Submitted:
20 February 2024
Posted:
21 February 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
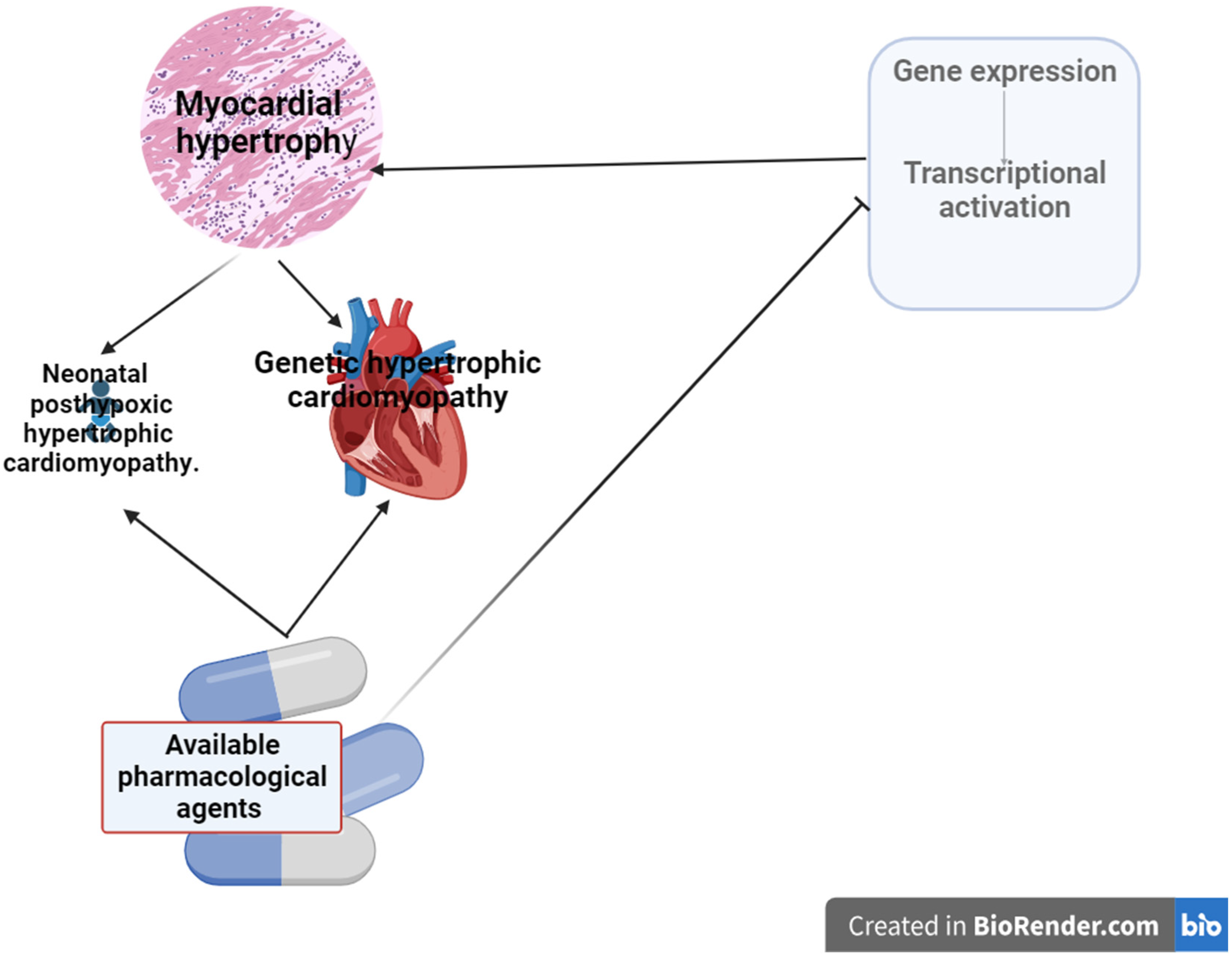
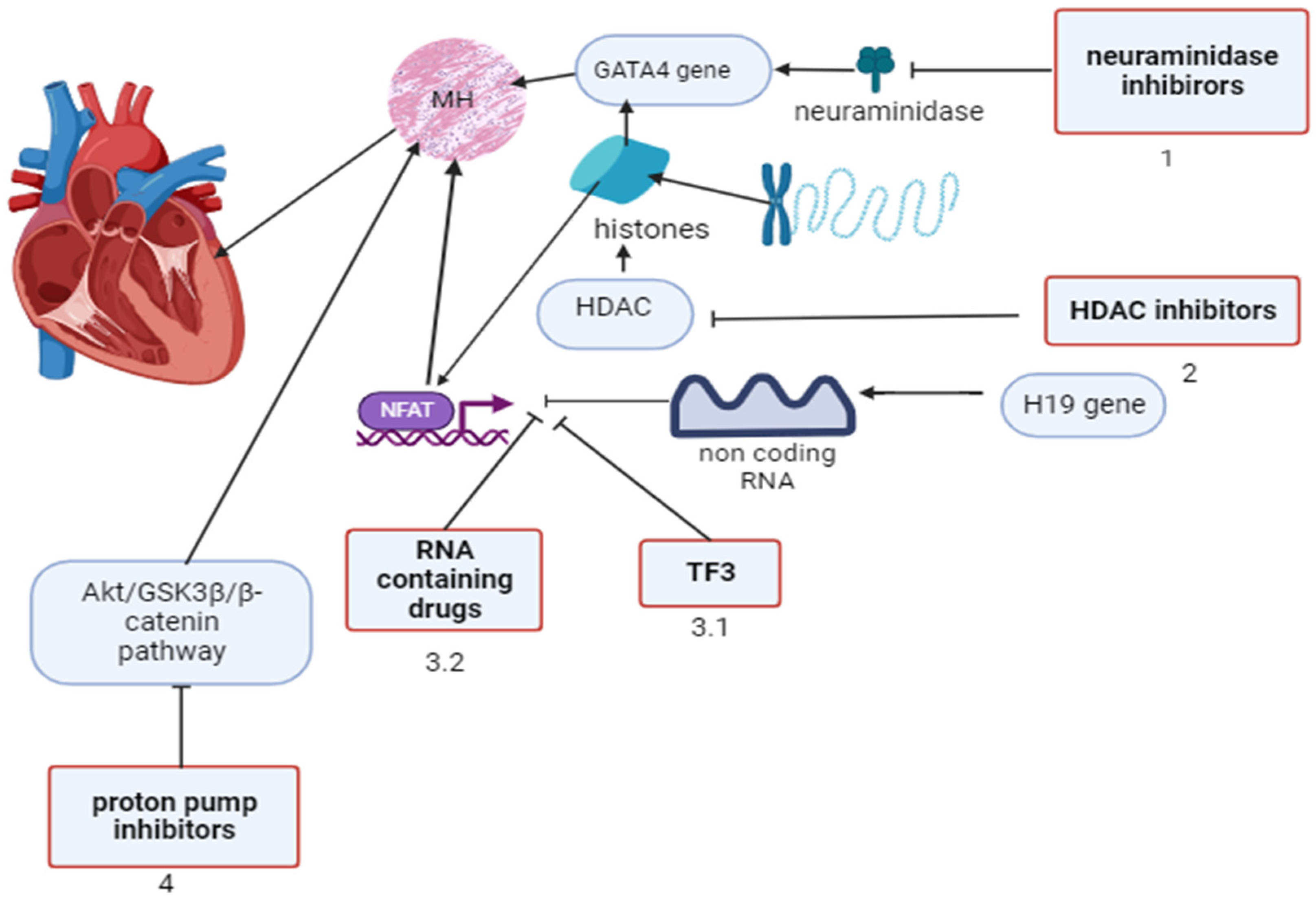
2. Regulatory factors of gene expression in myocardial hypertrophy and approaches to pharmacological modulation
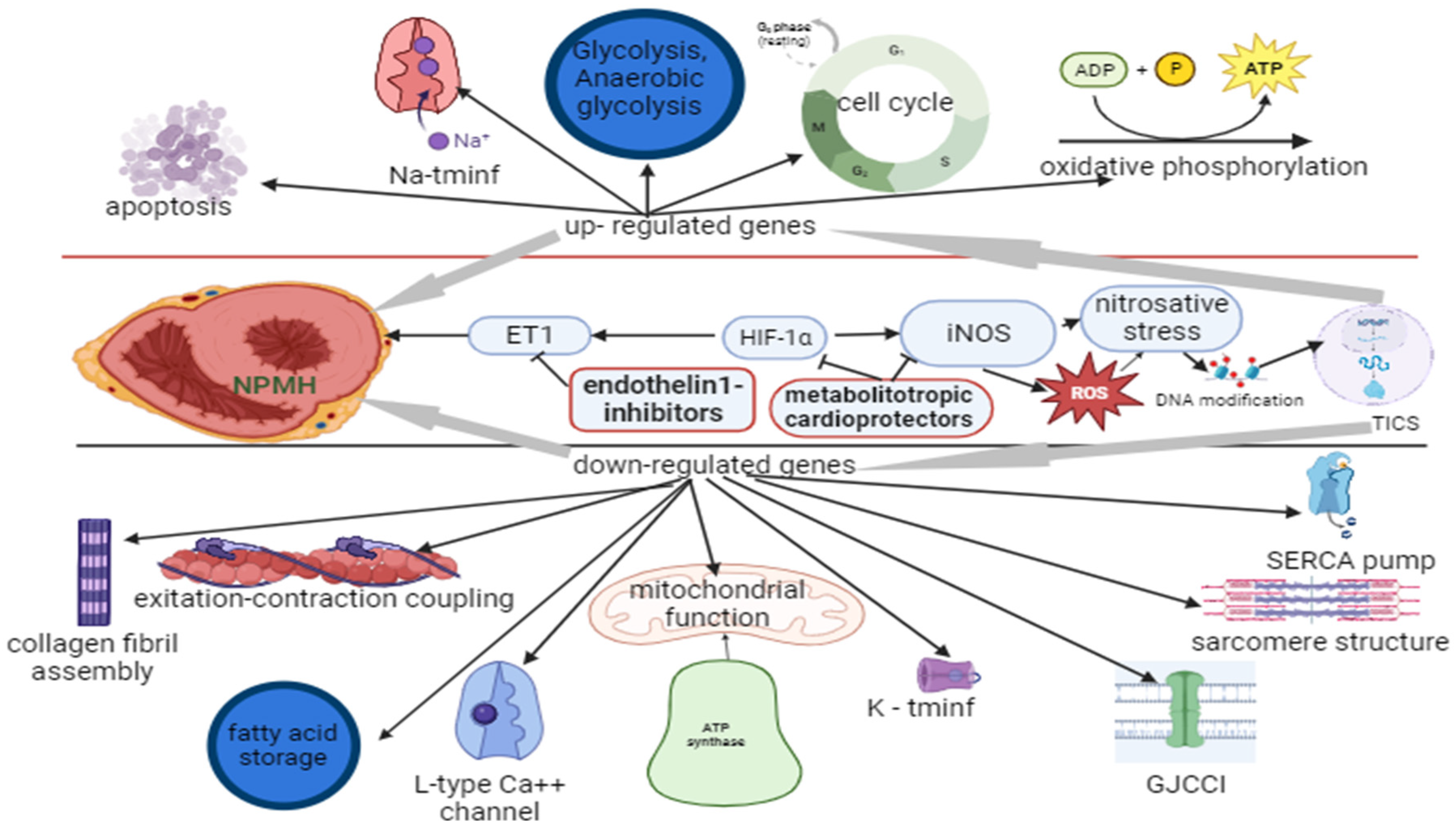
3. Neonatal posthypoxic myocardial hypertrophy, factors of gene expression regulation and pharmacological modulation.
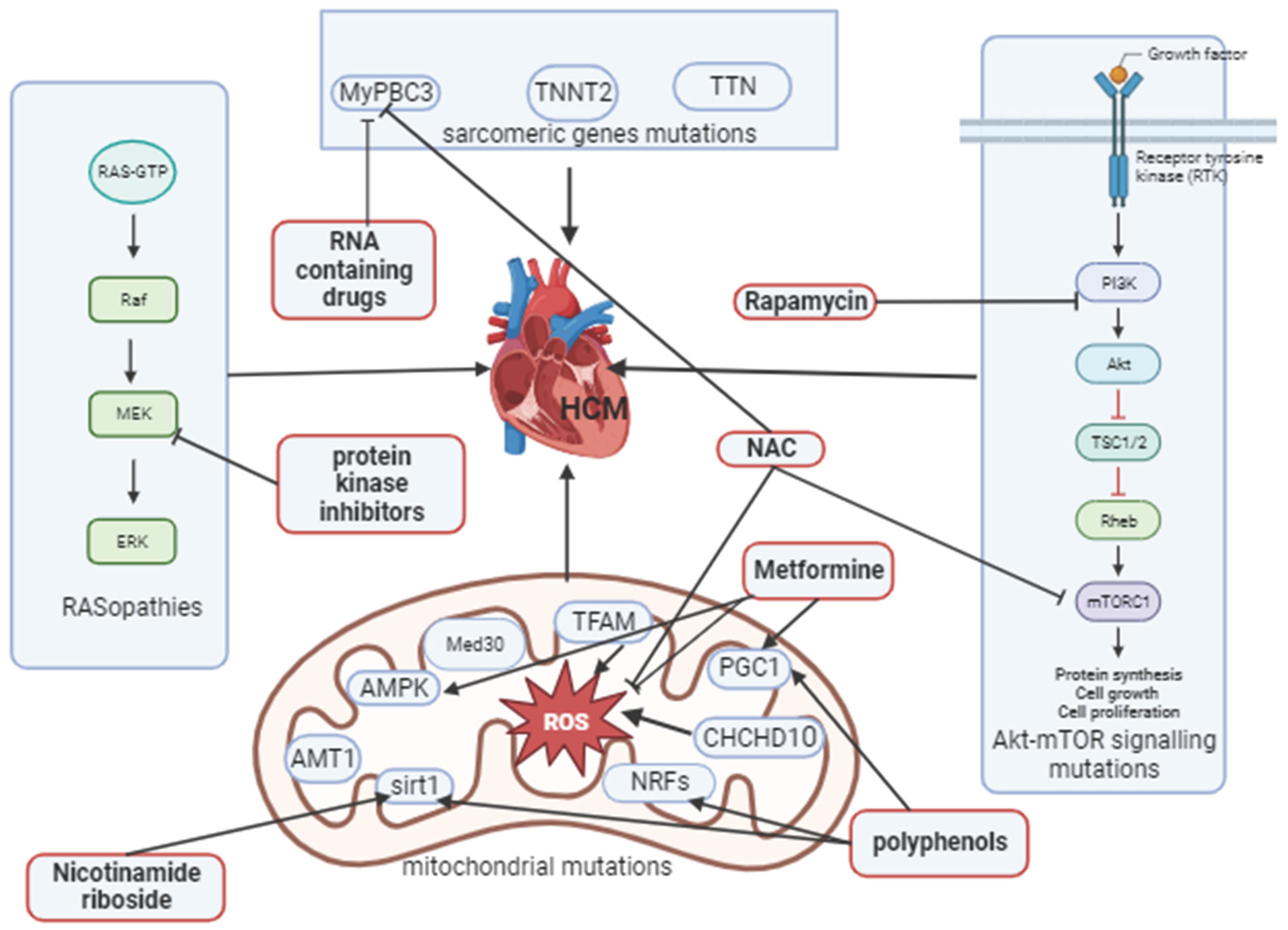
4. Hypertrophic cardiomyopathy, transcriptional signaling pathways and options for the treatment.
5. Conclusions and future perspectives.
Author Contributions
Funding
Conflicts of Interest
References
- Liu, L.; Hu, J.; Lei, H.; Qin, H.; Wang, C.; Gui, Y.; Xu, D. Regulatory T Cells in Pathological Cardiac Hypertrophy: Mechanisms and Therapeutic Potential. Cardiovasc. Drugs Ther. 2023, 37, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Shaw, R.M.; Nikolova, A.P. A Surprising Noncanonical Role for Calcineurin in Pressure-Induced Cardiac Hypertrophy. J. Am. Coll. Cardiol. 2018, 71, 668–669. [Google Scholar] [CrossRef] [PubMed]
- Mognol, G.; Carneiro, F.; Robbs, B.K.; Faget, B.V.; Viola, J.P.B. Cell cycle and apoptosis regulation by NFAT transcription factors: New roles for an old player. Cell Death Dis. 2016, 7, e2199. [Google Scholar] [CrossRef] [PubMed]
- Gunawan, F.; Gentile, A.; Gauvrit, S.; Didier YRBensimon-Brito SBensimon-Brito, A. NFATC1 Promotes Interstitial Cell Formation During Cardiac Valve Development in Zebrafish. Circ. Res. 2020, 126, 968. [Google Scholar] [CrossRef] [PubMed]
- Monaghan MG, Linneweh M, Liebscher S, Van Handel B, Layland SL, Schenke-Layland K. Endocardial-to-mesenchymal transformation and mesenchymal cell colonization at the onset of human cardiac valve development. Development. 2016 Feb 1;143(3):473-82. [CrossRef]
- Kamenshchyk A.V., Kamyshny A.M., Ivanko O.G. Gene expression of nuclear factor of activated T-cells in children with bicuspid aortic valve. Medical perspectives. 2016, V. 21, 3. - p. 29-33. http://nbuv.gov.ua/UJRN/Mp_2016_21_3_7.
- Kamenshchyk A, Gonchar M, Oksenych V, Kamyshnyi A. Association of Myocardial Changes and Gene Expression of the NFATC1 and NFATC4—Calcineurin Signaling Pathway in Children with Bicuspid Aortic Valve. Children. 2023; 10(9):1434. [CrossRef]
- Nakamura, M., Sadoshima, J. Mechanisms of physiological and pathological cardiac hypertrophy. Nat Rev Cardiol. 2018; 15, 387–407. [CrossRef]
- Marta Vigil-Garcia and others, Gene expression profiling of hypertrophic cardiomyocytes identifies new players in pathological remodelling, Cardiovascular Research, 2021; Volume 117, Issue 6, Pages 1532–1545. [CrossRef]
- Malay Chaklader, Beverly A. Rothermel. Calcineurin in the heart: New horizons for an old friend. Cellular Signalling. 2021; Volume 87, 110134. [CrossRef]
- Bazgir, F.; Nau, J.; Nakhaei-Rad, S.; Amin, E.; Wolf, M.J.; Saucerman, J.J.; Lorenz, K.; Ahmadian, M.R. The Microenvironment of the Pathogenesis of Cardiac Hypertrophy. Cells 2023; 12, 1780. [CrossRef]
- Janika Viereck and others, Targeting muscle-enriched long non-coding RNA H19 reverses pathological cardiac hypertrophy, European Heart Journal, 2020; Volume 41, Issue 36, 21, Pages 3462–3474.
- He, J., Luo, Y., Song, J., Tan, T., Zhu, H. Non-coding RNAs and Pathological Cardiac Hypertrophy. In: Xiao, J. (eds) Non-coding RNAs in Cardiovascular Diseases. Advances in Experimental Medicine and Biology, 2020; vol 1229. Springer, Singapore. [CrossRef]
- Zhang, X., Lei, F., Wang, X. M., Deng, K. Q., Ji, Y. X., Zhang, Y., et al. NULP1 Alleviates Cardiac Hypertrophy by Suppressing NFAT3 Transcriptional Activity. J. Am. Heart Assoc. 2020; 9 (16), e016419. [CrossRef]
- Zhou H, Xia C, Yang Y, Warusawitharana HK, Liu X, Tu Y. The Prevention Role of Theaflavin-3,3’-digallate in Angiotensin II Induced Pathological Cardiac Hypertrophy via CaN-NFAT Signal Pathway. Nutrients. 2022; 14(7):1391. [CrossRef]
- Yu Han, Jiali Nie, Dao Wen Wang, Li Ni. Mechanism of histone deacetylases in cardiac hypertrophy and its therapeutic inhibitors. Frontiers in Cardiovascular Medicine, 2022; Volume 9,. [CrossRef]
- Kolski-Andreaco A, Balut CM, Bertuccio CA, Wilson AS, Rivers WM, Liu X, Gandley RE, Straub AC, Butterworth MB, Binion D, Devor DC. Histone deacetylase inhibitors (HDACi) increase expression of KCa2.3 (SK3) in primary microvascular endothelial cells. Am J Physiol Cell Physiol. 2022; 322(3):C338-C353. [CrossRef]
- Chun P. Therapeutic effects of histone deacetylase inhibitors on heart disease. Arch Pharm Res. 2020; Dec;43(12):1276-1296. [CrossRef]
- Heimerl M, Sieve I, Ricke-Hoch M, Erschow S, Battmer K, Scherr M, Hilfiker-Kleiner D. Neuraminidase-1 promotes heart failure after ischemia/reperfusion injury by affecting cardiomyocytes and invading monocytes/macrophages. Basic Res Cardiol. 2020; Sep 25;115(6):62. PMID: 32975669; PMCID: PMC7519006. [CrossRef]
- Qian-Qian Chen and others, Neuraminidase 1 is a driver of experimental cardiac hypertrophy, European Heart Journal, 2021; Volume 42, Issue 36, 21, Pages 3770–3782. [CrossRef]
- Hairuo Lin and others, Lansoprazole alleviates pressure overload-induced cardiac hypertrophy and heart failure in mice by blocking the activation of β-catenin, Cardiovascular Research, 2020; Volume 116, Issue 1, Pages 101–113. [CrossRef]
- Dimitiru L, Dimitiru A, Stamatin M. Hypoxic Perinatal Cardiomyopathy-Diagnosis and Evolution Archives of Disease in Childhood. 2012; 97: A331. [CrossRef]
- L. Dimitriu , A.G. Dimitriu, Particular aspects of diagnosis and evolution of hypoxic myocardial injury in newborn infant, European Heart Journal, 2013; Volume 34, Issue suppl_1, P3011. [CrossRef]
- L. Dimitriu , A.G. Dimitriu, Detection of myocardial injury in perinatal asphyxia by echocardiography and cardiac biomarkers. Archives of Cardiovascular Diseases Supplements. 2021;Volume 13, Issue 1, 2021, Page 144. [CrossRef]
- Aye, C., Lewandowski, A., Lamata, P. et al. Disproportionate cardiac hypertrophy during early postnatal development in infants born preterm. Pediat Res. 2017; 82, 36–46. [CrossRef]
- Hutter D, Kingdom J, Jaeggi E. Causes and Mechanisms of Intrauterine Hypoxia and Its Impact on the Fetal Cardiovascular System: A Review. Int J Pediatr. 2010; [CrossRef]
- Chen, X., Zhang, L. & Wang, C. Prenatal hypoxia-induced epigenomic and transcriptomic reprogramming in rat fetal and adult offspring hearts. Sci Data. 2019;6,238. [CrossRef]
- Romanowicz J, Guerrelli D, Dhari Z, Mulvany C, Reilly M, Swift L, Vasandani N, Ramadan M, Leatherbury L, Ishibashi N, Posnack NG. Chronic perinatal hypoxia delays cardiac maturation in a mouse model for cyanotic congenital heart disease. Am J Physiol Heart Circ Physiol. 2021; 320(5):H1873-H1886. [CrossRef]
- Lei Huang, Xin Chen, Chiranjib Dasgupta, Wanqiu Chen, Rui Song, Charles Wang, Lubo Zhang, Foetal hypoxia impacts methylome and transcriptome in developmental programming of heart disease, Cardiovascular Research, 2019; Volume 115, Issue 8, 2019, Pages 1306–1319. [CrossRef]
- Paradis AN, Gay MS, Wilson CG, Zhang L Newborn Hypoxia/Anoxia Inhibits Cardiomyocyte Proliferation and Decreases Cardiomyocyte Endowment in the Developing Heart: Role of Endothelin-1. PLoS ONE. 2015; 10(2): e0116600. [CrossRef]
- Popazova O, Belenichev I, Abramov A, Bukhtiyarova N, Chereshniuk I, Skoryna D. Indicators of Bioelectrical Activity of the Rat Heart After Prenatal Hypoxia and Pharmacological Correction. Innov Biosyst Bioeng [Internet]. 2023; 6(3-4):148-60. Available from: http://ibb.kpi.ua/article/view/268504. /.
- Ferreiro CR, Chagas AC, Carvalho MH, Dantas AP, Jatene MB, Bento De Souza LC, Lemos Da Luz P. Influence of hypoxia on nitric oxide synthase activity and gene expression in children with congenital heart disease: a novel pathophysiological adaptive mechanism. Circulation. 2001 May 8;103(18):2272-6. [CrossRef]
- Belenichev I,. Abramov A. , Puzyrenko A., et al. Molecular mechanisms of myocardial damage in the hypertensive rats and hypertensive rats with metabolic disorders (diabetes mellitus, atherosclerosis). RRP. 2022; Vol.8 (4):25-33. [CrossRef]
- Niu, Y.; Kane, A.D.; Lusby, C.M.; Allison, B.J.; Chua, Y.Y.; Kaandorp, J.J.; Nevin-Dolan, R.; Ashmore, T.J.; Blackmore, H.L.; Derks, J.B.; et al. Maternal Allopurinol Prevents Cardiac Dysfunction in Adult Male Offspring Programmed by Chronic Hypoxia During Pregnancy. Hypertension. 2018, 72, 971–978. [Google Scholar] [CrossRef]
- Patterson AJ, Zhang L. Hypoxia and fetal heart development. Curr Mol Med. 2010; 10(7):653-66. [CrossRef]
- Hauton, D. Ousley, V. Prenatal hypoxia induces increased cardiac contractility on a background of decreased capillary density. BMC Cardiovasc Disord. 2009, 9, 1. [Google Scholar] [CrossRef] [PubMed]
- Hula, N.; Liu, R.; Spaans, F.; Pasha, M.; Quon, A.; Kirschenman, R.; Cooke, C.-L.M.; Davidge, S.T. The Long-Term Effects of Prenatal Hypoxia on Coronary Artery Function of the Male and Female Offspring. Biomedicines. 2022, 10, 3019. [Google Scholar] [CrossRef] [PubMed]
- De Pascali F, Hemann C, Samons K, Chen CA, Zweier JL. Hypoxia and reoxygenation induce endothelial nitric oxide synthase uncoupling in endothelial cells through tetrahydrobiopterin depletion and S-glutathionylation. Biochemistry. 2014;53(22):3679-88. [CrossRef]
- Zhang, Z., Yao, L., Yang, J., Wang, Z., & Du, G. PI3K/Akt and HIF-1 signaling pathway in hypoxia-ischemia (Review). Molecular Medicine Reports, 2018;18, 3547-3554. [CrossRef]
- Jung F, Palmer LA, Zhou N, Johns RA. Hypoxic regulation of inducible nitric oxide synthase via hypoxia inducible factor-1 in cardiac myocytes. Circ Res. 2000;86(3):319-25. [CrossRef]
- Belenichev I, Gorbachova S, Pavlov S, Bukhtiyarova N, Puzyrenko A, Brek O. Neurochemical status of nitric oxide in the settings of the norm, ishemic event of central nervous system, and pharmacological intervention. Georgian Med News. 2021; (315):169-176. [PubMed]
- Dattilo, S., Mancuso, C., Koverech, G. et al. Heat shock proteins and hormesis in the diagnosis and treatment of neurodegenerative diseases. Immun Ageing, 2015; 12, 20. [CrossRef]
- Belenichev IF, Aliyeva OG, Popazova OO, Bukhtiyarova NV. Involvement of heat shock proteins HSP70 in the mechanisms of endogenous neuroprotection: the prospect of using HSP70 modulators. Front Cell Neurosci. 2023;17:1131683. [CrossRef]
- Bogdan C. Nitric oxide and the regulation of gene expression. Trends Cell Biol. 2001; 11(2):66-75. [CrossRef]
- Kröncke KD. Nitrosative stress and transcription. Biol Chem. 2003;384(10-11):1365-77. [CrossRef]
- Bielenichev IF., Vizir VA.,, Mamchur VYo., Kuriat OV. Place of tiotriazoline in the gallery of modern metabolitotropic medicines. February. Zaporozhye Medical Journal. 2019; 21(1),118-128. [CrossRef]
- Anavi S, Tirosh O. iNOS as a metabolic enzyme under stress conditions. Free Radic Biol Med. 2020;146:16-35. [CrossRef]
- Hsu CN, Tain YL. Impact of Arginine Nutrition and Metabolism during Pregnancy on Offspring Outcomes. Nutrients. 2019; 11(7):1452. [CrossRef]
- Bednov A, Espinoza J, Betancourt A, Vedernikov Y, Belfort M, Yallampalli C. L-arginin prevents hypoxia-induced vasoconstriction in dual-perfused human placental cotyledons. Placenta. 2015;36(11):1254-9. [CrossRef]
- Kwon SH, Pimentel DR, Remondino A, Sawyer DB, Colucci WS. H(2)O(2) regulates cardiac myocyte phenotype via concentration-dependent activation of distinct kinase pathways. J Mol Cell Cardiol. 2003;35(6):615-21. [CrossRef]
- Amin JK, Xiao L, Pimental DR, Pagano PJ, Singh K, Sawyer DB, Colucci WS. Reactive oxygen species mediate alpha-adrenergic receptor-stimulated hypertrophy in adult rat ventricular myocytes. J Mol Cell Cardiol. 2001;33(1):131-9. [CrossRef]
- Hirotani S, Otsu K, Nishida K, Higuchi Y, Morita T, Nakayama H, Yamaguchi O, Mano T, Matsumura Y, Ueno H, Tada M, Hori M. Involvement of nuclear factor-kappaB and apoptosis signal-regulating kinase 1 in G-protein-coupled receptor agonist-induced cardiomyocyte hypertrophy. Circulation. 2002;105(4):509-15. [CrossRef]
- Belenichev IF, Bak1 PG., Popazova OO, Bukhtiyarova N, Yadlovsky OE. Nitric oxide- dependent mechanism of endothelial dysfunction formation is a promising target link for pharmacological management I Biopolymers and Cell. 2022 Vol. 38. N 3. P 145–157.
- Belenichev IF, Nagornaya EA, Gorbacheva SV, Gorchakova NA, Bukhtiyarova NV. Thiol-disulfide system: role in endogenous cyto-and organoprotection, pathways of pharmacological modulation. 2020; Euston Publishing House, Kiev, 232.
- Lipshultz SE, Law YM, Asante-Korang A, Austin ED, Dipchand AI, Everitt MD, et al. Cardiomyopathy in children: classification and diagnosis: a scientific statement from the American heart association. Circulation. 2019;140:e9–68. [CrossRef]
- Kumar S, Kumar V, Kim JJ. Sarcomeric Gene Variants and Their Role with Left Ventricular Dysfunction in Background of Coronary Artery Disease. Biomolecules. 2020; 12;10(3):442. [CrossRef]
- Ramírez CD, Padrón R. Cardiomiopatia hipertrófica familiar: genes, mutaciones y modelos animales. Revisión Familial hypertrophic cardiomyopathy: genes, mutations and animal models. A review. Invest Clin. 2004;45(1):69-99. Spanish. PMID: 15058760.
- Mearini G, Stimpel D, Geertz B, Weinberger F, Krämer E, Schlossarek S, Mourot-Filiatre J, Stoehr A, Dutsch A, Wijnker PJ, Braren I, Katus HA, Müller OJ, Voit T, Eschenhagen T, Carrier L. Mybpc3 gene therapy for neonatal cardiomyopathy enables long-term disease prevention in mice. Nat Commun. 2014; 2;5:5515. [CrossRef]
- Jiang J, Wakimoto H, Seidman JG, Seidman CE. Allele - specific silencing of mutant Myh6 transcripts in mice suppresses hypertrophic cardiomyopathy. Science. 2013; 342(6154):111-4. [CrossRef]
- Monda E, Rubino M, Lioncino M, Di Fraia F, Pacileo R, Verrillo F, Cirillo A, Caiazza M, Fusco A, Esposito A, Fimiani F, Palmiero G, Pacileo G, Calabrò P, Russo MG, Limongelli G. Hypertrophic Cardiomyopathy in Children: Pathophysiology, Diagnosis, and Treatment of Non-sarcomeric Causes. Front Pediatr. 2021;25;9:632293. [CrossRef]
- Pandit B, Sarkozy A, Pennacchio LA, Carta C, Oishi K, Martinelli S, et al. Gain-of-function RAF1 mutations cause Noonan and LEOPARD syndromes with hypertrophic cardiomyopathy. Nat Genet. 2007; 39:1007–12. [CrossRef]
- Calcagni G, Adorisio R, Martinelli S, Grutter G, Baban A, Versacci P, et al. Clinical presentation and natural history of hypertrophic cardiomyopathy in rasopathies. Heart Fail Clin. 2018; 14:225–35. [CrossRef]
- Wu X, Simpson J, Hong JH, Kim KH, Thavarajah NK, Backx PH, et al. MEK-ERK pathway modulation ameliorates disease phenotypes in a mouse model of Noonan syndrome associated with the Raf1(L613V) mutation. J Clin Invest. 2011; 121:1009–25. [CrossRef]
- Marin TM, Keith K, Davies B, Conner DA, Guha P, Kalaitzidis D, et al. Rapamycin reverses hypertrophic cardiomyopathy in a mouse model of LEOPARD syndrome-associated PTPN11 mutation. J Clin Invest. 2011; 121:1026–43. [CrossRef]
- Hahn A, Lauriol J, Thul J, Behnke-Hall K, Logeswaran T, Schänzer A, Böğürcü N, Garvalov BK, Zenker M, Gelb BD, von Gerlach S, Kandolf R, Kontaridis MI, Schranz D. Rapidly progressive hypertrophic cardiomyopathy in an infant with Noonan syndrome with multiple lentigines: palliative treatment with a rapamycin analog. Am J Med Genet A. 2015;167A(4):744-51. [CrossRef]
- Zhang J, Cao L, Tan Y, Zheng Y, Gui Y. N-acetylcysteine protects neonatal mice from ventricular hypertrophy induced by maternal obesity in a sex-specific manner. Biomed Pharmacother. 2021;133:110989. [CrossRef]
- Zafarullah M, Li WQ, Sylvester J, Ahmad M. Molecular mechanisms of N-acetylcysteine actions. Cell Mol Life Sci. 2003; 60(1):6-20. [CrossRef]
- Craven L, Alston CL, Taylor RW, Turnbull DM. Recent Advances in Mitochondrial Disease. Annu Rev Genomics Hum Genet. 2017; 31;18:257-275. [CrossRef]
- Brunel-Guitton C, Levtova A, Sasarman F. Mitochondrial Diseases and Cardiomyopathies. Can J Cardiol. 2015;31(11):1360-76. [CrossRef]
- Menezes MJ, Riley LG, Christodoulou J. Mitochondrial respiratory chain disorders in childhood: insights into diagnosis and management in the new era of genomic medicine. Biochim Biophys Acta. 2014;1840(4):1368-79. [CrossRef]
- Yang, J.; Chen, S. Duan, F., Wang, X., Zhang, X., Lian, B.; Kou, M., Chiang, Z., Li, Z., Lian, Q. Mitochondrial Cardiomyopathy: Molecular Epidemiology, Diagnosis, Models, and Therapeutic Management. Cells 2022, 11, 3511. [Google Scholar] [CrossRef] [PubMed]
- Driver, Christin, Bamitale, Kayode D. S., Kazi, Aniessa, Olla, Mehnaaz, Nyane, Ntsoaki A. B., Owira, Peter M. O. Cardioprotective Effects of Metformin. Journal of Cardiovascular Pharmacology. 2018; 72(2): p.121-127, August. [CrossRef]
- Dziubak A, Wójcicka G, Wojtak A, Bełtowski J. Metabolic Effects of Metformin in the Failing Heart. Int J Mol Sci. 2018; Sep 21;19(10):2869. [CrossRef]
- Li C, Mu N, Gu C, Liu M, Yang Z, Yin Y, Chen M, Wang Y, Han Y, Yu L, Ma H. Metformin mediates cardioprotection against aging-induced ischemic necroptosis. Aging Cell. 2020;19(2):e13096. [CrossRef]
- Zhu, Y., Zhu, L., Wang, X. et al. RNA-based therapeutics: an overview and prospectus. Cell Death Dis. 2022; 13, 644. [CrossRef]
- Palandri C, Santini L, Argirò A, Margara F, Doste R, Bueno-Orovio A, Olivotto I, Coppini R. Pharmacological Management of Hypertrophic Cardiomyopathy: From Bench to Bedside. Drugs. 2022; Jun;82(8):889-912. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



