Submitted:
15 March 2024
Posted:
18 March 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Generation of Constructs
2.2. qPCR and Sequence of Primers
2.3. Inhibition of Protein Expression by Small-Interfering RNAs (siRNAs)
2.4. Cell Culture and Transfection
2.5. In-House ELISA Measurement of Human PCSK9 Levels in Media
2.6. Western Blotting
2.7. Immunofluorescence Assay (IF)
2.8. PCSK9 – LDLR (EGF-AB peptide) Binding Assay
2.9. Modeling of PCSK9/HFE Complex
2.10. Modeling of the Interaction between PCSK9’s N-Terminus with HLA-C and Other HLA Memnbers
3. Results
3.1. In search for “Protein X”
3.2. HFE and HLA-C as New Targets and Regulators of Extracellular PCSK9
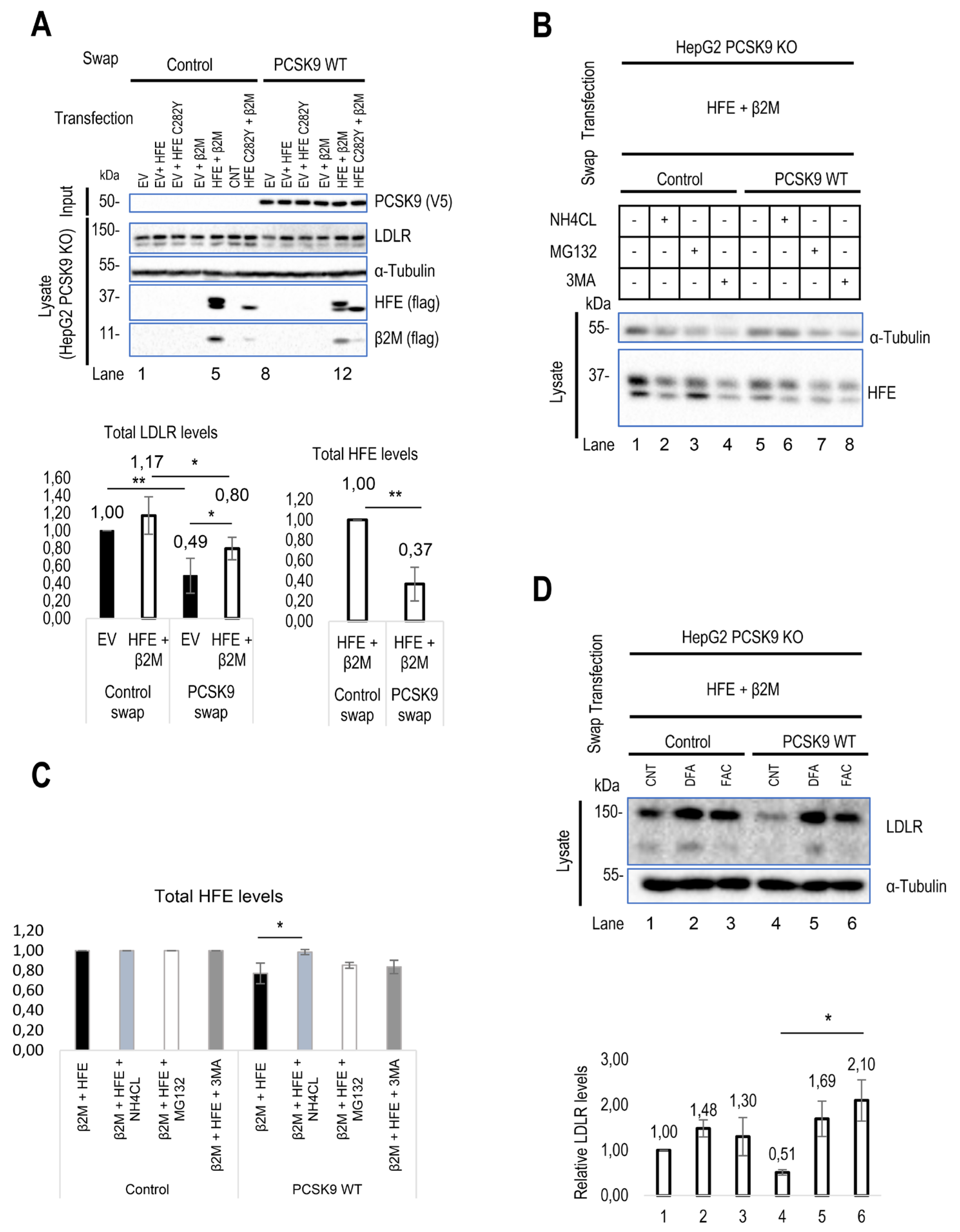
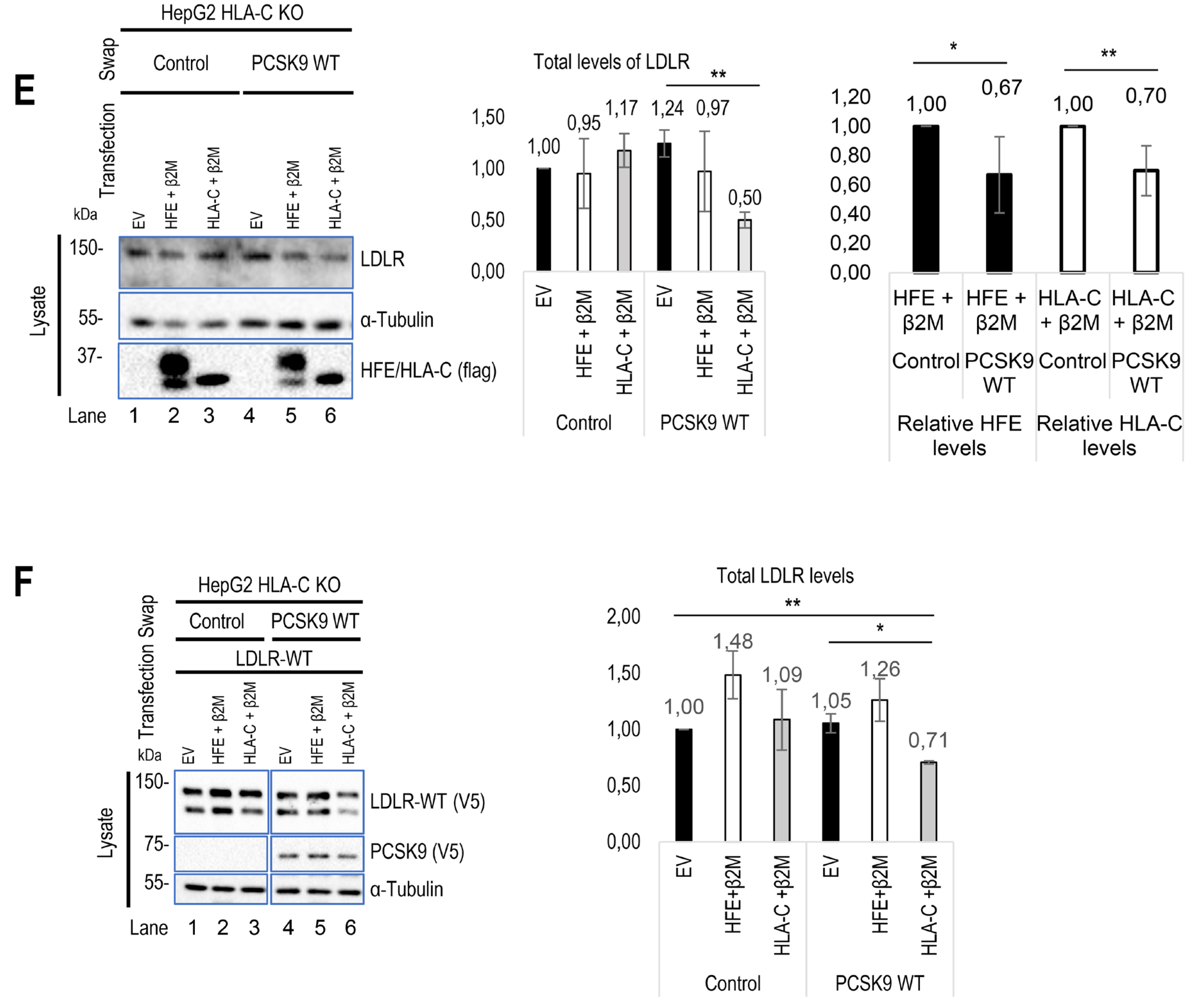
3.3. Interaction of PCSK9 with HFE and HLA-C
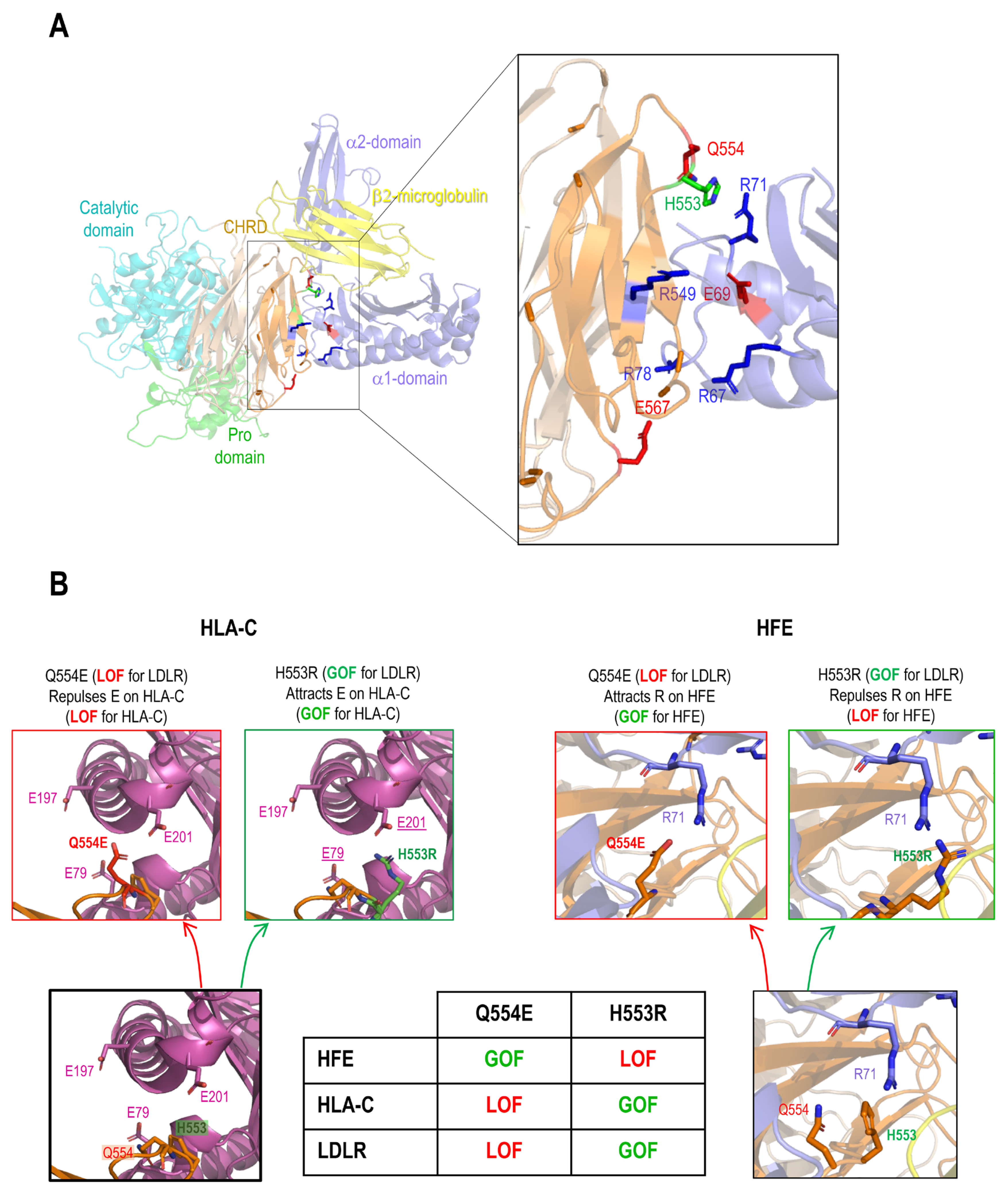
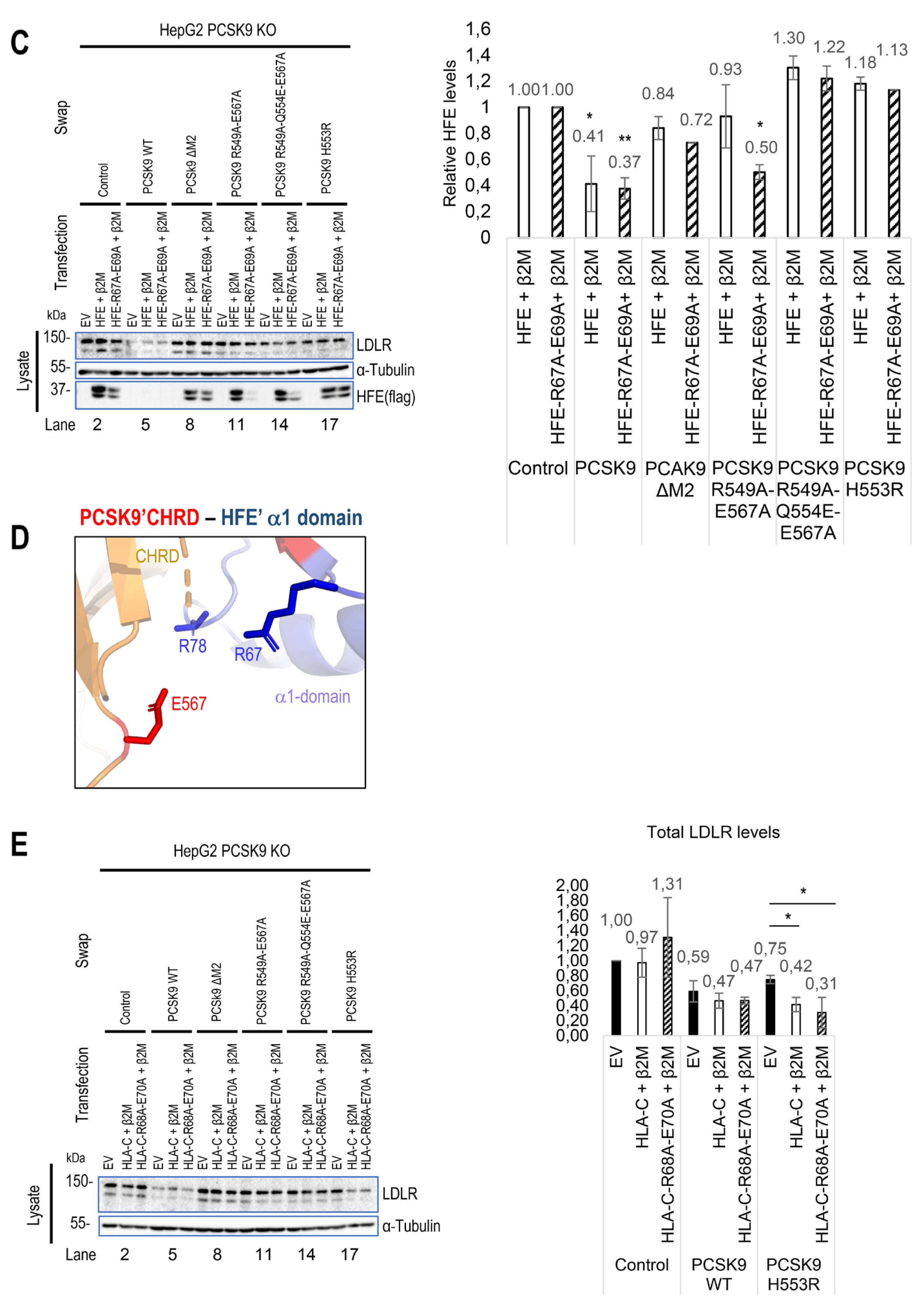
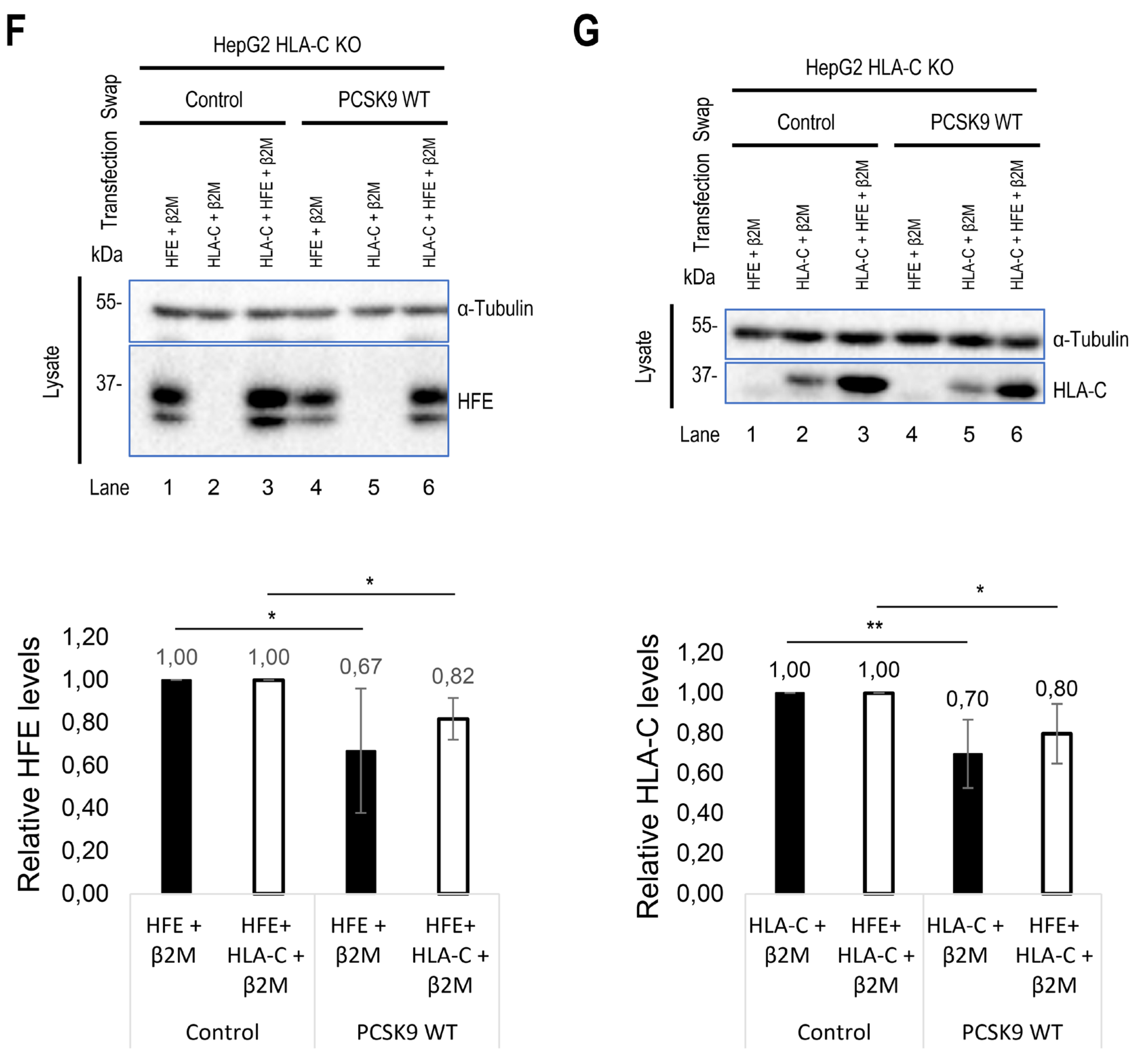
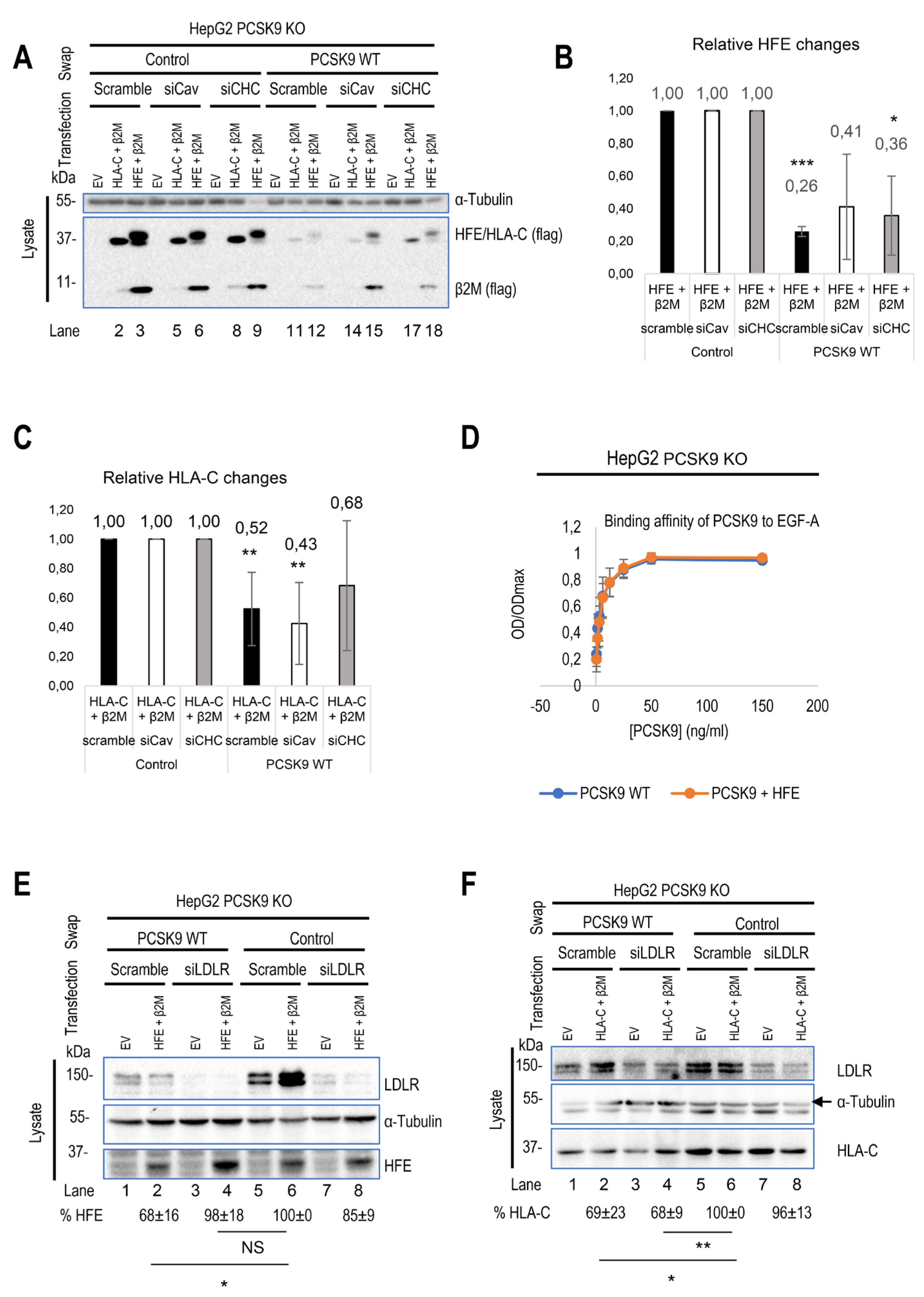
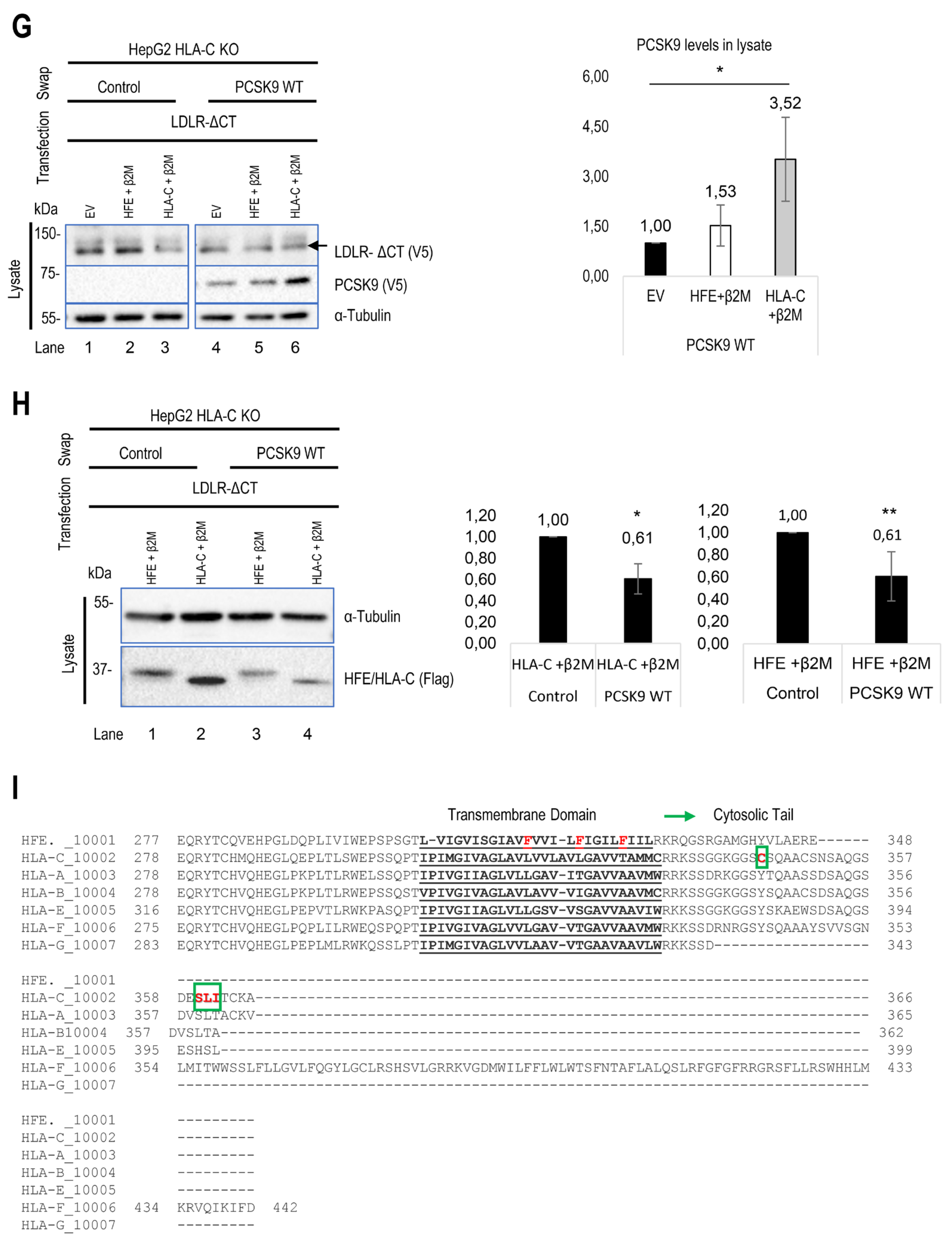
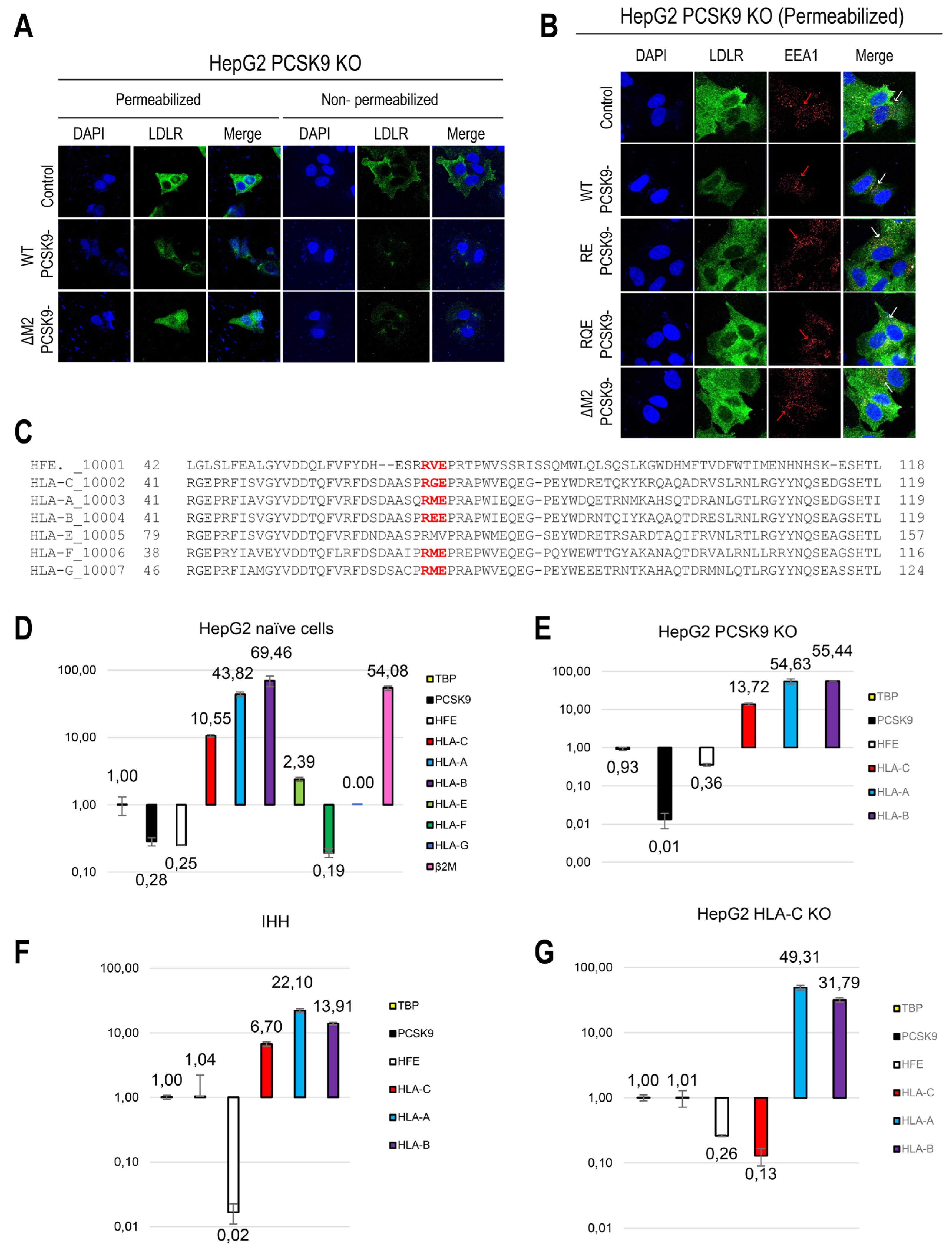
3.4. HFE and HLA-C Trafficking
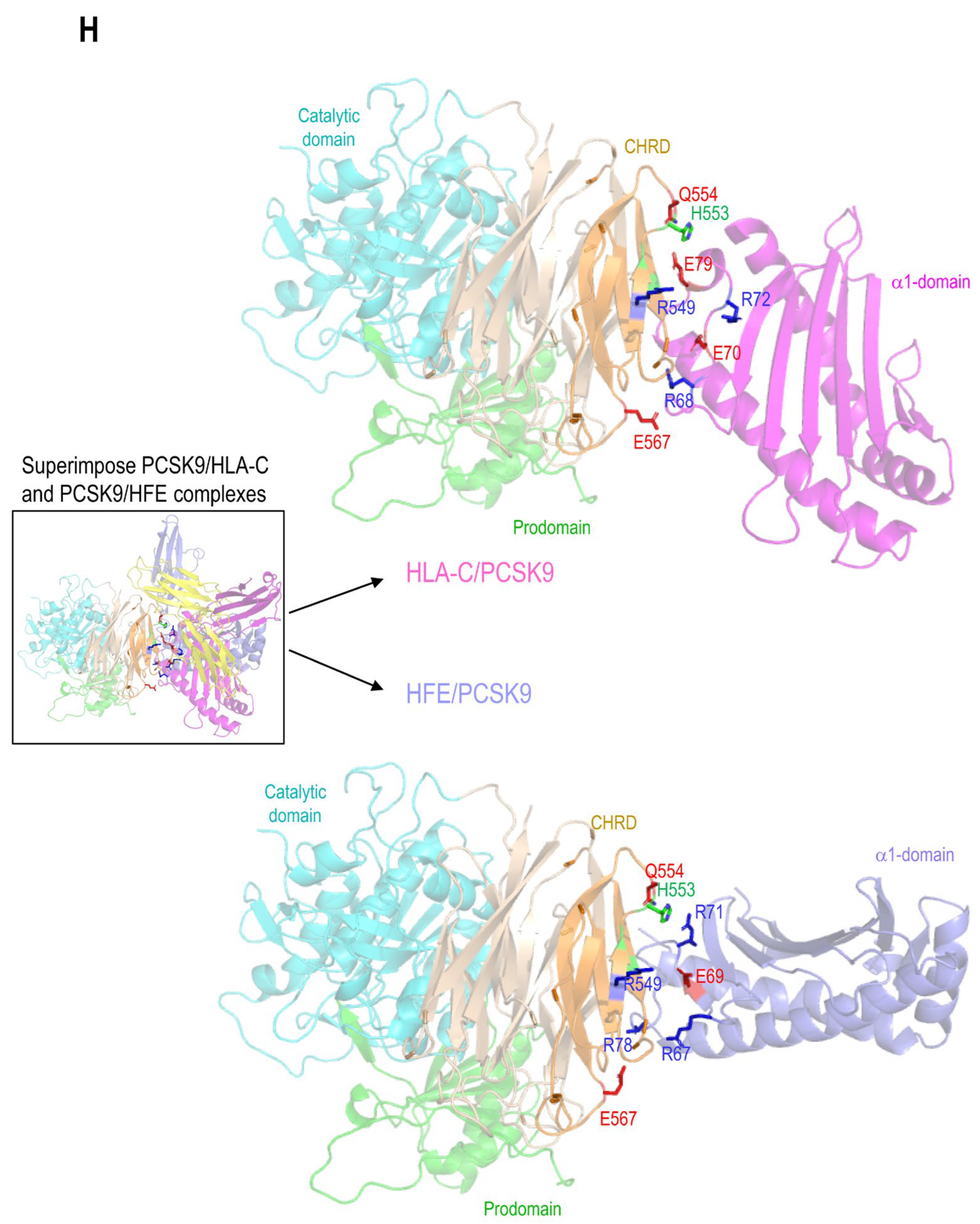
3.5. Modeling of the Interaction between PCSK9’s N-Terminus and HLA-C
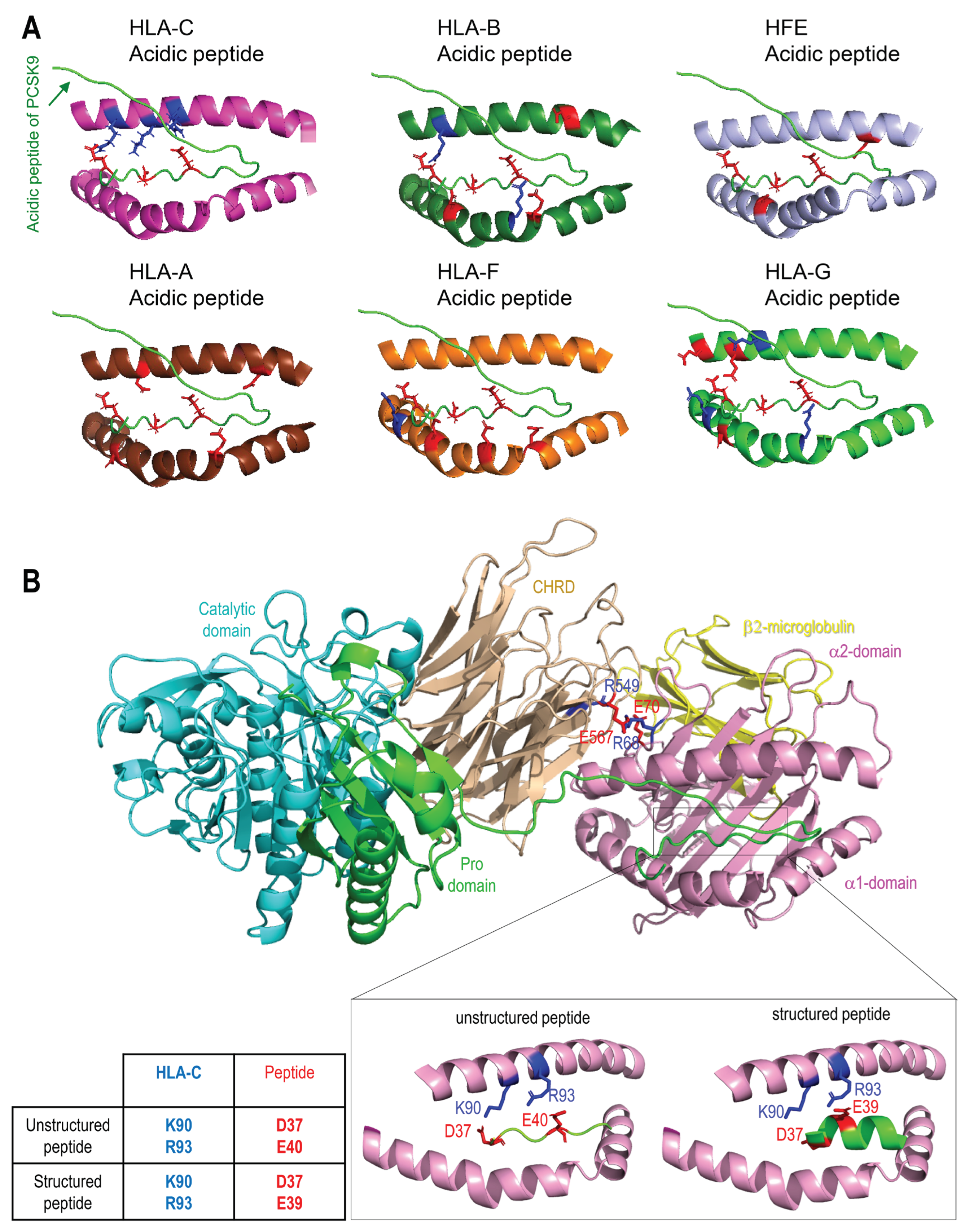
4. Discussion
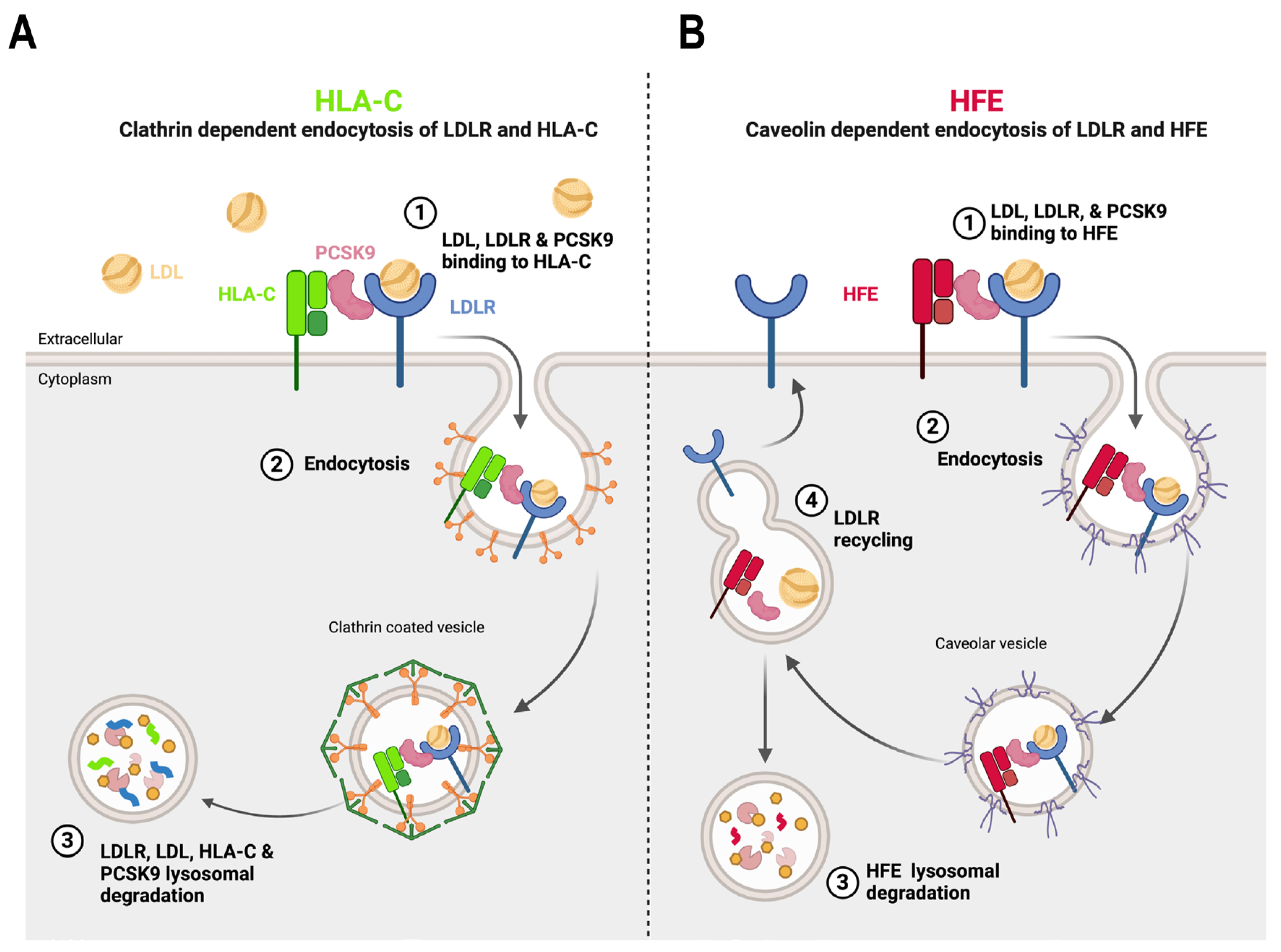
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Seidah, N.G.; Benjannet, S.; Wickham, L.; Marcinkiewicz, J.; Jasmin, S.B.; Stifani, S.; Basak, A.; Prat, A.; Chretien, M. The secretory proprotein convertase neural apoptosis-regulated convertase 1 (NARC-1): liver regeneration and neuronal differentiation. Proceedings of the National Academy of Sciences of the United States of America 2003, 100, 928-933. [CrossRef]
- Seidah, N.G. The PCSK9 discovery, an inactive protease with varied functions in hypercholesterolemia, viral infections, and cancer. J Lipid Res 2021, 62, 100130. [CrossRef]
- Seidah, N.G.; Prat, A. The biology and therapeutic targeting of the proprotein convertases. Nat Rev Drug Discov 2012, 11, 367-383. [CrossRef]
- Abifadel, M.; Varret, M.; Rabes, J.P.; Allard, D.; Ouguerram, K.; Devillers, M.; Cruaud, C.; Benjannet, S.; Wickham, L.; Erlich, D.; et al. Mutations in PCSK9 cause autosomal dominant hypercholesterolemia. Nature Genetics 2003, 34, 154-156. [CrossRef]
- Maxwell, K.N.; Breslow, J.L. Adenoviral-mediated expression of Pcsk9 in mice results in a low-density lipoprotein receptor knockout phenotype. Proceedings of the National Academy of Sciences of the United States of America 2004, 101, 7100-7105.
- Benjannet, S.; Rhainds, D.; Essalmani, R.; Mayne, J.; Wickham, L.; Jin, W.; Asselin, M.C.; Hamelin, J.; Varret, M.; Allard, D.; et al. NARC-1/PCSK9 and its natural mutants: zymogen cleavage and effects on the low density lipoprotein (LDL) receptor and LDL cholesterol. Journal of Biological Chemistry 2004, 279, 48865-48875. [CrossRef]
- McNutt, M.C.; Lagace, T.A.; Horton, J.D. Catalytic activity is not required for secreted PCSK9 to reduce low density lipoprotein receptors in HepG2 cells. Journal of Biological Chemistry 2007, 282, 20799-20803.
- Poirier, S.; Mayer, G.; Benjannet, S.; Bergeron, E.; Marcinkiewicz, J.; Nassoury, N.; Mayer, H.; Nimpf, J.; Prat, A.; Seidah, N.G. The proprotein convertase PCSK9 induces the degradation of low density lipoprotein receptor (LDLR) and its closest family members VLDLR and ApoER2. Journal of Biological Chemistry 2008, 283, 2363-2372.
- Seidah, N.G.; Abifadel, M.; Prost, S.; Boileau, C.; Prat, A. The Proprotein Convertases in Hypercholesterolemia and Cardiovascular Diseases: Emphasis on Proprotein Convertase Subtilisin/Kexin 9. Pharmacol Rev 2017, 69, 33-52. [CrossRef]
- Seidah, N.G.; Prat, A. The Multifaceted Biology of PCSK9. Endocr Rev 2022, 43, 558-582. [CrossRef]
- Lambert, G.; Sjouke, B.; Choque, B.; Kastelein, J.J.; Hovingh, G.K. The PCSK9 decade. J Lipid Res 2012, 53, 2515-2524. [CrossRef]
- Seidah, N.G.; Prat, A.; Pirillo, A.; Catapano, A.L.; Norata, G.D. Novel strategies to target proprotein convertase subtilisin kexin 9: beyond monoclonal antibodies. Cardiovasc Res 2019, 115, 510-518. [CrossRef]
- Oostveen, R.F.; Khera, A.V.; Kathiresan, S.; Stroes, E.S.G.; Fitzgerald, K.; Harms, M.J.; Oakes, B.L.; Kastelein, J.J.P. New Approaches for Targeting PCSK9: Small-Interfering Ribonucleic Acid and Genome Editing. Arterioscler Thromb Vasc Biol 2023, 43, 1081-1092. [CrossRef]
- Lee, R.G.; Mazzola, A.M.; Braun, M.C.; Platt, C.; Vafai, S.B.; Kathiresan, S.; Rohde, E.; Bellinger, A.M.; Khera, A.V. Efficacy and Safety of an Investigational Single-Course CRISPR Base-Editing Therapy Targeting PCSK9 in Nonhuman Primate and Mouse Models. Circulation 2023, 147, 242-253. [CrossRef]
- Davis, J.R.; Wang, X.; Witte, I.P.; Huang, T.P.; Levy, J.M.; Raguram, A.; Banskota, S.; Seidah, N.G.; Musunuru, K.; Liu, D.R. Efficient in vivo base editing via single adeno-associated viruses with size-optimized genomes encoding compact adenine base editors. Nat Biomed Eng 2022, 6, 1272-1283. [CrossRef]
- Cunningham, D.; Danley, D.E.; Geoghegan, K.F.; Griffor, M.C.; Hawkins, J.L.; Subashi, T.A.; Varghese, A.H.; Ammirati, M.J.; Culp, J.S.; Hoth, L.R.; et al. Structural and biophysical studies of PCSK9 and its mutants linked to familial hypercholesterolemia. Nat Struct Mol Biol. 2007, 14, 413-419. [CrossRef]
- Piper, D.E.; Jackson, S.; Liu, Q.; Romanow, W.G.; Shetterly, S.; Thibault, S.T.; Shan, B.; Walker, N.P. The Crystal Structure of PCSK9: A Regulator of Plasma LDL-Cholesterol. Structure. 2007, 15, 545-552.
- Holla, O.L.; Cameron, J.; Berge, K.E.; Ranheim, T.; Leren, T.P. Degradation of the LDL receptors by PCSK9 is not mediated by a secreted protein acted upon by PCSK9 extracellularly. BMC. Cell Biol. 2007, 8, 9-20.
- Zhang, D.W.; Garuti, R.; Tang, W.J.; Cohen, J.C.; Hobbs, H.H. Structural requirements for PCSK9-mediated degradation of the low-density lipoprotein receptor. Proceedings of the National Academy of Sciences of the United States of America 2008, 105, 13045-13050.
- Saavedra, Y.G.; Day, R.; Seidah, N.G. The M2 module of the Cys-His-rich domain (CHRD) of PCSK9 is needed for the extracellular low density lipoprotein receptor (LDLR) degradation pathway. J Biol. Chem. 2012, 287, 43492-43501.
- Poirier, S.; Mayer, G.; Poupon, V.; McPherson, P.S.; Desjardins, R.; Ly, K.; Asselin, M.C.; Day, R.; Duclos, F.J.; Witmer, M.; et al. Dissection of the endogenous cellular pathways of PCSK9-induced LDLR degradation: Evidence for an intracellular route. Journal of Biological Chemistry 2009, 284, 28856-28864. [CrossRef]
- Zaid, A.; Roubtsova, A.; Essalmani, R.; Marcinkiewicz, J.; Chamberland, A.; Hamelin, J.; Tremblay, M.; Jacques, H.; Jin, W.; Davignon, J.; et al. Proprotein convertase subtilisin/kexin type 9 (PCSK9): Hepatocyte-specific low-density lipoprotein receptor degradation and critical role in mouse liver regeneration. Hepatology 2008, 48, 646-654. [CrossRef]
- Fruchart Gaillard, C.; Ouadda, A.B.D.; Ciccone, L.; Girard, E.; Mikaeeli, S.; Evagelidis, A.; Le Devehat, M.; Susan-Resiga, D.; Lajeunesse, E.C.; Nozach, H.; et al. Molecular interactions of PCSK9 with an inhibitory nanobody, CAP1 and HLA-C: Functional regulation of LDLR levels. Mol Metab 2023, 67, 101662. [CrossRef]
- Devay, R.M.; Shelton, D.L.; Liang, H. Characterization of Proprotein Convertase Subtilisin/Kexin Type 9 (PCSK9) Trafficking Reveals a Novel Lysosomal Targeting Mechanism via Amyloid Precursor-like Protein 2 (APLP2). J Biol. Chem. 2013, 288, 10805-10818.
- Sparks, R.P.; Arango, A.S.; Jenkins, J.L.; Guida, W.C.; Tajkhorshid, E.; Sparks, C.E.; Sparks, J.D.; Fratti, R.A. An Allosteric Binding Site on Sortilin Regulates the Trafficking of VLDL, PCSK9, and LDLR in Hepatocytes. Biochemistry 2020, 59, 4321-4335. [CrossRef]
- Butkinaree, C.; Canuel, M.; Essalmani, R.; Poirier, S.; Benjannet, S.; Asselin, M.C.; Roubtsova, A.; Hamelin, J.; Marcinkiewicz, J.; Chamberland, A.; et al. Amyloid precursor-like protein 2 and sortilin do not regulate the PCSK9-mediated low density lipoprotein receptor degradation but interact with each other. Journal of Biological Chemistry 2015, 290, 18609-18620. [CrossRef]
- Jang, H.D.; Lee, S.E.; Yang, J.; Lee, H.C.; Shin, D.; Lee, H.; Lee, J.; Jin, S.; Kim, S.; Lee, S.J.; et al. Cyclase-associated protein 1 is a binding partner of proprotein convertase subtilisin/kexin type-9 and is required for the degradation of low-density lipoprotein receptors by proprotein convertase subtilisin/kexin type-9. Eur Heart J 2020, 41, 239-252. [CrossRef]
- Kotowski, I.K.; Pertsemlidis, A.; Luke, A.; Cooper, R.S.; Vega, G.L.; Cohen, J.C.; Hobbs, H.H. A Spectrum of PCSK9 Alleles Contributes to Plasma Levels of Low-Density Lipoprotein Cholesterol. American Journal of Human Genetics 2006, 78, 410-422. [CrossRef]
- Liu, X.; Bao, X.; Hu, M.; Chang, H.; Jiao, M.; Cheng, J.; Xie, L.; Huang, Q.; Li, F.; Li, C.Y. Inhibition of PCSK9 potentiates immune checkpoint therapy for cancer. Nature 2020, 588, 693-698. [CrossRef]
- Klein, J.; Sato, A. The HLA system. First of two parts. N Engl J Med 2000, 343, 702-709. [CrossRef]
- Stasiak, M.; Zawadzka-Starczewska, K.; Tymoniuk, B.; Stasiak, B.; Lewiński, A. Associations between Lipid Profiles and Graves' Orbitopathy can Be HLA-Dependent. Genes (Basel) 2023, 14. [CrossRef]
- Demetz, E.; Tymoszuk, P.; Hilbe, R.; Volani, C.; Haschka, D.; Heim, C.; Auer, K.; Lener, D.; Zeiger, L.B.; Pfeifhofer-Obermair, C.; et al. The haemochromatosis gene Hfe and Kupffer cells control LDL cholesterol homeostasis and impact on atherosclerosis development. Eur Heart J 2020, 41, 3949-3959. [CrossRef]
- Pankow, J.S.; Boerwinkle, E.; Adams, P.C.; Guallar, E.; Leiendecker-Foster, C.; Rogowski, J.; Eckfeldt, J.H. HFE C282Y homozygotes have reduced low-density lipoprotein cholesterol: the Atherosclerosis Risk in Communities (ARIC) Study. Transl Res 2008, 152, 3-10. [CrossRef]
- Susan-Resiga, D.; Girard, E.; Essalmani, R.; Roubtsova, A.; Marcinkiewicz, J.; Derbali, R.M.; Evagelidis, A.; Byun, J.H.; Lebeau, P.F.; Austin, R.C.; Seidah, N.G. Asialoglycoprotein receptor 1 is a novel PCSK9-independent ligand of liver LDLR cleaved by furin. J Biol Chem 2021, 297, 101177. [CrossRef]
- Kruse, V.; Hamann, C.; Monecke, S.; Cyganek, L.; Elsner, L.; Hübscher, D.; Walter, L.; Streckfuss-Bömeke, K.; Guan, K.; Dressel, R. Human Induced Pluripotent Stem Cells Are Targets for Allogeneic and Autologous Natural Killer (NK) Cells and Killing Is Partly Mediated by the Activating NK Receptor DNAM-1. PLoS One 2015, 10, e0125544. [CrossRef]
- Ben Djoudi Ouadda, A.; Gauthier, M.S.; Susan-Resiga, D.; Girard, E.; Essalmani, R.; Black, M.; Marcinkiewicz, J.; Forget, D.; Hamelin, J.; Evagelidis, A.; et al. Ser-Phosphorylation of PCSK9 (Proprotein Convertase Subtilisin-Kexin 9) by Fam20C (Family With Sequence Similarity 20, Member C) Kinase Enhances Its Ability to Degrade the LDLR (Low-Density Lipoprotein Receptor). Arterioscler Thromb Vasc Biol 2019, 39, 1996-2013. [CrossRef]
- Dubuc, G.; Tremblay, M.; Pare, G.; Jacques, H.; Hamelin, J.; Benjannet, S.; Boulet, L.; Genest, J.; Bernier, L.; Seidah, N.G.; Davignon, J. A new method for measurement of total plasma PCSK9: clinical applications. Journal of Lipid Research 2010, 51, 140-149.
- Lowry, O.H.; Rosebrough, N.J.; Farr, A.L.; Randall, R.J. Protein measurement with the Folin phenol reagent. Journal of Biological Chemistry 1951, 193, 265-275.
- Nassoury, N.; Blasiole, D.A.; Tebon, O.A.; Benjannet, S.; Hamelin, J.; Poupon, V.; McPherson, P.S.; Attie, A.D.; Prat, A.; Seidah, N.G. The Cellular Trafficking of the Secretory Proprotein Convertase PCSK9 and Its Dependence on the LDLR. Traffic. 2007, 8, 718-732. [CrossRef]
- Pettersen, E.F.; Goddard, T.D.; Huang, C.C.; Couch, G.S.; Greenblatt, D.M.; Meng, E.C.; Ferrin, T.E. UCSF Chimera--a visualization system for exploratory research and analysis. J Comput Chem 2004, 25, 1605-1612. [CrossRef]
- Singh, A.; Copeland, M.M.; Kundrotas, P.J.; Vakser, I.A. GRAMM Web Server for Protein Docking. Methods Mol Biol 2024, 2714, 101-112. [CrossRef]
- Kuhlman, B.; Dantas, G.; Ireton, G.C.; Varani, G.; Stoddard, B.L.; Baker, D. Design of a novel globular protein fold with atomic-level accuracy. Science 2003, 302, 1364-1368. [CrossRef]
- Strom, T.B.; Holla, O.L.; Tveten, K.; Cameron, J.; Berge, K.E.; Leren, T.P. Disrupted recycling of the low density lipoprotein receptor by PCSK9 is not mediated by residues of the cytoplasmic domain. Mol. Genet. Metab 2010, 101, 76-80.
- Holla, O.L.; Strom, T.B.; Cameron, J.; Berge, K.E.; Leren, T.P. A chimeric LDL receptor containing the cytoplasmic domain of the transferrin receptor is degraded by PCSK9. Mol. Genet. Metab 2010, 99, 149-156.
- Canuel, M.; Sun, X.; Asselin, M.C.; Paramithiotis, E.; Prat, A.; Seidah, N.G. Proprotein convertase subtilisin/kexin type 9 (PCSK9) can mediate degradation of the low density lipoprotein receptor-related protein 1 (LRP-1). PLoS ONE 2013, 8, e64145.
- Johnson, J.K.; Wright, P.W.; Li, H.; Anderson, S.K. Identification of trophoblast-specific elements in the HLA-C core promoter. Hla 2018, 92, 288-297. [CrossRef]
- Roubtsova, A.; Chamberland, A.; Marcinkiewicz, J.; Essalmani, R.; Fazel, A.; Bergeron, J.J.; Seidah, N.G.; Prat, A. PCSK9 deficiency unmasks a sex/tissue-specific subcellular distribution of the LDL and VLDL receptors in mice. Journal of Lipid Research 2015, 56, 2133-2142. [CrossRef]
- Lebron, J.A.; Bennett, M.J.; Vaughn, D.E.; Chirino, A.J.; Snow, P.M.; Mintier, G.A.; Feder, J.N.; Bjorkman, P.J. Crystal structure of the hemochromatosis protein HFE and characterization of its interaction with transferrin receptor. Cell 1998, 93, 111-123. [CrossRef]
- Gross, C.N.; Irrinki, A.; Feder, J.N.; Enns, C.A. Co-trafficking of HFE, a nonclassical major histocompatibility complex class I protein, with the transferrin receptor implies a role in intracellular iron regulation. J Biol Chem 1998, 273, 22068-22074. [CrossRef]
- Ganz, T. Hepcidin, a key regulator of iron metabolism and mediator of anemia of inflammation. Blood 2003, 102, 783-788.
- Zhang, A.S.; Davies, P.S.; Carlson, H.L.; Enns, C.A. Mechanisms of HFE-induced regulation of iron homeostasis: Insights from the W81A HFE mutation. Proc Natl Acad Sci U S A 2003, 100, 9500-9505. [CrossRef]
- Guillemot, J.; Asselin, M.C.; Susan-Resiga, D.; Essalmani, R.; Seidah, N.G. Deferoxamine stimulates LDLR expression and LDL uptake in HepG2 cells. Mol Nutr. Food Res. 2016, 60, 600-608.
- Parolini, F.; Biswas, P.; Serena, M.; Sironi, F.; Muraro, V.; Guizzardi, E.; Cazzoletti, L.; Scupoli, M.T.; Gibellini, D.; Ugolotti, E.; et al. Stability and Expression Levels of HLA-C on the Cell Membrane Modulate HIV-1 Infectivity. J Virol 2018, 92. [CrossRef]
- Mayer, G.; Poirier, S.; Seidah, N.G. Annexin A2 is a C-terminal PCSK9-binding protein that regulates endogenous low density lipoprotein receptor levels. Journal of Biological Chemistry 2008, 283, 31791-31801.
- Qian, Y.W.; Schmidt, R.J.; Zhang, Y.; Chu, S.; Lin, A.; Wang, H.; Wang, X.; Beyer, T.P.; Bensch, W.R.; Li, W.; et al. Secreted PCSK9 downregulates low density lipoprotein receptor through receptor-mediated endocytosis. Journal of Lipid Research 2007, 48, 1488-1498.
- Puddu, A.; Maggi, D. Emerging Role of Caveolin-1 in GLP-1 Action. Front Endocrinol (Lausanne) 2021, 12, 668012. [CrossRef]
- Mikaeeli, S.; Susan-Resiga, D.; Girard, E.; Ben Djoudi Ouadda, A.; Day, R.; Prost, S.; Seidah, N.G. Functional analysis of natural PCSK9 mutants in modern and archaic humans. Febs j 2020, 287, 515-528. [CrossRef]
- Ramalingam, T.S.; West, A.P., Jr.; Lebron, J.A.; Nangiana, J.S.; Hogan, T.H.; Enns, C.A.; Bjorkman, P.J. Binding to the transferrin receptor is required for endocytosis of HFE and regulation of iron homeostasis. Nat. Cell Biol. 2000, 2, 953-957.
- Ultsch, M.; Li, W.; Eigenbrot, C.; Di Lello, P.; Lipari, M.T.; Gerhardy, S.; AhYoung, A.P.; Quinn, J.; Franke, Y.; Chen, Y.; et al. Identification of a Helical Segment within the Intrinsically Disordered Region of the PCSK9 Prodomain. J Mol Biol 2019, 431, 885-903. [CrossRef]
- Seidah, N.G.; Garçon, D. Expanding Biology of PCSK9: Roles in Atherosclerosis and Beyond. Curr Atheroscler Rep 2022, 1-10. [CrossRef]
- Wang, R.; Liu, H.; He, P.; An, D.; Guo, X.; Zhang, X.; Feng, M. Inhibition of PCSK9 enhances the antitumor effect of PD-1 inhibitor in colorectal cancer by promoting the infiltration of CD8(+) T cells and the exclusion of Treg cells. Front Immunol 2022, 13, 947756. [CrossRef]
- Seidah, N.G.; Garcon, D. Expanding Biology of PCSK9: Roles in Atherosclerosis and Beyond. Curr Atheroscler Rep 2022, 24, 821-830. [CrossRef]
- Tan, P.K.; Waites, C.; Liu, Y.; Krantz, D.E.; Edwards, R.H. A leucine-based motif mediates the endocytosis of vesicular monoamine and acetylcholine transporters. J Biol Chem 1998, 273, 17351-17360. [CrossRef]
- Bennett, M.J.; Lebron, J.A.; Bjorkman, P.J. Crystal structure of the hereditary haemochromatosis protein HFE complexed with transferrin receptor. Nature 2000, 403, 46-53. [CrossRef]
- Di Guglielmo, G.M.; Le Roy, C.; Goodfellow, A.F.; Wrana, J.L. Distinct endocytic pathways regulate TGF-beta receptor signalling and turnover. Nat Cell Biol 2003, 5, 410-421. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



