Submitted:
16 January 2024
Posted:
17 January 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Reagents
2.1. Sample extraction
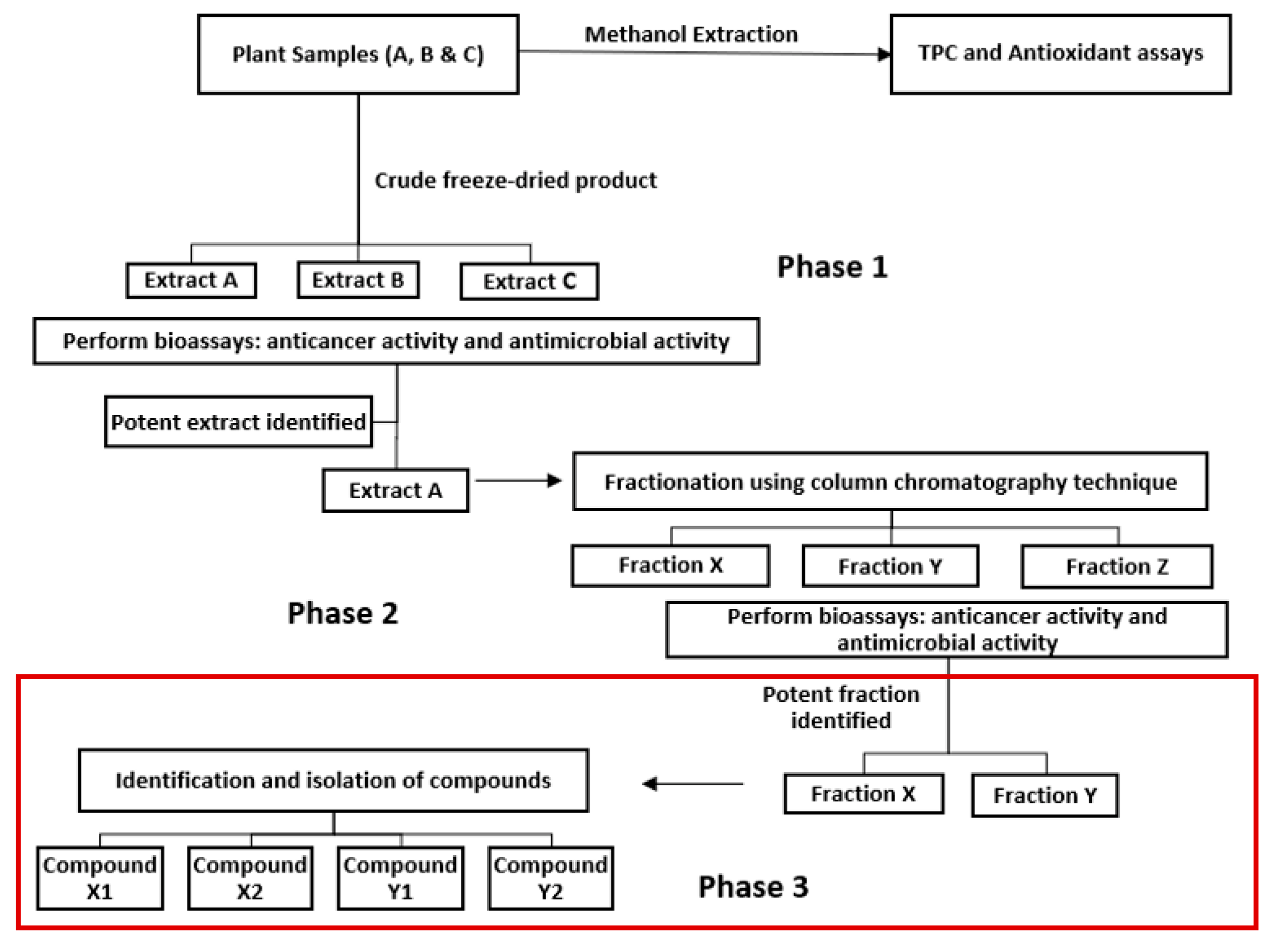
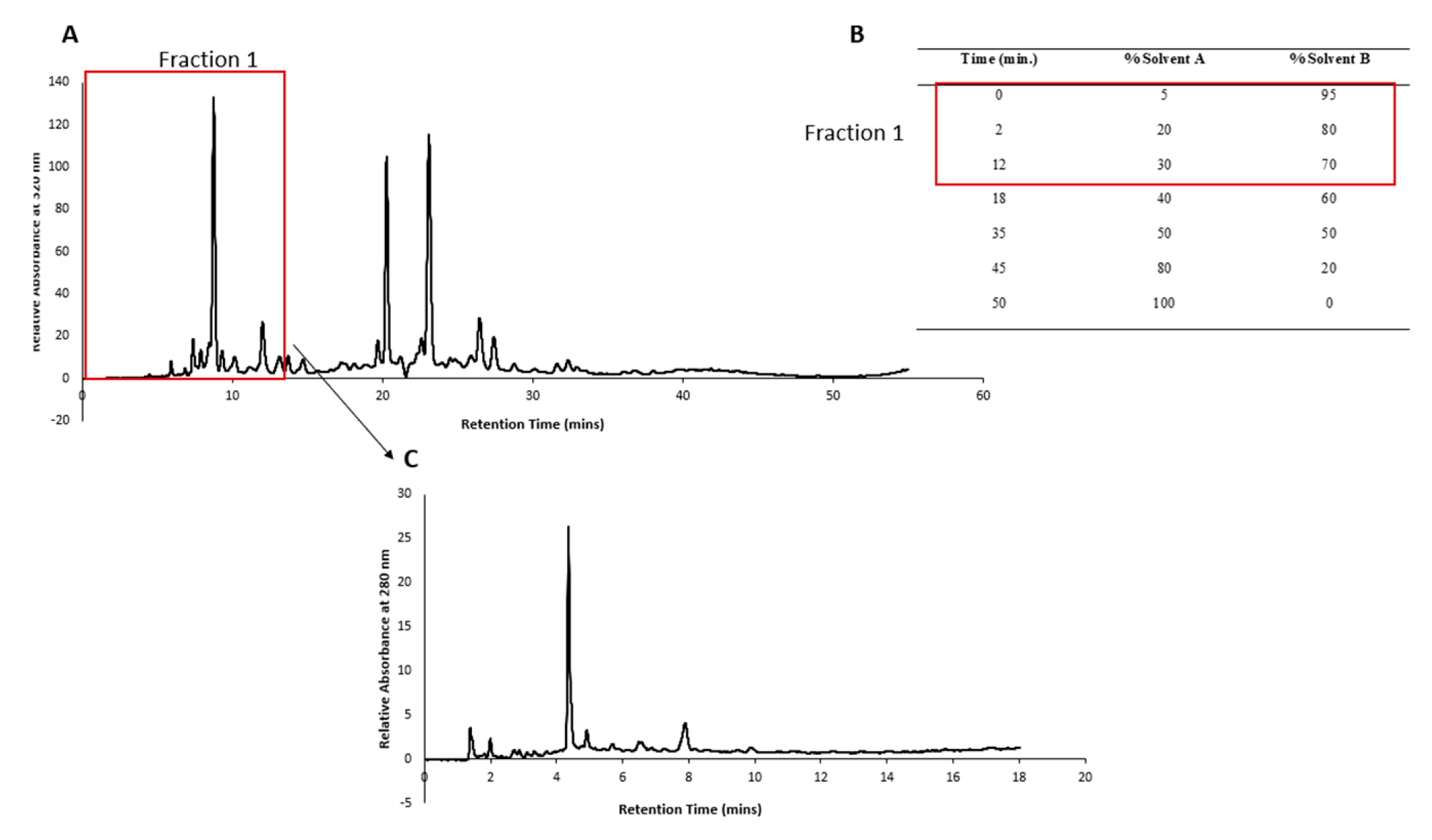
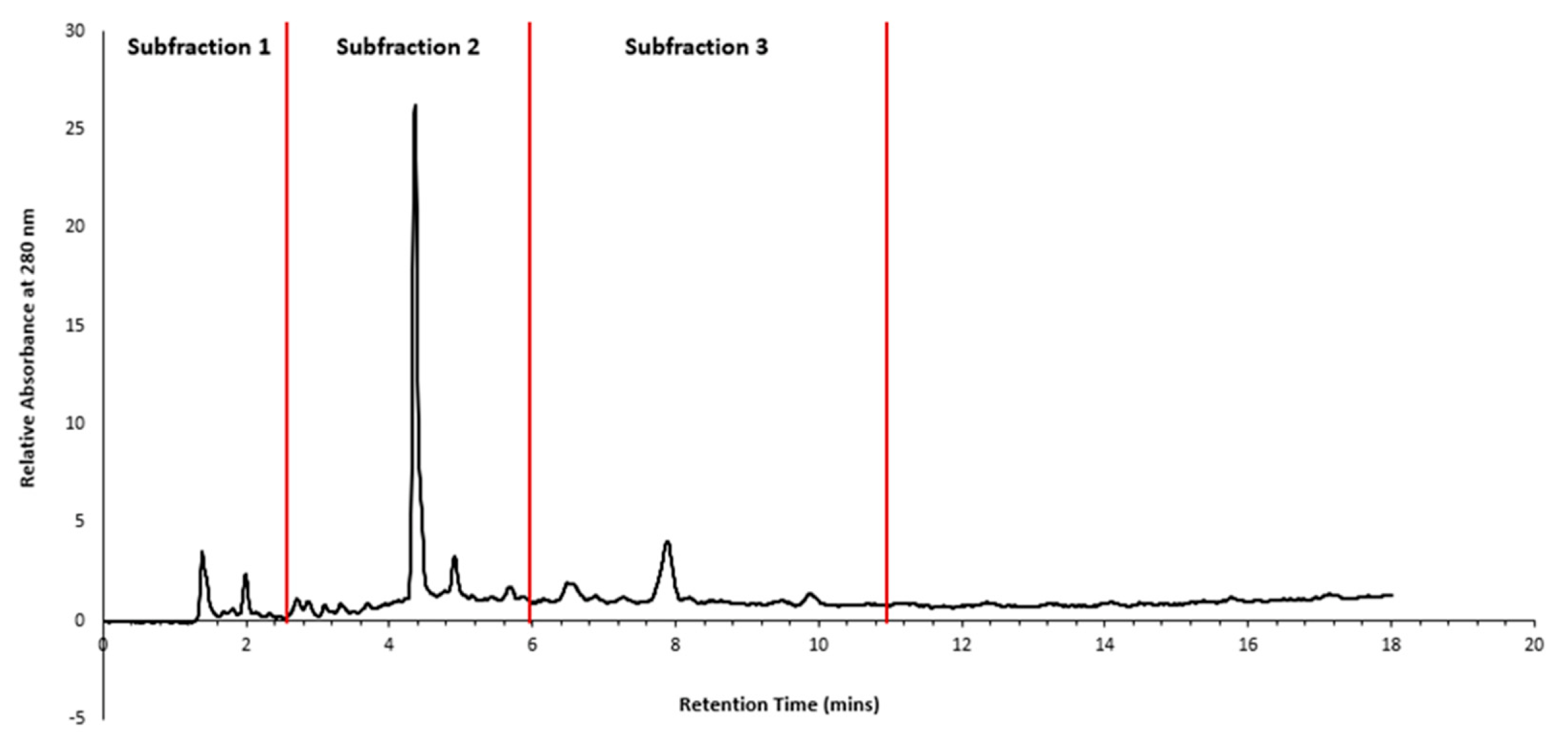
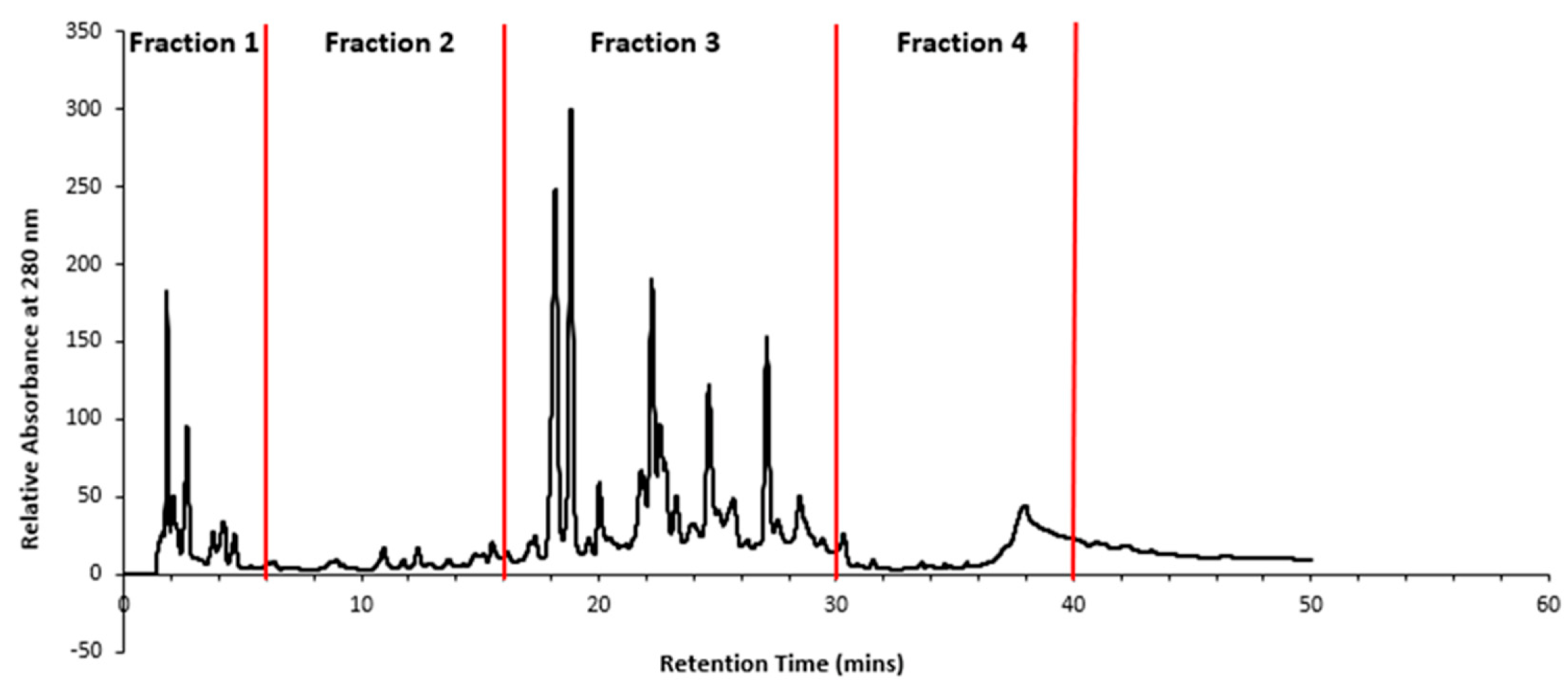
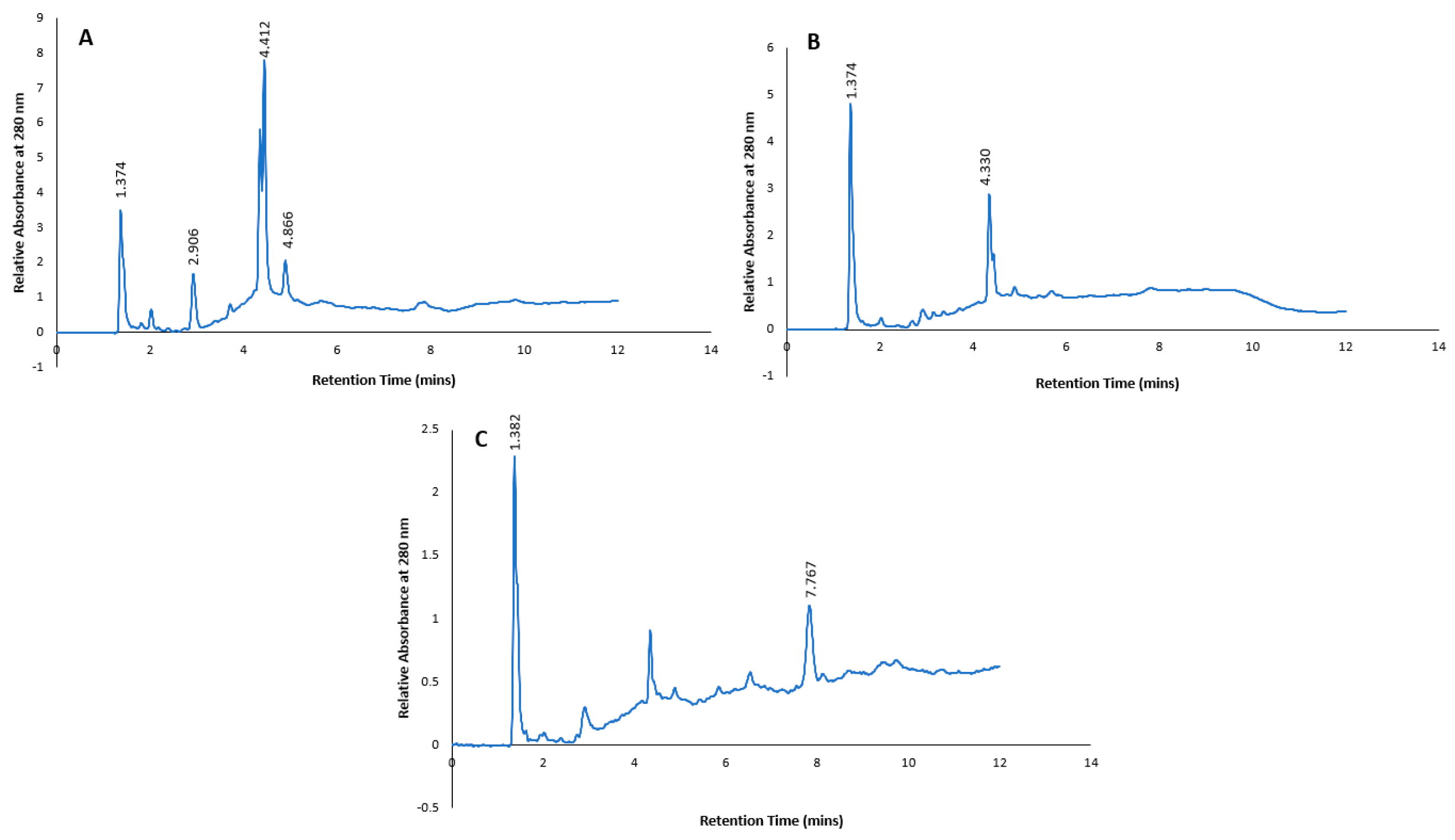
2.2. HPLC fractionation and sub-fractionation of P. angustifolium extract
2.3. HPLC fractionation of T. ferdinandiana flesh extract
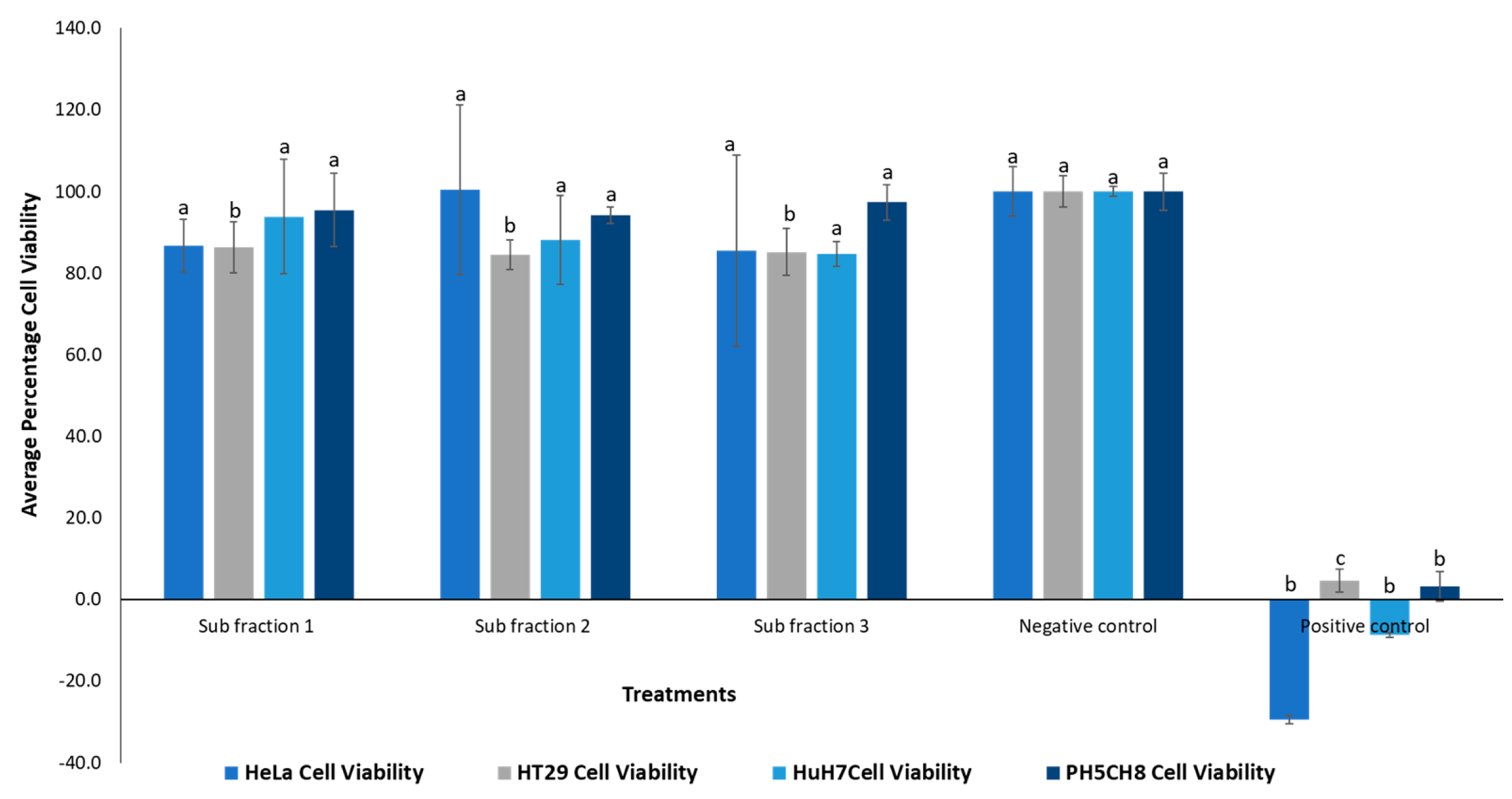
2.4. Cytotoxicity Assay
2.5. Antimicrobial Activity
2.6. LC-MS/MS Analysis of P. angustifolium fraction
2.7. GC-MS/MS Analysis of P. angustifolium fraction 1 and subfractions
2.8. Statistical Analysis
3. Results and discussion
3.1. Pittosporum angustifolium sub fractions
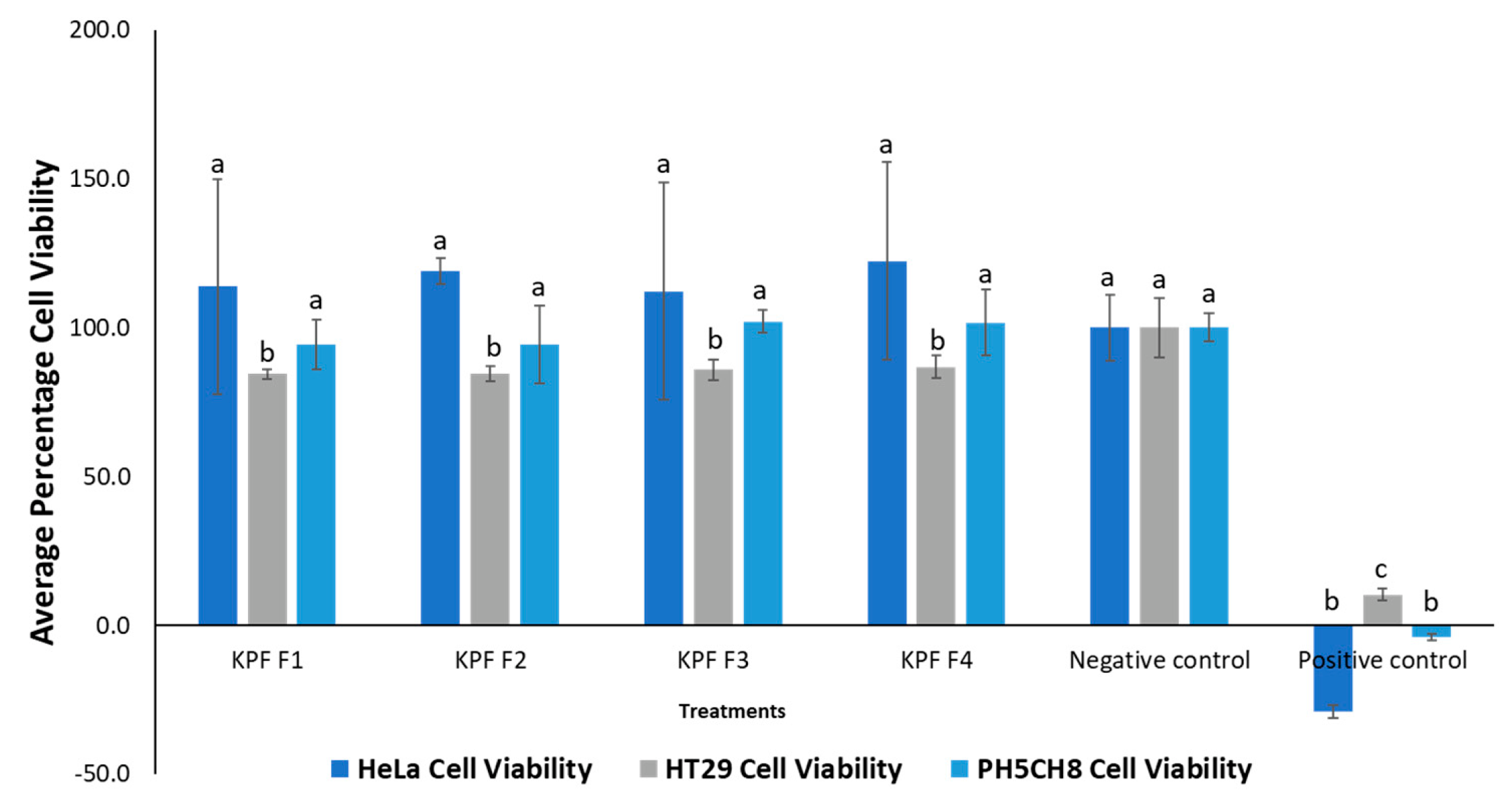
3.1.1. Cytotoxic Activity and HPLC Profiling
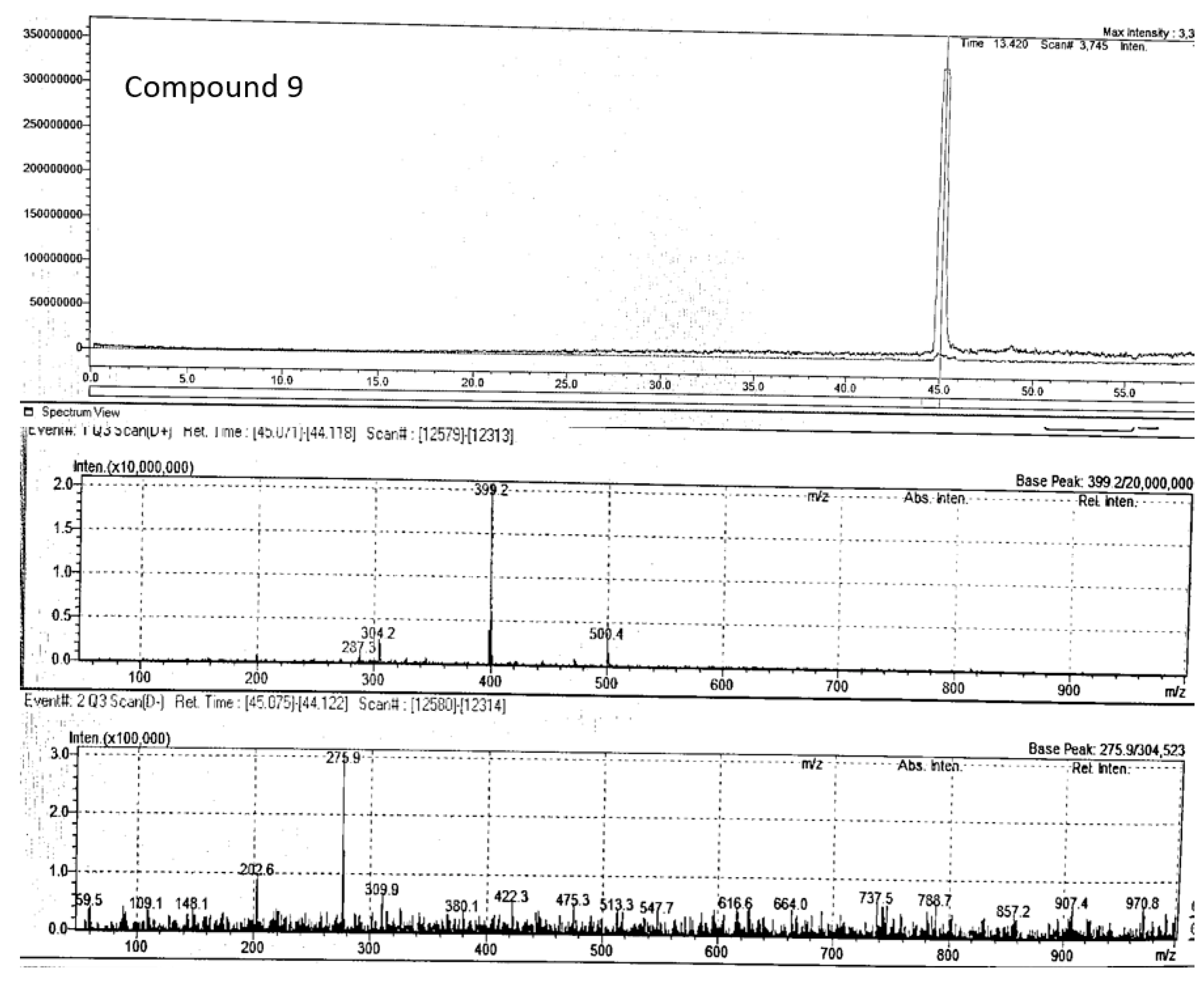
3.1.2. LC- MS Analysis of P. angustifolium Fraction 1
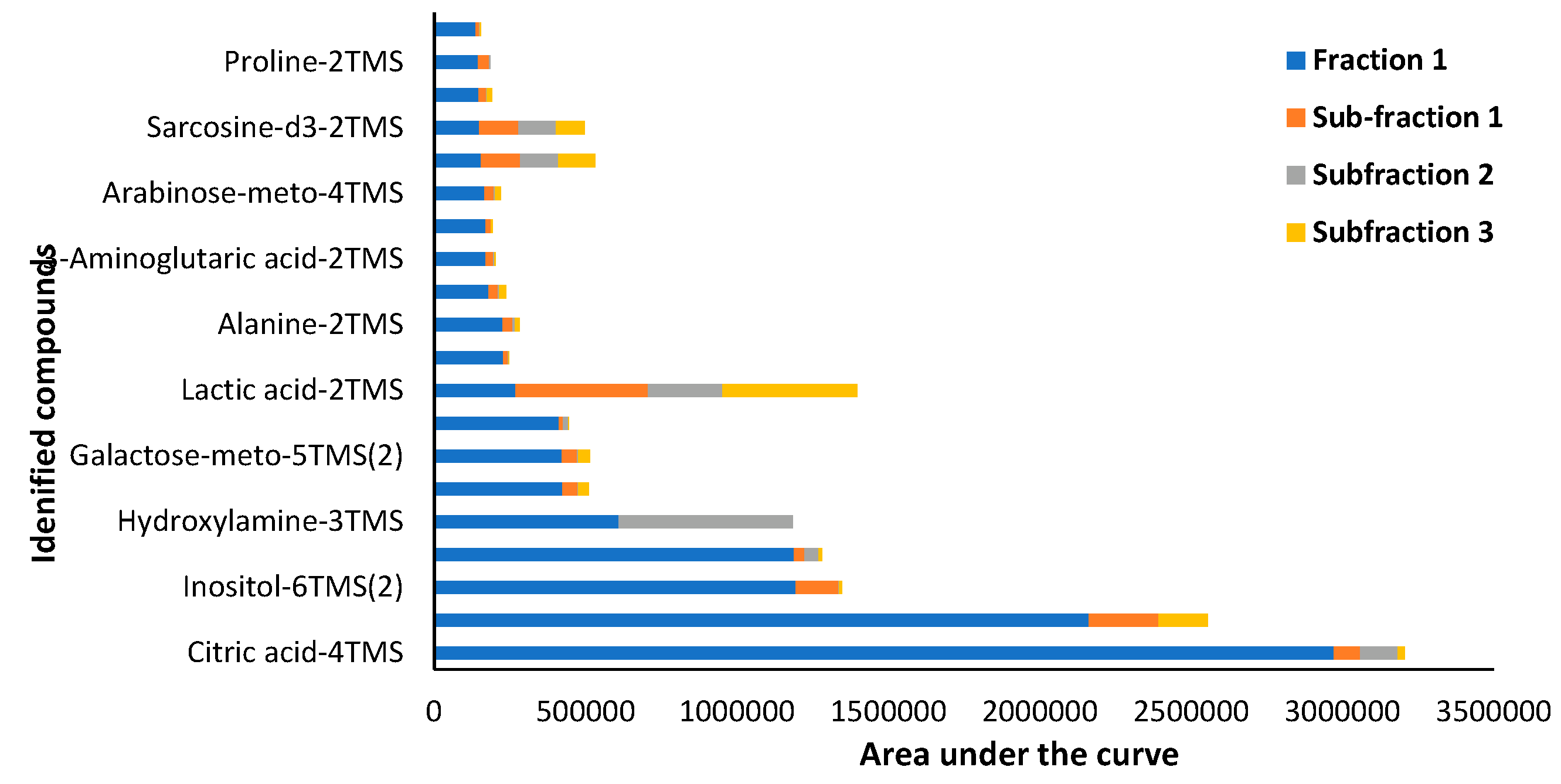
3.1.3. GC- MS Analysis of Pittosporum angustifolium Fraction 1 and sub-fractions
3.2. Terminalia ferdinandiana Fractions
3.2.1. Cytotoxic Activity
3.2.2. Antibacterial Activity
4. Conclusion
References
- Abdallah, M. S., Mustafa, M., Nallappan, M. A., Choi, S., Paik, J. H., & Rusea, G. (2021). Determination of Phenolics and Flavonoids of Some Useful Medicinal Plants and Bioassay-Guided Fractionation Substances of Sclerocarya birrea (A. Rich) Hochst Stem (Bark) Extract and Their Efficacy Against Salmonella typhi. Frontiers in Chemistry, 9(July), 1–13. [CrossRef]
- Akter, S., Netzel, M. E., Fletcher, M. T., Tinggi, U., & Sultanbawa, Y. (2018). Chemical and nutritional composition of terminalia ferdinandiana (kakadu plum) kernels: A novel nutrition source. Foods, 7(4). [CrossRef]
- Akter, S., Sultanbawa, Y., & Cozzolino, D. (2021). High throughput screening to determine the antibacterial activity of Terminalia ferdinandiana (Kakadu plum): A proof of concept. Journal of Microbiological Methods, 182(February), 106169. [CrossRef]
- Ali, A., Cottrell, J. J., & Dunshea, F. R. (2022). LC-MS/MS Characterization of Phenolic Metabolites and Their Antioxidant Activities from Australian Native Plants. Metabolites, 12(11). [CrossRef]
- Amaral, D. C., & Brown, P. H. (2022). Foliar Application of an Inositol-Based Plant Biostimulant Boosts Zinc Accumulation in Wheat Grains: A μ-X-Ray Fluorescence Case Study. Frontiers in Plant Science, 13(April), 1–9. [CrossRef]
- Bäcker, C., Jenett-Siems, K., Bodtke, A., & Lindequist, U. (2014). Polyphenolic compounds from the leaves of pittosporum angustifolium. Biochemical Systematics and Ecology, 55, 101–103. [CrossRef]
- Bäcker, C., Jenett-Siems, K., Siems, K., Wurster, M., Bodtke, A., & Lindequist, U. (2014). Cytotoxic saponins from the seeds of Pittosporum angustifolium. Zeitschrift Fur Naturforschung - Section C Journal of Biosciences, 69 C(5–6), 191–198. [CrossRef]
- Beh, C. C., & Teoh, W. H. (2022). Recent Advances in the Extraction of Pittosporum angustifolium Lodd. Used in Traditional Aboriginal Medicine: A Mini Review. Nutraceuticals, 2(2), 49–59. [CrossRef]
- Bizzarri, M., Fuso, A., Dinicola, S., Cucina, A., & Bevilacqua, A. (2016). Pharmacodynamics and pharmacokinetics of inositol(s) in health and disease. Expert Opinion on Drug Metabolism and Toxicology, 12(10), 1181–1196. [CrossRef]
- Blonk, B., & Cock, I. E. (2019). Interactive antimicrobial and toxicity profiles of Pittosporum angustifolium Lodd. extracts with conventional antimicrobials. Journal of Integrative Medicine, 17(4), 261–272. [CrossRef]
- Deo, P., Hewawasam, E., Karakoulakis, A., Claudie, D. J., Nelson, R., Simpson, B. S., Smith, N. M., & Semple, S. J. (2016). In vitro inhibitory activities of selected Australian medicinal plant extracts against protein glycation, angiotensin converting enzyme (ACE) and digestive enzymes linked to type II diabetes. BMC Complementary and Alternative Medicine, 16(435). [CrossRef]
- Feng, W., Li, M., Hao, Z., & Zhang, J. (2019). Analytical Methods of Isolation and Identification. In Phytochemicals in Human Health (p. 13). IntechOpen. [CrossRef]
- Gorman, J. T., Wurm, P. A. S., Vemuri, S., Brady, C., & Sultanbawa, Y. (2019). Kakadu Plum (Terminalia ferdinandiana) as a Sustainable Indigenous Agribusiness. Economic Botany, X, 1–18. [CrossRef]
- Jang, M. H., Piao, X. L., Kim, J. M., Kwon, S. W., Park, J. H., Raju, M., Kulkarni, Y. A., & Wairkar, S. (2008). Inhibition of cholinesterase and amyloid-&bgr; aggregation by resveratrol oligomers from Vitis amurensis. Journal of Functional Foods, 61(August), 103517.
- Jiao, Q. S., Xu, L. L., Zhang, J. Y., Wang, Z. J., Jiang, Y. Y., & Liu, B. (2018). Rapid Characterization and Identification of Non-Diterpenoid Constituents in Tinospora sinensis by HPLC-LTQ-Orbitrap MSn. Molecules, 23(2). [CrossRef]
- Kulkarni, S. K., & Dhir, A. (2010). Berberine: A Plant Alkaloid with Therapeutic Potential for Central Nervous System Disorders. Phytotherapy Research, 24, 317–324. [CrossRef]
- Liang, Y., Xie, L., Liu, K., Cao, Y., Dai, X., Wang, X., Lu, J., Zhang, X., & Li, X. (2021). Bergapten: A review of its pharmacology, pharmacokinetics, and toxicity. Phytotherapy Research, 35(11), 6131–6147. [CrossRef]
- Liu, J., Qu, L., Meng, L., & Shou, C. (2019). Topoisomerase inhibitors promote cancer cell motility via ROS-mediated activation of JAK2-STAT1-CXCL1 pathway. Journal of Experimental and Clinical Cancer Research, 38(1), 1–12. [CrossRef]
- Mani, J., Johnson, J., Hosking, H., Hoyos, B. E., Walsh, K. B., Neilsen, P., & Naiker, M. (2022). Bioassay Guided Fractionation Protocol for Determining Novel Active Compounds in Selected Australian Flora. Plants, 11(21), 2886. [CrossRef]
- Mani, J., Johnson, J., Hosking, H., Walsh, K., Neilsen, P., & Naiker, M. (2022a). In vitro Cytotoxic Properties of Crude Polar Extracts of Plants Sourced from Australia. Clinical Complementary Medicine and Pharmacology, 2(1), 100022. [CrossRef]
- Mani, J., Johnson, J., Hosking, H., Walsh, K., Neilsen, P., & Naiker, M. (2022b). In vitro Cytotoxic Properties of Crude Polar Extracts of Plants Sourced from Australia. Clinical Complementary Medicine and Pharmacology, 2(1), 100022. [CrossRef]
- Mekky, R. H., Abdel-Sattar, E., Segura-Carretero, A., & Del Mar Contreras, M. (2019). Phenolic compounds from sesame cake and antioxidant activity: A new insight for agri-food residues’ significance for sustainable development. Foods, 8(10). [CrossRef]
- Naiker, M., Anderson, S., Johnson, J. B., Mani, J. S., Wakeling, L., & Bowry, V. (2020). Loss of trans-resveratrol during storage and ageing of red wines. Australian Journal of Grape and Wine Research, 26(4), 385–387. [CrossRef]
- Noé, W., Murhekar, S., White, A., Davis, C., & Cock, I. E. (2019). Inhibition of the growth of human dermatophytic pathogens by selected australian and asian plants traditionally used to treat fungal infections. Journal de Mycologie Medicale, 29(4), 331–344. [CrossRef]
- Noor, S., Mohammad, T., Rub, M. A., Raza, A., Azum, N., Yadav, D. K., Hassan, M. I., & Asiri, A. M. (2022). Biomedical features and therapeutic potential of rosmarinic acid. Archives of Pharmacal Research, 45(4), 205–228. [CrossRef]
- Nour, V., Trandafir, I., & Cosmulescu, S. (2013). HPLC determination of phenolic acids, flavonoids and juglone in walnut leaves. Journal of Chromatographic Science, 51(9), 883–890. [CrossRef]
- Nunes, S., Madureira, A. R., Campos, D., Sarmento, B., Gomes, A. M., Pintado, M., & Reis, F. (2017). Therapeutic and nutraceutical potential of rosmarinic acid—Cytoprotective properties and pharmacokinetic profile. Critical Reviews in Food Science and Nutrition, 57(9), 1799–1806. [CrossRef]
- Omar, R., El-salam, M. A., Elsbaey, M., & Hassan, M. (2023). Fourteen immunomodulatory alkaloids and two prenylated phenylpropanoids with dual therapeutic approach for COVID-19 : molecular docking and dynamics studies. Journal of Biomolecular Structure and Dynamics, 0(0), 1–18. [CrossRef]
- Phan, A. D. T., Chaliha, M., Hong, H. T., Tinggi, U., Netzel, M. E., & Sultanbawa, Y. (2020). Nutritional value and antimicrobial activity of Pittosporum angustifolium (gumby gumby), an australian indigenous plant. Foods, 9(7). [CrossRef]
- Quetglas-Llabrés, M. M., Quispe, C., Herrera-Bravo, J., Catarino, M. D., Pereira, O. R., Cardoso, S. M., Dua, K., Chellappan, D. K., Pabreja, K., Satija, S., Mehta, M., Sureda, A., Martorell, M., Satmbekova, D., Yeskaliyeva, B., Sharifi-Rad, J., Rasool, N., Butnariu, M., Bagiu, I. C., … Cho, W. C. (2022). Pharmacological Properties of Bergapten: Mechanistic and Therapeutic Aspects. Oxidative Medicine and Cellular Longevity, 2022(December 2021). [CrossRef]
- Rabizadeh, F., Mirian, M. S., Doosti, R., Kiani-Anbouhi, R., & Eftekhari, E. (2022). Phytochemical Classification of Medicinal Plants Used in the Treatment of Kidney Disease Based on Traditional Persian Medicine. Evidence-Based Complementary and Alternative Medicine, 2022. [CrossRef]
- Rauf, A., Abu-Izneid, T., Khalil, A. A., Imran, M., Shah, Z. A., Bin Emran, T., Mitra, S., Khan, Z., Alhumaydhi, F. A., Aljohani, A. S. M., Khan, I., Rahman, M. M., Jeandet, P., & Gondal, T. A. (2021). Berberine as a potential anticancer agent: A comprehensive review. Molecules, 26(23), 1–19. [CrossRef]
- Samadi, P., Sarvarian, P., Gholipour, E., Asenjan, K. S., Aghebati-Maleki, L., Motavalli, R., Hojjat-Farsangi, M., & Yousefi, M. (2020). Berberine: A novel therapeutic strategy for cancer. IUBMB Life, 72(10), 2065–2079. [CrossRef]
- Siracusa, L., & Napoli, E. (2022). Novel Chemical and Biological Insights of Inositol Derivatives in Mediterranean Plants. Molecules, 27(5), 1525. [CrossRef]
- Sochor, J., Zitka, O., Skutkova, H., Pavlik, D., Babula, P., Krska, B., Horna, A., Adam, V., Provaznik, I., & Kizek, R. (2010). Content of phenolic compounds and antioxidant capacity in fruits of apricot genotypes. Molecules, 15(9), 6285–6305. [CrossRef]
- Tan, A. C., Konczak, I., Ramzan, I., & Sze, D. M. Y. (2011). Native Australian fruit polyphenols inhibit cell viability and induce apoptosis in human cancer cell lines. Nutrition and Cancer, 63(3), 444–455. [CrossRef]
- Tan, A. C., Konczak, I., Ramzan, I., Zabaras, D., & Sze, D. M. Y. (2011). Potential antioxidant, antiinflammatory, and proapoptotic anticancer activities of Kakadu plum and Illawarra plum polyphenolic fractions. Nutrition and Cancer, 63(7), 1074–1084. [CrossRef]
- Widyawati, T., Yusoff, N. A., Bello, I., Asmawi, M. Z., & Ahmad, M. (2022). Bioactivity-Guided Fractionation and Identification of Antidiabetic Compound of Syzygium polyanthum (Wight.)’s Leaf Extract in Streptozotocin-Induced Diabetic Rat Model. Molecules, 27(20). [CrossRef]
- Winnett, V., Sirdaarta, J., White, A., Clarke, F. M., & Cock, I. E. (2017). Inhibition of Klebsiella pneumoniae growth by selected Australian plants: natural approaches for the prevention and management of ankylosing spondylitis. Inflammopharmacology, 25(2), 223–235. [CrossRef]
- Zanatta, A. C., Vilegas, W., & Edrada-Ebel, R. (2021). UHPLC-(ESI)-HRMS and NMR-Based Metabolomics Approach to Access the Seasonality of Byrsonima intermedia and Serjania marginata From Brazilian Cerrado Flora Diversity. Frontiers in Chemistry, 9, 710025. [CrossRef]
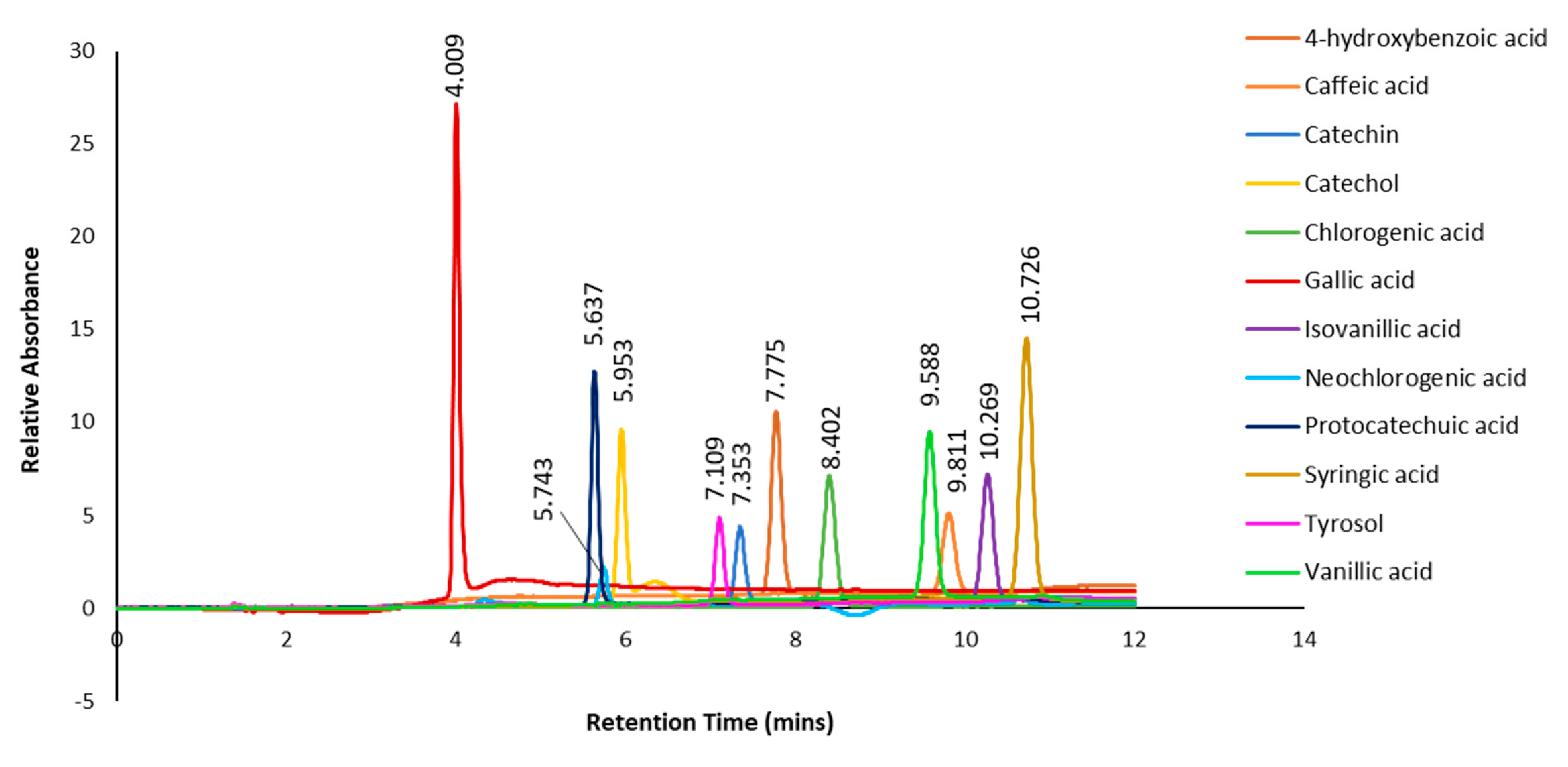
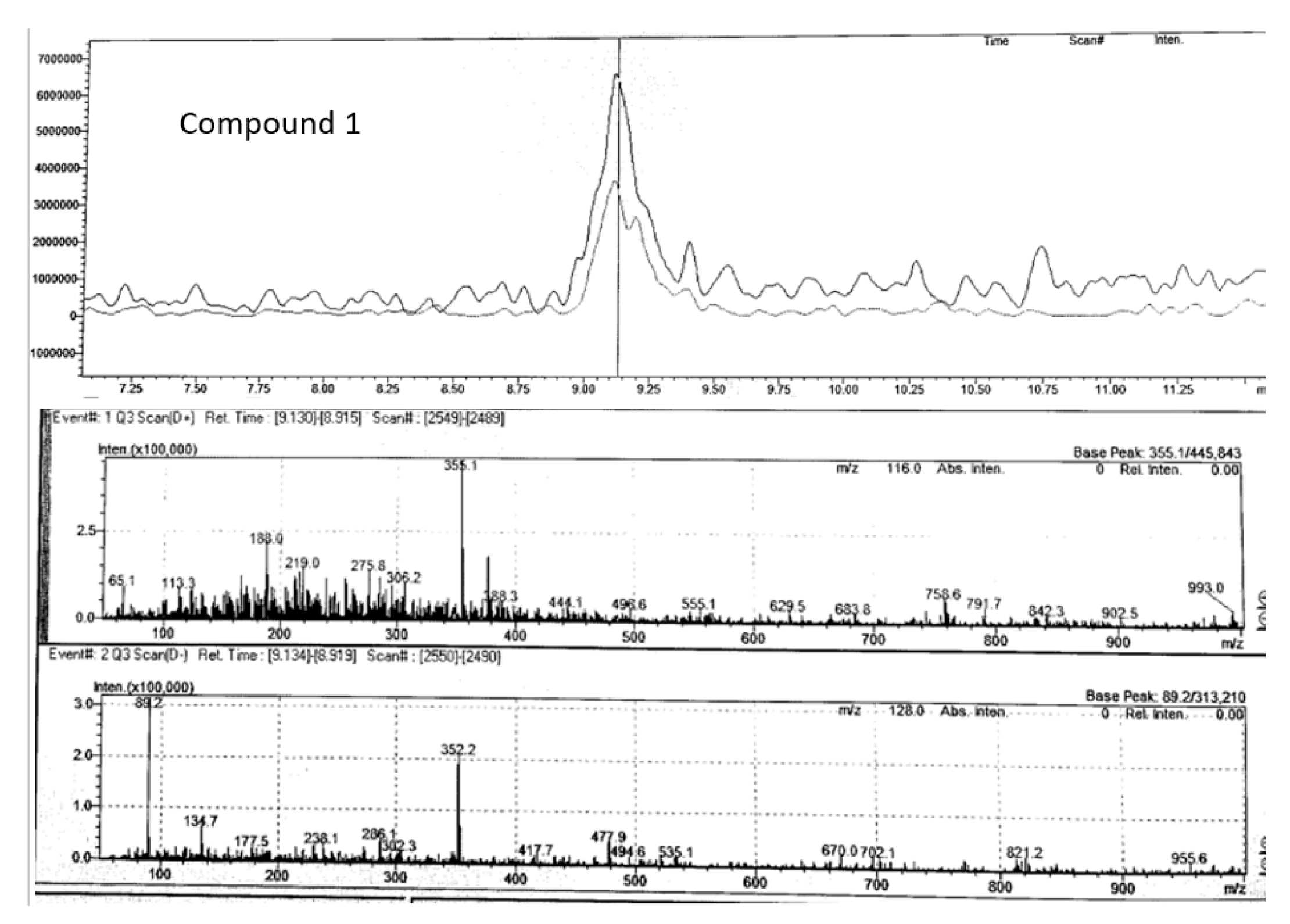
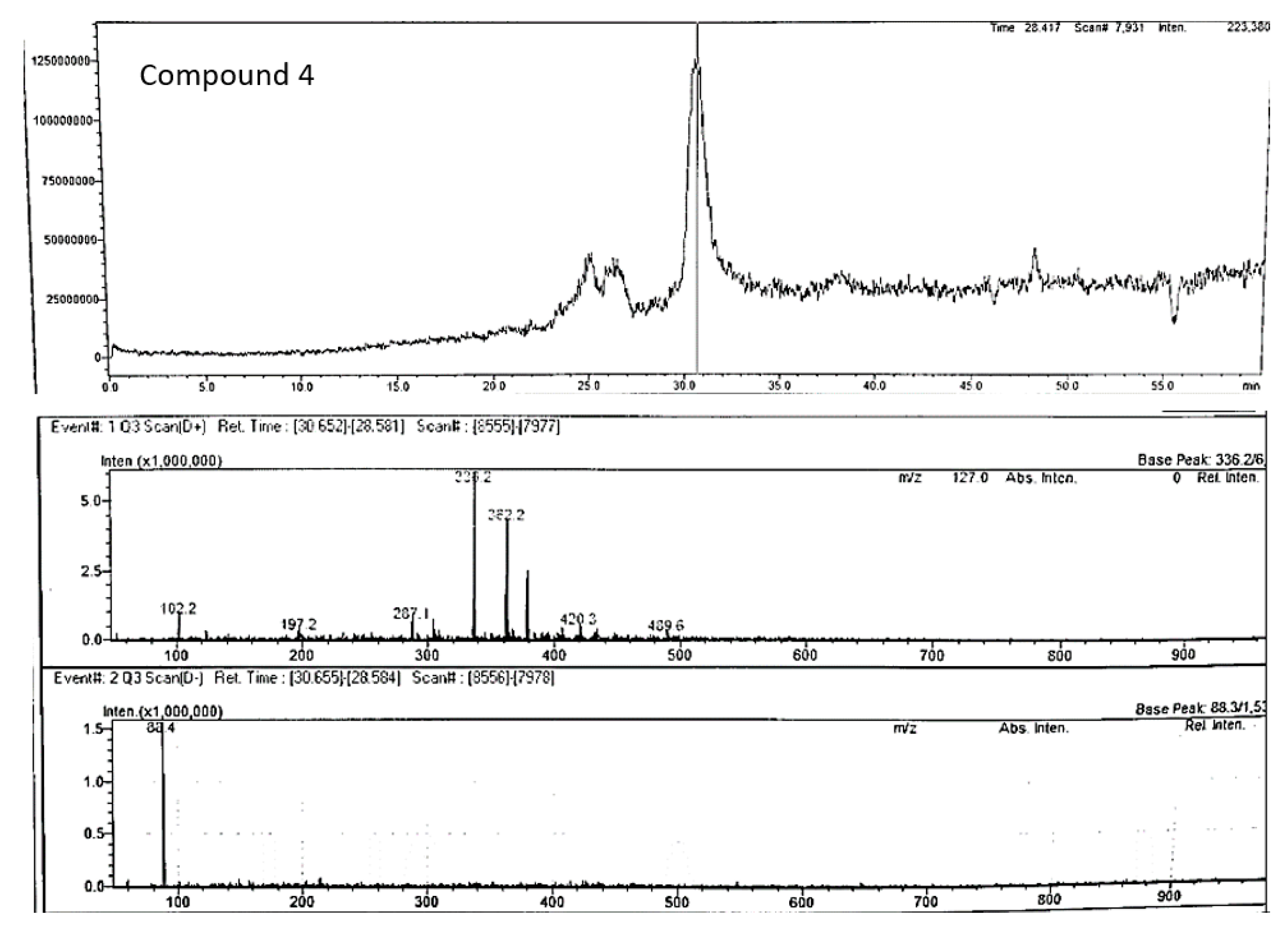
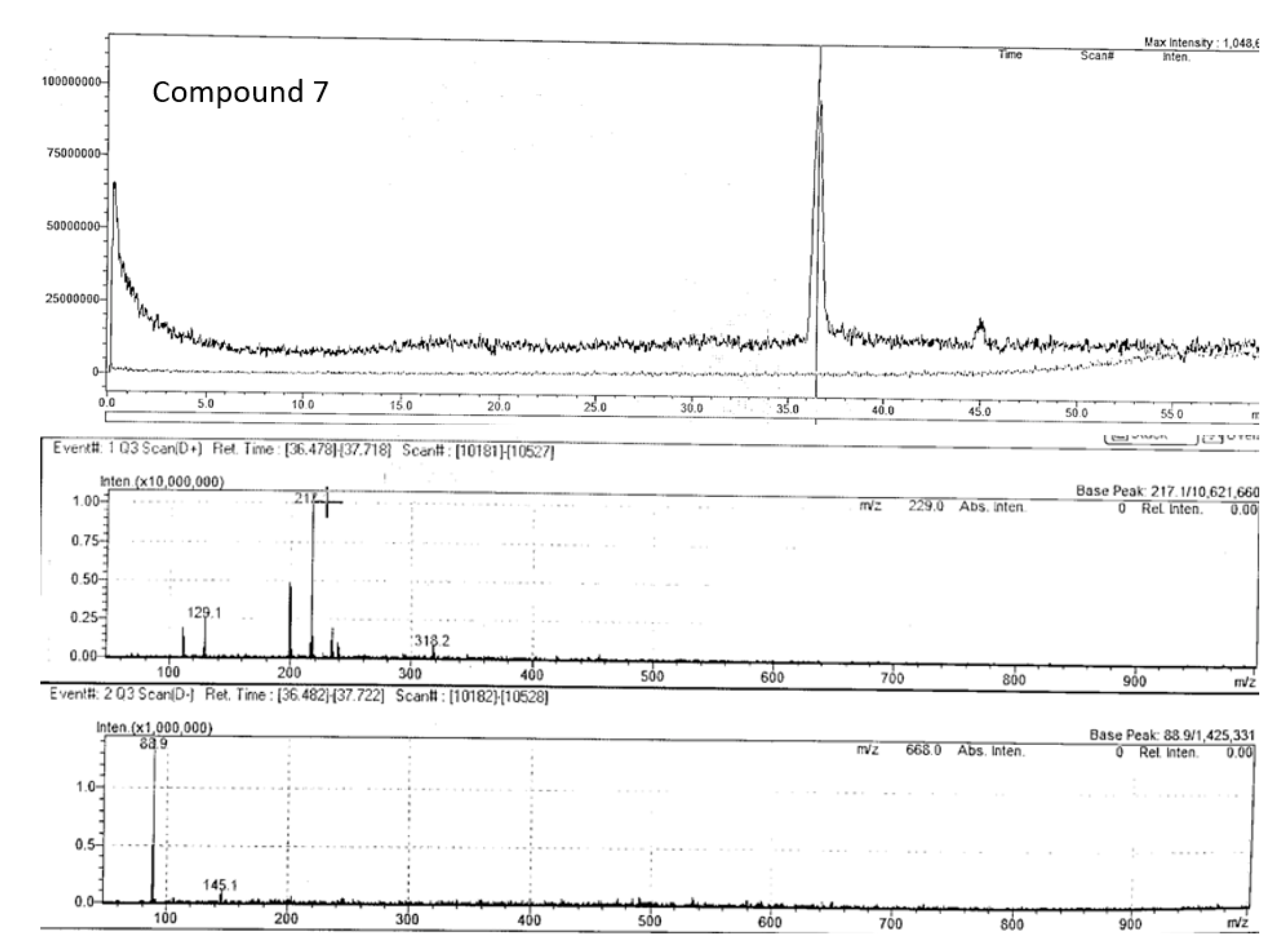
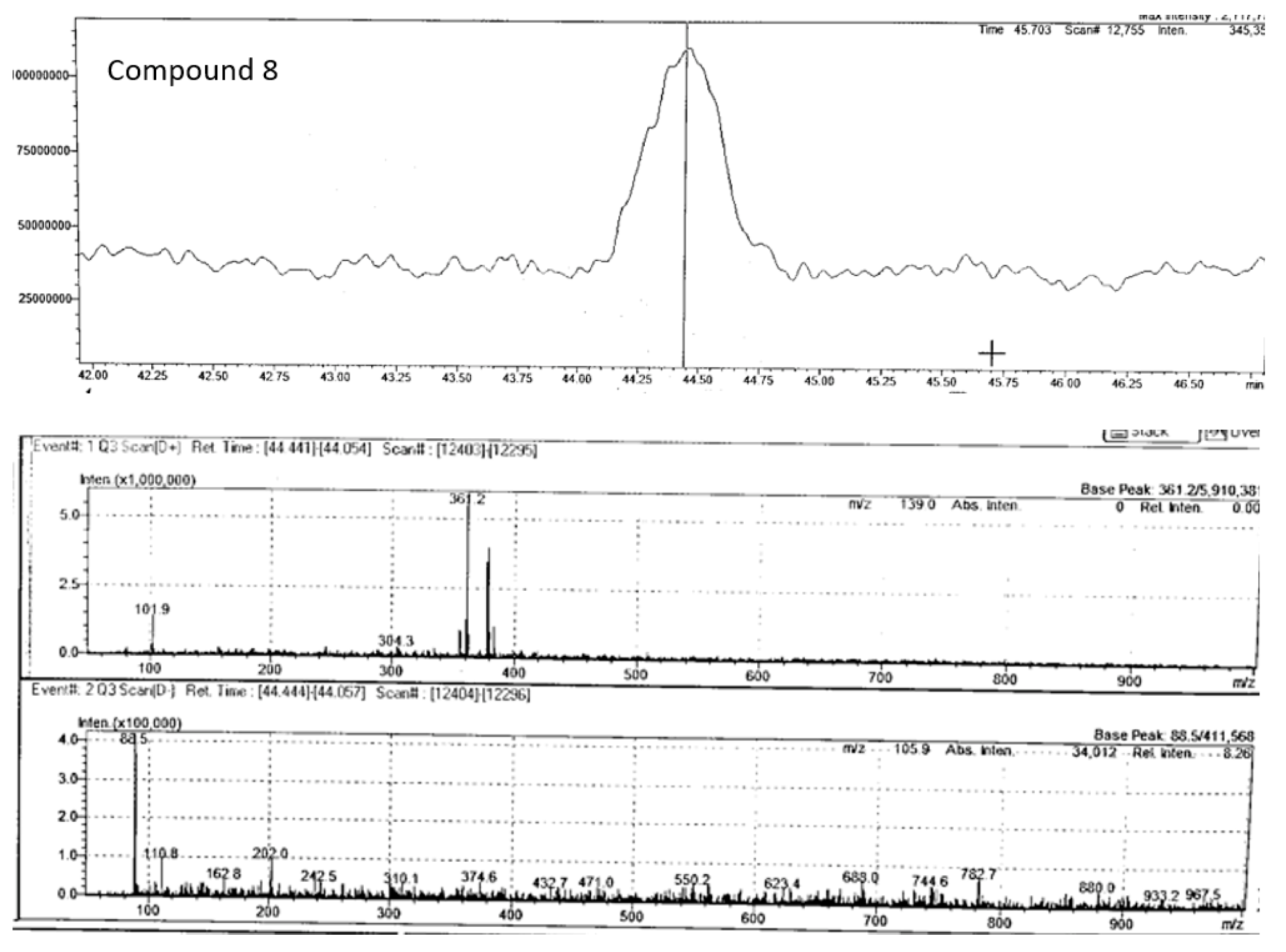
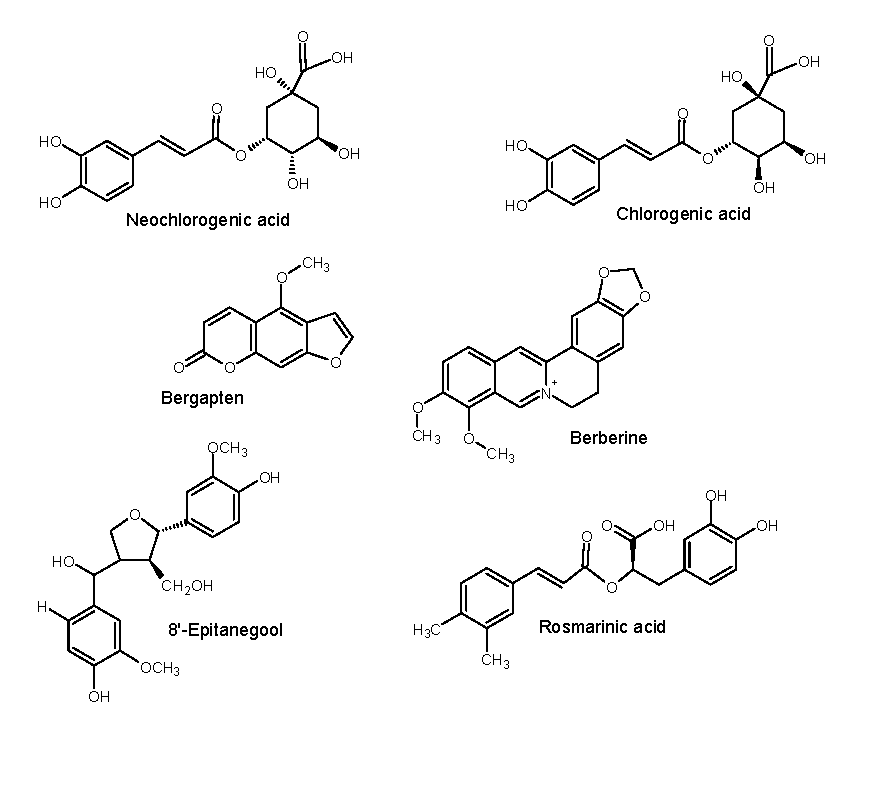
| Peak no. | Proposed compound | Molecular formula | RT (min) | Mode of ionisation | Molecular Weight (g/mol) | Observed Precursor mass (m/z) | Theoretical mass (m/z) | Product ions (MS/MS) | Literature |
|---|---|---|---|---|---|---|---|---|---|
| 1 | Chlorogenic acid | C16H18O9 | 9.13 | positive | 354.31 | 355.10 | 355.00* | 65.0, 188.0, 219.0, 275.8 | |
| 1 | Neochlorogenic acid | C16H18O9 | 9.13 | positive | 354.31 | 354.31 | 355.00 | 65.0, 188.0, 219.0, 275.8 | Xiao et al 2016 |
| 2 | Unidentified | Unidentified | 23.29 | negative | Unidentified | 229.10 | Unidentified | 157.1, 102.2 | |
| 3 | Unidentified | Unidentified | 29.15 | positive | Unidentified | 313.10 | Unidentified | 223.1, 158.2, 102.2 | |
| 4 | Berberine | C20H18NO4+ | 30.65 | positive | 336.4 | 336.20 | 336.12** | 287.2 | Jiao et al 2018 |
| 5 | Unidentified | Unidentified | 33.09 | positive | Unidentified | 378.2 | Unidentified | 102.1, 249.2 | |
| 6 | Unidentified | Unidentified | 34.11 | positive | Unidentified | 326.9 | Unidentified | 102.3, 185.1, 228.3 | |
| 7 | Bergapten | C12H8O4 | 36.48 | positive | 216.042 | 217.10 | 217.05* | 129.0, 202.0 | |
| 8 | Rosmarinic acid | C18H16O8 | 44.44 | positive | 360.3 | 361.20 | 361.09** | 181.05, 139.04 | |
| 9 | 8'-epitanegool | C20H24O7Na | 45.07 | positive | 399.39 | 399.2 | 399.14 | 287.3, 304.2 | Jiao et al 2018 |
| 10 | Unidentified | Unidentified | 46.07 | positive | Unidentified | 403.2 | Unidentified | 102.2, 329.2, 361 |
| Target | Fraction 1 | Sub-fraction 1 | Subfraction 2 | Subfraction 3 | Compound Class | |
|---|---|---|---|---|---|---|
| 1 | Citric acid-4TMS | 2971610 | 87281 | 124060 | 23465 | Carboxylic acids |
| 2 | Glucose-meto-5TMS(1) | 2162365 | 230045 | 0 | 163277 | Carbohydrate |
| 3 | Inositol-6TMS(2) | 1194216 | 141218 | 1123 | 10389 | Carbocyclic sugar |
| 4 | 2-Aminopimelic acid-3TMS | 1188393 | 35263 | 46722 | 10226 | Amino acid |
| 5 | Hydroxylamine-3TMS | 609044 | 0 | 574334 | 0 | Hydroxylamine |
| 6 | Glucose-meto-5TMS(2) | 423257 | 49736 | 3456 | 34808 | Carbohydrate |
| 7 | Galactose-meto-5TMS(2) | 422411 | 49066 | 3587 | 38804 | Carbohydrate |
| 8 | 1,5-13C2-Citric acid | 412327 | 13144 | 16485 | 3281 | Carboxylic acids |
| 9 | Lactic acid-2TMS | 268289 | 437458 | 247071 | 444661 | Carboxylic acids |
| 10 | 1,6-Anhydroglucose-3TMS | 228502 | 14470 | 1080 | 4143 | Carbohydrate |
| 11 | Alanine-2TMS | 225866 | 32076 | 8606 | 14237 | Amino acid |
| 12 | Xylose-meto-4TMS(1) | 178497 | 31305 | 4775 | 22749 | Carbohydrates |
| 13 | 3-Aminoglutaric acid-2TMS | 169217 | 26046 | 4177 | 2165 | Amino acid |
| 14 | 4-Aminobutyric acid-3TMS | 169048 | 17510 | 0 | 6009 | Amino acid |
| 15 | Arabinose-meto-4TMS | 165445 | 30249 | 4666 | 19117 | Carbohydrates |
| 16 | Palmitic acid-TMS | 154588 | 129131 | 126973 | 120981 | Saturated fatty acid |
| 17 | Sarcosine-d3-2TMS | 147242 | 130615 | 123520 | 94609 | Amino acid |
| 18 | Lyxose-meto-4TMS(2) | 146571 | 23892 | 3754 | 16430 | Carbohydrates |
| 19 | Proline-2TMS | 144359 | 37171 | 3022 | 0 | Amino acid |
| 20 | Malic acid-3TMS | 135932 | 12652 | 2318 | 3148 | Carboxylic acids |
| Fractions | Crystal product obtained (mg) | Concentrations of fractions tested (mg/mL) |
|---|---|---|
| KPF1 | 38.10 | 0.095 |
| KPF 2 | 169.00 | 0.423 |
| KPF 3 | 77.40 | 0.194 |
| KPF 4 | 17.60 | 0.044 |
| Kakadu plum fractions | Concentrations µg/mL |
Bacterial strain zone of inhibition (mm) | |||
|---|---|---|---|---|---|
| Gram positive | Gram negative | ||||
| S. aureus | E. coli | P. aeruginosa | S. typhimurium | ||
| 1 | 58.1 | 4.00 ± 0.71 | 0.00 | 0.00 | 0.00 |
| 2 | 169.0 | 4.30 ± 0.35 | 3.20 ± .10 | 2.10 ± 0.20 | 2.30 ± 0.40 |
| 3 | 77.9 | 0.00 | 0.00 | 0.00 | 0.00 |
| 4 | 17.6 | 4.20 ± 0.25 | 0.00 | 0.00 | 0.00 |
| Positive control (gentamicin) | 10 | 13.67 ± 0.58 | 16.33 ± 0.58 | 13.00± 0.10 | 12.67 ± 0.58 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




