Submitted:
22 March 2024
Posted:
25 March 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Evolution in NMR Metabolomics Research - Part I: Feasibility of Lung Cancer Detection via Blood Plasma Using NMR Spectroscopy
2.1. Feasibility: Lung Cancer Detection
2.1.1. Study Set-Up
2.1.2. Results
2.1.3. Supporting Evidence
2.2. Feasibility: Differentiation between Cancer Types: Lung Cancer versus Breast Cancer
2.2.1. Study Set-Up
2.2.2. Results
3. Evolution in NMR Metabolomics Research - Part II: Developments in Preanalytical Sample Preparation and NMR Measurement Procedure
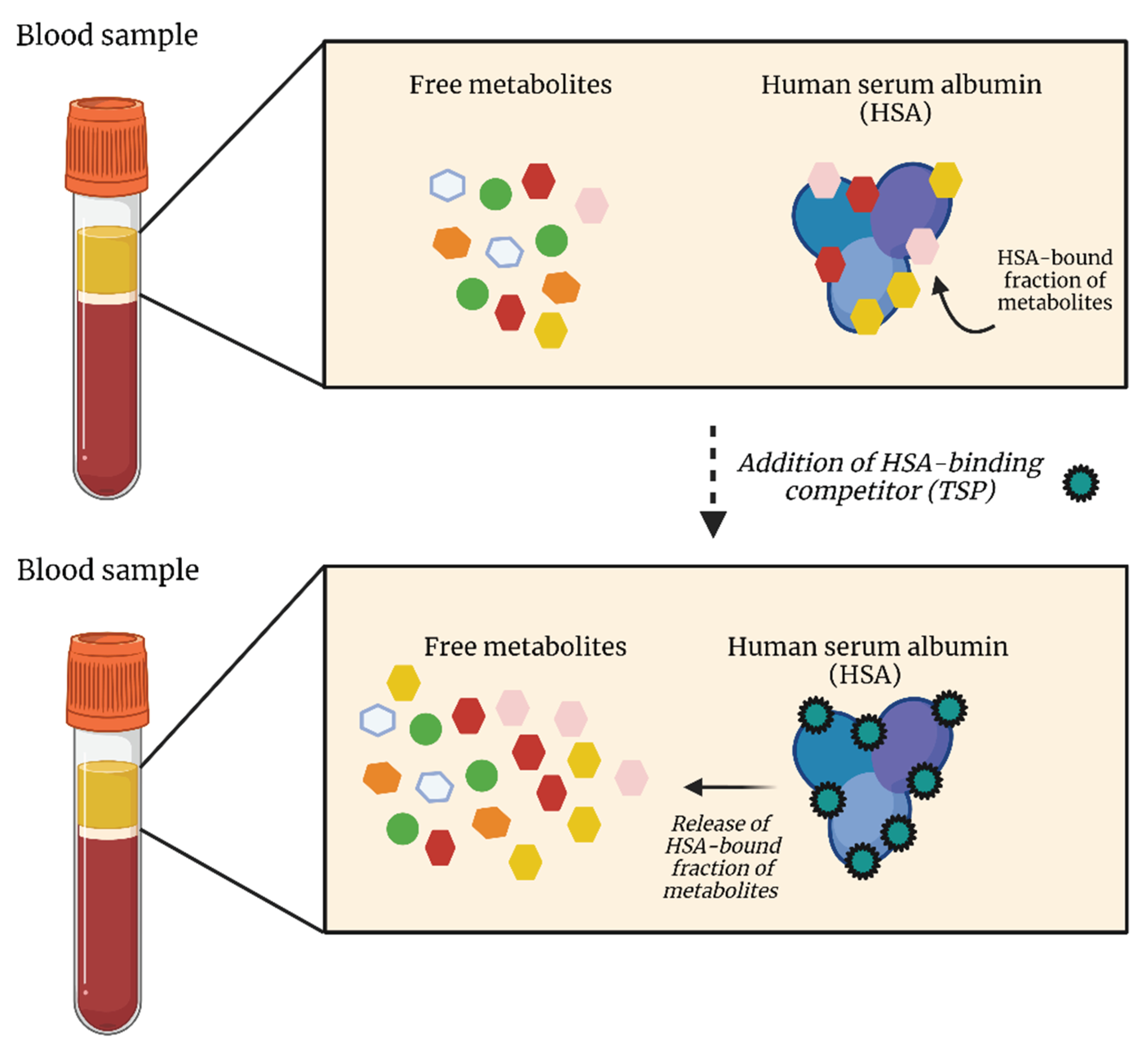
3.1. TSP as an HSA Binding Competitor and MA as an Internal Standard
3.2. Study Design
3.3. Results
4. Evolution in NMR Metabolomics Research - Part III: Plasma Biomarkers for Early-Stage NSCLC and Their Potential for Detection and Monitoring of NSCLC Recurrence
4.1. Study Design
4.2. Results
5. Discussion of Breakthroughs in NMR Metabolomics Research
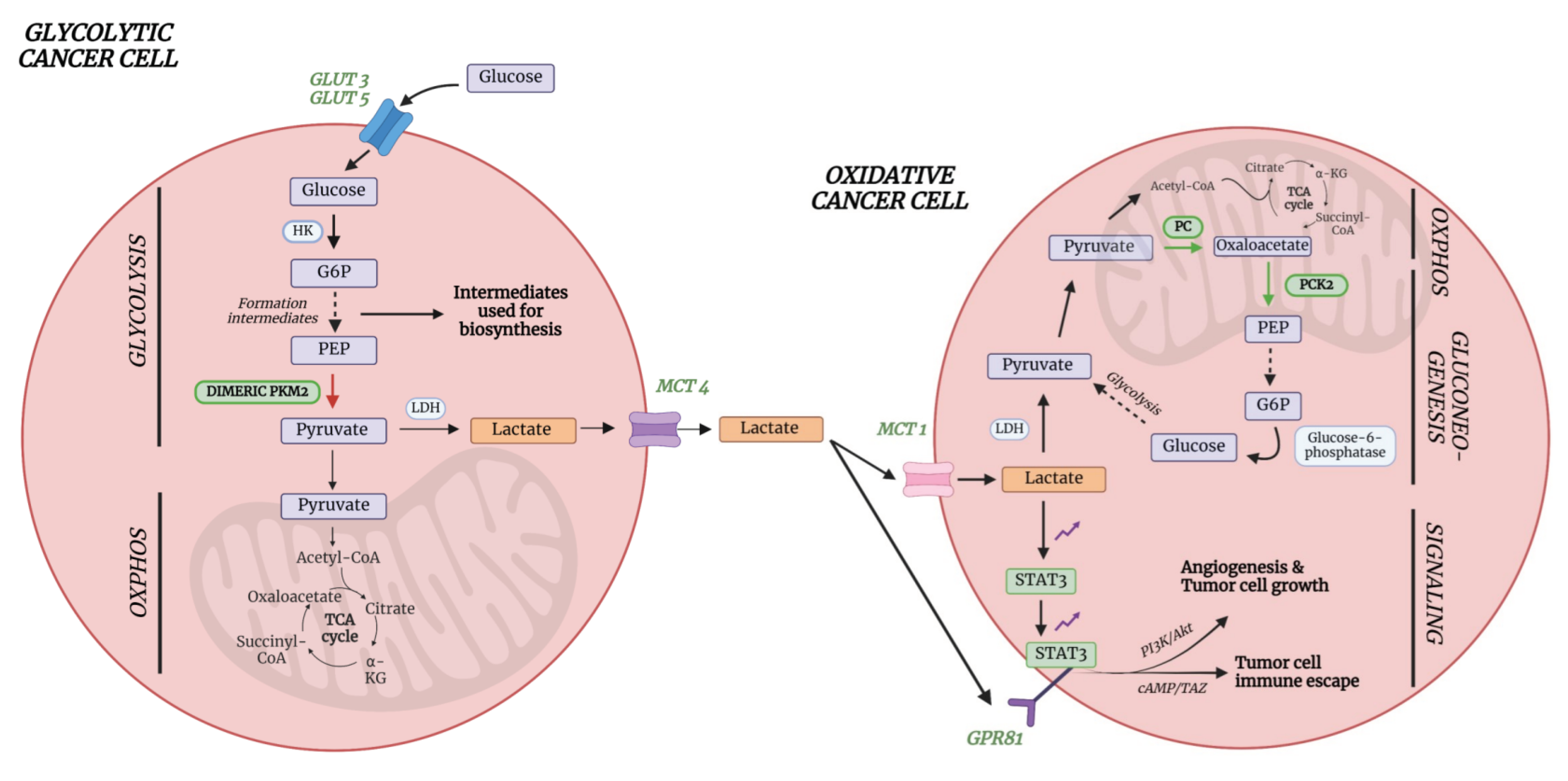
6. Metabolic Pathways Involved in (Lung) Cancer
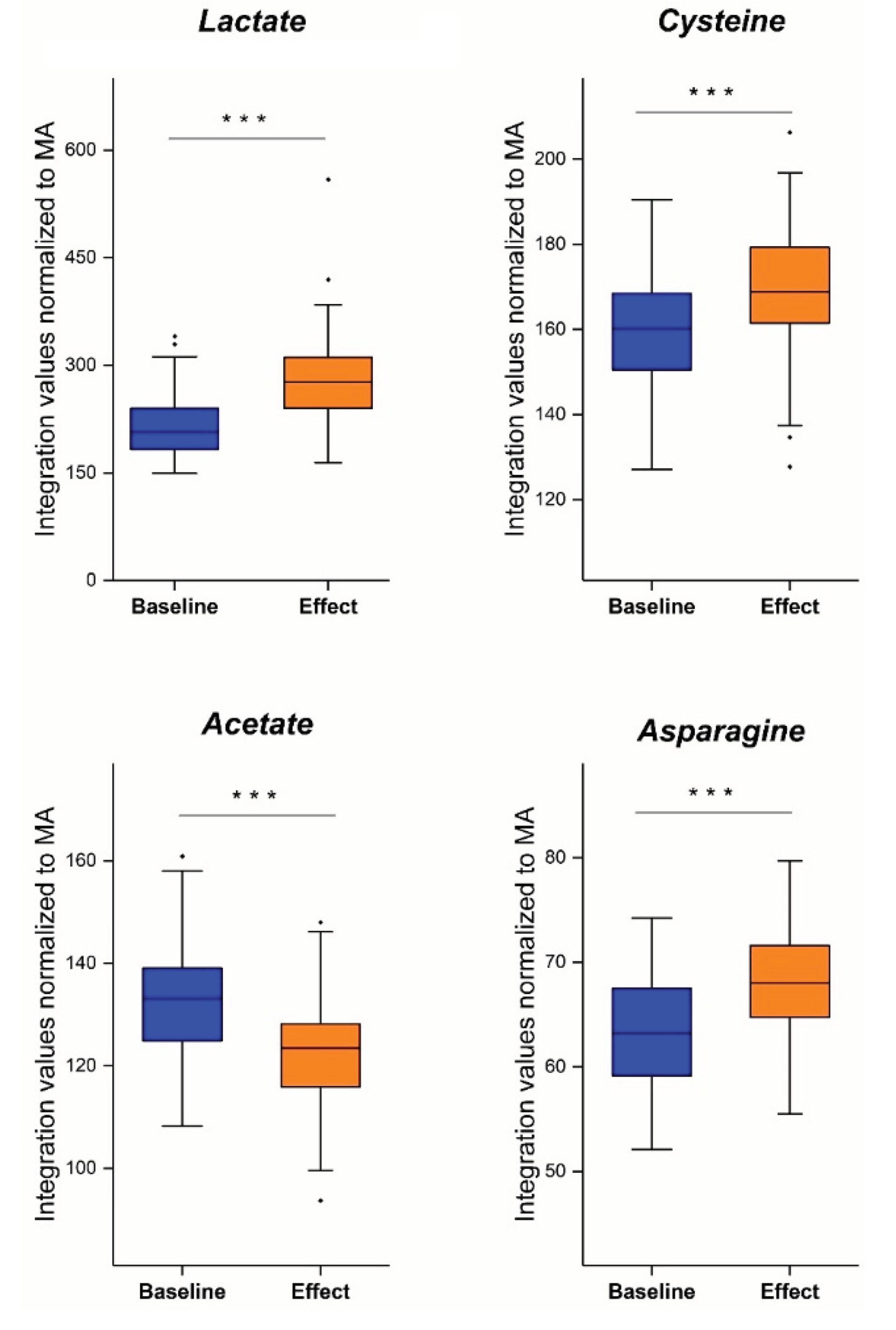
6.1. Lactate
6.2. Acetate and Amino Acids Cysteine and Asparagine
7. Conclusion and Future Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Hirsch, F.R.; Scagliotti, G.V.; Mulshine, J.L.; Kwon, R.; Curran, W.J.; Wu, Y.-L.; Paz-Ares, L. Lung cancer: current therapies and new targeted treatments. The Lancet 2017, 389, 299–311. [Google Scholar] [CrossRef] [PubMed]
- Wood De Fau - Kazerooni, E.A.; Kazerooni Ea Fau - Baum, S.L.; Baum Sl Fau - Eapen, G.A.; Eapen Ga Fau - Ettinger, D.S.; Ettinger Ds Fau - Hou, L.; Hou L Fau - Jackman, D.M.; Jackman Dm Fau - Klippenstein, D.; Klippenstein D Fau - Kumar, R.; Kumar R Fau - Lackner, R.P.; Lackner Rp Fau - Leard, L.E.; et al. Lung Cancer Screening, Version 3.2018, NCCN Clinical Practice Guidelines in Oncology. 2018. [CrossRef]
- Lancaster, H.; Heuvelmans, M.A.; Oudkerk, M. Low-dose computed tomography lung cancer screening: Clinical evidence and implementation research. 2022. [CrossRef]
- Aberle Dr Fau - Adams, A.M.; Adams Am Fau - Berg, C.D.; Berg Cd Fau - Black, W.C.; Black Wc Fau - Clapp, J.D.; Clapp Jd Fau - Fagerstrom, R.M.; Fagerstrom Rm Fau - Gareen, I.F.; Gareen If Fau - Gatsonis, C.; Gatsonis C Fau - Marcus, P.M.; Marcus Pm Fau - Sicks, J.D.; Sicks, J.D. Reduced lung-cancer mortality with low-dose computed tomographic screening. 2011. [CrossRef]
- Duffy, S.W.; Field, J. Mortality Reduction with Low-Dose CT Screening for Lung Cancer. 2020. [CrossRef]
- Oudkerk, M.; Liu, S.; Heuvelmans, M.A.; Walter, J.E.; Field, J.K. Lung cancer LDCT screening and mortality reduction — evidence, pitfalls and future perspectives. Nature Reviews Clinical Oncology 2021, 18, 135–151. [Google Scholar] [CrossRef] [PubMed]
- Berg, C.D.; Aberle, D.R.; Wood, D.E. Lung Cancer Screening: Promise and Pitfalls. American Society of Clinical Oncology Educational Book 2012, 450–457. [Google Scholar] [CrossRef] [PubMed]
- Farwell, M.D.; Pryma Da Fau - Mankoff, D.A.; Mankoff, D.A. PET/CT imaging in cancer: current applications and future directions. 2014. [CrossRef]
- Vanhove, K.; Giesen, P.; Owokotomo, O.E.; Mesotten, L.; Louis, E.; Shkedy, Z.; Thomeer, M.; Adriaensens, P. The plasma glutamate concentration as a complementary tool to differentiate benign PET-positive lung lesions from lung cancer. BMC Cancer 2018, 18, 868. [Google Scholar] [CrossRef] [PubMed]
- Rosenbaum, S.J.; Lind, T.; Antoch, G.; Bockisch, A. False-Positive FDG PET Uptake−the Role of PET/CT. European Radiology 2006, 16, 1054–1065. [Google Scholar] [CrossRef] [PubMed]
- Steven, B.Z.; Elizabeth, T.L.; Christopher, G.S.; David, H.A.; Paul, L.H.; Gregory, J.K.; Larry, G.K.; Leah, M.B. Preoperative PET and the Reduction of Unnecessary Surgery Among Newly Diagnosed Lung Cancer Patients in a Community Setting. Journal of Nuclear Medicine 2014, 55, 379. [Google Scholar] [CrossRef]
- Seijo, L.M.; Peled, N.; Ajona, D.; Boeri, M.; Field, J.K.; Sozzi, G.; Pio, R.; Zulueta, J.J.; Spira, A.; Massion, P.P.; et al. Biomarkers in Lung Cancer Screening: Achievements, Promises, and Challenges. 2019. [CrossRef]
- Clish, C.B. Metabolomics: an emerging but powerful tool for precision medicine. 2015. [CrossRef]
- Vanhove, K.; Derveaux, E.; Mesotten, L.; Thomeer, M.; Criel, M.; Mariën, H.; Adriaensens, P. Unraveling the Rewired Metabolism in Lung Cancer Using Quantitative NMR Metabolomics. International Journal of Molecular Sciences 2022, 23. [Google Scholar] [CrossRef] [PubMed]
- Noreldeen, H.A.A.; Liu, X.; Xu, G. Metabolomics of lung cancer: Analytical platforms and their applications. 2020. [CrossRef]
- Pascale, R.; Calvisi, D.F.; Simile, M.M.; Feo, C.; Feo, F. The Warburg Effect 97 Years after Its Discovery. LID - 10.3390/cancers12102819 [doi] LID - 2819. 2020. [CrossRef]
- Louis, E.; Adriaensens, P.; Guedens, W.; Bigirumurame, T.; Baeten, K.; Vanhove, K.; Vandeurzen, K.; Darquennes, K.; Vansteenkiste, J.; Dooms, C.; et al. Detection of Lung Cancer through Metabolic Changes Measured in Blood Plasma. 2016. [CrossRef]
- Louis, E.; Adriaensens, P.; Guedens, W.; Vanhove, K.; Vandeurzen, K.; Darquennes, K.; Vansteenkiste, J.; Dooms, C.; de Jonge, E.; Thomeer, M.; Mesotten, L. Metabolic phenotyping of human blood plasma: a powerful tool to discriminate between cancer types? 2016. [CrossRef]
- Derveaux, E.; Thomeer, M.; Mesotten, L.; Reekmans, G.; Adriaensens, P. Detection of Lung Cancer via Blood Plasma and (1)H-NMR Metabolomics: Validation by a Semi-Targeted and Quantitative Approach Using a Protein-Binding Competitor. LID - 10.3390/metabo11080537 [doi] LID - 537. 2021. [CrossRef]
- Derveaux, E.; Geubbelmans, M.; Criel, M.; Demedts, I.; Himpe, U.; Tournoy, K.; Vercauter, P.; Johansson, E.; Valkenborg, D.; Vanhove, K.; et al. NMR-Metabolomics Reveals a Metabolic Shift after Surgical Resection of Non-Small Cell Lung Cancer. LID - 10.3390/cancers15072127 [doi] LID - 2127. 2023. [CrossRef]
- Worley, B.; Powers, R. Multivariate Analysis in Metabolomics. 2013. [CrossRef]
- Puchades-Carrasco, L.; Jantus-Lewintre, E.; Pérez-Rambla, C.; García-García, F.; Lucas, R.; Calabuig, S.; Blasco, A.; Dopazo, J.; Camps, C.; Pineda-Lucena, A. Serum metabolomic profiling facilitates the non-invasive identification of metabolic biomarkers associated with the onset and progression of non-small cell lung cancer. 2016. [CrossRef]
- Rocha, C.M.; Carrola J Fau - Barros, A.S.; Barros As Fau - Gil, A.M.; Gil Am Fau - Goodfellow, B.J.; Goodfellow Bj Fau - Carreira, I.M.; Carreira Im Fau - Bernardo, J.; Bernardo J Fau - Gomes, A.; Gomes A Fau - Sousa, V.; Sousa V Fau - Carvalho, L.; Carvalho L Fau - Duarte, I.F.; Duarte, I.F. Metabolic signatures of lung cancer in biofluids: NMR-based metabonomics of blood plasma. 2011. [CrossRef]
- Xu, Y.; Goodacre, R. On Splitting Training and Validation Set: A Comparative Study of Cross-Validation, Bootstrap and Systematic Sampling for Estimating the Generalization Performance of Supervised Learning. Journal of Analysis and Testing 2018, 2, 249–262. [Google Scholar] [CrossRef] [PubMed]
- Guo, L.; Liu, J.; Lu, R. Subsampling bias and the best-discrepancy systematic cross validation. Science China Mathematics 2021, 64, 197–210. [Google Scholar] [CrossRef]
- Carrola, J.; Rocha Cm Fau - Barros, A.S.; Barros As Fau - Gil, A.M.; Gil Am Fau - Goodfellow, B.J.; Goodfellow Bj Fau - Carreira, I.M.; Carreira Im Fau - Bernardo, J.; Bernardo J Fau - Gomes, A.; Gomes A Fau - Sousa, V.; Sousa V Fau - Carvalho, L.; Carvalho L Fau - Duarte, I.F.; Duarte, I.F. Metabolic signatures of lung cancer in biofluids: NMR-based metabonomics of urine. 2011. [CrossRef]
- Louis, R.L., L.; Stinkens, K.; Mesotten, L.; de Jonge, E.; Thomeer, M.; Caenepeel, P.; Adriaensens, P. Metabolic Phenotyping of Blood Plasma by Proton Nuclear Magnetic Resonance to Discriminate between Colorectal Cancer, Breast Cancer and Lung cancer. Metabolomics 2016, 6. [Google Scholar]
- Pearce, J.T.; Athersuch Tj Fau - Ebbels, T.M.D.; Ebbels Tm Fau - Lindon, J.C.; Lindon Jc Fau - Nicholson, J.K.; Nicholson Jk Fau - Keun, H.C.; Keun, H.C. Robust algorithms for automated chemical shift calibration of 1D 1H NMR spectra of blood serum. 2008. [CrossRef]
- Zhang, X.; Zhu, X.; Wang, C.; Zhang, H.; Cai, Z. Non-targeted and targeted metabolomics approaches to diagnosing lung cancer and predicting patient prognosis. 2016. [CrossRef]
- Larkin, J.; Anthony, S.; Johanssen, V.; Yeo, T.; Sealey, M.; Yates, A.G.; Smith, C.; Claridge, T.; Nicholson, B.D.; Moreland, J.; et al. Metabolomic Biomarkers in Blood Samples Identify Cancers in a Mixed Population of Patients with Nonspecific Symptoms. 2022. [CrossRef]
- Gupta, D.; Lis, C.G. Pretreatment serum albumin as a predictor of cancer survival: a systematic review of the epidemiological literature. 2010. [CrossRef]
- Kowalczyk, T.; Kisluk, J.; Pietrowska, K.; Godzien, J.; Kozlowski, M.; Reszeć, J.; Sierko, E.; Naumnik, W.; Mróz, R.; Moniuszko, M.; et al. The Ability of Metabolomics to Discriminate Non-Small-Cell Lung Cancer Subtypes Depends on the Stage of the Disease and the Type of Material Studied. LID - 10.3390/cancers13133314 [doi] LID - 3314. 2021. [CrossRef]
- Ramteke, P.; Deb, A.; Shepal, V.; Bhat, M.K. Hyperglycemia Associated Metabolic and Molecular Alterations in Cancer Risk, Progression, Treatment, and Mortality. Cancers 2019, 11. [Google Scholar] [CrossRef] [PubMed]
- Kannampuzha, S.; Mukherjee, A.; Wanjari, U.; Gopalakrishnan, A.; Murali, R.; Namachivayam, A.; Renu, K.; Dey, A.; Vellingiri, B.; Madhyastha, H.; Ganesan, R. A Systematic Role of Metabolomics, Metabolic Pathways, and Chemical Metabolism in Lung Cancer. LID - 10.3390/vaccines11020381 [doi] LID - 381. 2023. [CrossRef]
- Vanhove, K.; Graulus, G.J.; Mesotten, L.; Thomeer, M.; Derveaux, E.; Noben, J.P.; Guedens, W.; Adriaensens, P. The Metabolic Landscape of Lung Cancer: New Insights in a Disturbed Glucose Metabolism. 2019. [CrossRef]
- Leithner, K.; Hrzenjak, A.; Trötzmüller, M.; Moustafa, T.; Köfeler, H.C.; Wohlkoenig, C.; Stacher, E.; Lindenmann, J.; Harris, A.L.; Olschewski, A.; Olschewski, H. PCK2 activation mediates an adaptive response to glucose depletion in lung cancer. 2015. [CrossRef]
- Mendes, C.; Serpa, J. Revisiting lactate dynamics in cancer—a metabolic expertise or an alternative attempt to survive? Journal of Molecular Medicine 2020, 98, 1397–1414. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Liu, H.; Ni, Y.; Shen, P.; Han, X. Lactate shuttle: from substance exchange to regulatory mechanism. Human Cell 2022, 35, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Smolle, E.; Leko, P.; Stacher-Priehse, E.; Brcic, L.; El-Heliebi, A.; Hofmann, L.; Quehenberger, F.; Hrzenjak, A.; Popper, H.H.; Olschewski, H.; Leithner, K. Distribution and prognostic significance of gluconeogenesis and glycolysis in lung cancer. 2020. [CrossRef]
- Kiesel, V.A.; Sheeley, M.P.; Coleman, M.F.; Cotul, E.K.; Donkin, S.S.; Hursting, S.D.; Wendt, M.K.; Teegarden, D. Pyruvate carboxylase and cancer progression. Cancer & Metabolism 2021, 9, 20. [Google Scholar] [CrossRef]
- Li, Z.; Cui, J. Targeting the lactic acid metabolic pathway for antitumor therapy. 2023. [CrossRef]
- Xie, Q.; Zhu, Z.; He, Y.; Zhang, Z.; Zhang, Y.; Wang, Y.; Luo, J.; Peng, T.; Cheng, F.; Gao, J.; et al. A lactate-induced Snail/STAT3 pathway drives GPR81 expression in lung cancer cells. 2019. [CrossRef]
- Feng, J.; Yang, H.; Zhang, Y.; Wei, H.; Zhu, Z.; Zhu, B.; Yang, M.; Cao, W.; Wang, L.; Wu, Z. Tumor cell-derived lactate induces TAZ-dependent upregulation of PD-L1 through GPR81 in human lung cancer cells. 2017. [CrossRef]
- Schug, Z.T.; Vande Voorde, J.; Gottlieb, E. The metabolic fate of acetate in cancer. Nature Reviews Cancer 2016, 16, 708–717. [Google Scholar] [CrossRef] [PubMed]
- Comerford, S.A.; Huang, Z.; Du, X.; Wang, Y.; Cai, L.; Witkiewicz, A.K.; Walters, H.; Tantawy, M.N.; Fu, A.; Manning, H.C.; et al. Acetate dependence of tumors. 2014. [CrossRef]
- Migita, T.; Narita T Fau - Nomura, K.; Nomura K Fau - Miyagi, E.; Miyagi E Fau - Inazuka, F.; Inazuka F Fau - Matsuura, M.; Matsuura M Fau - Ushijima, M.; Ushijima M Fau - Mashima, T.; Mashima T Fau - Seimiya, H.; Seimiya H Fau - Satoh, Y.; Satoh Y Fau - Okumura, S.; et al. ATP citrate lyase: activation and therapeutic implications in non-small cell lung cancer. 2008. [CrossRef]
- Bose, S.; Ramesh, V.; Locasale, J.W. Acetate Metabolism in Physiology, Cancer, and Beyond. 2019. [CrossRef]
- Horibata, Y.; Ando, H.; Itoh, M.; Sugimoto, H. Enzymatic and transcriptional regulation of the cytoplasmic acetyl-CoA hydrolase ACOT12. 2013. [CrossRef]
- Yoon, S.; Combs, J.; Falzone, A.; Prieto-Farigua, N.; Caldwell, S.; Ackerman, H.; Flores, E.; DeNicola, G. Comprehensive Metabolic Tracing Reveals the Origin and Catabolism of Cysteine in Mammalian Tissues and Tumors. 2023. [CrossRef]
- Vanhove, K.; Derveaux, E.; Graulus, G.; Mesotten, L.; Thomeer, M.; Noben, J.; Guedens, W.; Adriaensens, P. Glutamine Addiction and Therapeutic Strategies in Lung Cancer. LID - 10.3390/ijms20020252 [doi] LID - 252. 2019. [CrossRef]
- García-Gaytán, A.C.; Hernández-Abrego, A.; Díaz-Muñoz, M.; Méndez, I. Glutamatergic system components as potential biomarkers and therapeutic targets in cancer in non-neural organs. 2022. [CrossRef]
- Bonifácio, V.D.B.; Pereira, S.A.; Serpa, J.; Vicente, J.B. Cysteine metabolic circuitries: druggable targets in cancer. 2021. [CrossRef]
- Zhang, J.; Fan, J.; Venneti, S.; Cross, J.R.; Takagi, T.; Bhinder, B.; Djaballah, H.; Kanai, M.; Cheng, E.H.; Judkins, A.R.; et al. Asparagine plays a critical role in regulating cellular adaptation to glutamine depletion. 2014. [CrossRef]
- Zhang, J.; Fan, J.; Venneti, S.; Cross, Justin R. ; Takagi, T.; Bhinder, B.; Djaballah, H.; Kanai, M.; Cheng, Emily H.; Judkins, Alexander R.; et al. Asparagine Plays a Critical Role in Regulating Cellular Adaptation to Glutamine Depletion. Molecular Cell 2014, 56, 205–218. [Google Scholar] [CrossRef]
- Pavlova, N.N.; Hui, S.; Ghergurovich, J.M.; Fan, J.; Intlekofer, A.M.; White, R.M.; Rabinowitz, J.D.; Thompson, C.B.; Zhang, J. As Extracellular Glutamine Levels Decline, Asparagine Becomes an Essential Amino Acid. 2018. [CrossRef]
- Jiang, J.; Pavlova, N.N.; Zhang, J. Asparagine, a critical limiting metabolite during glutamine starvation. 2018. [CrossRef]
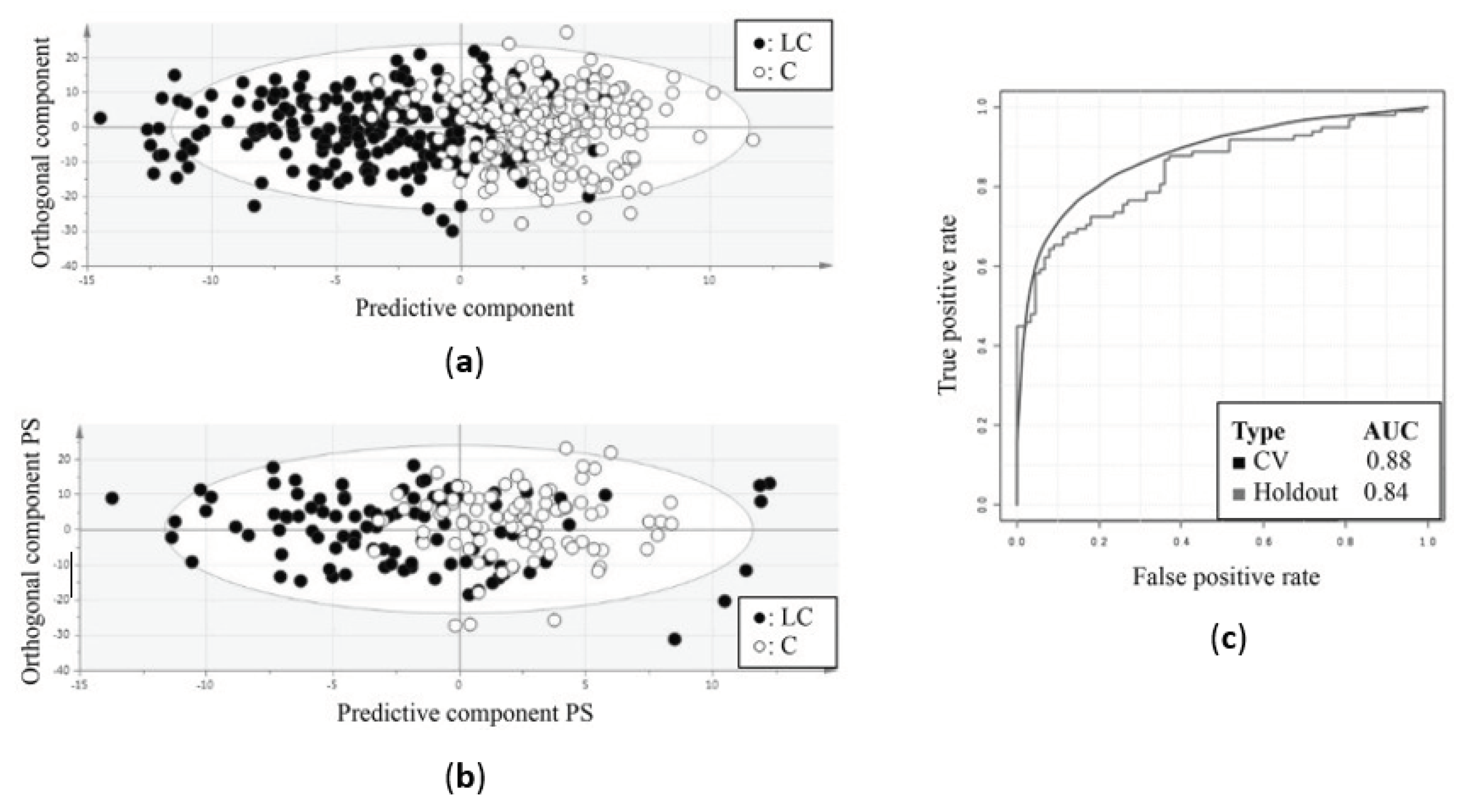
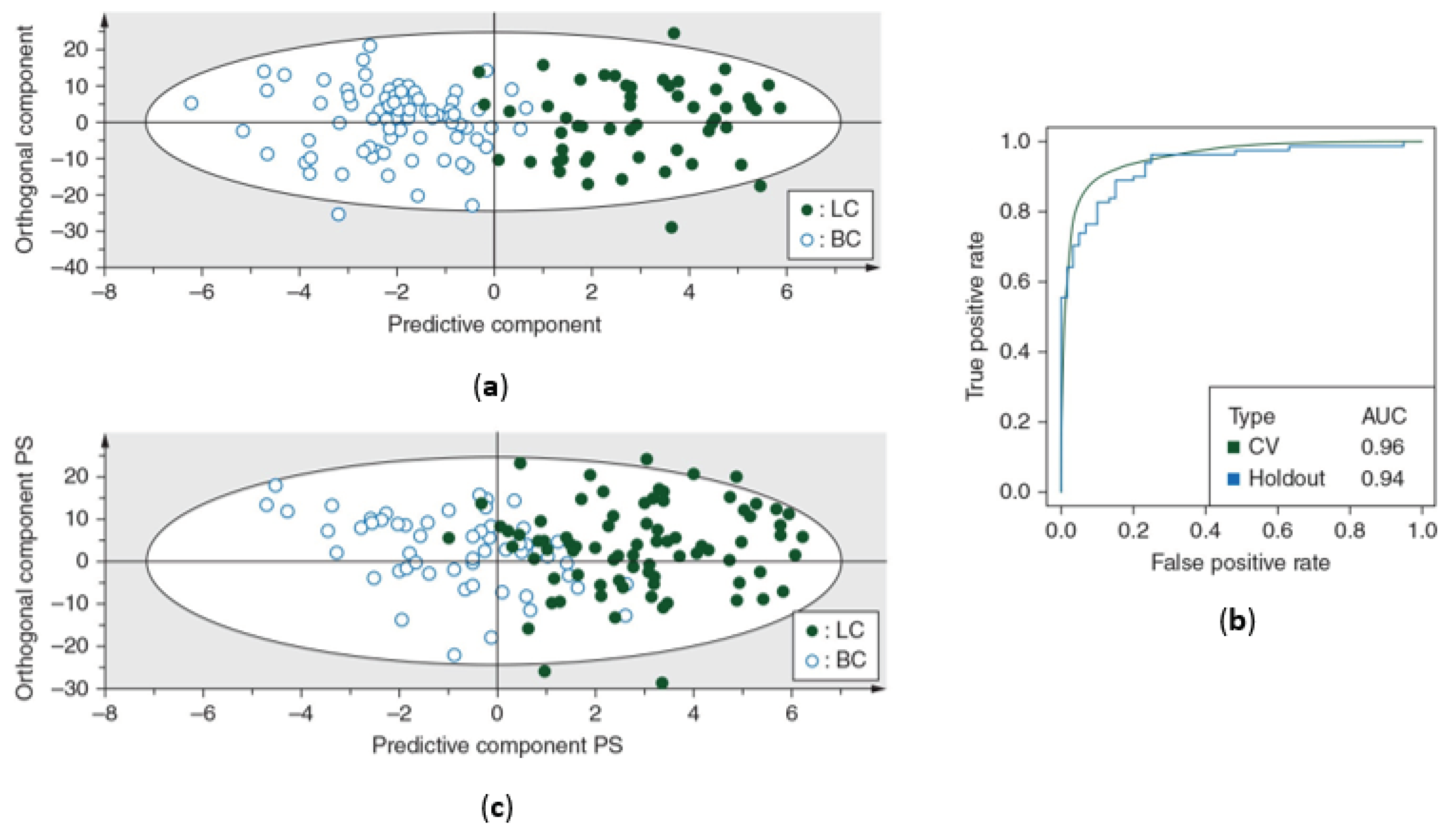
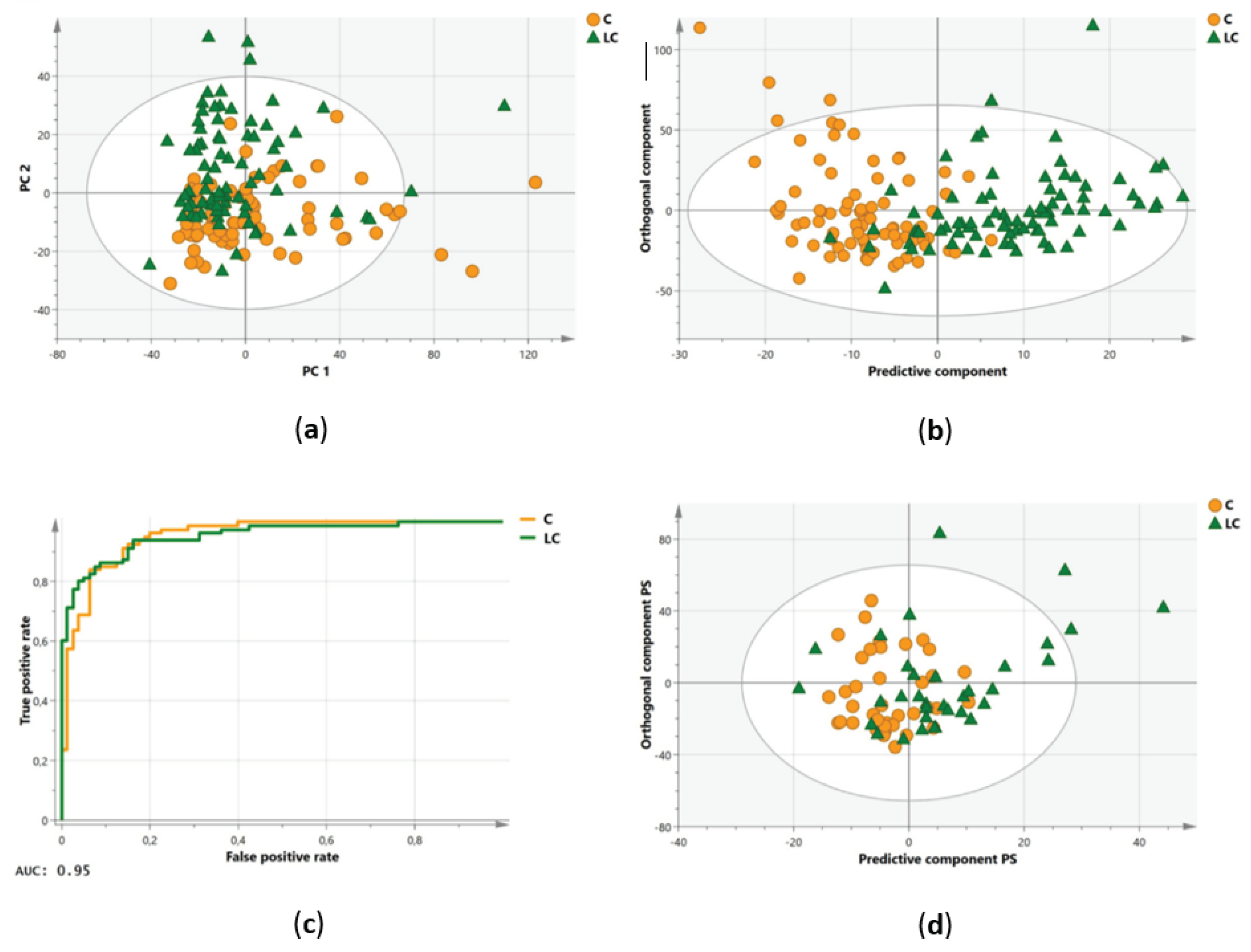
| Louis et al., 2016 [17] | ||||
| Training cohort | Validation cohort | |||
| LC | C | LC | C | |
| Number of subjects, n | 233 | 226 | 98 | 89 |
| Sensitivity (%) | 78 | 71 | ||
| Specificity (%) | 92 | 81 | ||
| R2X(Cum) | 0.864 | - | ||
| R2Y(Cum) | 0.477 | - | ||
| Q2(Cum) | 0.391 | - | ||
| AUC | 0.88 | 0.84 | ||
| Louis et al., 2016 [18] | ||||
| Training cohort | Validation cohort | |||
| LC | BC | LC | BC | |
| Number of subjects, n | 54 | 80 | 81 | 60 |
| Sensitivity (%) | 93 | 89 | ||
| Specificity (%) | 99 | 82 | ||
| R2X(Cum) | 0.82 | - | ||
| R2Y(Cum) | 0.73 | - | ||
| Q2(Cum) | 0.63 | - | ||
| AUC | 0.96 | 0.94 | ||
| Derveaux et al., 2021 [19] | ||||
| Training cohort | Validation cohort | |||
| LC | C | LC | C | |
| Number of subjects, n | 80 | 80 | 34 | 38 |
| Sensitivity (%) | 85 | 74 | ||
| Specificity (%) | 93 | 74 | ||
| R2X(Cum) | 0.861 | - | ||
| R2Y(Cum) | 0.581 | - | ||
| Q2(Cum) | 0.364 | - | ||
| AUC | 0.95 | - | ||
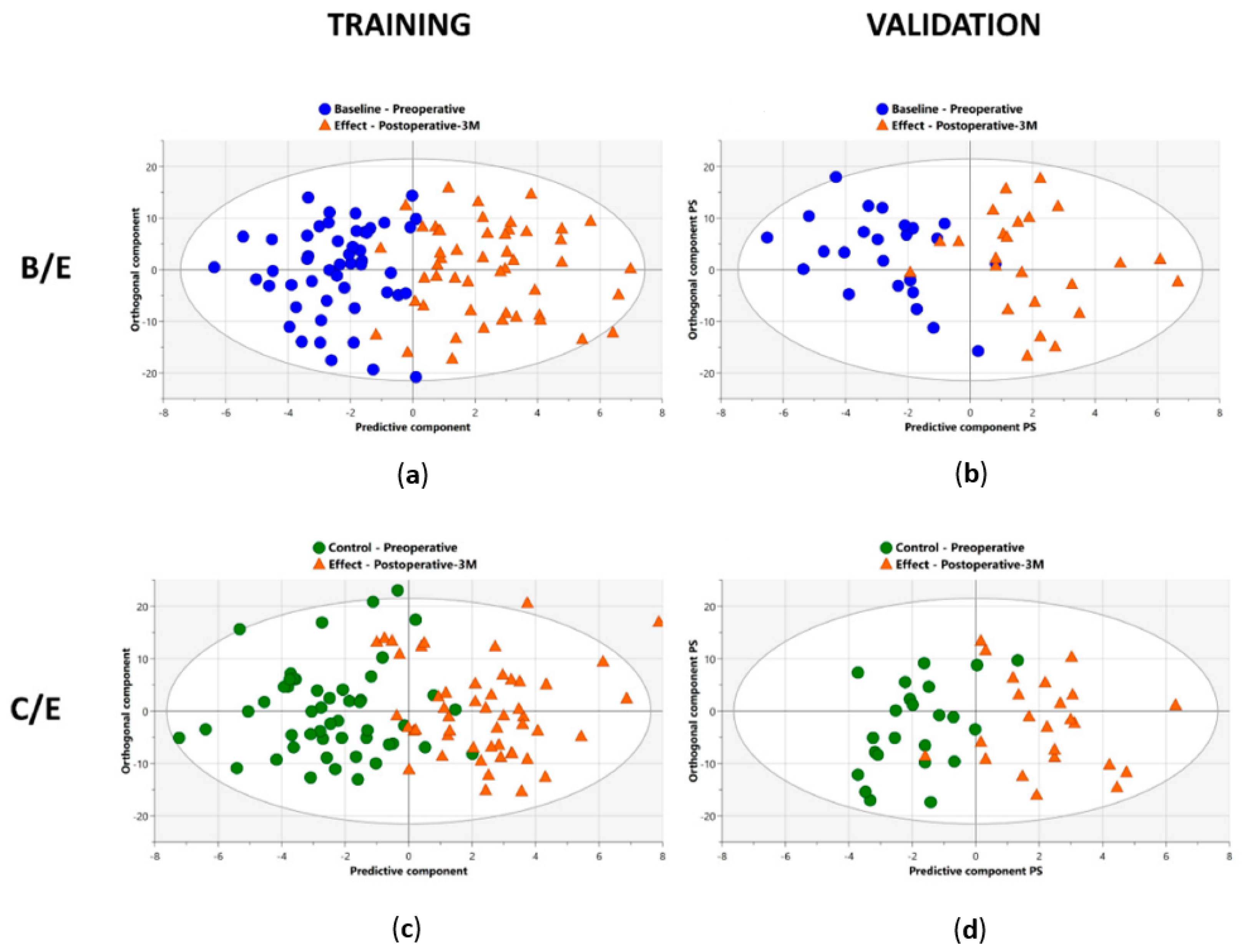
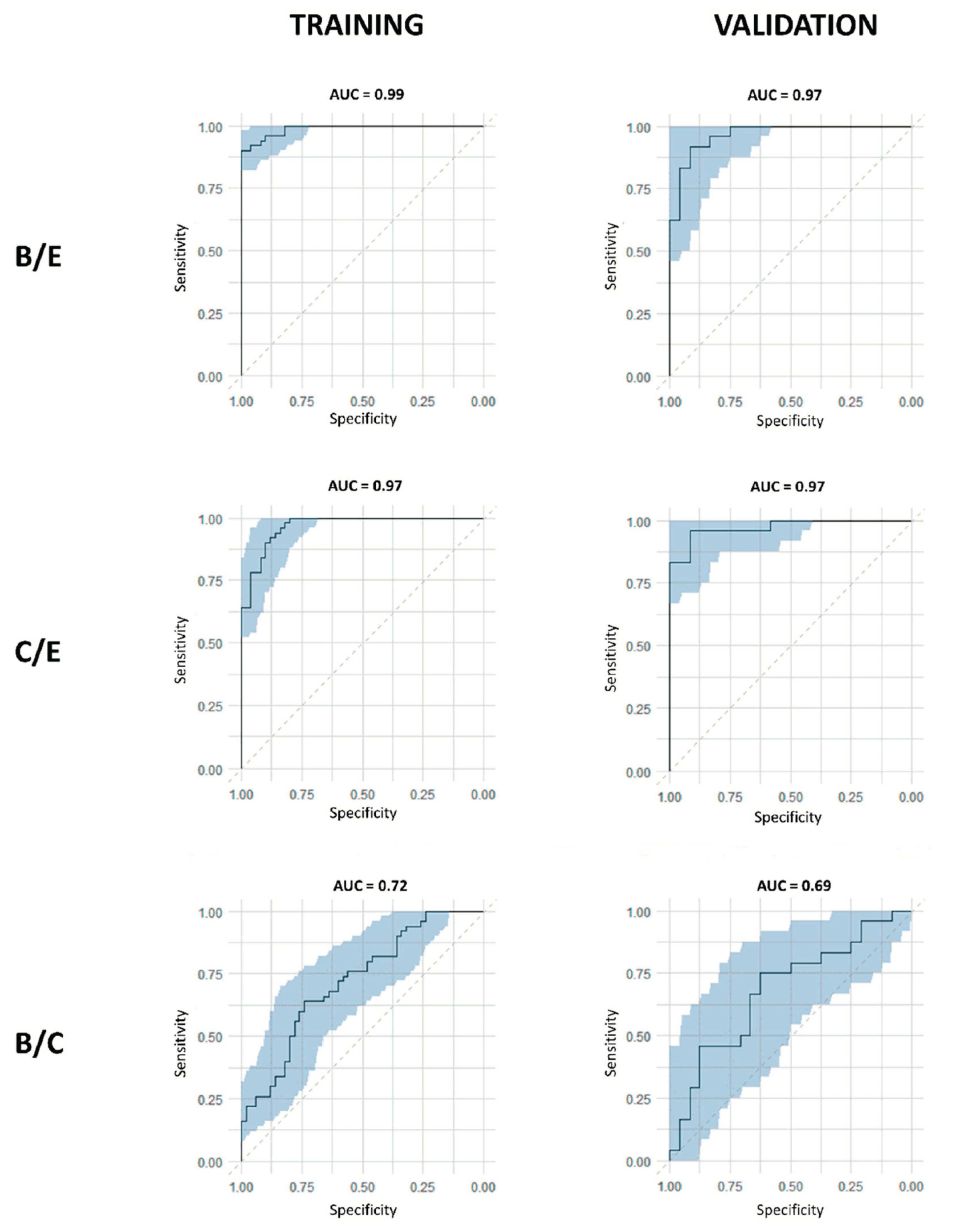
| Derveaux et al., 2023 [20] | ||||||
| B/E | C/E | B/E | ||||
| OPLS-DA | OPLS-EP | OPLS-DA | OPLS-EP | OPLS-DA | ||
| TRAINING COHORT | ||||||
| Number of subjects, n | 50 | 50 | 50 | 50 | 50 | |
| Sensitivity (%) | 92 | - | 88 | - | 74 | |
| Specificity (%) | 96 | - | 90 | - | 62 | |
| R2X(Cum) | 0.55 | 0.59 | 0.53 | 0.57 | 0.31 | |
| R2Y(Cum) | 0.67 | 0.89 | 0.61 | 0.83 | 0.15 | |
| Q2(Cum) | 0.42 | 0.76 | 0.36 | 0.60 | 0.08 | |
| AUC | 0.99 | - | 0.97 | - | 0.72 | |
| VALIDATION COHORT | ||||||
| Number of subjects, n | 24 | 24 | 23 | 23 | 23 | |
| Sensitivity (%) | 88 | - | 96 | - | 74 | |
| Specificity (%) | 92 | - | 91 | - | 43 | |
| AUC | 0.97 | - | 0.97 | - | 0.69 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




