Submitted:
22 January 2024
Posted:
22 January 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
3. Results
Acute and Chronic Complications
4. Discussion
5. Study Limitations
6. Conclusions
References
- Hui JYC. Epidemiology and Etiology of Sarcomas. Surg Clin North Am. 2016;96:901-14. [CrossRef]
- American Cancer Society. Cancer Facts & Figures 2020. Atlanta: American Cancer Society; 2020.
- Instituto Nacional de Cancerología. Anuario estadístico 2020. Vol. 18. Bogotá: INC; 2020.
- Pisters, P. W. , Weiss, M., & Maki, R. (2011). Soft-tissue sarcomas. Cancer Management: A Multidisciplinary Approach Medical, Surgical, & Radiation Oncology, 4th ed., UBM Medica LLC, Norwalk, CT.
- Gamboa AC, Gronchi A, Cardona K. Soft-tissue sarcoma in adults: An update on the current state of histiotype-specific management in an era of personalized medicine. CA Cancer J Clin. 2020;70(3):200-29. [CrossRef]
- Keung EZ, Chiang YJ, Voss RK, Cormier JN, Torres KE, Hunt KK, et al. Defining the incidence and clinical significance of lymph node metastasis in soft tissue sarcoma. Eur J Surg Oncol. 2018;44(1):170-7. [CrossRef]
- Sbaraglia M, Dei Tos AP. The pathology of soft tissue sarcomas. Radiol Medica. 2019;124(4):266-81. [CrossRef]
- Amin MB, Greene FL, Edge SB, Compton CC, Gershenwald JE, Brookland RK, et al. The Eighth Edition AJCC Cancer Staging Manual: Continuing to build a bridge from a population-based to a more “personalized” approach to cancer staging. CA Cancer J Clin. 2017;67(2):93-9. [CrossRef]
- Voss RK, Chiang YJ, Torres KE, Guadagnolo BA, Mann GN, Feig BW, et al. Adherence to National Comprehensive Cancer Network Guidelines is Associated with Improved Survival for Patients with Stage 2A and Stages 2B and 3 Extremity and Superficial Trunk Soft Tissue Sarcoma. Ann Surg Oncol. 2017;24(11):3271-8. [CrossRef]
- Billingsley KG, Lewis JJ, Leung DHY, Casper ES, Woodruff JM, Brennan MF. Multifactorial analysis of the survival of patients with distant metastasis arising from primary extremity sarcoma. Cancer. 1999;85(2):389-95.
- Wasif N, Smith CA, Tamurian RM, Christensen SD, Monjazeb AM, Martinez SR, et al. Influence of physician specialty on treatment recommendations in the multidisciplinary management of soft tissue sarcoma of the extremities. JAMA Surg. 2013;148(7):632-9. [CrossRef]
- Rosenberg SA, Tepper J, Glatstein E, Costa J, Baker A, Brennan M, et al. The treatment of soft-tissue sarcomas of the extremities: prospective randomized evaluations of (1) limb-sparing surgery plus radiation therapy compared with amputation and (2) the role of adjuvant chemotherapy. Ann Surg. 1982;196(3):305-15. [CrossRef]
- O’Sullivan B, Davis AM, Turcotte R, Bell R, Catton C, Chabot P, et al. Preoperative versus postoperative radiotherapy in soft-tissue sarcoma of the limbs: a randomised trial. Lancet. 2002;359(9325):2235-41. [CrossRef]
- Tierney JF, Mosseri V, Stewart LA, Souhami RL, Parmar MK. Adjuvant chemotherapy for soft-tissue sarcoma: review and meta-analysis of the published results of randomised clinical trials. Br J Cancer. 1995;72(2):469-75. [CrossRef]
- Blay JY, Honoré C, Stoeckle E, Meeus P, Jafari M, Gouin F, et al. Surgery in reference centers improves survival of sarcoma patients: a nationwide study. Ann Oncol. 2019;30(7):1143-53. [CrossRef]
- Demetri GD, Blay JY, Casali PG. Advances and controversies in the management of soft tissue sarcomas. Future Oncol. 2016;13(1S):3-11. [CrossRef]
- Weskamp P, Ufton D, Drysch M, Wagner JM, Dadras M, Lehnhardt M, et al. Risk Factors for Occurrence and Relapse of Soft Tissue Sarcoma. Cancers (Basel). 2022;14(5):1273. [CrossRef]
- Gamboa AC, Gronchi A, Cardona K. Soft-tissue sarcoma in adults: An update on the current state of histiotype-specific management in an era of personalized medicine. CA Cancer J Clin. 2020;70(3):200-29. [CrossRef]
- Coindre JM, Trojani M, Contesso G, David M, Rouesse J, Bui NB, et al. Reproducibility of a histopathologic grading system for adult soft tissue sarcoma. Cancer. 1986;58(2):306-9. [CrossRef]
- Brennan, M. F., Antonescu, C. R., Moraco, N., & Singer, S. (2014). Lessons learned from the study of 10,000 patients with soft tissue sarcoma. Annals of surgery, 260(3), 416.
- Coindre J-M, Terrier P, Bui NB, Bonichon O, Collin F, Le Doussal V, et al. Prognostic factors in adult patients with locally controlled soft tissue sarcoma. A study of 546 patients from the French Feredarion of Cancer Centers Sarcoma Group. J Clin Oncol. 1996;14(3):869-77. [CrossRef]
- Gaynor JJ, Tan CC, Casper ES, Collin CF, Friedrich C, Shiu M, et al. Refinement of clinicopathologic staging for localized soft tissue sarcoma of the extremity: A study of 423 adults. J Clin Oncol. 1992;10(8):1317-29. [CrossRef]
- Pisters PW, Leung DH, Woodruff J, Shi W, Brennan MF. Analysis of prognostic factors in 1,041 patients with localized soft tissue sarcomas of the extremities. J Clin Oncol. 1996;14(5):1679-89. [CrossRef]
- Stefanovski PD, Bidoli E, De Paoli A, Buonadonna A, Boz G, Libra M, et al. Prognostic factors in soft tissue sarcomas: A study of 395 patients. Eur J Surg Oncol. 2002;28(2):153-64. [CrossRef]
- Gronchi A, Casali PG, Mariani L, Miceli R, Fiore M, Lo Vullo S, et al. Status of surgical margins and prognosis in adult soft tissue sarcomas of the extremities: A series of patients treated at a single institution. J Clin Oncol. 2005;23(1):96-104. [CrossRef]
- Stojadinovic A, Leung DHY, Hoos A, Jaques DP, Lewis JJ, Brennan MF. Analysis of the prognostic significance of microscopic margins in 2,084 localized primary adult soft tissue sarcomas. Ann Surg. 2002;235(3):424-34. [CrossRef]
- Trovik CS, Bauer HCF, Alvegård TA, Anderson H, Blomqvist C, Berlin, et al. Surgical margins, local recurrence and metastasis in soft tissue sarcomas: 559 surgically-treated patients from the Scandinavian Sarcoma Group Register. Eur J Cancer. 2000;36(6):710-6. [CrossRef]
- Zagars GK, Ballo MT, Pisters PWT, Pollock RE, Patel SR, Benjamin RS, et al. Prognostic factors for patients with localized soft-tissue sarcoma treated with conservation surgery and radiation therapy: An analysis of 1225 patients. Cancer. 2003;97(10):2530-43. [CrossRef]
- Rosenthal J, Cardona K, Sayyid SK, Perricone AJ, Reimer N, Monson D, et al. Nodal metastases of soft tissue sarcomas: risk factors, imaging findings, and implications. Skeletal Radiol. 2020;49(2):221-9. [CrossRef]
- avis, A. M., O'Sullivan, B., Turcotte, R., Bell, R., Catton, C., Chabot, P., ... & Randomized, N. C. C. T. G. (2005). Late radiation morbidity following randomization to preoperative versus postoperative radiotherapy in extremity soft tissue sarcoma. Radiotherapy and oncology, 75(1), 48-53.
- Gingrich AA, Bateni SB, Monjazeb AM, Darrow M, Thorpe SW, Kirane AR, et al. Neoadjuvant Radiotherapy is Associated with R0 Resection and Improved Survival in Extremity Soft Tissue Sarcoma Patients Undergoing Surgery: An NCDB Analysis Alicia. Ann Surg Oncol. 2018;24(11):3252-63. [CrossRef]
- Broecker JS, Ethun CG, Monson DK, Lopez-Aguiar AG, Le N, McInnis M, et al. The Oncologic Impact of Postoperative Complications Following Resection of Truncal and Extremity Soft Tissue Sarcomas. Ann Surg Oncol. 2017;24(12):3574-86. [CrossRef]
- Schwartz A, Rebecca A, Smith A, Casey W, Ashman J, Gunderson L, et al. Risk factors for significant wound complications following wide resection of extremity soft tissue sarcomas. Clin Orthop Relat Res. 2013;471(11):3612-7. [CrossRef]
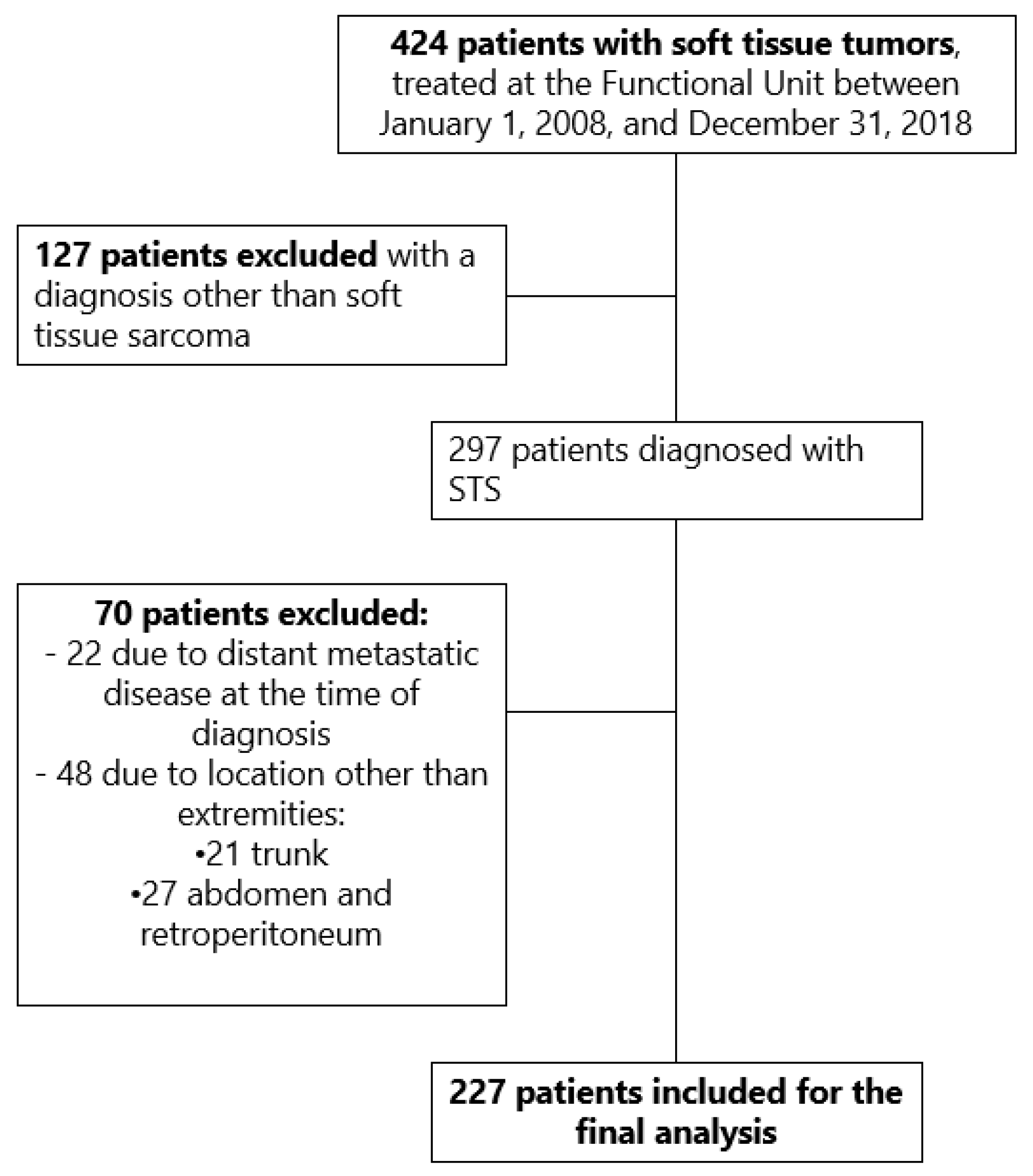
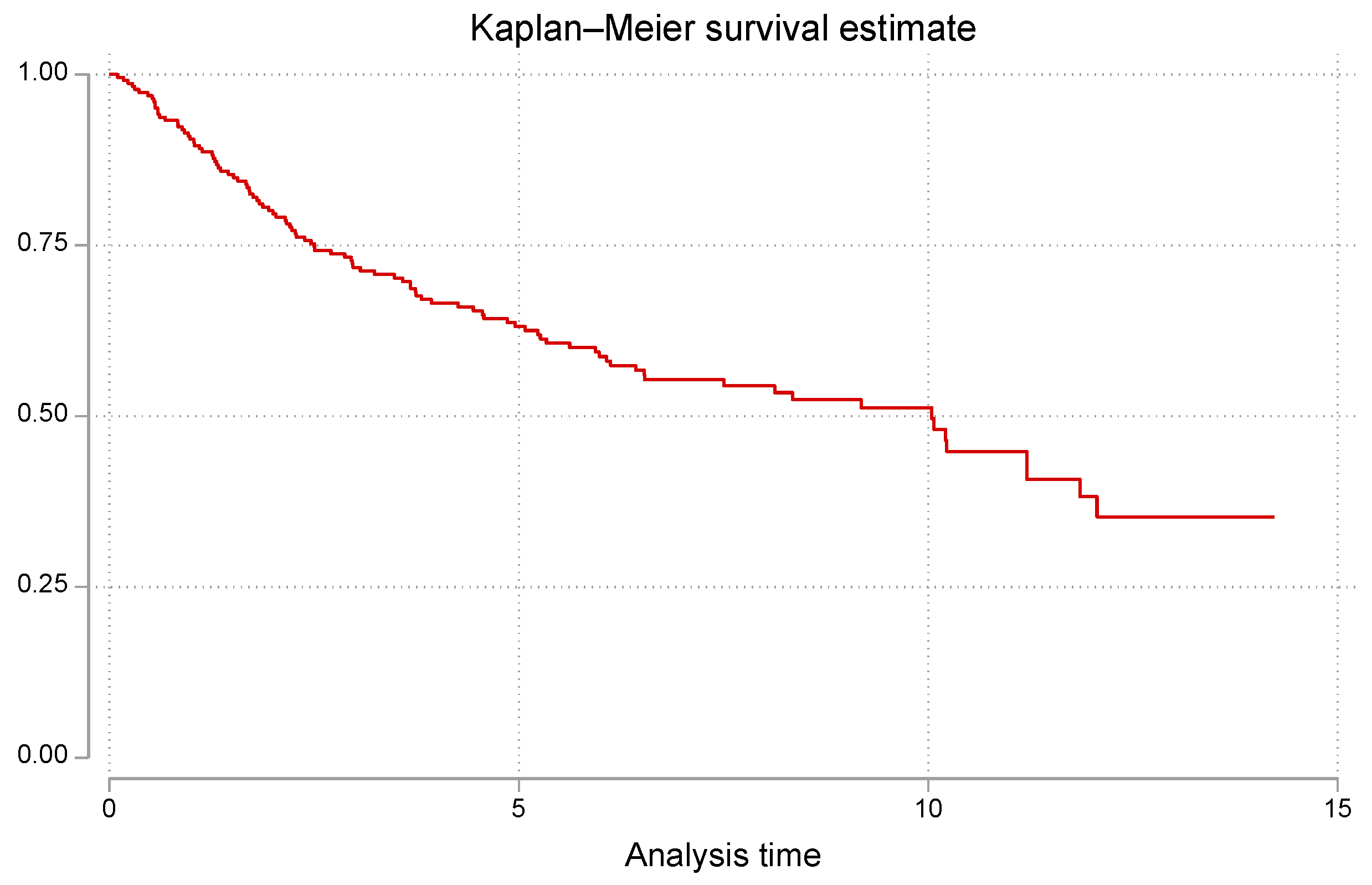
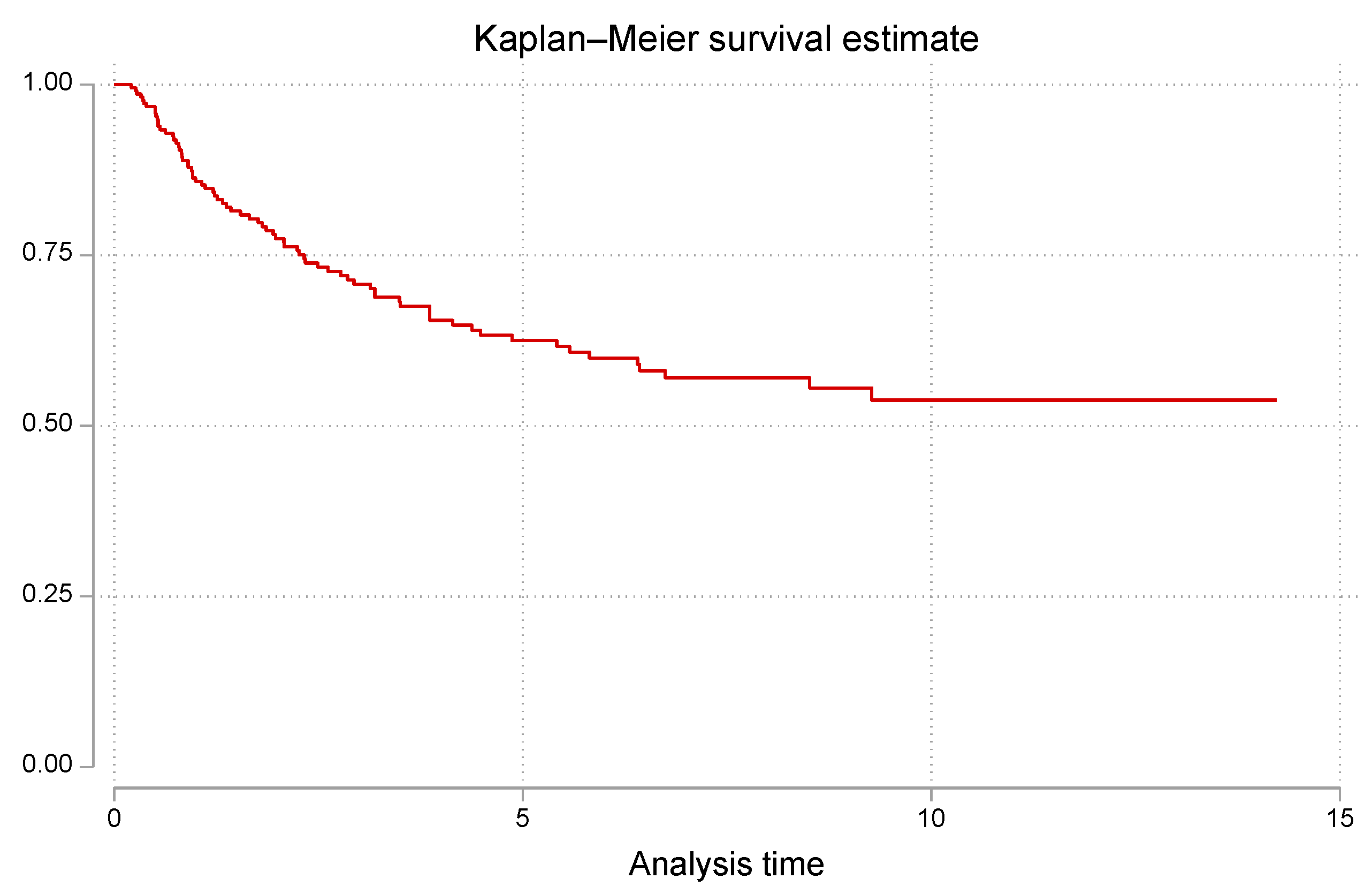
| Characteristics | Total of patients (n=227), n (%) |
|---|---|
| Median age (years) | 53 (18-89) |
|
Sex Men Women |
117 (51.5) 110 (48.5) |
|
Presentation type Primary without treatment Tumor persistence Tumor recurrence |
142 (62.5) 63 (27.8) 22 (9.7) |
|
Histologic type Well-differentiated liposarcoma Undifferentiated pleomorphic sarcoma Myxoid liposarcoma Synovial sarcoma Myxofibrosarcoma Leiomyosarcoma Malignant neural sheath tumor Others |
39 (17.2) 39 (17.2) 33 (14.5) 25 (11) 21 (9.3) 19 (8.4) 16 (7) 35 (15.4) |
|
Histologic grade I II III No data |
67 (29.5) 12 (5.3) 147 (64.8) 1 (0.4) |
|
Tumor size T1 T2 T3 T4 No data (Initial surgery outside the INC) |
53 (23.3) 64 (28.1) 40 (17.7) 65 (28.7) 5 (2.2) |
|
Clinical stage IA IB II IIIA IIIB IV (Lymph node involvement) Not applicable (Dermatofibrosarcoma protuberans) |
14 (6.2) 43 (18.9) 35 (15.4) 57 (25.1) 66 (29.1) 3 (1.3) 9 (3.9) |
|
Tumor location Thigh Leg Forearm Gluteus Foot Arm Shoulder Hand |
108 (47.7) 39 (17.2) 23 (10.1) 16 (7) 13 (5.7) 12 (5.3) 10 (4.4) 6 (2.6) |
| Administered treatment | Total of patients (n=228), n (%) |
|---|---|
|
Initial treatment type Surgical treatment Neoadjuvant RT ILP Neoadjuvant CHT |
170 (74.9) 27 (11.9) 23 (10.1) 7 (3.1) |
|
Type of primary tumor surgery Wide local resection Amputation Widening of margins of previous non-oncological surgery outside the INC Compartmental resection Marginal resection |
130 (57.4) 54 (23.8) 28 (12.3) 9 (3.9) 6 (2.6) |
|
Additional interventions Sentinel lymph node Lymph node dissection IORT |
19 (8.3) 18 (7.9) 14 (6.1) |
|
Positive margins in INC pathology No Yes Planned positive margins |
189 (83.3) 34 (14.9) 6 (17.7) |
|
Surgery to achieve negative margins Widening of margins |
11 (4.8) |
|
Adjuvant treatment Adjuvant CHT Adjuvant RT |
51 (22.4) 85 (37.3) |
| Variable | Overall Survival Hazard Ratio (IC95%) | Recurrence-Free Survival Hazard Ratio (IC95%) | ||
|---|---|---|---|---|
| Age | 1.01 (1–1.02) | p=0.048 | 0.99 (0.983–1.015) |
p=0.92 |
| Tumor size | 1.01 (0.99–1.04) | p=0.18 | 1.03 (0.996–1.056) | p=0.08 |
|
Histologic grade I II III |
Ref. 0.69 (0.007–0.67) 0.13 (0.01–1.1) |
p= 0.021 p=0.062 |
Ref. 1.97 (0.49–7.9) 5.36 (2.29–12.56) |
p=0.33 p<0.001 |
|
Type of primary tumor surgery Amputation Compartmental resection Wide local resection Marginal resection Widening of margins of previous non-oncological surgery outside the INC |
Ref. 0.69 (0.2–2.23) 0.48 (0.24–0.96) 1.81 (0.47–6.9) 0.36 (0.12–1.1) |
p=0.56 p=0.038 p=0.38 p=0.075 |
Ref. 0.68 (0.16–2.83) 0.56 (0.26–1.21) 2.16 (0.41–11.3) 0.16 (0.045–0.6) |
p=0.6 p=0.14 p=0.36 p=0.006 |
|
Positive margins in INC pathology Yes No |
1.11 (0.54–2.26) Ref. |
p=0.76 |
1.56 (0.75–3.2) Ref. |
p=0.23 |
|
Presentation type Primary without treatment Tumor persistence Tumor recurrence |
Ref. 0.95 (0.51–1.77) 0.83 (0.39–1.76) |
p=0.88 p=0.63 |
Ref. 2.34 (1.25–4.35) 1.62 (0.74–3.53) |
p=0.007 p=0.21 |
|
Initial treatment type Surgical treatment Neoadjuvant CHT Neoadjuvant RT ILP |
Ref. 1.18 (0.43–3.24) 0.7 (0.3–1.65) 0.75 (0.36–1.54) |
p=0.73 p=0.42 p=0.44 |
Ref. 0.68 (0.15–3.03) 0.86 (0.35–2.09) 0.62 (0.25–1.52) |
p=0.61 p=0.75 p=0.75 |
|
Adjuvant treatment Adjuvant CHT Yes No Adjuvant RT Yes No |
0.66 (0.38–1.14) Ref. 0.7 (0.38–1.27) Ref. |
p=0.14 p=0.24 |
1.14 (0.3–1.2) Ref. 0.6 (0.3–1.2) Ref. |
p=0.15 p=1.2 |
| Variable | Local Recurrence Hazard Ratio (IC95%) | Distant Recurrence Hazard Ratio (IC95%) | ||
|---|---|---|---|---|
| Age | 0.99 (0.77–1.02) | p=0.77 | 0.99 (0.983–1.019) |
p=0.93 |
| Tumor size |
1 (0.95–1.05) |
p=0.89 |
1.03 (1.007–1.067) |
p=0.015 |
|
Histologic grade I II III |
Ref. 1.1 (0.11–10.5) 6.09 (2.03–18.2) |
p=0.92 p=0.001 |
Ref. 2.21 (0.37–13.08) 4.53 (1.42–14.49) |
p=0.38 p=0.011 |
|
Type of primary tumor surgery Amputation Compartmental resection Wide local resection Marginal resection Widening of margins of previous non-oncological surgery outside the INC |
Ref. 3.43 (0.48–24.63) 1.69 (0.46–6.1) 8.88 (0.75–104.4) 0.51 (0.08–3.06) |
p=0.21 p=0.42 p=0.08 p=0.46 |
Ref. 0.48 (0.086–2.74) 0.37 (0.14–0.94) 0.78 (0.08–7.52) 0.07 (0.008–0.64) |
p=0.41 p=0.038 p=0.83 p=0.019 |
|
Positive margins in INC pathology Yes No |
1.74 (0.69–4.34) Ref. |
p=0.23 |
1 (0.37–2.72) Ref. |
p=0.98 |
|
Presentation type Primary without treatment Tumor persistence Tumor recurrence |
Ref. 2.85 (1.23–6.57) 1.6 (0.5–5.07) |
p=0.014 p=0.42 |
Ref. 1.49 (0.65–3.41) 1.42 (0.55–3.64) |
p=0.33 p=0.46 |
|
Initial treatment type Surgical treatment Neoadjuvant CHT Neoadjuvant RT ILP |
Ref. 0.81 (0.09–6.84) 0.77 (0.21–2.65) 1.02 (0.32–3.24) |
p=0.85 p=0.65 p=0.96 |
Ref. 1.35 (0.28–6.44) 0.98 (0.32–3.03) 0.50 (0.14–1.78) |
p=0.7 p=0.98 p=0.29 |
|
Adjuvant treatment Adjuvant CHT Yes No Adjuvant RT Yes No |
0.75 (0.33–1.69) Ref. 0.55 (0.22–1.4) Ref. |
p=0.49 p=0.21 |
1.25 (0.62–2.54) Ref. 0.82 (0.33–2.01) Ref. |
p=0.52 p=0.66 |
| Variable | Acute Complications Coefficient (β) (IC95%) | Chronic Complications Odds Ratio (IC95%) | ||
|---|---|---|---|---|
| Age | 0.017 (0.002–0.031) | p=0.022 | 1 (0.96–1.03) |
p=0.89 |
| Tumor size | 0.036 (0.007–0.065) | p=0.015 | 0.96 (0.87–1.07) | p=0.52 |
|
Histologic grade I II III |
Ref. 0.19 (0.82–1.22) 0.41 (0.19–1.02) |
p=0.7 p=0.18 |
Ref. 1.1 (0.07–15.76) 1.55 (0.3–7.9) |
p=0.94 p=0.59 |
|
Type of primary tumor surgery Amputation Compartmental resection Wide local resection Marginal resection Widening of margins of previous non-oncological surgery outside the INC |
Ref. 1.37 (0.1–2.63) 1.38 (0.43–2.33) 1.47 (-0.12–3.06) 1.69 (0.34–3.05) |
- p=0.03 p=0.004 p=0.07 p=0.014 |
Ref. - 2.49 (0.23–26.46) 7.33 (0.24–219.49) 0.77 (0.027–21.82) |
p=0.44 p=0.25 p=0.88 |
|
Positive margins in INC pathology Yes No |
Ref. -0.07 (-0.754–0.596) |
p=0.81 |
Ref. 0.62 (0.11–3.37) |
p=0.58 |
|
Presentation type Primary without treatment Tumor persistence Tumor recurrence |
Ref. -0.24 (-1.01–0.52) 0.95 (0.32–1.59) |
p=0.53 p=0.003 |
Ref. 1.53 (0.32–7.25) 3.35 (0.68–16.33) |
p=0.59 p=0.13 |
|
Initial treatment type Surgical treatment Neoadjuvant CHT Neoadjuvant RT ILP |
Ref. -0.14 (-1.63–1.34) 0.16 (-0.05–0.88) -0.21 (-1.18–0.76) |
p=0.84 p=0.65 p=0.67 |
Ref. 2.31 (0.18–29.12) 3.53 (0.44–28.42) 2.41 (0.37–15.68) |
p=0.51 p=0.23 p=0.35 |
|
Adjuvant treatment Adjuvant CHT Yes No Adjuvant RT Yes No |
0.02 (-0.57–0.62) Ref. -0.03 (-0.6–0.53) Ref. |
p=0.93 p=0.9 |
0.55 (0.11–2.64) Ref. 4.36 (0.98–19.26) Ref. |
p=0.45 p=0.052 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




