Submitted:
12 March 2024
Posted:
13 March 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction

2. Materials and Methods
2.1. Cell Culture
2.2. Drug Preparation
2.3. MTT (3-(4,5-Dimethylthiazol-2-yl)-2,5-Diphenyltetrazolium Bromide) Assay
2.5. Scratch Assay
2.6. Spheroid Invasion Assay & IF
2.7. Confocal Microscopy
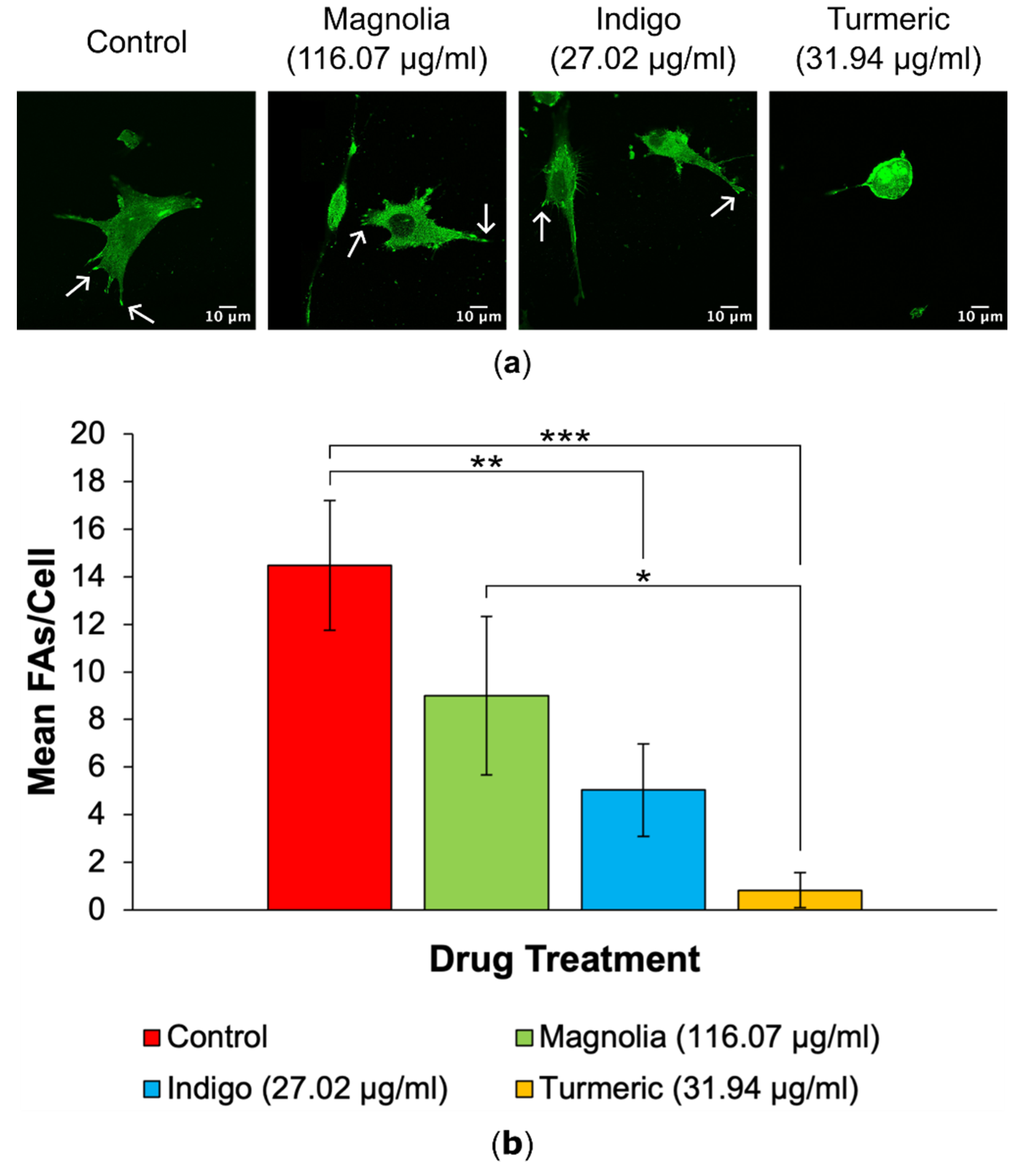
2.8. Focal Adhesion Analysis
2.9. Actin Localisation Analysis
2.10. Statistical Analysis
3. Results
3.1. MTT Assays
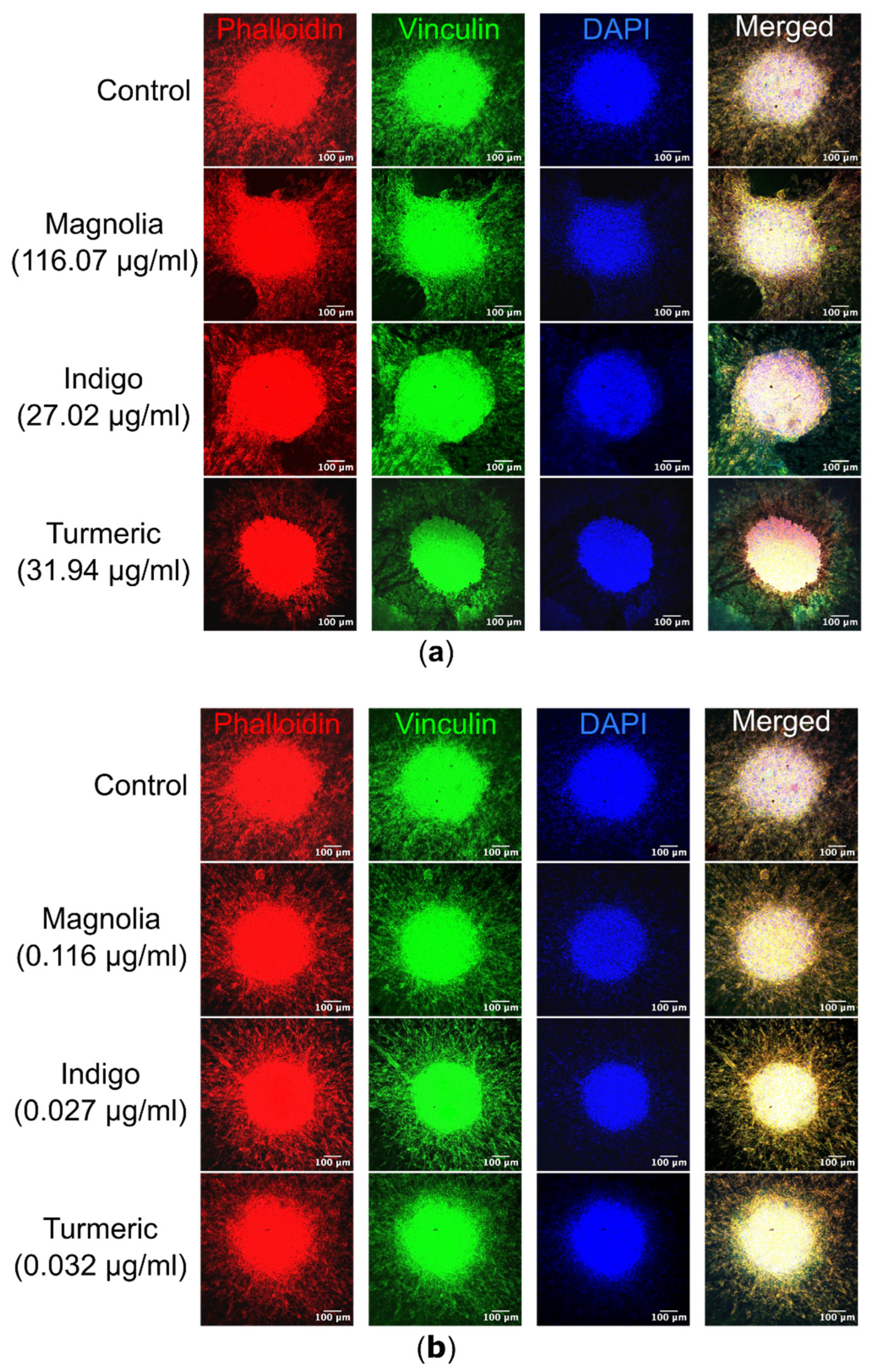
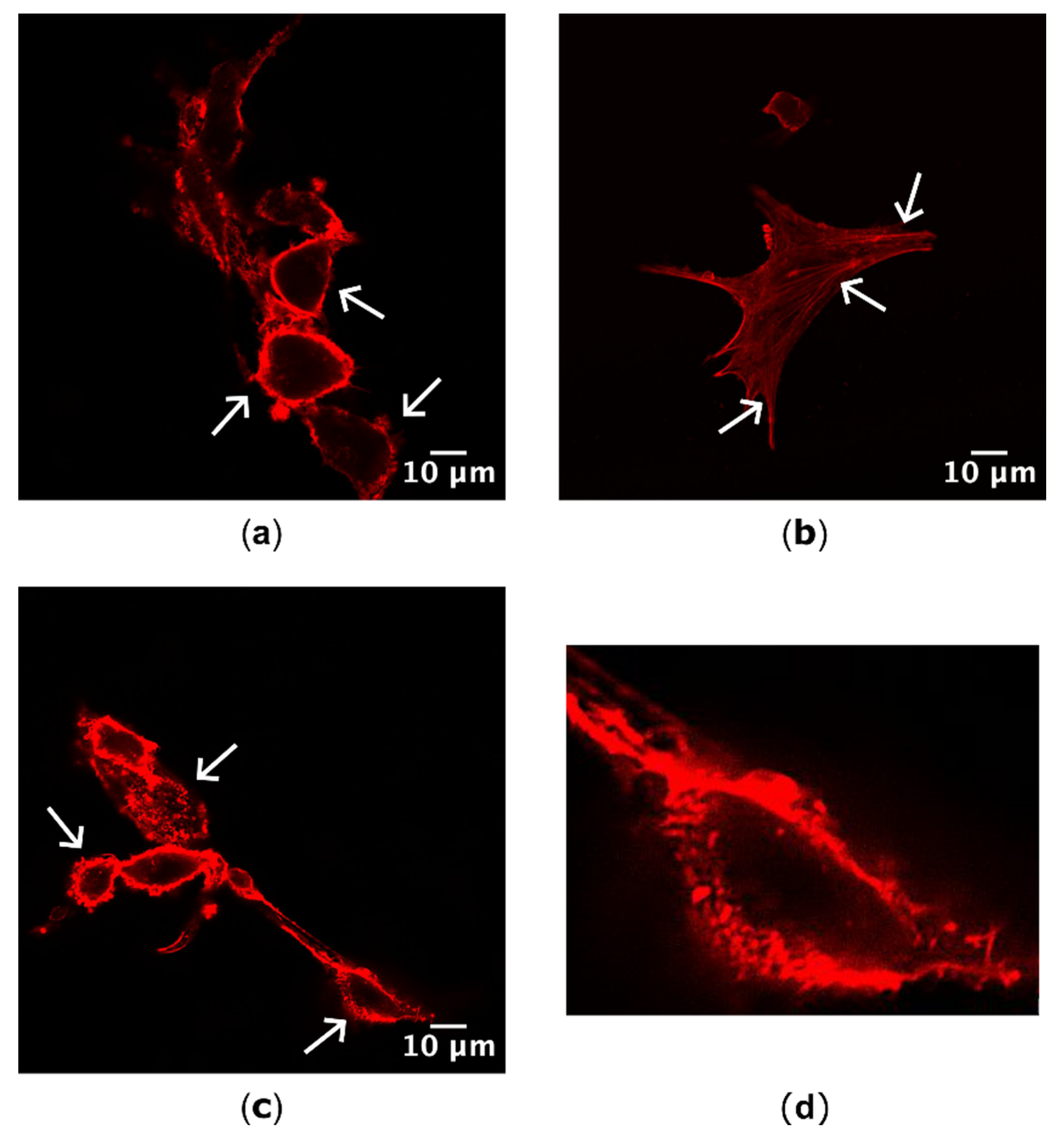
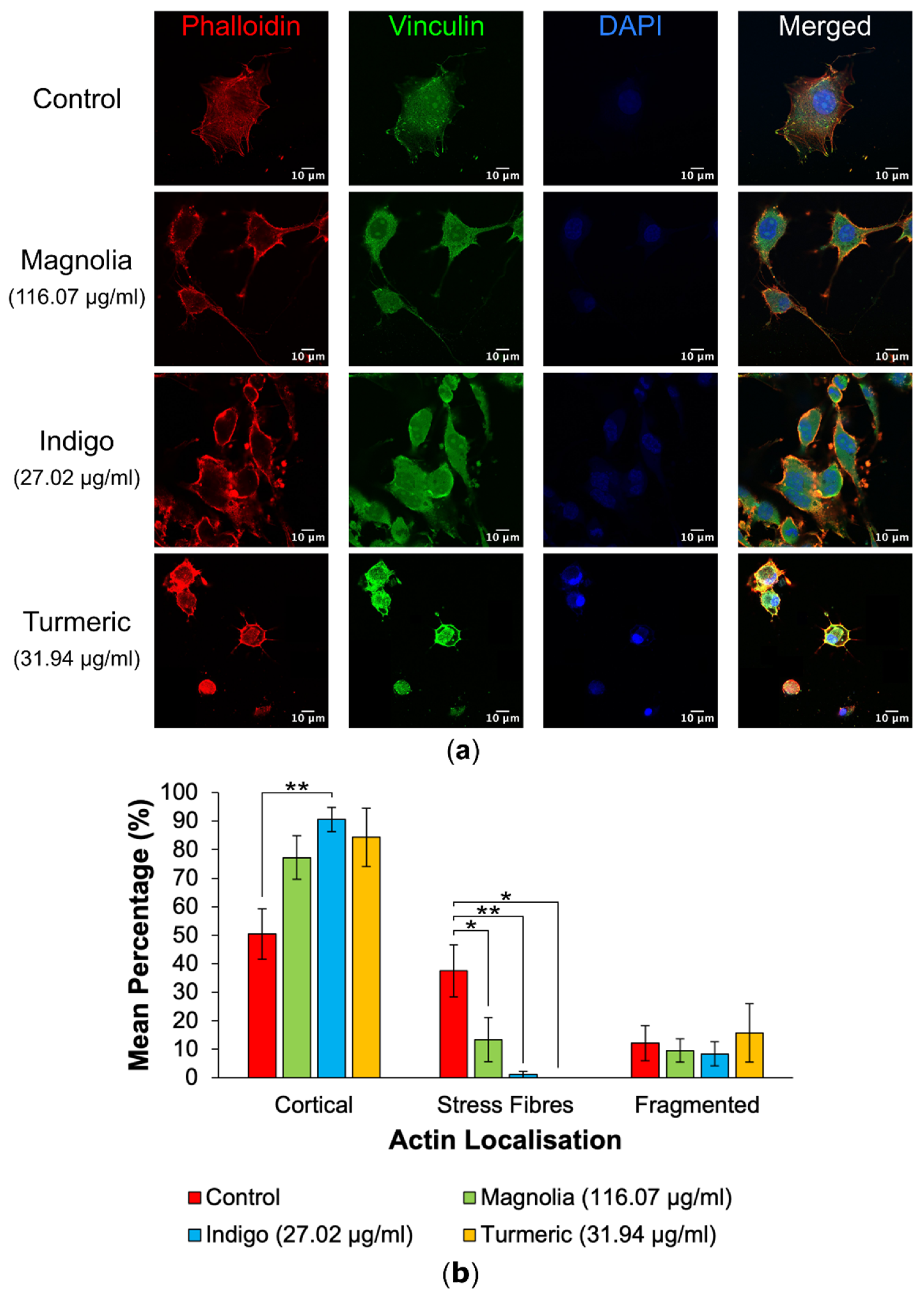
3.2.1. Actin Localization
3.2.2. FA Generation
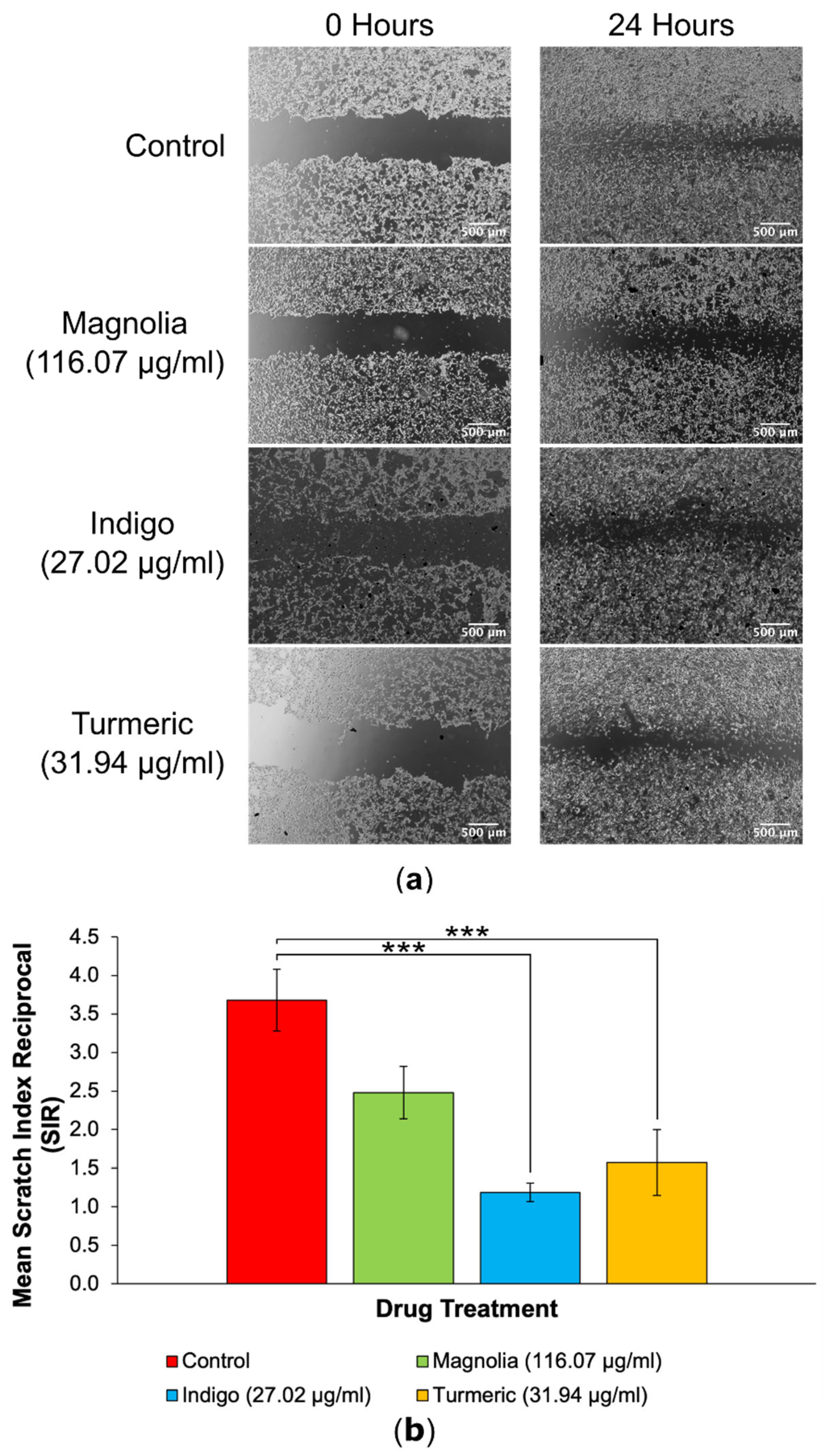
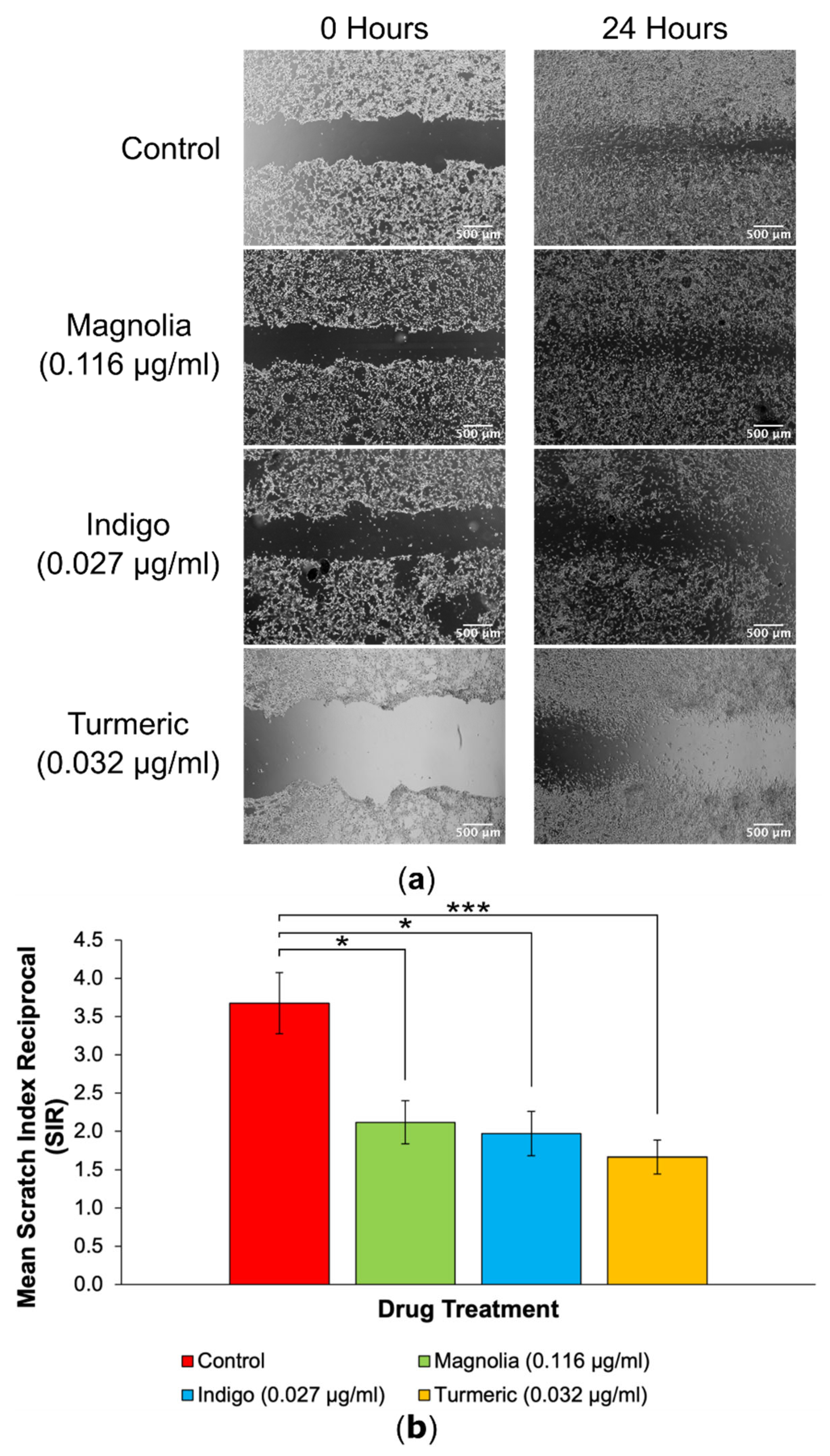
3.3. Scratch Assays
3.3.1. Drugs Administered at Low Concentration
3.3.2. Drugs Administered at High Concentration
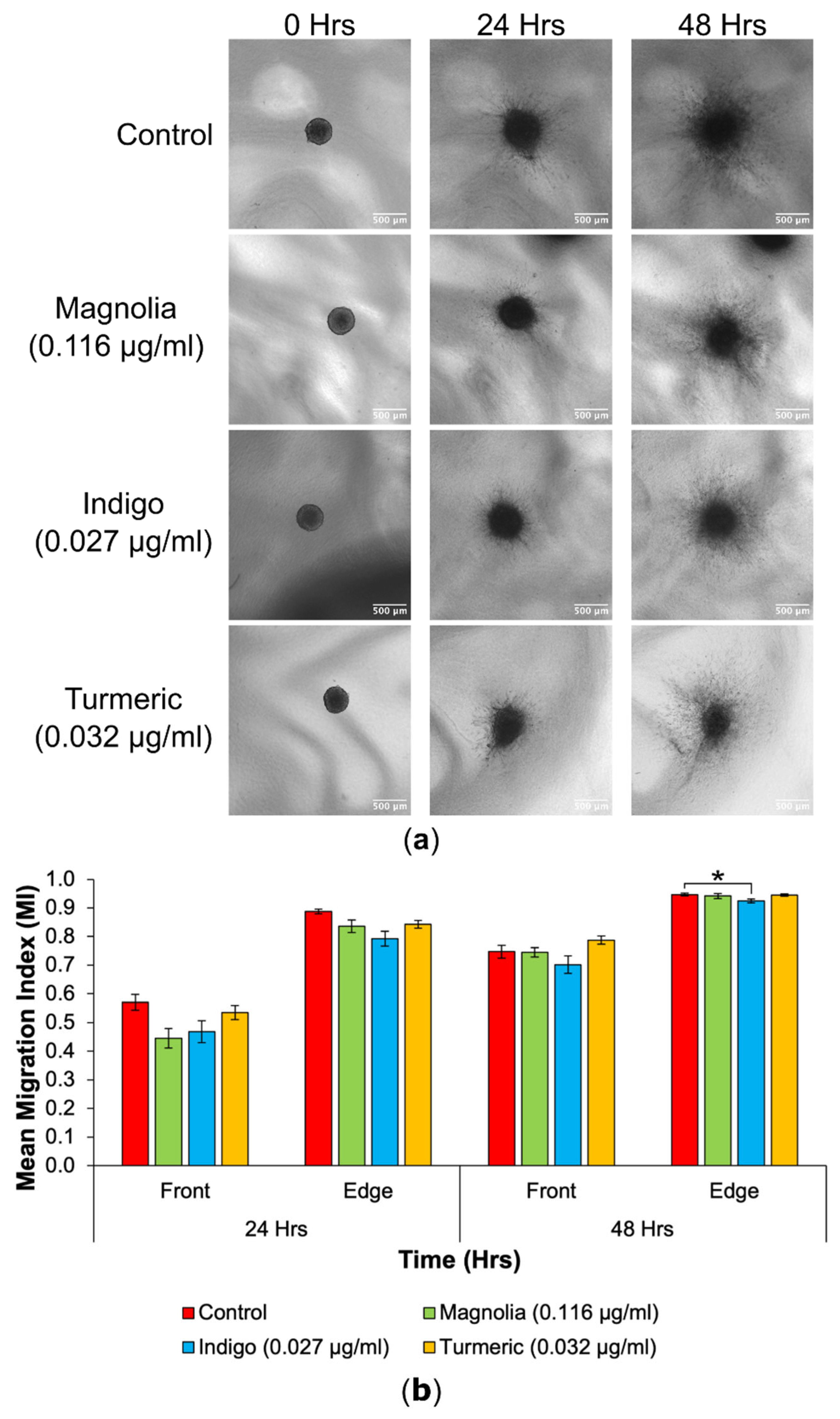
3.4. Spheroid Invasion Assays
3.4.1. Drugs Administered at Low Concentration
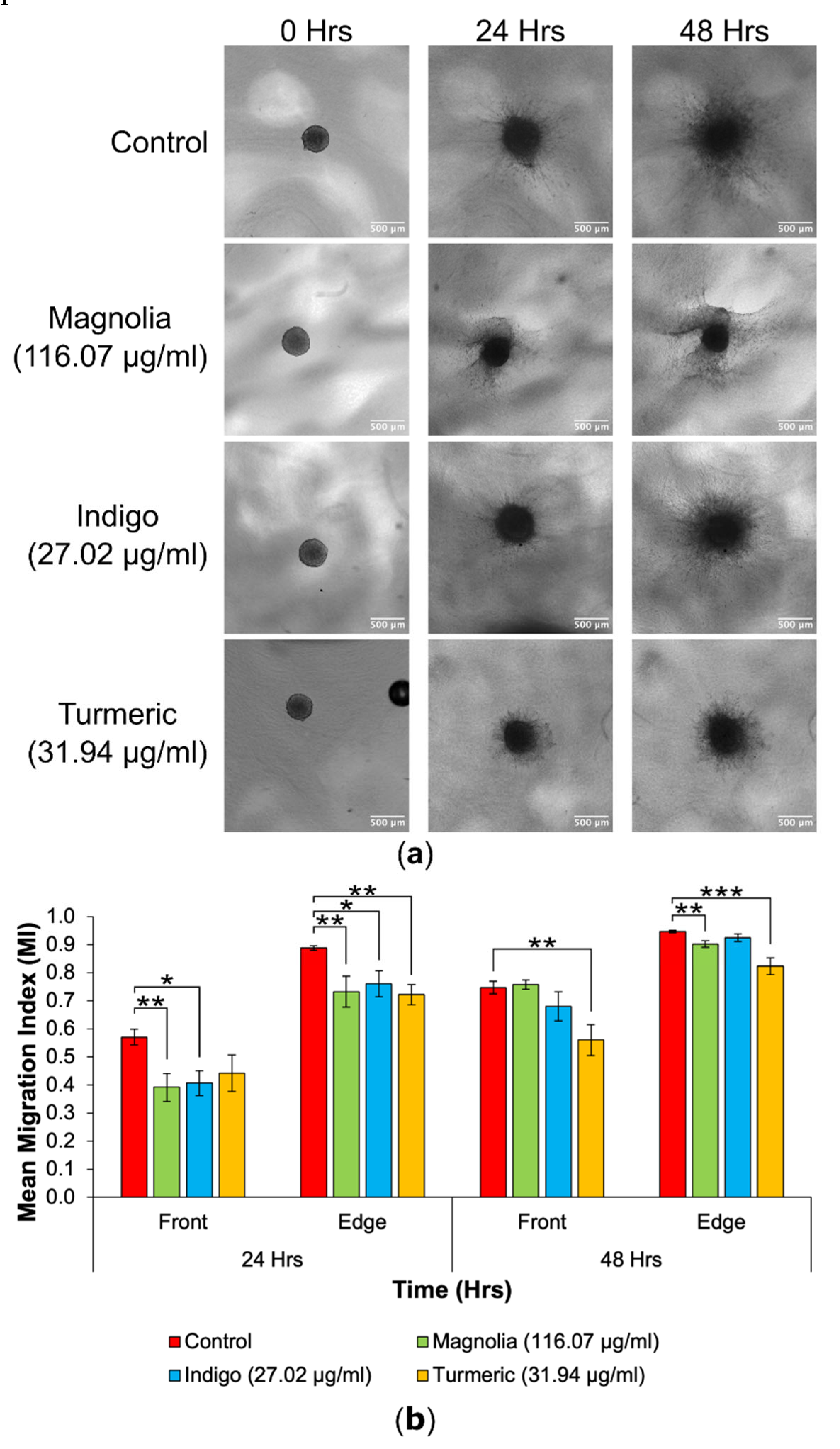
3.4.2. Drugs Administered at High Concentration
3.4.3. Spheroid Confocal Microscopy
4. Discussion
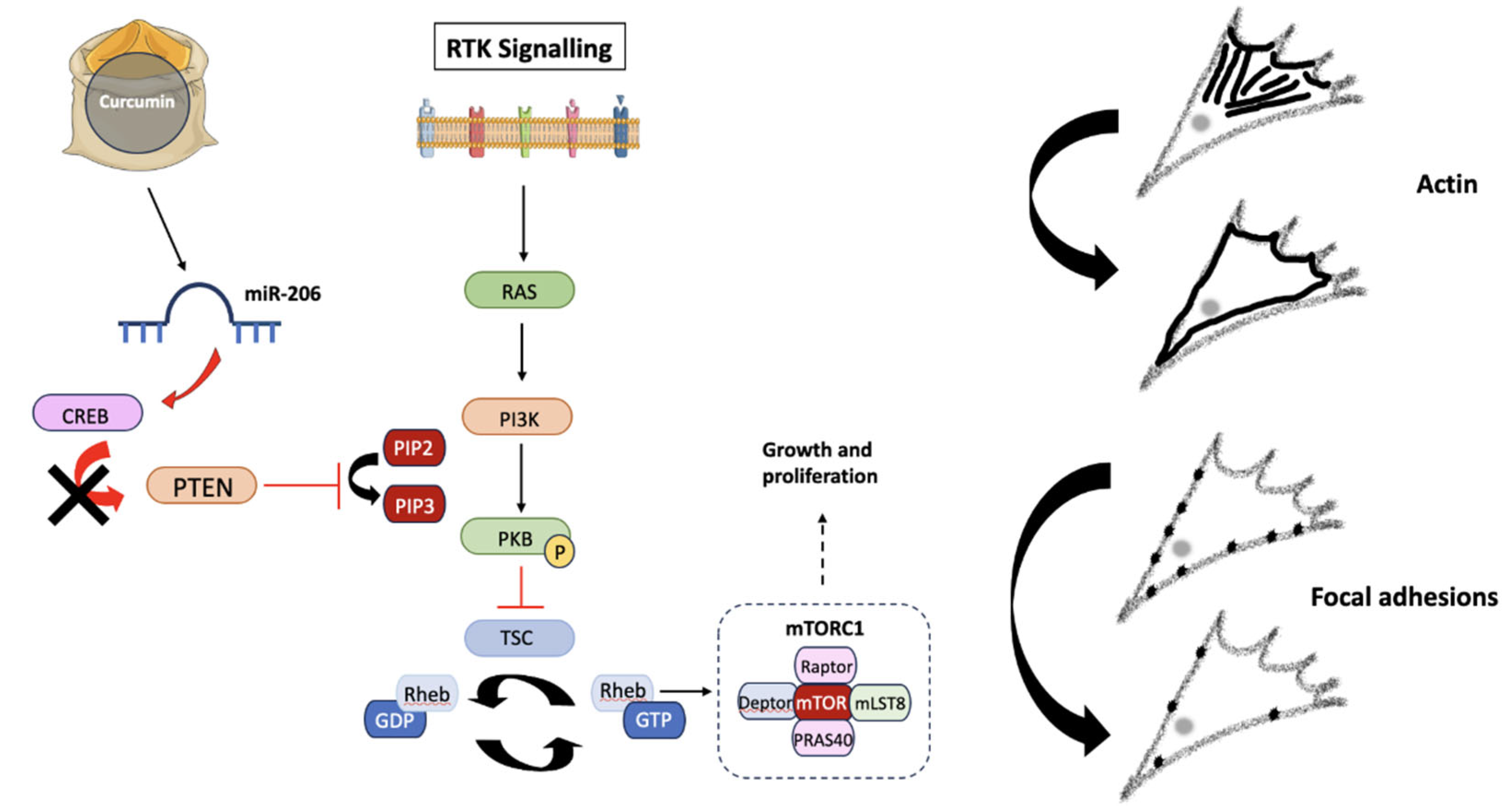
4.1. Turmeric
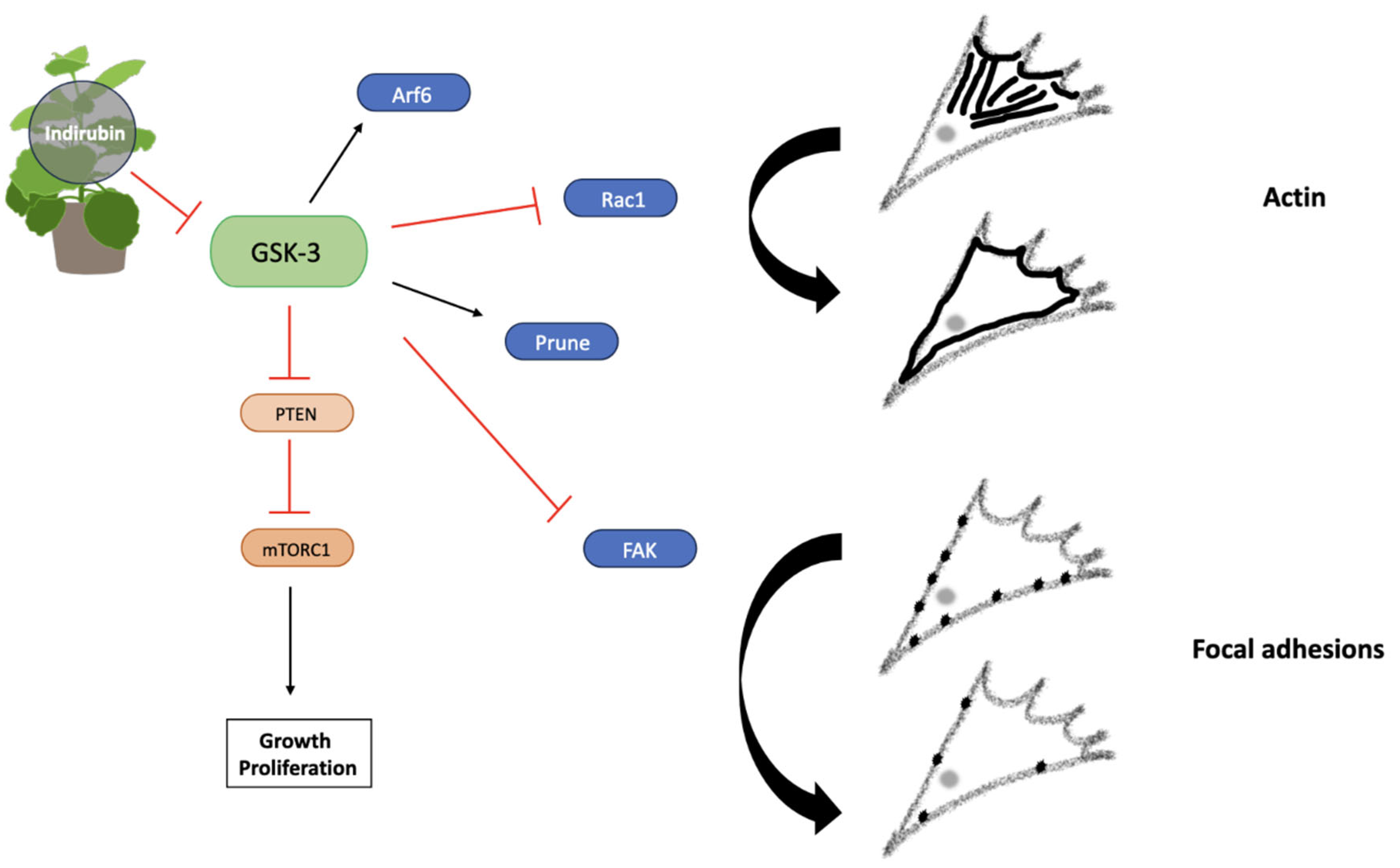
4.2. Indigo
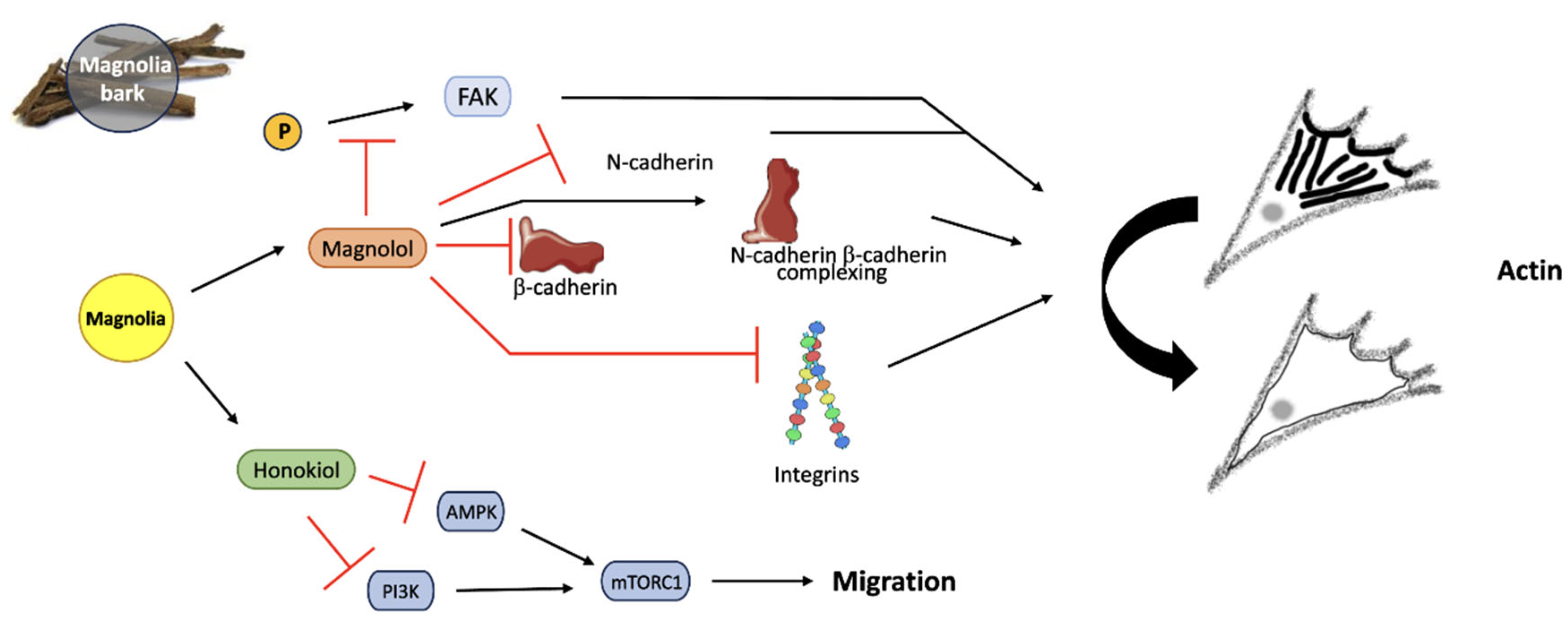
4.3. Magnolia
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Weller, M.; et al. Standards of care for treatment of recurrent glioblastoma--are we there yet? Neuro-Oncology 2013, 15, 4–27. [Google Scholar] [CrossRef] [PubMed]
- Thakkar, J.P.; et al. Epidemiologic and molecular prognostic review of glioblastoma. Cancer Epidemiology, Biomarkers & Prevention 2014, 23, 1985–1996. [Google Scholar]
- Minniti, G.; et al. Current status and recent advances in reirradiation of glioblastoma. Radiation Oncology 2021, 16. [Google Scholar] [CrossRef] [PubMed]
- Davis, M.E. Glioblastoma: Overview of Disease and Treatment. Clinical Journal of Oncology Nursing 2016, 20, 2–8. [Google Scholar] [CrossRef]
- Wu, W.; et al. Glioblastoma multiforme (GBM): An overview of current therapies and mechanisms of resistance. Pharmacological Research 2021, 171. [Google Scholar] [CrossRef] [PubMed]
- Singh, S.K. and R. Singh, Nanotherapy: targeting the tumour microenvironment. Nature Reviews Cancer 2022, 22, 258. [Google Scholar] [CrossRef] [PubMed]
- Mierke, C.T. Physical view on migration modes. Cell Adhesion & Migration 2015, 9, 367–379. [Google Scholar]
- O'Neill, G.M. The coordination between actin filaments and adhesion in mesenchymal migration. 3Cell Adhesion & Migration 2009, 3, 355–357. [Google Scholar]
- Wehrle-Haller, B. Structure and function of focal adhesions. Current Opinion in Cell Biology 2012, 24, 116–124. [Google Scholar] [CrossRef]
- Sen, T.; Samanta, S.K. Medicinal plants, human health and biodiversity: a broad review. Biotechnological Applications of Biodiversity 2015, 147, 59–110. [Google Scholar]
- Khan, T.; Gurav, P. PhytoNanotechnology: Enhancing Delivery of Plant Based Anti-cancer Drugs. Frontiers in Pharmacology 2018, 8. [Google Scholar] [CrossRef]
- Sarrica, A.; et al. Safety and Toxicology of Magnolol and Honokiol. Planta Medica 2018, 84, 1151–1164. [Google Scholar] [CrossRef] [PubMed]
- Khatoon, F.; et al. Pharmacological features, health benefits and clinical implications of Honokiol. Journal of Biomolecular Structure and Dynamics 2023, 41, 7511–7533. [Google Scholar] [PubMed]
- Banik, K.; et al. Honokiol for cancer therapeutics: A traditional medicine that can modulate multiple oncogenic targets. Pharmacological Research 2019, 144, 192–209. [Google Scholar] [CrossRef] [PubMed]
- Pan, J.; et al. Honokiol targets mitochondria to halt cancer progression and metastasis. Molecular Nutrition & Food Research 2016, 60, 1383–1395. [Google Scholar]
- Ranaware, A.M.; et al. Magnolol: A Neolignan from the Magnolia Family for the Prevention and Treatment of Cancer. International Journal of Molecular Sciences 2018, 19, 2362. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.-Y.; et al. Bioactivity, Health Benefits, and Related Molecular Mechanisms of Curcumin: Current Progress, Challenges, and Perspectives. Nutrients 2018, 10, 1553. [Google Scholar] [CrossRef] [PubMed]
- Astinfeshana, M.; et al. Curcumin inhibits angiogenesis in endothelial cells using downregulation of the PI3K/Akt signaling pathway. Food Bioscience 2019, 29, 86–93. [Google Scholar] [CrossRef]
- Sharifi-Rad, J.; et al. Turmeric and Its Major Compound Curcumin on Health: Bioactive Effects and Safety Profiles for Food, Pharmaceutical, Biotechnological and Medicinal Applications. Frontiers in Pharmacology 2020, 11. [Google Scholar] [CrossRef]
- Bojko, A.; et al. Modulatory Effects of Curcumin and Tyrphostins (AG494 and AG1478) on Growth Regulation and Viability of LN229 Human Brain Cancer Cells. Nutrition and Cancer 2015, 67, 1170–1182. [Google Scholar]
- Zanotto-Filho, A.; et al. The curry spice Curcumin selectively inhibits cancer cells growth in vitro and in preclinical model of glioblastoma. Journal of Nutritional Biochemistry 2012, 23, 591–601. [Google Scholar] [CrossRef] [PubMed]
- Cui, H.; et al. Recent Advancements in Natural Plant Colorants Used for Hair Dye Applications: A Review. Molecules 2022, 27, 8062. [Google Scholar] [CrossRef] [PubMed]
- Xie, Q.; et al. A novel realgar-indigo naturalis formula more effectively induces apoptosis in NB4 cells. Pakistan Journal of Pharmaceutical Sciences 2019, 32, 957–962. [Google Scholar] [PubMed]
- Williams, S.P.; et al. Indirubins Decrease Glioma Invasion by Blocking Migratory Phenotypes in Both the Tumor and Stromal Endothelial Cell Compartments. Cancer Research 2011, 71, 5374–5380. [Google Scholar] [CrossRef]
- Cockle, J.V.; et al. Cell migration in paediatric glioma; characterisation and potential therapeutic targeting. British Journal of Cancer 2015, 112, 693–703. [Google Scholar] [CrossRef] [PubMed]
- Song, H.; et al. Discovery of selective, antimetastatic and anti-cancer stem cell metallohelices via post-assembly modification. Chemical Science 2019, 10, 8547–8557. [Google Scholar] [CrossRef] [PubMed]
- Schindelin, J.; et al. Fiji: an open-source platform for biological-image analysis. Nature Methods 2012, 9, 676–682. [Google Scholar] [CrossRef] [PubMed]
- IBM Corp. Released 2021; IBM SPSS Statistics for Macintosh, Version 28.0; IBM Corp: Armonk, NY.
- Revach, O.-Y.; I. Grosheva, and B. Geiger, Biomechanical regulation of focal adhesion and invadopodia formation. Journal of Cell Science 2020, 133. [Google Scholar] [CrossRef]
- Alvarez-González, B.; et al. Cytoskeletal Mechanics Regulating Amoeboid Cell Locomotion. Applied Mechanics Reviews 2014, 66. [Google Scholar] [CrossRef]
- Rösel, D.; et al. Up-regulation of Rho/ROCK signaling in sarcoma cells drives invasion and increased generation of protrusive forces. Molecular Cancer Research 2008, 6, 1410–1420. [Google Scholar] [CrossRef]
- Kosla, J.; et al. Metastasis of aggressive amoeboid sarcoma cells is dependent on Rho/ROCK/MLC signaling. Cell Communication & Signalling 2013, 11. [Google Scholar]
- Matsuoka, T. and M. Yashiro, Rho/ROCK signaling in motility and metastasis of gastric cancer. World Journal of Gastroenterology 2014, 20, 13756–13766. [Google Scholar] [CrossRef] [PubMed]
- Ketchen, S.E.; et al. Drug Resistance in Glioma Cells Induced by a Mesenchymal-Amoeboid Migratory Switch. Biomedicines 2021, 10, 9–24. [Google Scholar] [CrossRef] [PubMed]
- Davoodvandi, A.; et al. Antimetastatic Effects of Curcumin in Oral and Gastrointestinal Cancers. Frontiers in Pharmacology 2021, 12. [Google Scholar] [CrossRef] [PubMed]
- Bravo-Cordero, J.J.; et al. Functions of cofilin in cell locomotion and invasion. Nature Reviews Molecular Cell Biology 2013, 14, 405–415. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; et al. Turmeric extract, with absorbable Curcumin, has potent anti-metastatic effect in vitro and in vivo. Phytomedicine 2018, 46, 131–141. [Google Scholar] [CrossRef] [PubMed]
- Mitra, S.K.; D.A. Hanson, and D.D. Schlaepfer. Focal adhesion kinase: in command and control of cell motility. Nature Reviews Molecular Cell Biology 2005, 6, 56–68. [Google Scholar] [CrossRef]
- Park, K.S.; et al. Anti-Migration and Anti-Invasion Effects of Curcumin via Suppression of Fascin Expression in Glioblastoma Cells. Brain Tumour Research and Treatment 2019, 7, 16–24. [Google Scholar] [CrossRef]
- Wang, N.; et al. Curcumin inhibits migration and invasion of non-small cell lung cancer cells through up-regulation of miR-206 and suppression of PI3K/AKT/mTOR signaling pathway. Acta Pharmaceutica 2020, 70, 399–409. [Google Scholar] [CrossRef]
- Bastola, S.; et al. Glioma-initiating cells at tumor edge gain signals from tumor core cells to promote their malignancy. Nature Communications 2020, 11. [Google Scholar] [CrossRef]
- Zhang, Y.; Q. Xia, and J. Lin. Identification of the potential oncogenes in glioblastoma based on bioinformatic analysis and elucidation of the underlying mechanisms. Oncology Reports 2018, 40, 715–725. [Google Scholar]
- Zhang, Z.; et al. PTEN regulates PLK1 and controls chromosomal stability during cell division. Cell Cycle 2016, 15, 2476–2485. [Google Scholar] [CrossRef] [PubMed]
- Hanahan, D. and R.A. Weinberg, Hallmarks of cancer: the next generation. Cell 2011, 144, 646–674. [Google Scholar] [CrossRef] [PubMed]
- He, Y.; et al. Targeting PI3K/Akt signal transduction for cancer therapy. Signal Transduction and Targeted Therapy 2021, 6. [Google Scholar] [CrossRef]
- Hemmings, B.A. and D.F. Restuccia, PI3K-PKB/Akt pathway. Cold Spring Harbor Perspectives in Biology 2012, 4. [Google Scholar] [CrossRef] [PubMed]
- Paplomata, E. and R. O'Regan, The PI3K/AKT/mTOR pathway in breast cancer: targets, trials and biomarkers. Therapeutic Advances in Medical Oncology 2014, 6, 154–166. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; et al. Complex roles of cAMP-PKA-CREB signaling in cancer. Experimental Hematology & Oncology 2020, 9. [Google Scholar]
- Yue, D. and X. Qin, miR-182 regulates trastuzumab resistance by targeting MET in breast cancer cells. Cancer Gene Therapy 2019, 26, 1–10. [Google Scholar] [CrossRef]
- Liu, R.; et al. PI3K/AKT pathway as a key link modulates the multidrug resistance of cancers. Cell Death & Disease 2020, 11. [Google Scholar]
- Mercedes, S.A.V.; et al. Decoding leader cells in collective cancer invasion. Nature Reviews Cancer 2021, 21, 592–604. [Google Scholar] [CrossRef]
- Minata, M.; et al. Phenotypic Plasticity of Invasive Edge Glioma Stem-like Cells in Response to Ionizing Radiation. Cell Reports 2019, 26, 1893–1905. [Google Scholar] [CrossRef] [PubMed]
- Rebl, H.; et al. Synergistic effect of plasma-activated medium and novel indirubin derivatives on human skin cancer cells by activation of the AhR pathway. Scientific Reports 2022, 12. [Google Scholar] [CrossRef]
- Peat, S.; et al. Characterisation of the anti-migratory activity of the 6-bromoindirubin-3’oxime (BIO) derivative VTIND42 in patient-derived GBM subpopulations. Neuro-Oncology 2019, 21. [Google Scholar] [CrossRef]
- Hajka, D.; et al. GSK3 as a Regulator of Cytoskeleton Architecture: Consequences for Health and Disease. Cells 2021, 10. [Google Scholar] [CrossRef] [PubMed]
- Sun, T.; M. Rodriguez, and L. Kim. RGlycogen synthase kinase 3 in the world of cell migration. Development, Growth & Differentiation 2009, 51, 735–742. [Google Scholar]
- Cheng, Y.-C.; et al. Magnolol Inhibits Human Glioblastoma Cell Migration by Regulating N-Cadherin. Journal of Neuropathology & Experimental Neurology 2018, 77, 426–436. [Google Scholar]
- Mrozik, K.M.; et al. N-cadherin in cancer metastasis, its emerging role in haematological malignancies and potential as a therapeutic target in cancer. BMC Cancer 2018, 18. [Google Scholar] [CrossRef]
- Loh, C.-Y.; et al. The E-Cadherin and N-Cadherin Switch in Epithelial-to-Mesenchymal Transition: Signaling, Therapeutic Implications, and Challenges. Cells 2019, 8. [Google Scholar] [CrossRef]
- Noh, M.-G.; et al. Prognostic significance of E-cadherin and N-cadherin expression in Gliomas. BMC Cancer 2017, 17. [Google Scholar] [CrossRef]
- Yeh, P.-S.; et al. Honokiol induces autophagy of neuroblastoma cells through activating the PI3K/Akt/mTOR and endoplasmic reticular stress/ERK1/2 signaling pathways and suppressing cell migration. Cancer Letters 2016, 370, 66–77. [Google Scholar] [CrossRef]
- Lee, J.S.; et al. Honokiol induces apoptosis and suppresses migration and invasion of ovarian carcinoma cells via AMPK/mTOR signaling pathway. International Journal of Molecular Medicine 2019, 43, 1969–1978. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



