Submitted:
21 February 2024
Posted:
23 February 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
1.1. Background and Rationale
1.2. Objectives
2. Materials and Methods
2.1. Study Design
2.2. Setting
2.3. Participants
2.4. Interventions
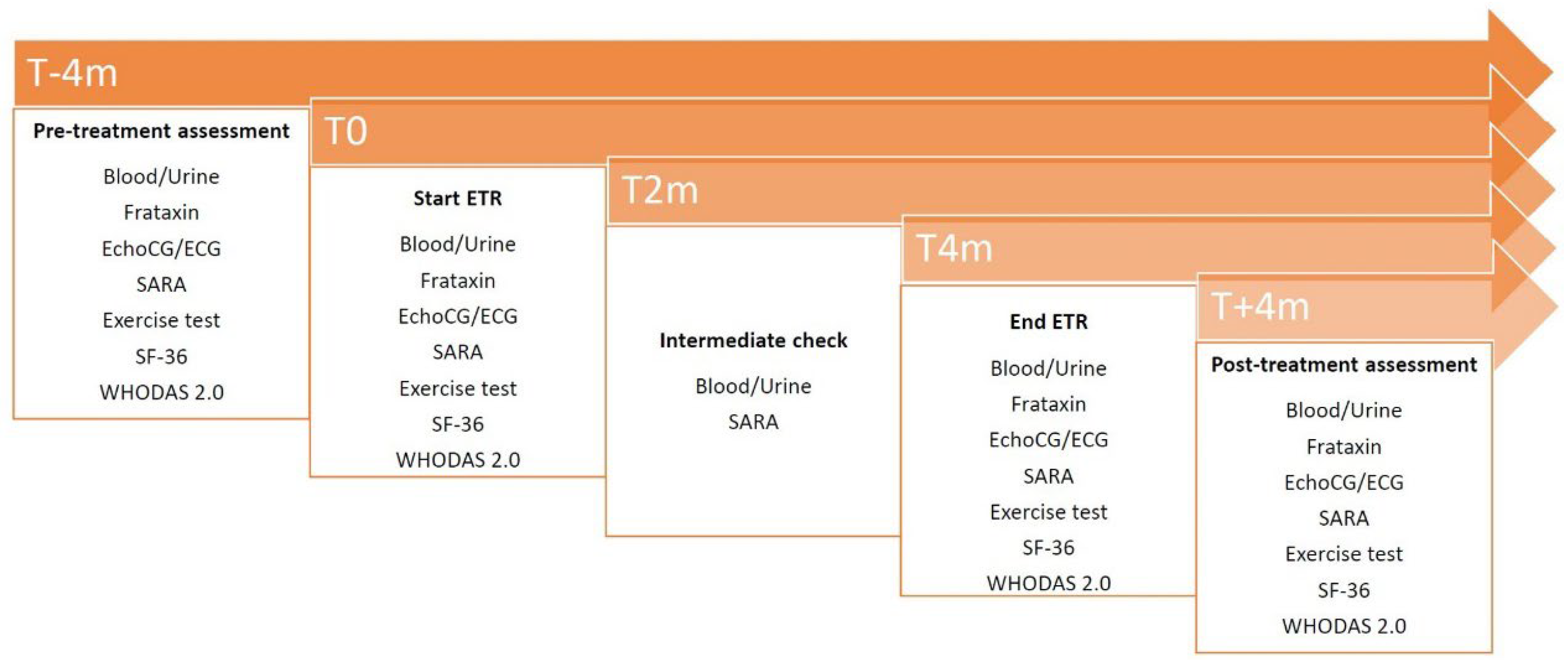
2.5. Study Endpoints
- peak workload (peak W);
- neurological progression measured with Scale for the assessment and Rating of Ataxia (SARA) [12];
- echocardiography (septal wall thickness) and electrocardiogram (Sokolow-Lyon index) parameters;
- frataxin protein levels in peripheral blood mononuclear cells and molecular analysis of frataxin mRNA translation efficiency;
- quality of life evaluated with Short Form Health Survey 36 (SF-36) [13];
- disability, assessed by World Health Organization Disability Assessment Schedule 2.0 (WHODAS 2.0) 36-item self-administered version [14].
2.5.1. Incremental Exercise Test
2.5.2. Neurological Measure
2.5.3. Cardiac Parameters
2.5.4. Health and Disability Measures
2.6. Sample Size Calculation
2.7. Randomization
2.8. Statistical Analysis
3. Results
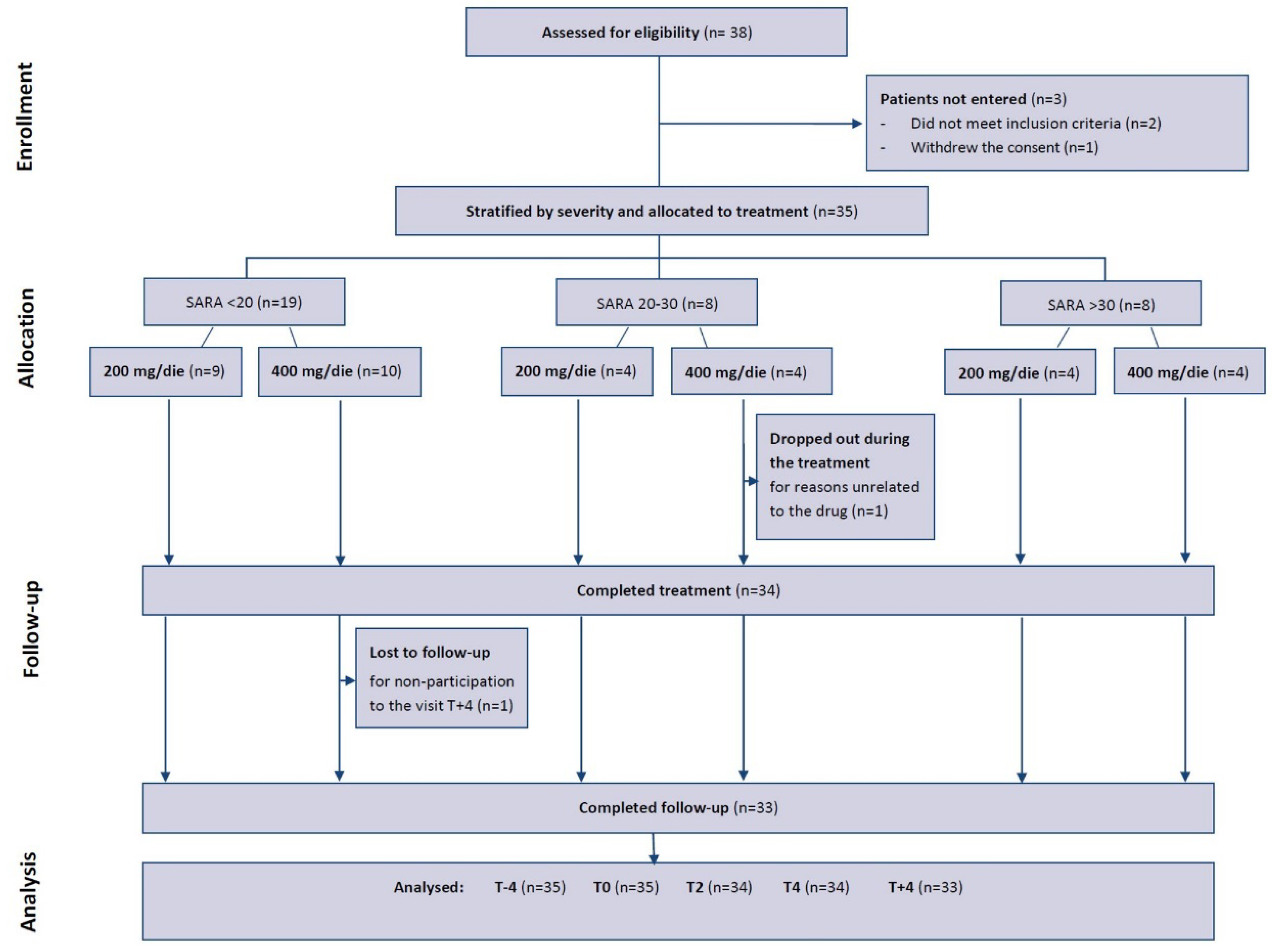
3.1. Participant Flow
3.2. Patient Features
3.3. Safety
3.4. Efficacy
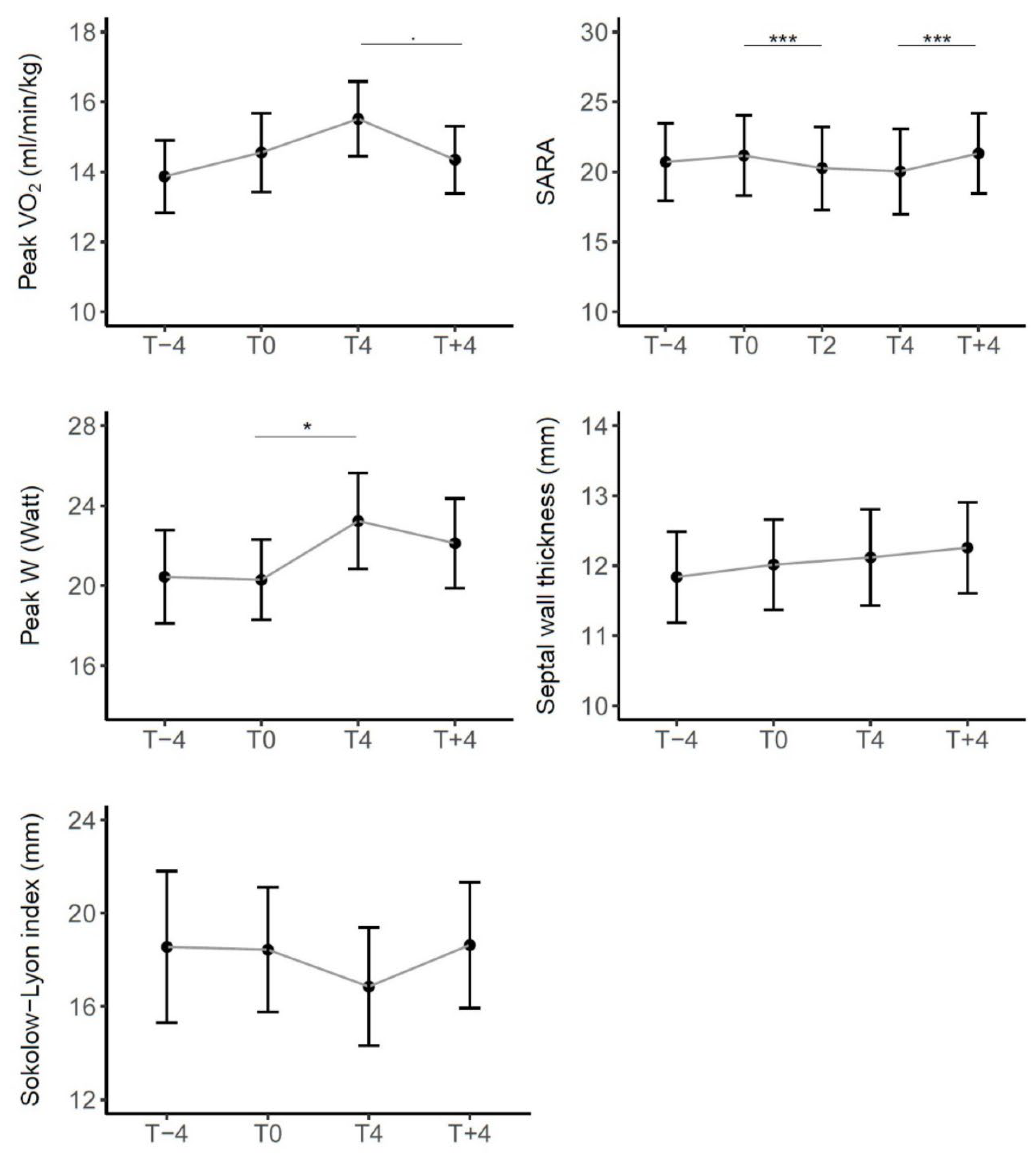
3.4.1. Variations in Clinical and Functional Outcomes
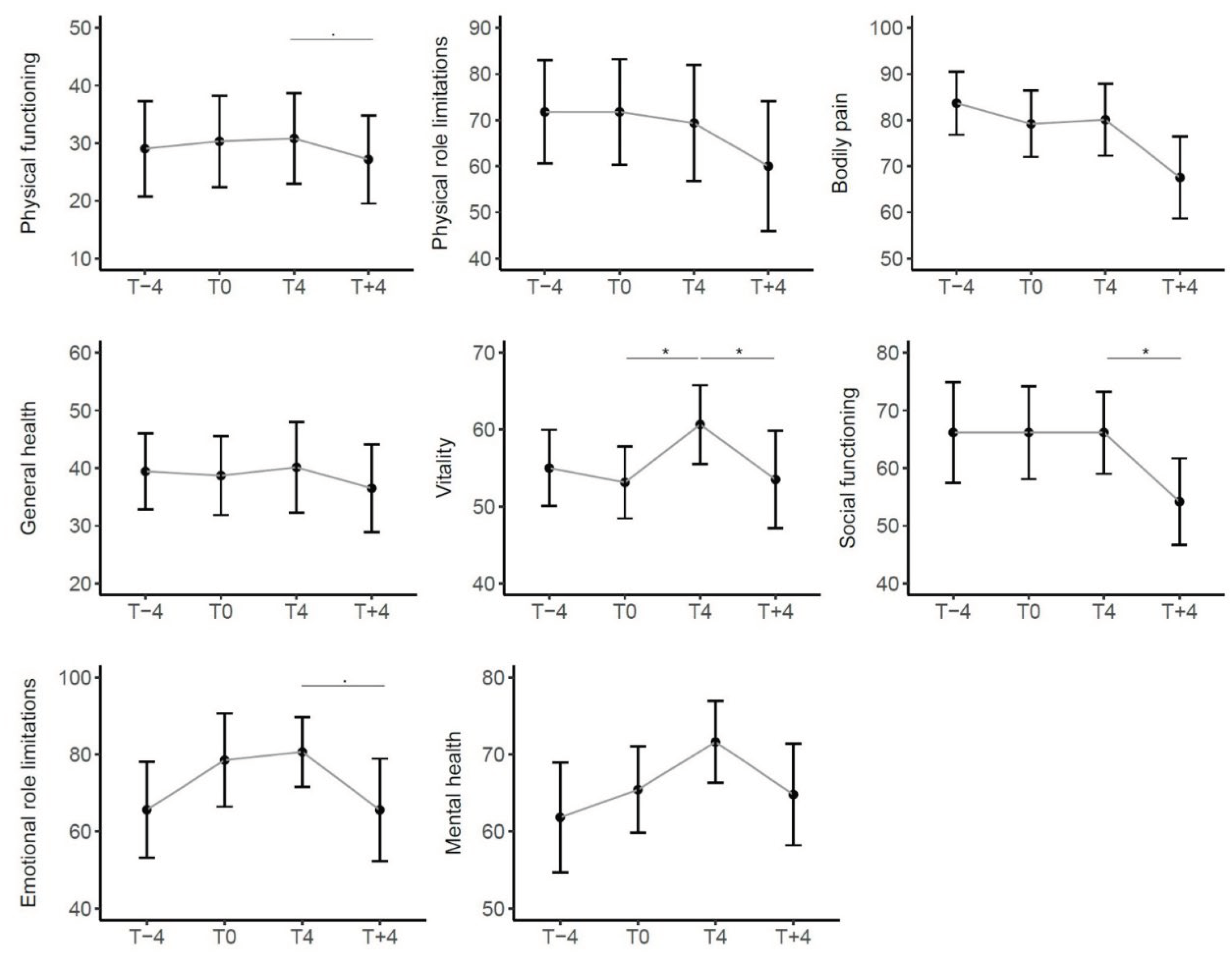
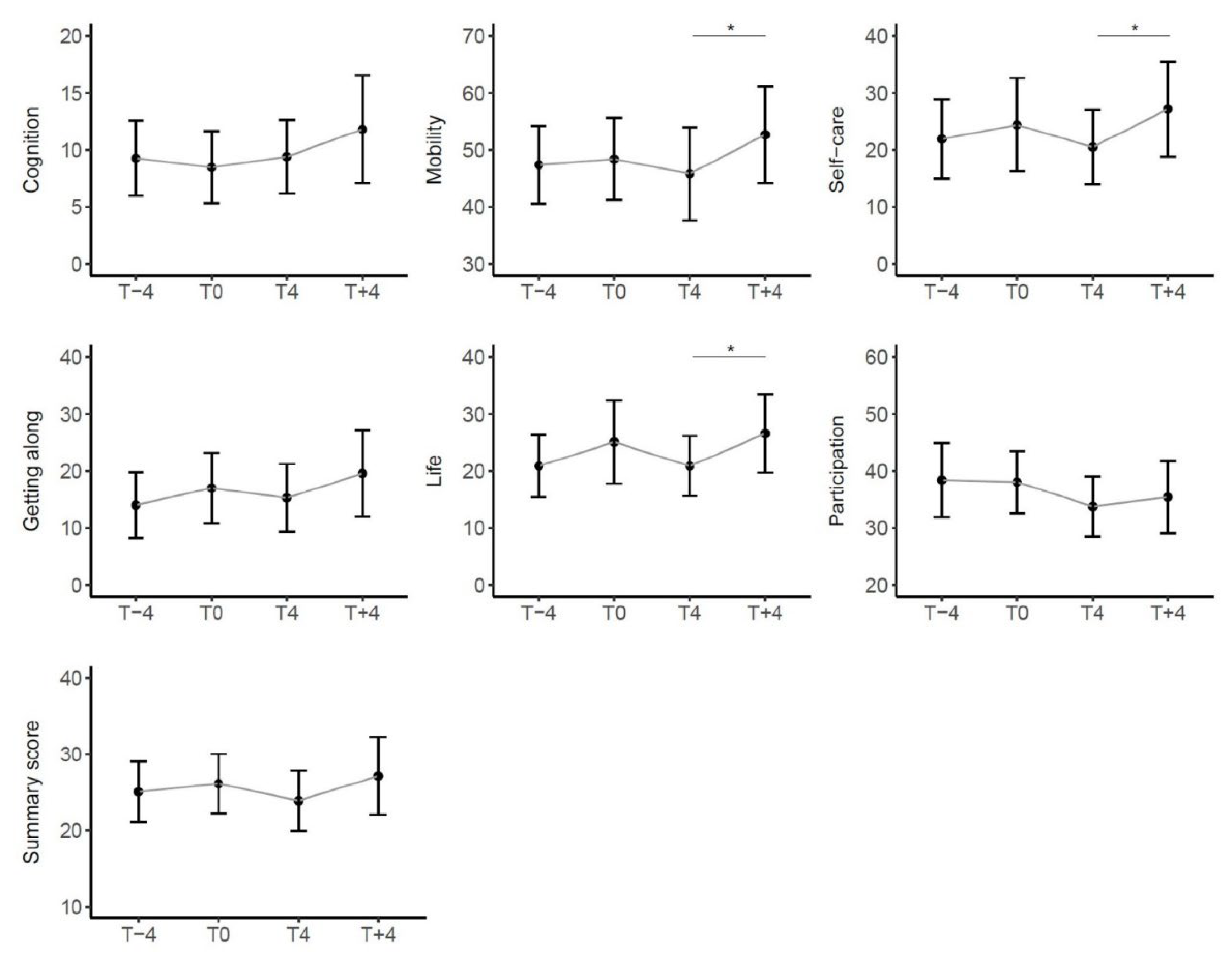
3.4.2. Variations in SF-36 and WHODAS 2.0 Questionnaires
3.4.3. Variations in Frataxin
4. Discussion
4.1. Limitations of the Study
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Campuzano, V.; Montermini, L.; Moltò, M.D.; Pianese, L.; Cossée, M.; Cavalcanti, F.; Monros, E.; Rodius, F.; Duclos, F.; Monticelli, A.; Zara, F.; Cañizares, J.; Koutnikova, H.; Bidichandani, S.I.; Gellera, C.; Brice, A.; Trouillas, P.; De Michele, G.; Filla, A.; De Frutos, R.; Palau, F.; Patel, P.I.; Di Donato, S.; Mandel, J.L.; Cocozza, S.; Koenig, M.; Pandolfo, M. Friedreich’s Ataxia: Autosomal Recessive Disease Caused by an Intronic GAA Triplet Repeat Expansion. Science 1996, 271, 1423–1427. [Google Scholar] [CrossRef] [PubMed]
- Schulz, J.B.; Dehmer, T.; Schöls, L.; Mende, H.; Hardt, C.; Vorgerd, M.; Bürk, K.; Matson, W.; Dichgans, J.; Beal, M.F.; Bogdanov, M.B. Oxidative Stress in Patients with Friedreich Ataxia. Neurology 2000, 55, 1719–1721. [Google Scholar] [CrossRef]
- Clark, E.; Johnson, J.; Dong, Y.N.; Mercado-Ayon, E.; Warren, N.; Zhai, M.; McMillan, E.; Salovin, A.; Lin, H.; Lynch, D.R. Role of Frataxin Protein Deficiency and Metabolic Dysfunction in Friedreich Ataxia, an Autosomal Recessive Mitochondrial Disease. Neuronal Signal. 2018, 2, NS20180060. [Google Scholar] [CrossRef] [PubMed]
- Galea, C.A.; Huq, A.; Lockhart, P.J.; Tai, G.; Corben, L.A.; Yiu, E.M.; Gurrin, L.C.; Lynch, D.R.; Gelbard, S.; Durr, A.; Pousset, F.; Parkinson, M.; Labrum, R.; Giunti, P.; Perlman, S.L.; Delatycki, M.B.; Evans-Galea, M.V. Compound Heterozygous FXN Mutations and Clinical Outcome in Friedreich Ataxia. Ann. Neurol. 2016, 79, 485–495. [Google Scholar] [CrossRef] [PubMed]
- Parkinson, M.H.; Boesch, S.; Nachbauer, W.; Mariotti, C.; Giunti, P. Clinical Features of Friedreich’s Ataxia: Classical and Atypical Phenotypes. J. Neurochem. 2013, 126 Suppl 1, 103–117. [Google Scholar] [CrossRef]
- Schulz, J.B.; Boesch, S.; Bürk, K.; Dürr, A.; Giunti, P.; Mariotti, C.; Pousset, F.; Schöls, L.; Vankan, P.; Pandolfo, M. Diagnosis and Treatment of Friedreich Ataxia: A European Perspective. Nat. Rev. Neurol. 2009, 5, 222–234. [Google Scholar] [CrossRef]
- Delatycki, M.B.; Corben, L.A. Clinical Features of Friedreich Ataxia. J. Child Neurol. 2012, 27, 1133–1137. [Google Scholar] [CrossRef]
- Alfedi, G.; Luffarelli, R.; Condò, I.; Pedini, G.; Mannucci, L.; Massaro, D.S.; Benini, M.; Toschi, N.; Alaimo, G.; Panarello, L.; Pacini, L.; Fortuni, S.; Serio, D.; Malisan, F.; Testi, R.; Rufini, A. Drug Repositioning Screening Identifies Etravirine as a Potential Therapeutic for Friedreich’s Ataxia. Mov. Disord. Off. J. Mov. Disord. Soc. 2019, 34, 323–334. [Google Scholar] [CrossRef]
- Lynch, D.R.; Farmer, J.; Hauser, L.; Blair, I.A.; Wang, Q.Q.; Mesaros, C.; Snyder, N.; Boesch, S.; Chin, M.; Delatycki, M.B.; Giunti, P.; Goldsberry, A.; Hoyle, C.; McBride, M.G.; Nachbauer, W.; O’Grady, M.; Perlman, S.; Subramony, S.H.; Wilmot, G.R.; Zesiewicz, T.; Meyer, C. Safety, Pharmacodynamics, and Potential Benefit of Omaveloxolone in Friedreich Ataxia. Ann. Clin. Transl. Neurol. 2018, 6, 15–26. [Google Scholar] [CrossRef] [PubMed]
- Lynch, D.R.; Chin, M.P.; Delatycki, M.B.; Subramony, S.H.; Corti, M.; Hoyle, J.C.; Boesch, S.; Nachbauer, W.; Mariotti, C.; Mathews, K.D.; Giunti, P.; Wilmot, G.; Zesiewicz, T.; Perlman, S.; Goldsberry, A.; O’Grady, M.; Meyer, C.J. Safety and Efficacy of Omaveloxolone in Friedreich Ataxia (MOXIe Study). Ann. Neurol. 2021, 89, 212–225. [Google Scholar] [CrossRef]
- Vavla, M.; D’Angelo, M.G.; Arrigoni, F.; Toschi, N.; Peruzzo, D.; Gandossini, S.; Russo, A.; Diella, E.; Tirelli, S.; Salati, R.; Scarpazza, P.; Luffarelli, R.; Fortuni, S.; Rufini, A.; Condò, I.; Testi, R.; Martinuzzi, A. Safety and Efficacy of Interferon γ in Friedreich’s Ataxia. Mov. Disord. Off. J. Mov. Disord. Soc. 2020, 35, 370–371. [Google Scholar] [CrossRef] [PubMed]
- Schmitz-Hübsch, T.; du Montcel, S.T.; Baliko, L.; Berciano, J.; Boesch, S.; Depondt, C.; Giunti, P.; Globas, C.; Infante, J.; Kang, J.-S.; Kremer, B.; Mariotti, C.; Melegh, B.; Pandolfo, M.; Rakowicz, M.; Ribai, P.; Rola, R.; Schöls, L.; Szymanski, S.; van de Warrenburg, B.P.; Dürr, A.; Klockgether, T.; Fancellu, R. Scale for the Assessment and Rating of Ataxia: Development of a New Clinical Scale. Neurology 2006, 66, 1717–1720. [Google Scholar] [CrossRef]
- Apolone, G.; Mosconi, P. The Italian SF-36 Health Survey: Translation, Validation and Norming. J. Clin. Epidemiol. 1998, 51, 1025–1036. [Google Scholar] [CrossRef] [PubMed]
- Ustün, T.B.; Chatterji, S.; Kostanjsek, N.; Rehm, J.; Kennedy, C.; Epping-Jordan, J.; Saxena, S.; von Korff, M.; Pull, C.; WHO/NIH Joint Project. Developing the World Health Organization Disability Assessment Schedule 2.0. Bull. World Health Organ. 2010, 88, 815–823. [Google Scholar] [CrossRef]
- Herdy, A.H.; Uhlendorf, D. Reference Values for Cardiopulmonary Exercise Testing for Sedentary and Active Men and Women. Arq. Bras. Cardiol. 2011, 96, 54–59. [Google Scholar] [CrossRef] [PubMed]
- Lintu, N.; Viitasalo, A.; Tompuri, T.; Veijalainen, A.; Hakulinen, M.; Laitinen, T.; Savonen, K.; Lakka, T.A. Cardiorespiratory Fitness, Respiratory Function and Hemodynamic Responses to Maximal Cycle Ergometer Exercise Test in Girls and Boys Aged 9-11 Years: The PANIC Study. Eur. J. Appl. Physiol. 2015, 115, 235–243. [Google Scholar] [CrossRef]
- Drinkard, B.E.; Keyser, R.E.; Paul, S.M.; Arena, R.; Plehn, J.F.; Yanovski, J.A.; Di Prospero, N.A. Exercise Capacity and Idebenone Intervention in Children and Adolescents with Friedreich Ataxia. Arch. Phys. Med. Rehabil. 2010, 91, 1044–1050. [Google Scholar] [CrossRef]
- Saccà, F.; Puorro, G.; Marsili, A.; Antenora, A.; Pane, C.; Casali, C.; Marcotulli, C.; Defazio, G.; Liuzzi, D.; Tatillo, C.; Cambriglia, D.M.; Schiano di Cola, G.; Giuliani, L.; Guardasole, V.; Salzano, A.; Ruvolo, A.; De Rosa, A.; Cittadini, A.; De Michele, G.; Filla, A. Long-Term Effect of Epoetin Alfa on Clinical and Biochemical Markers in Friedreich Ataxia. Mov. Disord. Off. J. Mov. Disord. Soc. 2016, 31, 734–741. [Google Scholar] [CrossRef]
- Pane, C.; Salzano, A.; Trinchillo, A.; Del Prete, C.; Casali, C.; Marcotulli, C.; Defazio, G.; Guardasole, V.; Vastarella, R.; Giallauria, F.; Puorro, G.; Marsili, A.; De Michele, G.; Filla, A.; Cittadini, A.; Saccà, F. Safety and Feasibility of Upper Limb Cardiopulmonary Exercise Test in Friedreich Ataxia. Eur. J. Prev. Cardiol. 2022, 29, 445–451. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing., 2022. https://www.R-project.org/.
- Roessler, H.I.; Knoers, N.V.A.M.; van Haelst, M.M.; van Haaften, G. Drug Repurposing for Rare Diseases. Trends Pharmacol. Sci. 2021, 42, 255–267. [Google Scholar] [CrossRef]
- Rufini, A.; Malisan, F.; Condò, I.; Testi, R. Drug Repositioning in Friedreich Ataxia. Front. Neurosci. 2022, 16, 814445. [Google Scholar] [CrossRef]
- Allavena, C.; Katlama, C.; Cotte, L.; Roger, P.M.; Delobel, P.; Cheret, A.; Duvivier, C.; Poizot-Martin, I.; Hoen, B.; Cabie, A.; Cheret, A.; Lahoulou, R.; Raffi, F.; Pugliese, P.; Dat’AIDS Study group. Long-Term Efficacy and Safety of Etravirine-Containing Regimens in a Real-Life Cohort of Treatment-Experienced HIV-1-Infected Patients. Infect. Dis. Lond. Engl. 2016, 48, 392–398. [Google Scholar] [CrossRef] [PubMed]
- Marelli, C.; Figoni, J.; Charles, P.; Anheim, M.; Tchikviladze, M.; Vincitorio, C.-M.; du Montcel, S.T.; Brice, A.; Golmard, J.L.; Dürr, A. Annual Change in Friedreich’s Ataxia Evaluated by the Scale for the Assessment and Rating of Ataxia (SARA) Is Independent of Disease Severity. Mov. Disord. Off. J. Mov. Disord. Soc. 2012, 27, 135–138. [Google Scholar] [CrossRef]
- Tanguy Melac, A.; Mariotti, C.; Filipovic Pierucci, A.; Giunti, P.; Arpa, J.; Boesch, S.; Klopstock, T.; Müller Vom Hagen, J.; Klockgether, T.; Bürk, K.; Schulz, J.B.; Reetz, K.; Pandolfo, M.; Durr, A.; Tezenas du Montcel, S.; EFACTS group. Friedreich and Dominant Ataxias: Quantitative Differences in Cerebellar Dysfunction Measurements. J. Neurol. Neurosurg. Psychiatry 2018, 89, 559–565. [Google Scholar] [CrossRef] [PubMed]
- Lynch, D.R.; Schadt, K.; Kichula, E. Etravirine in Friedreich’s Ataxia: Lessons from HIV? Mov. Disord. Off. J. Mov. Disord. Soc. 2019, 34, 305–306. [Google Scholar] [CrossRef] [PubMed]
- Reetz, K.; Dogan, I.; Hilgers, R.-D.; Giunti, P.; Mariotti, C.; Durr, A.; Boesch, S.; Klopstock, T.; de Rivera, F.J.R.; Schöls, L.; Klockgether, T.; Bürk, K.; Rai, M.; Pandolfo, M.; Schulz, J.B.; EFACTS Study Group. Progression Characteristics of the European Friedreich’s Ataxia Consortium for Translational Studies (EFACTS): A 2 Year Cohort Study. Lancet Neurol. 2016, 15, 1346–1354. [Google Scholar] [CrossRef] [PubMed]
- Patel, M.; Isaacs, C.J.; Seyer, L.; Brigatti, K.; Gelbard, S.; Strawser, C.; Foerster, D.; Shinnick, J.; Schadt, K.; Yiu, E.M.; Delatycki, M.B.; Perlman, S.; Wilmot, G.R.; Zesiewicz, T.; Mathews, K.; Gomez, C.M.; Yoon, G.; Subramony, S.H.; Brocht, A.; Farmer, J.; Lynch, D.R. Progression of Friedreich Ataxia: Quantitative Characterization over 5 Years. Ann. Clin. Transl. Neurol. 2016, 3, 684–694. [Google Scholar] [CrossRef]
- Reetz, K.; Dogan, I.; Hilgers, R.-D.; Giunti, P.; Parkinson, M.H.; Mariotti, C.; Nanetti, L.; Durr, A.; Ewenczyk, C.; Boesch, S.; Nachbauer, W.; Klopstock, T.; Stendel, C.; Rodríguez de Rivera Garrido, F.J.; Rummey, C.; Schöls, L.; Hayer, S.N.; Klockgether, T.; Giordano, I.; Didszun, C.; Rai, M.; Pandolfo, M.; Schulz, J.B.; EFACTS study group. Progression Characteristics of the European Friedreich’s Ataxia Consortium for Translational Studies (EFACTS): A 4-Year Cohort Study. Lancet Neurol. 2021, 20, 362–372. [Google Scholar] [CrossRef]
| Overall (N=35) | Dose 200 (N=17) | Dose 400 (N=18) | p-value | |
|---|---|---|---|---|
| Patient type, n (%) | >0.999 | |||
| Adult | 30 (85.7) | 15 (88.2) | 15 (83.3) | |
| Children | 5 (14.3) | 2 (11.8) | 3 (16.7) | |
| Male, n (%) | 16 (45.7) | 5 (29.4) | 11 (61.1) | 0.123 |
| Severity, n (%) | 0.988 | |||
| SARA <20 | 19 (54.3) | 9 (52.9) | 10 (55.6) | |
| SARA 20-30 | 8 (22.9) | 4 (23.5) | 4 (22.2) | |
| SARA >30 | 8 (22.9) | 4 (23.5) | 4 (22.2) | |
| GAA sr | 665.50 (189.02) | 670.87 (197.10) | 660.13 (187.35) | 0.880 |
| GAA lr | 862.89 (203.86) | 917.64 (226.81) | 808.14 (168.56) | 0.159 |
| Age at onset (years), mean (SD) | 11.83 (5.70) | 10.65 (4.20) | 12.94 (6.76) | 0.239 |
| Age (years), mean (SD) | 25.00 (7.14) | 23.53 (5.01) | 26.39 (8.61) | 0.242 |
| Disease duration (years), mean (SD) | 13.17 (6.00) | 12.88 (5.29) | 13.44 (6.75) | 0.786 |
| Years of education, median [IQR] | 13.00 [13.00, 16.50] | 14.00 [13.00, 17.00] | 13.00 [13.00, 15.50] | 0.458 |
| Months 1 e 2 | Months 3 e 4 | |
|---|---|---|
| Headache | 16 (47%) | 3 (9%) |
| Diarrhea | 7 (21%) | 1 (3%) |
| Nausea | 4 (12%) | 1 (3%) |
| Skin Rash | 4 (12%) | 0 |
| Other: | ||
| Fatigue | 3 (9%) | 0 |
| Hypotension | 2 (6%) | 0 |
| Tachycardia | 1 (3%) | 1 (3%) |
| Vomit | 1 (3%) | 1 (3%) |
| Constipation | 1 (3%) | 0 |
| Blurred vision | 1 (3%) | 0 |
| Sweating | 1 (3%) | 0 |
| Urinary urgency | 1 (3%) | 0 |
| Calf cramps | 1 (3%) | 0 |
| N (%) | |
|---|---|
| Total cholesterol + | 4 (12%) |
| Triglyceride + | 3 (9%) |
| AST/ALT + | 1 (3%) |
| Amylase + | 1 (3%) |
| White blood cells - | 1 (3%) |
| T0 vs T-4 | T2 vs T0 | T4 vs T2 | T4 vs T0 | T+4 vs T4 | |
|---|---|---|---|---|---|
| Start ETR vs Pre-treatment | Intermediate check vs Start ETR | End ETR vs Intermediate check |
End ETR vs Start ETR |
Post-treatment vs End ETR | |
| Peak VO2 | 0.685 (0.619) | 0.959 (0.224) | -1.145 (0.088) | ||
| SARA | 0.456 (0.243) | -0.897 (<0.001) | -0.235 (0.914) | -1.132 (<0.001) | 1.229 (<0.001) |
| Peak W | -0.147 (>0.999) | 2.941 (0.021) | -1.037 (0.905) | ||
| Septal wall thickness | 0.176 | 0.103 | 0.111 | ||
| Sokolow-Lyon index | -0.118 | -1.582 | 1.808 |
| SARA < 20 (N=19) | SARA 20-30 (N=7) | SARA > 30 (N=8) | Dose 200 (N=17) | Dose 400 (N=17) | |
|---|---|---|---|---|---|
| Peak VO2 | |||||
| delta T4 - T0 | 0.51 (2.26) | 1.81 (1.88) | 1.29 (2.45) | 0.80 (2.80) | 1.12 (1.55) |
| delta T+4 - T4 | -1.42 (2.84) | -1.21 (2.15) | -0.36 (1.18) | -0.99 (2.60) | -1.25 (2.19) |
| SARA | |||||
| delta T4 - T0 | -1.53 (1.03) | -1.21 (1.82) | -0.12 (0.35) | -0.97 (1.21) | -1.29 (1.30) |
| delta T+4 - T4 | 1.78 (1.23) | 1.14 (1.14) | 0.06 (0.50) | 0.94 (1.14) | 1.53 (1.36) |
| Peak W | |||||
| delta T4 - T0 | 2.95 (4.34) | 1.43 (2.76) | 4.25 (8.45) | 2.71 (3.24) | 3.18 (6.82) |
| delta T+4 - T4 | -1.00 (2.77) | 1.43 (1.90) | -3.00 (11.36) | -0.71 (2.91) | -1.25 (8.13) |
| Septal wall thickness | |||||
| delta T4 - T0 | 0.11 (0.86) | 0.43 (0.84) | -0.19 (1.07) | 0.06 (0.58) | 0.15 (1.16) |
| delta T+4 - T4 | 0.19 (0.86) | 0.07 (0.61) | 0.06 (0.68) | -0.09 (0.73) | 0.38 (0.72) |
| Sokolow-Lyon index | |||||
| delta T4 - T0 | -1.31 (3.64) | -3.29 (6.21) | -0.75 (2.82) | -2.00 (4.09) | -1.16 (4.17) |
| delta T+4 - T4 | 2.06 (3.40) | 2.00 (4.36) | 1.25 (2.49) | 1.65 (2.94) | 2.06 (3.82) |
| T4 vs T0 | T+4 vs T4 | |
|---|---|---|
| Peak VO2 | ||
| Severity (SARA 20-30) | 1.347 (0.196) | 0.212 (0.850) |
| Severity (SARA >30) | 0.715 (0.478) | 1.242 (0.268) |
| Dose (400) | 0.354 (0.661) | -0.205 (0.815) |
| SARA | ||
| Severity (SARA 20-30) | 0.282 (0.589) | -0.569 (0.234) |
| Severity (SARA >30) | 1.398 (0.010) | -1.552 (0.002) |
| Dose (400) | -0.304 (0.459) | 0.637 (0.094) |
| Peak W | ||
| Severity (SARA 20-30) | -1.485 (0.546) | 2.290 (0.403) |
| Severity (SARA >30) | 0.962 (0.689) | -2.742 (0.315) |
| Dose (400) | 0.189 (0.922) | -0.627 (0.770) |
| Septal wall thickness | ||
| Severity (SARA 20-30) | 0.335 (0.422) | -0.100 (0.769) |
| Severity (SARA >30) | -0.220 (0.589) | -0.198 (0.559) |
| Dose (400) | 0.147 (0.653) | 0.441 (0.108) |
| Sokolow-Lyon index | ||
| Severity (SARA 20-30) | -1.911 (0.317) | -0.034 (0.983) |
| Severity (SARA >30) | 0.444 (0.810) | -0.864 (0.588) |
| Dose (400) | 0.651 (0.662) | 0.408 (0.746) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




