Submitted:
12 January 2024
Posted:
15 January 2024
Read the latest preprint version here
Abstract
Keywords:
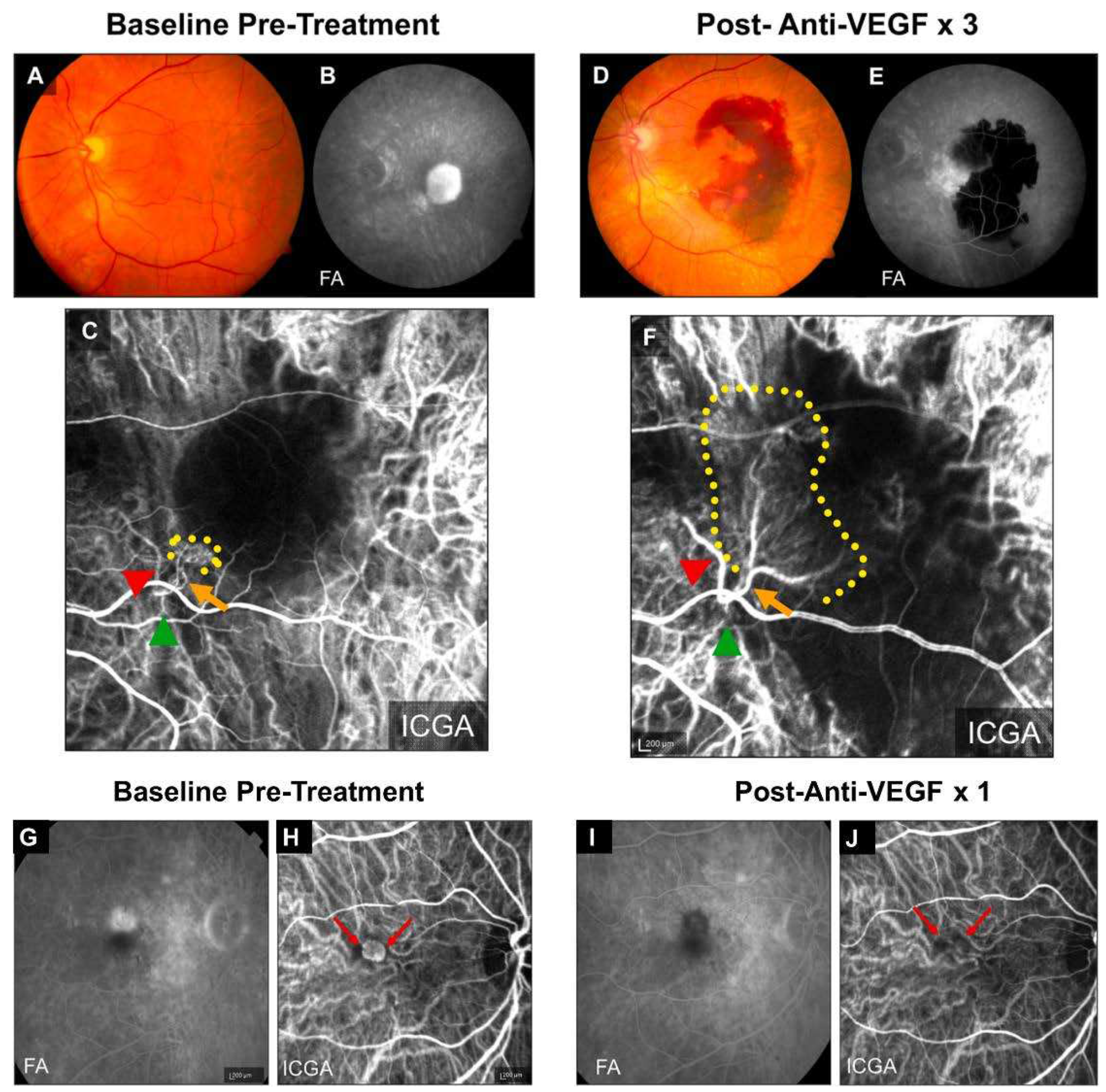
1. Limitation of Anti-VEGF Therapies
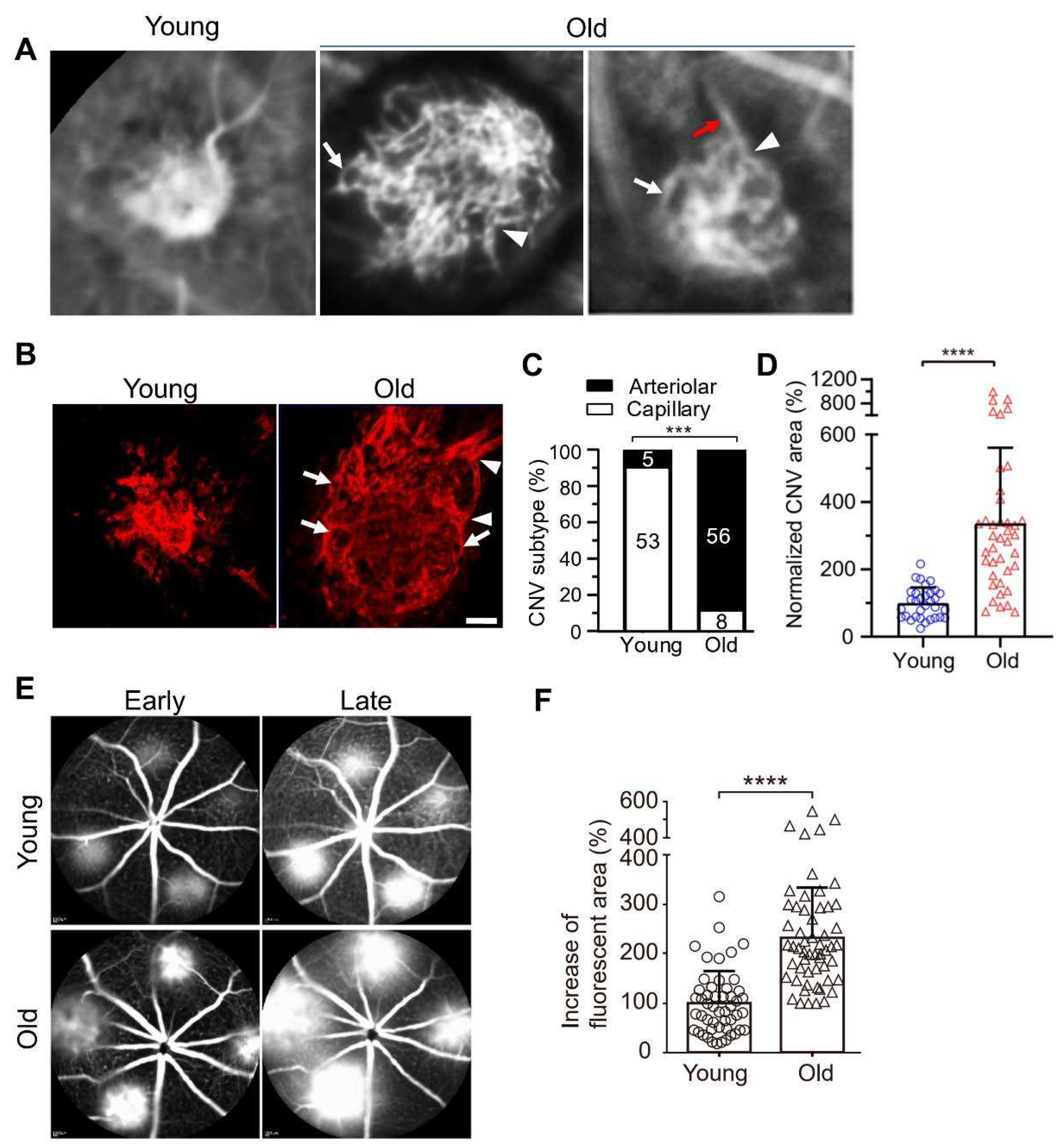
2. Animal Models of Anti-VEGF Resistance
3. Role of Macrophages in Anti-VEGF Resistance
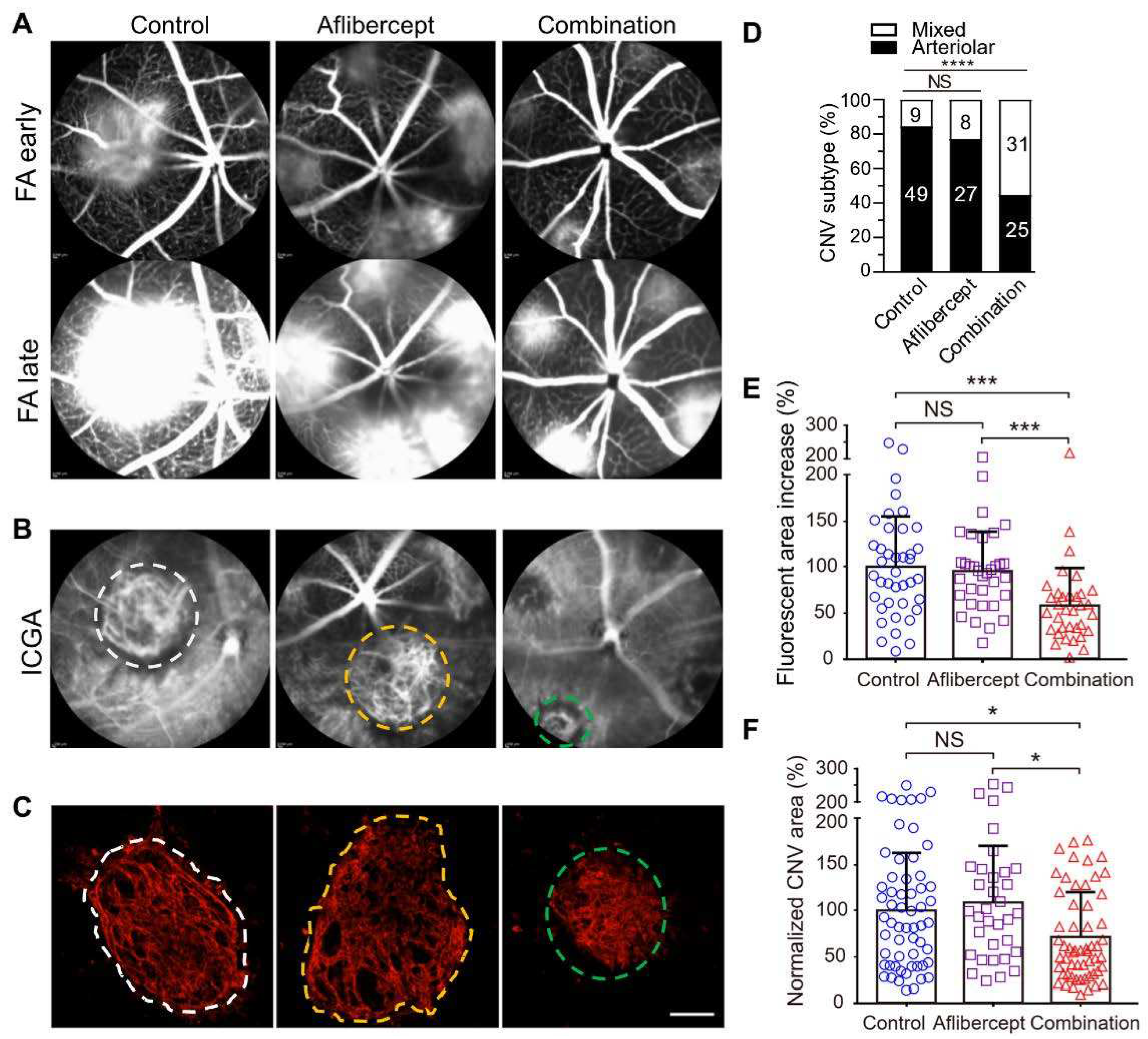
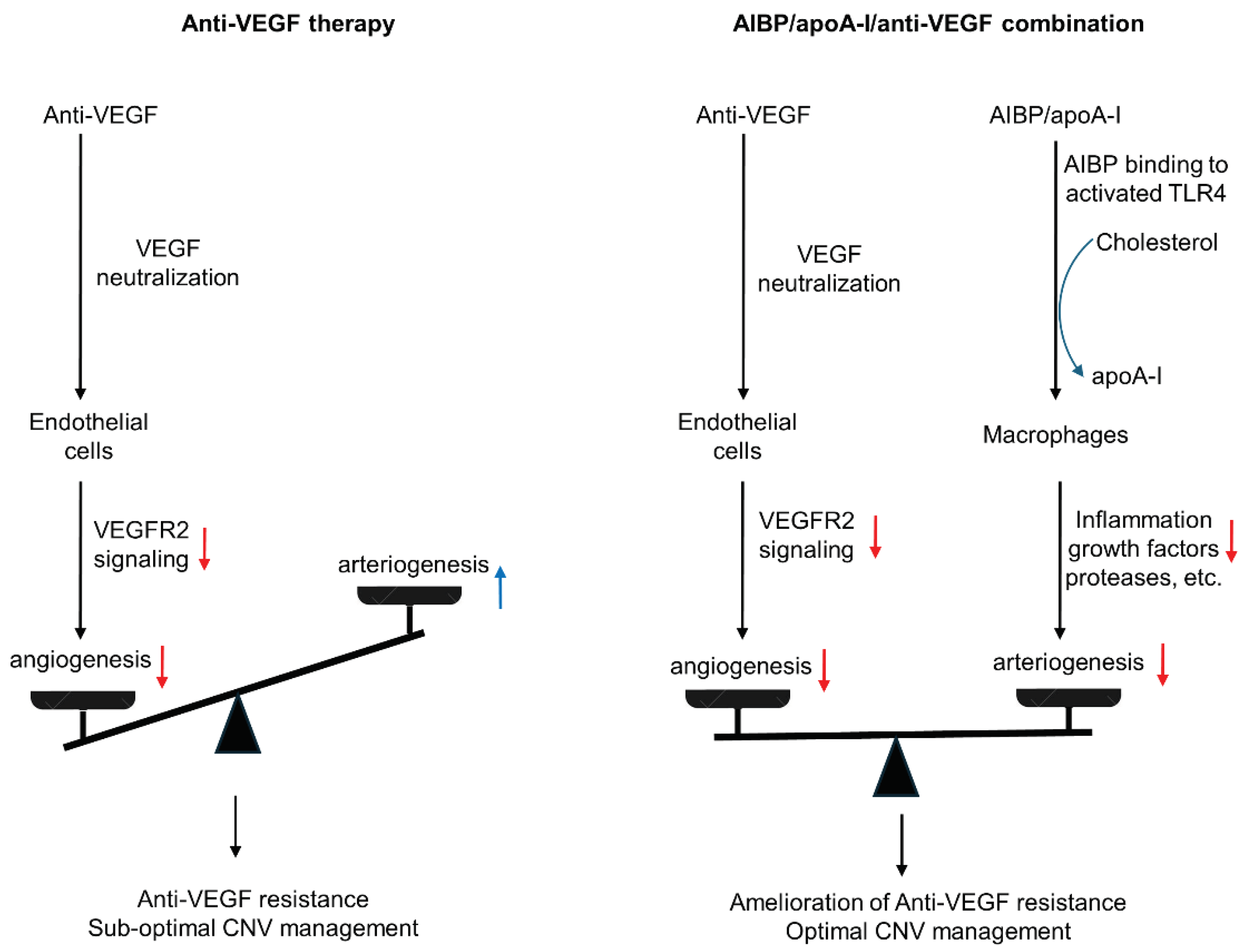
4. Treatment Strategies for Anti-VEGF Resistance by simultaneously Targeting Capillary and Arteriolar CNV
5. How Does the Combination Therapy Compare with Anti-VEGF Gene Therapy and Higher Dose Anti-VEGF Regimen Currently in Development?
6. Perspectives
Patent
Funding
Authors contributions
Conflict of interest’s statement
References
- Wong, W. L. et al. Global prevalence of age-related macular degeneration and disease burden projection for 2020 and 2040: a systematic review and meta-analysis. Lancet Glob. Health 2, e106–e116 (2014). [CrossRef]
- Bressler, N. M., Bressler, S. B. & Fine, S. L. Age-related macular degeneration. Surv. Ophthalmol. 32, 375–413 (1988). [CrossRef]
- Comparison of Age-related Macular Degeneration Treatments Trials (CATT) Research Group et al. Five-Year Outcomes with Anti-Vascular Endothelial Growth Factor Treatment of Neovascular Age-Related Macular Degeneration: The Comparison of Age-Related Macular Degeneration Treatments Trials. Ophthalmology 123, 1751–1761 (2016). [CrossRef]
- Yang, S., Zhao, J. & Sun, X. Resistance to anti-VEGF therapy in neovascular age-related macular degeneration: a comprehensive review. Drug Des. Devel. Ther. 10, 1857–1867 (2016). [CrossRef]
- Ehlken, C. et al. Switch of anti-VEGF agents is an option for nonresponders in the treatment of AMD. Eye Lond. Engl. 28, 538–545 (2014). [CrossRef]
- Heier, J. S. et al. Intravitreal aflibercept (VEGF trap-eye) in wet age-related macular degeneration. Ophthalmology 119, 2537–2548 (2012). [CrossRef]
- Rofagha, S. et al. Seven-year outcomes in ranibizumab-treated patients in ANCHOR, MARINA, and HORIZON: a multicenter cohort study (SEVEN-UP). Ophthalmology 120, 2292–2299 (2013). [CrossRef]
- Krebs, I. et al. Non-responders to treatment with antagonists of vascular endothelial growth factor in age-related macular degeneration. Br. J. Ophthalmol. 97, 1443–1446 (2013). [CrossRef]
- Mettu, P. S., Allingham, M. J. & Cousins, S. W. Incomplete response to Anti-VEGF therapy in neovascular AMD: Exploring disease mechanisms and therapeutic opportunities. Prog. Retin. Eye Res. 100906 (2020). [CrossRef]
- Otsuji, T. et al. Initial non-responders to ranibizumab in the treatment of age-related macular degeneration (AMD). Clin. Ophthalmol. Auckl. NZ 7, 1487–1490 (2013). [CrossRef]
- Cobos, E. et al. Association between CFH, CFB, ARMS2, SERPINF1, VEGFR1 and VEGF polymorphisms and anatomical and functional response to ranibizumab treatment in neovascular age-related macular degeneration. Acta Ophthalmol. (Copenh.) 96, e201–e212 (2018). [CrossRef]
- Kitchens, J. W. et al. A pharmacogenetics study to predict outcome in patients receiving anti-VEGF therapy in age related macular degeneration. Clin. Ophthalmol. Auckl. NZ 7, 1987–1993 (2013). [CrossRef]
- Rosenfeld, P. J. et al. Characteristics of Patients Losing Vision after 2 Years of Monthly Dosing in the Phase III Ranibizumab Clinical Trials. Ophthalmology 118, 523–530 (2011). [CrossRef]
- Spaide, R. F. Optical Coherence Tomography Angiography Signs of Vascular Abnormalization With Antiangiogenic Therapy for Choroidal Neovascularization. Am. J. Ophthalmol. 160, 6–16 (2015). [CrossRef]
- Sharma, D., Zachary, I. & Jia, H. Mechanisms of Acquired Resistance to Anti-VEGF Therapy for Neovascular Eye Diseases. Invest. Ophthalmol. Vis. Sci. 64, 28 (2023). [CrossRef]
- Zuber-Laskawiec, K., Kubicka-Trzaska, A., Karska-Basta, I., Pociej-Marciak, W. & Romanowska-Dixon, B. Non-responsiveness and tachyphylaxis to anti-vascular endothelial growth factor treatment in naive patients with exudative age-related macular degeneration. J. Physiol. Pharmacol. Off. J. Pol. Physiol. Soc. 70, (2019). [CrossRef]
- Hara, C. et al. Tachyphylaxis during treatment of exudative age-related macular degeneration with aflibercept. Graefes Arch. Clin. Exp. Ophthalmol. Albrecht Von Graefes Arch. Klin. Exp. Ophthalmol. 257, 2559–2569 (2019). [CrossRef]
- Schaal, S., Kaplan, H. J. & Tezel, T. H. Is there tachyphylaxis to intravitreal anti-vascular endothelial growth factor pharmacotherapy in age-related macular degeneration? Ophthalmology 115, 2199–2205 (2008). [CrossRef]
- Forooghian, F., Cukras, C., Meyerle, C. B., Chew, E. Y. & Wong, W. T. Tachyphylaxis after intravitreal bevacizumab for exudative age-related macular degeneration. Retina Phila. Pa 29, 723–731 (2009). [CrossRef]
- Hwang, R. Y., Santos, D. & Oliver, S. C. N. RATES OF EXUDATIVE RECURRENCE FOR EYES WITH INACTIVATED WET AGE-RELATED MACULAR DEGENERATION ON 12-WEEK INTERVAL DOSING WITH BEVACIZUMAB THERAPY. Retina Phila. Pa 40, 679–685 (2020). [CrossRef]
- Kuroda, Y. et al. Factors Associated with Recurrence of Age-Related Macular Degeneration after Anti-Vascular Endothelial Growth Factor Treatment: A Retrospective Cohort Study. Ophthalmology 122, 2303–2310 (2015). [CrossRef]
- Kim, J. H., Chang, Y. S., Kim, J. W., Kim, C. G. & Lee, D. W. RECURRENCE IN PATIENTS WITH TYPE 3 NEOVASCULARIZATION (RETINAL ANGIOMATOUS PROLIFERATION) AFTER INTRAVITREAL RANIBIZUMAB. Retina Phila. Pa 37, 1508–1515 (2017). [CrossRef]
- You, Q. S. et al. HIGH-DOSE HIGH-FREQUENCY AFLIBERCEPT FOR RECALCITRANT NEOVASCULAR AGE-RELATED MACULAR DEGENERATION. Retina Phila. Pa 38, 1156–1165 (2018). [CrossRef]
- Brown, D. M., Chen, E., Mariani, A., Major, J. C., & SAVE Study Group. Super-dose anti-VEGF (SAVE) trial: 2.0 mg intravitreal ranibizumab for recalcitrant neovascular macular degeneration-primary end point. Ophthalmology 120, 349–354 (2013). [CrossRef]
- Fung, A. T. et al. Pilot study to evaluate the role of high-dose ranibizumab 2.0 mg in the management of neovascular age-related macular degeneration in patients with persistent/recurrent macular fluid <30 days following treatment with intravitreal anti-VEGF therapy (the LAST Study). Eye Lond. Engl. 26, 1181–1187 (2012). [CrossRef]
- Chang, A. A. et al. Intravitreal aflibercept for treatment-resistant neovascular age-related macular degeneration. Ophthalmology 121, 188–192 (2014). [CrossRef]
- Marquis, L.-M. & Mantel, I. Beneficial switch from aflibercept to ranibizumab for the treatment of refractory neovascular age-related macular degeneration. Graefes Arch. Clin. Exp. Ophthalmol. Albrecht Von Graefes Arch. Klin. Exp. Ophthalmol. 258, 1591–1596 (2020). [CrossRef]
- Spooner, K. et al. Long-term outcomes of switching to aflibercept for treatment-resistant neovascular age-related macular degeneration. Acta Ophthalmol. (Copenh.) 97, e706–e712 (2019). [CrossRef]
- Broadhead, G. K., Keenan, T. D. L., Chew, E. Y., Wiley, H. E. & Cukras, C. A. Comparison of agents using higher dose anti-VEGF therapy for treatment-resistant neovascular age-related macular degeneration. Graefes Arch. Clin. Exp. Ophthalmol. Albrecht Von Graefes Arch. Klin. Exp. Ophthalmol. 260, 2239–2247 (2022). [CrossRef]
- Dunn, E. N., Hariprasad, S. M. & Sheth, V. S. An Overview of the Fovista and Rinucumab Trials and the Fate of Anti-PDGF Medications. Ophthalmic Surg. Lasers Imaging Retina 48, 100–104 (2017). [CrossRef]
- Apte, R. S., Chen, D. S. & Ferrara, N. VEGF in Signaling and Disease: Beyond Discovery and Development. Cell 176, 1248–1264 (2019). [CrossRef]
- Heier, J. S. et al. Efficacy, durability, and safety of intravitreal faricimab up to every 16 weeks for neovascular age-related macular degeneration (TENAYA and LUCERNE): two randomised, double-masked, phase 3, non-inferiority trials. The Lancet 399, 729–740 (2022). [CrossRef]
- Khachigian, L. M., Liew, G., Teo, K. Y. C., Wong, T. Y. & Mitchell, P. Emerging therapeutic strategies for unmet need in neovascular age-related macular degeneration. J. Transl. Med. 21, 133 (2023). [CrossRef]
- Kaiser, P. K. et al. Ranibizumab for predominantly classic neovascular age-related macular degeneration: subgroup analysis of first-year ANCHOR results. Am. J. Ophthalmol. 144, 850–857 (2007). [CrossRef]
- Finger, R. P., Wickremasinghe, S. S., Baird, P. N. & Guymer, R. H. Predictors of anti-VEGF treatment response in neovascular age-related macular degeneration. Surv. Ophthalmol. 59, 1–18 (2014). [CrossRef]
- Boyer, D. S. et al. Subgroup analysis of the MARINA study of ranibizumab in neovascular age-related macular degeneration. Ophthalmology 114, 246–252 (2007). [CrossRef]
- Lumbroso, B. et al. Optical Coherence Tomography Angiography Study of Choroidal Neovascularization Early Response after Treatment. Dev. Ophthalmol. 56, 77–85 (2016). [CrossRef]
- Zhu, L. et al. Combination of apolipoprotein-A-I/apolipoprotein-A-I binding protein and anti-VEGF treatment overcomes anti-VEGF resistance in choroidal neovascularization in mice. Commun. Biol. 3, 386 (2020). [CrossRef]
- Zhang, Z., Shen, M. M. & Fu, Y. Combination of AIBP, apoA-I, and Aflibercept Overcomes Anti-VEGF Resistance in Neovascular AMD by Inhibiting Arteriolar Choroidal Neovascularization. Invest. Ophthalmol. Vis. Sci. 63, 2 (2022). [CrossRef]
- Attarde, A., Riad, T. S., Zhang, Z., Ahir, M. & Fu, Y. Characterization of Vascular Morphology of Neovascular Age-Related Macular Degeneration by Indocyanine Green Angiography. JoVE J. Vis. Exp. e65682 (2023). [CrossRef]
- Suñer, I. J. et al. Nicotine increases size and severity of experimental choroidal neovascularization. Invest. Ophthalmol. Vis. Sci. 45, 311–317 (2004). [CrossRef]
- Cousins, S. W. et al. Macrophage Activation Associated with Chronic Murine Cytomegalovirus Infection Results in More Severe Experimental Choroidal Neovascularization. PLOS Pathog. 8, e1002671 (2012). [CrossRef]
- Mettu, P. S., Saloupis, P. & Cousins, S. W. PAMP Stimulation of Macrophages Promotes Neovascular Remodeling in Experimental Choroidal Neovascularization. Invest. Ophthalmol. Vis. Sci. 55, 1198 (2014).
- Wang, Z. et al. Genetic associations of anti-vascular endothelial growth factor therapy response in age-related macular degeneration: a systematic review and meta-analysis. Acta Ophthalmol. (Copenh.) 100, e669–e680 (2022). [CrossRef]
- Zhang, J., Liu, Z., Hu, S. & Qi, J. Meta-Analysis of the Pharmacogenetics of ARMS2 A69S Polymorphism and the Response to Advanced Age-Related Macular Degeneration. Ophthalmic Res. 64, 192–204 (2021). [CrossRef]
- Gourgouli, K. et al. Investigation of genetic base in the treatment of age-related macular degeneration. Int. Ophthalmol. 40, 985–997 (2020). [CrossRef]
- Nguyen, V. P. et al. Age differential response to bevacizumab therapy in choroidal neovascularization in rabbits. Exp. Eye Res. 223, 109215 (2022). [CrossRef]
- Grossniklaus, H. E. et al. Correlation of histologic 2-dimensional reconstruction and confocal scanning laser microscopic imaging of choroidal neovascularization in eyes with age-related maculopathy. Arch. Ophthalmol. Chic. Ill 1960 118, 625–629 (2000). [CrossRef]
- Nishimura, T., Goodnight, R., Prendergast, R. A. & Ryan, S. J. Activated macrophages in experimental subretinal neovascularization. Ophthalmol. J. Int. Ophtalmol. Int. J. Ophthalmol. Z. Augenheilkd. 200, 39–44 (1990). [CrossRef]
- Oh, H. et al. The potential angiogenic role of macrophages in the formation of choroidal neovascular membranes. Invest. Ophthalmol. Vis. Sci. 40, 1891–1898 (1999).
- Tatar, O. et al. Effect of bevacizumab on inflammation and proliferation in human choroidal neovascularization. Arch. Ophthalmol. Chic. Ill 1960 126, 782–790 (2008). [CrossRef]
- Subhi, Y. et al. Association of CD11b+ Monocytes and Anti-Vascular Endothelial Growth Factor Injections in Treatment of Neovascular Age-Related Macular Degeneration and Polypoidal Choroidal Vasculopathy. JAMA Ophthalmol. 137, 515–522 (2019). [CrossRef]
- Buschmann, I. & Schaper, W. Arteriogenesis Versus Angiogenesis: Two Mechanisms of Vessel Growth. News Physiol. Sci. Int. J. Physiol. Prod. Jointly Int. Union Physiol. Sci. Am. Physiol. Soc. 14, 121–125 (1999). [CrossRef]
- Heil, M., Eitenmüller, I., Schmitz-Rixen, T. & Schaper, W. Arteriogenesis versus angiogenesis: similarities and differences. J. Cell. Mol. Med. 10, 45–55 (2006). [CrossRef]
- Arras, M. et al. Monocyte activation in angiogenesis and collateral growth in the rabbit hindlimb. J. Clin. Invest. 101, 40–50 (1998). [CrossRef]
- la Sala, A., Pontecorvo, L., Agresta, A., Rosano, G. & Stabile, E. Regulation of collateral blood vessel development by the innate and adaptive immune system. Trends Mol. Med. 18, 494–501 (2012). [CrossRef]
- McLeod, D. S. et al. Distribution and Quantification of Choroidal Macrophages in Human Eyes With Age-Related Macular Degeneration. Invest. Ophthalmol. Vis. Sci. 57, 5843–5855 (2016). [CrossRef]
- Li, M. et al. Clinicopathologic Correlation of Anti-Vascular Endothelial Growth Factor-Treated Type 3 Neovascularization in Age-Related Macular Degeneration. Ophthalmology 125, 276–287 (2018). [CrossRef]
- Pang, C. E., Messinger, J. D., Zanzottera, E. C., Freund, K. B. & Curcio, C. A. The Onion Sign in Neovascular Age-Related Macular Degeneration Represents Cholesterol Crystals. Ophthalmology 122, 2316–2326 (2015). [CrossRef]
- Kamei, M. et al. Scavenger receptors for oxidized lipoprotein in age-related macular degeneration. Invest. Ophthalmol. Vis. Sci. 48, 1801–1807 (2007). [CrossRef]
- Lin, J. B. et al. Oxysterol Signatures Distinguish Age-Related Macular Degeneration from Physiologic Aging. EBioMedicine 32, 9–20 (2018). [CrossRef]
- Sene, A. et al. Impaired cholesterol efflux in senescent macrophages promotes age-related macular degeneration. Cell Metab. 17, 549–561 (2013). [CrossRef]
- Neale, B. M. et al. Genome-wide association study of advanced age-related macular degeneration identifies a role of the hepatic lipase gene (LIPC). Proc. Natl. Acad. Sci. U. S. A. 107, 7395–7400 (2010). [CrossRef]
- Espinosa-Heidmann, D. G. et al. Macrophage depletion diminishes lesion size and severity in experimental choroidal neovascularization. Invest. Ophthalmol. Vis. Sci. 44, 3586–3592 (2003). [CrossRef]
- Sakurai, E., Anand, A., Ambati, B. K., van Rooijen, N. & Ambati, J. Macrophage depletion inhibits experimental choroidal neovascularization. Invest Ophthalmol Vis Sci 44, 3578–85 (2003). [CrossRef]
- Ishida, S. et al. VEGF164-mediated inflammation is required for pathological, but not physiological, ischemia-induced retinal neovascularization. J Exp Med 198, 483–9 (2003). [CrossRef]
- Nagineni, C. N., Kommineni, V. K., William, A., Detrick, B. & Hooks, J. J. Regulation of VEGF expression in human retinal cells by cytokines: implications for the role of inflammation in age-related macular degeneration. J. Cell. Physiol. 227, 116–126 (2012). [CrossRef]
- Apte, R. S., Richter, J., Herndon, J. & Ferguson, T. A. Macrophages inhibit neovascularization in a murine model of age-related macular degeneration. PLoS Med. 3, e310 (2006). [CrossRef]
- Cherepanoff, S., McMenamin, P., Gillies, M. C., Kettle, E. & Sarks, S. H. Bruch’s membrane and choroidal macrophages in early and advanced age-related macular degeneration. Br. J. Ophthalmol. 94, 918–925 (2010). [CrossRef]
- Grossniklaus, H. E. et al. Macrophage and retinal pigment epithelium expression of angiogenic cytokines in choroidal neovascularization. Mol. Vis. 8, 119–126 (2002).
- Hagbi-Levi, S. et al. Proangiogenic characteristics of activated macrophages from patients with age-related macular degeneration. Neurobiol. Aging 51, 71–82 (2017). [CrossRef]
- Killingsworth, M. C., Sarks, J. P. & Sarks, S. H. Macrophages related to Bruch’s membrane in age-related macular degeneration. Eye Lond. Engl. 4 ( Pt 4), 613–621 (1990). [CrossRef]
- Lopez, P. F., Lambert, H. M., Grossniklaus, H. E. & Sternberg, P. Well-defined subfoveal choroidal neovascular membranes in age-related macular degeneration. Ophthalmology 100, 415–422 (1993). [CrossRef]
- Sarks, J. P., Sarks, S. H. & Killingsworth, M. C. Morphology of early choroidal neovascularisation in age-related macular degeneration: correlation with activity. Eye Lond. Engl. 11 ( Pt 4), 515–522 (1997). [CrossRef]
- Campa, C. et al. Inflammatory mediators and angiogenic factors in choroidal neovascularization: pathogenetic interactions and therapeutic implications. Mediators Inflamm. 2010, (2010). [CrossRef]
- Chen, M., Chan, C.-C. & Xu, H. Cholesterol homeostasis, macrophage malfunction and age-related macular degeneration. Ann. Transl. Med. 6, S55 (2018). [CrossRef]
- Kang, E. Y.-C. et al. Association of Statin Therapy With Prevention of Vision-Threatening Diabetic Retinopathy. JAMA Ophthalmol. 137, 363–371 (2019). [CrossRef]
- Chung, Y.-R. et al. Association of statin use and hypertriglyceridemia with diabetic macular edema in patients with type 2 diabetes and diabetic retinopathy. Cardiovasc. Diabetol. 16, 4 (2017). [CrossRef]
- Vail, D., Callaway, N. F., Ludwig, C. A., Saroj, N. & Moshfeghi, D. M. Lipid-Lowering Medications Are Associated with Lower Risk of Retinopathy and Ophthalmic Interventions among United States Patients with Diabetes. Am. J. Ophthalmol. 207, 378–384 (2019). [CrossRef]
- Vavvas, D. G. et al. Regression of Some High-risk Features of Age-related Macular Degeneration (AMD) in Patients Receiving Intensive Statin Treatment. EBioMedicine 5, 198–203 (2016). [CrossRef]
- Duvall, J. & Tso, M. O. Cellular mechanisms of resolution of drusen after laser coagulation. An experimental study. Arch. Ophthalmol. Chic. Ill 1960 103, 694–703 (1985). [CrossRef]
- Forrester, J. V. Macrophages eyed in macular degeneration. Nat. Med. 9, 1350–1351 (2003). [CrossRef]
- Cousins, S. W., Espinosa-Heidmann, D. G. & Csaky, K. G. Monocyte activation in patients with age-related macular degeneration: a biomarker of risk for choroidal neovascularization? Arch. Ophthalmol. Chic. Ill 1960 122, 1013–1018 (2004). [CrossRef]
- Miller, Y. I., Navia-Pelaez, J. M., Corr, M. & Yaksh, T. L. Lipid rafts in glial cells: role in neuroinflammation and pain processing. J. Lipid Res. 61, 655–666 (2020). [CrossRef]
- Navia-Pelaez, J. M. et al. Normalization of cholesterol metabolism in spinal microglia alleviates neuropathic pain. J. Exp. Med. 218, (2021). [CrossRef]
- Labrecque, L. et al. Regulation of Vascular Endothelial Growth Factor Receptor-2 Activity by Caveolin-1 and Plasma Membrane Cholesterol. Mol. Biol. Cell 14, 334–347 (2003). [CrossRef]
- Laurenzana, A. et al. Lipid rafts: integrated platforms for vascular organization offering therapeutic opportunities. Cell. Mol. Life Sci. CMLS 72, 1537–1557 (2015). [CrossRef]
- Pilarczyk, M. et al. Endothelium in Spots – High-Content Imaging of Lipid Rafts Clusters in db/db Mice. PLOS ONE 9, e106065 (2014). [CrossRef]
- Fang, L. et al. Control of angiogenesis by AIBP-mediated cholesterol efflux. Nature 498, 118–122 (2013). [CrossRef]
- Schneider, D. A. et al. AIBP protects against metabolic abnormalities and atherosclerosis. J. Lipid Res. (2018). [CrossRef]
- Zhang, M. et al. AIBP reduces atherosclerosis by promoting reverse cholesterol transport and ameliorating inflammation in apoE-/-mice. Atherosclerosis (2018). [CrossRef]
- Zhang, M. et al. Apolipoprotein A-1 binding protein promotes macrophage cholesterol efflux by facilitating apolipoprotein A-1 binding to ABCA1 and preventing ABCA1 degradation. Atherosclerosis 248, 149–159 (2016). [CrossRef]
- Dubrovsky, L. et al. Inhibition of HIV Replication by Apolipoprotein A-I Binding Protein Targeting the Lipid Rafts. mBio 11, (2020). [CrossRef]
- Gu, Q. et al. AIBP-mediated cholesterol efflux instructs hematopoietic stem and progenitor cell fate. Science 363, 1085–1088 (2019). [CrossRef]
- Choi, S.-H. et al. AIBP augments cholesterol efflux from alveolar macrophages to surfactant and reduces acute lung inflammation. JCI Insight 3, (2018). [CrossRef]
- Qiu, X., Luo, J. & Fang, L. AIBP, Angiogenesis, Hematopoiesis, and Atherogenesis. Curr. Atheroscler. Rep. 23, 1 (2020). [CrossRef]
- Woller, S. A. et al. Inhibition of Neuroinflammation by AIBP: Spinal Effects upon Facilitated Pain States. Cell Rep. 23, 2667–2677 (2018). [CrossRef]
- Corliss, B. A., Azimi, M. S., Munson, J. M., Peirce, S. M. & Murfee, W. L. Macrophages: An Inflammatory Link Between Angiogenesis and Lymphangiogenesis. Microcirc. N. Y. N 1994 23, 95–121 (2016). [CrossRef]
- Deindl, E. et al. Role of ischemia and of hypoxia-inducible genes in arteriogenesis after femoral artery occlusion in the rabbit. Circ. Res. 89, 779–786 (2001). [CrossRef]
- Schierling, W. et al. The role of angiogenic growth factors in arteriogenesis. J. Vasc. Res. 46, 365–374 (2009). [CrossRef]
- Wu, S. et al. Immunohistochemical study of the growth factors, aFGF, bFGF, PDGF-AB, VEGF-A and its receptor (Flk-1) during arteriogenesis. Mol. Cell. Biochem. 343, 223–229 (2010). [CrossRef]
- Abudukeremu, A. et al. Efficacy and Safety of High-Density Lipoprotein/Apolipoprotein A1 Replacement Therapy in Humans and Mice With Atherosclerosis: A Systematic Review and Meta-Analysis. Front. Cardiovasc. Med. 8, (2021). [CrossRef]
- Morton, J. et al. Strikingly Different Atheroprotective Effects of Apolipoprotein A-I in Early- Versus Late-Stage Atherosclerosis. JACC Basic Transl. Sci. 3, 187–199 (2018). [CrossRef]
- Shah, P. K. et al. High-dose recombinant apolipoprotein A-I(milano) mobilizes tissue cholesterol and rapidly reduces plaque lipid and macrophage content in apolipoprotein e-deficient mice. Potential implications for acute plaque stabilization. Circulation 103, 3047–3050 (2001). [CrossRef]
- Miyazaki, A. et al. Intravenous injection of rabbit apolipoprotein A-I inhibits the progression of atherosclerosis in cholesterol-fed rabbits. Arterioscler. Thromb. Vasc. Biol. 15, 1882–1888 (1995). [CrossRef]
- Tangirala, R. K. et al. Regression of atherosclerosis induced by liver-directed gene transfer of apolipoprotein A-I in mice. Circulation 100, 1816–1822 (1999). [CrossRef]
- Busbee, B. G. et al. Twelve-month efficacy and safety of 0.5 mg or 2.0 mg ranibizumab in patients with subfoveal neovascular age-related macular degeneration. Ophthalmology 120, 1046–1056 (2013). [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).



