1. Introduction
Historically, plastic surgeons rely on a step-like approach known as the “Reconstructive Ladder” when planning reconstruction procedures. Starting with the simplest technique available, this would then progressively escalate to more complex interventions when appropriate. [
1,
2] In 1994, Gotlieb and Krieger proposed the "Reconstructive Elevator," which allowed surgeons to select the most suitable primary reconstructive technique, regardless of complexity. [
3] Then, in a more recent development, came the "Reconstructive Grid," which considered factors such as wound complexity, surgeon expertise, available resources, and patient preferences when deciding on a method for wound closure. [
4,
5,
6]
Over the last twenty years, there has been a remarkable advancement in the development of skin substitutes, leading to their gradual integration into clinical practice. This evolution has significantly transformed the approach to managing soft tissue deficits in contemporary medical settings. [
7] Several studies have indicated that the use of skin substitutes is associated with a reduction in the time required for wound closure, [
8] and their inherent biological properties have been shown to effectively address a range of complexities in the wound care process, including inflammation, re-epithelialization, angiogenesis, wound contraction, and extracellular matrix remodeling. [
9] In our experience, skin substitutes can provide a temporal coverage when there is limited native tissue available, and can also offer a simpler alternative to complex reconstructive procedures in situations when they are contraindicated or unsafe, particularly in frail or unstable patients. [
10,
11] This review emphasizes the benefits of integrating skin substitutes as adjunctive components in complex reconstructive scenarios, without proposing specific standards for locoregional reconstruction.
2. Skin Substitutes for Reconstruction and Wound Care: Properties, and Types
2.1. Properties
Skin Substitutes have become essential for tissue reinforcement in reconstruction and wound care because of their regenerative properties, active modulation of the wound healing process, and remodeling of the extracellular matrix (ECM). [
12] When applied to the wound, these tridimensional scaffolds are quickly infiltrated by cellular components and growth factors that stimulate angiogenesis and help promote wound healing. [
13,
14] Studies have shown that wounds treated with dermal matrices exhibit increased expression of various growth factors such as EGF, FGF, PDGF, and TGF-β. [
7,
9,
15] This increase in expression is due to natural cytokines stored within the scaffold and their cumulative effect on the local production of molecules by native cells on the recipient tissue. [
11] Additionally, these matrices offer immediate protection of the native ECM against dehydration, microorganism colonization, exposure to toxins, and external environmental factors that can disrupt the healing process. [
15]
2.2. Types
There are two main forms, dermal and layered. Dermal contains only dermis components, while layered incorporates an additional semipermeable sheet, typically constructed from silicone, to mimic the native epidermis. These matrices can be classified as cellular or acellular depending on their cellular composition. Furthermore, they can be subcategorized based on their donor origin into autografts, allografts, and xenografts. Allografts come from cadaveric and neonatal donors, whereas xenografts are typically derived from bovine and porcine sources, but can also come from other species. Acellular grafts, which contain no living cells and are immunologically inert, are mandatory for xenografts to avoid an immunogenic host response. [
16] Acellular Dermal Matrices (ADMs) are obtained from allogeneic and xenogeneic donors, and retain numerous ECM components including collagen, elastin, laminin, hyaluronic acid, and glycosaminoglycans. [
16]
Allogenic ADMs are classified as banked human tissue by the Food and Drug Administration (FDA) because they are produced from cadaveric donor skin. Xenogeneic ADMs are classified as medical devices. Certain alternative products involve additional processing, such as collagen cross-linking, which aids in minimizing degradation by native collagenases. [
17] A recently developed scaffold derived from porcine urinary bladder extracellular matrix has been found to be advantageous in various respects. Studies have shown that they enhance progenitor cell migration, proliferation, and differentiation while promoting angiogenesis, reinnervation, and minimal foreign body reaction. [
18] Particulate and paste presentations are currently available and can be used to treat tunneled or irregular wounds, although they have comparatively shorter absorption times.
Cellular dermal matrices are composed primarily composed of human neonatal fibroblasts and keratinocytes cultured on a bovine collagen matrix or a biodegradable polyglactin mesh. They are mainly used in non-infected venous leg ulcers and neuropathic diabetic foot ulcers without tendon, muscle, capsule, or bone exposure.
3. Current Uses of Skin Substitutes
3.1. Scalp, Face, and Neck Reconstruction after Oncologic Resection, Trauma and Burns
Scalp reconstruction is a challenging task, specially following wide excisions for cancer treatment, which can result in defects that disrupt the blood supply of potential local flaps. Several articles in the literature discuss comprehensive algorithms for scalp reconstruction; however, such detailed approaches are beyond the scope of this review. [
19,
20,
21] Local and regional flaps typically provide stable coverage of scalp defects, but their availability may be limited due to prior surgery, radiotherapy or scarring (
Figure 1) [
22,
23] Free tissue flaps have high survival rates and may be necessary for large scalp defects, while can result in donor-site morbidity, increased hospitalization length and higher overall cost. [
24,
25] However, when temporary coverage is indicated or a previous reconstruction attempt has failed, skin substitutes become viable alternatives. [
26,
27,
28,
29] In full-thickness defects with exposed skull, skin substitutes can be considered an initial step in the reconstructive strategy. A single-stage reconstruction can be achieved by placing a dermal matrix and a thin split-thickness skin graft (STSG) over burred external table, while some authors recommend to hold skin grafting for about six weeks, until granulation tissue is optimal for graft take. [
30] When possible, vascularized pericranial flaps can be mobilized to cover the exposed skull before skin substitute placement. [
31,
32,
33] Cost-analysis studies have demonstrated that treating scalp defects larger than 100 cm
2 with the use of dermal matrices is more cost-effective than free and local flaps. [
34] Furthermore, the use of temporal synthetic biodegradable matrices has proven beneficial in facilitating the closure of large and infected scalp defects. [
35]
The many aesthetics subunits of the face require complex reconstructions after cancer resection defects, trauma or as a result of burns. Defects over the lower eyelids, inner cantus, cheek, and neck can potentially benefit from incorporating skin substitutes as adjunct to reconstructive procedures to either temporally or permanently assist in covering the defect. For example, application of dehydrated human amniotic membrane over defects in the lower eyelids, or a bilayered dermal matrix over defects on the inner cantus has shown promising results providing stable closure of the wounds and improving overall scar healing pain scores. [
36] Similarly, large neck defects requiring complex regional flap, skin substitutes such as bilayered dermal matrices and others, can facilitate and reinforce the closure and temporalize the wound bed in preparation for subsequent skin grafting. (
Figure 2).
Facial burns, especially in infants and children, benefit from the application of certain skin substitutes such as human amniotic membrane. Studies has shown that the regenerative characteristics of these biomaterials are safe and enhance the wound healing process in this vulnerable population where donor skin is limited or not available. [
37]
3.2. Upper Extremities Reconstruction after Burns, Trauma and Chronic Wounds.
Traumatic and burn injuries to the upper extremity, particularly the hand, pose significant challenges, as they are often associated with high rates of disability and morbidity, often necessitating multiple and complex reconstructive procedures. [
38,
39,
40] In the context of hand burns, digit scar contractures are a common occurrence, and the standard of care often involves the utilization of local flaps and full-thickness skin grafts for scar release and return of range of motion. The incorporation of skin substitutes, specifically bilayer dermal matrices, can serve as a valuable adjunct to these procedures, providing temporary and definitive coverage for secondary defects and prior to the application of skin grafts. [
41] For extensive upper extremity burn wounds, an increasingly popular strategy involves the combination of skin grafts with dermal matrices. This approach has shown promising results in enhancing the quality and elasticity of the skin, and ultimately improving the resultant range of motion of the affected joints. The key to this improvement lies in the introduction of a regenerative scaffold of elastin and collagen into the wound bed that serves as a template for new tissue growth. [
42,
43,
44,
45] Furthermore, when this approach is used in conjunction with negative pressure wound therapy (NPWT), the outcomes are even more promising, resulting in a superior scar appearance compared to the use of skin grafts alone. [
46]
Traumatic fingertip injuries are common. The decision between nonoperative and operative management depends on specific criteria. Secondary intention healing is indicated in patients without exposed bone or tendon and less than 2 cm of skin loss, or children with exposed bone. Operative interventions, including primary closure, full-thickness skin grafting, and flap reconstruction, are tailored based on the extent of tissue loss and exposure of bone or tendon. The goal of fingertip reconstruction lies in restoration of sensate and durable fingertips with adequate bone support for nail growth. Improper treatment may lead to stiffness, long-term functional loss and hook nail deformity. [
47] The adjunct use of skin substitutes for second intention healing of fingertip injuries is a feasible option. This approach has led to promising results and is associated with better scar quality, shorter surgical times and hospital stays, and lower surgical costs. [
48] Additionally, it has been linked to improved range of motion and sensory recovery, with no significant differences in patient satisfaction compared to traditional flaps. [
49] This approach can also be considered in cases with exposed tendons and bones according to some authors. [
50]
Chronic and infected upper extremity wounds are difficult. Treatment involves reconstruction after full course of antibiotics and serial debridement. Under these circumstances, the resulting defects are often extensive and complex. The use of temporary skin substitutes, such as synthetic biodegradable polyurethane matrices, has proven to be beneficial. It increases the success rates of reconstruction and reduces morbidity in patients with chronic wounds, including those complicated by osteomyelitis. [
51,
52,
53] Another example noteworthy to highlight is the management of severe axillary hidradenitis suppurativa, which necessitates extensive full thickness skin resection of the axillary region, resulting in undesirable scarring and contracture despite local flaps and application of full thickness skin grafts. [
54] Studies have shown that applying a bilayer dermal matrix followed by skin grafts has positive outcomes including low recurrence rate, improved range of motion at the shoulder, better aesthetic results, and lower pain scores compared to skin grafts alone. [
55,
56,
57,
58]
3.3. Applications on Lower Extremities Reconstruction
Most chronic non-healing wounds in the lower extremities are the consequence of multiple conditions, including venous insufficiency, diabetic foot ulcers, osteomyelitis, peripheral artery disease, deep burns, necrotizing infections, tumor resection defects, and severe trauma. Providing care for these wounds is particularly challenging, as patients are subject to significant disability and recurrence. [
59] Reconstruction involves the use of local or free flaps with favorable outcomes and high rates of limb salvage. Nevertheless, difficulties can arise due to limited availability of donor tissues, particularly in complex cases involving significant soft tissue loss. [
60,
61] Skin substitutes can be used as temporary biological coverage, for wound bed preparation for future skin grafting, or in conjunction to flaps in complex wounds with satisfactory results. [
62] (
Figure 3) Moreover, skin substitutes alone can achieve reconstruction success of 70-80%, which is remarkable compared to the 91% and 93% for local and free flaps, respectively. [
63,
64] Also, most skin substitutes are available off-the-shelf, making their use convenient especially in urgent situations. [
62]
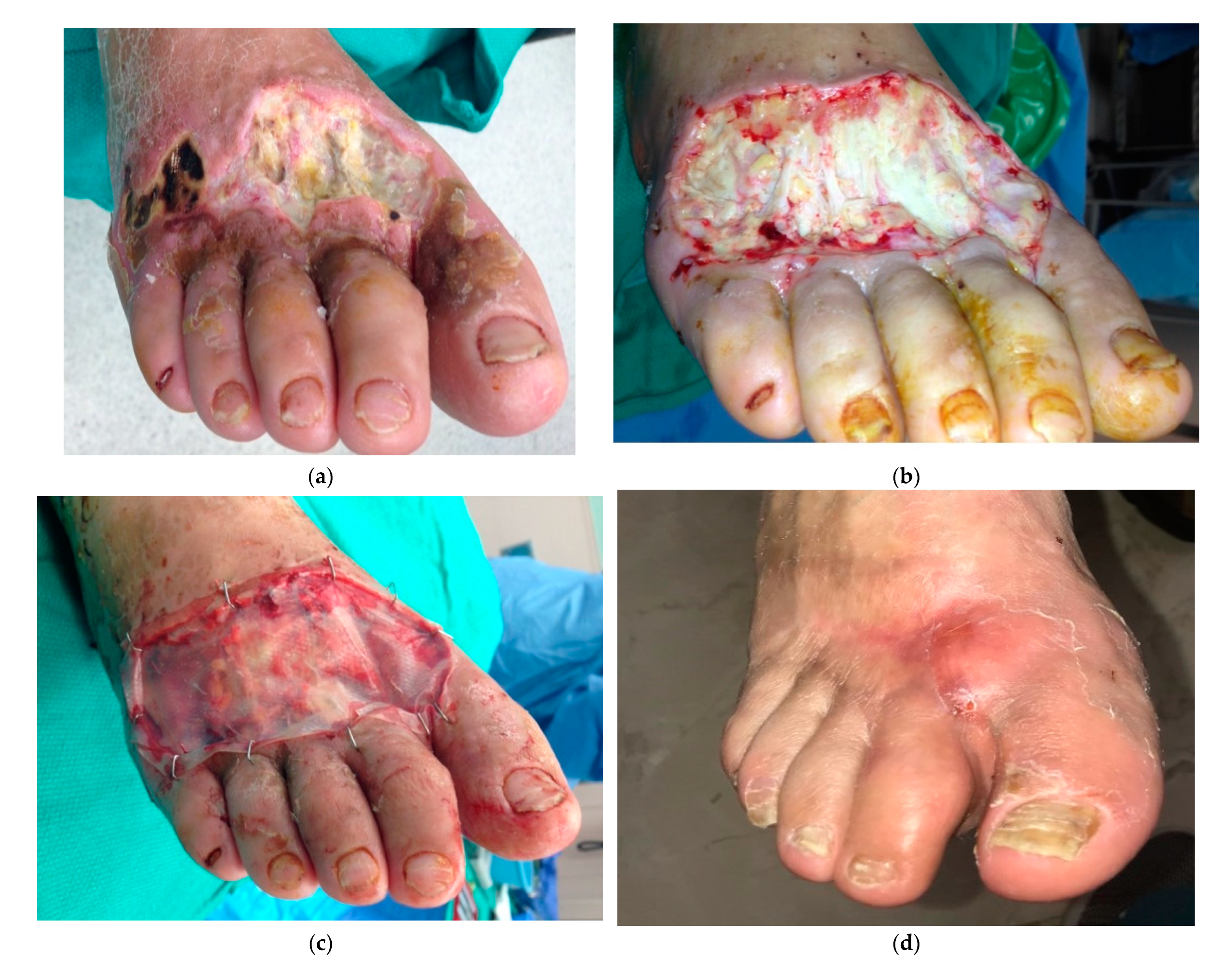
In foot and ankle traumatic wounds, adjunct use of dermal matrices with flaps has shown a 25% reduction in healing time, and are a feasible alternative for coverage after flap failure. [
65] Similarly, in chronic venous ulcers bilayered dermal matrices have shown advantageous results decreasing healing time compared to controls treated with standard wound care. [
66] (
Figure 4) In a similar way, diabetic foot ulcers that failed to heal with traditional dressings can see positive outcomes when treated with 2 ± 1.4 applications of fetal bovine acellular dermal matrix. [
67] Furthermore, studies indicate that skin substitutes can boost tissue oxygen pressure in these poorly vascularized wound beds [
68] (
Figure 5).
3.4. Applications on Trunk and Spinal Reconstruction
The use of dermal matrices in abdominal reconstruction has become increasingly common, as local and free flaps are utilized for repair of large and complex abdominal wall defects following oncologic resections and catastrophic abdominal complications. [
69,
70] Literature contains detailed strategies that incorporate the use of autologous tissue, with or without synthetic and biological materials. [
71,
72] Nonetheless, it should be noted that characterizing these strategies is not the focus of the current review; rather, our main objective is to highlight the promising results observed when using skin substitutes in combination with autologous tissue for full-thickness abdominal wall defects. [
73,
74,
75] Indications for employing dermal matrices or other biomaterials as surgical meshes or regeneration scaffolds include previous failed reconstructions and contaminated surgical field. [
76,
77,
78] (
Figure 6) Biological dermal matrices promote revascularization and integrate into native tissues more quickly than synthetic materials. [
79,
80] This allows for the formation of a robust tissue layer that promotes lower rates of extrusion, visceral erosion, intra-abdominal adhesion formation, and infections when compared to synthetic meshes. [
81,
82] Also, when utilized for abdominal wall hernia repair, current research recommends the use of retromuscular or underlay mesh placement due to their lower risk of hernia recurrence. [
83,
84,
85] Hybrid meshes, which have been recently developed, combine biological materials with a permanent synthetic component to create a durable mesh that facilitates tissue ingrowth and lower foreign-body reaction. [
86] The addition of biosynthetic or biological material could reduce the need for permanent materials in abdominal wall repairs, providing better tissue integration and infection protection. [
87] Furthermore, the use of biosynthetic meshes in certain scenarios has resulted in improved outcomes and reduced costs compared to biologicals alone. [
88,
89,
90]
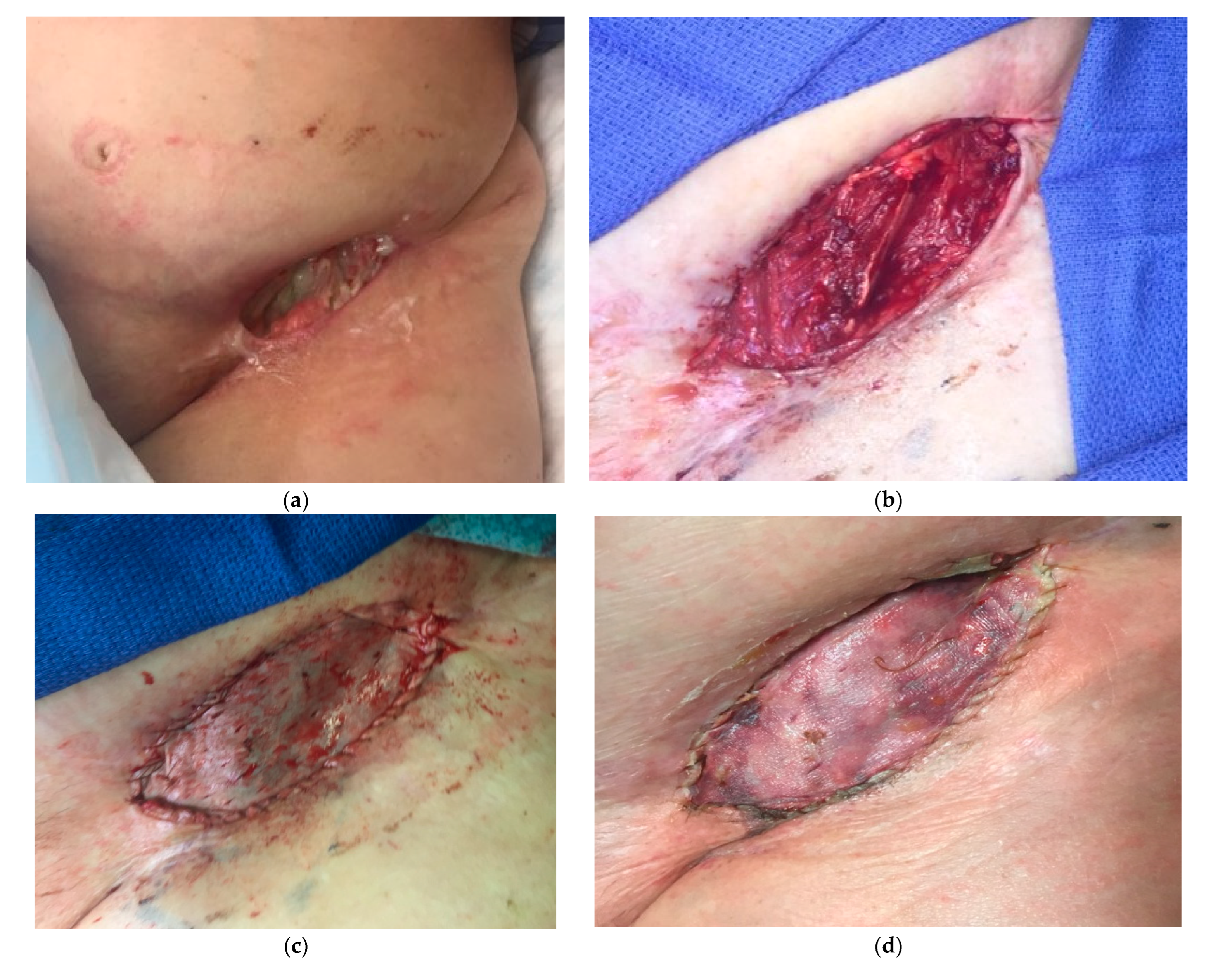
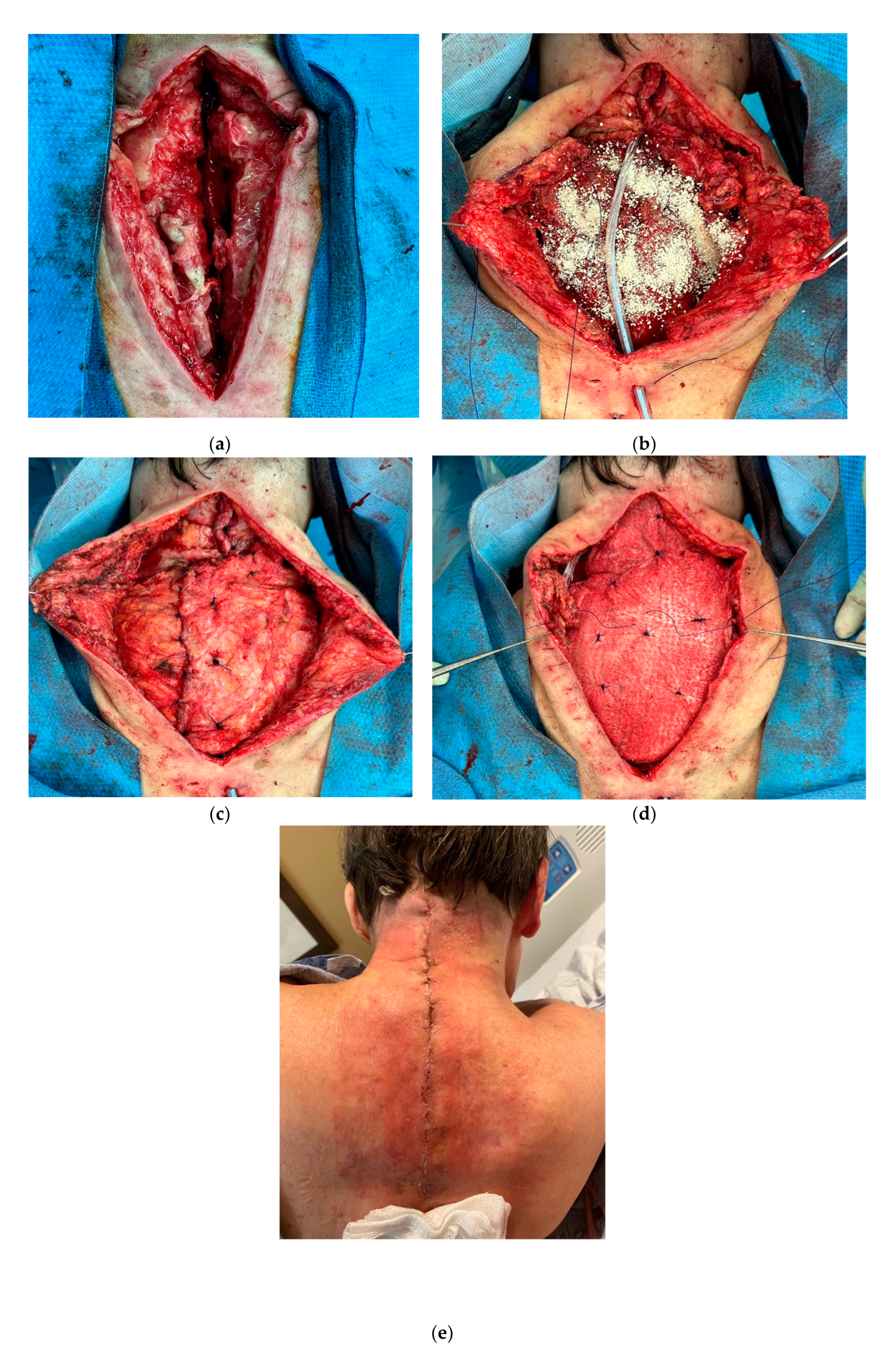
In the same way, immediate reconstruction following complex spinal surgery and oncologic spinal wounds can benefit from the adjunct use of a particulate extracellular matrices along with local muscular flaps. These presentations are morcellated forms of extracellular matrices from different sources, and can assist in obliterating the resultant dead space between the dura and paravertebral muscles flaps. [
91] Additionally, other local flaps used for spine reconstruction such as the trapezium and latissimus, can be reinforced with application of extracellular matrices with favorable results. (
Figure 7)
4. The Future of Skin Substitutes
The reconstructive ladder and subsequent models have provided valuable guidance in making optimal choices to expedite patient healing and achieve a balance between function and aesthetic. [
92] Current scientific and technological advances have facilitated the development of numerous skin substitutes that enhance and modulate the wound healing process through biomodulation effects. [
93,
94,
95] This include immune cell recruitment, increased essential cytokines and growth factors, and modified molecular interactions within the wound. [
96,
97,
98] More recently, tridimensional bioprinting has enabled the production of synthetic skin embedded with cells and bioactive molecules, resulting in increased cytokine production at the wound site. This accelerates healing by stimulating cell proliferation, promoting macrophage differentiation, and enhancing neovascularization. [
99,
100] Additionally, gene editing technology applied to novel skin substitutes has shown potential for accelerated skin regeneration by targeting growth factors and pluripotent cells. [
101,
102] Despite these advancements, the challenges surrounding skin substitutes in clinical practice are multifaceted. One hurdle is the incomplete integration with the host tissue, as achieving proper vascularization and cellular interactions is crucial for successful wound healing. Another is the uncertain long-term stability of these substitutes, as well as the absence of native skin elements such as epidermal appendages, intrinsic vasculature, innervation, and the capacity to produce melanin. [
9] Additionally, high costs of skin substitutes presents challenges for generalized availability in low-income and uninsured patients. In contrast, cost analysis studies comparing skin substitutes to traditional wound care strategies have demonstrated a beneficial economic impact, owing to decreased number of emergency visits and readmissions, shorter hospitalizations, and improved limb salvage rates. [
103] Moreover, regulatory considerations associated with the development and commercialization of these products add to their complexity. Overcoming these challenges will require continuous collaboration between scientists, clinicians, industry, and regulatory authorities.
In conclusion, recent technological advancements in skin substitutes have been pivotal in filling the gaps within reconstructive algorithms. These biomaterials offer solutions for reconstructive surgery and wound care while reducing overall treatment costs. Further research and development in this field will likely lead to additional advances in the effectiveness and accessibility of skin substitutes for patients.
Author Contributions
(I) Conception and design: PJF; GMH; KCM; RC; (II) Provision of study materials or patients: PJF; GMH; RC; (III) Collection and assembly of data: KCM; KMS; WS; SSA; SP; MD; EG. (IV) Data analysis and interpretation: PJF; KCM; RC; (V) Manuscript writing: All authors; (VI) Final approval of manuscript: All authors
Funding
This research received no external funding.
Informed Consent Statement
All patients receiving treatment involving skin substitutes and biological materials under the care of the senior author (RC) have undergone a thorough informed consent process. Prior to any intervention, these patients have been provided with comprehensive information about the nature, purpose, potential risks, and expected outcomes of the proposed procedures.
Conflicts of Interest
All authors declare no conflict of interest, except for Dr. Ricardo Castrellon, who presently serves as a key opinion leader for Integra LifeSciences®. However, every effort has been made to guarantee an objective and unbiased presentation of the research findings. Any opinions expressed by Dr. Castrellon are their own and should be assessed in light of the disclosed conflict of interest.
References
- Janis, J.E.; Kwon, R.K.; Attinger, C.E. The new reconstructive ladder: modifications to the traditional model [published correction appears in Plast Reconstr Surg. 2020 Nov;146(5):1212]. Plast. Reconstr. Surg. 2011, 127, 205S–212S. [Google Scholar] [CrossRef] [PubMed]
- Bennett, N.; Choudhary, S. Why Climb a Ladder When You Can Take the Elevator? Plast. Reconstr. Surg. 2000, 105, 2266–2266. [Google Scholar] [CrossRef] [PubMed]
- Gottlieb, L.J.M.; Krieger, L.M.M. From the Reconstructive Ladder to the Reconstructive Elevator. Plast. Reconstr. Surg. 1994, 93, 1503–4. [Google Scholar] [CrossRef] [PubMed]
- Giordano, V.; Napoli, S.; Quercioli, F.; Mori, A.; Dini, M. The Solar System Model for the Reconstructive Ladder. Plast. Reconstr. Surg. 2011, 128, 336–337. [Google Scholar] [CrossRef] [PubMed]
- Knobloch, K.; Vogt, P.M. The Reconstructive Clockwork of the Twenty-First Century: An Extension of the Concept of the Reconstructive Ladder and Reconstructive Elevator. Plast. Reconstr. Surg. 2010, 126, 220e–222e. [Google Scholar] [CrossRef] [PubMed]
- Mohapatra, D.P.; Thiruvoth, F.M. Reconstruction 2.0: Restructuring the Reconstructive Ladder. Plast. Reconstr. Surg. 2021, 147, 572e–573e. [Google Scholar] [CrossRef]
- Oualla-Bachiri, W.; Fernández-González, A.; Quiñones-Vico, M.I.; Arias-Santiago, S. From Grafts to Human Bioengineered Vascularized Skin Substitutes. Int. J. Mol. Sci. 2020, 21, 8197. [Google Scholar] [CrossRef] [PubMed]
- Gushiken, L.F.S.; Beserra, F.P.; Bastos, J.K.; Jackson, C.J.; Pellizzon, C.H. Cutaneous Wound Healing: An Update from Physiopathology to Current Therapies. Life 2021, 11, 665. [Google Scholar] [CrossRef] [PubMed]
- Dearman, B.L.; Boyce, S.T.; Greenwood, J.E. Advances in Skin Tissue Bioengineering and the Challenges of Clinical Translation. Front. Surg. 2021, 8. [Google Scholar] [CrossRef] [PubMed]
- Przekora, A. A Concise Review on Tissue Engineered Artificial Skin Grafts for Chronic Wound Treatment: Can We Reconstruct Functional Skin Tissue In Vitro? Cells 2020, 9, 1622. [Google Scholar] [CrossRef] [PubMed]
- Petrie, K.; Cox, C.T.; Becker, B.C.; MacKay, B.J. Clinical applications of acellular dermal matrices: A review. Scars, Burn. Heal. 2022, 8. [Google Scholar] [CrossRef] [PubMed]
- Hicks, K.E.; Huynh, M.N.; Jeschke, M.; Malic, C. Dermal regenerative matrix use in burn patients: A systematic review. J. Plast. Reconstr. Aesthetic Surg. 2019, 72, 1741–1751. [Google Scholar] [CrossRef] [PubMed]
- Panayi, A.C.; Orgill, D.P. Current Use of Biological Scaffolds in Plastic Surgery. Plast. Reconstr. Surg. 2019, 143, 209–220. [Google Scholar] [CrossRef] [PubMed]
- Tenenhaus, M.; Rennekampff, H.-O. Current Concepts in Tissue Engineering: Skin and Wound. Plast. Reconstr. Surg. 2016, 138, 42S–50S. [Google Scholar] [CrossRef]
- du Plessis, M.; Cottler, P.S.; Campbell, C.A. Acellular Dermal Matrix Favorably Modulates the Healing Response after Surgery. Plast. Reconstr. Surg. 2022, 150, 290E–299E. [Google Scholar] [CrossRef] [PubMed]
- Hughes, O.B.; Rakosi, A.; Macquhae, F.; Herskovitz, I.; Fox, J.D.; Kirsner, R.S. A Review of Cellular and Acellular Matrix Products: Indications, Techniques, and Outcomes. Plast. Reconstr. Surg. 2016, 138, 138S–147S. [Google Scholar] [CrossRef] [PubMed]
- Mazari, F.A.K.; Wattoo, G.M.; Kazzazi, N.H.; Kolar, K.M.; Olubowale, O.O.; Rogers, C.E.; Azmy, I.A. The Comparison of Strattice and SurgiMend in Acellular Dermal Matrix–Assisted, Implant-Based Immediate Breast Reconstruction. Plast. Reconstr. Surg. 2018, 141, 283–293. [Google Scholar] [CrossRef]
- Parcells, A.L.; Abernathie, B.; Datiashvili, R. The use of urinary bladder matrix in the treatment of complicated open wounds. . 2014, 26, 189–96. [Google Scholar]
- Leedy, J.E.; Janis, J.E.; Rohrich, R.J. Reconstruction of Acquired Scalp Defects: An Algorithmic Approach. Plast. Reconstr. Surg. 2005, 116, 54e–72e. [Google Scholar] [CrossRef]
- Desai, S.C.; Sand, J.P.; Sharon, J.D.; Branham, G.; Nussenbaum, B. Scalp Reconstruction: an algorithmic approach and systematic review. JAMA Facial Plast. Surg. 2015, 17, 56–66. [Google Scholar] [CrossRef] [PubMed]
- Harirah, M.; Sanniec, K.M.; Yates, T.B.; Harirah, O.B.; Thornton, J.F. Scalp Reconstruction after Mohs Cancer Excision: Lessons Learned from More Than 900 Consecutive Cases. Plast. Reconstr. Surg. 2021, 147, 1165–1175. [Google Scholar] [CrossRef]
- Zayakova, Y.; Stanev, A.; Mihailov, H.; Pashaliev, N. Application of Local Axial Flaps to Scalp Reconstruction. Arch. Plast. Surg. 2013, 40, 564–569. [Google Scholar] [CrossRef] [PubMed]
- Brawley, C.C.; Sidle, D. Scalp reconstructive flaps. Plast. Aesthetic Res. 2022, 9, 6. [Google Scholar] [CrossRef]
- Furnas, H.; Lineaweaver, W.C.; Alpert, B.S.; Buncke, H.J. Scalp Reconstruction by Microvascular Free Tissue Transfer. Ann. Plast. Surg. 1990, 24, 431–444. [Google Scholar] [CrossRef] [PubMed]
- Lembo, F.; Cecchino, L.R.; Parisi, D.; Portincasa, A. Utility of a New Artificial Dermis as a Successful Tool in Face and Scalp Reconstruction for Skin Cancer: Analysis of the Efficacy, Safety, and Aesthetic Outcomes. Dermatol. Res. Pr. 2020, 2020, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Brawley, C.C.; Sidle, D. Scalp reconstructive flaps. Plast. Aesthetic Res. 2022, 9, 6. [Google Scholar] [CrossRef]
- Othman, S.B.; Shakir, S.; Azoury, S.C.; Lukowiak, T.B.; Shin, T.M.M.; Sobanko, J.F.; Miller, C.J.; Etzkorn, J.R.; Fischer, J.P.M.; Kovach, S.J. Utility of Dermal Wound Matrices Compared with Local-Tissue Rearrangement and Free Flap Reconstruction for Oncologic Scalp Wounds: A Multidisciplinary Dual Matched-Pair Analysis. Plast. Reconstr. Surg. 2022, 149, 469–480. [Google Scholar] [CrossRef] [PubMed]
- Richardson, M.A.; Lange, J.P.; Jordan, J.R. Reconstruction of Full-Thickness Scalp Defects Using a Dermal Regeneration Template. JAMA Facial Plast. Surg. 2016, 18, 62–67. [Google Scholar] [CrossRef]
- Magnoni, C.; De Santis, G.; Fraccalvieri, M.; Bellini, P.; Portincasa, A.; Giacomelli, L.; Papa, G. Integra in Scalp Reconstruction After Tumor Excision: Recommendations From a Multidisciplinary Advisory Board. J. Craniofacial Surg. 2019, 30, 2416–2420. [Google Scholar] [CrossRef] [PubMed]
- Chun, Y.S.; Verma, K. Single-Stage Full-Thickness Scalp Reconstruction Using Acellular Dermal Matrix and Skin Graft. 2011, 11, e4. [Google Scholar] [PubMed]
- Chaiyasate, K.M.; Oliver, L.N.D.; Kreitzberg, S.A.D.; Lyons, M.; Goldman, J.; Lu, S.M.M.; Bastiaans, T.D.; Lumley, C.D.; Sachanandani, N.S. Use of Pericranial Flaps with Dermal Substitute for Scalp Reconstruction: A Case Series. Plast. Reconstr. Surg. - Glob. Open 2020, 8, e3011. [Google Scholar] [CrossRef] [PubMed]
- Richardson, M.A.; Lange, J.P.; Jordan, J.R. Reconstruction of Full-Thickness Scalp Defects Using a Dermal Regeneration Template. JAMA Facial Plast. Surg. 2016, 18, 62–67. [Google Scholar] [CrossRef] [PubMed]
- Dessy, L.A.; Mazzocchi, M.; Rizzo, M.I.; Onesti, M.G.; Scuderi, G. Scalp reconstruction using dermal induction template: state of the art and personal experience. 2013, 27, 153–8. [Google Scholar]
- Schiavon, M.; Francescon, M.; Drigo, D.; Salloum, G.; Baraziol, R.; Tesei, J.; Fraccalanza, E.; Barbone, F. The Use of Integra Dermal Regeneration Template Versus Flaps for Reconstruction of Full-Thickness Scalp Defects Involving the Calvaria: A Cost–Benefit Analysis. Aesthetic Plast. Surg. 2016, 40, 901–907. [Google Scholar] [CrossRef] [PubMed]
- Patel, N.K.; Tipps, J.A.B.; Graham, E.M.B.; Taylor, J.A.; Mendenhall, S.D. Reconstruction of a Near-total Scalp Avulsion with NovoSorb Biodegradable Temporizing Matrix: Pediatric Case Report. Plast. Reconstr. Surg. - Glob. Open 2022, 10, e4717. [Google Scholar] [CrossRef] [PubMed]
- Wisco, O.J. Case series: The use of a dehydrated human amnion/chorion membrane allograft to enhance healing in the repair of lower eyelid defects after Mohs micrographic surgery. JAAD Case Rep. 2016, 2, 294–297. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.; Xiong, A.B.; He, X.C.; Bin Ding, X.; Tian, X.L.; Li, Y.; Yan, H. Efficacy and feasibility of amniotic membrane for the treatment of burn wounds: A meta-analysis. J. Trauma: Inj. Infect. Crit. Care 2020, 90, 744–755. [Google Scholar] [CrossRef]
- Engrav LH, Dutcher KA, Nakamura DY. Rating burn impairment. Clin Plast Surg. 1992 Jul;19(3):569-98. [PubMed]
- Askari, M.; Cohen, M.J.; Grossman, P.H.; Kulber, D.A. The Use of Acellular Dermal Matrix in Release of Burn Contracture Scars in the Hand. Plast. Reconstr. Surg. 2011, 127, 1593–1599. [Google Scholar] [CrossRef] [PubMed]
- Bhavsar, D.; Tenenhaus, M. The Use of Acellular Dermal Matrix for Coverage of Exposed Joint and Extensor Mechanism in Thermally Injured Patients With Few Options. 2008, 8, e33. 8.
- Ellis, C.V.; Kulber, D.A. Acellular Dermal Matrices in Hand Reconstruction. Plast. Reconstr. Surg. 2012, 130, 256S–269S. [Google Scholar] [CrossRef] [PubMed]
- Hicks, K.E.; Huynh, M.N.; Jeschke, M.; Malic, C. Dermal regenerative matrix use in burn patients: A systematic review. J. Plast. Reconstr. Aesthetic Surg. 2019, 72, 1741–1751. [Google Scholar] [CrossRef]
- Boháč, M.; Danišovič; Koller, J.; Dragúňová, J.; Varga, I. What happens to an acellular dermal matrix after implantation in the human body? A histological and electron microscopic study. Eur. J. Histochem. 2018, 62, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Keane, T.J.; Horejs, C.-M.; Stevens, M.M. Scarring vs. functional healing: Matrix-based strategies to regulate tissue repair. Adv. Drug Deliv. Rev. 2018, 129, 407–419. [Google Scholar] [CrossRef] [PubMed]
- Kreymerman, P.A.; Andres, L.A.; Lucas, H.D.; Silverman, A.L.; Smith, A.A. Reconstruction of the Burned Hand. Plast. Reconstr. Surg. 2011, 127, 752–759. [Google Scholar] [CrossRef] [PubMed]
- Yi, J.W.; Kim, J.K. Prospective Randomized Comparison of Scar Appearances between Cograft of Acellular Dermal Matrix with Autologous Split-Thickness Skin and Autologous Split-Thickness Skin Graft Alone for Full-Thickness Skin Defects of the Extremities. Plast. Reconstr. Surg. 2015, 135, 609e–616e. [Google Scholar] [CrossRef] [PubMed]
- Kawaiah, A.; Thakur, M.; Garg, S.; Kawasmi, S.H.; Hassan, A. Fingertip Injuries and Amputations: A Review of the Literature. Cureus 2020, 12, e8291. [Google Scholar] [CrossRef] [PubMed]
- Namgoong, S.; Jung, J.E.; Han, S.-K.; Jeong, S.-H.; Dhong, E.-S. Potential of Tissue-Engineered and Artificial Dermis Grafts for Fingertip Reconstruction. Plast. Reconstr. Surg. 2020, 146, 1082–1095. [Google Scholar] [CrossRef] [PubMed]
- You, H.-J.; Han, S.-K. Cell Therapy for Wound Healing. J. Korean Med Sci. 2014, 29, 311–319. [Google Scholar] [CrossRef] [PubMed]
- Rehim SA, Singhal M, Chung KC. Dermal skin substitutes for upper limb reconstruction: current status, indications, and contraindications. Hand Clin 2014; 30: 239–52, vii.
- Cheng, C.; Kwiecien, G.J.; Rowe, D.J.; Gatherwright, J.R.; Chepla, K.J. Reconstruction of Chronic Wounds Secondary to Injectable Drug Use with a Biodegradable Temporizing Matrix. Plast. Reconstr. Surg. - Glob. Open 2021, 9, e3678. [Google Scholar] [CrossRef] [PubMed]
- Solanki, N.S.; York, B.; Gao, Y.; Baker, P.; She, R.B.W. A consecutive case series of defects reconstructed using NovoSorb® Biodegradable Temporising Matrix: Initial experience and early results. J. Plast. Reconstr. Aesthetic Surg. 2020, 73, 1845–1853. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Lim, P.; Stanley, E.; Lee, G.; Lin, S.; Neoh, D.; Liew, J.; Ng, S.K. Experience with NovoSorb® Biodegradable Temporising Matrix in reconstruction of complex wounds. ANZ J. Surg. 2021, 91, 1744–1750. [Google Scholar] [CrossRef] [PubMed]
- Taylor, E.M.; Hamaguchi, R.B.; Kramer, K.M.; Kimball, A.B.M.; Orgill, D.P.M. Plastic Surgical Management of Hidradenitis Suppurativa. Plast. Reconstr. Surg. 2021, 147, 479–491. [Google Scholar] [CrossRef] [PubMed]
- Gonzaga, T.A.; Endorf, F.W.; Mohr, W.J.; Ahrenholz, D.H. Novel Surgical Approach for Axillary Hidradenitis Suppurativa Using a Bilayer Dermal Regeneration Template: a retrospective case study. J. Burn. Care Res. 2013, 34, 51–57. [Google Scholar] [CrossRef] [PubMed]
- Kraft, C.; Pearson, G. Axillary Hidradenitis Reconstruction Using a Dermal Regeneration Template. Wounds : a Compend. Clin. Res. Pr. 2022, 34, 43–46. [Google Scholar] [CrossRef] [PubMed]
- Amendola, F.; Cottone, G.; Alessandri-Bonetti, M.; Borelli, F.; Catapano, S.; Carbonaro, R.; Riccardi, F.; Vaienti, L. Reconstruction of the Axillary Region after Excision of Hidradenitis Suppurativa: A Systematic Review. Indian J. Plast. Surg. 2022, 56, 006–012. [Google Scholar] [CrossRef]
- Frame, J.D.; Still, J.; Lakhel-LeCoadou, A.; Carstens, M.H.; Lorenz, C.; Orlet, H.; Spence, R.; Berger, A.C.; Dantzer, E.; Burd, A. Use of Dermal Regeneration Template in Contracture Release Procedures: A Multicenter Evaluation. Plast. Reconstr. Surg. 2004, 113, 1330–1338. [Google Scholar] [CrossRef] [PubMed]
- Deldar, R.; Merle, C.; Attinger, C.E.; Evans, K.K. Soft tissue coverage of lower extremity defects: pearls and pitfalls in the chronic wound population. Plast. Aesthetic Res. 2022, 9, 13. [Google Scholar] [CrossRef]
- Lachica, R.D. Evidence-Based Medicine: Management of Acute Lower Extremity Trauma. Plast. Reconstr. Surg. 2017, 139, 287e–301e. [Google Scholar] [CrossRef] [PubMed]
- Tork, S.; Jefferson, R.C.; Janis, J.E. Acellular Dermal Matrices: Applications in Plastic Surgery. Semin. Plast. Surg. 2019, 33, 173–184. [Google Scholar] [CrossRef]
- Kim, Y.H.; Hwang, K.T.; Kim, K.H.; Sung, I.H.; Kim, S.W. Application of acellular human dermis and skin grafts for lower extremity reconstruction. J. Wound Care 2019, 28, S12–S17. [Google Scholar] [CrossRef] [PubMed]
- Kozak, G.M.; Hsu, J.Y.; Broach, R.B.; Shakir, S.; Calvert, C.; Stranix, J.T.; Messa, C.; Levin, L.S.; Serletti, J.M.; Kovach, S.J.; et al. Comparative Effectiveness Analysis of Complex Lower Extremity Reconstruction: Outcomes and Costs for Biologically Based, Local Tissue Rearrangement, and Free Flap Reconstruction. Plast. Reconstr. Surg. 2020, 145, 608E–616E. [Google Scholar] [CrossRef]
- Graham, G.P.; Helmer, S.D.; Haan, J.M.; Khandelwal, A. The Use of Integra® Dermal Regeneration Template in the Reconstruction of Traumatic Degloving Injuries. J. Burn. Care Res. 2013, 34, 261–266. [Google Scholar] [CrossRef] [PubMed]
- Pontell, M.E.; Saad, N.; Winters, B.S.; Daniel, J.N.; Saad, A. Reverse Sural Adipofascial Flaps with Acellular Dermal Matrix and Negative-Pressure Wound Therapy. Adv. Ski. Wound Care 2018, 31, 612–617. [Google Scholar] [CrossRef] [PubMed]
- Cazzell, S. A Randomized Controlled Trial Comparing a Human Acellular Dermal Matrix Versus Conventional Care for the Treatment of Venous Leg Ulcers. Wounds 2019, 31, 68–74. [Google Scholar] [PubMed]
- Kavros SJ, Dutra T, Gonzalez-Cruz R, et al. The use of PriMatrix, a fetal bovine acellular dermal matrix, in healing chronic diabetic foot ulcers: a prospective multicenter study. Adv Skin Wound Care 2014; 27: 356–362.
- Papa, G.; Spazzapan, L.; Pangos, M.; Delpin, A.; Arnez, Z.M. Compared to coverage by STSG grafts only reconstruction by the dermal substitute Integra® plus STSG increases TcPO2 values in diabetic feet at 3 and 6 months after reconstruction*. G Chir. 2014, 35, 141–5. [Google Scholar] [CrossRef]
- Baumann, D.P.; Roubaud, M.S. Flap Reconstruction of the Abdominal Wall. Semin. Plast. Surg. 2018, 32, 133–140. [Google Scholar] [CrossRef] [PubMed]
- Maxwell, D.W.; Hart, A.M.; Keifer, O.P.; Halani, S.H.; Losken, A. A Comparison of Acellular Dermal Matrices in Abdominal Wall Reconstruction. Ann. Plast. Surg. 2019, 82, 435–440. [Google Scholar] [CrossRef] [PubMed]
- Rohrich RJ, Lowe JB, Hackney FL, Bowman JL, Hobar PC. An algorithm for abdominal wall reconstruction. Plast Reconstr Surg 2000; 105:202e16.
- Kolker, A.R.; Brown, D.J.; Redstone, J.S.; Scarpinato, V.M.; Wallack, M.K. Multilayer Reconstruction of Abdominal Wall Defects With Acellular Dermal Allograft (AlloDerm) and Component Separation. Ann. Plast. Surg. 2005, 55, 36–42. [Google Scholar] [CrossRef] [PubMed]
- Buinewicz, B.; Rosen, B.D. Acellular Cadaveric Dermis (AlloDerm): A New Alternative for Abdominal Hernia Repair. Ann. Plast. Surg. 2004, 52, 188–194. [Google Scholar] [CrossRef]
- de Moya, M.A.; Dunham, M.; Inaba, K.; Bahouth, H.; Alam, H.B.; Sultan, B.; Namias, N. Long-Term Outcome of Acellular Dermal Matrix When Used for Large Traumatic Open Abdomen. J. Trauma: Inj. Infect. Crit. Care 2008, 65, 349–353. [Google Scholar] [CrossRef]
- Garvey, P.B.; Giordano, S.A.; Baumann, D.P.; Liu, J.; Butler, C.E. Long-Term Outcomes after Abdominal Wall Reconstruction with Acellular Dermal Matrix. J. Am. Coll. Surg. 2017, 224, 341–350. [Google Scholar] [CrossRef] [PubMed]
- Garvey, P.B.; Martinez, R.A.; Baumann, D.P.; Liu, J.; Butler, C.E. Outcomes of Abdominal Wall Reconstruction with Acellular Dermal Matrix Are Not Affected by Wound Contamination. J. Am. Coll. Surg. 2014, 219, 853–864. [Google Scholar] [CrossRef] [PubMed]
- Taibi, A.; Derbal, S.; Durand; Fontanier, S.D.; Christou, N.; Fredon, F.; Bouvier, S.; Fabre, A.; Rivaille, T.; Valleix, D.; Mathonnet, M. Implantation of biologic mesh in ventral hernia repair-does it make sense? Surg. Endosc. 2021, 35, 702–709. [Google Scholar] [CrossRef] [PubMed]
- Sasse, K.C.; Warner, D.L.; Ackerman, E.; Brandt, J. Parastomal hernia repair with urinary bladder matrix grafts: A case series. Int. J. Case Rep. Images 2016, 7, 85–91. [Google Scholar] [CrossRef]
- Butler, C.E.; Langstein, H.N.; Kronowitz, S.J. Pelvic, Abdominal, and Chest Wall Reconstruction with AlloDerm in Patients at Increased Risk for Mesh-Related Complications. Plast. Reconstr. Surg. 2005, 116, 1263–1275. [Google Scholar] [CrossRef] [PubMed]
- Costa, A.; Adamo, S.; Gossetti, F.; D’Amore, L.; Ceci, F.; Negro, P.; Bruzzone, P.; Amore, D. Ceci Biological Scaffolds for Abdominal Wall Repair: Future in Clinical Application? Materials 2019, 12, 2375. [Google Scholar] [CrossRef] [PubMed]
- Hsu, P.W.; Salgado, C.J.; Kent, K.; Finnegan, M.; Pello, M.; Simons, R.; Atabek, U.; Kann, B. Evaluation of porcine dermal collagen (Permacol) used in abdominal wall reconstruction. J. Plast. Reconstr. Aesthetic Surg. 2009, 62, 1484–1489. [Google Scholar] [CrossRef] [PubMed]
- Beale, E.W.; Hoxworth, R.E.; Livingston, E.H.; Trussler, A.P. The role of biologic mesh in abdominal wall reconstruction: a systematic review of the current literature. Am. J. Surg. 2012, 204, 510–517. [Google Scholar] [CrossRef] [PubMed]
- Sosin, M.; Nahabedian, M.Y.; Bhanot, P. The Perfect Plane: A Systematic Review of Mesh Location and Outcomes, Update 2018. Plast. Reconstr. Surg. 2018, 142, 107S–116S. [Google Scholar] [CrossRef] [PubMed]
- Albino, F.P.; Patel, K.M.; Nahabedian, M.Y.; Sosin, M.; Attinger, C.E.; Bhanot, P. Does Mesh Location Matter in Abdominal Wall Reconstruction? A Systematic Review of the Literature and a Summary of Recommendations. Plast. Reconstr. Surg. 2013, 132, 1295–1304. [Google Scholar] [CrossRef] [PubMed]
- Fuenmayor, P.; Lujan, H.J.; Plasencia, G.; Karmaker, A.; Mata, W.; Vecin, N. Robotic-assisted ventral and incisional hernia repair with hernia defect closure and intraperitoneal onlay mesh (IPOM) experience. J. Robot. Surg. 2020, 14, 695–701. [Google Scholar] [CrossRef] [PubMed]
- Rosen MJ, Bauer JJ, Harmaty M, et al. Multicenter, prospective, longitudinal study of the recurrence, surgical site infection, and quality of life after contaminated ventral hernia repair using biosynthetic absorbable mesh: the COBRA study. Ann Surg. 2017; 265:205–211.
- Rosen, M.J.; Krpata, D.M.; Petro, C.C.; Carbonell, A.; Warren, J.; Poulose, B.K.; Costanzo, A.; Tu, C.; Blatnik, J.; Prabhu, A.S. Biologic vs Synthetic Mesh for Single-stage Repair of Contaminated Ventral Hernias: A Randomized Clinical Trial. JAMA Surg. 2022, 157, 293–301. [Google Scholar] [CrossRef]
- Reid, C.M.; Jacobsen, G.R. A Current Review of Hybrid Meshes in Abdominal Wall Reconstruction. Plast. Reconstr. Surg. 2018, 142, 92S–96S. [Google Scholar] [CrossRef] [PubMed]
- Irvine, S.M.; Cayzer, J.; Todd, E.M.; Lun, S.; Floden, E.W.; Negron, L.; Fisher, J.N.; Dempsey, S.G.; Alexander, A.; Hill, M.C.; et al. Quantification of in vitro and in vivo angiogenesis stimulated by ovine forestomach matrix biomaterial. Biomaterials 2011, 32, 6351–6361. [Google Scholar] [CrossRef] [PubMed]
- Bittner, J.G.; El-Hayek, K.; Strong, A.T.; LaPinska, M.P.; Yoo, J.S.; Pauli, E.M.; Kroh, M. First human use of hybrid synthetic/biologic mesh in ventral hernia repair: a multicenter trial. Surg. Endosc. 2017, 32, 1123–1130. [Google Scholar] [CrossRef] [PubMed]
- Wright, M.A.B.; Weinstein, A.L.M.; Bernstein, J.L.; Franck, P.; Lara, D.O.B.; Samadi, A.B.; Cohen, L.E.; Härtl, R.; Baaj, A.A.; Spector, J.A. Muscle Flap Closure following Complex Spine Surgery: A Decade of Experience. Plast. Reconstr. Surg. 2020, 146, 642e–650e. [Google Scholar] [CrossRef] [PubMed]
- The New Reconstructive Ladder: Modifications to the Traditional Model: Correction. Plast Reconstr Surg. 2020;146(5):1212.
- Hosseini, M.; Brown, J.; Shafiee, A. Strategies to Induce Blood Vessel Ingrowth into Skin Grafts and Tissue-Engineered Substitutes. Tissue Eng. Part C: Methods 2022, 28, 113–126. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, A.; Simões, S.; Ascenso, A.; Reis, C.P. Therapeutic advances in wound healing. J. Dermatol. Treat. 2020, 33, 2–22. [Google Scholar] [CrossRef] [PubMed]
- Oualla-Bachiri, W.; Fernández-González, A.; Quiñones-Vico, M.I.; Arias-Santiago, S. From Grafts to Human Bioengineered Vascularized Skin Substitutes. Int. J. Mol. Sci. 2020, 21, 8197. [Google Scholar] [CrossRef] [PubMed]
- Krzyszczyk, P.; Schloss, R.; Palmer, A.; Berthiaume, F. The Role of Macrophages in Acute and Chronic Wound Healing and Interventions to Promote Pro-wound Healing Phenotypes. Front. Physiol. 2018, 9, 419. [Google Scholar] [CrossRef] [PubMed]
- Finnson, K.W.; McLean, S.; Di Guglielmo, G.M.; Philip, A. Dynamics of Transforming Growth Factor Beta Signaling in Wound Healing and Scarring. Adv. Wound Care 2013, 2, 195–214. [Google Scholar] [CrossRef] [PubMed]
- Moretti, L.; Stalfort, J.; Barker, T.H.; Abebayehu, D. The interplay of fibroblasts, the extracellular matrix, and inflammation in scar formation. J. Biol. Chem. 2022, 298, 101530. [Google Scholar] [CrossRef] [PubMed]
- Maita, K.C.; Avila, F.R.; A Torres-Guzman, R.; Garcia, J.P.; Eldaly, A.S.; Palmieri, L.; Emam, O.S.; Ho, O.; Forte, A.J. Local anti-inflammatory effect and immunomodulatory activity of chitosan-based dressing in skin wound healing: A systematic review. . 2022, 8, 488–498. [Google Scholar] [PubMed]
- Fayyazbakhsh, F.; Leu, M.C. A Brief Review on 3D Bioprinted Skin Substitutes. Procedia Manuf. 2020, 48, 790–796. [Google Scholar] [CrossRef]
- Roh, D.S.; Li, E.B.-H.; Liao, E.C. CRISPR Craft: DNA Editing the Reconstructive Ladder. Plast. Reconstr. Surg. 2018, 142, 1355–1364. [Google Scholar] [CrossRef] [PubMed]
- Pang, C.; Fan, K.S.; Wei, L.; Kolar, M.K. Gene therapy in wound healing using nanotechnology. Wound Repair Regen. 2021, 29, 225–239. [Google Scholar] [CrossRef] [PubMed]
- Samsell, B.; McLean, J.; Cazzell, S.; Dorsch, K.; Moyer, P.M.; Moore, M. Health economics for treatment of diabetic foot ulcers: a cost-effectiveness analysis of eight skin substitutes. J. Wound Care 2019, 28, S14–S26. [Google Scholar] [CrossRef] [PubMed]
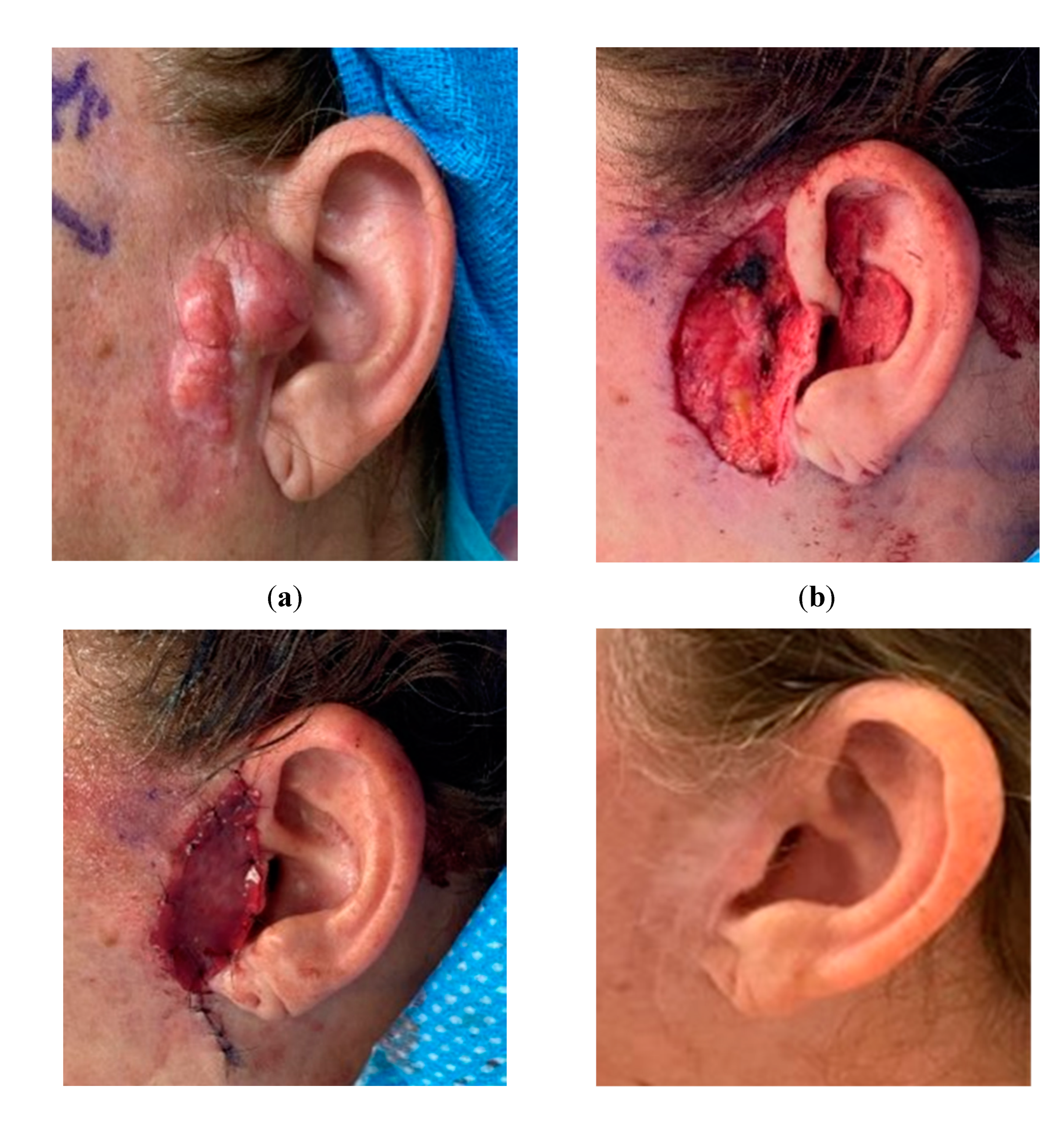
Figure 1.
Use of bilayer dermal regeneration template in the face. a. A 50-year-old female with a preauricular benign histiocytoma from a facelift scar. b. Resection of the lesion resulted in a full-thickness defect of 6 x 4 cm. c. The absence of facial skin laxity due to the previous facelift did not allow the advancement of a flap over the defect, which was covered with a bilayered dermal regeneration template d. Successful wound healing with acceptable aesthetic result.
Figure 1.
Use of bilayer dermal regeneration template in the face. a. A 50-year-old female with a preauricular benign histiocytoma from a facelift scar. b. Resection of the lesion resulted in a full-thickness defect of 6 x 4 cm. c. The absence of facial skin laxity due to the previous facelift did not allow the advancement of a flap over the defect, which was covered with a bilayered dermal regeneration template d. Successful wound healing with acceptable aesthetic result.
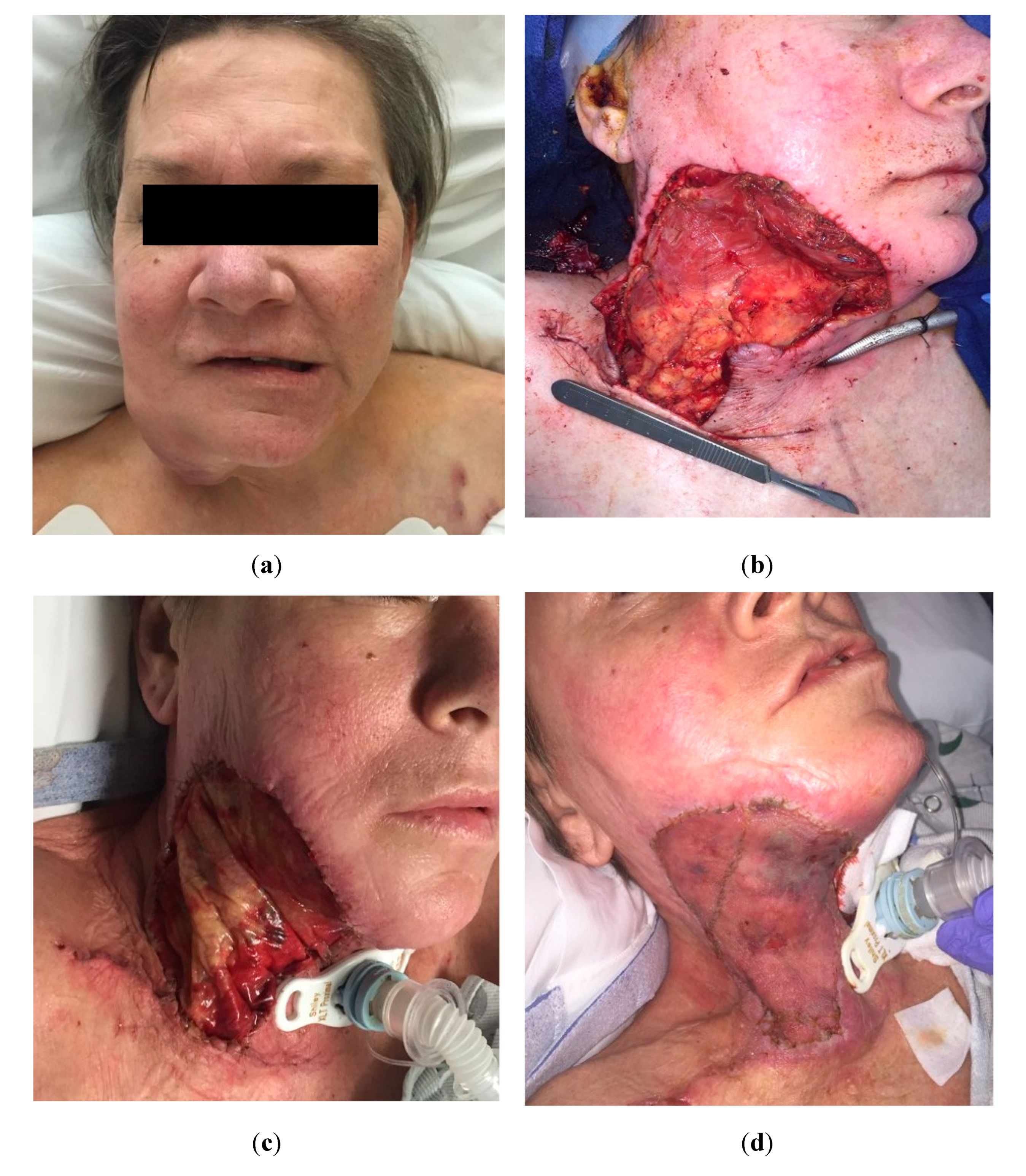
Figure 2.
Utilization of bilayer dermal regeneration template, negative pressure wound therapy, and split-thickness skin graft in the neck. a. A 60-year-old female with an oropharyngeal squamous cell carcinoma extending to the neck. b Extensive mandibular resection defect covered with a pediculated pectoralis major muscle flap. c Bilayer dermal regeneration template applied on the exposed muscle flap. d Skin graft three weeks later, provided stable coverage for adjuvant radiotherapy.
Figure 2.
Utilization of bilayer dermal regeneration template, negative pressure wound therapy, and split-thickness skin graft in the neck. a. A 60-year-old female with an oropharyngeal squamous cell carcinoma extending to the neck. b Extensive mandibular resection defect covered with a pediculated pectoralis major muscle flap. c Bilayer dermal regeneration template applied on the exposed muscle flap. d Skin graft three weeks later, provided stable coverage for adjuvant radiotherapy.
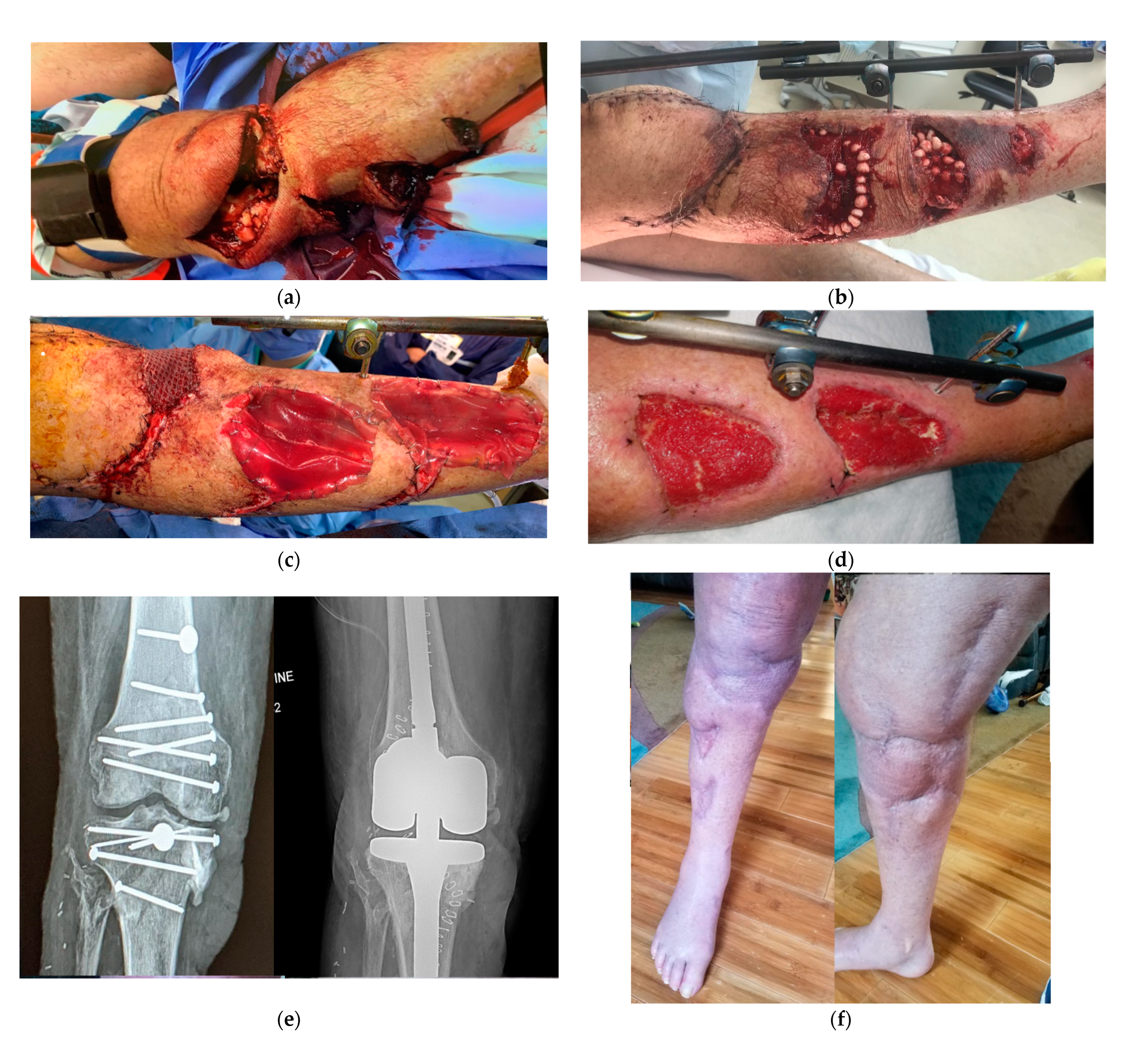
Figure 3.
Bilayer dermal regeneration template, particulate urinary bladder matrix, and split-thickness skin grafts for coverage of a complex lower extremity injury. a. A mid-60s male with a propeller injury. Vascular and orthopedic intervention was required due to Gustilo IIIC tibial fracture. b. The patient underwent multiple debridement, application of wound antibiotic beads, and negative pressure wound dressing. c. A large knee defect was covered with a reverse gracilis muscle flap and skin graft, while two large defects over the leg were temporarily cover with bilayered dermal matrix. d. Wound bed optimized for skin graft take. e. Patient required total knee arthroplasty 12 months later due to the severity of the injury. f. Patient ultimately had a full restoration of function.
Figure 3.
Bilayer dermal regeneration template, particulate urinary bladder matrix, and split-thickness skin grafts for coverage of a complex lower extremity injury. a. A mid-60s male with a propeller injury. Vascular and orthopedic intervention was required due to Gustilo IIIC tibial fracture. b. The patient underwent multiple debridement, application of wound antibiotic beads, and negative pressure wound dressing. c. A large knee defect was covered with a reverse gracilis muscle flap and skin graft, while two large defects over the leg were temporarily cover with bilayered dermal matrix. d. Wound bed optimized for skin graft take. e. Patient required total knee arthroplasty 12 months later due to the severity of the injury. f. Patient ultimately had a full restoration of function.
Figure 4.
Particulate and laminated urinary bladder matrix under negative pressure wound therapy to treat lower extremity venous ulcer. a. A mid-40s morbidly obese female with a large infected venous ulcer that failed to improve after months of wound care and pressure dressing. b. IV antibiotics, surgical debridement, and wound preparation were performed before skin substitute application. c. A split-thickness skin graft provided final coverage, improving patient's quality of life.
Figure 4.
Particulate and laminated urinary bladder matrix under negative pressure wound therapy to treat lower extremity venous ulcer. a. A mid-40s morbidly obese female with a large infected venous ulcer that failed to improve after months of wound care and pressure dressing. b. IV antibiotics, surgical debridement, and wound preparation were performed before skin substitute application. c. A split-thickness skin graft provided final coverage, improving patient's quality of life.
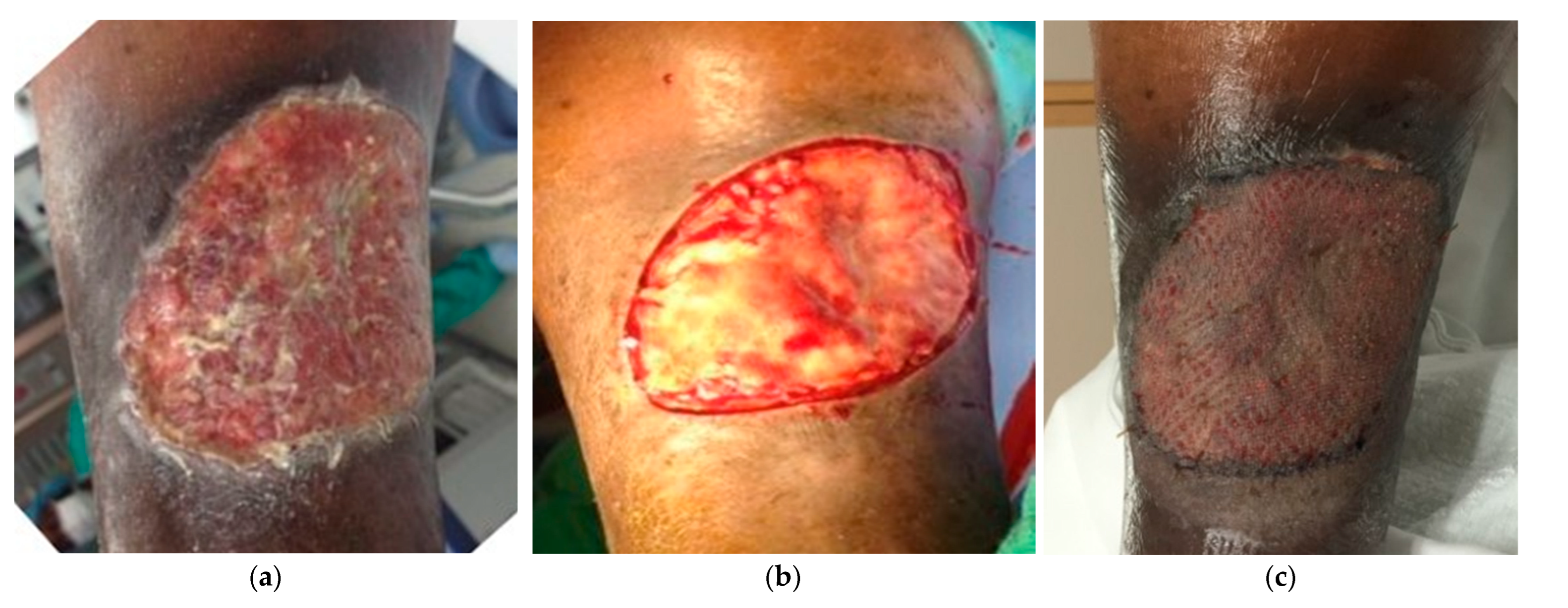
Figure 5.
Application of porcine urinary bladder matrix and split-thickness skin graft in a diabetic foot ulcer. a. 28-year-old male, diabetic type I, with a necrotizing infection. b. Multiple debridements were performed for local infection control exposing the extensor tendons. c. Urinary bladder ECM applied over the wound. d. Complete healing and foot salvage despite a poor initial prognosis.
Figure 5.
Application of porcine urinary bladder matrix and split-thickness skin graft in a diabetic foot ulcer. a. 28-year-old male, diabetic type I, with a necrotizing infection. b. Multiple debridements were performed for local infection control exposing the extensor tendons. c. Urinary bladder ECM applied over the wound. d. Complete healing and foot salvage despite a poor initial prognosis.
Figure 6.
Urinary bladder matrix reinforcement of pediculated gracilis muscle flap. a. 43-year-old female with recurrent melanoma and previous radiation to left groin. Presented with a non-healing, infected, and painful wound, with failed previous reconstruction attempts. b. Gracilis muscle flap was used to fill the volume defect. c. UBM covering temporarily an irregular wound bed. d. Wound ultimately covered with a skin graft. Despite successful wound management, patient's unfortunate passing was attributed to disease progression. .
Figure 6.
Urinary bladder matrix reinforcement of pediculated gracilis muscle flap. a. 43-year-old female with recurrent melanoma and previous radiation to left groin. Presented with a non-healing, infected, and painful wound, with failed previous reconstruction attempts. b. Gracilis muscle flap was used to fill the volume defect. c. UBM covering temporarily an irregular wound bed. d. Wound ultimately covered with a skin graft. Despite successful wound management, patient's unfortunate passing was attributed to disease progression. .
Figure 7.
Particulate extracellular matrix with paravertebral and trapezium muscle flaps. a. 66-year-old female with multiple cervical spine surgeries, complicated with hardware infection. b, c, d. Following hardware removal and debridement, reconstruction was completed with paravertebral and trapezium muscle flaps, reinforced with morselized and laminated ECM allograft. e. Wound healing after 12 days.
Figure 7.
Particulate extracellular matrix with paravertebral and trapezium muscle flaps. a. 66-year-old female with multiple cervical spine surgeries, complicated with hardware infection. b, c, d. Following hardware removal and debridement, reconstruction was completed with paravertebral and trapezium muscle flaps, reinforced with morselized and laminated ECM allograft. e. Wound healing after 12 days.
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).