Submitted:
08 January 2024
Posted:
11 January 2024
Read the latest preprint version here
Abstract
Keywords:
1. Introduction
2. Materials and Methods
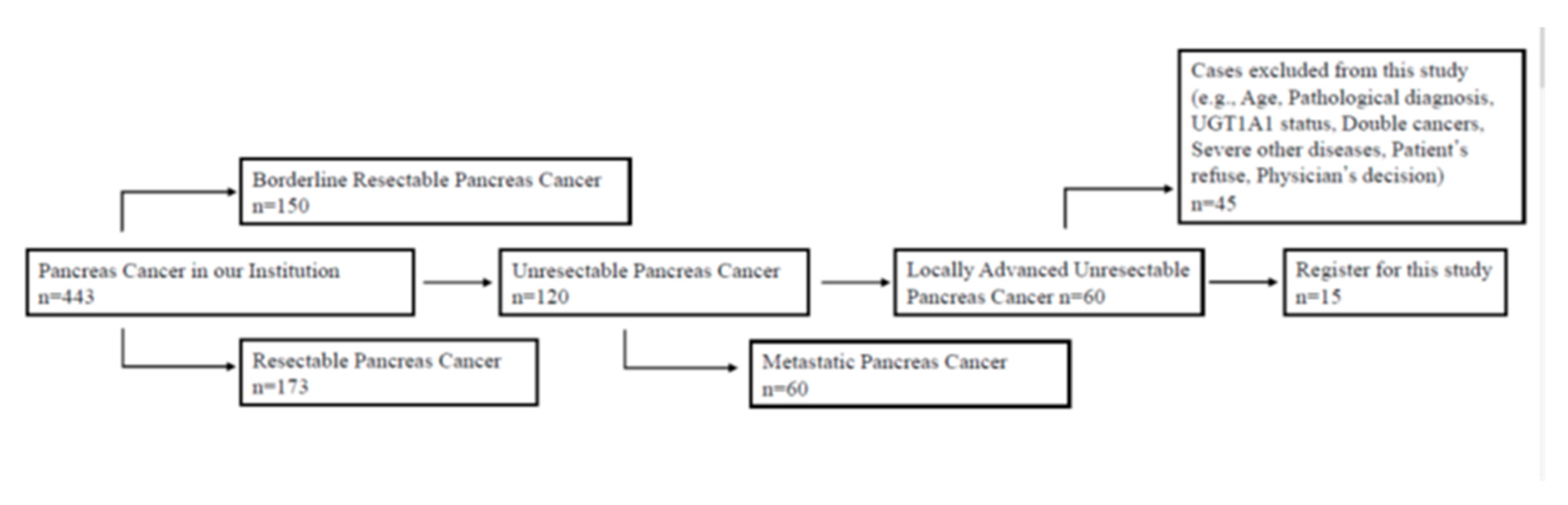
2.1. Study Design
2.2. Inclusion Criteria
2.3. Exclusion Criteria
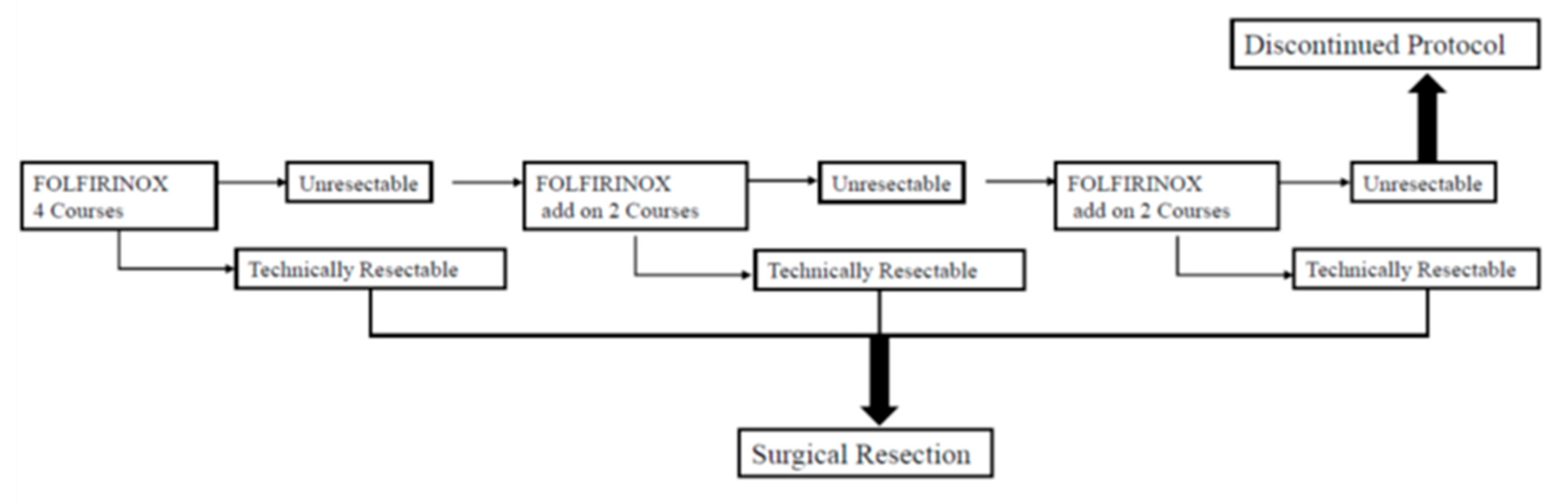
2.4. Treatment Protocol
2.5. Decision for conversion surgery
2.6. Statistical Analysis
3. Results
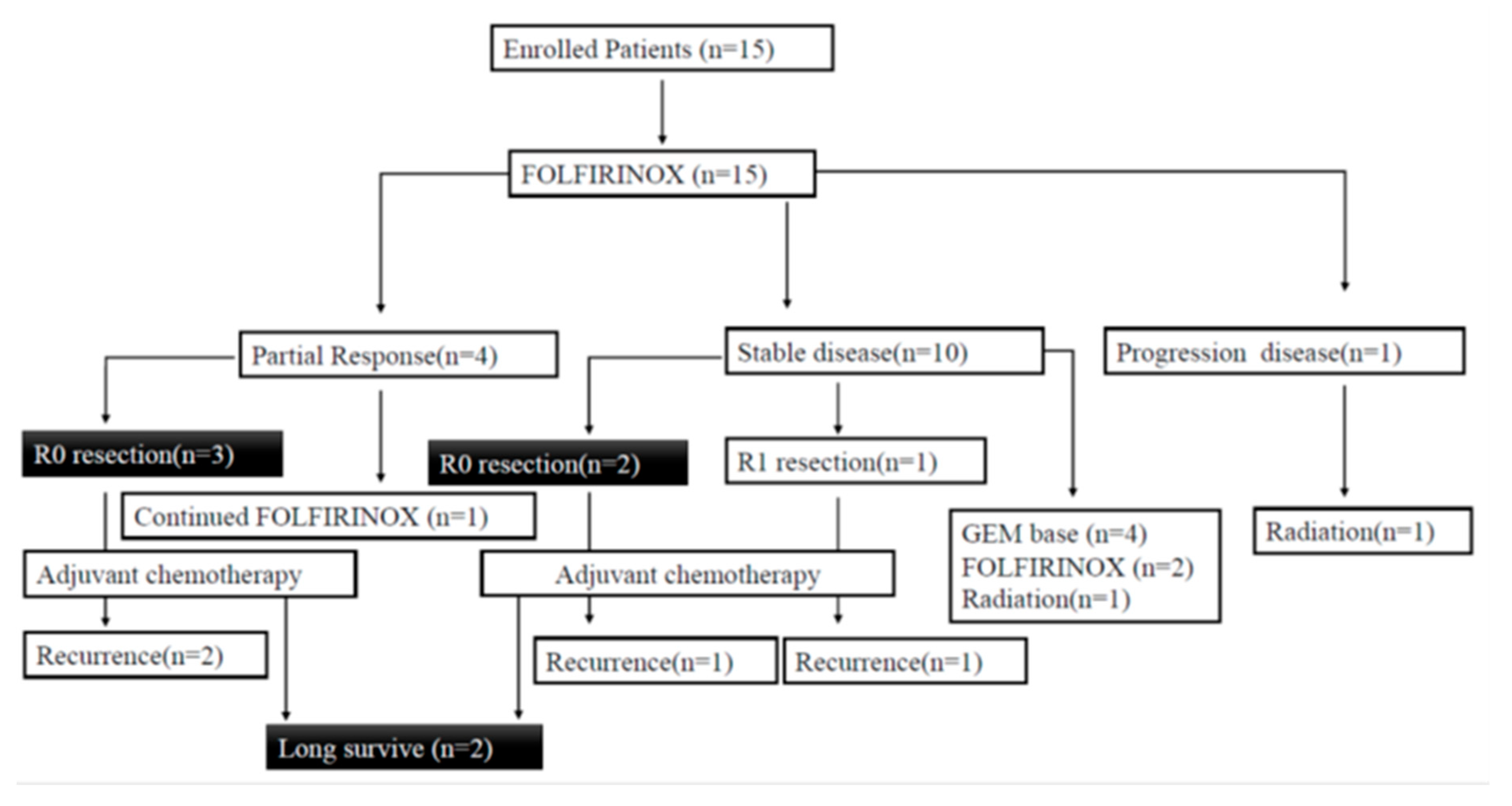
3.1. Patients
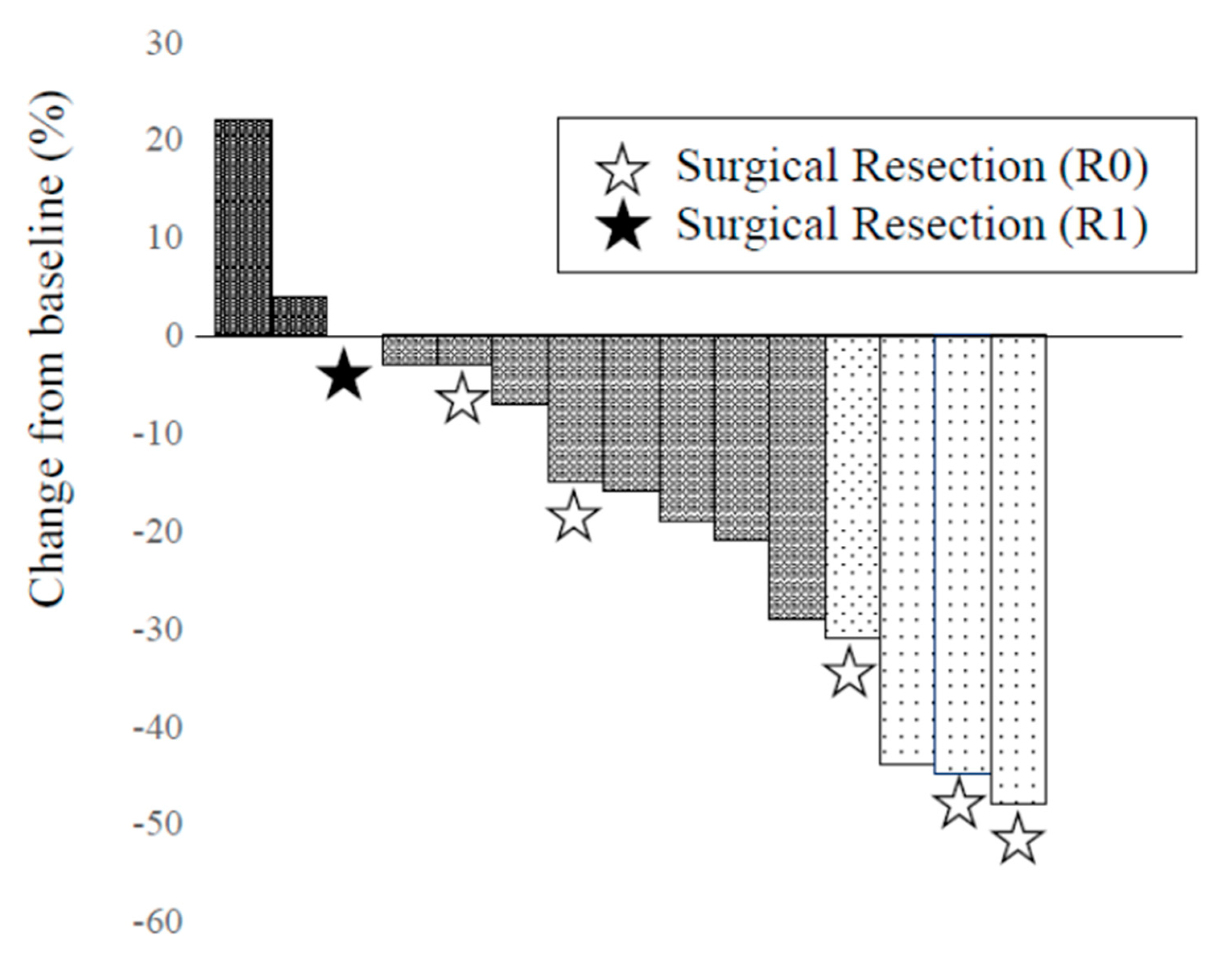
3.2. Efficacy of Chemotherapy
3.3. Toxicity of Chemotherapy
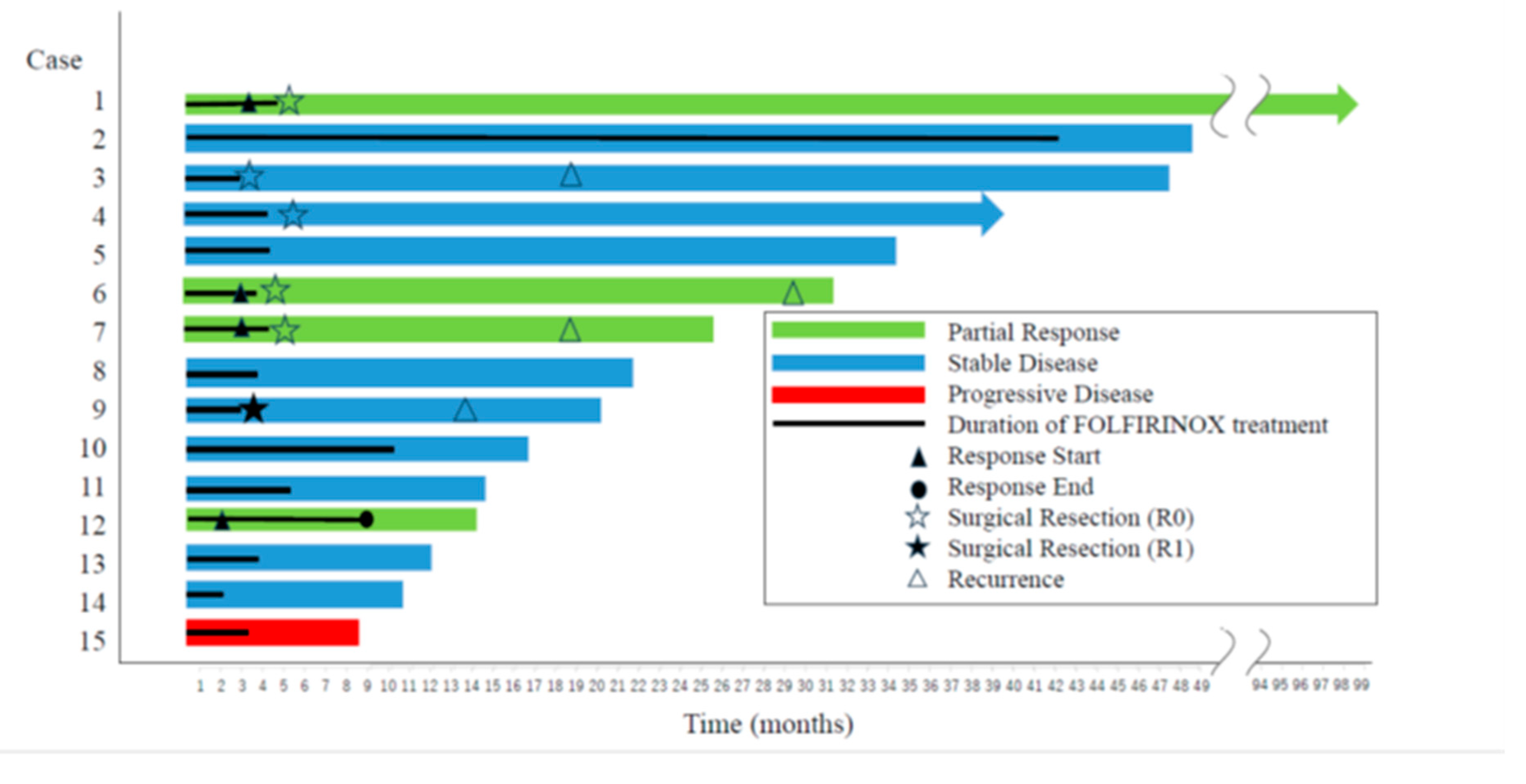
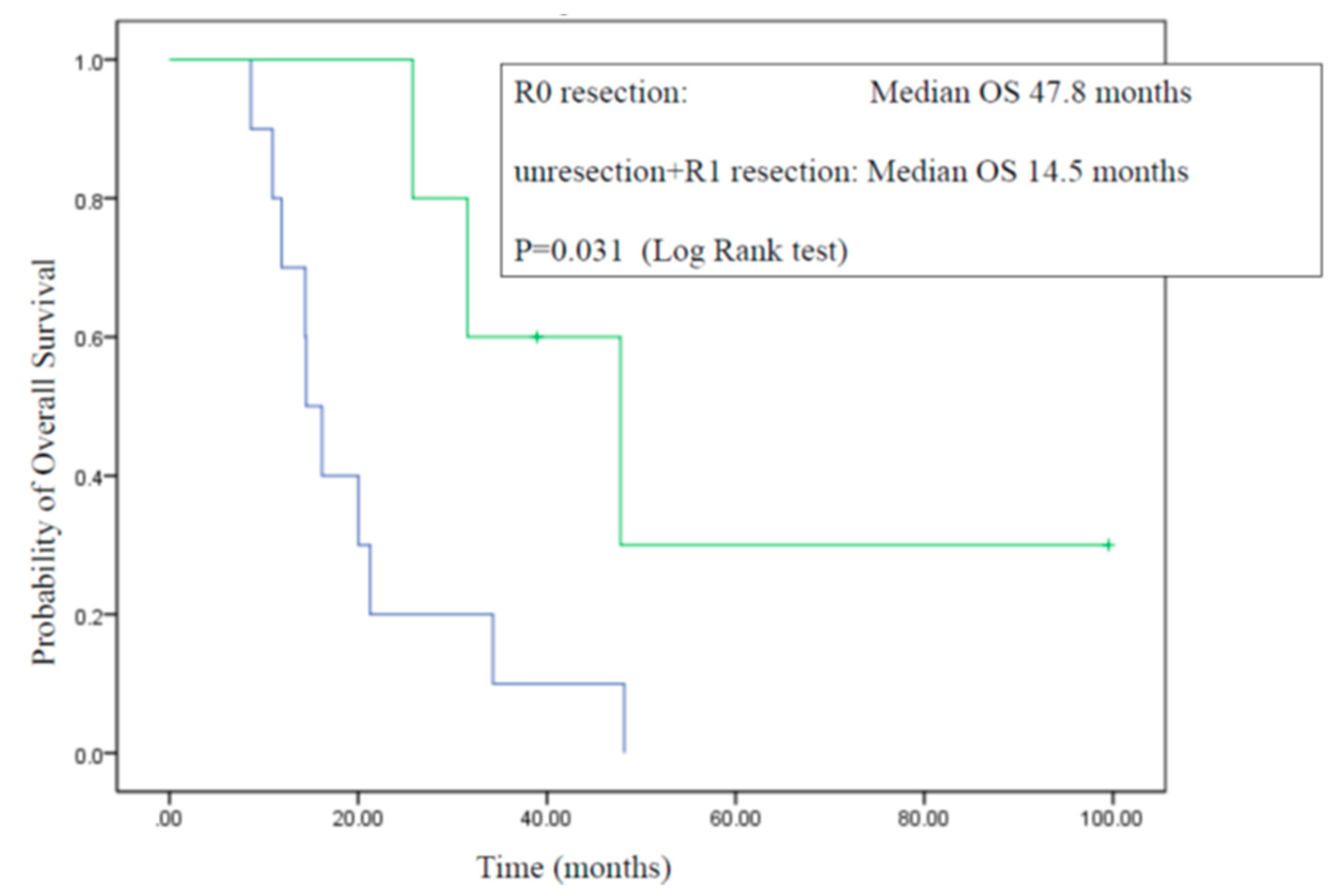
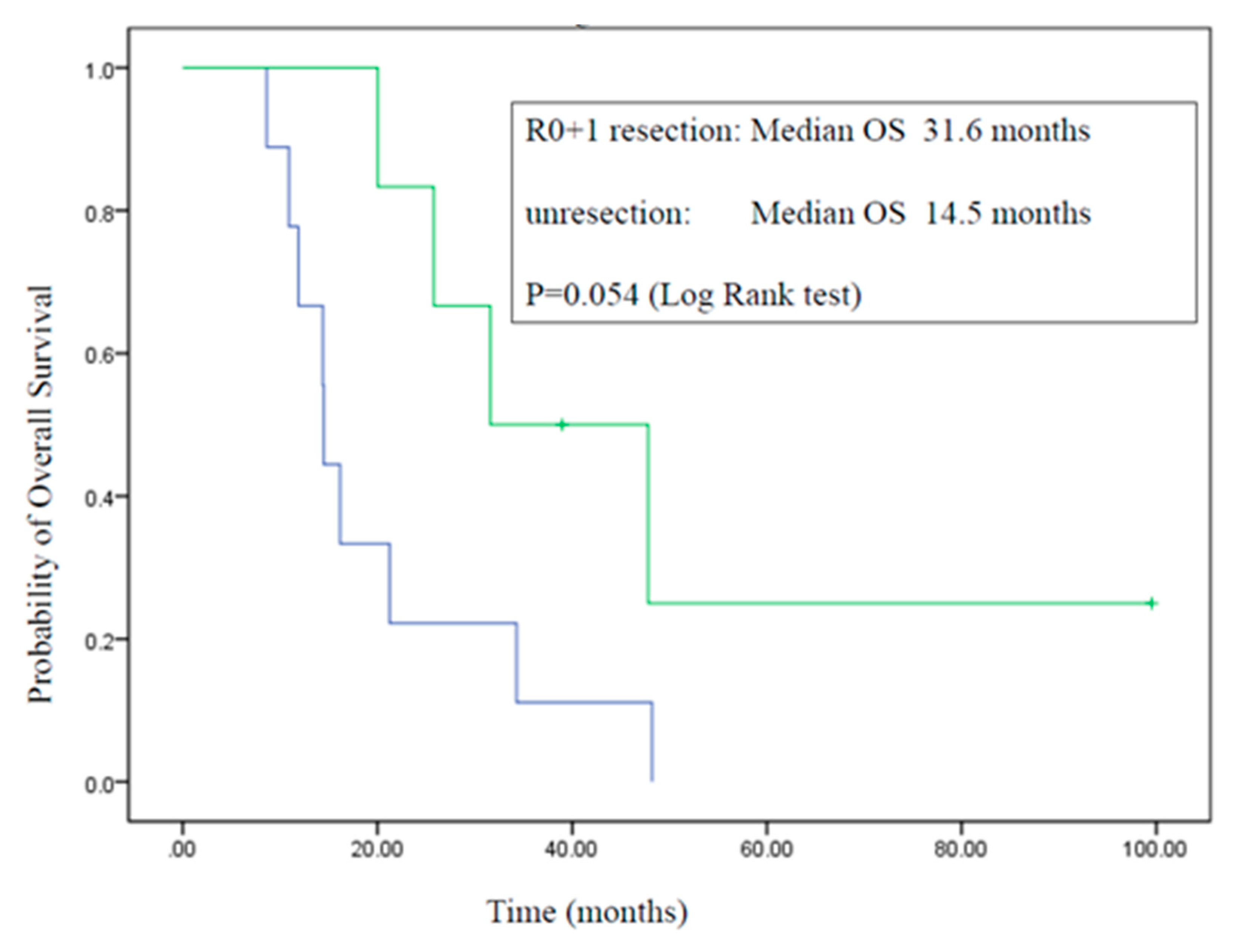
3.4. Surgery
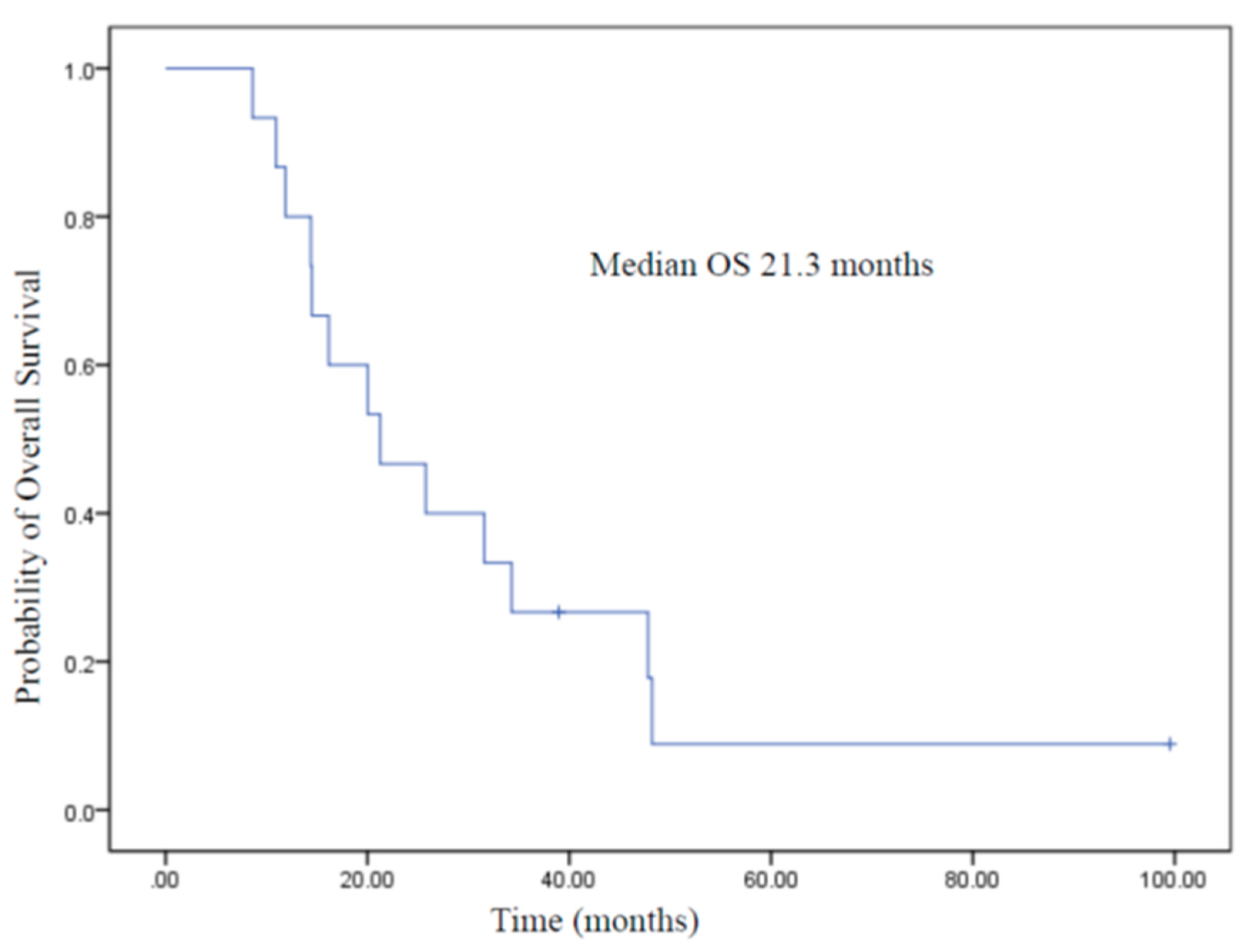
3.5. Overall Survival
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Siegel, R.L.; Miller, K.D.; Fuchs, H.E.; Jemal, A. Cancer statistics, 2021. CA Cancer J Clin 2021, 71, 7–33. [Google Scholar] [CrossRef] [PubMed]
- Rahib, L.; Smith, B.D.; Aizenberg, R.; Rosenzweig, A.B.; Fleshman, J.M.; Matrisian, L.M. Projecting cancer incidence and deaths to 2030: the unexpected burden of thyroid, liver, and pancreas cancers in the United States. Cancer Res 2014, 74, 2913–2921. [Google Scholar] [CrossRef] [PubMed]
- Gillen, A.S.; Schuster, T.; Meyer Zum Büschenfelde, C.; Friess, H.; Kleeff, J. Preoperative/neoadjuvant therapy in pancreatic cancer: a systematic review and meta-analysis of response and resection percentages. PLoS Med, 2010; 7, e1000267. [Google Scholar]
- Vauthey, J.N.; Ahpba, D.E.; SSO/SSAT. AHPBA/SSO/SSAT consensus conference on research and borderline research pancreatic cancer: A rationale and overview of the conference. Ann Surg Oncol Consensus Conference on Research and Borderline Research Pancreatic Cancer: Rationale and Overview of the Conference. Ann Surg Oncol 2009, 16, 1725–1726. [Google Scholar] [CrossRef] [PubMed]
- Allen, P.J.; Kuk, D.; Castillo, C. F-D.; Basturk, O.; Wolfgang, C.L.; Cameron, J.L.; Lillemoe, K.D.; Ferrone, C.R.; Morales-Oyarvide, V.; He, J.; Weiss, M.J.; Hruban, R.H.; Gönen, M.; Klimstra, D.S.; Mino-Kenudson, M. Multi-institutional Validation Study of the American Joint Commission on Cancer (8th edition): Changes for T and N Staging in Patients With Pancreatic Adenocarcinoma. Ann Surg 2017, 265, 185–191. [Google Scholar] [CrossRef] [PubMed]
- Gilbert, J. W.; Wolpin, B.; Clancy, T.; Wang, J.; Mamon, H.; Shinagare, A. B.; Jagannathan, J.; Rosenthal, M. Borderline resectable pancreatic cancer: conceptual evolution and current approach to image-based classification. Ann Oncol 2017, 28, 2067–2076. [Google Scholar] [CrossRef] [PubMed]
- Sohal, D.P.S.; Duong, M.; Ahmad, S.A.; Gandhi, N.S.; Beg, M.S.; Wang-Gillam, A.; Wade, J.L.; Chiorean, E.G.; Guthrie, K.A.; Lowy, A.M.; Philip, P.A.; Hochster, H.S. Efficacy of perioperative chemotherapy for resectable pancreatic adenocarcinoma: a phase 2 randomized clinical trial. JAMA Oncol 2021, 7, 421–427. [Google Scholar] [CrossRef] [PubMed]
- Murphy, J.E.; Wo, J.Y.; Ryan, D.P.; Jiang, W.; Yeap, B.Y.; Drapek, L.C.; Blaszkowsky, L.S.; Kwak, E.L.; Allen, J.N.; Clark, J.W.; Faris, J.E.; Zhu, A.X.; Goyal, L.; Lillemoe, K.D.; DeLaney, T.F.; Castillo, C. F-D.; Ferrone, C.R.; Hong, T.S. Total neoadjuvant therapy with folfirinox followed by individualized chemoradiotherapy for borderline resectable pancreatic adenocarcinoma: A phase 2 clinical trial. JAMA Oncol 2018, 4, 963–969. [Google Scholar] [CrossRef]
- Chawla, A.; Molina, G.; Pak, L.M.; Rosenthal, M.; Mancias, J.D.; Clancy, T.E.; Wolpin, B.M.; Wang, J. Neoadjuvant therapy is associated with improved survival in borderline resectable pancreatic cancer. Ann Surg Oncol 2020, 27, 1191–1200. [Google Scholar] [CrossRef]
- Jang, J.Y.; Han, Y.; Lee, H.; Kim, S.W.; Kwon, W.; Lee, K.H.; Oh, D.Y.; Chie, E.K.; Lee, J.M.; Heo, J.S.; Park, J.O.; Lim, D.H.; Kim, S.H.; Park, S.J.; Lee, W.J.; Koh, Y.H.; Park, J.S.; Yoon, D.S.; Lee, I.J.; Choi, S.H. Oncological benefits of neoadjuvant chemoradiation with gemcitabine versus upfront surgery in patients with borderline resectable pancreatic cancer: a prospective, randomized, open-label, multicenter phase 2/3 trial. Ann Surg 2018, 268, 215–222. [Google Scholar] [CrossRef]
- Sultana, A.; Tudur Smith, C.; Cunningham, D.; Starling, N.; Tait, D.; Neoptolemos, J.P.; Ghaneh, P. Systematic review, including meta-analyses on the management of locally advanced pancreatic cancer using radiation/combined modality therapy. Br J Cancer 2007, 96, 1183–1190. [Google Scholar] [CrossRef]
- Suker, M.; Beumer, B.R.; Sadot, E.; Marthey, L.; Faris, J.E.; Mellon, E.A.; El-Rayes, B.F.; Wang-Gillam, A.; Lacy, J.; Hosein, P.J.; Moorcraft, S.Y.; Conroy, T.; Hohla, F.; Allen, P.; Taieb, J.; Hong, T.S.; Shridhar, R.; Chau, I.; van Eijck, C.H.; Koerkamp, B.G. FOLFIRINOX therapy for locally advanced pancreatic cancer: A systematic review and patient-level meta-analysis. Lancet Oncol 2016, 17, 801–810. [Google Scholar] [CrossRef]
- Sadot, E.; Doussot, A.; O’Reilly, E.M.; Lowery, M.A.; Goodman, K.A.; Do, R.K.; Tang, L.H.; Gönen, M.; D’Angelica, M.I.; DeMatteo, R.P.; Kingham, T.P.; Jarnagin, W.R.; Allen, P.J. FOLFIRINOX induction therapy for stage 3 pancreatic adenocarcinoma. Ann Surg Oncol 2015, 22, 3512–3521. [Google Scholar] [CrossRef]
- NCCN Clinical Guideline, 2021.
- Cardenes, H.R.; Moore, A.M.; Johnson, C.S.; Yu, M.; Helft, P.; Chiorean, E.G.; Vinson, J.; Howard, T.J.; Stephens, A.W.; Tai, D.F.; Loehrer Sr, P.J. Phase II study of gemcitabine in combination with radiation therapy in patients with localized, unresectable pancreatic cancer: A Hoosier Oncology Group study. Am J Clin Oncol 2011, 34, 460–465. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.L.; Kim, S.C.; Kim, J.H.; Lee, S.S.; Kim, T.W.; Park, D.H.; Seo, D.W.; Lee, S.K.; Kim, M.H.; Kim, J.H.; Park, J.H.; Shin, S.H.; Han, D.J. Prospective efficacy and safety of neoadjuvant gemcitabine and capecitabine combination chemotherapy for borderline resectable or unresectable locally advanced pancreatic adenocarcinoma. Surgery 2012, 152, 851–862. [Google Scholar] [CrossRef]
- Strobel, O.; Berens, V.; Hinz, U.; Hartwig, W.; Hackert, T.; Bergmann, F.; Debus, J.; Jäger, D.; Büchler, M.W.; Werner, J. Resection after neoadjuvant therapy for locally advanced, “unresectable” pancreatic cancer. Surgery 2012, 152 (suppl 1), S33–42. [Google Scholar] [CrossRef] [PubMed]
- Arvold, N.D.; Ryan, D.P.; Niemierko, A.; Blaszkowsky, L.S.; Kwak, E.L.; Wo, J.Y.; Allen, J.N.; Clark, J.W.; Wadlow, R.C.; Zhu, A.X.; Castillo, C. F-D.; Hong, T.S. Long-term outcomes of neoadjuvant chemotherapy before chemoradiation for locally advanced pancreatic cancer. Cancer 2012, 118, 3026–3035. [Google Scholar] [CrossRef]
- Leone, F.; Gatti, M.; Massucco, P.; Colombi, F.; Sperti, E.; Campanella, D.; Regge, D.; Gabriele, P.; Capussotti, L.; Aglietta, M. Induction gemcitabine and oxaliplatin therapy followed by a twice-weekly infusion of gemcitabine and concurrent external-beam radiation for neoadjuvant treatment of locally advanced pancreatic cancer: a single institutional experience. Cancer 2013, 119, 277–284. [Google Scholar] [CrossRef]
- Hosein, P.J.; Macintyre, J.; Kawamura, C.; Maldonado, J.C.; Ernani, V.; Loaiza-Bonilla, A.; Narayanan, G.; Ribeiro, A.; Portelance, L.; Merchan, J.R.; Levi, J.U.; Rocha-Lima, C.M. Neoadjuvant FOLFIRINOX in unresectable or borderline-resectable locally advanced pancreatic adenocarcinoma: A retrospective study BMC Cancer 2012, 12, 199. [CrossRef]
- Boone, B.A.; Steve, J.; Krasinskas, A.M.; Zureikat, A.H.; Lembersky, B.C.; Gibson, M.K.; Stoller, R.G.; Zeh, H.J.; Bahary, N. Outcome with FOLFIRINOX for borderline resectable and locally unresectable pancreatic cancer. J Surg Oncol 2013, 108, 236–241. [Google Scholar] [CrossRef]
- Faris, J.E.; Blaszkowsky, L.S.; Mcdermott, S.; Guimaraes, A.R.; Szymonifka, J.; Huynh, M.A.; Ferrone, C.R.; Wargo, J.A.; Allen, J.N.; Dias, L.E.; Kwak, E.L.; Lillemoe, K.D.; Thayer, S.P.; Murphy, J.E.; Zhu, A.X.; Sahani, D.V.; Wo, J.Y.; Clark, J.W.; Castillo, C.F-D.; Ryan, D.P.; Hong, T.S. FOLFIRINOX in locally advanced pancreatic cancer: The Massachusetts General Hospital Cancer Center experience. Oncologist 2013, 18, 543–548. [CrossRef] [PubMed]
- Blazer, M.; Wu, C.; Goldberg, R.M.; et al. Modified FOLFIRINOX is tolerable and effective in patients with borderline resectable (BRPC) and locally advanced unresectable (LAURPC) pancreatic cancer. Gastrointestinal Cancer Symposium. 2014; Vol. B48, abstr. 275.
- SWOG statistical Tools: one arm binominal. Available online: http://www.swogstat.org/stat/public/one_binomial.htm.
- Blazer, M.; Wu, C.; Goldberg, R.M.; Phillips, G.; Schmidt, C.; Muscarella, P.; Wuthrick, E.; Williams, T.M.; Reardon, J.; Ellison, E.C.; Bloomston, M.; Bekaii-Saab, T. Neoadjuvant-modified (m) FOLFIRINOX for locally advanced unresectable (LAPC) and borderline resectable (BRPC) adenocarcinoma Ann Surg Oncol 2015, 22, 1153–1159. [CrossRef]
- Petrelli, F.; Coinu, A.; Borgonovo, K.; Cabiddu, M.; Ghilardi, M.; Lonati, V.; Aitini, E.; Barni, S.; Gruppo Italiano per lo Studio dei Carcinomi dell’Apparato Digerente (GISCAD). Gruppo Italiano per lo Studio dei Carcinomi dell’Apparato Digerente (GISCAD). FOLFIRINOX-based neoadjuvant therapy in borderline resectable or unresectable pancreatic cancer: a meta-analytical review of published studies. Pancreas 2015, 44, 515–521. [Google Scholar] [CrossRef]
- Eshmuminov, D.; Aminjonov, B.; Palm, R.F.; Malleo, G.; Schmocker, R.K.; Abdallah, R.; Yoo, C.; Shaib, W.L.; Schneider, M.A.; Rangelova, E.; Choi, Y.J.; Kim, H.; Rose, J.B.; Patel, S.; Wilson, G.C.; Maloney, S.; Timmermann, L.; Sahora, K.; Rössler, F.; Lopez-Lopez, V.; Boyer, E.; Maggino, L.; Malinka, T.; Park, J.Y.; Katz, M.H.G.; Prakash, L.; Ahmad, S.A.; Helton, S.; Jang, J-Y. Hoffe, S.E.; Salvia, R.; Taieb, J.; He, J.; Clavien, P-A.; Held, U.; Lehmann, K. FOLFIRINOX or gemcitabine-based chemotherapy for borderline resectable and locally advanced pancreatic cancer: A multi-institutional, patient-level, meta-analysis and systematic review. Ann Surg Oncol 2023, 30, 4417–4428. [Google Scholar] [CrossRef] [PubMed]
- Janssen, Q.P.; Buettner, S.; Suker, M.; Beumer, B.R.; Addeo, P.; Bachellier, P.; Bahary, N.; Bekaii-Saab, T.; Bali, M.A.; Besselink, M.G.; Boone, B.A.; Chau, I.; Clarke, S.; Dillhoff, M.; El-Rayes, B.F.; Frakes, J.M.; Grose, D.; Hosein, P.J.; Jamieson, N.B.; Javed, A.A.; Khan, K.; Kim, K-P. Kim, S.C.; Kim, S.S.; Ko, A.H.; Lacy, J.; Margonis, G.A.; McCarter, M.D.; McKay, C.J.; Mellon, E.A.; Moorcraft, S.Y.; Okada, K-I.; Paniccia, A.; Parikh, P.J.; Peters, N.A.; Rabl, H.; Samra, J.; Tinchon, C.; van Tienhoven, G.; van Veldhuisen, E.; Wang-Gillam, A.; Weiss, M.J.; Wilmink, J.W.; Yamaue, H.; Homs, M.Y.V.; van Eijck, C.H.J.; Katz, M.H.G.; Koerkamp, B.G. Neoadjuvant FOLFIRINOX in patients with borderline resectable pancreatic cancer: A systematic review and patient-level meta-analysis. J Natl Cancer Inst 2019, 111, 782–794. [Google Scholar] [CrossRef] [PubMed]
- Satoi, S.; Yamaue, H.; Kato, K.; Takahashi, S.; Hirono, S.; Takeda, S.; Eguchi, H.; Sho, M.; Wada, K.; Shinchi, H.; Kwon, A.H.; Hirano, S.; Kinoshita, T.; Nakao, A.; Nagano, H.; Nakajima, Y.; Sano, K.; Miyazaki, M.; Takada, T. Role of adjuvant surgery for patients with initially unresectable pancreatic cancer with a long-term favorable response to nonsurgical anticancer treatments: results of a project study for pancreatic surgery by the Japanese Society of Hepato-Biliary-Pancreatic Surgery. J Hepatobiliary Pancreat Sci 2013, 20, 590–600. [Google Scholar] [CrossRef] [PubMed]
- Teriaca, M.A.; Loi, M.; Suker, M.; Eskens, F.A.L.M.; van Eijck, C.H.J.; Nuyttens, J.J. A phase II study of stereotactic radiotherapy after FOLFIRINOX for locally advanced pancreatic cancer (LAPC-1 trial): long-term outcome. Radiother Oncol 2021, 155, 232–236. [Google Scholar] [CrossRef] [PubMed]
| (n = 15) | |
| Clinical characteristics | |
| Age, median (range) | 64 (44-73) |
| Sex, Male / Female, n (%) | 12 (80.0) / 3 (20.0) |
| ECOG Performance Status 0/1, n (%) | 15 (100)/ 0 (0) |
| BSA median (range) | 1.75 (1.46–2.13) |
| Smoking, ever smoker/ never smoker, n (%) | 10 (66.7)/ 5 (33.3) |
| DM yes/no, n (%) | 5(33.3)/ 10 (66.7) |
| CEA (ng/ml), median (range) | 2.5 (0.6–43.1) |
| CA19-9 (U/ml), median (range) | 44 (1.2–13549) |
| Bile duct Stent Placement, yes/no, n (%) | 4 (26.7)/ 11 (73.3) |
| UGT1A1 (*6/*28) | |
| wild/wild, n (%) | 5 (33.3) |
| wild/heterozygous, n (%) | 6 (40) |
| heterozygous/wild, n (%) | 4 26.7) |
| Radiological findings | |
| Tumor diameter (mm), median (range) | 35 (24–72) |
| Tumor location (Ph/ Pbt), n (%) | 9 (60)/ 6 (40) |
| Arterial invasion, n (%) | 15 (100) |
| Portal invasion, n (%) | 12 (80) |
| Bile duct invasion, n (%) | 5 (33.3) |
| Duodenum invasion, n (%) | 1 (6.7) |
| Other organ invasion, n (%) | 0 (0) |
| JPS Stage (6th edition) n (4a: 4b) | 10:05 |
| UICC Stage (7th edition) n (3: 4) | 15:00 |
| Treatment course, median (rage) | 6 (3-8) |
| Relative dose intensity and dose intensity L-OHP (%), (mg/m2), mean (rage) CPT-11 (%), (mg/m2), mean (rage) 5-FU bolus (%), (mg/m2), man (rage) 5-FU continuous (%), (mg/m2), mean (rage) |
75, 63.75 (43-80) 71.9, 129.47 (84.7-158.8) 13.3, 53.3 (0-235.3) 76.9, 1846.2 (1411.8-2258.9) |
| RECIST (CR/PR/SD/PD), n (%) | 0 (0)/ 4 (26.7)/ 10 (66.7)/ 1 (6.7) |
| Tumor Shrinkage ration (%), median (range) | 16 (-22- 48) |
| Decrease in the CEA ration, (%), median (range) | -41.7 (-304~52.1) |
| Decrease in the CA19-9 ratio (%), median (range) | 38.6 (-425~92.3) |
| Hematological Adverse events (All grade/ grade>3), n (%) |
15 (100)/ 10 (66.7) |
| Non-hematological Adverse events (All grade/ grade>3) |
15 (100)/ 6 (40) |
| All grades, n (%) | Grade 3/4, n (%) | |
| Hematological toxicity | ||
| Anemia | 15 (100) | 0 (0) |
| Thrombocytopenia | 10 (66.7) | 0 (0) |
| Leukopenia | 14 (93.3) | 3 (20) |
| Neutropenia | 14 (93.3) | 10 (66.7) |
| Febrile neutropenia | 1 (6.7) | 1 (6.7) |
| Non-hematological toxicity | ||
| Bilirubin | 1 (8.7) | 0 (0) |
| AST | 6 (40) | 1 (6.7) |
| ALP | 6 (40) | 0 (0) |
| Alb | 15 (100) | 0 (0) |
| Creatinine | 1 (6.7) | 0 (0) |
| Hyponatremia | 9 (60) | 1 (6.7) |
| Fever | 5 (33.3) | 0 (0) |
| Nausea | 14 (93.3) | 2 (13.3) |
| Vomiting | 0 (0) | 0 (0) |
| Diarrhea | 9 (60) | 1 (6.7) |
| Appetite loss | 15 (100) | 2 (13.3) |
| Sensory neuropathy | 15 (100) | 1 (6.7) |
| Stomatitis | 6 (40) | 0 (0) |
| Hand foot syndrome | 2 (13.3) | 0 (0) |
| Acute cholangitis | 1 (6.7) | 1 (6.7) |
| Acute pancreatitis | 1 (6.7) | 1 (6.7) |
| Pneumonia | 1 (6.7) | 1 (6.7) |
| Periodontitis | 1 (6.7) | 0 (0) |
| Lymphadenopathy | 1 (6.7) | 0 (0) |
| Surgical procedure (PD: DP: TP) | 6 (5: 1: 0) |
| Operation time (min), median (range) | 646 (534–1033) |
| Arterial resection, n (%) | 3 (20) |
| Portal vein resection, n (%) | 4 (26.7) |
| Blood loss (ml), median (range) | 1600 (400–8853) |
| Pathological type (well: mod: poor: other) | 6 (2: 4: 0: 0) |
| Evans Grade (1: 2: 3: 4) | (3: 2: 1: 0) |
| Decrease in the CEA ratio (%), median (range) | -18.5 (-304–52.1) |
| Decrease in the CA19-9 ratio (%), median (range) | 48.65 (-73.7–92.3) |
| Pathological node positive, n (%) | 2 (33.3) |
| R (0: 1) | 6 (5: 1) |
| Recurrence, n (%) | 4 (66.7) |
| Major complication, n (%) | 1 (6.7) |
| JPS Stage (6th edition) n (2: 3: 4a: 4b) | 6 (1: 1: 2: 2) |
| UICC Stage (7th edition) (1b: 2a: 2b: 3: 4) | 6 (1: 3: 1: 0: 1) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




