Submitted:
11 January 2024
Posted:
12 January 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Reagents and Instruments
2.2. Experimental Methods
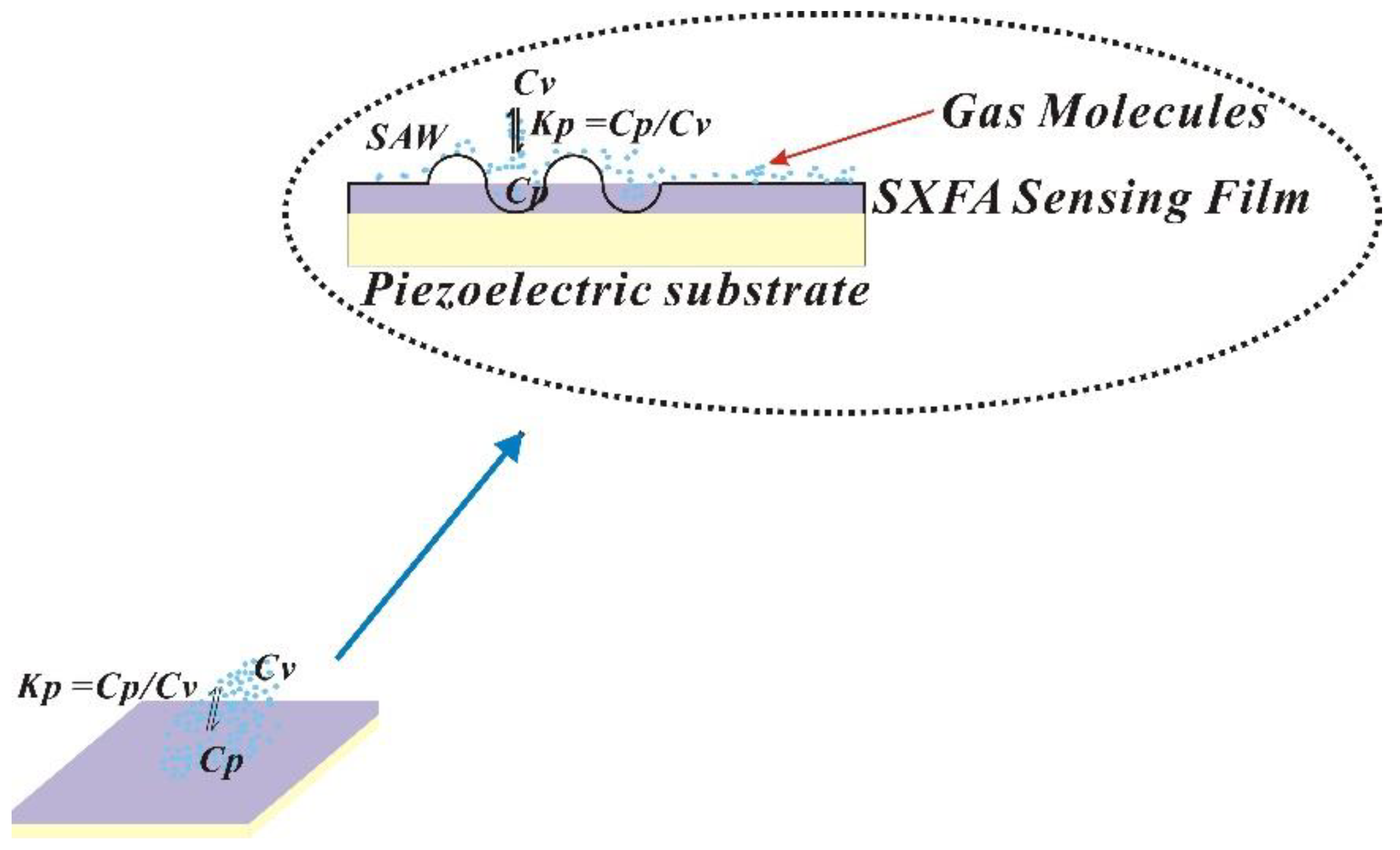
2.2.1. Mechanism of Interaction Between Polymer and Gas Molecules
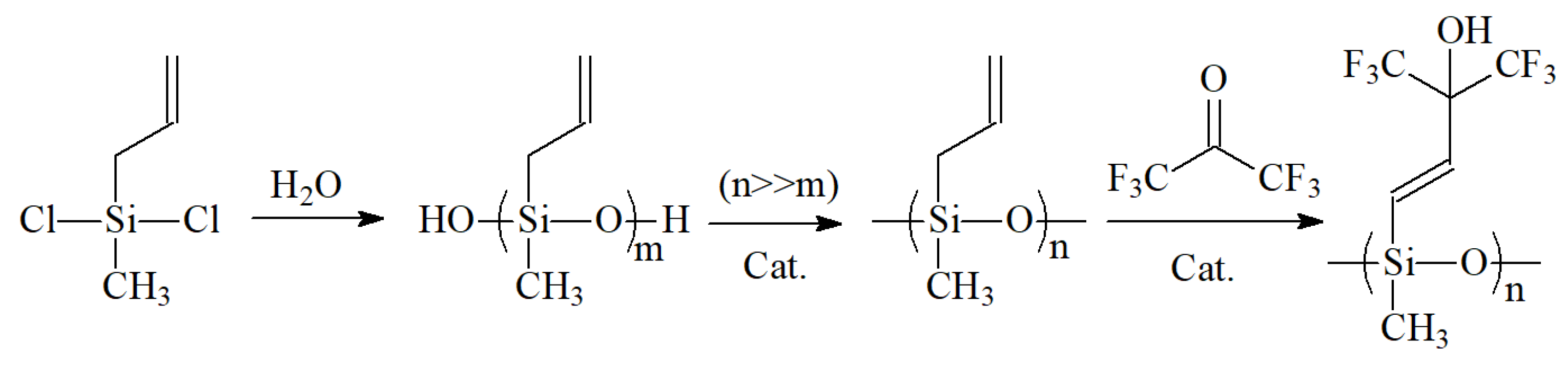
2.2.2. Synthesis Route of Hexafluoro-2-hydroxyisopropyl Polysiloxane
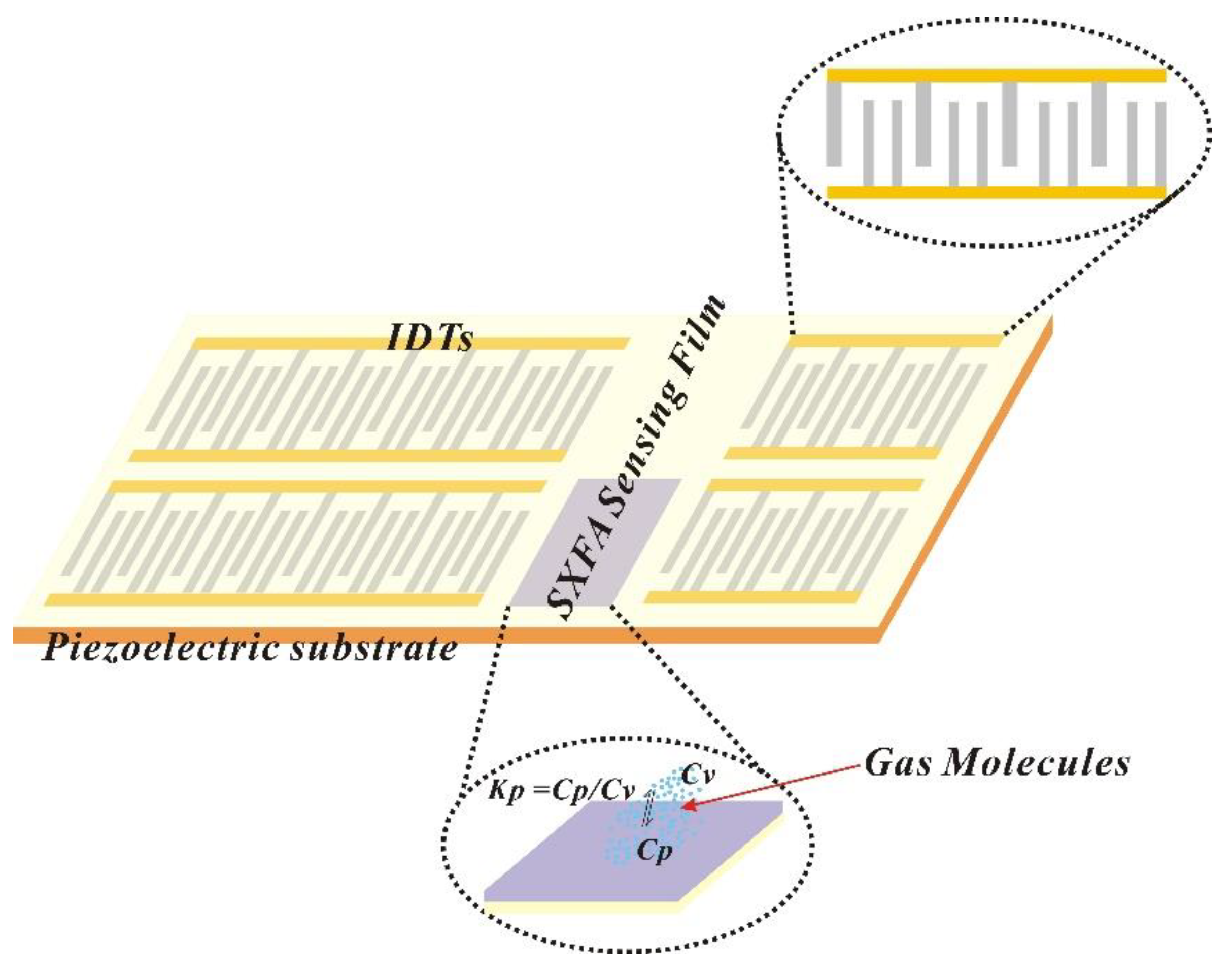
2.2.3. Preparation of SAW-SXFA Sensor and Its Detection of Organophosphine Agents
3. Results and Discussions
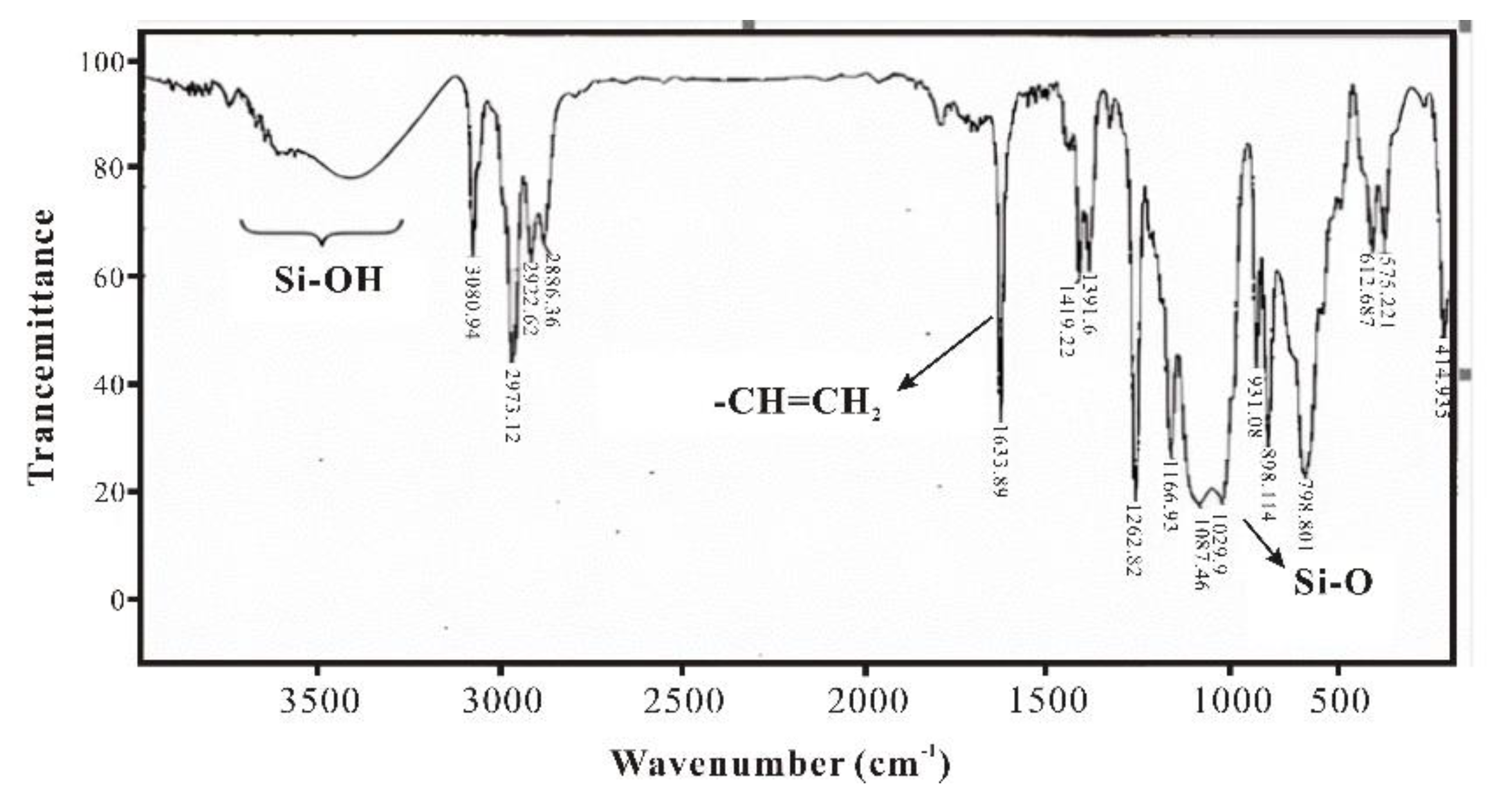
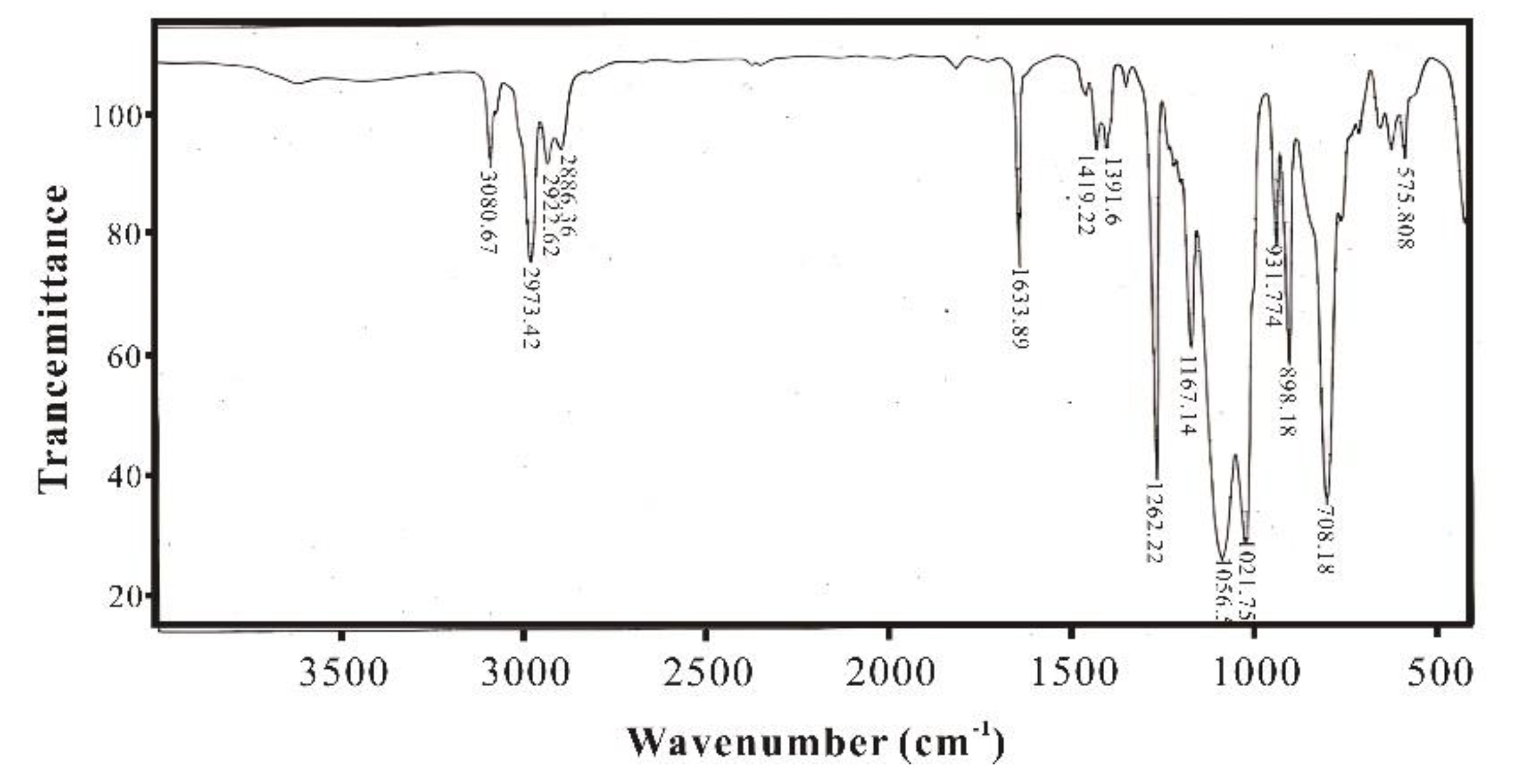
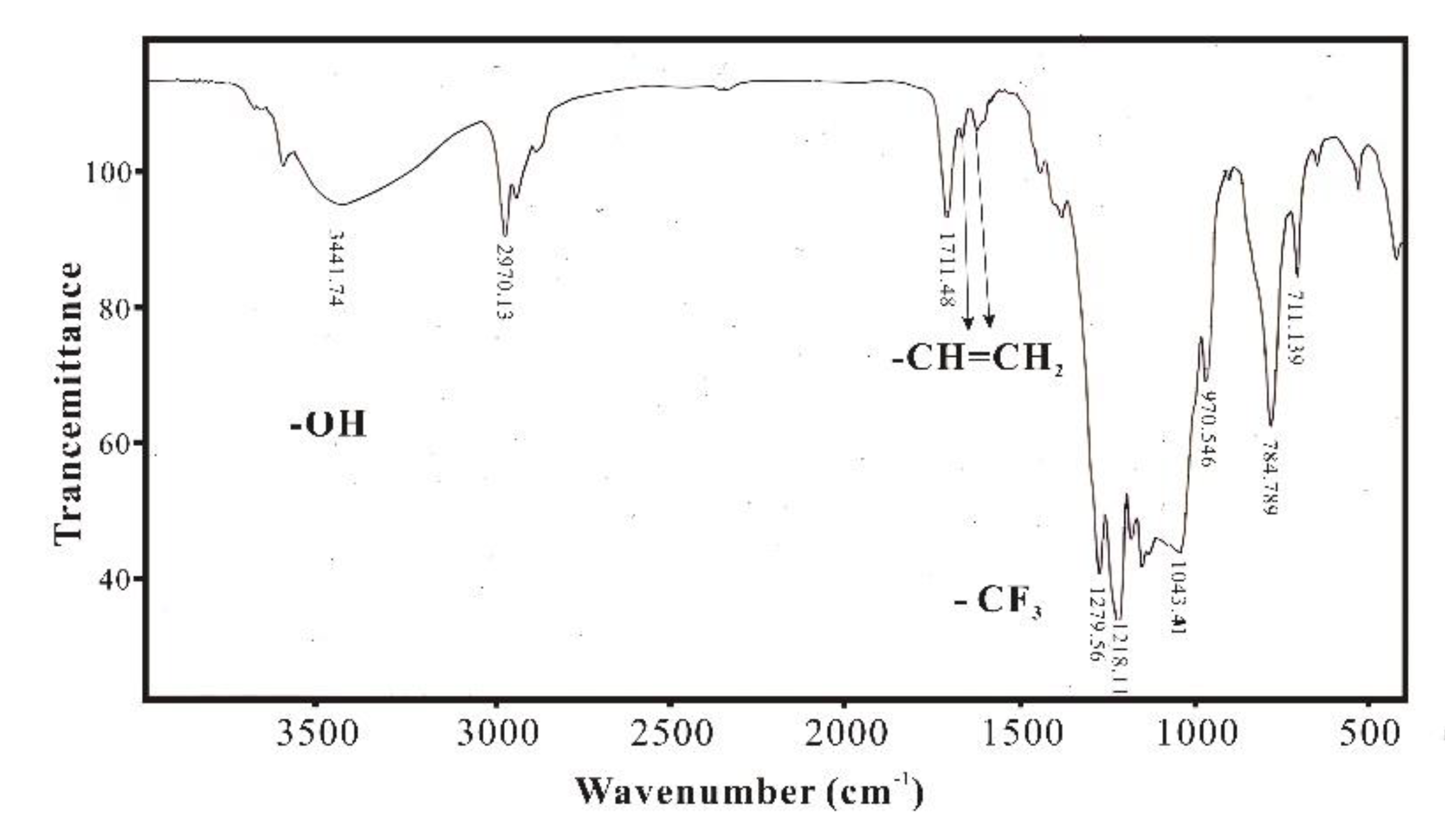
3.1. Infrared Spectroscopy Characterization and Analysis of SXFA Material
3.2. Detection of Organophosphorus Agents
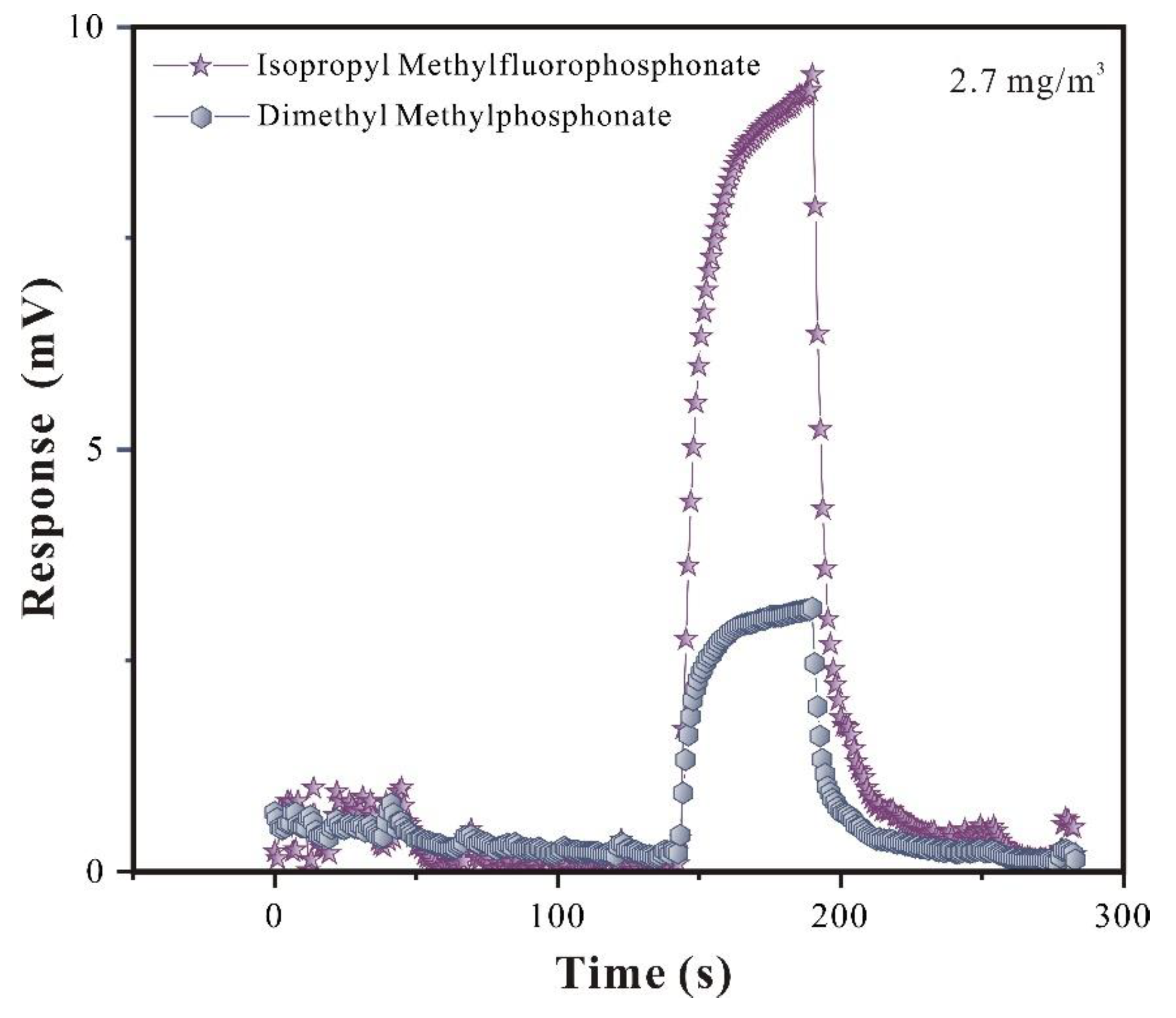
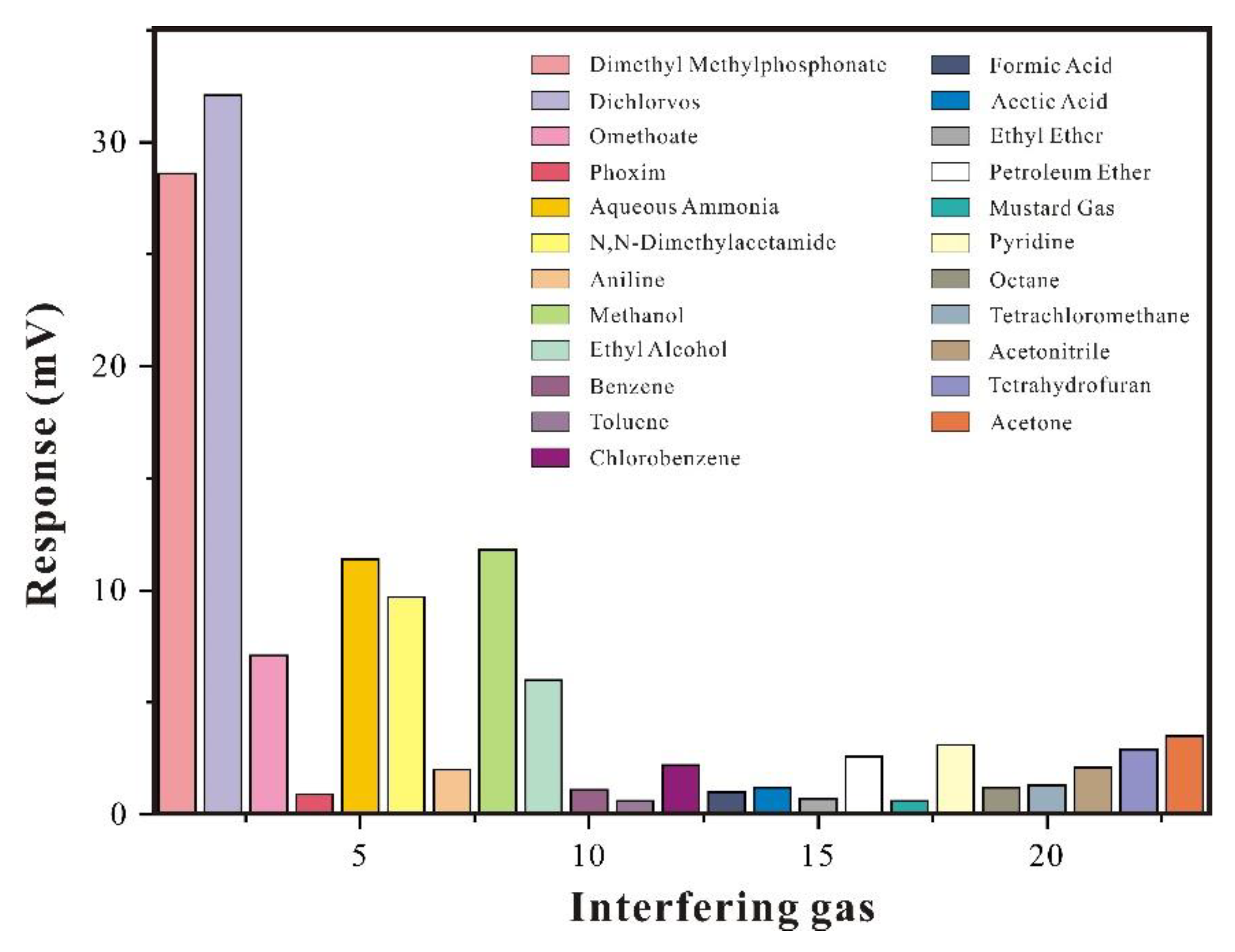
3.2.1. Selective Analysis of SAW-SXFA Sensor
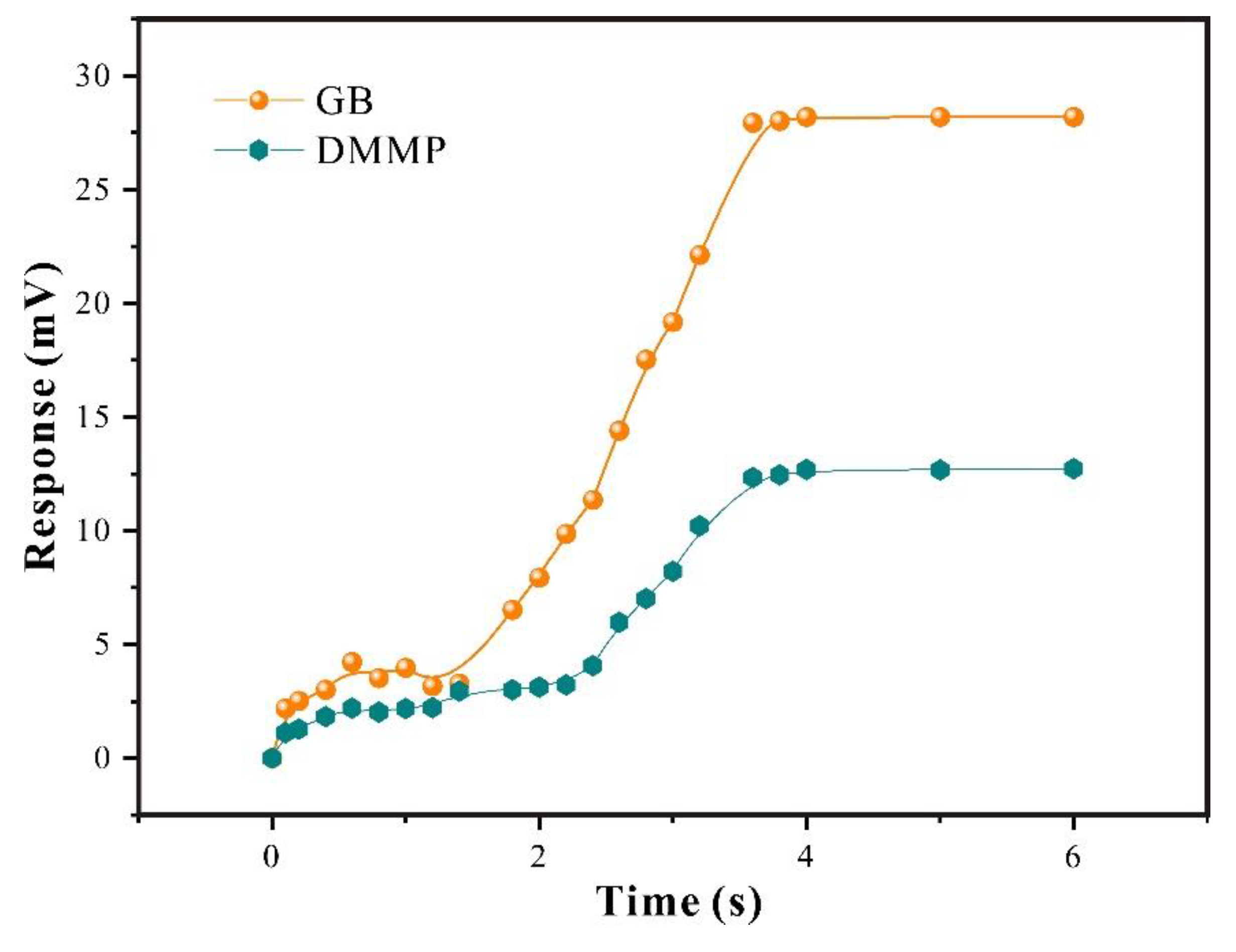
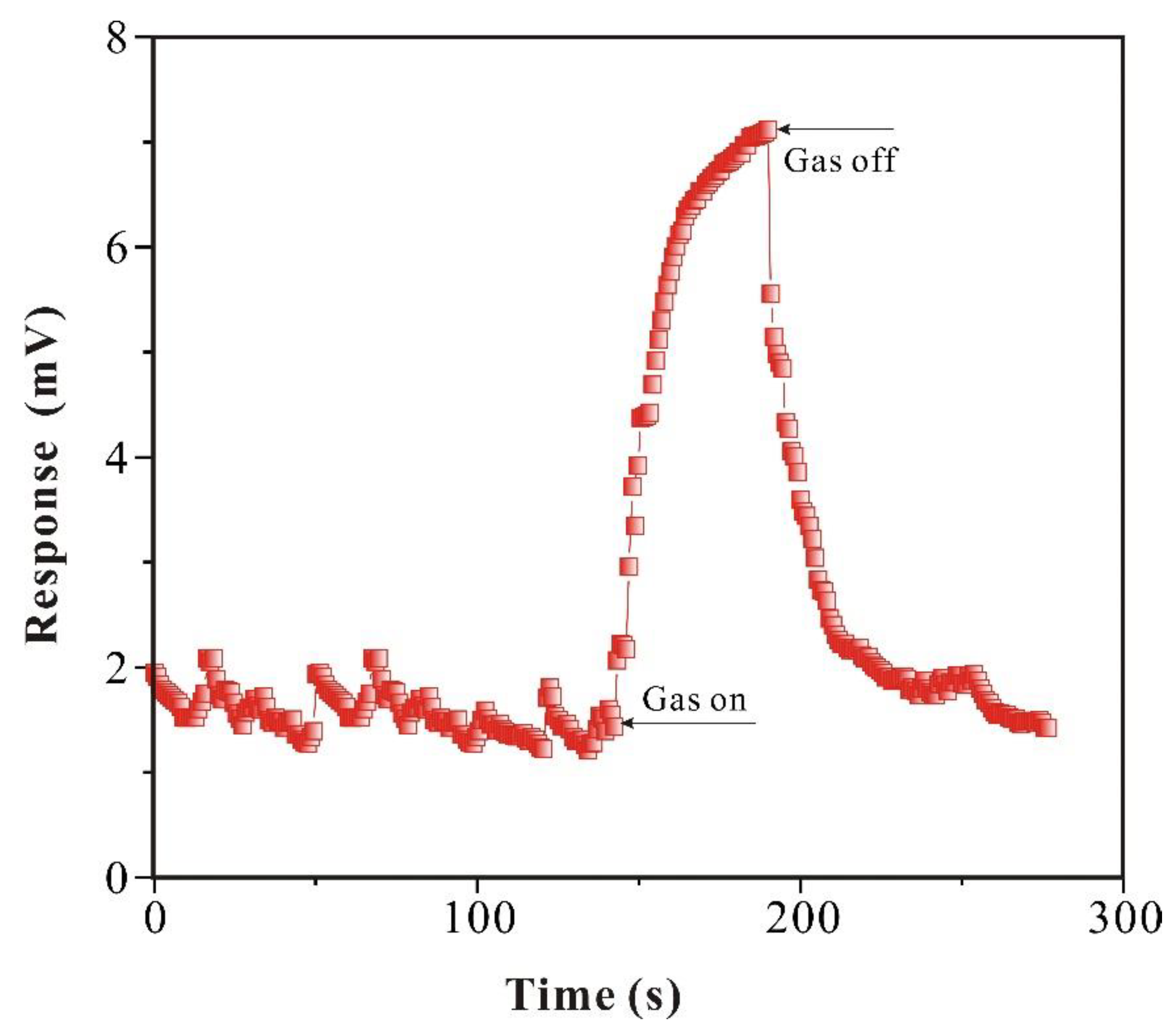
3.2.2. Analysis of Response of SAW-SXFA Sensor
3.2.3. Detection Limit of SAW-SXFA Sensor
| Intensity (mg/m3) | Response (mV) | Recovery (mV) | Recovery Rate (%) |
|---|---|---|---|
| 0.1 | 2.168 | 0.02 | 98 |
| 0.2 | 2.509 | 0.05 | 95 |
| 0.4 | 3.002 | 0.09 | 91 |
| 0.6 | 4.216 | 0.08 | 92 |
| 0.8 | 3.523 | 0.12 | 88 |
| 1.0 | 3.972 | 0.12 | 88 |
| 1.4 | 3.283 | 0.14 | 86 |
| 1.8 | 6.515 | 0.20 | 80 |
| 2.2 | 9.859 | 0.23 | 77 |
| 2.6 | 14.394 | 0.27 | 73 |
| 3.0 | 19.172 | 0.30 | 70 |
| 3. 4 | 27.605 | 0.35 | 65 |
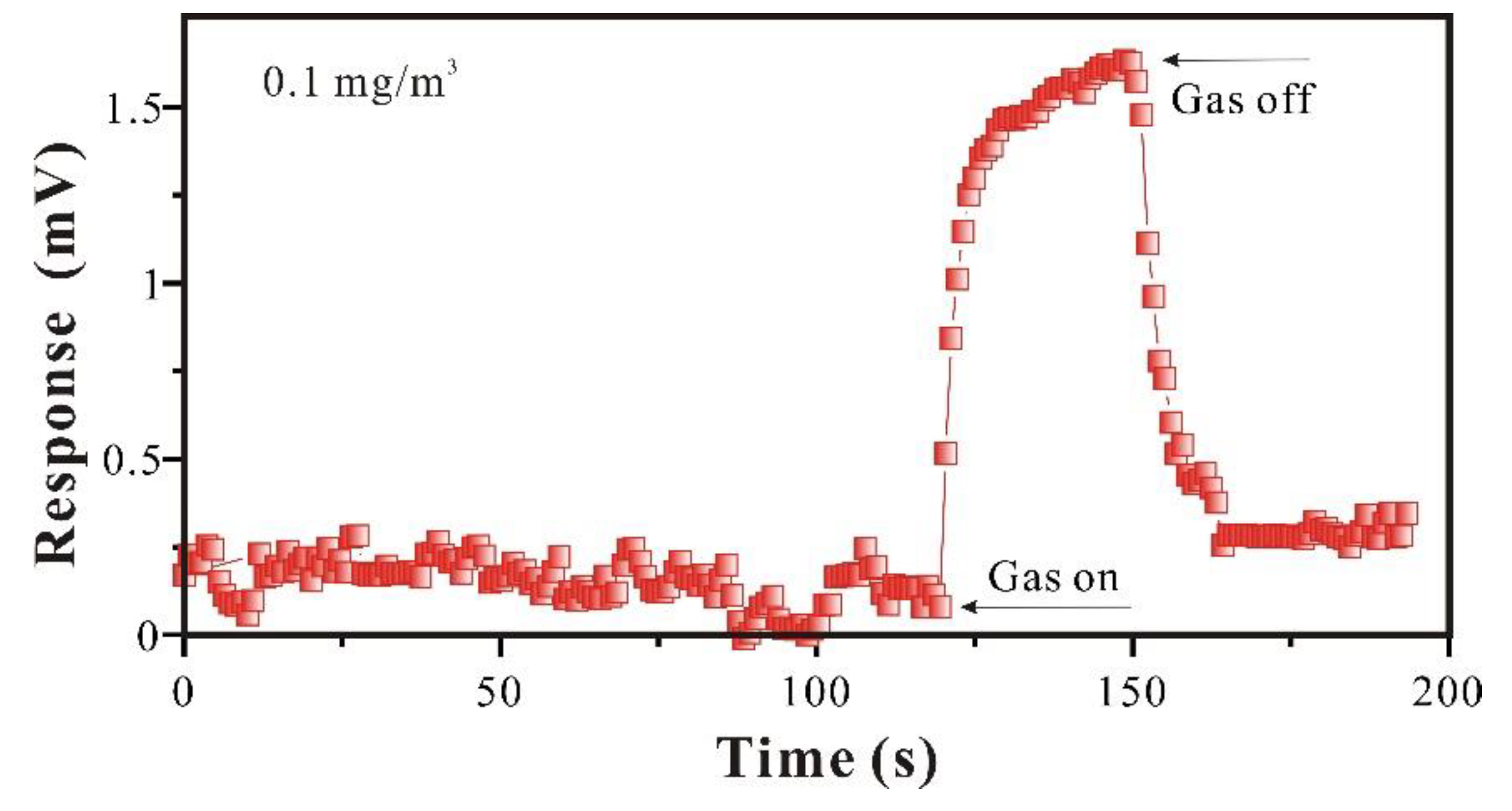
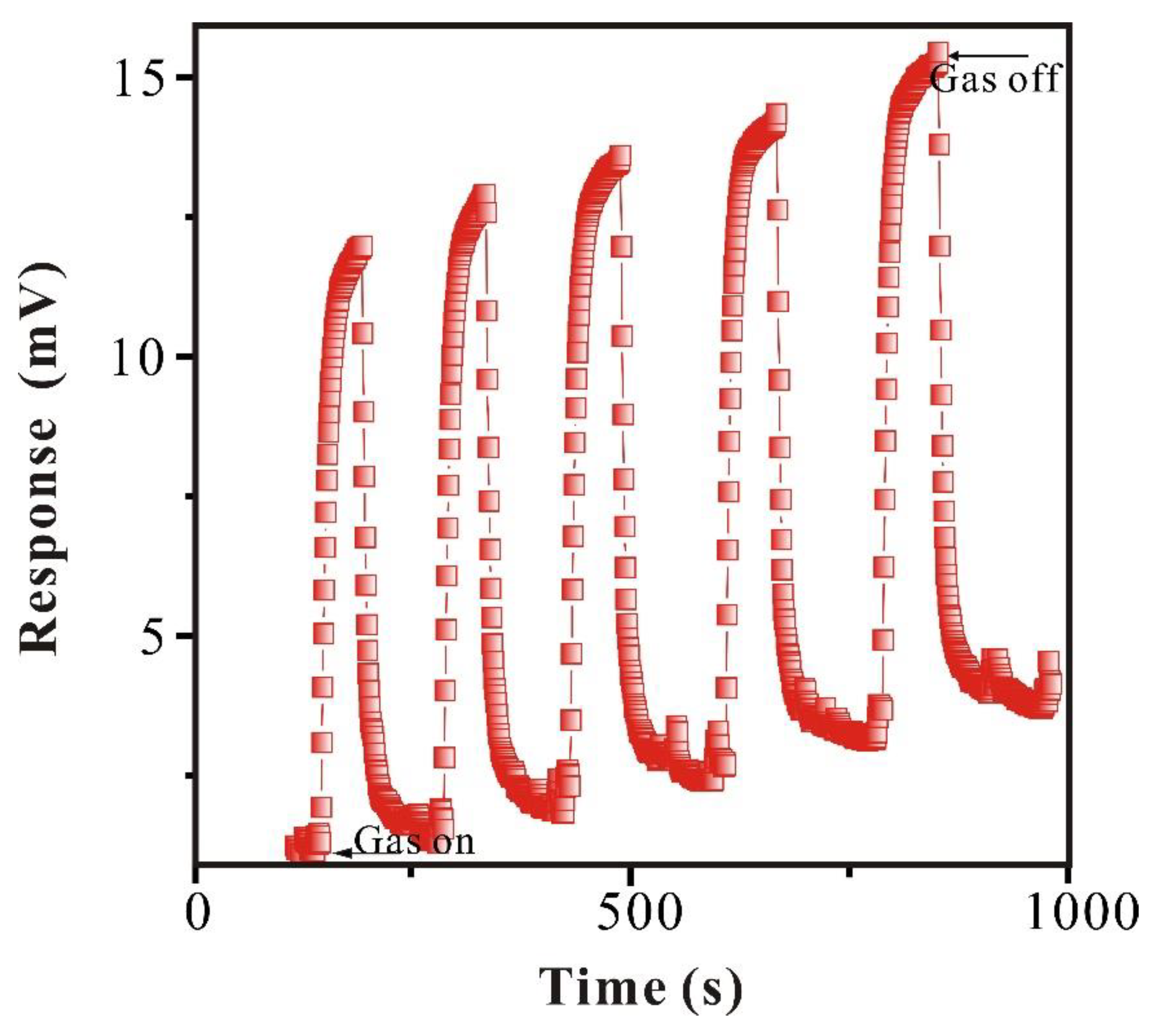
3.2.3. Reproducibility Study of SAW-SXFA Sensor
3.2.4. Interference Gas Research
4. Conclusions
Author Contributions
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Abu-Qare, A.W.; Abou-Donia, M.B. Sarin: health effects, metabolism, and methods of analysis. Food Chem. Toxicol. 2002, 40, 1327–1333. [Google Scholar] [CrossRef]
- Lee, E.C. Clinical manifestations of sarin nerve gas exposure. Jama-J. Am. Med. Assoc. 2003, 290, 659–662. [Google Scholar] [CrossRef]
- Tokuda, Y.; Kikuchi, M.; Takahashi, O.; Stein, G.H. Prehospital management of sarin nerve gas terrorism in urban settings: 10 years of progress after the Tokyo subway sarin attack. 2006 , 68, 2, 193–202.
- Yoo, J.; Kim, D.; Yang, H.; Lee, M.; Kim, S.O.; Ko, H.J.; Hong, S.; Park, T.H. Olfactory receptor-based CNT-FET sensor for the detection of DMMP as a simulant of sarin.Sensors and Actuators B-Chemical, 2022, 354.
- Hamel, M; Hamoniaux, J; Rocha, L; Normand, S. Ppb detection of Sarin surrogate in liquid solutions. Chemical, Biological, Radiological, Nuclear, and Explosives (CBRNE) Sensing XIV. 2013, 8710.
- O’Neill, H.J.; Brubaker, K.L.; Schneider, J.F.; Sytsma, L.F.; Kimmell, T.A. Development of an analytical methodology for sarin (GB) and soman (GD) in various military-related-wastes. J. Chromatogr. A 2002, 962, 183–195. [Google Scholar] [CrossRef]
- Maziejuk, M.; Ceremuga, M.; Szyposzynska, M.; Sikora, T.; Zalewska, A. Identification of organophosphate nerve agents by the DMS detector. Sens. Actuators B-Chem. 2015, 213, 368–374. [Google Scholar] [CrossRef]
- Black, R.M.; Clarke, R.J.; Read, R.W.; Reid, M.T.J. Application of gas-chromatography mass-spectrometry and Gas-chromatography tandem mass-spectrometry to the analysis of chemical warfare samples, found to contain residues of the nerve agent sarin, sulfur mustard and their degradation products. J. Chromatogr. A 1994, 662, 301–321. [Google Scholar] [CrossRef]
- William, H.K. Pizoelectric Sorption Detector. Anal. Chem. 1964, 36, 1735–1739. [Google Scholar]
- Stevenson, A.C.; Mehta, H.M.; Sethi, R.S.; Cheran, L.E.; Thompson, M.; Davies, I.; Lowe, C.R. Gigahertz surface acoustic wave probe for chemical analysis. Analyst 2001, 126, 1619–1624. [Google Scholar] [CrossRef]
- Fahim, F.; Mainuddin, M.; Mittal, U.; Kumar, J.; Nimal, A.T. Novel SAW CWA Detector Using Temperature Programmed Desorption. IEEE Sens. J. 2021, 21, 2915–5922. [Google Scholar] [CrossRef]
- Raj, V.B.; Singh, H.; Nimal, A.T.; Sharma, M.U.; Gupta, V. Oxide thin films (ZnO, TeO2, SnO2, and TiO2) based surface acoustic wave (SAW) E-nose for the detection of chemical warfare agents. Sens. Actuators B-Chem. 2013, 178, 647. [Google Scholar] [CrossRef]
- Pan, Y.; Zhang, G.; Guo, T.; Liu, X.; Liu, X.; Zhang, C.; Yang, J.; Cao, B.; Zhang, C.; Wang, W. Environmental characteristics of surface acoustic wave devices for sensing organophosphorus vapor. Sens. Actuators B Chem. 2020, 315, 127986. [Google Scholar] [CrossRef]
- Pan, Y.; Qin, M.; Wang, P.; Yang, L.; Zhang, L.; Yan, C.; Zhang, C.; Wang, W. Interface and Sensitive Characteristics of the Viscoelastic Film Used in a Surface Acoustic Wave Gas Sensor. ACS Sens. 2022, 7, 612–621. [Google Scholar] [CrossRef]
- Matatagui, D.; Martí, J.; Fernández, M.J.; Fontecha, J.L.; Gutiérrez, J.; Gràcia, I.; Cané, C.; Horrillo, M.C. Chemical warfare agents simulants detection with an optimized SAW sensor array. Sens. Actuators B-Chem. 2011, 154, 199–205. [Google Scholar] [CrossRef]
- Lama, S.; Kim, J.; Ramesh, S.; Lee, Y.J.; Kim, J.; Kim, J.H. Highly Sensitive Hybrid Nanostructures for Dimethyl Methyl Phosphonate Detection. Micromachines 2021, 12, 6. [Google Scholar] [CrossRef]
- Lurz, F.; Ostertag, T.; Scheiner, B.; Weigel, R.; Koelpin, A. Reader Architectures for Wireless Surface Acoustic Wave Sensors. Sensors 2018, 18, 6. [Google Scholar] [CrossRef]
- Palla-Papavlu, A.; Voicu, S.I.; Dinescu, M. Sensitive Materials and Coating Technologies for Surface Acoustic Wave Sensors. Chemosensors 2021, 9, 5. [Google Scholar] [CrossRef]
- Liu, X.; Wang, W.; Zhang, Y.; Pan, Y.; Liang, Y.; Li, J. Enhanced Sensitivity of a Hydrogen Sulfide Sensor Based on Surface Acoustic Waves at Room Temperature. Sensors 2018, 18, 11. [Google Scholar] [CrossRef] [PubMed]
- Wen, W.; He, S.; Li, S.; Liu, M.; Pan, Y. Advances in SXFA-Coated SAW Chemical Sensors for Organophosphorous Compound Detection. Sensors 2011, 11, 1526–1541. [Google Scholar]
- Grate, J.W. Hydrogen Bond Acidic Polymers for Surface Acoustic Wave Vapor Sensors and Arrays. Anal. Chem. 1999, 71, 1033–1040. [Google Scholar] [CrossRef] [PubMed]
- Abraham, M.H.; Andonian-Haftvan, J.; Du, C.M.; Diart, V.; Whiting, G.S.; Grate, J.W.; Andrew, M.R. Hydrogen bonding. Part 29. Characterization of 14 sorbent coatings for chemical microsensors using a new solvation equation. J. Chem. Soc. Perkin Trans. 1995, 2, 369–378. [Google Scholar] [CrossRef]
- Freudenberg, J.; Schickfus, M.V.; Hunklinger, S. A SAW immunosensor for operation in liquid using a SiO2 protective layer. Sens. Actuators B Chem. 2001, 76, 147–151. [Google Scholar] [CrossRef]
- Kumar, K.V.; Gadipelli, S.; Wood, B.; Ramisetty, K.A.; Stewart, A.A.; Howard, C.A.; Brett, D.J.L.; Rodriguez-Reinoso, F. Characterization of the adsorption site energies and heterogeneous surfaces of porous materials. J. Mater. Chem. A 2019, 7, 17. [Google Scholar] [CrossRef]
- Al-Ghouti, M.A; Da'ana, D.A. Guidelines for the use and interpretation of adsorption isotherm models: A review. J. Hazard. Mater. 2020, 393. [Google Scholar] [CrossRef] [PubMed]
- Kindlund, A.; Sundgren, H.; Lundstrm, I. Quartz crystal gas monitor with a gas concentrating stage. Sens. Actuators 1984, 6, 1–17. [Google Scholar] [CrossRef]
- Finklea, H.O.; Phillippi, M.A.; Lompert, E.; Grate, J.W. Highly sorbent films derived from ni(scn)2(4-picoline)4 for the detection of chlorinated and aromatic hydrocarbons with quartz crystal microbalance sensors. Anal. Chem. 1998, 70, 1268–76. [Google Scholar] [CrossRef] [PubMed]
- Gregory, C.F.; Stephen, J.M. Materials characterization using surface acoustic wave devices. Appl. Spectrosc. Rev. 1991, 26, 73–149. [Google Scholar]
- Grate, J.W.; Wenzel, S.W.; White, R.M. Flexural plate wave device for chemical analysis. Anal. Chem. 1991, 63, 1552–1561. [Google Scholar] [CrossRef]
- Grate, J.W.; Klusty, M.; Mcgill, R.A.; Abraham, M.H.; Whiting, G.; Andonian-Haftvan, J. The predominant role of swelling-induced modulus changes of the sorbent phase in determining the responses of polymer-coated surface acoustic wave vapor sensors. Anal. Chem. 1992, 64, 610–624. [Google Scholar] [CrossRef]
- Tascon, M.; Romero, L.M.; Acquaviva, A.; Keunchkarian, S.; Castells, C. Determinations of gas-liquid partition coefficients using capillary chromatographic columns. alkanols in squalane. J. Chromatogr. A 2013, 1294, 130–136. [Google Scholar] [CrossRef] [PubMed]
- Abraham, M.H.; RosÉS, M.; Poole, C.F.; Poole, S.K. Hydrogen bonding. 42. characterization of reversed-phase high-performance liquid chromatographic c18 stationary phases. J. Phys. Org. Chem. 2015, 10, 358–368. [Google Scholar] [CrossRef]
- Grate, J.W.; Patrash, S.J.; Abraham, M.H. Method for estimating polymer-coated acoustic wave vapor sensor responses. Anal. Chem. 1995, 67, 2162–2169. [Google Scholar] [CrossRef]
- Grate, J.W.; Kaganove, S.N.; Bhethanabotla, V.R. Comparisons of polymer/gas partition coefficients calculated from responses of thickness shear mode and surface acoustic wave vapor sensors. Anal. Chem. 1998, 70, 199–203. [Google Scholar] [CrossRef] [PubMed]
| Polymer | b/a | b/s | a/s | s/a | b+a+s | Dispersibility |
|---|---|---|---|---|---|---|
| SXFA | 6.07 | 7.08 | 1.17 | 0.86 | 5.55 | 0.13 |
| Experiment No | Response (mV) | Recovery (mV) |
|---|---|---|
| 1 | 10.845 | 9.696 |
| 2 | 10.619 | 10.073 |
| 3 | 10.767 | 10.176 |
| 4 | 10.921 | 9.729 |
| 5 | 10.831 | 9.741 |
| Average (mV) | 10.797 | 9.883 |
| Standard Deviation (mV) | 0.11 | 0.22 |
| Discrete coefficient | 0.01 | 0.022 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




