnology of Environmental Stress Tolerance in Plants: Volume 1: Secondary Metabolites in Environmental Stress.
- Mahapatra, D.K.; Bharti, S.K.; Asati, V. Chalcone scaffolds as anti-infective agents: Structural and molecular target perspectives. Eur. J. Med. Chem. 2015, 101, 496–524. [Google Scholar] [CrossRef] [PubMed]
- Morsy, N.M.; Hassan, A.S. Synthesis, reactions, and applications of chalcones: A review. European Journal of Chemistry. 2022, 13, 241–252. [Google Scholar] [CrossRef]
- Balan-Porcăraşu, M.; Roman, G. Novel chalcone analogs derived from 4-(benzyloxy)benzaldehyde. Ovidius University Annals of Chemistry. 2023, 34, 112–120. [Google Scholar] [CrossRef]
- Zeng, L.; Zhang, R.Y.; Meng TLou, Z.C. C. Determination of nine flavonoids and coumarins in licorice root by high-performance liquid chromatography. J Chromatogr. 1990, 513, 247–254. [Google Scholar] [CrossRef]
- Burda, S.; Oleszek, W.; Lee, C.Y. Phenolic compounds and their changes in apples during maturation and cold storage. J Agric Food Chem 1998, 38, 945–948. [Google Scholar] [CrossRef]
- Suaârez, B.; Picinelli, A.; Moreno, J.; Mangas, J.J. J. Changes in phenolic composition of apple juices by HPLC with direct injection. J Sci Food Agric 1998, 78, 461–465. [Google Scholar] [CrossRef]
- Zou, H.; Han, L.; Yuan, M.; Zhang, M.; Zhou, L.; Wang, Y. Sequence Analysis and Functional Verification of the Effects of Three Key Structural Genes, PdTHC2’GT, PdCHS and PdCHI, on the Isosalipurposide Synthesis Pathway in Paeonia delavayi var. lutea. International Journal of Mol. Sci. 2022, 23, 5696. [Google Scholar] [CrossRef] [PubMed]
- Krauze-Baranowska, M.; Pobłocka-Olech, M.; Głód, D.; Wiwart, M.; Zieliński, J.; Migas, P. HPLC of Flavanones and Chalcones in different species and clones of Salix. Acta Poloniae Pharmaceutica - Drug Research 2014, 70, 27–34. [Google Scholar]
- Guvenalp, Z.; Ozbek, H.; Karadayi, M.; Gulluce, M.; Kuruuzum-Uz, A.; Salih, B.; Demirezer, O. Two antigenotoxic chalcone glycosides from Mentha longifolia subsp. Longifolia. Pharm Biol. 2015, 53, 888–896. [Google Scholar] [CrossRef] [PubMed]
- Chen, Q.; Li, Y.; Chen, Z. Separation, identification, and quantification of active constituents in Fructus Psoraleae by high-performance liquid chromatography with UV, ion trap mass spectrometry, and electrochemical detection. Journal of Pharmaceutical Analysis. 2012, 2, 143–151. [Google Scholar] [CrossRef] [PubMed]
- Pobłocka-Olech, L. Zastosowanie metod chromatograficznych w badaniach składu chemicznego kory niektórych gatunków i klonów wierzby. Doctoral dissertation. 2016.
- Dhaliwal, J.S.; Moshawih, S.; Goh, K.W.; Loy, M.J.; Hossain, M.S.; Hermansyah, A.; Kotra, V.; Kifli, N.; Goh, H.P.; Dhaliwal, S.K.S.; et al. Pharmacotherapeutics Applications and Chemistry of Chalcone Derivatives. Molecules 2022, 27, 7062. [Google Scholar] [CrossRef] [PubMed]
- Hassan, S.M.; Hassan, S.A.; Abbas, A.K.; Najem, M.R.; Jber, N.R. R. Synthesis of heterocyclic and study activities in agriculture as anti-dubas on date palm trees via cholinesterase inhibitors. GSC Advanced Research and Reviews 2023, 16, 094–101. [Google Scholar] [CrossRef]
- Ma, T.; Nie, L.J.; Li, H.M.; Huo, Q.; Zhang, Y.X.; Wu, C.Z. Z. Determination of isobavachalcone in rat plasma by LC-MS/MS and its application to a pharmacokinetic study. J Pharm Biomed Anal. 2015, 107, 50–55. [Google Scholar] [CrossRef]
- Krittanaia, S.; Pichetpongtorn, P.; Sakamoto, S.; Waraporn, P. Monoclonal antibody-based immunoassay for the specific quantification of licochalcone A: an active chalcone in licorice. Food And Agricultural Immunology. 2022, 33, 220–234. [Google Scholar] [CrossRef]
- Yerragunta, V.; Suman, D.; Anusha, V.; Patil, P.; Samhitha, T. A review on chalcones and its importance. PharmaTutor 2013, 1, 54–55. [Google Scholar]
- Perera, H.; Meepagala, K.M.; Fronczek, F.R.; Cook, D.D.; Wedge, D.E.; Duke, S.O. O. Bioassay-Guided Isolation and Structure Elucidation of Fungicidal and Herbicidal Compounds from Ambrosia salsola (Asteraceae). Molecules 2019, 24, 835. [Google Scholar] [CrossRef] [PubMed]
- Yun, M.S.; Chen, W.; Deng, F.; Yogo, Y. Selective growth suppression of five annual plant species by chalcone and naringenin correlates with the total amount of 4coumarate: Coenzyme A ligase. Weed Biology and Management 2009, 9, 27–37. [Google Scholar] [CrossRef]
- Nguyen, G.T.T.; Erlenkamp, G.; Jäck, O.; Küberl, A.; Bott, M.; Fiorani, F.; Gohlke, H.; Groth, G. Chalcone-based selective inhibitors of a C4 plant key enzyme as novel potential herbicides. Scientific reports 2016, 6, 27333. [Google Scholar] [CrossRef] [PubMed]
- Díaz-Tielas, C.; Graña, E.; Sotelo, T.; Reigosa, M.J.; Sánchez-Moreiras, A.M. The natural compound trans-chalcone induces programmed cell death in Arabidopsis thaliana roots. Plant, Cell and Environment 2012, 35, 1500–1517. [Google Scholar] [CrossRef] [PubMed]
- Smailagić, D.; Banjac, N.; Ninković, S.; Savić, J.; Ćosić, T.; Pěnčík, A.; Ćalić, D; Bogdanović, M. ; Trajković, M.; Stanišić, M. New insights into the activity of apple dihydrochalcone phloretin: disturbance of auxin homeostasis as physiological basis of phloretin phytotoxic action. Frontiers in Plant Science 2022, 13, 875528. [Google Scholar] [CrossRef]
- Gupta, D.; Jain, D.K. Chalcone derivatives as potential antifungal agents: Synthesis, and antifungal activity. J. Adv. Pharm. Technol. Res. 2015, 6, 114–117. [Google Scholar] [CrossRef] [PubMed]
- Svetaz, L.; Tapia, A.; López, S.N.; Furlán, R.L.; Petenatti, E.; Pioli, R.; Schmeda-Hirschmann, G.; Zacchino, S.A. A. Antifungal chalcones and new caffeic acid esters from Zuccagnia punctata acting against soybean infecting fungi. Journal of Agricultural and Food chemistry 2004, 52, 3297–3300. [Google Scholar] [CrossRef] [PubMed]
- Badaracco, P.; Sortino, M.; Pioli, R.N. N. Study plant-origin in compounds with potential antifungal action against pathogens of cultivated plants. Chilean journal of agricultural & animal sciences 2020, 36, 244–252. [Google Scholar]
- Oleszek, M.; Pecio, Ł.; Kozachok, S.; Lachowska-Filipiuk, Ż.; Oszust, K.; Frąc, M. Phytochemicals of apple pomace as prospect bio-fungicide agents against mycotoxigenic fungal species—In vitro experiments. Toxins 2019, 11, 361. [Google Scholar] [CrossRef]
- Zhou, Q.; Tang, X.; Chen, S.; Zhan, W.; Hu, D.; Zhou, R.; Sun, N.; YongJun, W.; Xue, W. Design, synthesis, and antifungal activity of novel chalcone derivatives containing a piperazine fragment. Journal of Agricultural and Food Chemistry 2022, 70, 1029–1036. [Google Scholar] [CrossRef] [PubMed]
- Malhotra, B.; Onyilagha, J.C.; Bohm, B.A.; Towers GH, N.; James, D.; Harborne, J.B.; French, C.J. J. Inhibition of tomato ringspot virus by flavonoids. Phytochemistry 1996, 43, 1271–1276. [Google Scholar] [CrossRef]
- Onyilagha, J.C.; Malhotra, B.; Elder, M.; French, C.J.; Towers, G.N. N. Comparative studies of inhibitory activities of chalcones on tomato ringspot virus (ToRSV). Canadian Journal of Plant Pathology 1997, 19, 133–137. [Google Scholar] [CrossRef]
- Dong, L.R.; Hu, D.Y.; Wu, Z.X.; Chen, J.X.; Song, B.A. Study of the synthesis, antiviral bioactivity and interaction mechanisms of novel chalcone derivatives that contain the 1, 1-dichloropropene moiety. Chinese Chemical Letters 2017, 28, 1566–1570. [Google Scholar] [CrossRef]
- Zhou, D.; Xie, D.; He, F.; Song, B.; Hu, D. Antiviral properties and interaction of novel chalcone derivatives containing a purine and benzenesulfonamide moiety. Bioorganic & medicinal chemistry letters 2018, 28, 2091–2097. [Google Scholar]
- Zhang, W.; Mao, P.; Yuan Ch Zhang, Y.; Zhang, T.; Liu, Y.; Tian, J.; Xue, W. Design, synthesis and antiviral activities of chalcone derivatives containing pyrimidine. Journal of Saudi Chemical Society 2023, 27, 101590. [Google Scholar] [CrossRef]
- Silva, F.J.; Campos, V.P.; Oliveira, D.F.; Gomes, V.A.; Barros, A.F.; Din, Z.U.; Rodrigues-Filho, E. Chalcone analogues: Synthesis, activity against Meloidogyne incognita, and in silico interaction with cytochrome P450. Journal of Phytopathology 2019, 167, 197–208. [Google Scholar] [CrossRef]
- Cao, X.; Qiu, D.; Zhang, R.; Li, Z.; Xu, X. Synthesis, nematicidal evaluation, and SAR study of benzofuran derivatives containing 2-carbonyl thiophene. Chinese Chemical Letters 2023, 34, 107800. [Google Scholar] [CrossRef]
- Attar, S.; O’Brien, Z.; Alhaddad, H.; Golden, M.L.; Calderón-Urrea, A. Ferrocenyl chalcones versus organic chalcones: a comparative study of their nematocidal activity. Bioorganic & medicinal chemistry 2011, 19, 2055–2073. [Google Scholar]
- Stompor, M.; Dancewicz, K.; Gabrys, B.; Anioł, M. Insect antifeedant potential of xanthohumol, isoxanthohumol, and their derivatives. Journal of agricultural and food chemistry 2015, 63, 6749–6756. [Google Scholar] [CrossRef]
- Shakil, N.A.; Saxena, D.B. Isolation and structure of cordifolin, a novel insecticidal oxygenated chalcone, from the stem of Tinospora cordifolia Miers. Natural Product Communications 2006, 1, 553–556. [Google Scholar] [CrossRef]
- Hidalgo, J.R.; Santillán, M.; Parellada, E.A.; Khyaliya, P.; Neske, A.; Ameta, K.L. Synthetic bis-and mono-chalcones with insecticide effects on Spodoptera frugiperda (Lepidoptera: Noctuidae). International journal of pest management 2020, 66, 116–121. [Google Scholar] [CrossRef]
- Devi, A.P.; Alsulimani, A.; Hidalgo, J.R.; Neske, A.; Sayyed, R.Z.; Hassan, M.; Elshazly, H. Bis-and mono-substituted Chalcones exert anti-feedant and toxic effects on fall armyworm Spodoptera frugiperda. Saudi Journal of Biological Sciences 2021, 28, 5754–5759. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



