Submitted:
01 April 2024
Posted:
01 April 2024
You are already at the latest version
Abstract
Keywords:
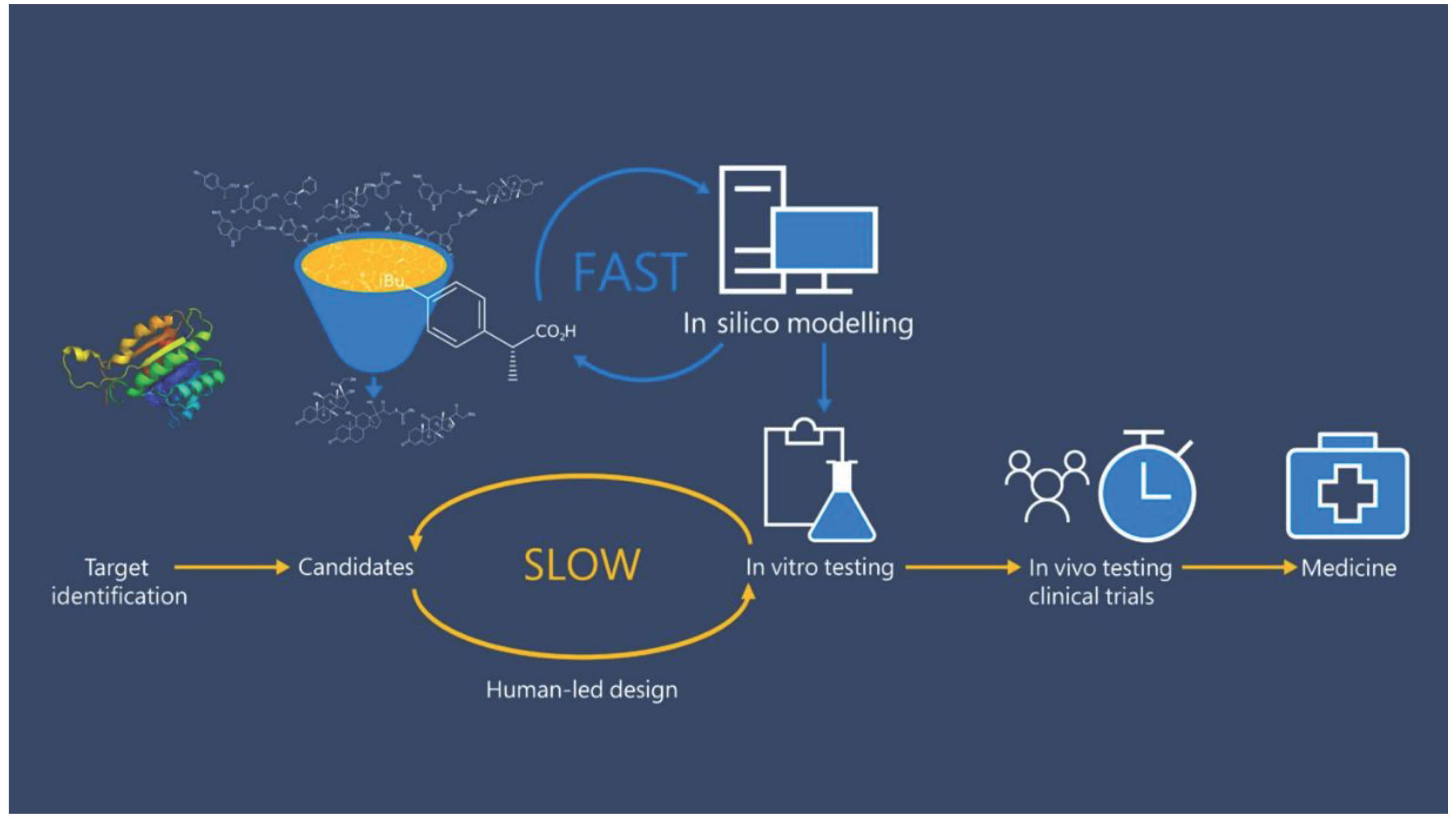
1. Introduction
2. Traditional Methods vs AI-driven Lead Optimization:
3. Key Future Optimization Strategies:
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Biswas, R.; Basu, A.; Nandy, A.; Deb, A.; Haque, K.M.G.; Chanda, D. Drug Discovery and Drug Identification using AI. 2020 Indo – Taiwan 2nd International Conference on Computing, Analytics and Networks (Indo-Taiwan ICAN) 2020, 49-51.
- McNair, D. Artificial Intelligence and Machine Learning for Lead-to-Candidate Decision-Making and Beyond. Annual review of pharmacology and toxicology 2022. [Google Scholar] [CrossRef] [PubMed]
- Tiwari, P.C.; Pal, R.; Chaudhary, M.J.; Nath, R. Artificial intelligence revolutionizing drug development: Exploring opportunities and challenges. Drug Development Research 2023, 84, 1652–1663. [Google Scholar] [CrossRef] [PubMed]
- Unogwu, O.J.; Ike, M.; Joktan, O.O. Employing Artificial Intelligence Methods in Drug Development: A New Era in Medicine. Mesopotamian Journal of Artificial Intelligence in Healthcare 2023. [Google Scholar] [CrossRef] [PubMed]
- Schwardt, O.; Kolb, H.C.; Ernst, B. Drug discovery today. Current topics in medicinal chemistry 2003, 3, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Zieliński, A. AI and the future of pharmaceutical research. arXiv 2021. arXiv, 2107; arXiv:2107.03896.
- Xie, W.; Cheng, X.; Ding, Z.; Deng, R.; Gu, D. Abstract 278: Artificial intelligence accelerate drug discovery. Cancer Chemistry 2021. [Google Scholar] [CrossRef]
- Kenakin, T. Predicting therapeutic value in the lead optimization phase of drug discovery. Nat Rev Drug Discov 2003, 2, 429–438. [Google Scholar] [CrossRef] [PubMed]
- Hoffer, L.; Muller, C.; Roche, P.; Morelli, X. Chemistry-driven Hit-to-lead Optimization Guided by Structure-based Approaches. Mol Inform 2018, 37, e1800059. [Google Scholar] [CrossRef]
- Broccatelli, F.; C, E.C.A.H.; Wright, M. Strategies to optimize drug half-life in lead candidate identification. Expert Opin Drug Discov 2019, 14, 221–230. [Google Scholar] [CrossRef] [PubMed]
- Braggio, S.; Montanari, D.; Rossi, T.; Ratti, E. Drug efficiency: a new concept to guide lead optimization programs towards the selection of better clinical candidates. Expert Opin Drug Discov 2010, 5, 609–618. [Google Scholar] [CrossRef]
- Das, B.; Baidya, A.T.K.; Mathew, A.T.; Yadav, A.K.; Kumar, R. Structural modification aimed for improving solubility of lead compounds in early phase drug discovery. Bioorg Med Chem 2022, 56, 116614. [Google Scholar] [CrossRef] [PubMed]
- Yadav, S.; Ahamad, S.; Gupta, D.; Mathur, P. Lead optimization, pharmacophore development and scaffold design of protein kinase CK2 inhibitors as potential COVID-19 therapeutics. J Biomol Struct Dyn 2023, 41, 1811–1827. [Google Scholar] [CrossRef] [PubMed]
- Varma, M.V.; Khandavilli, S.; Ashokraj, Y.; Jain, A.; Dhanikula, A.; Sood, A.; Thomas, N.S.; Pillai, O.; Sharma, P.; Gandhi, R.; et al. Biopharmaceutic classification system: a scientific framework for pharmacokinetic optimization in drug research. Curr Drug Metab 2004, 5, 375–388. [Google Scholar] [CrossRef] [PubMed]
- Pedreira, J.G.B.; Franco, L.S.; Barreiro, E.J. Chemical Intuition in Drug Design and Discovery. Curr Top Med Chem 2019, 19, 1679–1693. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Kogej, T.; Engkvist, O. Cheminformatics in Drug Discovery, an Industrial Perspective. Mol Inform 2018, 37, e1800041. [Google Scholar] [CrossRef] [PubMed]
- AxDrug. AI & Computational Chemistry powered Drug Discovery Platform as a Service - Immunocure CRO. Availabe online: (accessed on 30-01-2024).
- Vora, L.K.; Gholap, A.D.; Jetha, K.; Thakur, R.R.S.; Solanki, H.K.; Chavda, V.P. Artificial Intelligence in Pharmaceutical Technology and Drug Delivery Design. Pharmaceutics 2023, 15. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Deng, Z.; Xu, X.; Feng, Y.; Junliang, S. Application of Artificial Intelligence in Drug-Drug Interactions Prediction: A Review. J Chem Inf Model 2023. [CrossRef]
- Li, G.; Lin, P.; Wang, K.; Gu, C.C.; Kusari, S. Artificial intelligence-guided discovery of anticancer lead compounds from plants and associated microorganisms. Trends Cancer 2022, 8, 65–80. [Google Scholar] [CrossRef] [PubMed]
- McNair, D. Artificial Intelligence and Machine Learning for Lead-to-Candidate Decision-Making and Beyond. Annu Rev Pharmacol Toxicol 2023, 63, 77–97. [Google Scholar] [CrossRef] [PubMed]
- Moshawih, S.; Goh, H.P.; Kifli, N.; Idris, A.C.; Yassin, H.; Kotra, V.; Goh, K.W.; Liew, K.B.; Ming, L.C. Synergy between machine learning and natural products cheminformatics: Application to the lead discovery of anthraquinone derivatives. Chem Biol Drug Des 2022, 100, 185–217. [Google Scholar] [CrossRef] [PubMed]
- Saifi, I.; Bhat, B.A.; Hamdani, S.S.; Bhat, U.Y.; Lobato-Tapia, C.A.; Mir, M.A.; Dar, T.U.H.; Ganie, S.A. Artificial intelligence and cheminformatics tools: a contribution to the drug development and chemical science. J Biomol Struct Dyn 2023. [CrossRef]
- Liu, T.; Zuo, R.; Wang, J.; Huangtao, Z.; Wang, B.; Sun, L.; Wang, S.; Li, B.; Zhu, Z.; Pan, Y. Cardiovascular disease preventive effects of aspirin combined with different statins in the United States general population. Sci Rep 2023, 13, 4585. [Google Scholar] [CrossRef] [PubMed]
- Liu, Q.; Li, Y.; Niu, Z.; Zong, Y.; Wang, M.; Yao, L.; Lu, Z.; Liao, Q.; Zhao, Y. Atorvastatin (Lipitor) attenuates the effects of aspirin on pancreatic cancerogenesis and the chemotherapeutic efficacy of gemcitabine on pancreatic cancer by promoting M2 polarized tumor associated macrophages. J Exp Clin Cancer Res 2016, 35, 33. [Google Scholar] [CrossRef] [PubMed]
- Mahtta, D.; Ramsey, D.J.; Al Rifai, M.; Nasir, K.; Samad, Z.; Aguilar, D.; Jneid, H.; Ballantyne, C.M.; Petersen, L.A.; Virani, S.S. Evaluation of Aspirin and Statin Therapy Use and Adherence in Patients With Premature Atherosclerotic Cardiovascular Disease. JAMA Netw Open 2020, 3, e2011051. [Google Scholar] [CrossRef] [PubMed]
- Kang, K.M.; Jang, Y.; Lee, S.S.; Jin, M.S.; Jun, C.D.; Kim, M.; Kim, Y.C. Discovery of antiviral SARS-CoV-2 main protease inhibitors by structure-guided hit-to-lead optimization of carmofur. Eur J Med Chem 2023, 260, 115720. [Google Scholar] [CrossRef] [PubMed]
- Mengist, H.M.; Mekonnen, D.; Mohammed, A.; Shi, R.; Jin, T. Potency, Safety, and Pharmacokinetic Profiles of Potential Inhibitors Targeting SARS-CoV-2 Main Protease. Front Pharmacol 2020, 11, 630500. [Google Scholar] [CrossRef] [PubMed]
- Najmi, A.; Javed, S.A.; Al Bratty, M.; Alhazmi, H.A. Modern Approaches in the Discovery and Development of Plant-Based Natural Products and Their Analogues as Potential Therapeutic Agents. Molecules 2022, 27. [Google Scholar] [CrossRef] [PubMed]
- Karuppasamy, M.K.M.a.M. Fundamental considerations in drug design. Computer Aided Drug Design (CADD): From Ligand-Based Methods to Structure-Based Approaches, 17–55 2022. [CrossRef]
- de Souza Neto, L.R.; Moreira-Filho, J.T.; Neves, B.J.; Maidana, R.; Guimaraes, A.C.R.; Furnham, N.; Andrade, C.H.; Silva, F.P., Jr. In silico Strategies to Support Fragment-to-Lead Optimization in Drug Discovery. Front Chem 2020, 8, 93. [Google Scholar] [CrossRef] [PubMed]
- Grebner, C.; Matter, H.; Hessler, G. Artificial Intelligence in Compound Design. Methods Mol Biol 2022, 2390, 349–382. [Google Scholar] [CrossRef]
- Langevin, M.; Minoux, H.; Levesque, M.; Bianciotto, M. Scaffold-Constrained Molecular Generation. J Chem Inf Model 2020, 60, 5637–5646. [Google Scholar] [CrossRef]
- Cavasotto, C.N. Binding Free Energy Calculation Using Quantum Mechanics Aimed for Drug Lead Optimization. Methods Mol Biol 2020, 2114, 257–268. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.Q.; Chen, H.Y.; Dai, W.J.; Lv, Q.J.; Chen, C.Y. Artificial Intelligence Approach to Find Lead Compounds for Treating Tumors. J Phys Chem Lett 2019, 10, 4382–4400. [Google Scholar] [CrossRef] [PubMed]
- Joshi, R.P.; Kumar, N. Artificial Intelligence for Autonomous Molecular Design: A Perspective. Molecules 2021, 26. [Google Scholar] [CrossRef] [PubMed]
- Loos, N.H.C.; Sparidans, R.W.; Heydari, P.; Bui, V.; Lebre, M.C.; Beijnen, J.H.; Schinkel, A.H. The ABCB1 and ABCG2 efflux transporters limit brain disposition of the SYK inhibitors entospletinib and lanraplenib. Toxicol Appl Pharmacol 2024, 485, 116911. [Google Scholar] [CrossRef] [PubMed]
- Blomgren, P.; Chandrasekhar, J.; Di Paolo, J.A.; Fung, W.; Geng, G.; Ip, C.; Jones, R.; Kropf, J.E.; Lansdon, E.B.; Lee, S.; et al. Discovery of Lanraplenib (GS-9876): A Once-Daily Spleen Tyrosine Kinase Inhibitor for Autoimmune Diseases. ACS Med Chem Lett 2020, 11, 506–513. [Google Scholar] [CrossRef] [PubMed]
- Bleicher, L.S., Van Daelen, T., Honeycutt, J. D., Hassan, M., Chandrasekhar, J., Shirley, W., Tsui, V., & Schmitz, U. . Enhanced utility of AI/ML methods during lead optimization by inclusion of 3D ligand information. Frontiers in Drug Discovery, 2, 1074797. [CrossRef]
- Hoffman, S.C. , Chenthamarakshan, V., Wadhawan, K. et al. Optimizing molecules using efficient queries from property evaluations. Nat Mach Intell 4, 21–31. [CrossRef]
- Zhu, W.; Liu, X.; Li, Q.; Gao, F.; Liu, T.; Chen, X.; Zhang, M.; Aliper, A.; Ren, F.; Ding, X.; et al. Discovery of novel and selective SIK2 inhibitors by the application of AlphaFold structures and generative models. Bioorg Med Chem 2023, 91, 117414. [Google Scholar] [CrossRef] [PubMed]
- Ivanenkov, Y.A.; Polykovskiy, D.; Bezrukov, D.; Zagribelnyy, B.; Aladinskiy, V.; Kamya, P.; Aliper, A.; Ren, F.; Zhavoronkov, A. Chemistry42: An AI-Driven Platform for Molecular Design and Optimization. J Chem Inf Model 2023, 63, 695–701. [Google Scholar] [CrossRef] [PubMed]
- Ren, F.; Ding, X.; Zheng, M.; Korzinkin, M.; Cai, X.; Zhu, W.; Mantsyzov, A.; Aliper, A.; Aladinskiy, V.; Cao, Z.; et al. AlphaFold accelerates artificial intelligence powered drug discovery: efficient discovery of a novel CDK20 small molecule inhibitor. Chem Sci 2023, 14, 1443–1452. [Google Scholar] [CrossRef] [PubMed]
- Turon, G.; Hlozek, J.; Woodland, J.G.; Kumar, A.; Chibale, K.; Duran-Frigola, M. First fully-automated AI/ML virtual screening cascade implemented at a drug discovery centre in Africa. Nat Commun 2023, 14, 5736. [Google Scholar] [CrossRef] [PubMed]
- Narayanan, H.; Dingfelder, F.; Condado Morales, I.; Patel, B.; Heding, K.E.; Bjelke, J.R.; Egebjerg, T.; Butte, A.; Sokolov, M.; Lorenzen, N.; et al. Design of Biopharmaceutical Formulations Accelerated by Machine Learning. Mol Pharm 2021, 18, 3843–3853. [Google Scholar] [CrossRef] [PubMed]
- Mousaei, M.; Kudaibergenova, M.; MacKerell, A.D., Jr.; Noskov, S. Assessing hERG1 Blockade from Bayesian Machine-Learning-Optimized Site Identification by Ligand Competitive Saturation Simulations. J Chem Inf Model 2020, 60, 6489–6501. [Google Scholar] [CrossRef] [PubMed]
- Hughes, J.H.; Long-Boyle, J.; Keizer, R.J. Maximum a posteriori Bayesian methods out-perform non-compartmental analysis for busulfan precision dosing. J Pharmacokinet Pharmacodyn 2024. [CrossRef]
- Lee, J.W.; Maria-Solano, M.A.; Vu, T.N.L.; Yoon, S.; Choi, S. Big data and artificial intelligence (AI) methodologies for computer-aided drug design (CADD). Biochem Soc Trans 2022, 50, 241–252. [Google Scholar] [CrossRef] [PubMed]
- Thakur, A.; Mishra, A.P.; Panda, B.; Rodriguez, D.C.S.; Gaurav, I.; Majhi, B. Application of Artificial Intelligence in Pharmaceutical and Biomedical Studies. Curr Pharm Des 2020, 26, 3569–3578. [Google Scholar] [CrossRef] [PubMed]
- Ryeznik, Y.; Sverdlov, O.; Svensson, E.M.; Montepiedra, G.; Hooker, A.C.; Wong, W.K. Pharmacometrics meets statistics-A synergy for modern drug development. CPT Pharmacometrics Syst Pharmacol 2021, 10, 1134–1149. [Google Scholar] [CrossRef] [PubMed]
- Moore, C.M.; MaWhinney, S.; Carlson, N.E.; Kreidler, S. A Bayesian natural cubic B-spline varying coefficient method for non-ignorable dropout. BMC Med Res Methodol 2020, 20, 250. [Google Scholar] [CrossRef] [PubMed]
- Dr. Christoph Grebner, D.H.M., Dr. Daniel Kofink, Dr. Jan Wenzel, Dr. Friedemann Schmidt, Dr. Gerhard Hessler. Application of Deep Neural Network Models in Drug Discovery Programs. Wiley 2021, ChemMedChem, Volume 16, Issue 24. [CrossRef]
- Jaeger, S.; Fulle, S.; Turk, S. Mol2vec: Unsupervised Machine Learning Approach with Chemical Intuition. J Chem Inf Model 2018, 58, 27–35. [Google Scholar] [CrossRef] [PubMed]
- Yu, L.; Zhou, D.; Gao, L.; Zha, Y. Prediction of drug response in multilayer networks based on fusion of multiomics data. Methods 2021, 192, 85–92. [Google Scholar] [CrossRef] [PubMed]
- Amendola, G.; Ettari, R.; Previti, S.; Di Chio, C.; Messere, A.; Di Maro, S.; Hammerschmidt, S.J.; Zimmer, C.; Zimmermann, R.A.; Schirmeister, T.; et al. Lead Discovery of SARS-CoV-2 Main Protease Inhibitors through Covalent Docking-Based Virtual Screening. J Chem Inf Model 2021, 61, 2062–2073. [Google Scholar] [CrossRef] [PubMed]
- Gusev, F.; Gutkin, E.; Kurnikova, M.G.; Isayev, O. Active Learning Guided Drug Design Lead Optimization Based on Relative Binding Free Energy Modeling. J Chem Inf Model 2023, 63, 583–594. [Google Scholar] [CrossRef] [PubMed]
- Niclas Ståhl, G.F. , Alexander Karlsson, Gunnar Mathiason, and Jonas Boström. Deep Reinforcement Learning for Multiparameter Optimization in de novo Drug Design. American Chemical Society Publications 2019, 7, 3166–3176. [Google Scholar] [CrossRef]
- Quentin Perron, O.M., Hamza Tajmouati, Adam Skiredj, Anne Rojas, Arnaud Gohier, Pierre Ducrot, Marie-Pierre Bourguignon, Patricia Sansilvestri-Morel, Nicolas Do Huu, Françoise Gellibert, Yann Gaston-Mathé. Deep generative models for ligand-based de novo design applied to multi-parametric optimization. Wiley 2022. [CrossRef]
- Zhong, F.; Xing, J.; Li, X.; Liu, X.; Fu, Z.; Xiong, Z.; Lu, D.; Wu, X.; Zhao, J.; Tan, X.; et al. Artificial intelligence in drug design. Sci China Life Sci 2018, 61, 1191–1204. [Google Scholar] [CrossRef]
- Nayarisseri, A.; Khandelwal, R.; Tanwar, P.; Madhavi, M.; Sharma, D.; Thakur, G.; Speck-Planche, A.; Singh, S.K. Artificial Intelligence, Big Data and Machine Learning Approaches in Precision Medicine & Drug Discovery. Curr Drug Targets 2021, 22, 631–655. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



