Submitted:
08 February 2024
Posted:
08 February 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
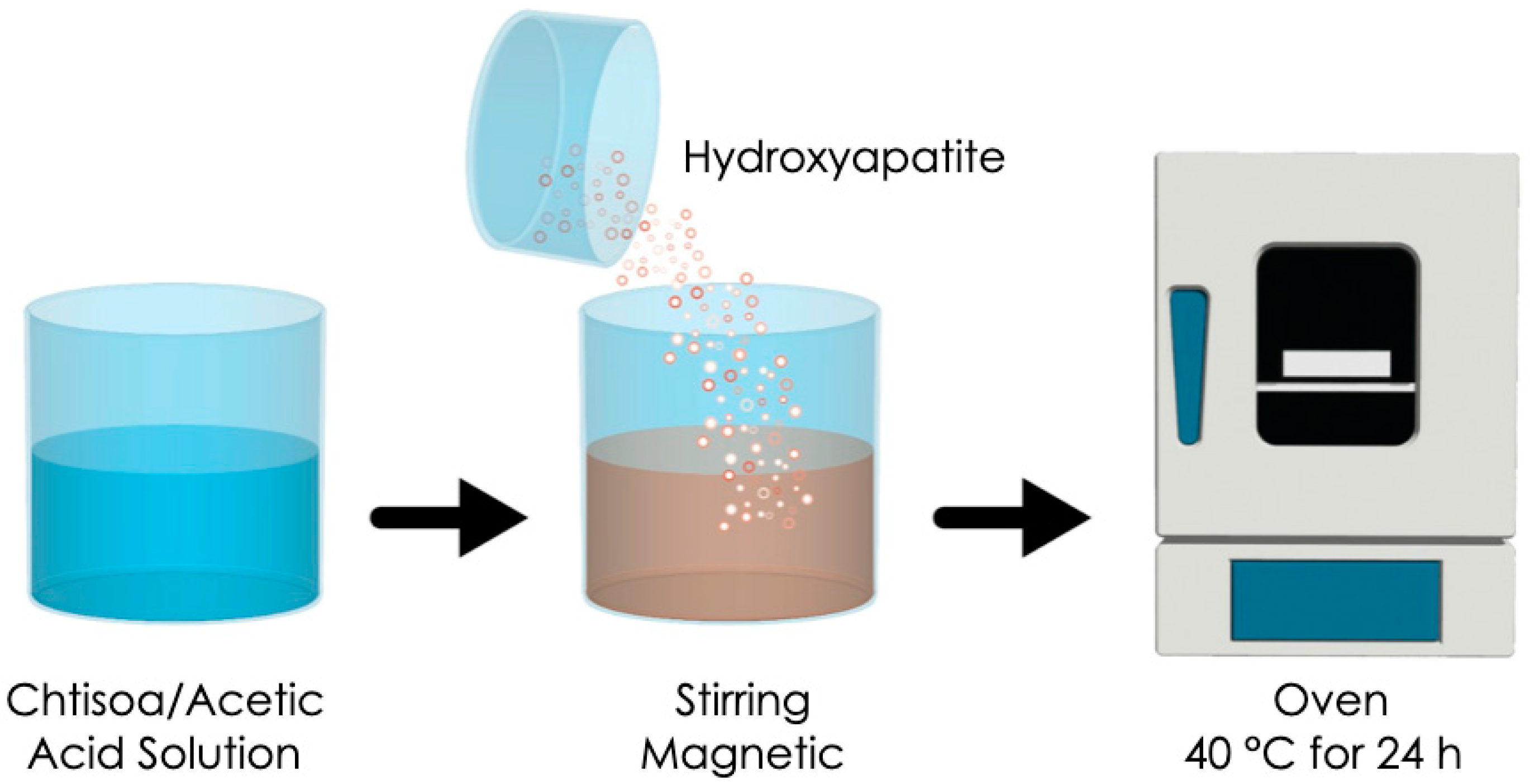
2. Materials and Methods
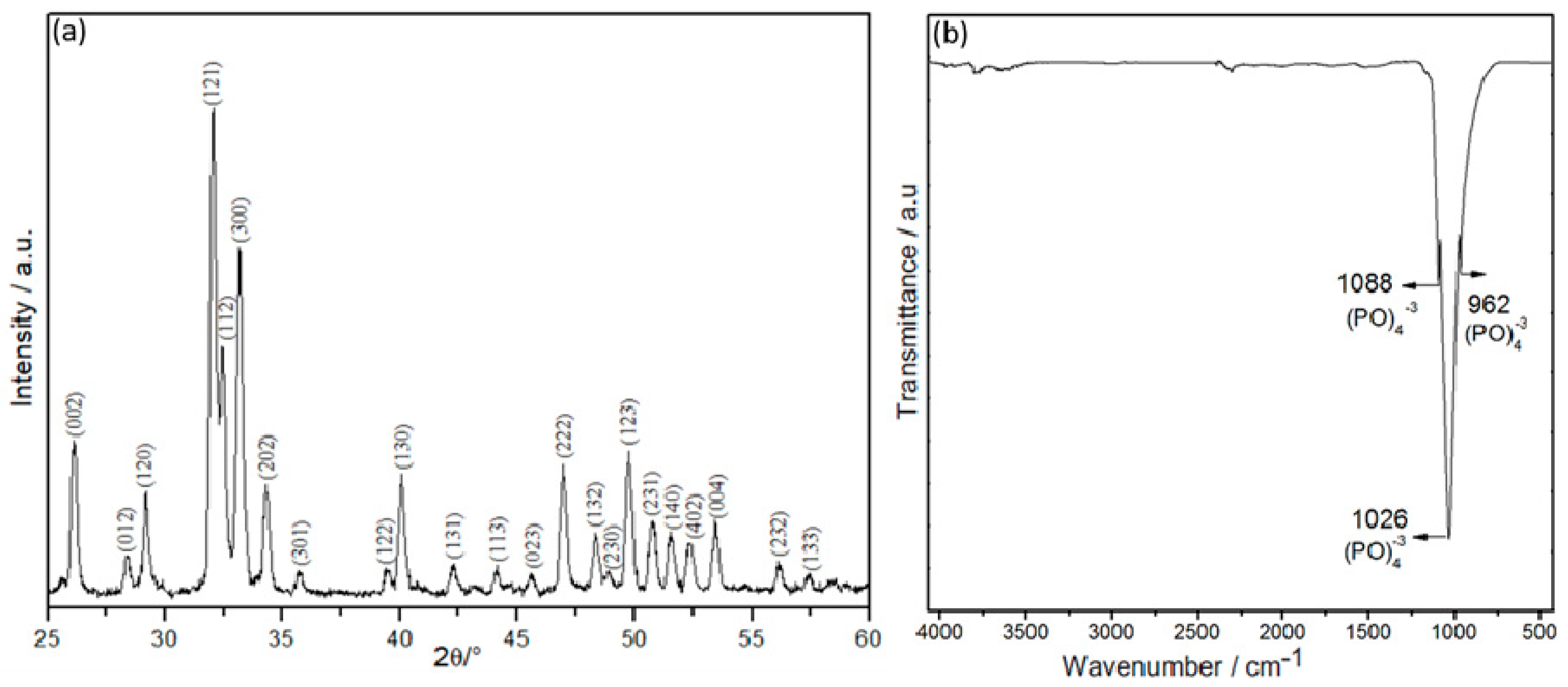
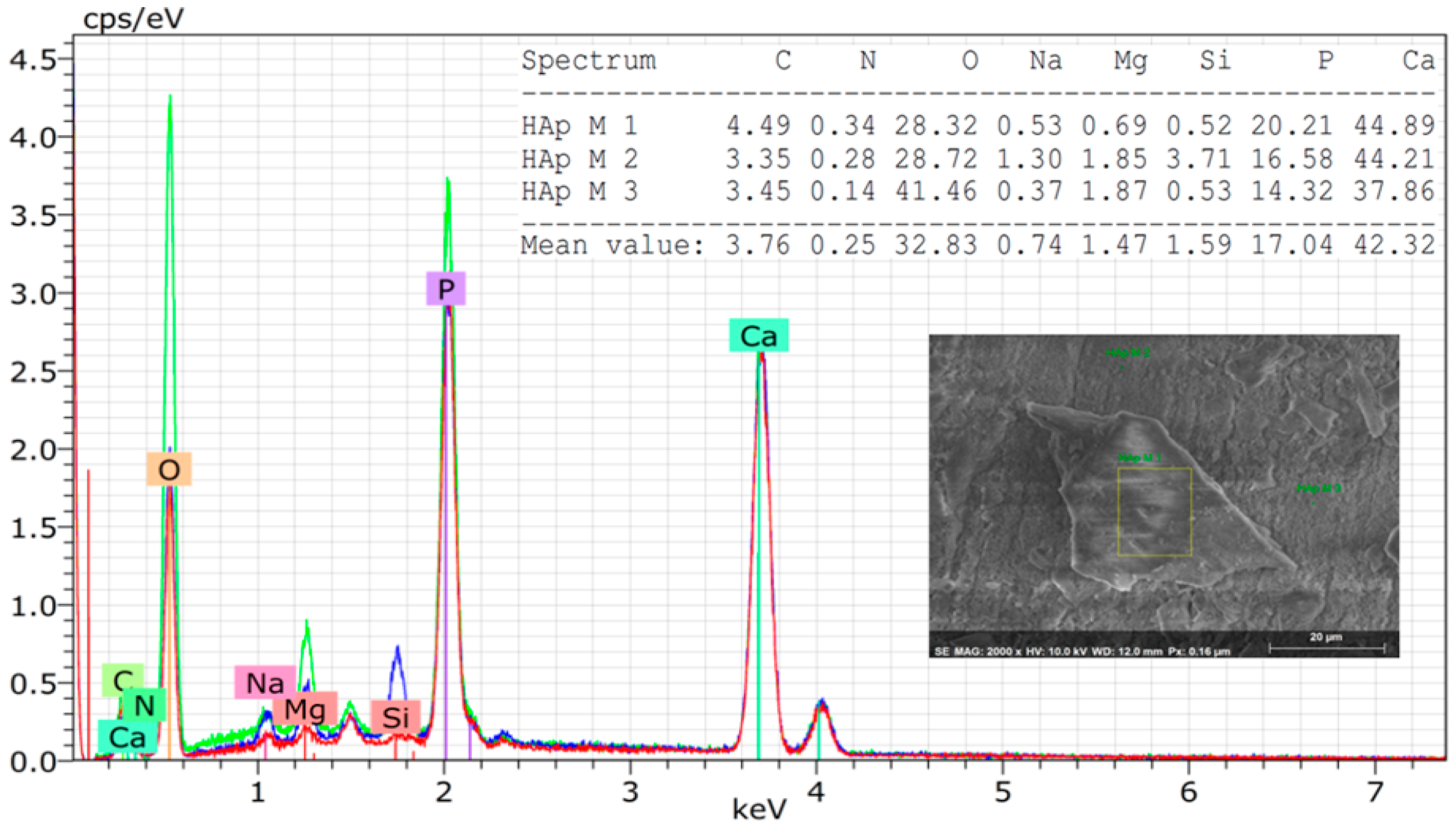
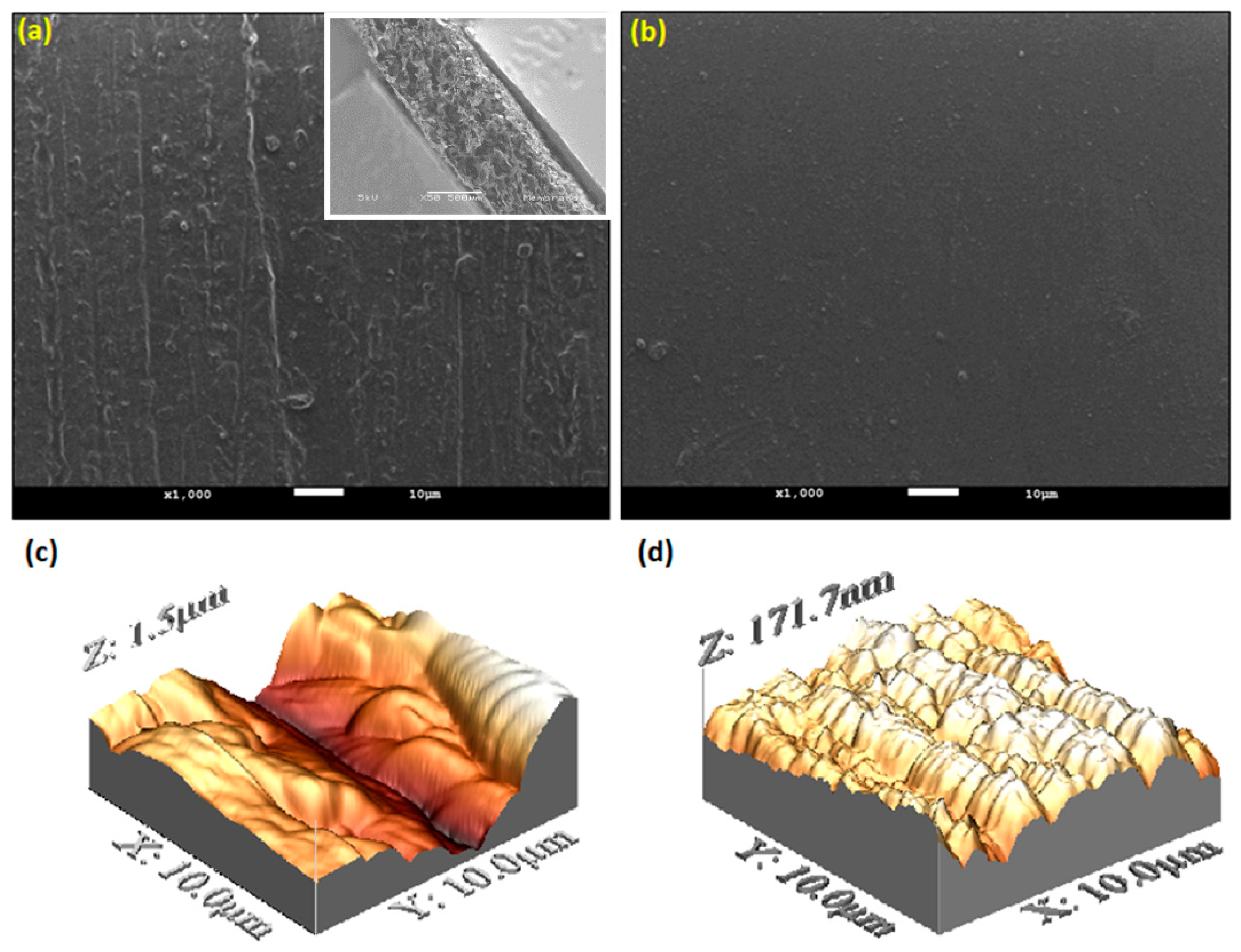
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Wakida-Kusunoki, A.T; Ruiz-Carus, R.; Amador del Angel, L.E. Amazon sailfin catfish, Pterygoplichthys pardalis (Castelnau, 1855) (Loricariidae), another exotic species established in Southeastern Mexico. Southwestern Natur. 2007, 52, 141–144. [Google Scholar] [CrossRef]
- Wu, L. W.; Liu, C. C.; Lin, S. M. Identification of exotic sailfin catfish species (Pterygoplichthys, Loricariidae) in Taiwan based on morphology and mtDNA sequences. Zoolog. St. 2011, 50, 235–246. [Google Scholar] [CrossRef] [PubMed]
- Capps, K.A.; Nico, L.G.; Mendoza-Carranza, M.; Arévalo-Frías, W.; Ropicki, A.; Heilpern, S.A.; Rodiles-Hernández, R. Salinity tolerance of non-native suckermouth armoured catfish (Loricariidae: Pterygoplichthys) in Southeastern Mexico: Implications for invasion and dispersal. Aqu. Cons.: Mar. Freshw. Ecosyst. 2011, 21, 528–540. [Google Scholar] [CrossRef]
- Hoover, J.J.; Killgore, K.J.; Cofrancesco, A.F. Suckermouth catfishes: Threats to aquatic ecosystems of the United States? Aqu. Nuis. Spec Res. Bull. 2004, 4, 1–9. [Google Scholar]
- Cheng, H.; Chabok, R.; Guan, X.; Chawla, A.; Li, Y. Synergistic interplay between the two major bone minerals, hydroxyapatite and whitlockite nanoparticles, for osteogenic differentiation of mesenchymal stem cells. Acta Biomater. 2018, 69, 342–351. [Google Scholar] [CrossRef] [PubMed]
- Bee, S.; Hamid, Z.A.A. Characterization of chicken bone waste-derived hydroxyapatite and its functionality on chitosan membrane for guided bone regeneration. Compos. Part B. 2019, 163, 562–73. [Google Scholar] [CrossRef]
- De Tullio, I.; Caputi, S.; Perfetti, G.; Mavriqi, L.; Wismeijer, D.; Traini, T. A Human Clinical and Histomorphometrical Study on Different Resorbable and Non-Resorbable Bone Substitutes Used in Post-Extractive Sites. Preliminary Results. Materials (Basel). 2019, 12, 2408. [Google Scholar] [CrossRef]
- Monika, Š. Substituted hydroxyapatites for biomedical applications: A review. Ceram. Int. 2015, 41, 9203–9231. [Google Scholar] [CrossRef]
- Luna-Domínguez, J.H.; Téllez-Jiménez, H.; Hernández-Cocoletzi, H.; García-Hernández, M.; Melo-Banda, J.A. , Nygren, H. Development and in vivo response of hydroxyapatite/whitlockite from chicken bones as bone substitute using a chitosan membrane for guided bone regeneration. Ceram. Int. 2018, 44, 22583–22591. [Google Scholar] [CrossRef]
- Mondal, S.; Mondal, B.; Dey, A.; Mukhopadhyay, S.S. Studies on Processing and Characterization of Hydroxyapatite Bio-materials from Different Bio Wastes. J. Miner. Mater. Charact. Eng. 2012, 11, 55–67. [Google Scholar] [CrossRef]
- Saber-Samandari, S.; Saber-Samandari, S.; Ghonjizade-Samani, F.; Aghazadeh, J.; Sadeghi, A. Bioactivity evaluation of novel nanocomposite scaffolds for bone tissue engineering: The impact of hydroxyapatite. Ceram. Int. 2016, 42, 11055–11062. [Google Scholar] [CrossRef]
- Pon-On, W.; Suntornsaratoon, P.; Charoenphandhu, N.; Thongbunchoo, J.; Krishnamra, N.; Tang, I.M. Hydroxyapatite from the fish scale for potential use as a bone scaffold or regenerative material. Mater. Sci. Eng. C. 2016, 62, 183–189. [Google Scholar] [CrossRef]
- Kamalanathan, P.; Ramesh, S.; Bang, L.T.; Niakan, A.; Tan, C.Y.; Purbolaksono, J. Synthesis and sintering of hydroxyapatite derived from eggshells as a calcium precursor. Ceram Int. 2014, 40, 16349–16359. [Google Scholar] [CrossRef]
- Elgali, I.; Omar, O.; Dahlin, C.; Guided, T.P. Guided bone regeneration: materials and biological mechanisms revisited. Eur J Oral Sci. 2017, 315–37. [Google Scholar] [CrossRef]
- Irinakis, T. Rationale for socket preservation after extraction of a single-rooted tooth when planning for future implant placement. J Can Dent Assoc (Tor). 2007, 72, 917–22. [Google Scholar]
- Fenbo, M.; Xingyu. X.; Bin, T. Strontium chondroitin sulfate/silk fibroin blend membrane containing microporous structure modulates macrophage responses for guided bone regeneration. Carbohydr. Polym. 2019, 213, 266–75. [Google Scholar] [CrossRef]
- Rocchietta, I.; Simion, M.; Hoffmann, M.; Trisciuoglio, D.; Benigni, M.; Dahlin, C. Vertical Bone Augmentation with an Autogenous Block or Particles in Combination with Guided Bone Regeneration : A Clinical and Histological Preliminary Study in Humans. Clin Implant Dent Relat Res. 2016, 18, 19–29. [Google Scholar] [CrossRef]
- Wang, J.; Wang, L.; Zhou, Z.; Lai, H.; Xu, P.; Liao, L.; Wei, J. Biodegradable Polymer Membranes Applied in Guided Bone/Tissue Regeneration: A Review. Polym. 2016, 8, 115–135. [Google Scholar] [CrossRef]
- Atrian, M.; Kharaziha, M.; Emadi, R.; Alihosseini, F. Silk-Laponite® fibrous membranes for bone tissue engineering. Appl Clay Sci. 2019, 174, 90–9. [Google Scholar] [CrossRef]
- Stankovic, D.; Labudovic-Borovic, M.; Radosavljevic, R.; Marinkovic, M.; Isenovic, E.R. Use of acellular collagen matrix for the closure of the open oral wound in bone regeneration. J. Stomatol. Oral Maxillofac. Surg. 2018, 119, 446–9. [Google Scholar] [CrossRef]
- Masoudi Rad, M.; Nouri Khorasani, S.; Ghasemi-Mobarakeh, L.; Prabhakaran, M.P.; Foroughi, M.R.; Kharaziha, M.; Saadat-kish, N.; Ramakrishna, S. Fabrication and characterization of two-layered nanofibrous membrane for guided bone and tissue regeneration application. Mater. Sci. Eng. C. 2017, 80, 75–87. [Google Scholar] [CrossRef] [PubMed]
- Arancibia, R.; Marutana, C.; Silva, D.; Tobar, N.; Tapia, C.; Salazar, J.C.; Martínez, J.; Smith, P.C. Effects of Chitosan Particles in Periodontal Pathogens and Gingival Fibroblasts. J. Dent. Res. 2013, 92, 740–5. [Google Scholar] [CrossRef] [PubMed]
- Venkatesan, J.; Kim, S.K. Chitosan composites for bone tissue engineering - An overview. Mar. Drugs. 2010, 8, 2252–66. [Google Scholar] [CrossRef] [PubMed]
- Acevedo, C.A.; Olguín, Y.; Briceño, M.; Forero, J.C.; Osses, N.; Díaz-Calderón, P.; Jaques, A.; Ortiz, R. Design of a biodegradable UV-irradiated gelatin-chitosan/nanocomposed membrane with osteogenic ability for application in bone regen-eration. Mater. Sci. Eng. C. 2019, 99, 875–86. [Google Scholar] [CrossRef] [PubMed]
- Munhoz, M.A.S.; Hirata, H.H.; Plepis, A.M.G.; Martins, V.C.A. , Cunha, M.R. Use of collagen/chitosan sponges mineralized with hydroxyapatite for the repair of cranial defects in rats. Injury. 2018, 49, 2154–60. [Google Scholar] [CrossRef]
- Pereira, I.C.; Duarte, A.S.; Neto, A.S.; Ferreira, J.M.F. Chitosan and polyethylene glycol-based membranes with antibacterial properties for tissue regeneration. Mater. Sci. Eng. C. 2019, 96, 606–15. [Google Scholar] [CrossRef]
- Cheng, Y.; Hu, Z.; Zhao, Y.; Zou, Z.; Lu, S.; Zhang, B.; Li, S. Sponges of Carboxymethyl Chitosan Grafted with Collagen Peptides for Wound Healing. Int. J. Mol. Sci. 2019, 20, 3890–3902. [Google Scholar] [CrossRef]
- Chen, P.; Liu, L.; Pan, J.; Mei, J.; Li, C.; Zheng, Y. Biomimetic composite scaffold of hydroxyapatite/gelatin-chitosan core-shell nanofibers for bone tissue engineering. Mater. Sci. Eng. C. 2019, 97, 325–35. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Tai, H.; Fu, E.; Don, T. Guided bone regeneration activity of different calcium phosphate/chitosan hybrid membranes. Int J Biol Macromol. 2019, 126, 159–69. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.S.; Baek, S.D.; Venkatesan, J.; Bhatnagar, I.; Chang, H.K.; Kim, H.T.; Kim, S.K. In vivo study of chitosan-natural nano-hydroxyapatite scaffolds for bone tissue regeneration. Int J Biol Macromol. 2014, 67, 360–6. [Google Scholar] [CrossRef]
- Park, K.; Yun, Y.; Kim, S.E.; Song, H. The Effect of Alendronate Loaded Biphasic Calcium Phosphate Scaffolds on Bone Regeneration in a Rat Tibial Defect Model. Int. J. Mol. Sci. 2015, 16, 26738–26753. [Google Scholar] [CrossRef]
- Saigo, L.; Kumar, V.; Liu, Y.; Lim, J.; Teoh, S.H.; Goh, B.T. A pilot study: Clinical efficacy of novel polycaprolactone-tricalcium phosphate membrane for guided bone regeneration in rabbit calvarial defect model. J. Oral Maxillofac. Surgery, Med. Pathol. 2018, 30, 212–219. [Google Scholar] [CrossRef]
- Poinern, G.J.E.; Brundavanam, R.; Le. X.T.; Djordjevic, S.; Prokic, M.; Fawcett, D. Thermal and ultrasonic influence in the formation of nanometer scale hydroxyapatite bio-ceramic. Int. J. Nanomedicine 2011, 2011, 2083–2095. [Google Scholar] [CrossRef] [PubMed]
- Venkatesan, J.; Kim, S.K. Effect of Temperature on Isolation and Characterization of Hydroxyapatite from Tuna (Thunnus obesus) Bone. Mater. 2010, 3, 4761–4772. [Google Scholar] [CrossRef]
- Camargo, H.A.N.; de Lima, S.A.; Gemelli, E. Synthesis and Characterization of Hydroxyapatite/TiO2n Nanocomposites for Bone Tissue Regeneration. Am. J. Biomed. Eng. 2012, 2, 41–7. [Google Scholar] [CrossRef]
- Bee, S.L.; Mariatti, M.; Ahmad, N.; Yahaya, B.H.; Hamid, Z.A.A. Effect of the calcination temperature on the properties of natural hydroxyapatite derived from chicken bone wastes. Mater. Today Proc. 2019, 16, 1876–85. [Google Scholar] [CrossRef]
- Sunil, B.R.; Jagannatham, M. Producing hydroxyapatite from fish bones by heat treatment. Mater. Lett. 2016, 185, 411–4. [Google Scholar] [CrossRef]
- Shi, P.; Liu, M.; Fan, F.; Yu, C.; Lu, W.; Du, M. Characterization of natural hydroxyapatite originated from fish bone and its biocompatibility with osteoblasts. Mater. Sci. Eng. C. 2018, 90, 706–12. [Google Scholar] [CrossRef]
- Zainol, I.; Adenan, N.H.; Rahim, N.A.; Jaafar, C.N.A. Extraction of natural hydroxyapatite from tilapia fish scales using alkaline treatment. Mater Today Proc. 2019, 16, 1942–1948. [Google Scholar] [CrossRef]
- Henríquez-Távara, N.; García-Molina, J.A.; Machado, G.; Carvalho-Lobato, P.; Belmonte-Calderón, A.; Serra-Renom, I.; Manzanares-Céspedes, M.C. Análisis semicuantitativo del calcio y fósforo en los tejidos calcificados de la mandíbula. Biomec. 2002, 10, 5–13. [Google Scholar] [CrossRef]
- Mohd Pu’ad, N.A.S.; Koshy, P.; Abdullah, H.Z.; Idris, M.I.; Lee, T.C. Syntheses of hydroxyapatite from natural sources. Heli-yon. 2019, 5, e01588. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Q.; Ablikim, Z.; Chen, T.; Cai, Q.; Xia, J.; Jiang, D.; Wang, S. The preparation and characterization of HA/β-TCP biphasic ceramics from fish bones. Ceram Int. 2017, 43, 12213–20. [Google Scholar] [CrossRef]
- Tai, H.; Fu, E.; Don, T. Calcium phosphates synthesized by reverse emulsion method for the preparation of chitosan composite membranes. Carbohydr Polym. 2012, 88, 904–11. [Google Scholar] [CrossRef]
- Chu, C.; Deng. J.; Xiang, L.; Wu, Y.; Wei, X.; Qu, Y.; Man, Y. Evaluation of epigallocatechin-3-gallate (EGCG) cross-linked collagen membranes and concerns on osteoblasts. Mater. Sci. Eng. C. 2016, 67, 386–94. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, S.; Bagchi, B.; Bhandary, S.; Kool, A.; Hoque, N.A.; Biswas, P.; Pal, K.; Thakuy, P.; Das, K.; Karmakar, P.; Das, S. Antimicrobial and biocompatible fluorescent hydroxyapatite-chitosan nanocomposite films for biomedical applications. Coll. Surf. B: Bioint. 2018, 171, 300–307. [Google Scholar] [CrossRef]
- Matinfar, M.; Mesgar, A.S.; Mohammadi, Z. Evaluation of physicochemical, mechanical and biological properties of chitosan/carboxymethyl cellulose reinforced with multiphasic calcium phosphate whisker-like fibers for bone tissue engineering. Mater. Sci. Eng. C. 2019, 100, 341–53. [Google Scholar] [CrossRef]
- Félix, A.; de Almeida Filho, E.; da Silva Rigo, E.C. , Ortega Boschi, A. Synthesis of chitosan/hydroxyapatite membranes coated with hydroxycarbonate apatite for guided tissue regeneration purposes. Appl. Surf. Sci. 2011, 257, 3888–92. [Google Scholar] [CrossRef]
- Sathiyavimal, S.; Vasantharaj, S.; Lewisoscar, F.; Pugazhendhi, A.; Subashkumar, R. Biosynthesis and characterization of hydroxyapatite and its composite (hydroxyapatite-gelatin-chitosan-fibrin-bone ash) for bone tissue engineering applications. Int. J. Biol. Macromol. 2019, 129, 844–52. [Google Scholar] [CrossRef]
- Ortolani, E.; Quadrini, F.; Bellisario, D.; Santo, L; Polimeni, A. ; Santarsiero, A. Mechanical qualification of collagen membranes used in dentistry. Ann. Ist. Super. Sanità. 2015, 51, 229–235. [Google Scholar] [CrossRef]
- Guo, M.; Chu, Z.; Yao, J.; Feng, W.; Wang, Y.; Wang, L.; Fan, Y. The effects of tensile stress on the degradation of biodegradable PLGA membranes: A quantitative study. Polym. Degrad. Stabyl. 2016, 124, 95–100. [Google Scholar] [CrossRef]
- Wu, H; Ji, D. ; Chang, W.; Yang, J.; Lee, S. Chitosan-based polyelectrolyte complex scaffolds with antibacterial properties for treating dental bone defects. Mater. Sci. Eng. C. 2012, 32, 207–214. [Google Scholar] [CrossRef]
- Kitayama, S.; Wong, L.O.; Ma, L.; Hao, J.; Kasugai, S.; Lang, N.P.; Mattheos, N. Regeneration of rabbit calvarial defects using biphasic calcium phosphate and a strontium hydroxyapatite-containing collagen membrane. Clin. Oral Implants Res. 2016, 27, e206–e214. [Google Scholar] [CrossRef] [PubMed]
- Vijayakumar, P.; Vathaluru, S.; Abisegapriyan, S.; Sherine, J.; Ramakrishna, S.; Hasbi, M.; Rahim, A.; Mohd, M.; Jose, R.; Reddy, J. Ramification of zinc oxide doped hydroxyapatite biocomposites for the mineralization of osteoblasts. Mater. Sci. Eng. C. 2019, 96, 337–46. [Google Scholar] [CrossRef]
- Nygren, H.; Bigdeli, N.; Ilver, I.; Malmberg, P. Mg-corrosion, hydroxyapatite, and bone healing. Biointerphases. 2017, 12, 02C407–1. [Google Scholar] [CrossRef]
- Liao, S.; Watari, F.; Zhu,Y. ; Uo, M.; Akasaka, T.; Wang, W.; Cui, F. The degradation of the three-layered nano-carbonated hydroxyapatite/collagen/PLGA composite membrane in vitro. Dental Materials. 2007, 23, 1120–1128. [Google Scholar] [CrossRef] [PubMed]

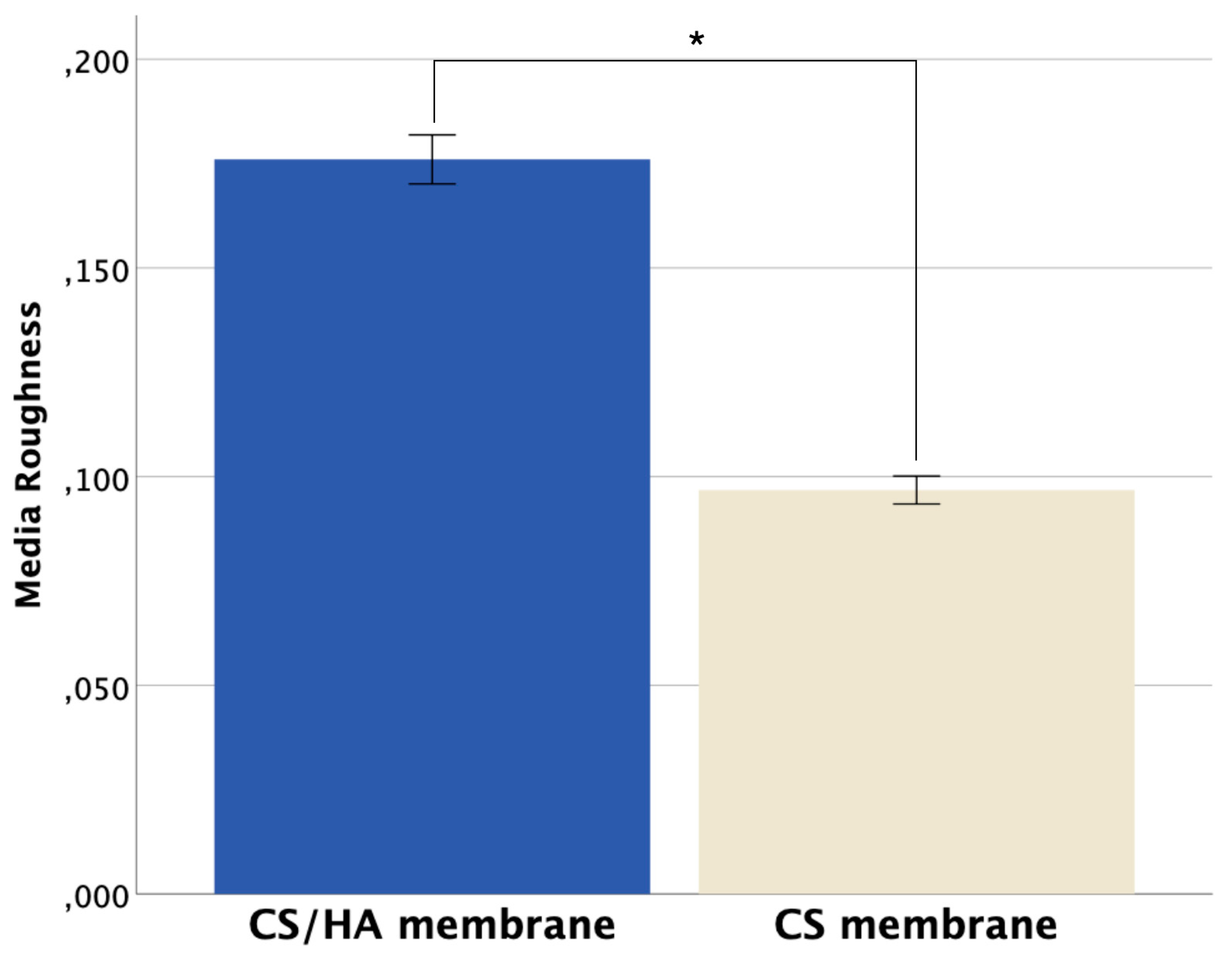
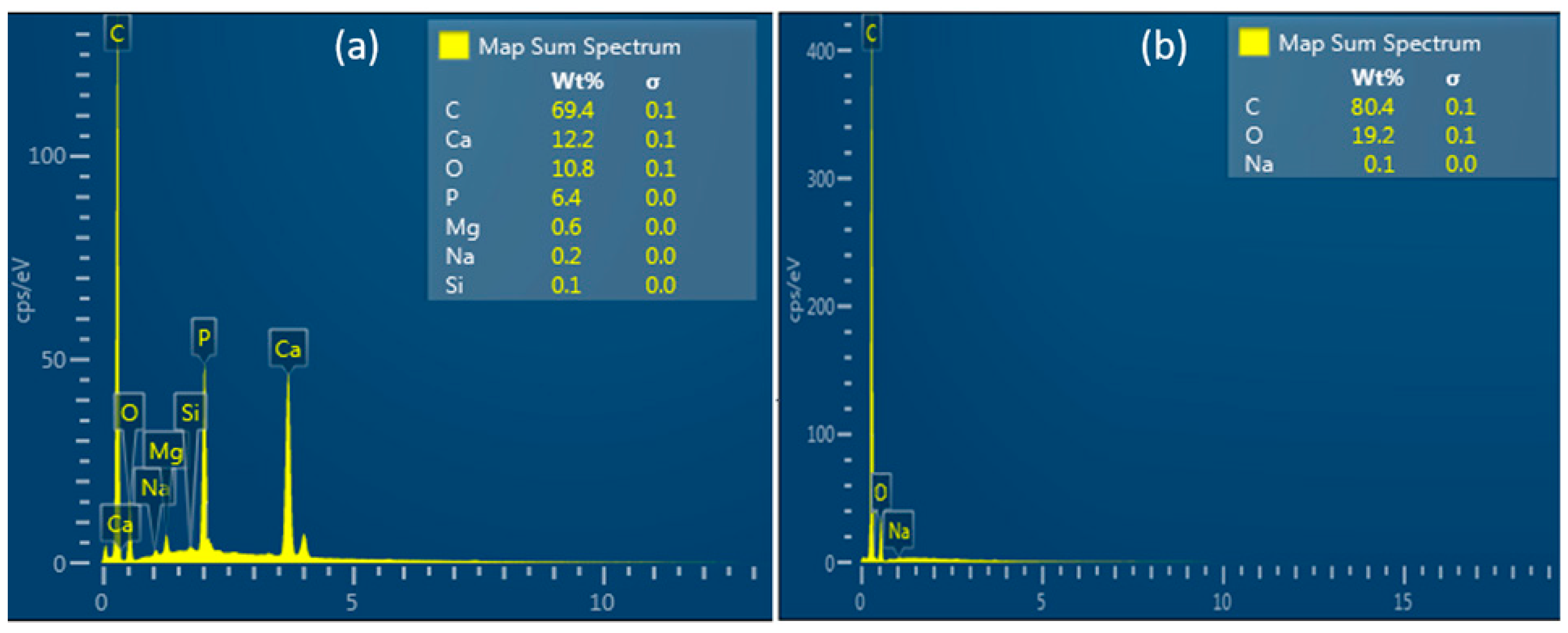
| CS/HA Membrane | CS Membrane | p value | |
|---|---|---|---|
| UTS (MPa) | 5.4 ± 0.62 | 3.3 ± 0.46 | <0.001* |
| Ɛb (%) | 92.4 ± 5.2 | 54 ± 6.1 | <0.001* |
| CS/HA Membrane | CS Membrane | p value | |
|---|---|---|---|
| Weight loss (%) | 54.2 ± 7.1% | 43.7 ± 5.92% | <0.001* |
| Swelling Ratio (%) | 62.5 ± 3.0% | 54.4 ± 3.3% | 0.004* |
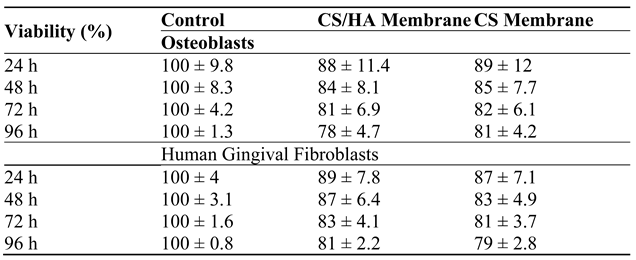
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




