Submitted:
10 January 2024
Posted:
11 January 2024
You are already at the latest version
Abstract
Keywords:
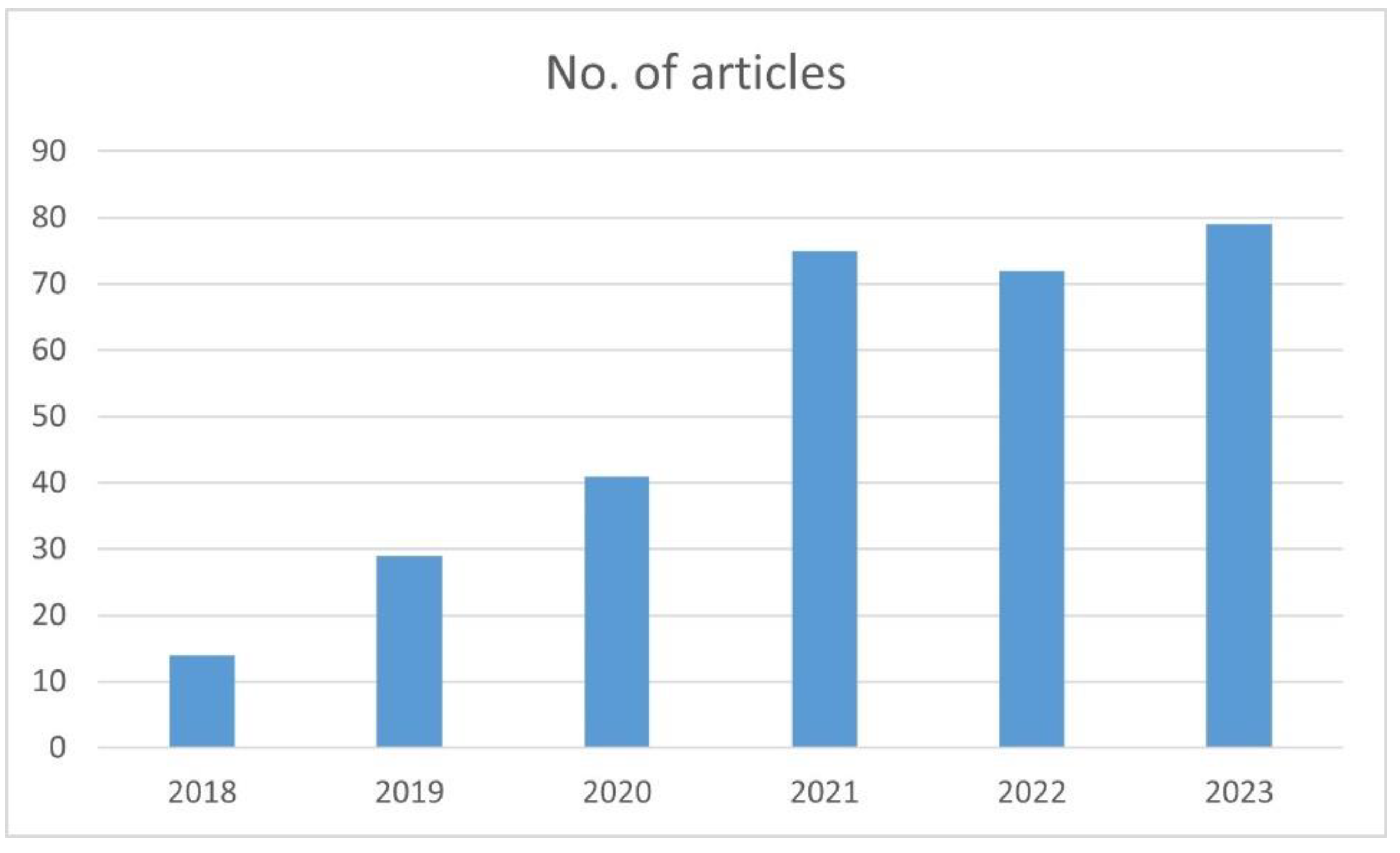
1. Introduction
2. Imaging modalities
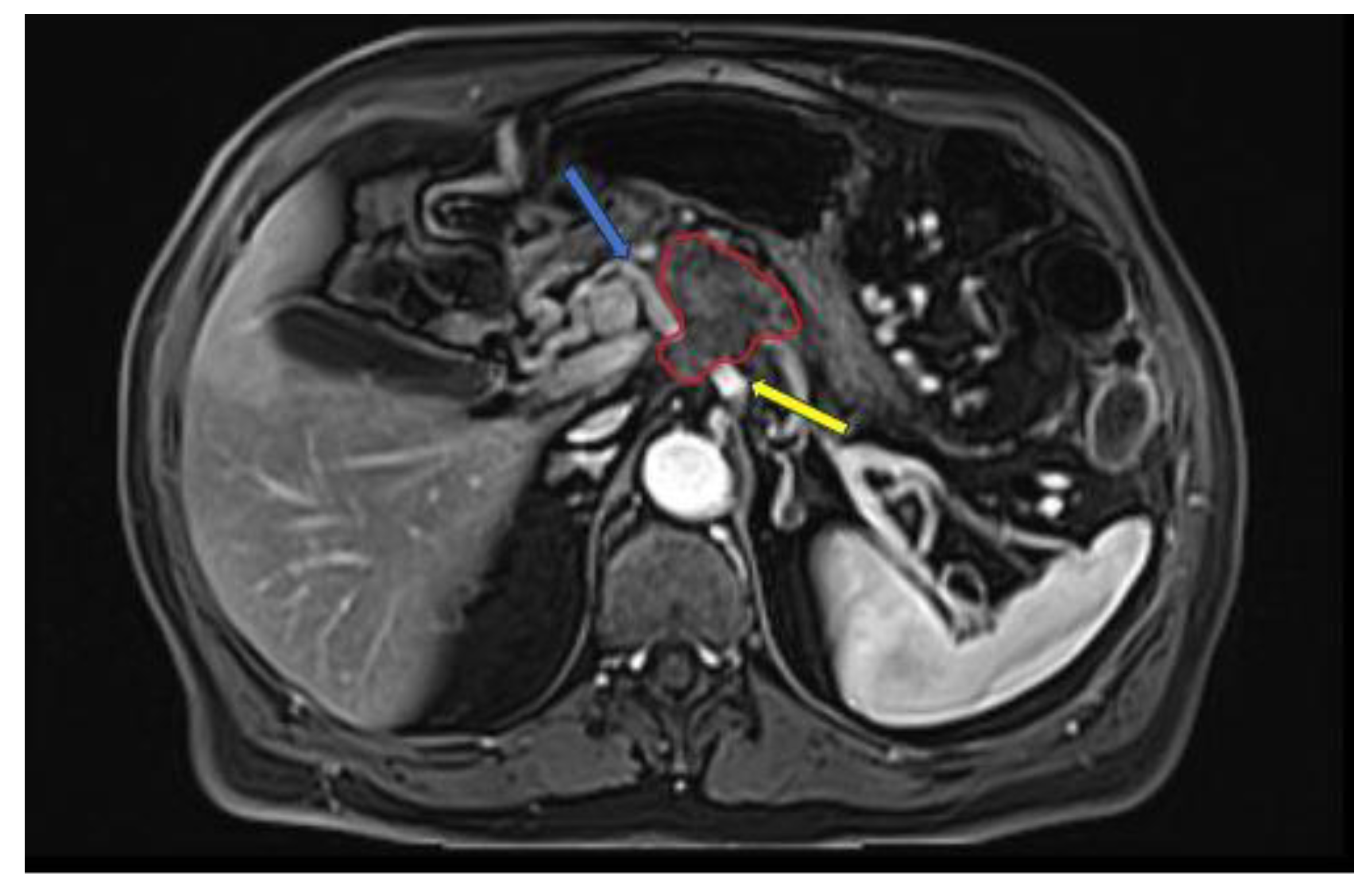
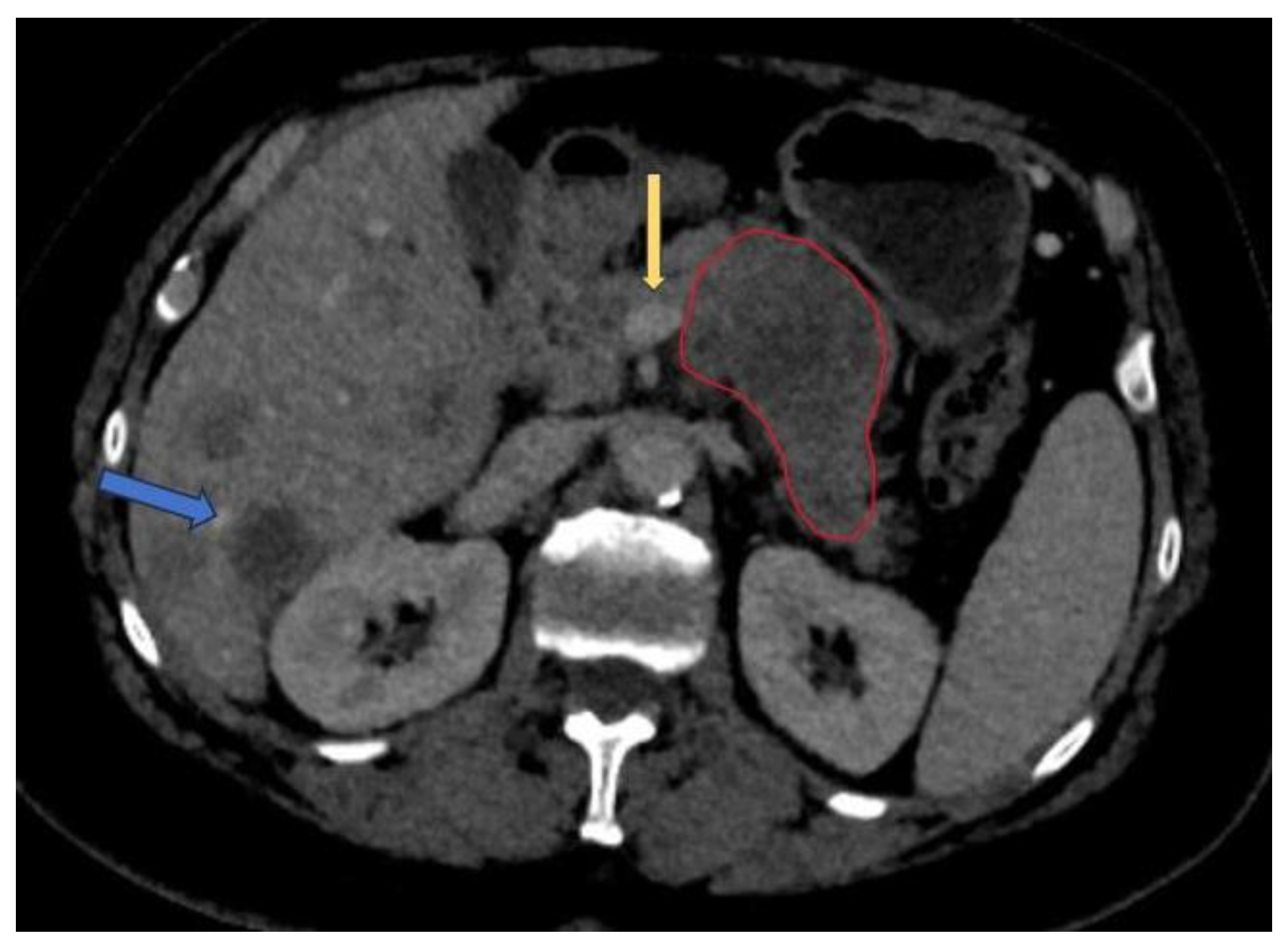
3. AI applications in PDAC
3.1. Detection
| Author | Year | Modality | Approach | Sensitivity |
|---|---|---|---|---|
| Cao et al.[38] | 2023 | Non-contrast CT | Deep learning | 92.9% |
| Korifatis et al.[39] | 2023 | CECT | 3D-CNN | 75% |
| Alves et al.[44] | 2022 | CECT | CNN | N.A. |
| Wang et al.[46] | 2022 | CECT | Radiomics | 84% |
| Mukherjee et al.[45] | 2022 | CECT | Radiomics-ML | 95.5% |
| Viviers et al.[49] | 2022 | CECT | U-Net-Like Deep CNN | 99% |
| Man et al.[43] | 2020 | Non-contrast CT | CNN | 91.58% |
| Chu et al.[40] | 2019 | CECT | Deep learning | 94,1% |
| Chu et al.[41] | 2019 | CECT | Radiomics | 100% |
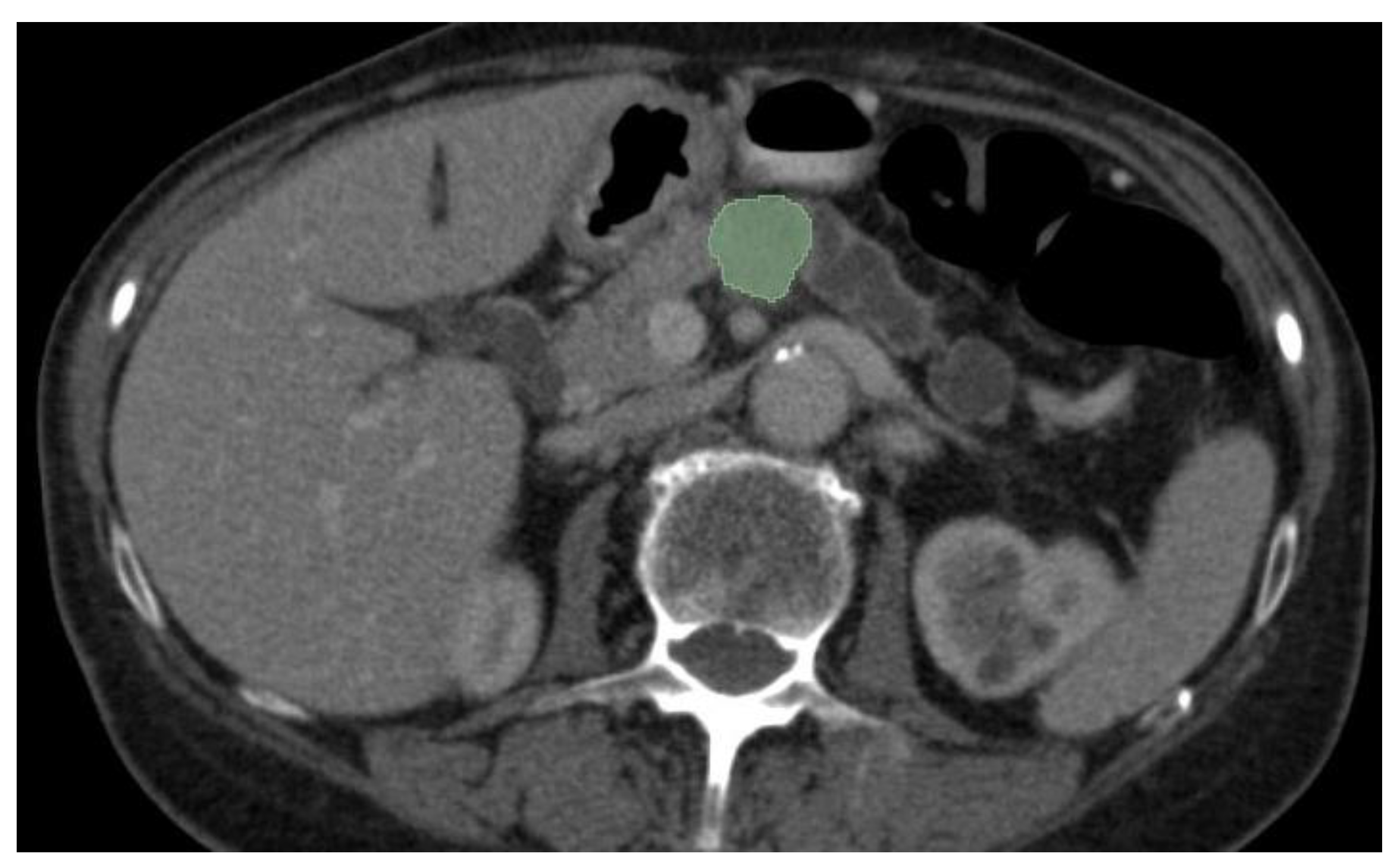
3.2. Segmentation
| Author | Year | Modality | AI Method | DICE score |
|---|---|---|---|---|
| Ni et al.[57] | 2023 | CECT | AX-Unet | 85,9% |
| Mahmoudi et al.[62] | 2022 | CECT | Hybrid 3D-CNN model | 60,6% |
| Tureckova et al.[58] | 2020 | CECT | VNet-AG-DSV | 52,99% |
| Zhou et al.[61] | 2019 | CECT | 3D-ResDSN-multi-HPN | 63.94% |
| Isensee et al.[60] | 2018 | CECT | nnU-Net | 52,12% |
3.2. Classification
3.2.1. Differential diagnosis
| Author | Year | Modality | Scope | Approach | AUC |
|---|---|---|---|---|---|
| Lu et al.[64] | 2023 | CECT | fAPI vs PDAC | Radiomics | 0.83 |
| Shi et al.[69] | 2023 | MRI | PDAC vs PNEN and SPN | CNN | 0.839 |
| Zhang et al.[71] | 2022 | CECT | PDAC vs pNET | Radiomics | 0.930 |
| Anai et al.[66] | 2022 | CECT | fAPI vs PDAC | Radiomics | 0.920 |
| Deng et al.[67] | 2021 | MRI | MFCP vs PDAC | Radiomics | 0.962 |
| Ren et al.[68] | 2020 | CECT | PASC vs PDAC | Radiomics | 0.82 |
| Park et al.[65] | 2020 | CECT | AIP vs PDAC | Radiomics | 0.975 |
3.2.2. Histopathological subtype and genomic features
| Author | Year | Modality | Scope | Approach | Performance |
|---|---|---|---|---|---|
| Cen et al.[80] | 2023 | CECT | Histopathological grade | Radiomics | AUC 0.76 |
| Deng et al.[87] | 2022 | MRI | MUC4 expression | Radiomics | AUC 0.861 |
| Hinzpeter et al.[82] | 2022 | CECT | Correlation with driver gene mutations | Radiomics | Youden Index 0.56 (KRAS), 0.67 (TP53), 0.5 (SMAD4 and CDKN2A) |
| Gao et al.[83] | 2021 | MRI | TP53 mutation | Radiomics | AUC 0.96 |
| Meng et al.[89] | 2021 | MRI | FAP expression | Radiomics | AUC 0.77 |
| Iwatate et al.[88] | 2020 | CECT | P53 and PD-L1 expression | Radiomics | AUC 0.795 and 0.683 |
| Qiu et al.[79] | 2019 | CECT | Histopathological grade | Radiomics | Accuracy 86% |
3.2.3. Prognosis
4. Discussion
5. Conclusion
Author Contributions
Funding
Conflicts of Interest
References
- R. L. Siegel, K. D. Miller, N. S. Wagle, and A. Jemal, “Cancer statistics, 2023,” CA Cancer J Clin, vol. 73, no. 1, pp. 17–48, Jan. 2023. [CrossRef]
- American Cancer Society, Facts & Figures 2023.
- B. Boursi et al., “A Clinical Prediction Model to Assess Risk for Pancreatic Cancer Among Patients With New-Onset Diabetes,” Gastroenterology, vol. 152, no. 4, pp. 840-850.e3, Mar. 2017. [CrossRef]
- A. P. Klein et al., “An Absolute Risk Model to Identify Individuals at Elevated Risk for Pancreatic Cancer in the General Population,” PLoS One, vol. 8, no. 9, p. e72311, Sep. 2013. [CrossRef]
- R. Pannala, A. Basu, G. M. Petersen, and S. T. Chari, “New-onset diabetes: a potential clue to the early diagnosis of pancreatic cancer,” Lancet Oncol, vol. 10, no. 1, pp. 88–95, Jan. 2009. [CrossRef]
- W. Muhammad et al., “Pancreatic Cancer Prediction Through an Artificial Neural Network,” Front Artif Intell, vol. 2, May 2019. [CrossRef]
- A. Adamska, A. Domenichini, and M. Falasca, “Pancreatic Ductal Adenocarcinoma: Current and Evolving Therapies,” Int J Mol Sci, vol. 18, no. 7, p. 1338, Jun. 2017. [CrossRef]
- S. Solomon, S. Das, R. Brand, and D. C. Whitcomb, “Inherited Pancreatic Cancer Syndromes,” The Cancer Journal, vol. 18, no. 6, pp. 485–491, Nov. 2012. [CrossRef]
- V. Rosato, J. Polesel, C. Bosetti, D. Serraino, E. Negri, and C. La Vecchia, “Population Attributable Risk for Pancreatic Cancer in Northern Italy,” Pancreas, vol. 44, no. 2, pp. 216–220, Mar. 2015. [CrossRef]
- V. Rosato, J. Polesel, C. Bosetti, D. Serraino, E. Negri, and C. La Vecchia, “Population Attributable Risk for Pancreatic Cancer in Northern Italy,” Pancreas, vol. 44, no. 2, pp. 216–220, Mar. 2015. [CrossRef]
- L. Marti-Bonmati et al., “Pancreatic cancer, radiomics and artificial intelligence,” British Journal of Radiology, vol. 95, no. 1137. British Institute of Radiology, Sep. 01, 2022. [CrossRef]
- B. H. Kann, A. Hosny, and H. J. W. L. Aerts, “Artificial intelligence for clinical oncology,” Cancer Cell, vol. 39, no. 7, pp. 916–927, Jul. 2021. [CrossRef]
- H. Hayashi et al., “Recent advances in artificial intelligence for pancreatic ductal adenocarcinoma,” World Journal of Gastroenterology, vol. 27, no. 43. Baishideng Publishing Group Inc, pp. 7480–7496, Nov. 21, 2021. [CrossRef]
- S. Do, K. D. Song, and J. W. Chung, “Basics of Deep Learning: A Radiologist’s Guide to Understanding Published Radiology Articles on Deep Learning,” Korean J Radiol, vol. 21, no. 1, p. 33, 2020. [CrossRef]
- V. Couteaux et al., “Kidney cortex segmentation in 2D CT with U-Nets ensemble aggregation,” Diagn Interv Imaging, vol. 100, no. 4, pp. 211–217, Apr. 2019. [CrossRef]
- P. Roca et al., “Artificial intelligence to predict clinical disability in patients with multiple sclerosis using FLAIR MRI,” Diagn Interv Imaging, vol. 101, no. 12, pp. 795–802, Dec. 2020. [CrossRef]
- S. Rastegar et al., “Radiomics for classification of bone mineral loss: A machine learning study,” Diagn Interv Imaging, vol. 101, no. 9, pp. 599–610, Sep. 2020. [CrossRef]
- S. S. F. Yip and H. J. W. L. Aerts, “Applications and limitations of radiomics,” Phys Med Biol, vol. 61, no. 13, pp. R150–R166, Jul. 2016. [CrossRef]
- R. J. Gillies, P. E. Kinahan, and H. Hricak, “Radiomics: Images Are More than Pictures, They Are Data,” Radiology, vol. 278, no. 2, pp. 563–577, Feb. 2016. [CrossRef]
- L. Shi et al., “Radiomics for Response and Outcome Assessment for Non-Small Cell Lung Cancer,” Technol Cancer Res Treat, vol. 17, p. 153303381878278, Jan. 2018. [CrossRef]
- X. Zheng et al., “Deep learning radiomics can predict axillary lymph node status in early-stage breast cancer,” Nat Commun, vol. 11, no. 1, p. 1236, Mar. 2020. [CrossRef]
- A. S. Tagliafico, M. Piana, D. Schenone, R. Lai, A. M. Massone, and N. Houssami, “Overview of radiomics in breast cancer diagnosis and prognostication,” The Breast, vol. 49, pp. 74–80, Feb. 2020. [CrossRef]
- Y. Huang et al., “Development and Validation of a Radiomics Nomogram for Preoperative Prediction of Lymph Node Metastasis in Colorectal Cancer,” Journal of Clinical Oncology, vol. 34, no. 18, pp. 2157–2164, Jun. 2016. [CrossRef]
- NCCN, “https://www.nccn.org/professionals/physician_gls/pdf/pancreatic.pdf,” Accesed on 10.12.2023.
- R. R. Almeida, G. C. Lo, M. Patino, B. Bizzo, R. Canellas, and D. V. Sahani, “Advances in Pancreatic CT Imaging,” American Journal of Roentgenology, vol. 211, no. 1, pp. 52–66, Jul. 2018. [CrossRef]
- H. Schoellnast et al., “Improvement of parenchymal and vascular enhancement using saline flush and power injection for multiple-detector-row abdominal CT,” Eur Radiol, vol. 14, no. 4, pp. 659–664, Apr. 2004. [CrossRef]
- Frank Wessels, Otto van Delden, and Robin Smithuis, “Pancreatic Cancer - CT staging 2.0,” https://radiologyassistant.nl/abdomen/pancreas/pancreas-carcinoma-1.
- H. S. Park, J. M. Lee, H. K. Choi, S. H. Hong, J. K. Han, and B. I. Choi, “Preoperative evaluation of pancreatic cancer: Comparison of gadolinium-enhanced dynamic MRI with MR cholangiopancreatography versus MDCT,” Journal of Magnetic Resonance Imaging, vol. 30, no. 3, pp. 586–595, Sep. 2009. [CrossRef]
- W. Schima and R. Függer, “Evaluation of focal pancreatic masses: comparison of mangafodipir-enhanced MR imaging and contrast-enhanced helical CT,” Eur Radiol, vol. 12, no. 12, pp. 2998–3008, Dec. 2002. [CrossRef]
- H. S. Park, J. M. Lee, H. K. Choi, S. H. Hong, J. K. Han, and B. I. Choi, “Preoperative evaluation of pancreatic cancer: Comparison of gadolinium-enhanced dynamic MRI with MR cholangiopancreatography versus MDCT,” Journal of Magnetic Resonance Imaging, vol. 30, no. 3, pp. 586–595, Sep. 2009. [CrossRef]
- N. Horvat, D. E. Ryan, M. D. LaGratta, P. M. Shah, and R. K. Do, “Imaging for pancreatic ductal adenocarcinoma,” Chin Clin Oncol, vol. 6, no. 6, pp. 62–62, Dec. 2017. [CrossRef]
- F.-M. Chen, J.-M. Ni, Z.-Y. Zhang, L. Zhang, B. Li, and C.-J. Jiang, “Presurgical Evaluation of Pancreatic Cancer: A Comprehensive Imaging Comparison of CT Versus MRI,” American Journal of Roentgenology, vol. 206, no. 3, pp. 526–535, Mar. 2016. [CrossRef]
- S. Rickes, K. Unkrodt, H. Neye, K. W. Ocran, and W. Wermke, “Differentiation of Pancreatic Tumours by Conventional Ultrasound, Unenhanced and Echo-Enhanced Power Doppler Sonography,” Scand J Gastroenterol, vol. 37, no. 11, pp. 1313–1320, Jan. 2002. [CrossRef]
- E. S. Lee, “Imaging diagnosis of pancreatic cancer: A state-of-the-art review,” World J Gastroenterol, vol. 20, no. 24, p. 7864, 2014. [CrossRef]
- M. Dbouk et al., “The Multicenter Cancer of Pancreas Screening Study: Impact on Stage and Survival,” Journal of Clinical Oncology, vol. 40, no. 28, pp. 3257–3266, Oct. 2022. [CrossRef]
- M. Ramaekers et al., “Computer-Aided Detection for Pancreatic Cancer Diagnosis: Radiological Challenges and Future Directions,” J Clin Med, vol. 12, no. 13, p. 4209, Jun. 2023. [CrossRef]
- K. M. Jang, S. H. Kim, Y. K. Kim, K. D. Song, S. J. Lee, and D. Choi, “Missed pancreatic ductal adenocarcinoma: Assessment of early imaging findings on prediagnostic magnetic resonance imaging,” Eur J Radiol, vol. 84, no. 8, pp. 1473–1479, Aug. 2015. [CrossRef]
- K. Cao et al., “Large-scale pancreatic cancer detection via non-contrast CT and deep learning,” Nat Med, Nov. 2023. [CrossRef]
- P. Korfiatis et al., “Automated Artificial Intelligence Model Trained on a Large Data Set Can Detect Pancreas Cancer on Diagnostic Computed Tomography Scans As Well As Visually Occult Preinvasive Cancer on Prediagnostic Computed Tomography Scans,” Gastroenterology, vol. 165, no. 6, pp. 1533-1546.e4, Dec. 2023. [CrossRef]
- L. C. Chu et al., “Application of Deep Learning to Pancreatic Cancer Detection: Lessons Learned From Our Initial Experience,” Journal of the American College of Radiology, vol. 16, no. 9, pp. 1338–1342, Sep. 2019. [CrossRef]
- L. C. Chu et al., “Utility of CT Radiomics Features in Differentiation of Pancreatic Ductal Adenocarcinoma From Normal Pancreatic Tissue,” American Journal of Roentgenology, vol. 213, no. 2, pp. 349–357, Aug. 2019. [CrossRef]
- L. C. Chu et al., “Diagnostic performance of commercially available vs. in-house radiomics software in classification of CT images from patients with pancreatic ductal adenocarcinoma vs. healthy controls,” Abdominal Radiology, vol. 45, no. 8, pp. 2469–2475, Aug. 2020. [CrossRef]
- H. Ma et al., “Construction of a convolutional neural network classifier developed by computed tomography images for pancreatic cancer diagnosis,” World J Gastroenterol, vol. 26, no. 34, pp. 5156–5168, Sep. 2020. [CrossRef]
- N. Alves, M. Schuurmans, G. Litjens, J. S. Bosma, J. Hermans, and H. Huisman, “Fully Automatic Deep Learning Framework for Pancreatic Ductal Adenocarcinoma Detection on Computed Tomography,” Cancers (Basel), vol. 14, no. 2, p. 376, Jan. 2022. [CrossRef]
- S. Mukherjee et al., “Radiomics-based Machine-learning Models Can Detect Pancreatic Cancer on Prediagnostic Computed Tomography Scans at a Substantial Lead Time Before Clinical Diagnosis,” Gastroenterology, vol. 163, no. 5, pp. 1435-1446.e3, Nov. 2022. [CrossRef]
- S. Wang et al., “Compute Tomography Radiomics Analysis on Whole Pancreas Between Healthy Individual and Pancreatic Ductal Adenocarcinoma Patients: Uncertainty Analysis and Predictive Modeling,” Technol Cancer Res Treat, vol. 21, 2022. [CrossRef]
- P.-T. Chen et al., “Radiomic Features at CT Can Distinguish Pancreatic Cancer from Noncancerous Pancreas,” Radiol Imaging Cancer, vol. 3, no. 4, p. e210010, Jul. 2021. [CrossRef]
- L. C. Chu et al., “Utility of CT Radiomics Features in Differentiation of Pancreatic Ductal Adenocarcinoma From Normal Pancreatic Tissue,” American Journal of Roentgenology, vol. 213, no. 2, pp. 349–357, Aug. 2019. [CrossRef]
- C. G. A. Viviers et al., “Improved Pancreatic Tumor Detection by Utilizing Clinically-Relevant Secondary Features,” Aug. 2022, [Online]. Available: http://arxiv.org/abs/2208.03581.
- E. M. Weisberg et al., “Deep lessons learned: Radiology, oncology, pathology, and computer science experts unite around artificial intelligence to strive for earlier pancreatic cancer diagnosis,” Diagn Interv Imaging, vol. 101, no. 2, pp. 111–115, Feb. 2020. [CrossRef]
- A. Farag, L. Lu, H. R. Roth, J. Liu, E. Turkbey, and R. M. Summers, “A Bottom-Up Approach for Pancreas Segmentation Using Cascaded Superpixels and (Deep) Image Patch Labeling,” IEEE Transactions on Image Processing, vol. 26, no. 1, pp. 386–399, Jan. 2017. [CrossRef]
- Y. Zhou et al., “Hyper-Pairing Network for Multi-phase Pancreatic Ductal Adenocarcinoma Segmentation,” 2019, pp. 155–163. [CrossRef]
- Z. Zhu, Y. Xia, L. Xie, E. K. Fishman, and A. L. Yuille, “Multi-Scale Coarse-to-Fine Segmentation for Screening Pancreatic Ductal Adenocarcinoma,” Jul. 2018.
- M. Barat et al., “Artificial intelligence: a critical review of current applications in pancreatic imaging,” Japanese Journal of Radiology, vol. 39, no. 6. Springer Japan, pp. 514–523, Jun. 01, 2021. [CrossRef]
- L.-C. Chen, G. Papandreou, I. Kokkinos, K. Murphy, and A. L. Yuille, “DeepLab: Semantic Image Segmentation with Deep Convolutional Nets, Atrous Convolution, and Fully Connected CRFs,” IEEE Trans Pattern Anal Mach Intell, vol. 40, no. 4, pp. 834–848, Apr. 2018. [CrossRef]
- F. Chollet, “Xception: Deep Learning with Depthwise Separable Convolutions,” in 2017 IEEE Conference on Computer Vision and Pattern Recognition (CVPR), IEEE, Jul. 2017, pp. 1800–1807. [CrossRef]
- H. Ni et al., “Predicting Recurrence in Pancreatic Ductal Adenocarcinoma after Radical Surgery Using an AX-Unet Pancreas Segmentation Model and Dynamic Nomogram,” Bioengineering, vol. 10, no. 7, Jul. 2023. [CrossRef]
- A. Turečková, T. Tureček, Z. Komínková Oplatková, and A. Rodríguez-Sánchez, “Improving CT Image Tumor Segmentation Through Deep Supervision and Attentional Gates,” Front Robot AI, vol. 7, Aug. 2020. [CrossRef]
- A. L. Simpson et al., “A large annotated medical image dataset for the development and evaluation of segmentation algorithms,” Feb. 2019.
- F. Isensee et al., “nnU-Net: Self-adapting Framework for U-Net-Based Medical Image Segmentation,” Sep. 2018.
- Y. Zhou et al., “Hyper-Pairing Network for Multi-phase Pancreatic Ductal Adenocarcinoma Segmentation,” 2019, pp. 155–163. [CrossRef]
- T. Mahmoudi et al., “Segmentation of pancreatic ductal adenocarcinoma (PDAC) and surrounding vessels in CT images using deep convolutional neural networks and texture descriptors,” Sci Rep, vol. 12, no. 1, p. 3092, Feb. 2022. [CrossRef]
- C. Shen et al., “A cascaded fully convolutional network framework for dilated pancreatic duct segmentation,” Int J Comput Assist Radiol Surg, vol. 17, no. 2, pp. 343–354, Feb. 2022. [CrossRef]
- J. Lu, N. Jiang, Y. Zhang, and D. Li, “A CT based radiomics nomogram for differentiation between focal-type autoimmune pancreatitis and pancreatic ductal adenocarcinoma,” Front Oncol, vol. 13, Mar. 2023. [CrossRef]
- S. Park et al., “Differentiating autoimmune pancreatitis from pancreatic ductal adenocarcinoma with CT radiomics features,” Diagn Interv Imaging, vol. 101, no. 9, pp. 555–564, Sep. 2020. [CrossRef]
- K. Anai et al., “The effect of CT texture-based analysis using machine learning approaches on radiologists’ performance in differentiating focal-type autoimmune pancreatitis and pancreatic duct carcinoma,” Jpn J Radiol, vol. 40, no. 11, pp. 1156–1165, Nov. 2022. [CrossRef]
- Y. Deng et al., “Radiomics Model Based on MR Images to Discriminate Pancreatic Ductal Adenocarcinoma and Mass-Forming Chronic Pancreatitis Lesions,” Front Oncol, vol. 11, Mar. 2021. [CrossRef]
- S. Ren et al., “Computed Tomography-Based Radiomics Signature for the Preoperative Differentiation of Pancreatic Adenosquamous Carcinoma From Pancreatic Ductal Adenocarcinoma,” Front Oncol, vol. 10, Aug. 2020. [CrossRef]
- Y. J. Shi et al., “Histogram array and convolutional neural network of DWI for differentiating pancreatic ductal adenocarcinomas from solid pseudopapillary neoplasms and neuroendocrine neoplasms,” Clin Imaging, vol. 96, pp. 15–22, Apr. 2023. [CrossRef]
- G. Younan, “Pancreas Solid Tumors,” Surgical Clinics of North America, vol. 100, no. 3, pp. 565–580, Jun. 2020. [CrossRef]
- T. Zhang et al., “Radiomics Combined with Multiple Machine Learning Algorithms in Differentiating Pancreatic Ductal Adenocarcinoma from Pancreatic Neuroendocrine Tumor: More Hands Produce a Stronger Flame,” J Clin Med, vol. 11, no. 22, Nov. 2022. [CrossRef]
- N. Macías et al., “Histologic Tumor Grade and Preoperative Bilary Drainage are the Unique Independent Prognostic Factors of Survival in Pancreatic Ductal Adenocarcinoma Patients After Pancreaticoduodenectomy,” J Clin Gastroenterol, vol. 52, no. 2, pp. e11–e17, Feb. 2018. [CrossRef]
- K. F. D. Kuhlmann et al., “Surgical treatment of pancreatic adenocarcinoma,” Eur J Cancer, vol. 40, no. 4, pp. 549–558, Mar. 2004. [CrossRef]
- S. H. Han et al., “Actual long-term outcome of T1 and T2 pancreatic ductal adenocarcinoma after surgical resection,” International Journal of Surgery, vol. 40, pp. 68–72, Apr. 2017. [CrossRef]
- M. Schuurmans, N. Alves, P. Vendittelli, H. Huisman, and J. Hermans, “Setting the Research Agenda for Clinical Artificial Intelligence in Pancreatic Adenocarcinoma Imaging,” Cancers (Basel), vol. 14, no. 14, p. 3498, Jul. 2022. [CrossRef]
- H. Fu et al., “Automatic Pancreatic Ductal Adenocarcinoma Detection in Whole Slide Images Using Deep Convolutional Neural Networks,” Front Oncol, vol. 11, Jun. 2021. [CrossRef]
- P. Sántha et al., “Morphological Heterogeneity in Pancreatic Cancer Reflects Structural and Functional Divergence,” Cancers (Basel), vol. 13, no. 4, p. 895, Feb. 2021. [CrossRef]
- V. Baxi, R. Edwards, M. Montalto, and S. Saha, “Digital pathology and artificial intelligence in translational medicine and clinical practice,” Modern Pathology, vol. 35, no. 1, pp. 23–32, Jan. 2022. [CrossRef]
- W. Qiu et al., “Pancreatic ductal adenocarcinoma: Machine learning–based quantitative computed tomography texture analysis for prediction of histopathological grade,” Cancer Manag Res, vol. 11, pp. 9253–9264, 2019. [CrossRef]
- C. Cen et al., “Clinical-radiomics nomogram using contrast-enhanced CT to predict histological grade and survival in pancreatic ductal adenocarcinoma,” Front Oncol, vol. 13, 2023. [CrossRef]
- A. Makohon-Moore and C. A. Iacobuzio-Donahue, “Pancreatic cancer biology and genetics from an evolutionary perspective,” Nat Rev Cancer, vol. 16, no. 9, pp. 553–565, Sep. 2016. [CrossRef]
- R. Hinzpeter et al., “CT Radiomics and Whole Genome Sequencing in Patients with Pancreatic Ductal Adenocarcinoma: Predictive Radiogenomics Modeling,” Cancers (Basel), vol. 14, no. 24, Dec. 2022. [CrossRef]
- J. Gao et al., “Differentiating TP53 Mutation Status in Pancreatic Ductal Adenocarcinoma Using Multiparametric MRI-Derived Radiomics,” Front Oncol, vol. 11, May 2021. [CrossRef]
- P. Chaturvedi et al., “MUC4 Mucin Potentiates Pancreatic Tumor Cell Proliferation, Survival, and Invasive Properties and Interferes with Its Interaction to Extracellular Matrix Proteins,” Molecular Cancer Research, vol. 5, no. 4, pp. 309–320, Apr. 2007. [CrossRef]
- M. Mimeault, S. L. Johansson, S. Senapati, N. Momi, S. Chakraborty, and S. K. Batra, “MUC4 down-regulation reverses chemoresistance of pancreatic cancer stem/progenitor cells and their progenies,” Cancer Lett, vol. 295, no. 1, pp. 69–84, Sep. 2010. [CrossRef]
- M. Saitou, “MUC4 expression is a novel prognostic factor in patients with invasive ductal carcinoma of the pancreas,” J Clin Pathol, vol. 58, no. 8, pp. 845–852, Aug. 2005. [CrossRef]
- Y. Deng et al., “Radiomics models based on multi-sequence MRI for preoperative evaluation of MUC4 status in pancreatic ductal adenocarcinoma: a preliminary study,” Quant Imaging Med Surg, vol. 12, no. 11, pp. 5129–5139, Nov. 2022. [CrossRef]
- Y. Iwatate et al., “Radiogenomics for predicting p53 status, PD-L1 expression, and prognosis with machine learning in pancreatic cancer,” Br J Cancer, vol. 123, no. 8, pp. 1253–1261, Oct. 2020. [CrossRef]
- Y. Meng et al., “Noncontrast Magnetic Resonance Radiomics and Multilayer Perceptron Network Classifier: An approach for Predicting Fibroblast Activation Protein Expression in Patients With Pancreatic Ductal Adenocarcinoma,” Journal of Magnetic Resonance Imaging, vol. 54, no. 5, pp. 1432–1443, Nov. 2021. [CrossRef]
- I. Vezakis et al., “An Automated Prognostic Model for Pancreatic Ductal Adenocarcinoma,” Genes (Basel), vol. 14, no. 9, Sep. 2023. [CrossRef]
- X. Xu, J. Qu, Y. Zhang, X. Qian, T. Chen, and Y. Liu, “Development and validation of an MRI-radiomics nomogram for the prognosis of pancreatic ductal adenocarcinoma,” Front Oncol, vol. 13, 2023. [CrossRef]
- H. Qiu et al., “Original Article A novel preoperative MRI-based radiomics nomogram outperforms traditional models for prognostic prediction in pancreatic ductal adenocarcinoma,” 2022. [Online]. Available: www.ajcr.us/.
- X. Li et al., “Preoperative recurrence prediction in pancreatic ductal adenocarcinoma after radical resection using radiomics of diagnostic computed tomography,” EClinicalMedicine, vol. 43, Jan. 2022. [CrossRef]
- T. Xie, X. Wang, M. Li, T. Tong, X. Yu, and Z. Zhou, “Pancreatic ductal adenocarcinoma: a radiomics nomogram outperforms clinical model and TNM staging for survival estimation after curative resection,” Eur Radiol, vol. 30, no. 5, pp. 2513–2524, May 2020. [CrossRef]
- S. Park et al., “CT Radiomics–Based Preoperative Survival Prediction in Patients With Pancreatic Ductal Adenocarcinoma,” American Journal of Roentgenology, vol. 217, no. 5, pp. 1104–1112, Nov. 2021. [CrossRef]
- F. Chen et al., “Radiomics-Assisted Presurgical Prediction for Surgical Portal Vein-Superior Mesenteric Vein Invasion in Pancreatic Ductal Adenocarcinoma,” Front Oncol, vol. 10, Nov. 2020. [CrossRef]
- J. K. Jang et al., “CT-determined resectability of borderline resectable and unresectable pancreatic adenocarcinoma following FOLFIRINOX therapy,” Eur Radiol, vol. 31, no. 2, pp. 813–823, Feb. 2021. [CrossRef]
- M. Zins, C. Matos, and C. Cassinotto, “Pancreatic Adenocarcinoma Staging in the Era of Preoperative Chemotherapy and Radiation Therapy,” Radiology, vol. 287, no. 2, pp. 374–390, May 2018. [CrossRef]
- L. Shi, L. Wang, C. Wu, Y. Wei, Y. Zhang, and J. Chen, “Preoperative Prediction of Lymph Node Metastasis of Pancreatic Ductal Adenocarcinoma Based on a Radiomics Nomogram of Dual-Parametric MRI Imaging,” Front Oncol, vol. 12, Jul. 2022. [CrossRef]
- Y. Bian et al., “Radiomics nomogram for the preoperative prediction of lymph node metastasis in pancreatic ductal adenocarcinoma,” Cancer Imaging, vol. 22, no. 1, Dec. 2022. [CrossRef]
- J. Chang et al., “Machine-learning based investigation of prognostic indicators for oncological outcome of pancreatic ductal adenocarcinoma,” Front Oncol, vol. 12, Dec. 2022. [CrossRef]
| Resectable | Borderline | Locally Advanced |
|---|---|---|
|
Arterial No contact |
Head/uncinate process: | Head/uncinate process: |
| Tumor contact with common hepatic artery without extension to celiac artery (CA) or hepatic artery bifurcation. Tumor contact with SMA ≤180°. Tumor contact with variant arterial anatomy. |
>180° SMA or CA | |
|
Body/tail: Tumor contact with the CA ≤180° |
Body/tail: >180° SMA or CA or ≤180° CA and aortic involvement |
|
|
Venous ≤180° without contour irregularity |
>180° or with contour irregularity/thrombosis resection & reconstruction possible. Tumor contact with IVC |
Unreconstructible SMV/PV due to tumor involvement or occlusion (can be due to tumor or bland thrombus) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




