Submitted:
22 January 2024
Posted:
22 January 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Methods
2.2.1. Cell cultures
2.2.2. Assessment of Mitochondrial Function in Microtiter Wells
2.2.3. Oxygen consumption and extracellular acidification rates
2.2.4. Mitochondrial DNA content
2.2.5. SOD2 mRNA Expression
2.2.6. Statistical analysis
3. Results
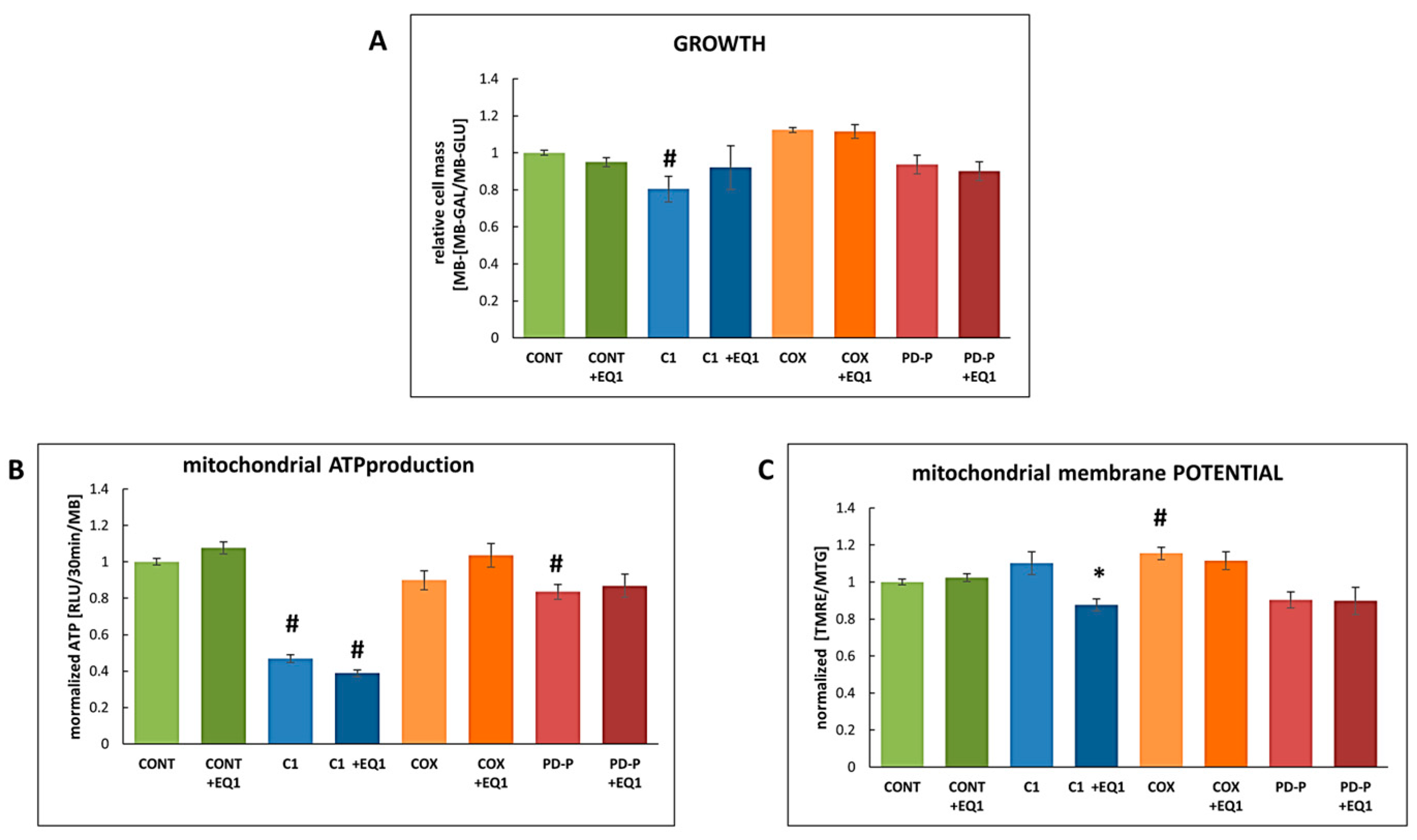
3.1. The Effect of EQ on mitochondrial parameters
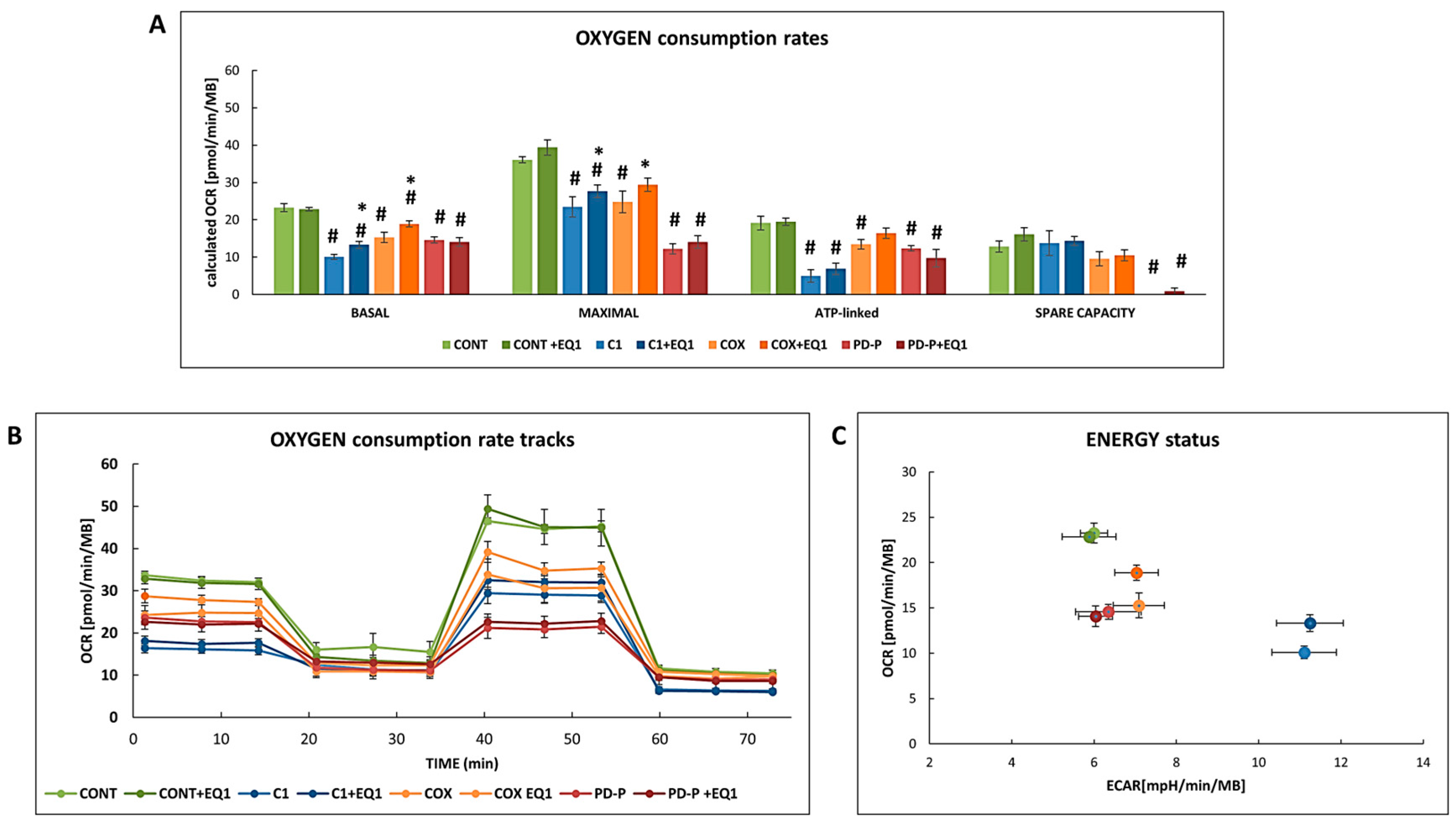
3.2. The Effect of EQ on bioenergetics
3.3. The Effect of EQ on cellular ROS production and mitochondrial content
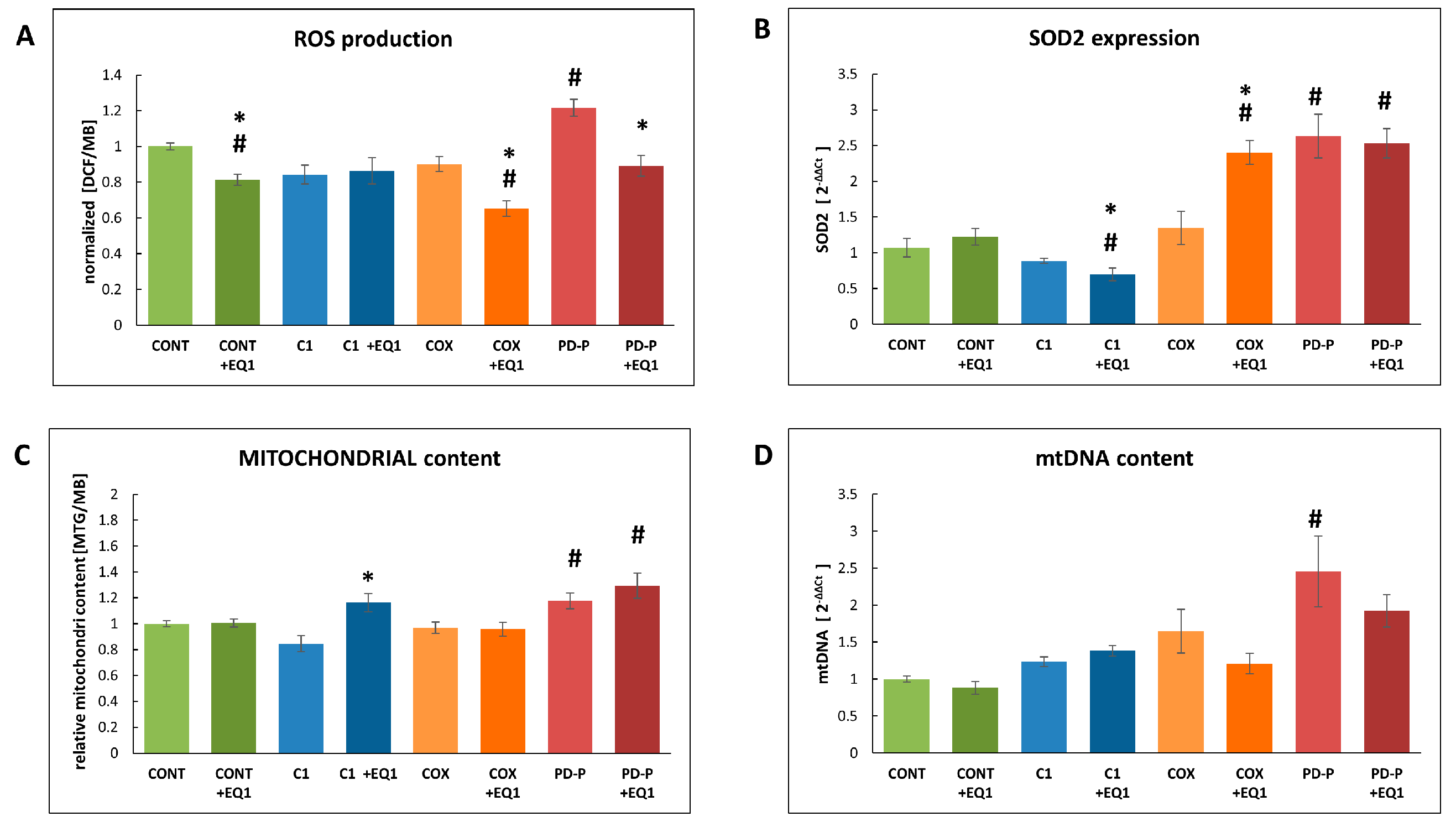
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- de Koning,A.J. (2002). The antioxidants ethoxyquin and its analogues: a review International Journal of Food Properties, 2002,5(2) 451-461. [CrossRef]
- Zhu, J., Chen, W., Mi, R., Zhou, C., Reed, N., & Höke, A. (2013). Ethoxyquin prevents chemotherapy-induced neurotoxicity via Hsp90 modulation. Annals of neurology, 74(6),2013, 893–904. [CrossRef]
- Cetinkaya-Fisgin, A., Zhu, J., Luan, X., Kim, J. S., Oh, B., Brayton, C., Alt, J., Rais, R., Slusher, B., & Höke, A. (2021). Development of EQ-6, a Novel Analogue of Ethoxyquin to Prevent Chemotherapy-Induced Peripheral Neuropathy. Neurotherapeutics: the journal of the American Society for Experimental NeuroTherapeutics, 18(3), 2061–2072. [CrossRef]
- Jeong, Y. E., Rajbhandari, L., Kim, B. W., Venkatesan, A., & Hoke, A. (2023). Downregulation of SF3B2 protects CNS neurons in models of multiple sclerosis. Annals of clinical and translational neurology, 10(2), 246–265. [CrossRef]
- Iskusnykh, I. Y., Kryl'skii, E. D., Brazhnikova, D. A., Popova, T. N., Shikhaliev, K. S., Shulgin, K. K., Matasova, L. V., Popov, S. S., Zhaglin, D. A., Zakharova, A. A., Popova, N. R., & Fattakhov, N. (2021). Novel Antioxidant, Deethylated Ethoxyquin, Protects against Carbon Tetrachloride Induced Hepatotoxicity in Rats by Inhibiting NLRP3 Inflammasome Activation and Apoptosis. Antioxidants (Basel, Switzerland), 10(1), 122. [CrossRef]
- Saada, A., Edvardson, S., Rapoport, M., Shaag, A., Amry, K., Miller, C., Lorberboum-Galski, H., & Elpeleg, O. (2008). C6ORF66 is an assembly factor of mitochondrial complex I. American journal of human genetics, 82(1), 32–38. [CrossRef]
- Abu-Libdeh, B., Douiev, L., Amro, S., Shahrour, M., Ta-Shma, A., Miller, C., Elpeleg, O., & Saada, A. (2017). Mutation in the COX4I1 gene is associated with short stature, poor weight gain and increased chromosomal breaks, simulating Fanconi anemia. European journal of human genetics : EJHG, 25(10), 1142–1146. [CrossRef]
- Mor-Shaked, H., Paz-Ebstein, E., Basal, A., Ben-Haim, S., Grobe, H., Heymann, S., Israel, Z., Namnah, M., Nitzan, A., Rosenbluh, C., Saada, A., Tzur, T., Yanovsky-Dagan, S., Zaidel-Bar, R., Harel, T., & Arkadir, D. (2021). Levodopa-responsive dystonia caused by biallelic PRKN exon inversion invisible to exome sequencing. Brain communications, 3(3), fcab197. [CrossRef]
- Yu-Wai-Man, P., Soiferman, D., Moore, D. G., Burté, F., & Saada, A. (2017). Evaluating the therapeutic potential of idebenone and related quinone analogues in Leber hereditary optic neuropathy. Mitochondrion, 36, 36–42. (. [CrossRef]
- Zighan, M., Arkadir, D., Douiev, L., Keller, G., Miller, C., & Saada, A. (2022). Variable effects of omaveloxolone (RTA408) on primary fibroblasts with mitochondrial defects. Frontiers in molecular biosciences, 9, 890653. (. [CrossRef]
- Venegas, V., Wang, J., Dimmock, D., and Wong, L. J. (2011). Real-time quantitative PCR analysis of mitochondrial DNA content. Curr. Protoc. Hum. Genet. 19. 19.17. [CrossRef]
- Islam, H., Bonafiglia, J. T., Turnbull, P. C., Simpson, C. A., Perry, C. G. R., & Gurd, B. J. (2020). The impact of acute and chronic exercise on Nrf2 expression in relation to markers of mitochondrial biogenesis in human skeletal muscle. European journal of applied physiology, 120(1), 149–160. [CrossRef]
- Golubitzky, A., Dan, P., Weissman, S., Link, G., Wikstrom, J. D., & Saada, A. (2011). Screening for active small molecules in mitochondrial complex I deficient patient's fibroblasts, reveals AICAR as the most beneficial compound. PloS one, 6(10), e26883. [CrossRef]
- Pitkänen, S., Feigenbaum, A., Laframboise, R., & Robinson, B. H. (1996). NADH-coenzyme Q reductase (complex I) deficiency: heterogeneity in phenotype and biochemical findings. Journal of inherited metabolic disease, 19(5), 675–686. [CrossRef]
- Palma, F. R., Gantner, B. N., Sakiyama, M. J., Kayzuka, C., Shukla, S., Lacchini, R., Cunniff, B., & Bonini, M. G. (2023). ROS production by mitochondria: function or dysfunction?. Oncogene. [CrossRef]
- Aquilina et al.(2015) .Safety and efficacy of ethoxyquin (6-ethoxy-1,2-dihydro-2,2,4-trimethylquinoline) for all animal species EFSA Journal;13(11):4272. [CrossRef]
- Murphy M. P. (2009). How mitochondria produce reactive oxygen species. The Biochemical journal, 417(1), 1–13. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).



