Submitted:
15 January 2024
Posted:
16 January 2024
You are already at the latest version
Abstract
Keywords:
Introduction
Methods
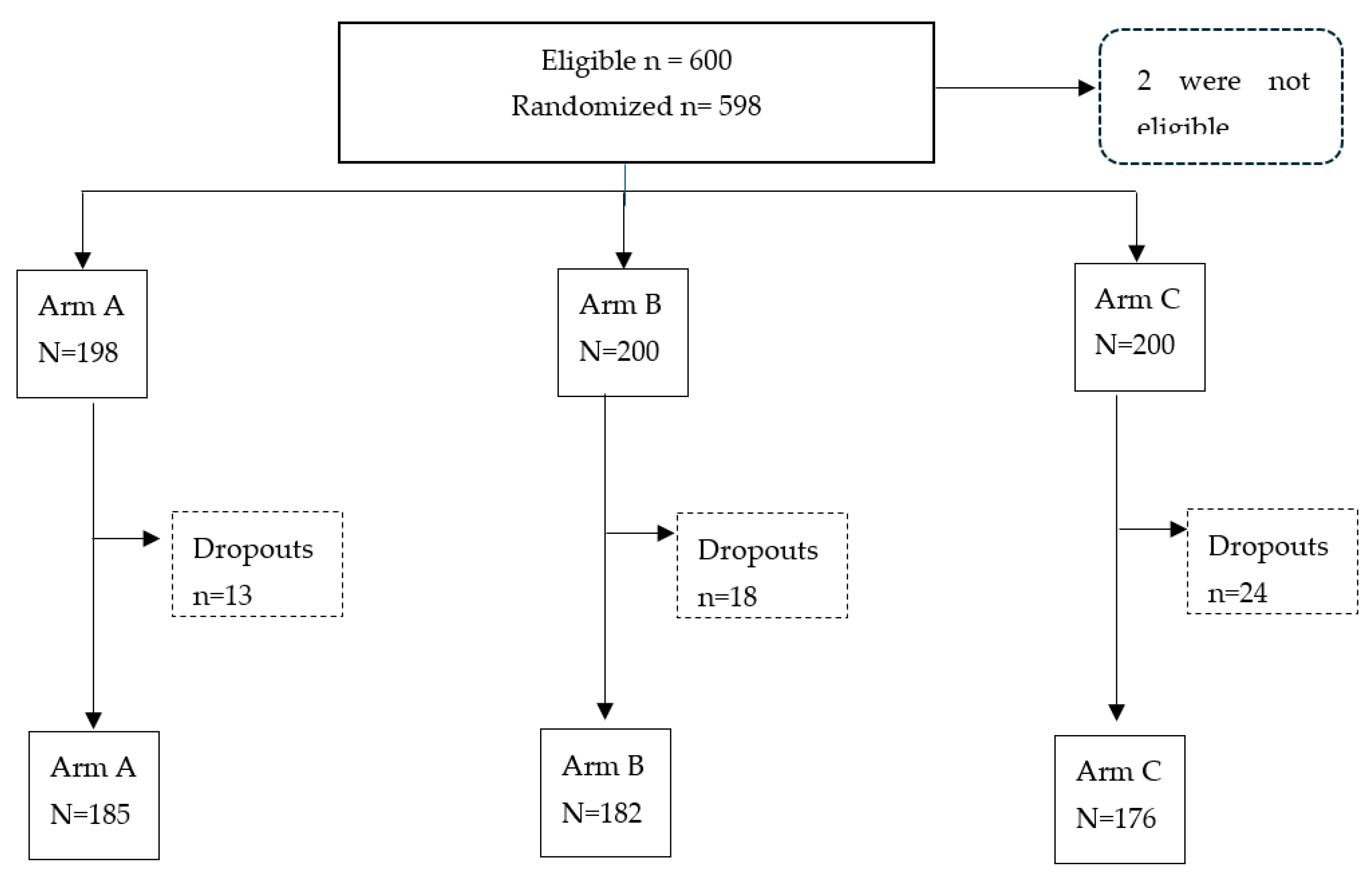
Enrolment of study Participants
Study Design
Administered Vaccines
Statistical Analysis
Results
Baseline Characteristics
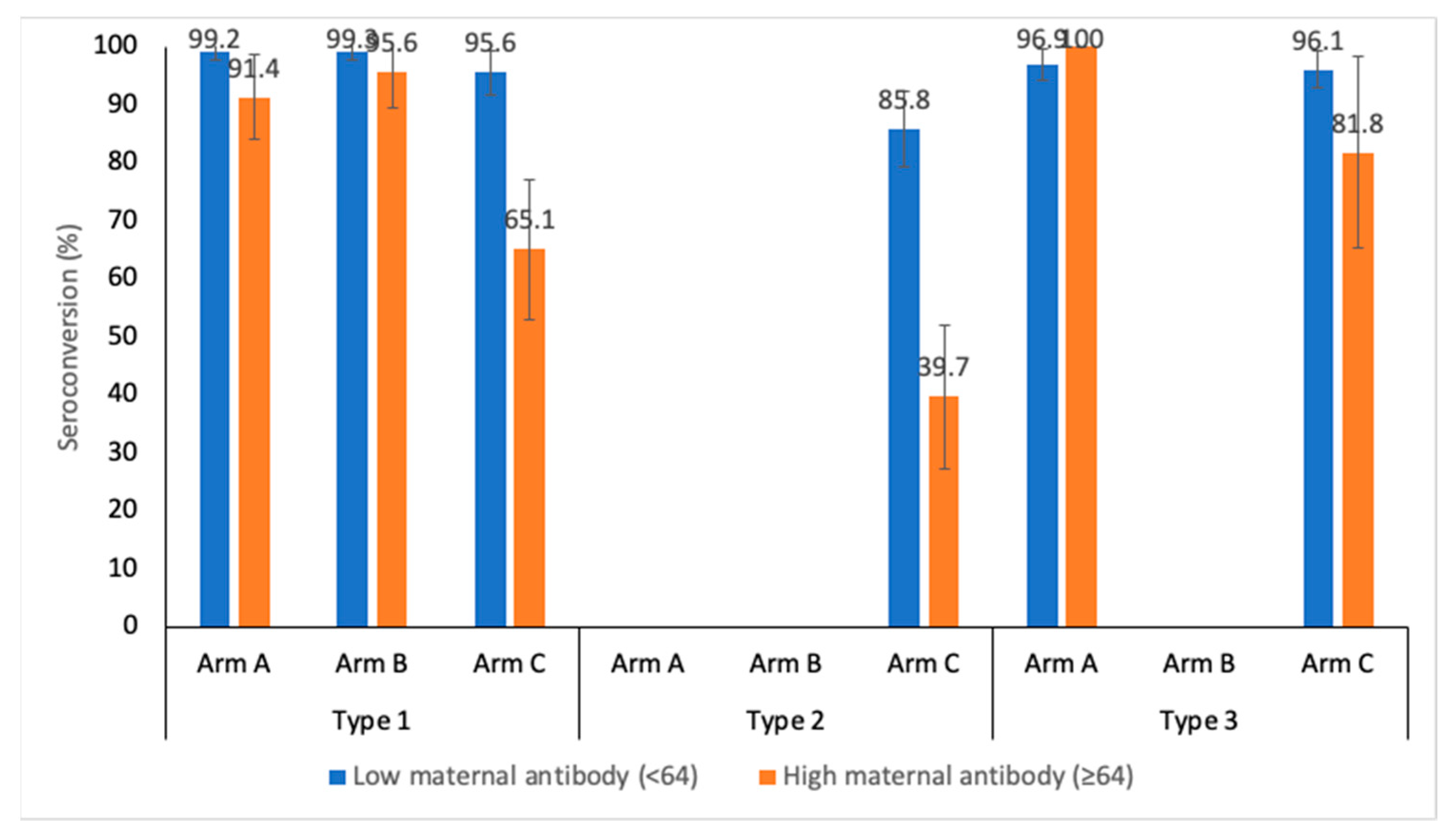
Seroconversion
Median titers
Immune response of concomitant EasyFive TT
Discussion
Immunogenicity of IPV schedule
Concomitant vaccines
Limitations
Conclusions
Funding
Acknowledgments
References
- World Health Organization. Global Polio Eradication Initiative. Semiannual report on the progress against the Polio Eradication and Endgame Strategic Plan, Geneva, Switzerland. [Internet]. 2017 [cited 2019 Jan 23]. Available from: http://polioeradication.org/wp-content/uploads/2017/12/WHO-Polio-Donor-Report-january-june-2017-web-30112017.pdf.
- GPEI-Global eradication of wild poliovirus type 2 declared [Internet]. [cited 2019 Jan 23]. Available from: http://polioeradication.org/news-post/global-eradication-of-wild-poliovirus-type-2-declared/.
- Kew, O.M.; Cochi, S.L.; Jafari, H.S.; Wassilak, S.G.F.; Mast, E.E.; Diop, O.M.; Tangermann, R.H.; Armstrong, G.L. ; Centers for Disease Control and Prevention (CDC) Possible eradication of wild poliovirus type 3--worldwide, 2012. MMWR Morb. Mortal. Wkly. Rep. 2014, 63, 1031–1033. [Google Scholar] [PubMed]
- GPEI-Endemic Countries [Internet]. [cited 2019 Jul 22]. Available from: http://polioeradication.org/where-we-work/polio-endemic-countries/.
- GPEI-Pakistan [Internet]. [cited 2023 Jul 26]. Available from: https://polioeradication.org/where-we-work/pakistan/.
- GPEI-Afghanistan [Internet]. [cited 2023 Jul 26]. Available from: https://polioeradication.org/where-we-work/afghanistan/.
- Ekwebelem, O.C.; Nnorom-Dike, O.V.; Aborode, A.T.; Ekwebelem, N.C.; Aleke, J.C.; Ofielu, E.S. Eradication of wild poliovirus in Nigeria: Lessons learnt. Public Health Pract. 2021, 2, 100144. [Google Scholar] [CrossRef]
- GPEI-Mozambique [Internet]. [cited 2023 Jul 17]. Available from: https://polioeradication.org/where-we-work/mozambique/.
- GPEI-GPEI Strategy 2022-2026 [Internet]. [cited 2023 Jul 17]. Available from: https://polioeradication.org/gpei-strategy-2022-2026/.
- Macklin, G.R.; O’Reilly, K.M.; Grassly, N.C.; Edmunds, W.J.; Mach, O.; Santhana Gopala Krishnan, R.; Voorman, A.; Vertefeuille, J.F.; Abdelwahab, J.; Gumede, N.; et al. Evolving epidemiology of poliovirus serotype 2 following withdrawal of the serotype 2 oral poliovirus vaccine. Science 2020, 368, 401–405. [Google Scholar] [CrossRef] [PubMed]
- GPEI-Polio Eradication and Endgame Strategic Plan 2013–2018 [Internet]. [cited 2023 Jul 17]. Available from: https://polioeradication.org/who-we-are/strategic-plan-2013-2018/.
- Garon, J.; Seib, K.; Orenstein, W.A.; Ramirez Gonzalez, A.; Chang Blanc, D.; Zaffran, M.; Patel, M. Polio endgame: the global switch from tOPV to bOPV. Expert Rev. Vaccines 2016, 15, 693–708. [Google Scholar] [CrossRef] [PubMed]
- GPEI-Global synchronisation and the switch [Internet]. [cited 2019 Jan 23]. Available from: http://polioeradication.org/news-post/global-synchronisation-and-the-switch/.
- Estívariz, C.F.; Jafari, H.; Sutter, R.W.; John, T.J.; Jain, V.; Agarwal, A.; Verma, H.; Pallansch, M.A.; Singh, A.P.; Guirguis, S.; et al. Immunogenicity of supplemental doses of poliovirus vaccine for children aged 6-9 months in Moradabad, India: a community-based, randomised controlled trial. Lancet Infect. Dis. 2012, 12, 128–135. [Google Scholar] [CrossRef] [PubMed]
- Sutter, R.W.; Bahl, S.; Deshpande, J.M.; Verma, H.; Ahmad, M.; Venugopal, P.; Rao, J.V.; Agarkhedkar, S.; Lalwani, S.K.; Kunwar, A.; et al. Immunogenicity of a new routine vaccination schedule for global poliomyelitis prevention: an open-label, randomised controlled trial. Lancet 2015, 386, 2413–2421. [Google Scholar] [CrossRef] [PubMed]
- John, T.J.; Vashishtha, V.M. Eradicating poliomyelitis: India’s journey from hyperendemic to polio-free status. Indian J. Med. Res. 2013, 137, 881–894. [Google Scholar] [PubMed]
- GPEI-Afghanistan [Internet]. [cited 2019 Feb 8]. Available from: http://polioeradication.org/where-we-work/afghanistan/.
- GPEI-Pakistan [Internet]. [cited 2019 Feb 8]. Available from: http://polioeradication.org/where-we-work/pakistan/.
- SAS Institute Inc. 2014. SAS 9.4 Language Reference: Concepts, Third Edition. Cary, NC: SAS Institute Inc. ISBN 978-I-62959-308-1.
- Weldon, W.C.; Oberste, M.S.; Pallansch, M.A. Standardized Methods for Detection of Poliovirus Antibodies. Methods Mol. Biol. Clifton NJ 2016, 1387, 145–176. [Google Scholar] [CrossRef]
- Cáceres, V.M.; Sutter, R.W. Sabin monovalent oral polio vaccines: review of past experiences and their potential use after polio eradication. Clin. Infect. Dis. Off. Publ. Infect. Dis. Soc. Am. 2001, 33, 531–541. [Google Scholar] [CrossRef]
- John, T.J.; Jain, H.; Ravishankar, K.; Amaresh, A.; Verma, H.; Deshpande, J.; Pallansch, M.A.; Singh, A.P.; Sreevatsava, M.; Burton, A.; et al. Monovalent type 1 oral poliovirus vaccine among infants in India: report of two randomized double-blind controlled clinical trials. Vaccine 2011, 29, 5793–5801. [Google Scholar] [CrossRef] [PubMed]
- Mir, F.; Quadri, F.; Mach, O.; Ahmed, I.; Bhatti, Z.; Khan, A.; Rehman, N. ur; Durry, E.; Salama, M.; Oberste, S.M.; et al. Monovalent type-1 oral poliovirus vaccine given at short intervals in Pakistan: a randomised controlled, four-arm, open-label, non-inferiority trial. Lancet Infect. Dis. 2015, 15, 889–897. [Google Scholar] [CrossRef] [PubMed]
- Huyen, D.T.T.; Anh, D.D.; Trung, N.T.; Hong, D.T.; Thanh, T.T.; Truong, L.N.; Jeyaseelan, V.; Lopez Cavestany, R.; Hendley, W.S.; Mainou, B.A.; et al. Inactivated Poliovirus Vaccine Closing the Type 2 Immunity Gap in Vietnam. J. Pediatr. Infect. Dis. Soc. 2022, 11, 413–416. [Google Scholar] [CrossRef]
- Tagbo, B.N.; Verma, H.; Mahmud, Z.M.; Ernest, K.; Nnani, R.O.; Chukwubike, C.; Craig, K.T.; Hamisu, A.; Weldon, W.C.; Oberste, S.M.; et al. Randomized Controlled Clinical Trial of Bivalent Oral Poliovirus Vaccine and Inactivated Poliovirus Vaccine in Nigerian Children. J. Infect. Dis. 2022, 226, 299–307. [Google Scholar] [CrossRef] [PubMed]
- Bravo, L.C.; Carlos, J.C.; Gatchalian, S.R.; Montellano, M.E.B.; Tabora, C.F.C.B.; Thierry-Carstensen, B.; Tingskov, P.N.; Sørensen, C.; Wachmann, H.; Bandyopadhyay, A.S.; et al. Immunogenicity and safety of an adjuvanted inactivated polio vaccine, IPV-Al, compared to standard IPV: A phase 3 observer-blinded, randomised, controlled trial in infants vaccinated at 6, 10, 14 weeks and 9 months of age. Vaccine 2020, 38, 530–538. [Google Scholar] [CrossRef] [PubMed]
- WHO Collaborative Study. Combined immunization of infants with oral and inactivated poliovirus vaccines: results of a randomized trial in The Gambia, Oman, and Thailand. WHO Collaborative Study Group on Oral and Inactivated Poliovirus Vaccines. Bull. World Health Organ. 1996, 74, 253–268. [Google Scholar]
- Cohen-Abbo, A.; Culley, B.S.; Reed, G.W.; Sannella, E.C.; Mace, R.L.; Robertson, S.E.; Wright, P.F. Seroresponse to trivalent oral poliovirus vaccine as a function of dosage interval. Pediatr. Infect. Dis. J. 1995, 14, 100–106. [Google Scholar] [CrossRef] [PubMed]
| Study Arm→ | Arm A (bOPV+IPV) |
Arm B (mOPV1+IPV) |
Arm C (IPV only) |
|
|---|---|---|---|---|
| Study Visit Schedule↓ | Study Vaccines & Samples↓ | |||
| Birth | Sample | Cord blood | Cord blood | Cord blood |
| Vaccine | bOPV | mOPV1 | __ | |
| 6* weeks | Sample | __ | __ | __ |
| Vaccine | bOPV | mOPV1 | IPV | |
| 10* weeks | Sample | __ | __ | __ |
| Vaccine | bOPV | mOPV1 | IPV | |
| 14* weeks | Sample | Blood | Blood | Blood |
| Vaccine | bOPV+IPV | mOPV1+IPV | IPV | |
| 18 weeks | Sample | Blood | Blood | Blood |
| Vaccine | IPV | IPV | IPV | |
| 22 weeks | Sample | Blood | Blood | Blood |
| Characteristics | Arm A N=198 |
Arm B N=200 |
Arm C N=200 |
|||||
|---|---|---|---|---|---|---|---|---|
| n | % | n | % | n | % | |||
| Female Child vs Male Child | 104 | 52.5 | 87 | 43.5 | 93 | 46.7 | ||
| Hindu Religion vs Other religion | 181 | 91.4 | 175 | 87.5 | 176 | 88.0 | ||
|
Father’s education Illiterate Primary/Middle High school/ Higher secondary Graduate/Higher |
8 62 84 44 |
4.0 31.3 42.4 22.2 |
12 55 89 41 |
6.1 27.9 45.2 20.8 |
10 53 86 50 |
5.0 26.6 43.2 25.1 |
||
| Normal weeks of Gestational vs not normal | 198 | 100.0 | 200 | 100.0 | 200 | 100.0 | ||
| Normal Apgar score vs abnormal Apgar score | 198 | 100.0 | 200 | 100.0 | 200 | 100.0 | ||
|
Baseline Seroprevalence Type 1 Type 2 Type 3 |
172 182 130 |
86.9 91.9 65.7 |
167 168 126 |
83.5 84.0 63.0 |
174 174 123 |
87.0 87.0 61.5 |
||
|
Baseline Titers Type 1 Median (95% CI) Type 2 Median (95% CI) Type 3 Median (95% CI) |
29 (23-45) 36 (36-45) 14 (11-18) |
29 (23-36) 36 (29-45) 11 (10-14) |
29 (23-36) 45 (36-45) 11 (9-14) |
|||||
| Arm A (N=185) | Arm B (N=182) | Arm C (N=176) | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| n | % | 95% CI | n | % | 95% CI | n | % | 95% CI | ||
|
14 weeks Type 1 |
179 |
96.8 |
93.1-98.5 |
179 |
98.4 |
95-3-99.4 |
149 |
84.7 |
78.6-89.4 |
|
| Type 2 | 40 | 21.6 | 16.3-28.1 | 40 | 22.0 | 16.6-28.5 | 122 | 69.3 | 62.1-75.6 | |
| Type 3 | 180 | 97.3 | 93.8-98.8 | 43 | 23.6 | 18.0-30.3 | 166 | 94.3 | 89.9-96.9 | |
|
18 weeks Type 1 |
184 |
99.5 |
97.0-99.9 |
182 |
100.0 |
97.9-100.0 |
169 |
96.0 |
92.0-98.1 |
|
| Type 2 | 148 | 80.0 | 73.7-85.1 | 140 | 76.9 | 70.3-82.4 | 164 | 93.2 | 88.5-96.1 | |
| Type 3 | 185 | 100.0 | 98.0-100.0 | 149 | 81.9 | 75.6-86.8 | 175 | 99.4 | 96.9-99.9 | |
|
22 weeks Type 1 |
185 |
100.0 |
98.0-100.0 |
182 |
100.0 |
97.9-100.0 |
171 |
97.2 |
93.5-98.9 |
|
| Type 2 | 181 | 97.8 | 94.6-99.2 | 176 | 96.7 | 93.0-98.5 | 170 | 96.6 | 92.8-98.4 | |
| Type 3 | 185 | 100.0 | 98.0-100.0 | 180 | 98.9 | 96.1-99.7 | 176 | 100.0 | 97.9-100.0 | |
| Type 1 | Type 2 | Type 3 | ||||||||
| A vs B | A vs C | B vs C | A vs B | A vs C | B vs C | A vs B | A vs C | B vs C | ||
| 14 weeks | 0.323 | <0.001 | <0.001 | 0.162 | ||||||
| 18 weeks | 0.824 | 0.024 | 0.006 | 0.522 | <0.001 | <0.001 | <0.001 | 0.891 | <0.001 | |
| 22 weeks | 0.022 | 0.023 | 0.525 | 0.495 | 0.959 | 0.153 | 0.729 | |||
| Titres | Arm A | Arm B | Arm C | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Median | 95% CI | Median | 95% CI | Median | 95% CI | |||||||
|
14 weeks Type 1 Type 2 Type 3 |
724 7 287 |
(574-818) (6-7) (228-362) |
724 6 6 |
(724-724) (6-7) (6-6) |
91 45 228 |
(91-114) (36-57) (181-287) |
||||||
|
18 weeks Type 1 Type 2 Type 3 |
1149 18 724 |
(912-≥1448) (16-23) (574-781) |
1149 23 36 |
(1149-1152) (18-29) (23-57) |
144 72 287 |
(114-181) (57-91) (228-362) |
||||||
|
22 weeks Type 1 Type 2 Type 3 |
912 57 724 |
(724-1149) (51-72) (724-724) |
1149 72 287 |
(912-1176) (57-72) (228-362) |
228 114 362 |
(181-269) (91-144) (287-410) |
||||||
| Type 1 | Type 2 | Type 3 | ||||||||||
| A vs B | A vs C | B vs C | A vs B | A vs C | B vs C | A vs B | A vs C | B vs C | ||||
| 14 weeks | 0.203 | <0.001 | <0.001 | 0.654 | <0.001 | <0.001 | <0.001 | 0.006 | <0.001 | |||
| 18 weeks | 1.000 | <0.001 | <0.001 | 0.778 | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 | |||
| 22 weeks | 0.084 | <0.001 | <0.001 | 0.876 | <0.001 | <0.001 | <0.001 | <0.001 | 0.103 | |||
| Antibody | GMT | |
|---|---|---|
| Pre-vaccination | Post vaccination | |
| Anti-diphtheria (IU/ml) | 0.093 ( 0.086-0.102) | 0.471 (0.435-0.513) |
| Anti-tetanus (IU/ml) | 1.58 (1.123-2.051) | 2.184 ( 1.87-2.55) |
| Anti-PRP (lg/ml) | 0.64 (0.592-0.690) | 2.91 ( 2.59-3.26) |
| Anti-HBs Ag (mIU/ml) | 0.00088 (0.001-0.001) | 471.31 (438.61-506.46) |
| Anti-PT (lg/ml) | 2.89 (2.30-4.06) | 16.54 (13.55-20.86) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




