Submitted:
29 February 2024
Posted:
29 February 2024
You are already at the latest version
Abstract
Keywords:
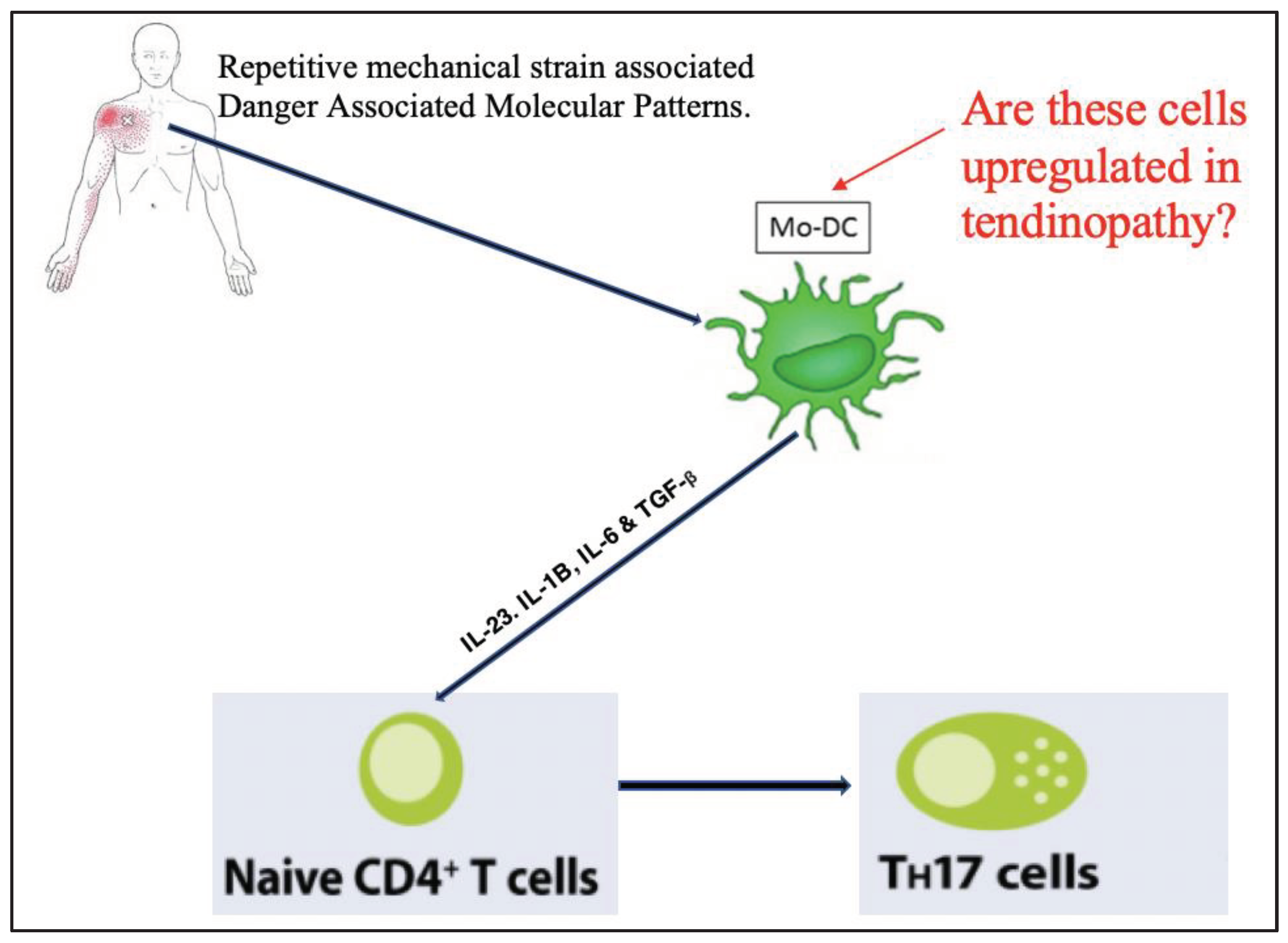
1. Introduction
2. Results
2.1. Patient Demographics
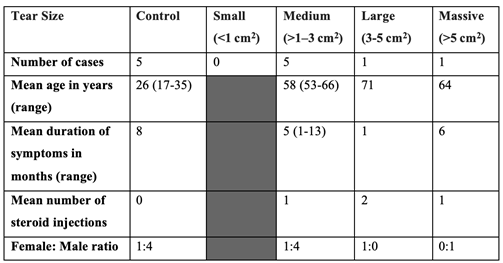 |
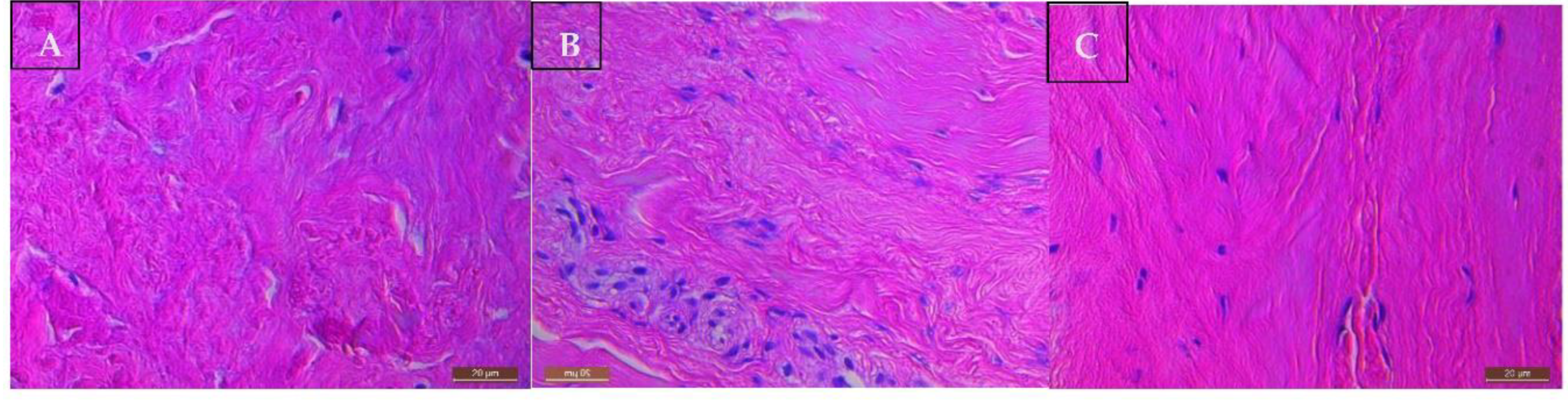
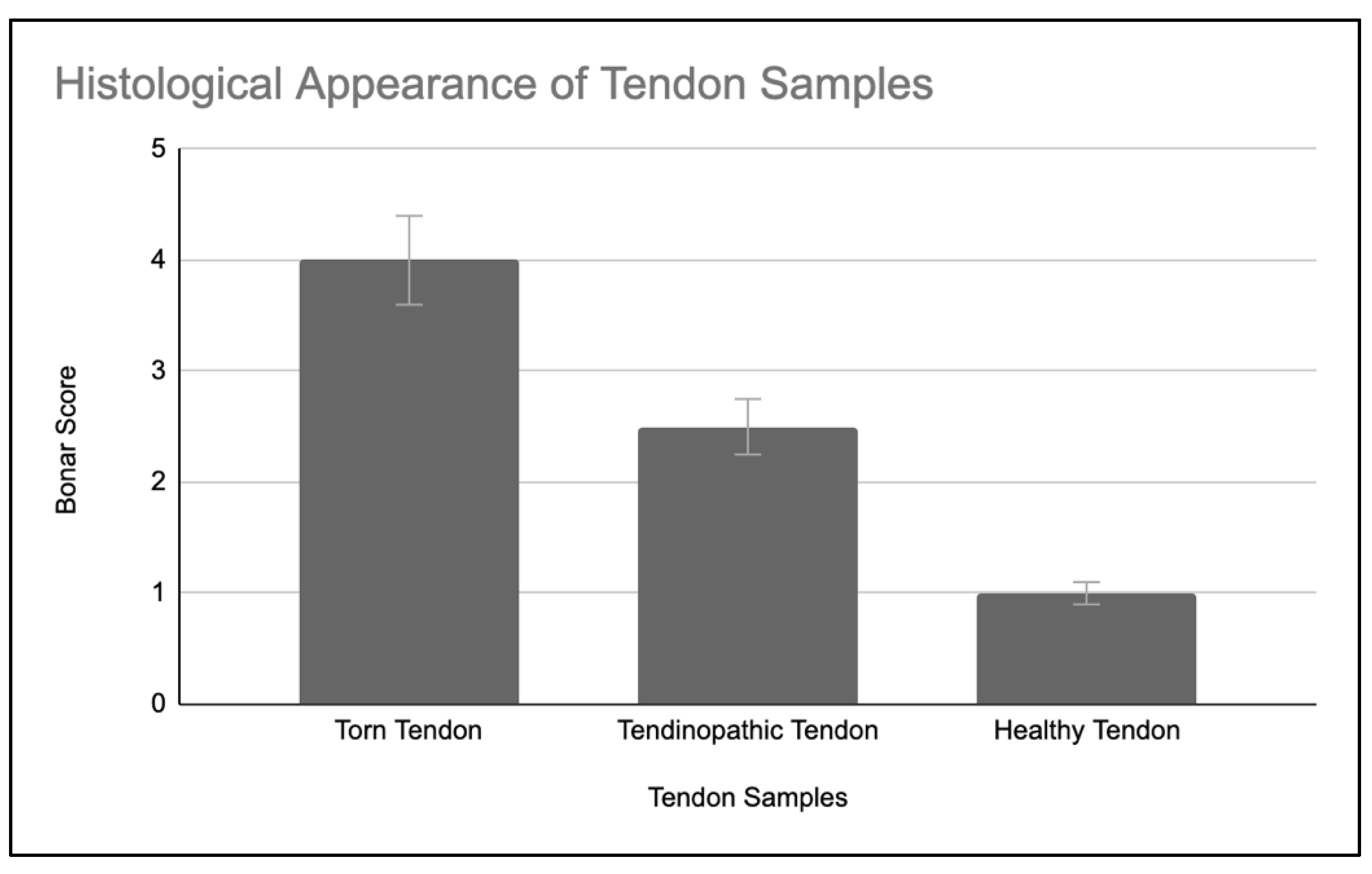
2.2. Histological Appearance
2.3. Inflammatory Cell Changes
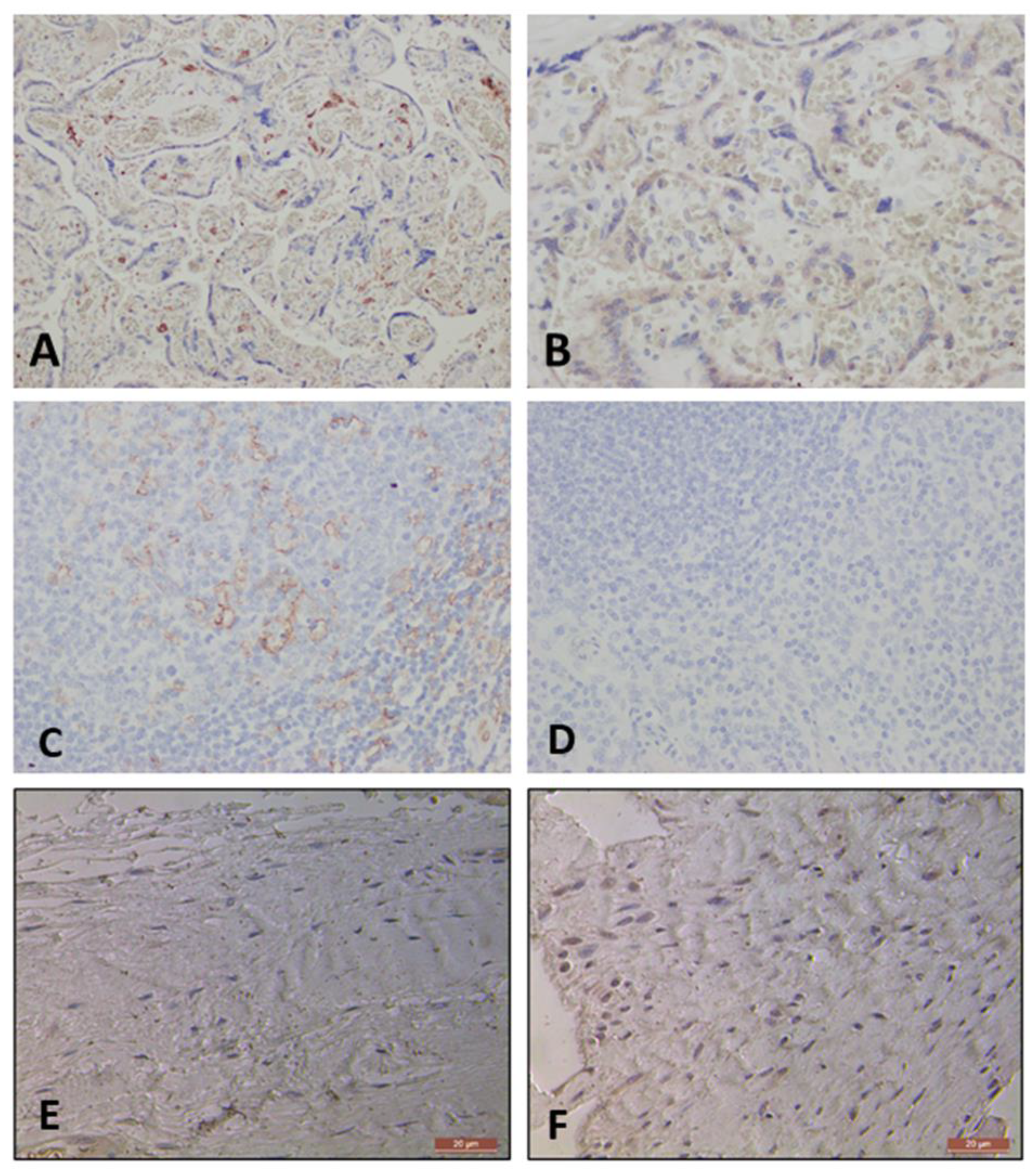
2.3.1. Testing Viability of Antibodies CD206 and CD11c
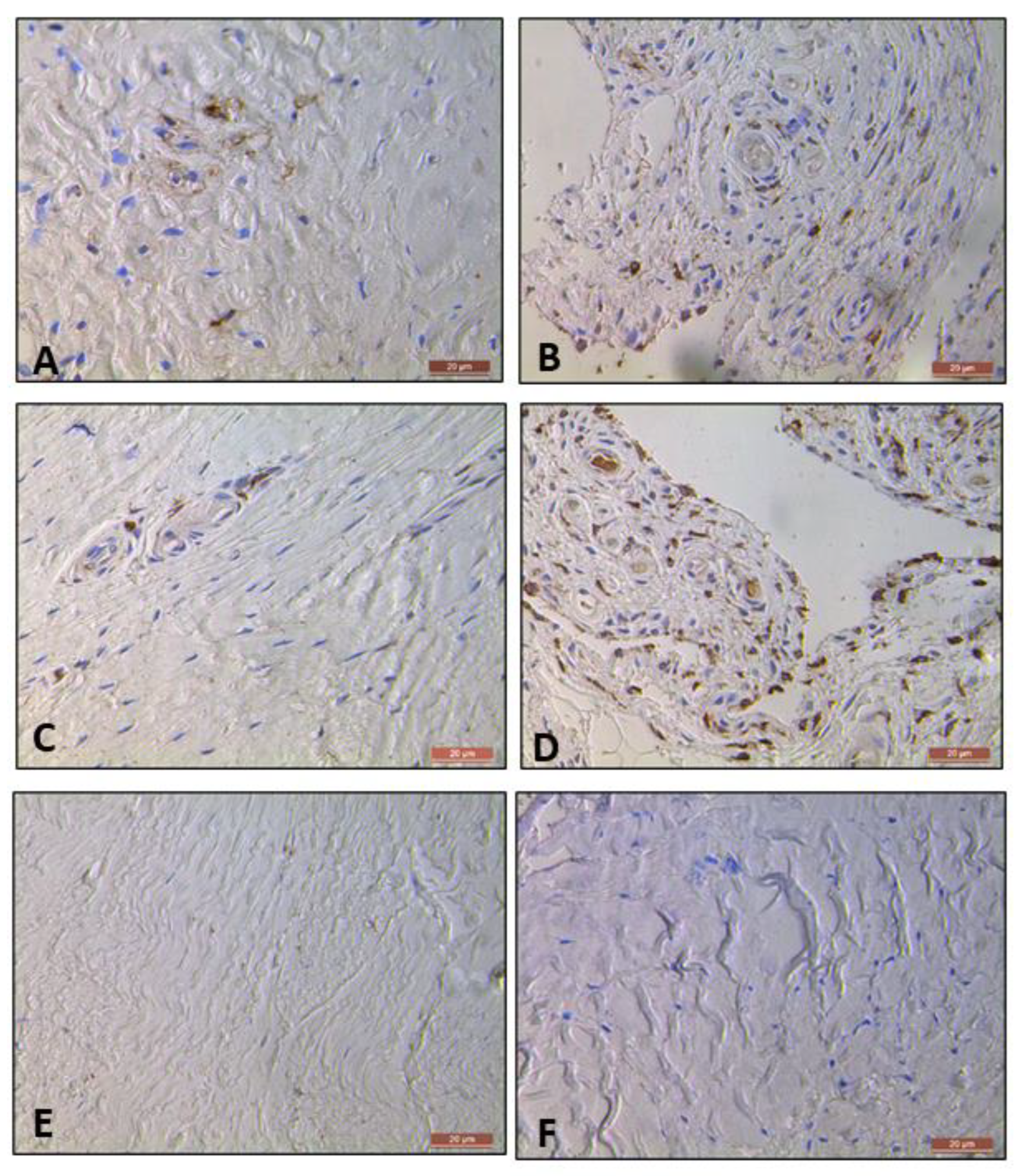
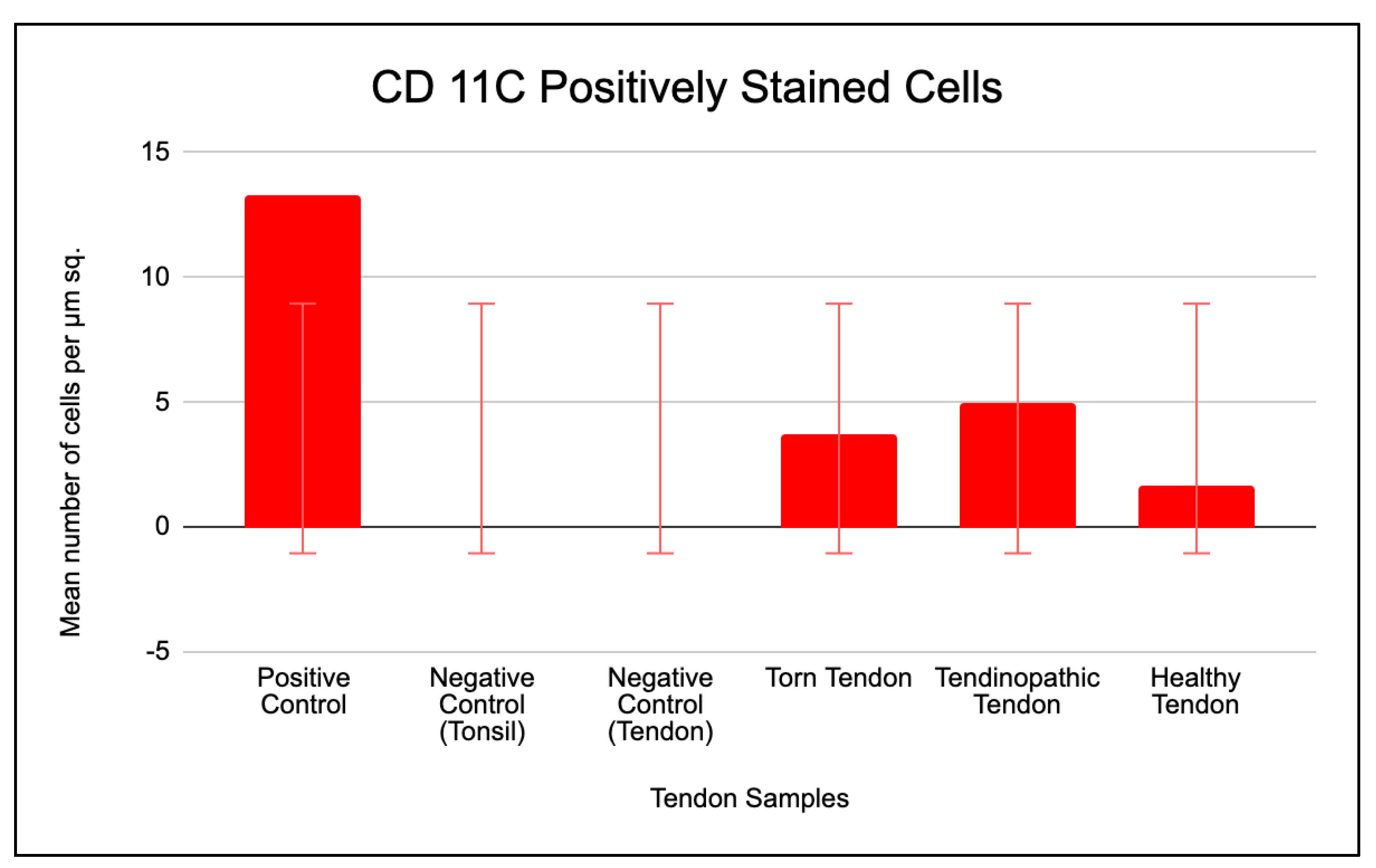
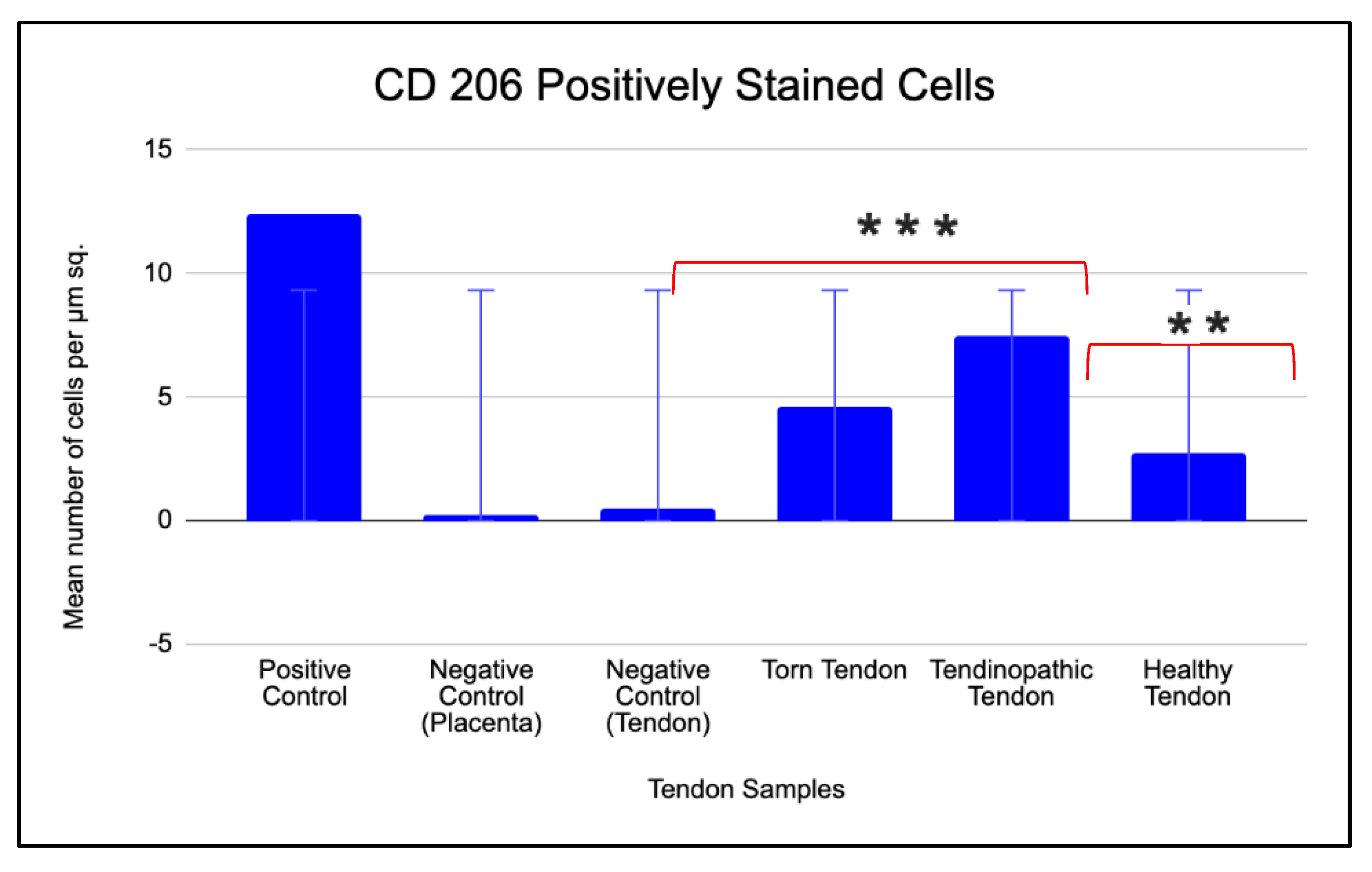
2.3.2. Prevalence of CD 206 and CD 11c Cells in Torn, Tendinopathic and Healthy Tendons
2.3.3. Comparison of CD 206 and CD 11C Cell Staining in Torn, Tendinopathic and Healthy Tendons
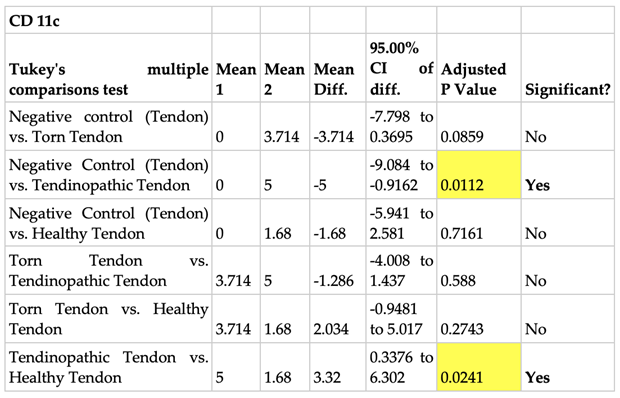 |
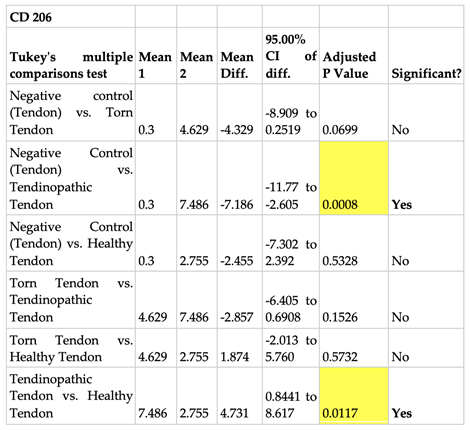 |
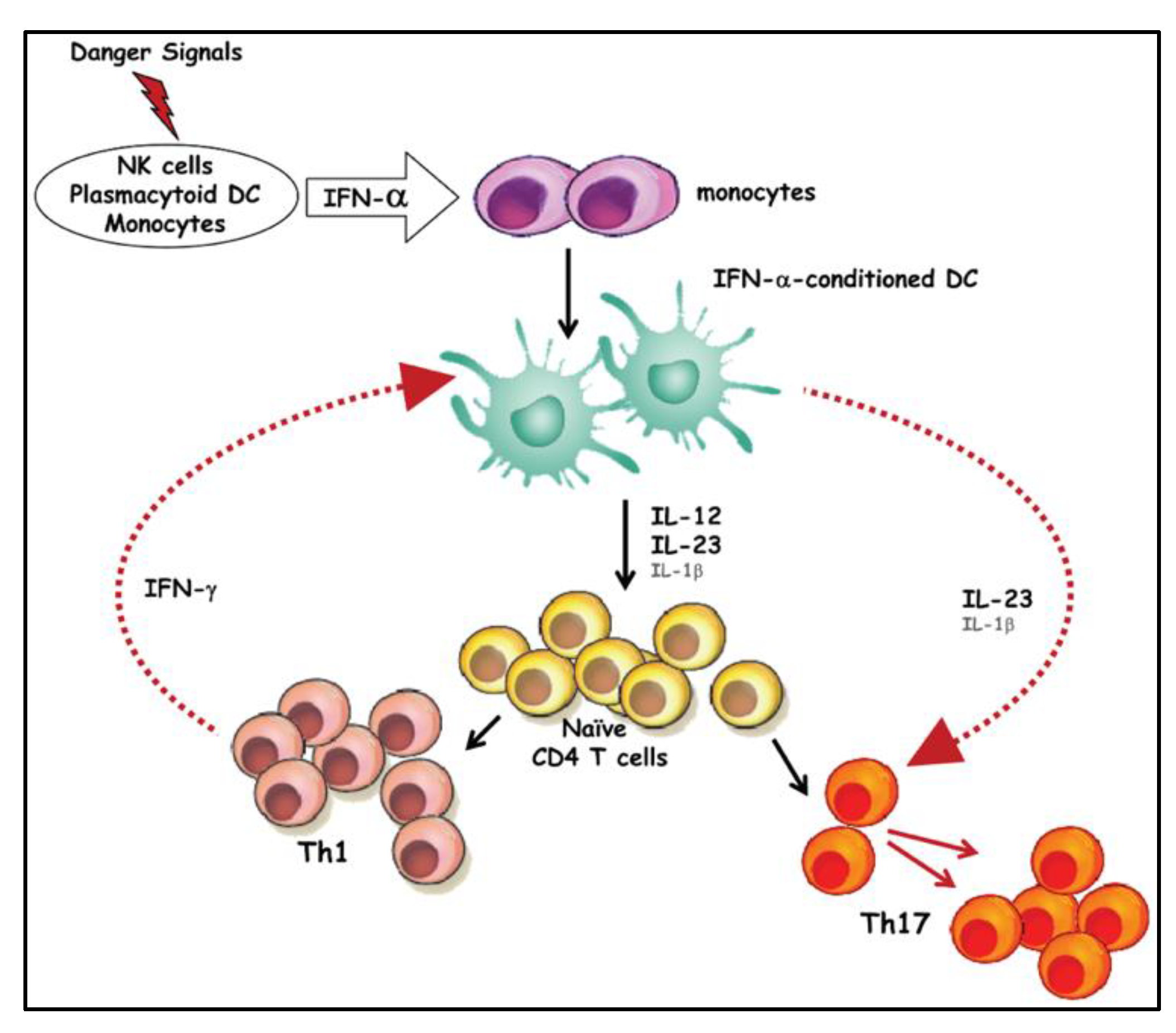
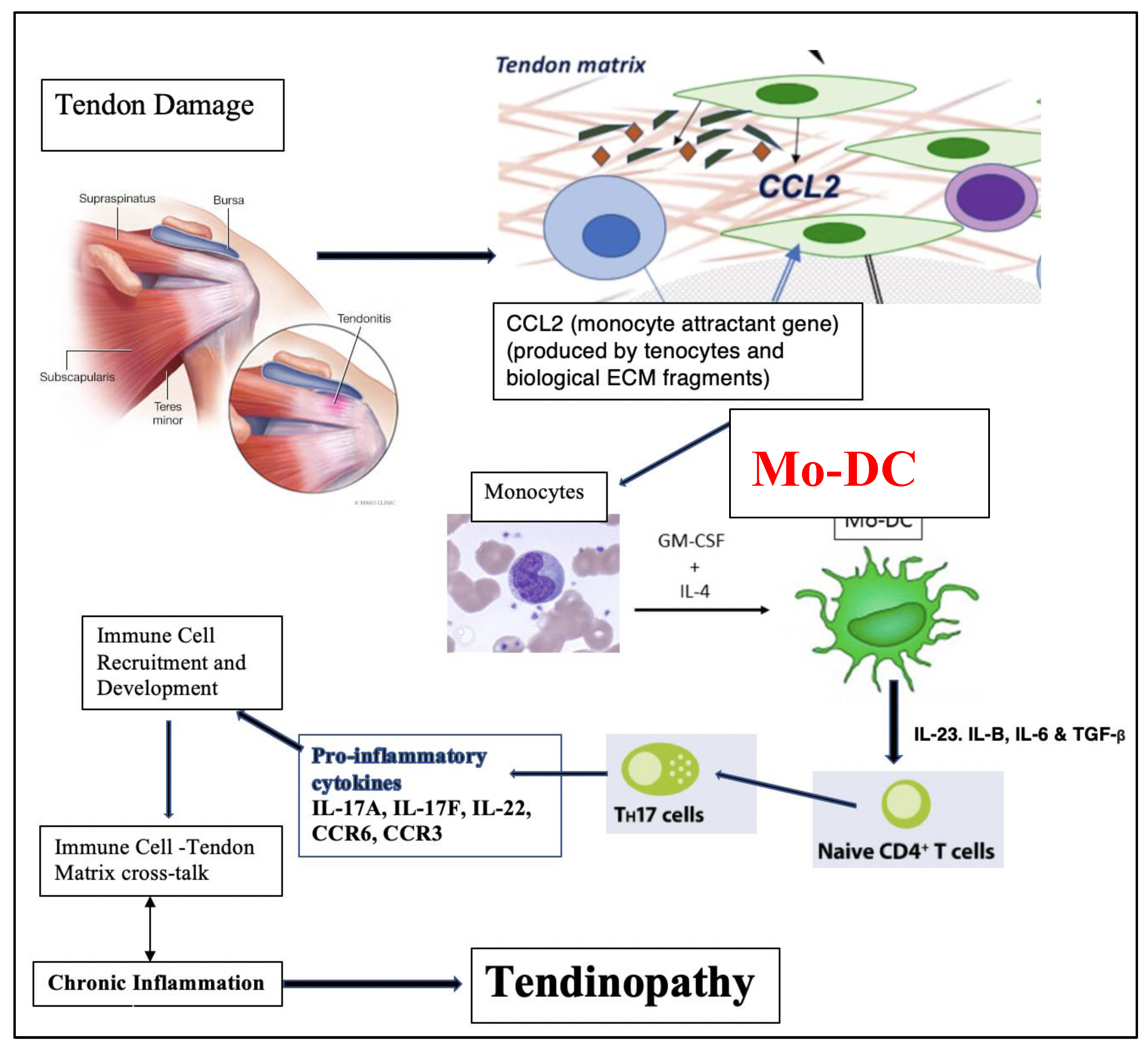
3. Discussion
4. Materials and Methods
Tissue Collection and Preparation
Histology and Immunohistochemistry Techniques
Control Staining
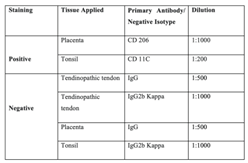 |
Image Analysis
Data Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Ackermann, P.W. and P. Renström, Tendinopathy in sport. Sports Health, 2012. 4(3): p. 193-201. [CrossRef]
- Maffulli, N., J. Wong, and L.C. Almekinders, Types and epidemiology of tendinopathy. Clin Sports Med, 2003. 22(4): p. 675-92. [CrossRef]
- Herring, S.A. and K.L. Nilson, Introduction to overuse injuries. Clin Sports Med, 1987. 6(2): p. 225-39. [CrossRef]
- Millar, N.L., G.A.C. Murrell, and I.B. McInnes, Inflammatory mechanisms in tendinopathy – towards translation. Nature Reviews Rheumatology, 2017. 13(2): p. 110-122. [CrossRef]
- Fredberg, U. and K. Stengaard-Pedersen, Chronic tendinopathy tissue pathology, pain mechanisms, and etiology with a special focus on inflammation. Scandinavian Journal of Medicine & Science in Sports, 2008. 18(1): p. 3-15.
- Riley, G., Tendinopathy--from basic science to treatment. Nat Clin Pract Rheumatol, 2008. 4(2): p. 82-9.
- Hopkins, C., et al., Critical review on the socio-economic impact of tendinopathy. Asia-Pacific journal of sports medicine, arthroscopy, rehabilitation and technology, 2016. 4: p. 9-20. [CrossRef]
- Zhang, J. and J.H. Wang, Mechanobiological response of tendon stem cells: implications of tendon homeostasis and pathogenesis of tendinopathy. J Orthop Res, 2010. 28(5): p. 639-43. [CrossRef]
- Abate, M., et al., Pathogenesis of tendinopathies: inflammation or degeneration? Arthritis research & therapy, 2009. 11(3): p. 235-235.
- Kannus, P. and L. Jozsa, Histopathological changes preceding spontaneous rupture of a tendon. A controlled study of 891 patients. J Bone Joint Surg Am, 1991. 73(10): p. 1507-25. [CrossRef]
- Kannus, P. and L. Józsa, Histopathological changes preceding spontaneous rupture of a tendon. A controlled study of 891 patients. JBJS, 1991. 73(10): p. 1507-1525. [CrossRef]
- Khan, K.M., et al., Histopathology of Common Tendinopathies. Sports Medicine, 1999. 27(6): p. 393-408. [CrossRef]
- Loiacono, et al., Tendinopathy: Pathophysiology, Therapeutic Options, and Role of Nutraceutics. A Narrative Literature Review. Medicina, 2019. 55(8): p. 447.
- Renström, P.A.a.W., S.L.-Y. , Tendinopathy: A Major Medical Problem in Sport, in Tendinopathy in Athletes. 2007. p. 1-9.
- Riley, G., The pathogenesis of tendinopathy. A molecular perspective. Rheumatology (Oxford), 2004. 43(2): p. 131-42. [CrossRef]
- Khan, K.M., et al., Histopathology of common tendinopathies. Update and implications for clinical management. Sports Med, 1999. 27(6): p. 393-408.
- Perry, S.M., et al., Inflammatory and angiogenic mRNA levels are altered in a supraspinatus tendon overuse animal model. Journal of Shoulder and Elbow Surgery, 2005. 14(1, Supplement): p. S79-S83. [CrossRef]
- Millar, N.L., et al., IL-17A mediates inflammatory and tissue remodelling events in early human tendinopathy. Scientific Reports, 2016. 6(1): p. 27149. [CrossRef]
- Jomaa, G., et al., A systematic review of inflammatory cells and markers in human tendinopathy. BMC Musculoskeletal Disorders, 2020. 21(1). [CrossRef]
- Millar, N.L., et al., Heat shock protein and apoptosis in supraspinatus tendinopathy. Clinical orthopaedics and related research, 2008. 466(7): p. 1569-1576. [CrossRef]
- Jomaa, G., et al., A systematic review of inflammatory cells and markers in human tendinopathy. BMC Musculoskeletal Disorders, 2020. 21(1): p. 78. [CrossRef]
- Stolk, M., et al., New insights into tenocyte-immune cell interplay in an in vitro model of inflammation. Scientific Reports, 2017. 7(1): p. 9801. [CrossRef]
- Giancola, R., et al., CD200 as a Potential New Player in Inflammation during Rotator Cuff Tendon Injury/Repair: An In Vitro Model. Int J Mol Sci, 2022. 23(23). [CrossRef]
- Millar, N.L., et al., Inflammation is Present in Early Human Tendinopathy. The American Journal of Sports Medicine, 2010. 38(10): p. 2085-2091. [CrossRef]
- Dakin, S.G., J. Dudhia, and R.K.W. Smith, Resolving an inflammatory concept: the importance of inflammation and resolution in tendinopathy. Veterinary immunology and immunopathology, 2014. 158(3-4): p. 121-127. [CrossRef]
- Dakin, S.G., et al., Chronic inflammation is a feature of Achilles tendinopathy and rupture. British Journal of Sports Medicine, 2018. 52(6): p. 359-367. [CrossRef]
- Millar, N.L., et al., Cytokines and apoptosis in supraspinatus tendinopathy. J Bone Joint Surg Br, 2009. 91(3): p. 417-24. [CrossRef]
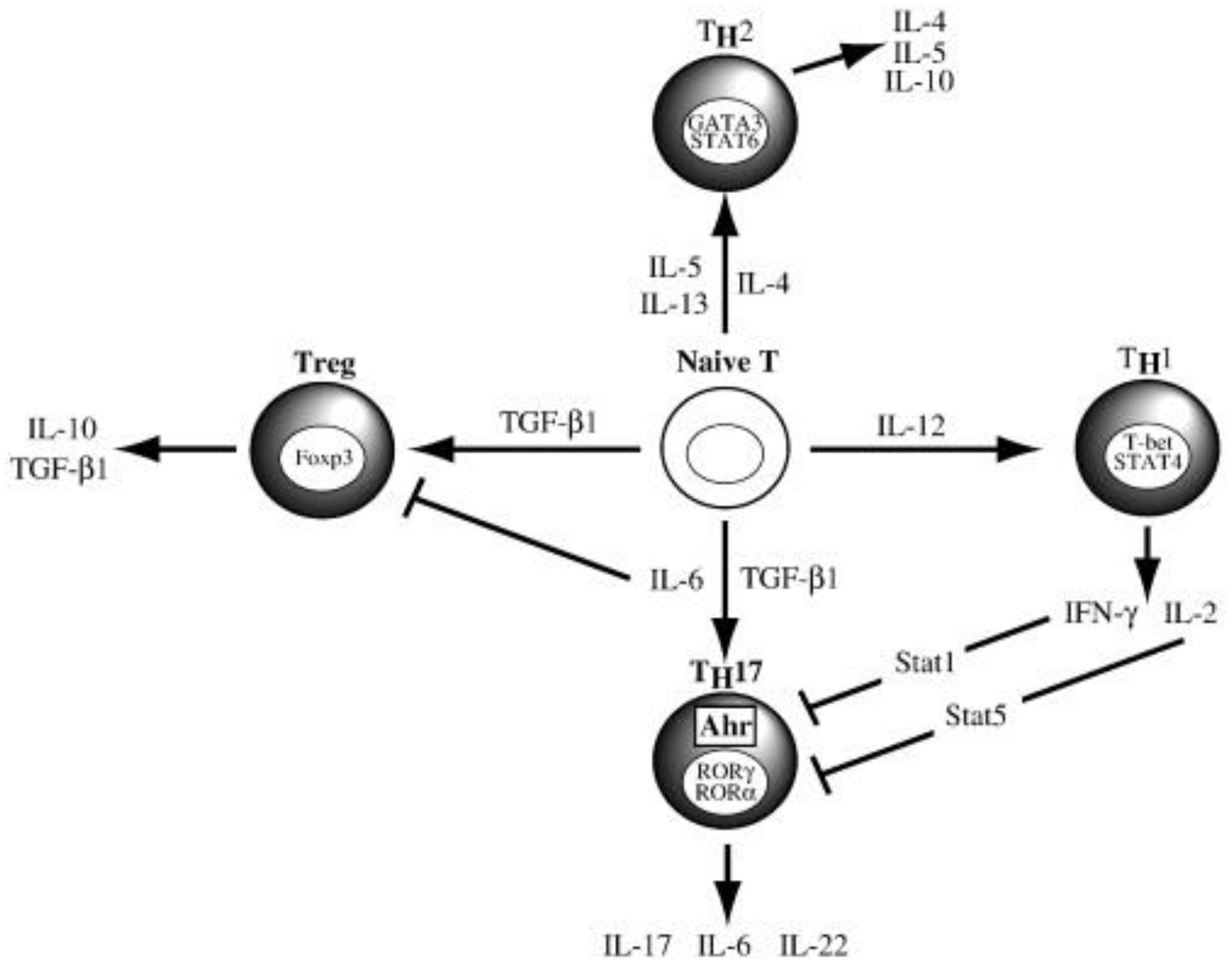
- Annunziato, F., et al., The phenotype of human Th17 cells and their precursors, the cytokines that mediate their differentiation and the role of Th17 cells in inflammation. Int Immunol, 2008. 20(11): p. 1361-8. [CrossRef]
- Estrada-Capetillo, L., et al., Induction of Th17 lymphocytes and Treg cells by monocyte-derived dendritic cells in patients with rheumatoid arthritis and systemic lupus erythematosus. Clinical & developmental immunology, 2013. 2013: p. 584303-584303. [CrossRef]
- Segura, E., et al., Human Inflammatory Dendritic Cells Induce Th17 Cell Differentiation. Immunity, 2013. 38(2): p. 336-348. [CrossRef]
- Cua, D.J. and C.M. Tato, Innate IL-17-producing cells: the sentinels of the immune system. Nature Reviews Immunology, 2010. 10(7): p. 479-489. [CrossRef]
- Kimura, A. and T. Kishimoto, Th17 cells in inflammation. International Immunopharmacology, 2011. 11(3): p. 319-322.
- Dhodapkar, K.M., et al., Dendritic cells mediate the induction of polyfunctional human IL17-producing cells (Th17-1 cells) enriched in the bone marrow of patients with myeloma. Blood, 2008. 112(7): p. 2878-2885. [CrossRef]
- Collin, M. and V. Bigley, Human dendritic cell subsets: an update. Immunology, 2018. 154(1): p. 3-20. [CrossRef]
- Persson, Emma K., et al., IRF4 Transcription-Factor-Dependent CD103+CD11b+ Dendritic Cells Drive Mucosal T Helper 17 Cell Differentiation. Immunity, 2013. 38(5): p. 958-969. [CrossRef]
- Bailey, S.L., et al., CNS myeloid DCs presenting endogenous myelin peptides 'preferentially' polarize CD4+ TH-17 cells in relapsing EAE. Nature Immunology, 2007. 8(2): p. 172-180. [CrossRef]
- Vaknin-Dembinsky, A., K. Balashov, and H.L. Weiner, IL-23 Is Increased in Dendritic Cells in Multiple Sclerosis and Down-Regulation of IL-23 by Antisense Oligos Increases Dendritic Cell IL-10 Production. The Journal of Immunology, 2006. 176(12): p. 7768-7774. [CrossRef]
- Farkas, Á. and L. Kemény, Monocyte-derived interferon-alpha primed dendritic cells in the pathogenesis of psoriasis: New pieces in the puzzle. International Immunopharmacology, 2012. 13(2): p. 215-218. [CrossRef]
- Siddiqui, K.R.R., S. Laffont, and F. Powrie, E-Cadherin Marks a Subset of Inflammatory Dendritic Cells that Promote T Cell-Mediated Colitis. Immunity, 2010. 32(4): p. 557-567. [CrossRef]
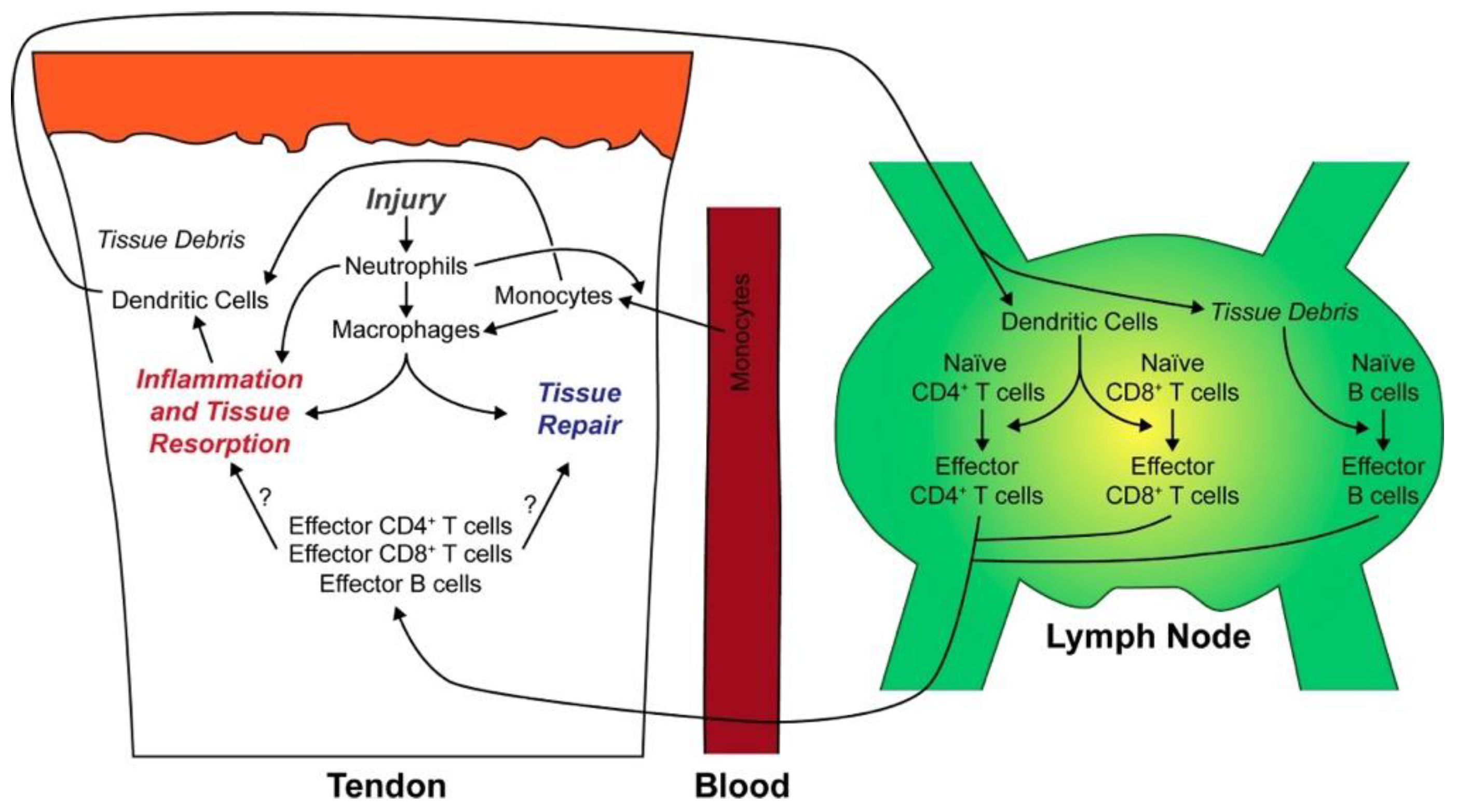
- Noah, A.C., et al., Adaptive and innate immune cell responses in tendons and lymph nodes after tendon injury and repair. Journal of Applied Physiology, 2020. 128(3): p. 473-482. [CrossRef]
- Crowe, L.A.N., et al., S100A8 & S100A9: Alarmin mediated inflammation in tendinopathy. Scientific reports, 2019. 9(1): p. 1463-1463. [CrossRef]
- Campbell, A.L., et al., IL-21 receptor expression in human tendinopathy. Mediators of inflammation, 2014. 2014: p. 481206-481206. [CrossRef]
- Murrell, G.A.C., Using nitric oxide to treat tendinopathy. British Journal of Sports Medicine, 2007. 41(4): p. 227-231. [CrossRef]
- Scott, A., et al., Tenocyte responses to mechanical loading in vivo: A role for local insulin-like growth factor 1 signaling in early tendinosis in rats. Arthritis & Rheumatism, 2007. 56(3): p. 871-881. [CrossRef]
- Wang, F., G.A. Murrell, and M.X. Wang, Oxidative stress-induced c-Jun N-terminal kinase (JNK) activation in tendon cells upregulates MMP1 mRNA and protein expression. J Orthop Res, 2007. 25(3): p. 378-89. [CrossRef]
- Xu, Y., F. Bonar, and G.A.C. Murrell, Neoinnervation in Rotator Cuff Tendinopathy. Sports Medicine and Arthroscopy Review, 2011. 19(4): p. 354-359. [CrossRef]
- Wu, X.L., et al., Arthroscopic Undersurface Rotator Cuff Repair. Techniques in Shoulder & Elbow Surgery, 2009. 10(3): p. 112-118. [CrossRef]
- Fu, S.-C., et al., Deciphering the pathogenesis of tendinopathy: a three-stages process. Sports medicine, arthroscopy, rehabilitation, therapy & technology : SMARTT, 2010. 2: p. 30-30. [CrossRef]
- Summers, K.L., et al., Phenotypic characterization of five dendritic cell subsets in human tonsils. The American journal of pathology, 2001. 159(1): p. 285-295. [CrossRef]
- Schliefsteiner, C., et al., Human Placental Hofbauer Cells Maintain an Anti-inflammatory M2 Phenotype despite the Presence of Gestational Diabetes Mellitus. Frontiers in immunology, 2017. 8: p. 888-888. [CrossRef]
- Hewitt, S.M., et al., Controls for immunohistochemistry: the Histochemical Society's standards of practice for validation of immunohistochemical assays. The journal of histochemistry and cytochemistry : official journal of the Histochemistry Society, 2014. 62(10): p. 693-697.
- Coillard, A. and E. Segura, In vivo Differentiation of Human Monocytes. Frontiers in Immunology, 2019. 10(1907). [CrossRef]
- Qu, C., et al., Monocyte-derived dendritic cells: targets as potent antigen-presenting cells for the design of vaccines against infectious diseases. International Journal of Infectious Diseases, 2014. 19: p. 1-5. [CrossRef]
- León, B., M. López-Bravo, and C. Ardavín, Monocyte-Derived Dendritic Cells Formed at the Infection Site Control the Induction of Protective T Helper 1 Responses against Leishmania. Immunity, 2007. 26(4): p. 519-531. [CrossRef]
- Le Borgne, M., et al., Dendritic Cells Rapidly Recruited into Epithelial Tissues via CCR6/CCL20 Are Responsible for CD8+ T Cell Crosspriming In Vivo. Immunity, 2006. 24(2): p. 191-201.
- Qu, C., et al., MHC Class I/Peptide Transfer between Dendritic Cells Overcomes Poor Cross-Presentation by Monocyte-Derived APCs That Engulf Dying Cells. The Journal of Immunology, 2009. 182(6): p. 3650-3659. [CrossRef]
- Sugg, K.B., et al., Changes in macrophage phenotype and induction of epithelial-to-mesenchymal transition genes following acute Achilles tenotomy and repair. Journal of Orthopaedic Research, 2014. 32(7): p. 944-951. [CrossRef]
- Acosta-Rodriguez, E.V., et al., Interleukins 1beta and 6 but not transforming growth factor-beta are essential for the differentiation of interleukin 17-producing human T helper cells. Nat Immunol, 2007. 8(9): p. 942-9.
- Manel, N., D. Unutmaz, and D.R. Littman, The differentiation of human T(H)-17 cells requires transforming growth factor-beta and induction of the nuclear receptor RORgammat. Nature immunology, 2008. 9(6): p. 641-649.
- Wilson, N.J., et al., Development, cytokine profile and function of human interleukin 17-producing helper T cells. Nat Immunol, 2007. 8(9): p. 950-7.
- Volpe, E., et al., A critical function for transforming growth factor-beta, interleukin 23 and proinflammatory cytokines in driving and modulating human T(H)-17 responses. Nat Immunol, 2008. 9(6): p. 650-7.
- Zaba, L.C., et al., Psoriasis Is Characterized by Accumulation of Immunostimulatory and Th1/Th17 Cell-Polarizing Myeloid Dendritic Cells. Journal of Investigative Dermatology, 2009. 129(1): p. 79-88.
- Santini, S.M., et al., Interferon-α-Conditioned Human Monocytes Combine a Th1-Orienting Attitude with the Induction of Autologous Th17 Responses: Role of IL-23 and IL-12. PLOS ONE, 2011. 6(2): p. e17364. [CrossRef]
- Farkas, A. and L. Kemény, Monocyte-derived interferon-alpha primed dendritic cells in the pathogenesis of psoriasis: new pieces in the puzzle. Int Immunopharmacol, 2012. 13(2): p. 215-8.

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




