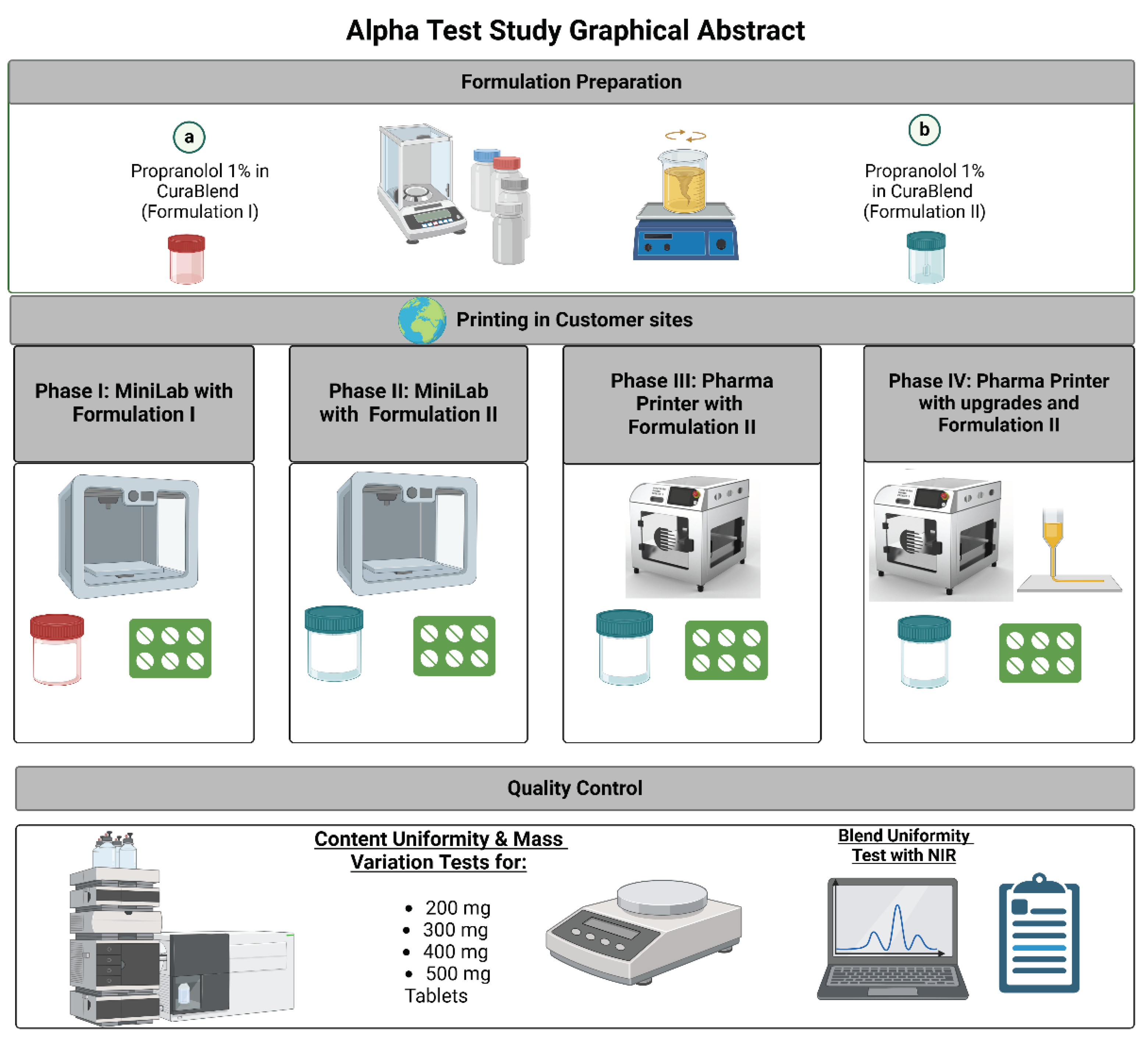
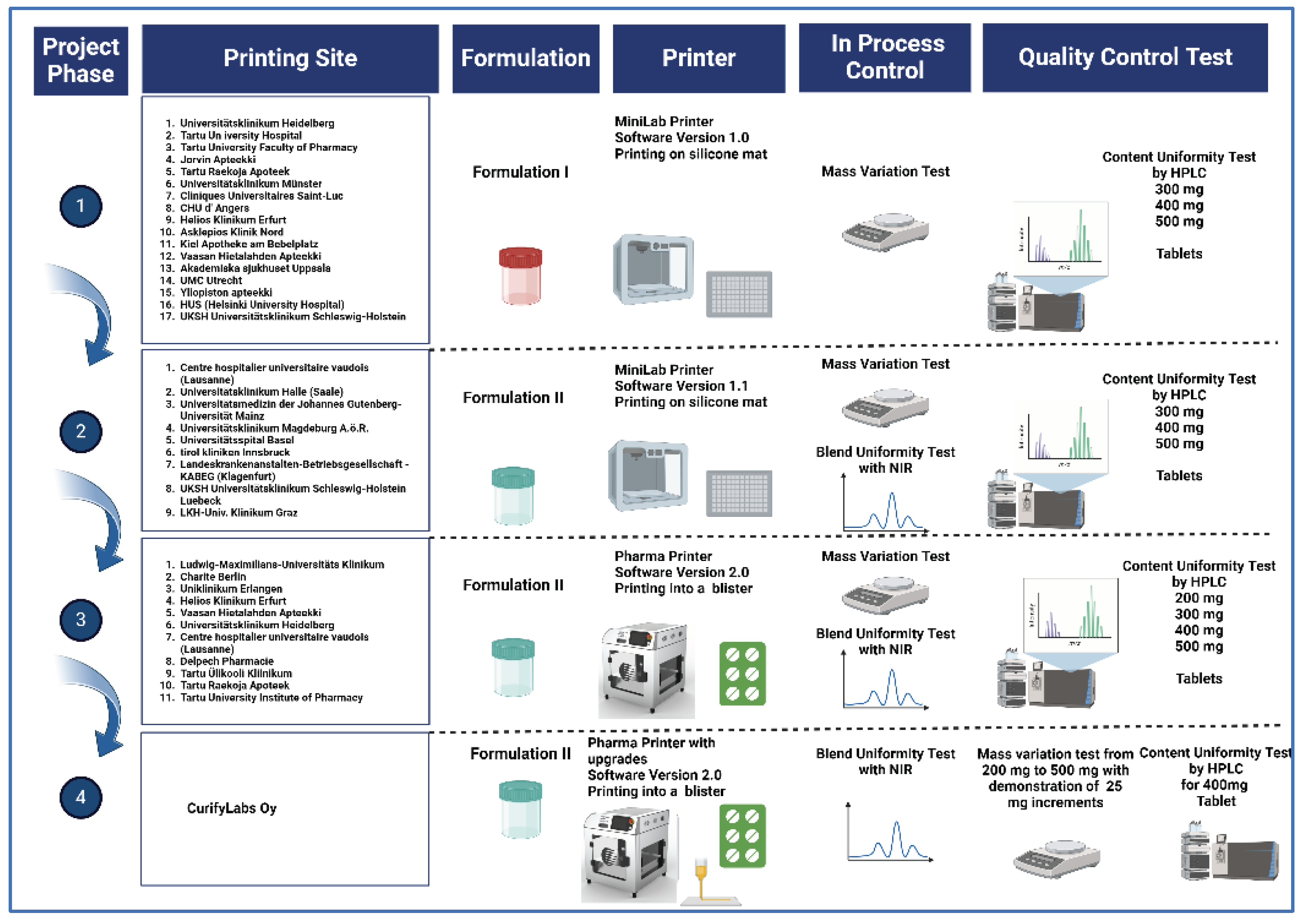
4.1. Deposition Accuracy with Mass Variation
4.1.1. Group I: MiniLab without PS Formulation
In the study conducted for Group I using MiniLab with Fom I in 17 different compounding pharmacy sites across Europe, the focus was on dosing accuracy by assessing mass variation. The experiment involved the alpha testing of MiniLab equipment and the application of Formulation 1. The analysis of printing accuracy was conducted for three different weight categories (500 mg, 400 mg, and 300 mg) across the sites.
Table 2 below provides descriptive statistics summarizing the results.
The descriptive statistics include the number of observations (N), minimum and maximum values, mean, and standard deviation for each weight category. The study indicates that, on average, the dosing accuracy for the 500 mg tablets was 93.8%, demonstrating a relatively high level of accuracy with minimal variation. Similarly, the 400 mg and 300 mg tablets exhibited mean accuracies of 89.2% and 91.2%, respectively. The standard deviations suggest the degree of variability within each weight category.
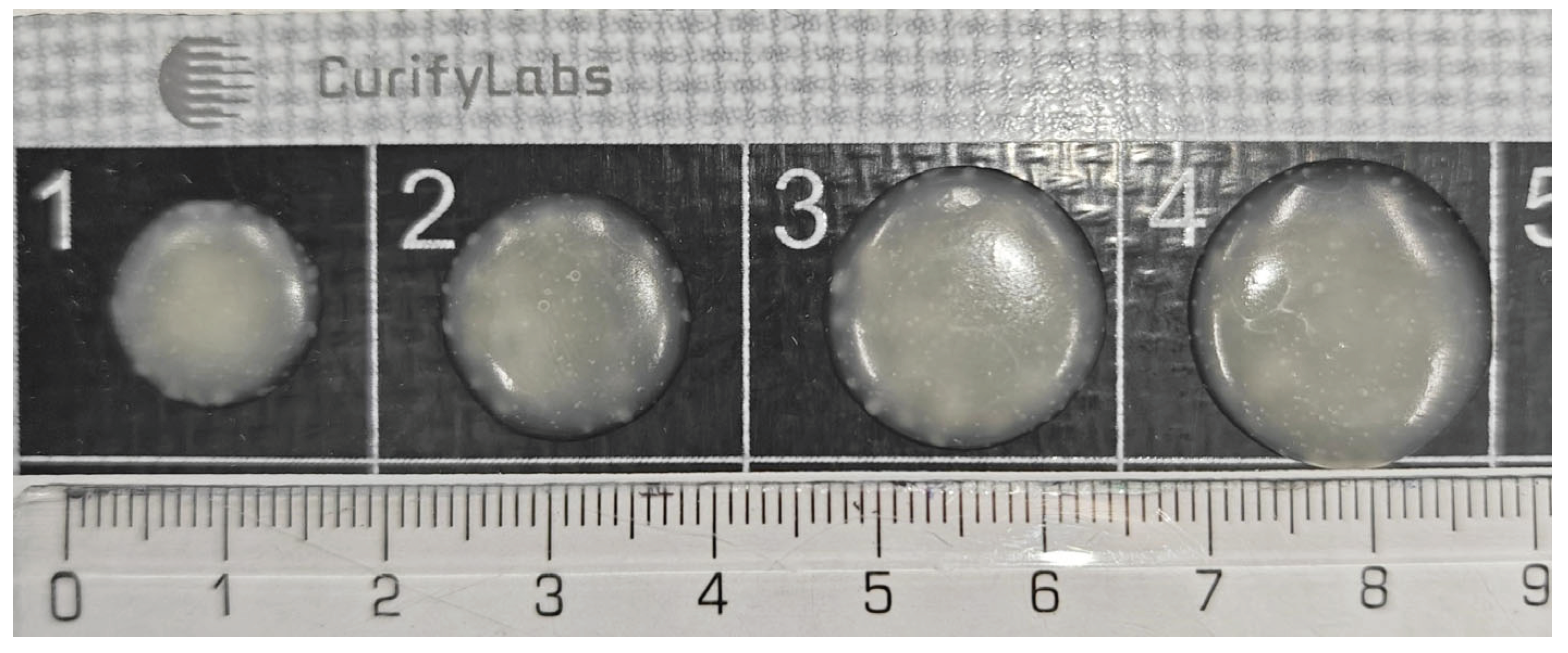
Figure 6 shows a picture of the appearance of the tablets produced in this study. A total of 2448 tablets were made in this phase of the study.
4.1.2. Group II: MiniLab with PS Formulation
Research was done for Group II at nine different pharmacies in Europe using MiniLab with Formulation II again assessing mass variation. The study involved testing MiniLab equipment and using Formulation 2. They looked at dosing accuracy for three tablet weights (500 mg, 400 mg, and 300 mg) at each site.
Table 3 below summarizes the basic info about dosing accuracy for different tablet weights in Group II. The total number of tablets manufactured was 1296.
This data shows the number of observations, range of values, average accuracy, and variability for each tablet weight. The study found that, on average, the printing accuracy for 500 mg tablets was 95.8%, indicating a high level of accuracy with little variation. Similarly, the average accuracies for 400 mg and 300 mg tablets were 91.2% and 90.7%, respectively. The variability shows how much the accuracy differs within each tablet weight category.
4.1.3. Group III: Pharma Printer with PS Formulation
For Group III, the research examined the precision of tablet deposition across various weights: 500 mg, 400 mg, 300 mg, and 200 mg. Here are the key statistics presented in table 4. The total number of tablets printed in this phase was 2112.
Table 4.
descriptive statistics of average deposition accuracy across different tablet weights in group III.
Table 4.
descriptive statistics of average deposition accuracy across different tablet weights in group III.
| Tablet Weight |
N |
Minimum (%) |
Maximum (%) |
Mean (%) |
Std. Deviation |
| 500 mg |
11 |
93.8 |
100.0 |
98.5 |
2.1 |
| 400 mg |
11 |
95.8 |
100.0 |
98.9 |
1.7 |
| 300 mg |
11 |
93.8 |
100.0 |
98.5 |
2.1 |
| 200 mg |
11 |
95.8 |
100.0 |
99.1 |
1.4 |
The precision of dosing ranged from 93.8% to 100.0%, with mean accuracies between 98.5% and 99.1%. Standard deviations indicate the level of variability in dosing precision within each weight category, which remained relatively low. These results offer valuable insights into the dosing performance of the Pharma Printer with formulation incorporating PS (Form II) across different tablet weights. The observed desposition accuracies highlight the reliability of the equipment across the 11 compounding pharmacy sites, contributing to a comprehensive understanding of its effectiveness in pharmaceutical manufacturing processes. The addition of the 200 mg tablets as a new feature further enhanced the capabilities of the Pharma Printer. In this step tablets were dosed directly into a blister pack as shown in
Figure 7.
The results from phases 3 and 4 when employing the Pharma Printer indicate a rapid deposition process. The time to deposit a tablet is around 1-3 seconds per dose including the weight measurement with a scale which makes it a very rapid approach for compounding and allowing 100% weight control of each dose. Bendicho-Lavilla [
18] reported about a 3D printing approach recently in which in-process weight control was employed. In the study the most rapid time for one individual tablet printing plus weighing was 50 s per a 250 mg tablet and 75 s for a 750 mg tablet. In comparison the smallest (200mg) tablet manufacture with weight control took 1 s with the Pharma Printer making it 50 times faster than the reported approach. Previously, Lafeber et al [
21] reported about a study with 3D printed sildenafil and furosemide tablets where the manufacturing method produces tablets with a speed of approximately 1-1.5 tablets per minute i.e. up to 100 tablets per hour without any in process weight control. As a comparison the 3D printing approaches reported produce roughly 1-1.5 tablets per minute and the automated extrusion-based dosing technology introduced in this study produces 30-60 quality controlled tablets per minute.
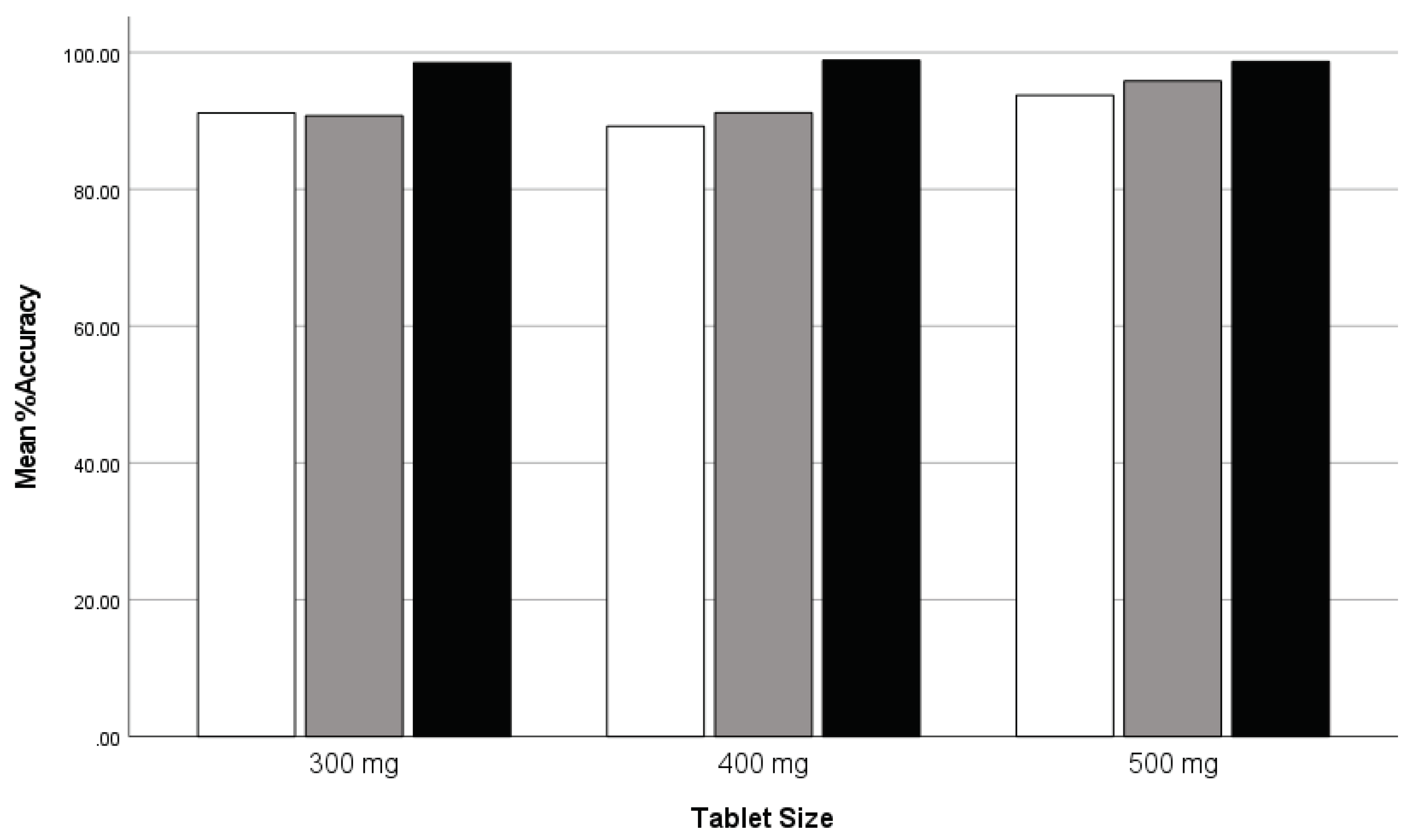
4.1.4. Comparison of Printing Accuracy Across Multiple Tablet Sizes
This study investigated the mean accuracy scores across three different tablet sizes (300 mg, 400 mg, and 500 mg) among three distinct groups. Groups I and II utilized the MiniLab printer, while Group III employed the latest version of the Pharma printer. The analysis aimed to assess the impact of printer performance on dosing accuracy with mass variation, particularly focusing on dosing accuracy. In total 3744 tablets were manufactured in the MinLab group and 2112 tablets in the Pharma Printer group.
ANOVA results revealed significant differences in mean accuracy scores among the groups for all tablet sizes (Accuracy300: F = 7.838, p = .002; Accuracy400: F = 7.860, p = .002; Accuracy500: F = 3.951, p = .029), suggesting variations in accuracy performance. Robust tests of equality of the mean values further supported these findings, highlighting significant differences in accuracy performance among the groups (p < .001 for both Accuracy300 and Accuracy400; p < .001 for Accuracy500). Post hoc tests indicated that Group 3 consistently demonstrated significantly higher mean accuracy scores compared to Groups I and II across all tablet sizes. These findings suggest that the utilization of the most advanced version of technology, i.e. the Pharma Printer in Group III led to superior accuracy performance in dosing compared to the MiniLab printers used by Groups I and II. The study reveals that dosing accuracy with mass variation is indeed dependent on printer performance. The utilization of the latest version of the Pharma Printer in Group III resulted in superior accuracy performance in dosing compared to the MiniLab printers used by Groups I and II. No comparison study was conducted for 200 mg tablets as data was available only for Group III using the Pharma printer.
Table 5.
Comparison of mean accuracy scores for tablet sizes (300 mg, 400 mg, and 500 mg) across three groups.
Table 5.
Comparison of mean accuracy scores for tablet sizes (300 mg, 400 mg, and 500 mg) across three groups.
| Tablet Size |
Comparison |
Mean Difference |
Standard Error |
Sig. |
95% Confidence Interval |
| 300 mg |
Group I vs. Group II |
0.43425 |
2.16209 |
.980 |
[-5.0999, 5.9684] |
| Group I vs. Group III |
7.30898* |
2.02951 |
.004 |
[2.1142, 12.5038] |
| Group II vs. Group III |
7.74323* |
2.35738 |
.009 |
[1.7092, 13.7773] |
| 400 mg |
Group I vs. Group II |
-1.98797 |
2.63780 |
.755 |
[-8.7398, 4.7639] |
| Group I vs. Group III |
9.64717* |
2.47605 |
.002 |
[3.3094, 15.9850] |
| Group II vs. Group III |
7.65919* |
2.87606 |
.040 |
[0.2975, 15.0209] |
| 500 mg |
Group I vs. Group II |
-2.54497 |
2.53453 |
.608 |
[-3.9425, 9.0325] |
| Group I vs. Group III |
4.92513 |
2.37911 |
.133 |
[-1.1645, 11.0148] |
| Group II vs. Group III |
-7.47010* |
2.76346 |
.037 |
[-14.5436, -0.3966] * |
Figure 8.
Clustered bar graph: comparison of mean weight accuracy across tablet sizes for three groups: group I (white), group II (gray), and group III (black).
Figure 8.
Clustered bar graph: comparison of mean weight accuracy across tablet sizes for three groups: group I (white), group II (gray), and group III (black).
4.3. Content Uniformity test by HPLC
The comprehensive analysis of content uniformity (CU) demonstrates consistent drug content across the varied tablet sizes. Each tablet size, from 200 mg to 500 mg, exhibits a narrow distribution of drug content, as evidenced by the low relative standard deviation (RSD%) values. This uniformity is crucial to ensure that each tablet delivers the intended dosage. The Acceptance Value (AV), a critical parameter indicating content uniformity, is well within acceptable limits for all tablet sizes. The AVG values, reflecting the average drug content, and SD values, indicative of variability, are also within established parameters, underscoring the uniformity of the formulation.
The content uniformity analysis establishes that the 1% propranolol HCl with 1% Polysorbate in CuraBlend® tablets maintain a uniform composition across different tablet sizes. The consistently low RSD%, AV, AVG, and SD values affirm the precision and reliability of the manufacturing process. This outcome reinforces the confidence in the quality and uniform drug delivery capabilities of the tablets, contributing to the overall quality and safety of the pharmaceutical product.
4.3.1. Group I: MiniLab with Formulation I
For the content uniformity (%CU) analysis, each tablet size underwent testing with a sample size of 170. The %CU values varied, with the minimum and maximum percentages falling within acceptable ranges: 79.0% to 115.9% for 500 mg tablets, 79.5% to 118.1% for 400 mg tablets, and 84.6% to 111.9% for 300 mg tablets. The mean %CU values were 102.9% for 500 mg tablets, 103.5% for 400 mg tablets, and 103.1% for 300 mg tablets. While the mean values for all tablet sizes are within the acceptance criteria, the standard deviations (7.7, 6.7, and 6.9, respectively) suggest some variability in content uniformity.
Table 6.
Content uniformity (CU) and acceptance value (AV) analysis for different tablet sizes - group I (MiniLab without PS formulation).
Table 6.
Content uniformity (CU) and acceptance value (AV) analysis for different tablet sizes - group I (MiniLab without PS formulation).
| Variable |
N |
Minimum (%) |
Maximum (%) |
Mean (%) |
Std. Deviation |
| %CU 500 mg |
170 |
79.0 |
115.9 |
102.9 |
7.7 |
| %CU 400 mg |
170 |
79.5 |
118.1 |
103.5 |
6.7 |
| %CU 300 mg |
170 |
84.6 |
111.9 |
103.1 |
6.9 |
| AV 500 mg |
17 |
2.0 |
27.0 |
16.5 |
6.2 |
| AV 400 mg |
17 |
9.0 |
26.0 |
16.8 |
4.7 |
| AV 300 mg |
17 |
5.0 |
26.0 |
16.6 |
6.1 |
The acceptance value data shows variability within the content uniformity (CU) test across different tablet sizes. Across all tablet sizes (500 mg, 400 mg, and 300 mg), the mean AV values are relatively consistent, ranging from approximately 16.47 to 16.76. However, the ranges of AV values vary within each tablet size group, indicating some variability in content uniformity results.
4.3.2. Group II: MiniLab with Formulation II
The data illustrates CU results for tablet sizes of 500 mg, 400mg, and 300 mg, with additional insight into the AV measurements. Notably, these formulations incorporated polysorbate to enhance the mixing of the API with the formulation, aimed at improving CU results.
Across all tablet sizes, the mean %CU values align closely with the acceptable range of 85.0% to 115.0%. Specifically, the mean %CU values are as follows: 103.4% for 500 mg tablets, 103.4% for 400 mg tablets, and 103.1% for 300 mg tablets. The standard deviations for %CU are relatively low, ranging from approximately 3.9 to 4.9, indicating consistent content uniformity results within each tablet size group.
Furthermore, the AV data provides insight into the variability within the CU test. For the 500 mg tablets, the AV ranges from 2.0 to 17.0, with a mean value of 11.4 and a standard deviation of 5.4. Similarly, for the 400 mg tablets, the AV ranges from 6.0 to 15.0, with a mean value of 10.8 and a standard deviation of 3.3. Additionally, for the 300 mg tablets, the AV ranges from 8.0 to 19.0, with a mean value of 13.5 and a standard deviation of 3.2. These findings suggest variability in content uniformity results across different tablet sizes, potentially influenced by the incorporation of polysorbate to enhance mixing.
Table 7.
Content uniformity (CU) and acceptance value (AV) analysis for different tablet sizes - group II (MiniLab with PS formulation).
Table 7.
Content uniformity (CU) and acceptance value (AV) analysis for different tablet sizes - group II (MiniLab with PS formulation).
| Variable |
N |
Minimum (%) |
Maximum (%) |
Mean (%) |
Std. Deviation |
| %CU 500 mg |
80 |
85.6 |
111.3 |
103.4 |
4.7 |
| %CU 400 mg |
80 |
89.7 |
107.8 |
103.4 |
3.9 |
| %CU 300 mg |
80 |
87.8 |
111.3 |
103.1 |
4.9 |
| AV 500 mg |
8 |
2.0 |
17.0 |
11.4 |
5.4 |
| AV 400 mg |
8 |
6.0 |
15.0 |
10.8 |
3.3 |
| AV 300 mg |
8 |
8.0 |
19.0 |
13.5 |
3.2 |
4.3.3. Group III: Pharma Printer with Formulation II
The data for group 3 presents content uniformity (CU) results for tablet sizes of 500 mg, 400 mg, 300 mg, and 200 mg, along with corresponding Acceptance Value (AV) measurements (
Table 8). It is important to note that the formulations for group 3 are consistent with those of group 2, utilizing a Pharma Printer for production.
Across all tablet sizes, the mean %CU values fall within the acceptable range of 85.0% to 115.0%. Specifically, the mean %CU values are as follows: 103.2% for 500 mg tablets, 103.5% for 400 mg tablets, 103.2% for 300 mg tablets, and 104.3% for 200 mg tablets. The standard deviations for %CU vary slightly, ranging from approximately 3.4 to 3.9, indicating consistent content uniformity results within each tablet size group.
Additionally, the AV data provides insight into the variability within the CU test. For the 500 mg tablets, the AV ranges from 3.0 to 14.0, with a mean value of 8.2 and a standard deviation of 4.2. Similarly, for the 400 mg tablets, the AV ranges from 3.0 to 16.0, with a mean value of 8.6 and a standard deviation of 5.0. Furthermore, for the 300 mg tablets, the AV ranges from 3.00 to 16.0, with a mean value of 9.2 and a standard deviation of 5.6. Finally, for the 200 mg tablets, the AV ranges from 4.0 to 15.0, with a mean value of 8.8 and a standard deviation of 4.2.
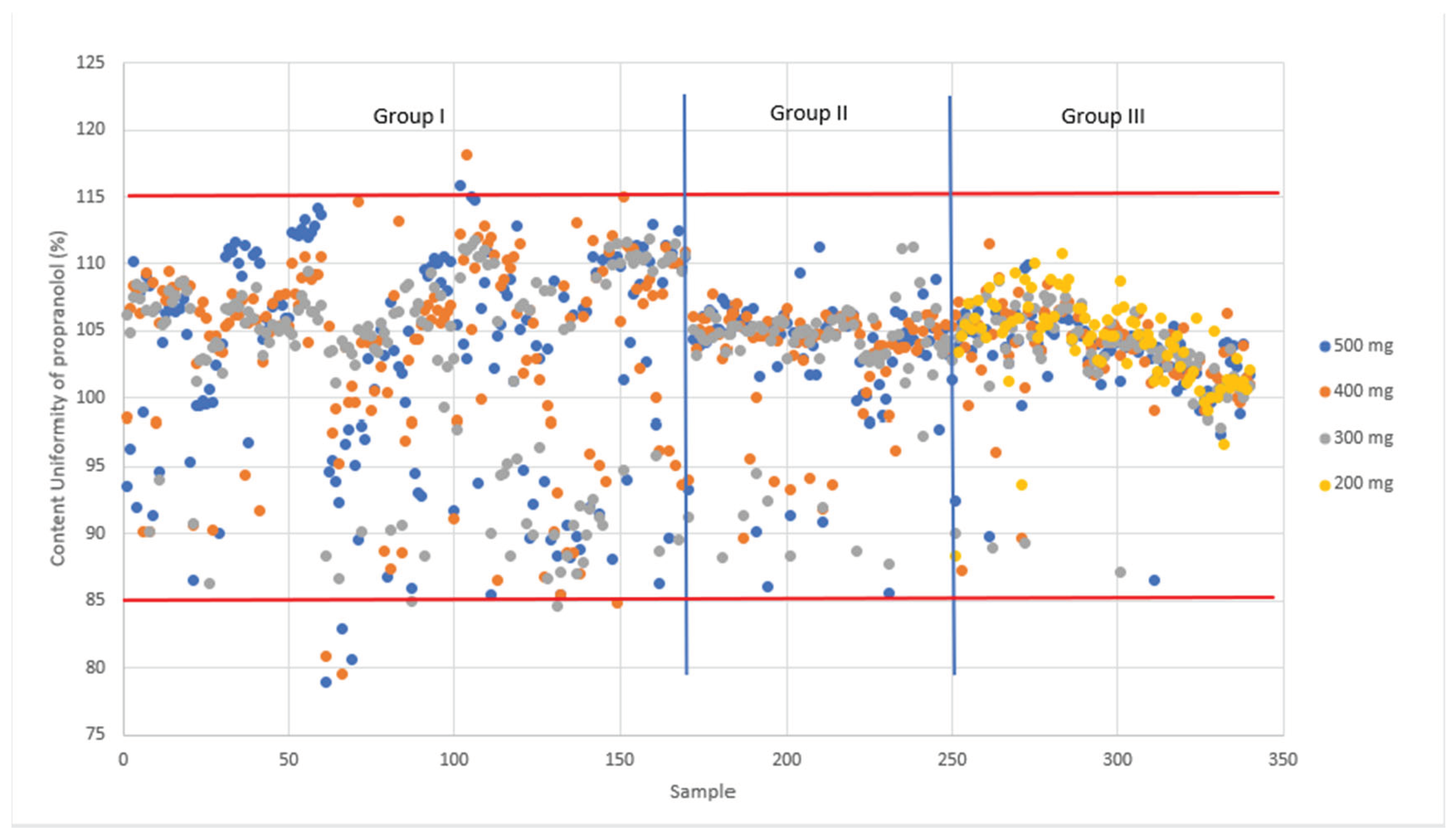
Figure 9 nicely shows the tighter CU values as the study progresses from Phase 1 to Phase 3. The spread in the results is much larger in phase 1 and 2 compared phase 3.
4.3.3. Comparison of Acceptance Value of content uniformity test Across Multiple Tablet Sizes: Between Group I, II and III
This study investigated the mean Acceptance Value (AV) across three distinct tablet sizes (300 mg, 400 mg, and 500 mg) among three groups, each employing different formulations and printers. Group I utilized formulations without polysorbate and the MiniLab printer, while Groups II and III employed formulations with polysorbate, with Group III additionally utilizing the latest Pharma Printer. The analysis aimed to discern the impact of polysorbate inclusion in formulations and printer type on AV with content uniformity, specifically focusing on AV of content uniformity test.
ANOVA outcomes unveiled noteworthy variances in mean AV among the groups for all tablet sizes (AV300: F = 5.483, p = .009; AV400: F = 11.223, p < .001; AV500: F = 6.834, p = .003), indicating differences in AV performance. Robust tests of equality of means further validated these findings, underscoring significant disparities in AV performance among the groups (p = .024 for AV300, p = .001 for AV400, and p = .004 for AV500).
Post hoc tests revealed that Group III consistently exhibited notably lower mean AV compared to Groups I and II across all tablet sizes. These findings suggest that the utilization of polysorbate-containing formulations and the latest Pharma printer in Group III led to superior AV performance compared to the MiniLab printers used by Groups I and II with similar formulations. This highlights the combined influence of formulation composition and printer performance on AV measurements with content uniformity, with the Pharma printer demonstrating enhanced accuracy in dosing.
The study underscores the interplay of both polysorbate inclusion in formulations and printer type on AV with content uniformity. The utilization of polysorbate-containing formulations and the Pharma Printer in Group III resulted in superior AV performance compared to Groups I and II. These findings underscore the importance of optimizing both formulation composition and printer type for improved accuracy in dosing pharmaceutical products.
Table 9.
Comparison of mean acceptance value (AV) for tablet sizes (300 mg, 400 mg, and 500 mg) across three groups.
Table 9.
Comparison of mean acceptance value (AV) for tablet sizes (300 mg, 400 mg, and 500 mg) across three groups.
| Tablet Size |
Comparison |
Mean Difference (Group 1 vs. Group 2) |
Mean Difference (Group 1 vs. Group 3) |
Mean Difference (Group 2 vs. Group 3) |
Standard Error |
p-value (Group 1 vs. Group 2) |
p-value (Group 1 vs. Group 3) |
p-value (Group 2 vs. Group 3) |
Significant at 0.05 Level |
| 300 mg |
Group I vs. Group II |
2.2 |
6.2 |
4.2 |
1.4 |
0.016 |
0.001 |
0.065 |
Yes |
| Group I vs. Group III |
7.3 |
- |
5.2 |
1.2 |
0.003 |
- |
0.029 |
Yes |
| Group II vs. Group III |
5.1 |
- |
- |
1.8 |
0.022 |
- |
- |
No |
| 400 mg |
Group I vs. Group II |
5.0 |
7.9 |
2.9 |
1.6 |
0.081 |
0.003 |
0.159 |
No |
| Group I vs. Group III |
9.0 |
- |
3.0 |
1.5 |
0.001 |
- |
0.067 |
Yes |
| Group II vs. Group III |
4.0 |
- |
- |
1.8 |
0.037 |
- |
- |
No |
| 500 mg |
Group I vs. Group II |
4.0 |
7.8 |
3.8 |
1.6 |
0.039 |
0.004 |
0.041 |
Yes |
| Group I vs. Group III |
7.8 |
- |
3.8 |
1.5 |
0.010 |
- |
0.043 |
Yes |
| Group II vs. Group III |
3.8 |
- |
- |
1.7 |
0.044 |
- |
- |
Yes |
No comparison study was conducted for 200 mg tablets, as data was only available for Group III utilizing the Pharma Printer.
4.3.4. Group IV: Pharma Printer with hardware updates and Formulation II
The data from Group IV as shown in
Table 10 showcases exceptional precision in dosing across different tablet sizes. This part of the study was done in the R&D laboratory of CurifyLabs. The Pharma Printer was employed with a more rigid syringe holder and dosing weight increments of 25 mg were introduced. The testing protocol with 3x 16 tablets for 200, 300, 400 and 500 mg targets weight was tested, and dosing was also demonstrated with target weights of 225, 275 and 425 mg respectively, which are three randomly chosen the targets weights in between the fixed values in the protocol. All in all, 336 tablets were dosed in this phase. The mean values for mass variation (MV) demonstrate consistent accuracy, with minimal deviations observed for all. The accuracy as defined per Ph. Eur. limits for the tablets in all target weight classes was 100%. For the 225 mg tablets the mass variation ranges from 218.0 to 230.0, with a mean of 224.5 and a standard deviation of 2.2. Similarly, for the 275 mg tablets, the mass variation remains tightly clustered, ranging from 264.0 to 281.0, with a mean of 274.0 mg and a standard deviation of 3.1. The 425 mg tablets also exhibit precision in dosing, with values concentrated around the mean of 423.6 and a standard deviation of 4.3.
These findings highlight the effectiveness of the Pharma printer's new syringe attachment and dose in ensuring precise control of the dosing. The narrow ranges and minimal deviations in mass variation across all tablet sizes indicate that the printer can accurately dispense doses within the specified ranges. It's worth noting that this test and the selection of tablet weights were based on a challenge test aimed at checking the printer's performance with specific tablet weights, such as 225 mg, 275 mg, and 425 mg. This level of accuracy is crucial in pharmaceutical manufacturing to guarantee that patients receive the intended dosage of medication consistently.
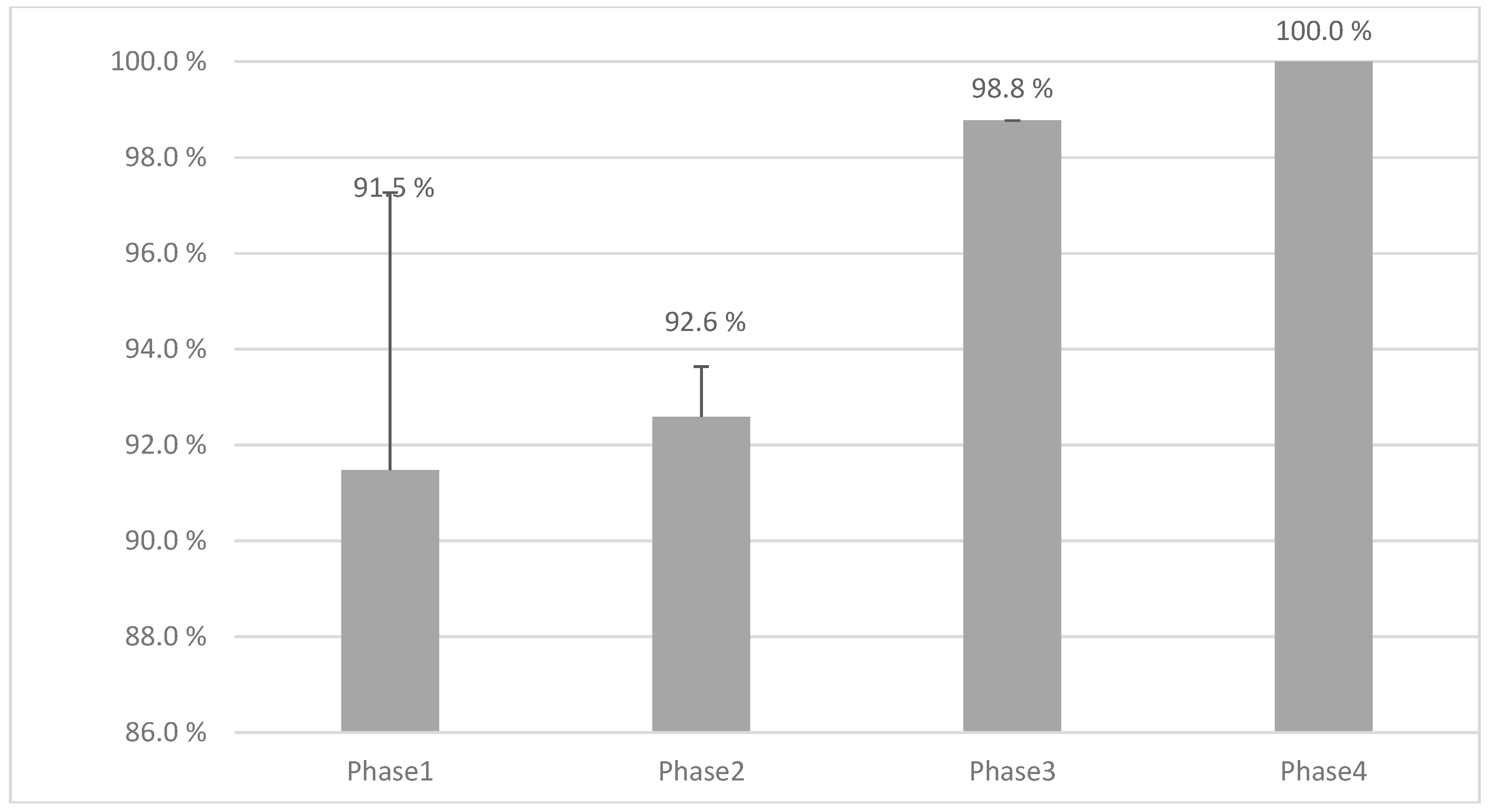
Figure 10 shows the dosing accuracy (%) and standard deviations across all phases of the study showing 100 % accuracy in the final step when all improvements have been implemented. Phase1 and 2 were performed with the MiniLab and Phases 3 and 4 with the Pharma Printer indicating a high dependency of the hardware features to the accuracy dosing weight.
The acceptance value, a critical parameter in pharmaceutical analysis denoting the uniformity of dosage units, was calculated to be 2. Coupled with a standard deviation of 0.85, these metrics offer a comprehensive understanding of the variability and consistency within the tablet batch. This finding underscores the robustness and precision of the manufacturing process, particularly with the implementation of hardware updates in the Pharma Printer. It reflects a diligent adherence to quality control measures, ensuring that each tablet within the batch delivers the intended dose with reliability and accuracy (
Table 11).
4.4. Blend Uniformity test by NIR Spectroscopy
In earlier studies NIR spectroscopy has been used for instance for quantitative measurement of accurate dosing of theophylline [
22], levothyroxine and prednisolone using inkjet printing [
23]. Recently Seani-Viano et al. [
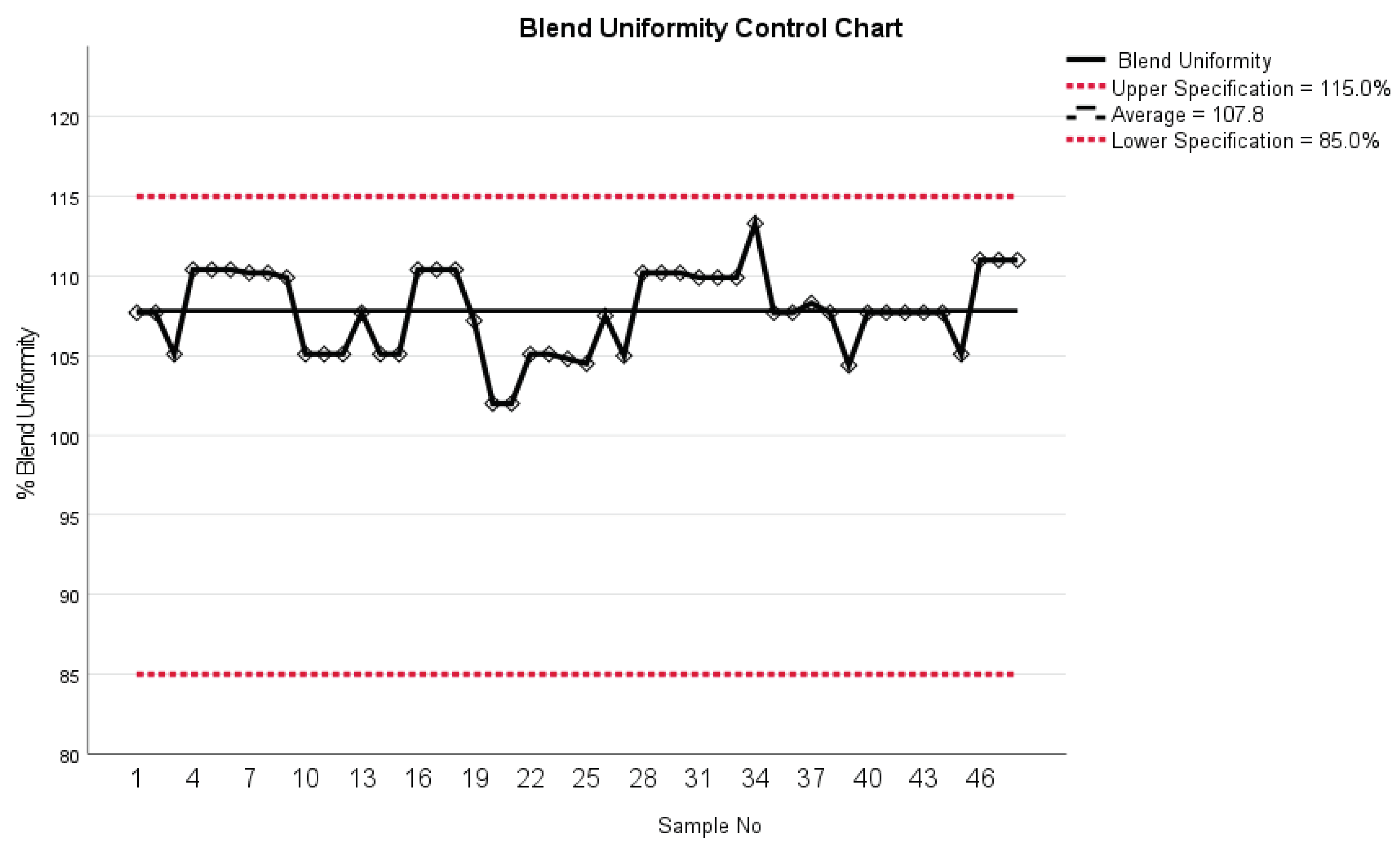
24] reported about a case study of 3D printed efavirenz tablets and successful real time NIR measurements of them. Blend uniformity results indicated a mean of 107.8% with a standard deviation of 2.6% (
Figure 11). This data falls within the specified range of 90 to 110%, aligning with the internal specification for blend uniformity. Thus, it meets the acceptance criteria for content uniformity, which typically ranges from 85% to 115%.
However, it's important to note that some values slightly exceed the upper limit of 110%. This may be attributed to inadequate mixing during the mixing process, resulting in localized areas with higher concentrations of the active ingredient. While these outliers are minimal, they underscore the significance of thorough blending to ensure homogeneity across the entire batch. Furthermore, the ongoing improvement of the Partial Least Squares (PLS) model for blend uniformity remains a continuous process. This involves refining the model-building process to enhance its accuracy and predictive capabilities. The CU and AV values form the HPLC analyses confirm the high quality of blend uniformity. It is important to understand that the NIR analysis utilizes a very small sample amount approximately 25-50 mg of sample.
4.6. Stability Study
In the stability study, the appearance of the tablets from both Formulation I and Formulation II was meticulously evaluated over the initial three-month period. The tablets maintained their anticipated off-white, soft chewable form with a vanilla flavour, confirming adherence to the specified characteristics. This visual conformity underscores the stability of the formulations in preserving their physical attributes throughout the assessment duration.
Assessing the drug potency through the assay results revealed a consistent performance for both formulations. Formulation II exhibited values of 105.5% at zero months, 107.6% at the first month, and 105.7% at the third month. The observed values within the assessed period consistently fell within the acceptance range of 90.0-110.0%. This indicates that both formulations maintain their specified potency limits, ensuring the sustained efficacy of the medication.
Drawing conclusions from the stability data, it can be asserted that the 1% Propranolol HCl with 1% Polysorbate in CuraBlend® tablets, represented by both Formulation I and Formulation II, exhibits stability for at least three months under the recommended storage conditions (Room temperature). The confirmation of appearance conformity and consistent drug potency over time provides a robust foundation for the formulations' reliability and potential for long-term use.
Examining the specific attributes of each formulation, Formulation II showcased conformity in appearance throughout the three-month period, with pH values consistently within the range of 4.9 to 5.0. In contrast, Formulation I maintained its appearance and exhibited pH values within the specified range of 4.5 to 5.5. However, the assay results for Formulation I indicated a gradual decrease in drug potency, with values declining from 95.3% at zero months to 92.3% at the third month.
Table 11 provides a concise overview of the stability study results for both Formulation I and Formulation II, encompassing appearance, pH values, and drug potency at zero, first, and third months.
Table 12.
Stability report for tablets from formulation I and formulation II. An overview of the stability study results for both formulation I and formulation II, encompassing appearance, pH values, and drug potency at zero, first, and third months.
Table 12.
Stability report for tablets from formulation I and formulation II. An overview of the stability study results for both formulation I and formulation II, encompassing appearance, pH values, and drug potency at zero, first, and third months.
| Test |
Specification |
Zero Month |
First Month |
Third Month |
| Formulation I |
| Appearance |
Off-white, soft chewable tablet with vanilla flavor |
Conforms |
Conforms |
Conforms |
| pH |
4.5-5.5 |
5.0 |
5.0 |
5.0 |
| Assay |
90.0-110.0% |
95.3% |
95.2% |
92.3% |
| Formulation II |
| Appearance |
Off-white, soft chewable tablet with vanilla flavor |
Conforms |
Conforms |
Conforms |
| pH |
4.5-5.5 |
4.9 |
4.9 |
5.0 |
| Assay |
90.0-110.0% |
105.5% |
107.6% |
105.7% |
4.9. General Considerations
For streamlining processes using automated technology for compounding purposes the is a need to have ready-made excipient bases available that can be utilized in a versatile way in extemporaneous drug manufacturing. If pharmacies need to develop these themselves a considerable amount of resources need to be put on this and this is not typically possible in compounding pharmacies or hospital pharmacies due to lack of resources. It is of course essential that these excipient bases or pharma inks are of the highest quality and are GMP manufactured. Moreover, the suitability for paeditaric use needs to be assured. Van Kampen et al. [
26] wrote brief overview of carrier materials currently used in pharmaceutical extrusion-based printing studies of medicines for pediatrics and they elaborated on a way how to guide in carrier material selection in this type of applications [
27,
28,
29,
30]. Many studies have addressed the importance of paediatric considerations and the STEP (Safety and Toxicity of Excipients for Paediatrics) Database [
31] and FDAs equivalent database [
32] are relevant sources in the context.
As we delve deeper into the digital transformation of manufacturing, leveraging cutting-edge technologies like artificial intelligence (AI) and the Internet of Things (IoT) becomes increasingly central. These advancements are paving the way for highly personalized and automated production methods across both public and private sectors. Within this landscape, three-dimensional (3D) printing emerges as an intriguing innovation, especially in crafting complex, tailored products with new functionalities [
33,
34,
35,
36,
37,
38,
39].
The application of 3D printing in the pharmaceutical sector, specifically in drug compounding, can replace current manual methods when speed, accuracy, quality control and user friendliness are fulfilled. While research into 3D-printed pharmaceuticals has been extensive, aiming to transform traditional manufacturing processes, the technology's role in personalizing drug delivery systems is not without challenges. The concept of tailoring medication to individual patient needs through 3D printing—adjusting drug shapes, sizes, and dosages, or adding specific release functionalities—remains complex and not fully realized. The practicality of implementing such personalized production on a wider scale, especially for standard medication compounding, reveals the technology's constraints in terms of efficiency and adaptability.
However, while 3D printing offers remarkable possibilities for creating tailored drugs for individual needs, it is an efficient solution only when speed and ease of use are addressed. Straightforward automated extrusion-based material deposition technologies and GMP manufactured excipient bases as presented in this multisite study offer a more viable alternative for rapid automated compounding. These type methods are precise and excel in speed, and the simplification of quality control processes, making them better suited for the fast-paced production of standard pharmaceuticals that are needed in normal compounding scenarios where the produced dosage forms should be similar in function (bioequivalent) as the market authorized products. This distinction underscores the need to choose the right manufacturing technology based on the specific requirements of drug production, whether seeking customization and complexity or efficiency, standardization and scalability in drug compounding.