Submitted:
09 January 2024
Posted:
10 January 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Antigen and Reagents
2.2. Study cohort and ethical statements
2.3. Detection of anti-SARS-Cov-2 IgG antibodies.
2.4. Detection of SARS-Cov-2 IgG subclasses.
2.5. Neutralization Assay
2.6. Statistical analysis
3. Results and Discussion
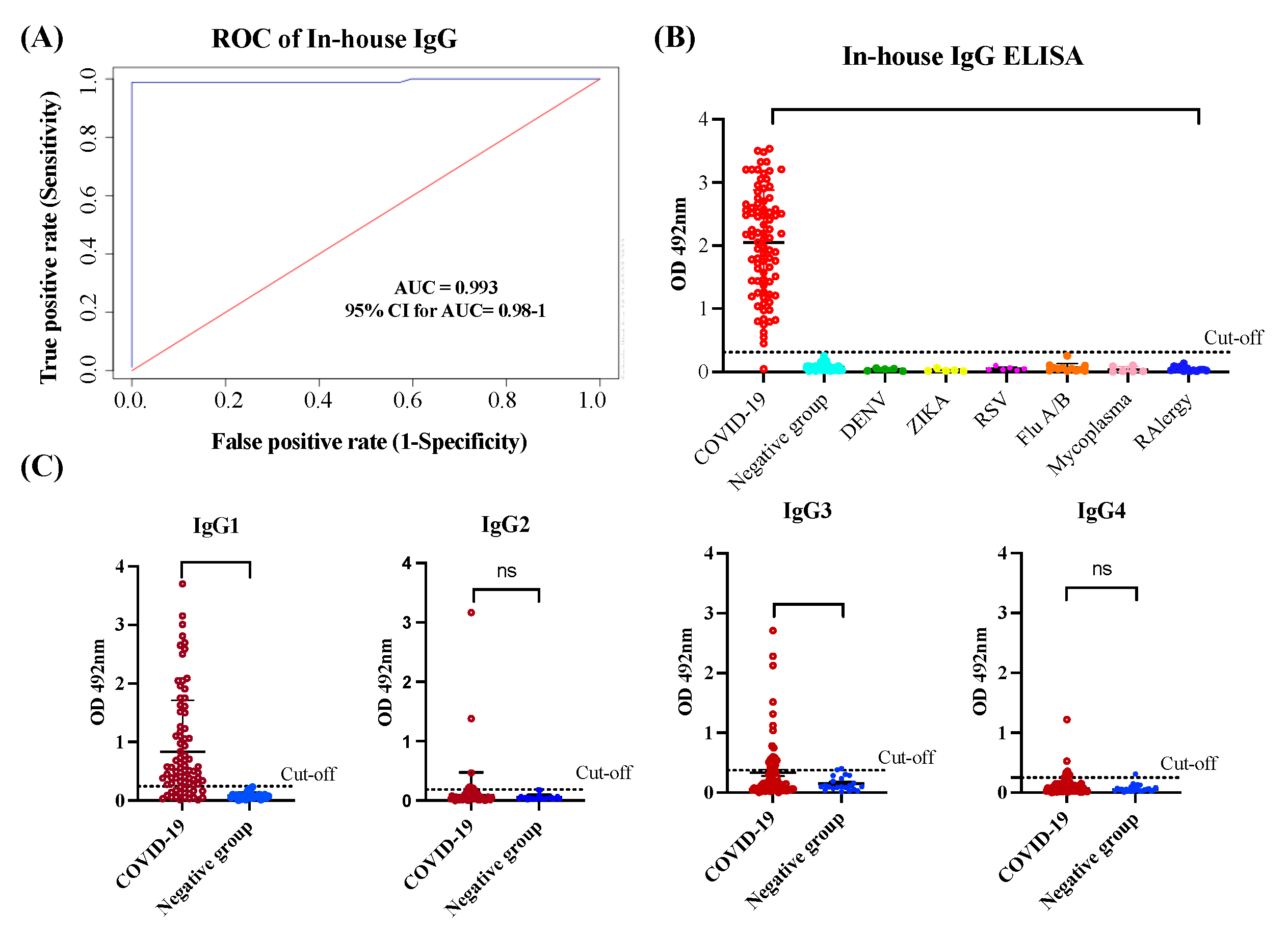
3.1. In-house IgG and IgG-isotypes ELISA performance assessment
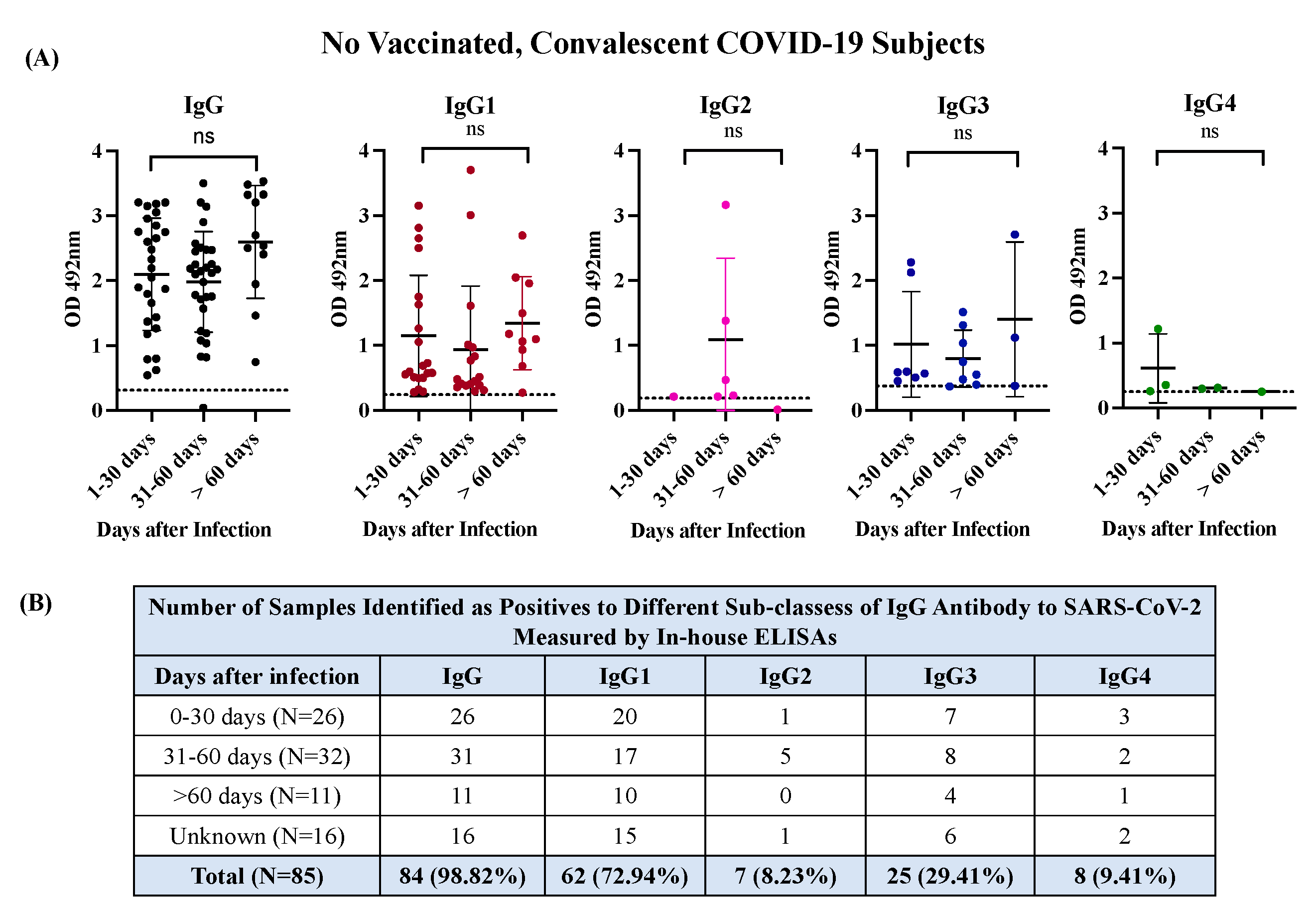
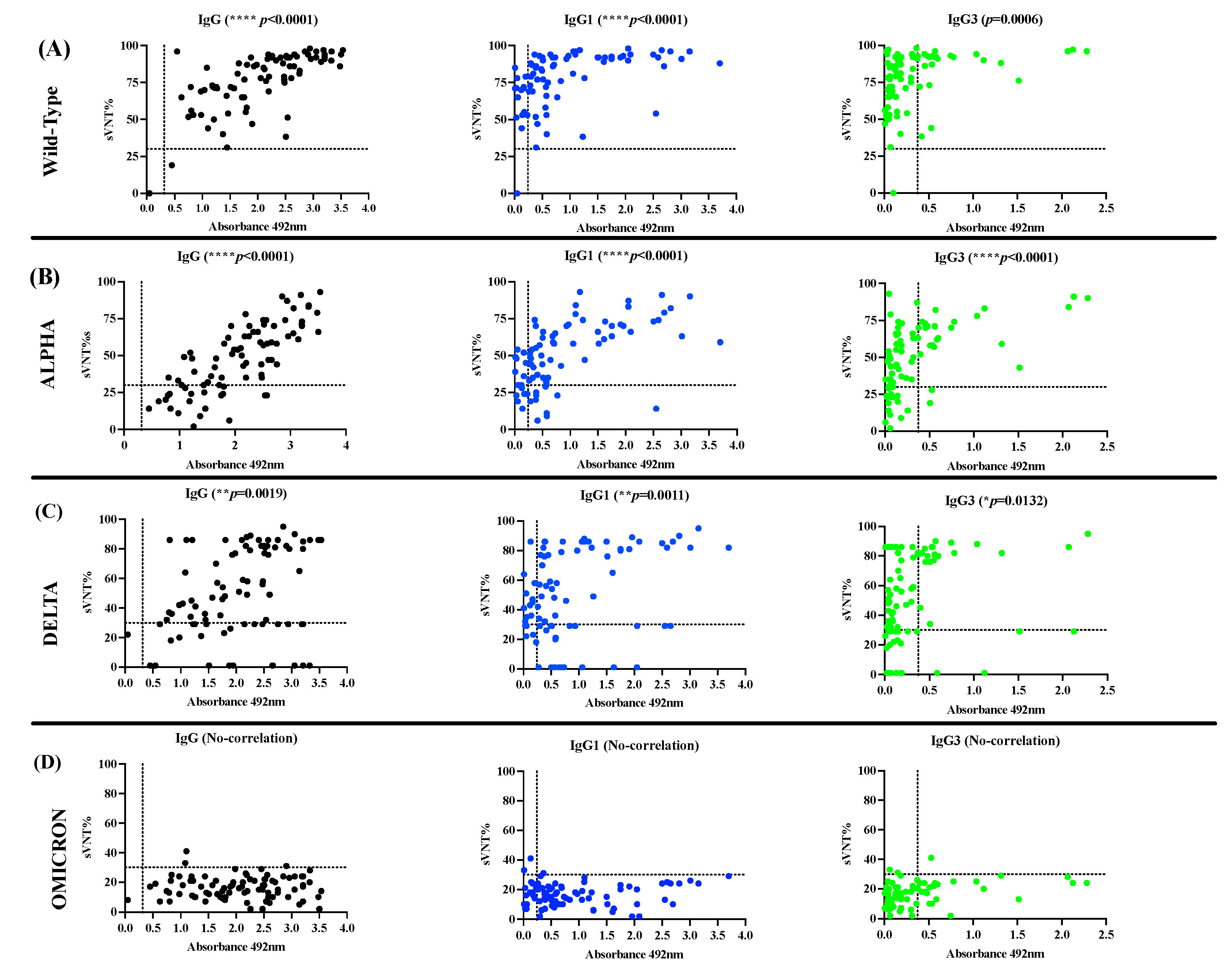
3.2. The antibody response in unvaccinated convalescent COVID-19 subjects is dominated by IgG1 and IgG3 isotypes, which neutralize the Wild-type strain and the VOC Alpha and Delta but are poorly effective against Omicron.
-
Kappa interpretation values:
- 0.01 - 0.2 slight agreement
- 0.21 - 0.4 fair agreement
- 0.41 - 0.6 moderate agreement
- 0.61 - 0.8 substantial agreement
- 0.81 – 1.0 almost perfect or perfect agreement
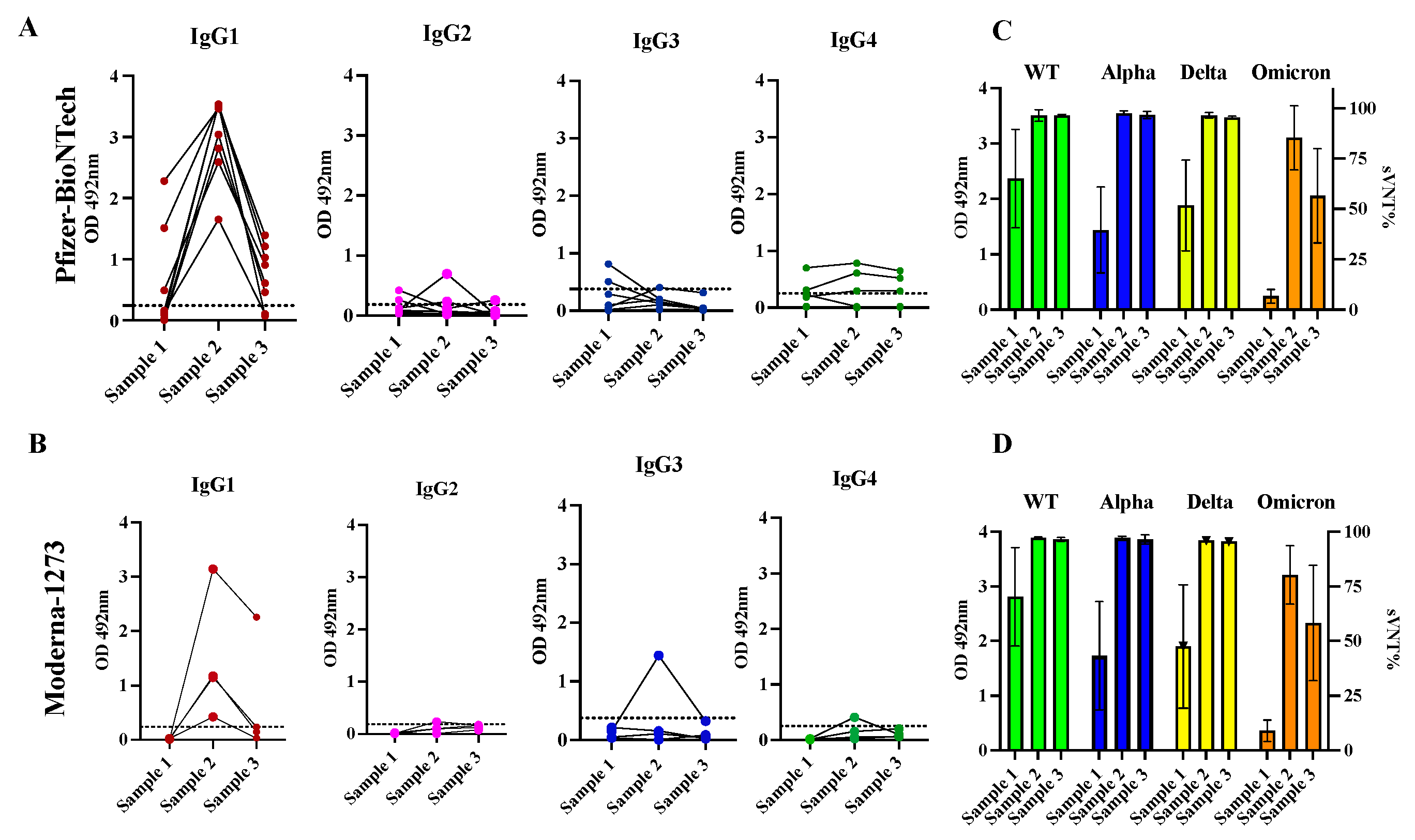
3.3. The antibody response in previously infected subjects that received two doses of Pfizer-BioNTech or Moderna-1273 is dominated by the IgG1 isotype that although declined fast, exhibited potent neutralizing activity against the VOC Alpha, Delta, and Omicron.
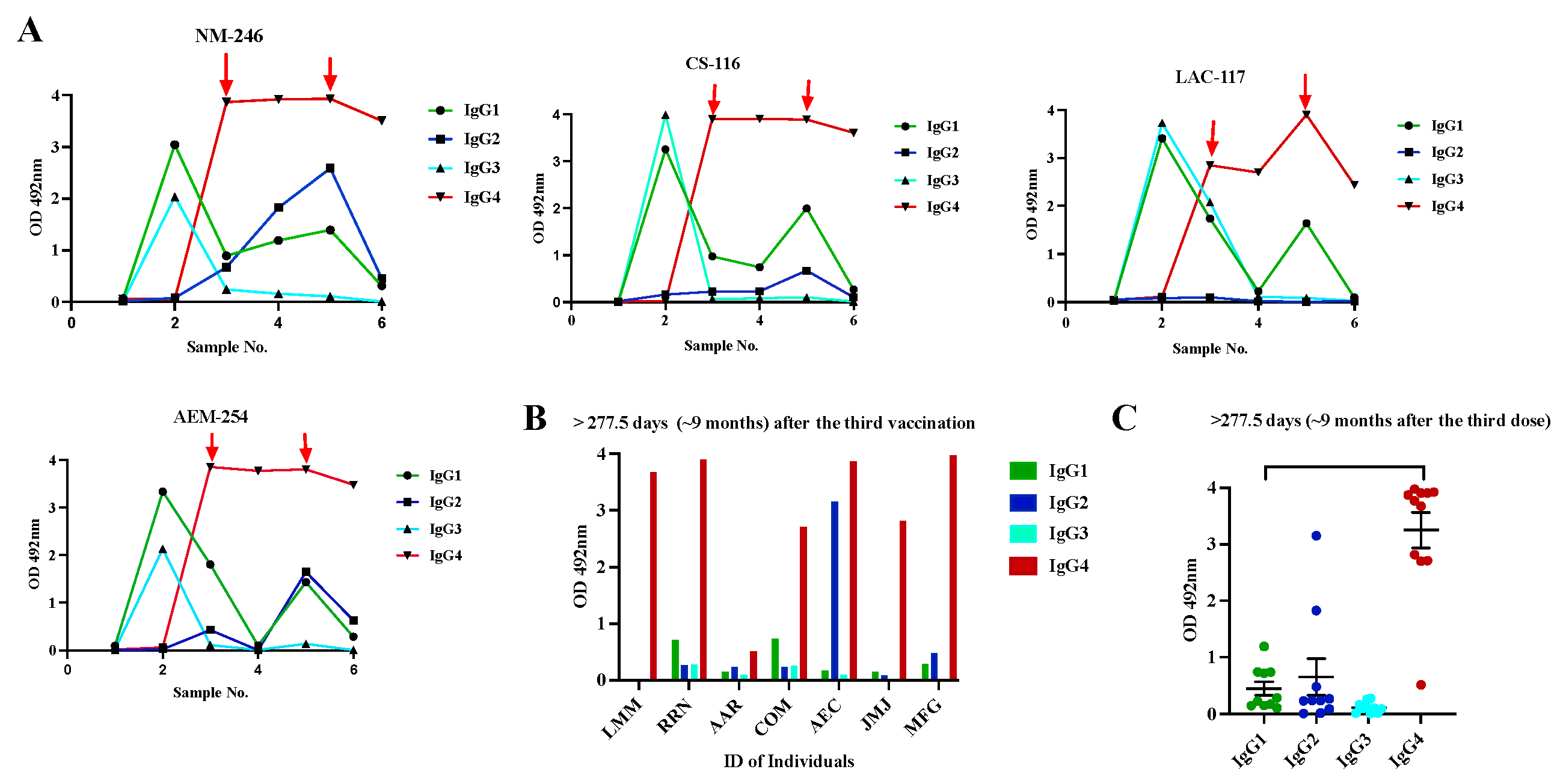
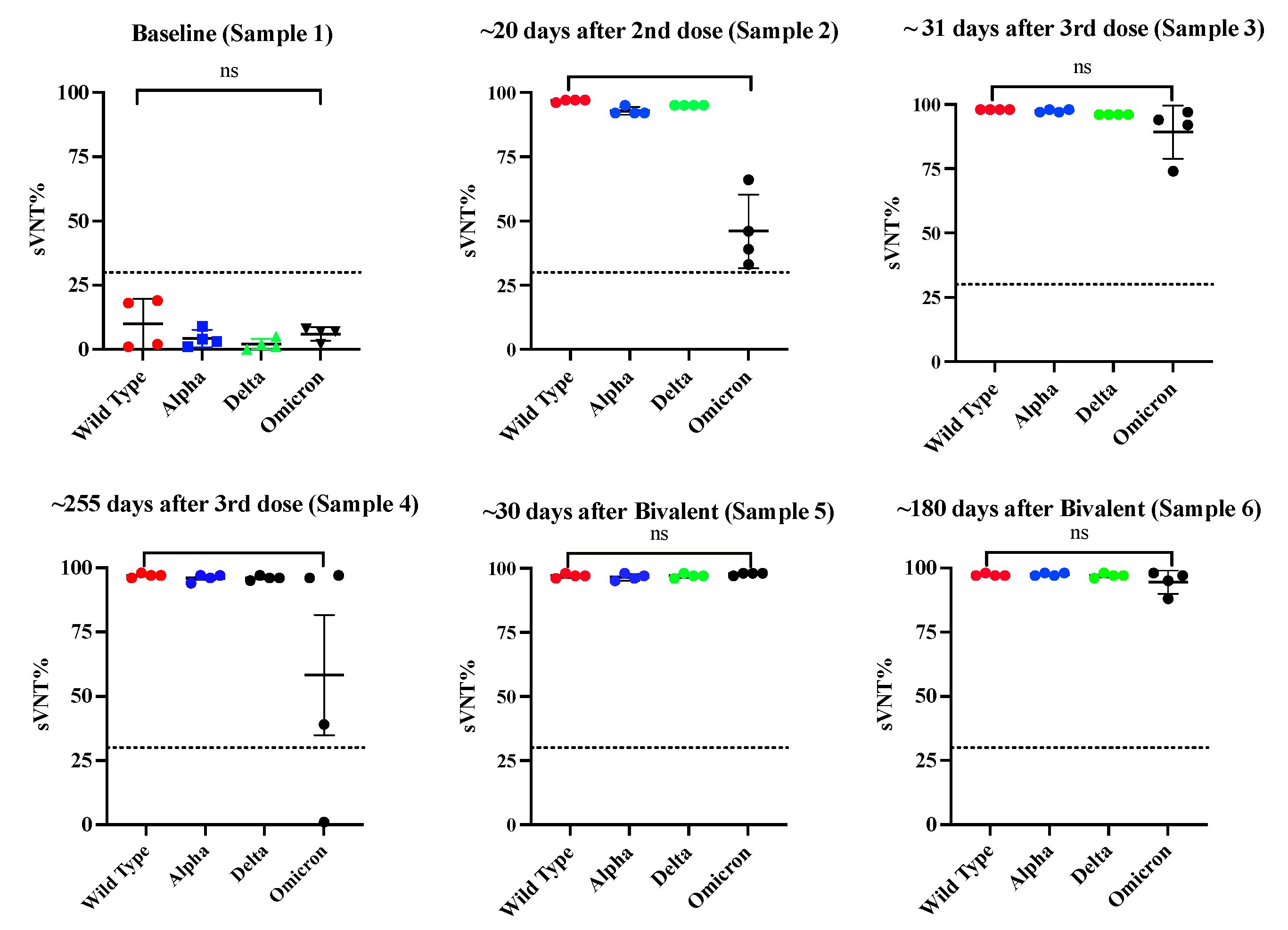
3.4. Class switch toward IgG4 occurs in subjects that receive multiple doses of Pfizer-BioNTech vaccine, which is sustained over the time.
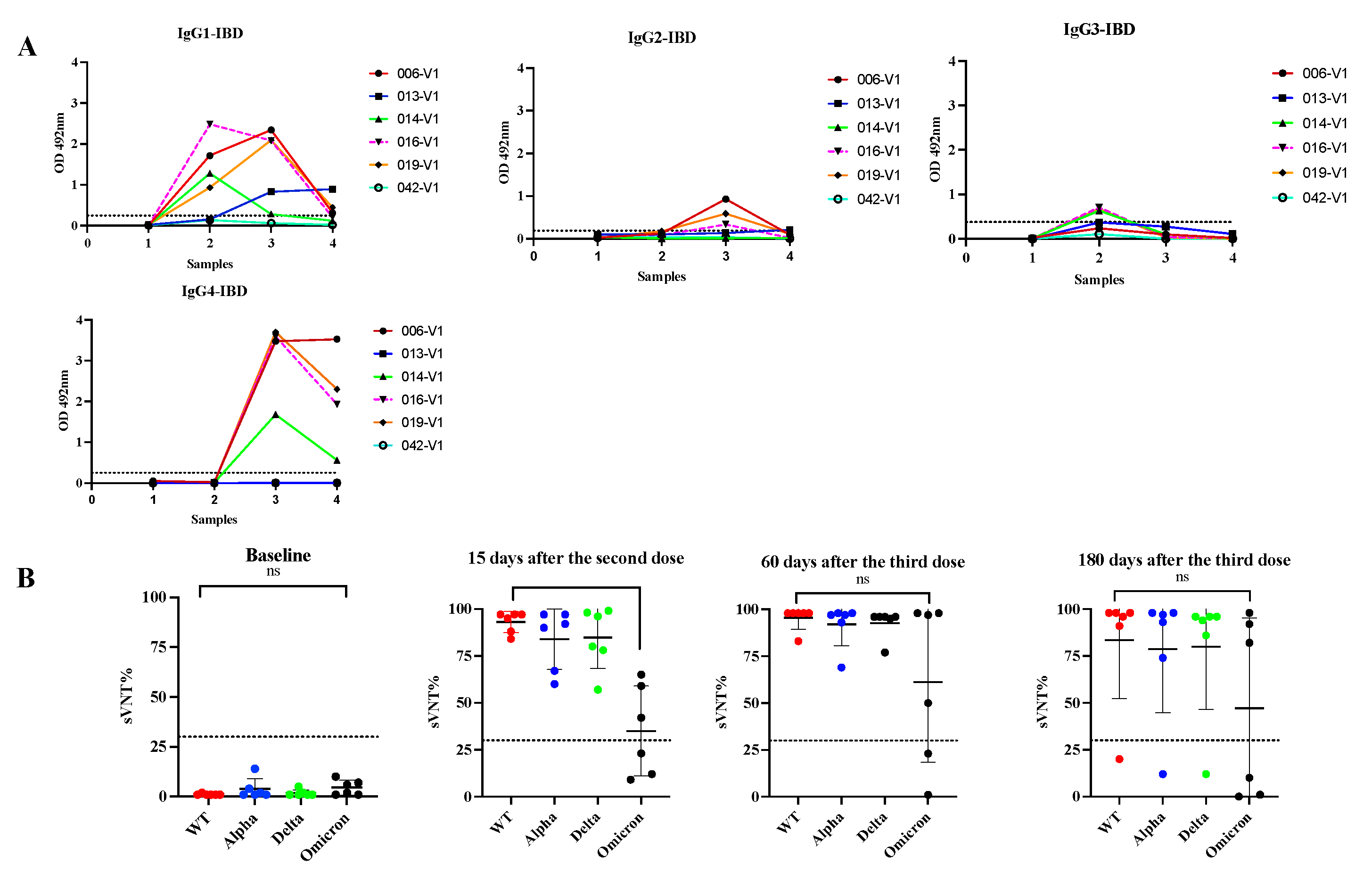
3.5. Switch class toward IgG4 is also observed in subjects with Inflammatory Bowel Diseases with multiple vaccinations.
4. Limitations of the study
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- WHO. 2022. Available online: https://covid19.who.int/ (accessed on 29 March 2022).
- Soto-Canetti G, Garcia L, Julia AE, Gordian EI, Bartolomei JA, Camareno N, Rodriguez JF, Montoya M. 2022. Developing a Case Investigation and Contact-Tracing System in Puerto Rico, 2020. Am J Public Health 112:223-226. [CrossRef]
- Amanat F, Stadlbauer D, Strohmeier S, Nguyen THO, Chromikova V, McMahon M, Jiang K, Arunkumar GA, Jurczyszak D, Polanco J, Bermudez-Gonzalez M, Kleiner G, Aydillo T, Miorin L, Fierer DS, Lugo LA, Kojic EM, Stoever J, Liu STH, Cunningham-Rundles C, Felgner PL, Moran T, Garcia-Sastre A, Caplivski D, Cheng AC, Kedzierska K, Vapalahti O, Hepojoki JM, Simon V, Krammer F. 2020. A serological assay to detect SARS-CoV-2 seroconversion in humans. Nat Med 26:1033-1036. [CrossRef]
- Lisboa Bastos M, Tavaziva G, Abidi SK, Campbell JR, Haraoui LP, Johnston JC, Lan Z, Law S, MacLean E, Trajman A, Menzies D, Benedetti A, Ahmad Khan F. 2020. Diagnostic accuracy of serological tests for covid-19: systematic review and meta-analysis. BMJ 370:m2516. [CrossRef]
- Ramirez-Reveco A, Velasquez G, Aros C, Navarrete G, Villarroel-Espindola F, Navarrete M, Fica A, Plaza A, Castro N, Verdugo C, Acosta-Jamett G, Verdugo CC. 2023. Performance estimation of two in-house ELISA assays for COVID-19 surveillance through the combined detection of anti-SARS-CoV-2 IgA, IgM, and IgG immunoglobulin isotypes. PLoS One 18:e0270388. eCollection 2023. [CrossRef]
- Rungrojcharoenkit K, Suthangkornkul R, Utennam D, Buddhari D, Pinpaiboon S, Mongkolsirichaikul D, Fernandez S, Jones AR, Cotrone TS, Hunsawong T. 2023. Standardization of in-house anti-IgG and IgA ELISAs for the detection of COVID-19. PLoS One 18:e0287107. eCollection 2023. [CrossRef]
- Sariol CAA, Pantoja P, Serrano-Collazo C, Rosa-Arocho T, Armina-Rodriguez A, Cruz L, Stone ETT, Arana T, Climent C, Latoni G, Atehortua D, Pabon-Carrero C, Pinto AKK, Brien JDD, Espino AMM. 2021. Function Is More Reliable than Quantity to Follow Up the Humoral Response to the Receptor-Binding Domain of SARS-CoV-2-Spike Protein after Natural Infection or COVID-19 Vaccination. Viruses 13 (10):1972. [CrossRef]
- Lopez-Marte P, Soto-Gonzalez A, Ramos-Tollinchi L, Torres-Jorge S, Ferre M, Rodriguez-Martino E, Torres EA, Sariol CA. 2022. Inefficient Induction of Neutralizing Antibodies against SARS-CoV-2 Variants in Patients with Inflammatory Bowel Disease on Anti-Tumor Necrosis Factor Therapy after Receiving a Third mRNA Vaccine Dose. Vaccines (Basel) 10(8):1301. [CrossRef]
- Sariol CA, Serrano-Collazo C, Ortiz EJ, Pantoja P, Cruz L, Arana T, Atehortua D, Pabon-Carrero C, Espino AM. 2021. Limited Impact of Delta Variant's Mutations on the Effectiveness of Neutralization Conferred by Natural Infection or COVID-19 Vaccines in a Latino Population. Viruses 13(12):2405. [CrossRef]
- Li K, Huang B, Wu M, Zhong A, Li L, Cai Y, Wang Z, Wu L, Zhu M, Li J, Wang Z, Wu W, Li W, Bosco B, Gan Z, Qiao Q, Wu J, Wang Q, Wang S, Xia X. 2020. Dynamic changes in anti-SARS-CoV-2 antibodies during SARS-CoV-2 infection and recovery from COVID-19. Nat Commun 11(1):6044. [CrossRef]
- Hashem AM, Algaissi A, Almahboub SA, Alfaleh MA, Abujamel TS, Alamri SS, Alluhaybi KA, Hobani HI, AlHarbi RH, Alsulaiman RM, ElAssouli MZ, Hala S, Alharbi NK, Alhabbab RY, AlSaieedi AA, Abdulaal WH, Bukhari A, Al-Somali AA, Alofi FS, Khogeer AA, Pain A, Alkayyal AA, Almontashiri NAM, Ahmad BM, Li X. 2020. Early Humoral Response Correlates with Disease Severity and Outcomes in COVID-19 Patients. Viruses 12(12):1390. [CrossRef]
- Trieu MC, Bansal A, Madsen A, Zhou F, Saevik M, Vahokoski J, Brokstad KA, Krammer F, Tondel C, Mohn KGI, Blomberg B, Langeland N, Cox RJ, Bergen C-RG. 2021. SARS-CoV-2-Specific Neutralizing Antibody Responses in Norwegian Health Care Workers After the First Wave of COVID-19 Pandemic: A Prospective Cohort Study. J Infect Dis 223:589-599. [CrossRef]
- Wang, P. 2021. Significance of IgA antibody testing for early detection of SARS-CoV-2. J Med Virol 93:1888-1889. Epub 2020 Dec 17. [CrossRef]
- Yu HQ, Sun BQ, Fang ZF, Zhao JC, Liu XY, Li YM, Sun XZ, Liang HF, Zhong B, Huang ZF, Zheng PY, Tian LF, Qu HQ, Liu DC, Wang EY, Xiao XJ, Li SY, Ye F, Guan L, Hu DS, Hakonarson H, Liu ZG, Zhong NS. 2020. Distinct features of SARS-CoV-2-specific IgA response in COVID-19 patients. Eur Respir J 56(2):2001526. Print 2020 Aug. [CrossRef]
- Xiang T, Liang B, Fang Y, Lu S, Li S, Wang H, Li H, Yang X, Shen S, Zhu B, Wang B, Wu J, Liu J, Lu M, Yang D, Dittmer U, Trilling M, Deng F, Zheng X. 2021. Declining Levels of Neutralizing Antibodies Against SARS-CoV-2 in Convalescent COVID-19 Patients One Year Post Symptom Onset. Front Immunol 12:708523. eCollection 2021. [CrossRef]
- Zhu L, Xu X, Zhu B, Guo X, Xu K, Song C, Fu J, Yu H, Kong X, Peng J, Huang H, Zou X, Ding Y, Bao C, Zhu F, Hu Z, Wu M, Shen H. 2021. Kinetics of SARS-CoV-2 Specific and Neutralizing Antibodies over Seven Months after Symptom Onset in COVID-19 Patients. Microbiol Spectr 9:e0059021. Epub 2021 Sep 22. [CrossRef]
- Hantz, S. 2020. [Biological diagnosis of Sars-CoV-2 infection: strategies and interpretation of results]. Rev Francoph Lab 2020:48-56. Epub 2020 Oct 31. [CrossRef]
- Vidarsson G, Dekkers G, Rispens T. 2014. IgG subclasses and allotypes: from structure to effector functions. Front Immunol 5:520. eCollection 2014. [CrossRef]
- Husain-Syed F, Vadasz I, Wilhelm J, Walmrath HD, Seeger W, Birk HW, Jennert B, Dietrich H, Herold S, Trauth J, Tello K, Sander M, Morty RE, Slanina H, Schuttler CG, Ziebuhr J, Kassoumeh S, Ronco C, Ferrari F, Warnatz K, Stahl K, Seeliger B, Hoeper MM, Welte T, David S. 2021. Immunoglobulin deficiency as an indicator of disease severity in patients with COVID-19. Am J Physiol Lung Cell Mol Physiol 320:L590-L599. Epub 2020 Nov 25. [CrossRef]
- Patil HP, Rane PS, Shrivastava S, Palkar S, Lalwani S, Mishra AC, Arankalle VA. 2021. Antibody (IgA, IgG, and IgG Subtype) Responses to SARS-CoV-2 in Severe and Nonsevere COVID-19 Patients. Viral Immunol 34:201-209. Epub 2021 Mar 3. [CrossRef]
- Yates JL, Ehrbar DJ, Hunt DT, Girardin RC, Dupuis AP, 2nd, Payne AF, Sowizral M, Varney S, Kulas KE, Demarest VL, Howard KM, Carson K, Hales M, Ejemel M, Li Q, Wang Y, Peredo-Wende R, Ramani A, Singh G, Strle K, Mantis NJ, McDonough KA, Lee WT. 2021. Serological analysis reveals an imbalanced IgG subclass composition associated with COVID-19 disease severity. Cell Rep Med 2:100329. Epub 2021 Jun 15. [CrossRef]
- Kellner C, Otte A., Cappuzzello E., Klausz K., Peipp M.. 2017. Modulating cytotoxic effector functions by Fc engineering to improve cancer tharpy. Transfus Med Hemother 44:327-336. Epub 2017 Sep 8. [CrossRef]
- Damelang T, Rogerson S.J., Kent S.J., Chung A.W.. 2019. Role of IgG3 in infections diseases. Trends Immunol 40:197-211. Epub 2019 Feb 10. [CrossRef]
- Tejedor Vaquero S, de Campos-Mata L, Ramada JM, Diaz P, Navarro-Barriuso J, Ribas-Llaurado C, Rodrigo Melero N, Carolis C, Cerutti A, Gimeno R, Magri G. 2021. The mRNA-1273 Vaccine Induces Cross-Variant Antibody Responses to SARS-CoV-2 With Distinct Profiles in Individuals With or Without Pre-Existing Immunity. Front Immunol 12:737083. eCollection 2021. [CrossRef]
- Rahman S, Rahman MM, Miah M, Begum MN, Sarmin M, Mahfuz M, Hossain ME, Rahman MZ, Chisti MJ, Ahmed T, Arifeen SE, Rahman M. 2022. COVID-19 reinfections among naturally infected and vaccinated individuals. Sci Rep 12:1438. [CrossRef]
- Ren X, Zhou J, Guo J, Hao C, Zheng M, Zhang R, Huang Q, Yao X, Li R, Jin Y. 2022. Reinfection in patients with COVID-19: a systematic review. Glob Health Res Policy 7(1):12. [CrossRef]
- Fernandes Q, Inchakalody VP, Merhi M, Mestiri S, Taib N, Moustafa Abo El-Ella D, Bedhiafi T, Raza A, Al-Zaidan L, Mohsen MO, Yousuf Al-Nesf MA, Hssain AA, Yassine HM, Bachmann MF, Uddin S, Dermime S. 2022. Emerging COVID-19 variants and their impact on SARS-CoV-2 diagnosis, therapeutics and vaccines. Ann Med 54(1):524-540. [CrossRef]
- Espino AM, Pantoja P., Sariol C.A. 2021. Validation and performance of a quantitative IgG assay for the screening of SARS-CoV-2 antibodies. bioRxv https: doi.org/10.1101/2020.06.11.146332.
- Taylor SC, Hurst B, Charlton CL, Bailey A, Kanji JN, McCarthy MK, Morrison TE, Huey L, Annen K, DomBourian MG, Knight V. 2021. A New SARS-CoV-2 Dual-Purpose Serology Test: Highly Accurate Infection Tracing and Neutralizing Antibody Response Detection. J Clin Microbiol 59(4): e02438-20. Print 2021 Mar 19. [CrossRef]
- Hosmer Jr DW, Lemeshow S., Sturdivant, R.X. 2013. Applied Logistic Regression. John Wiley & Sons.
- Obermeier P, Muehlhans S, Hoppe C, Karsch K, Tief F, Seeber L, Chen X, Conrad T, Boettcher S, Diedrich S, Rath B. 2016. Enabling Precision Medicine With Digital Case Classification at the Point-of-Care. EBioMedicine 4:191-6. eCollection 2016 Feb. [CrossRef]
- Linnet, K. 1993. Evaluation of regression procedures for methods comparison studies. Clin Chem 39:424-32.
- J. C. 1960. A coefficient of agreement for nominal scales. Educational and Psycological Measurement 20:37-46.
- Wieckowska B, Kubiak KB, Jozwiak P, Moryson W, Stawinska-Witoszynska B. 2022. Cohen's Kappa Coefficient as a Measure to Assess Classification Improvement following the Addition of a New Marker to a Regression Model. Int J Environ Res Public Health 19(16):10213. [CrossRef]
- Landis JR, Koch GG. 1977. An application of hierarchical kappa-type statistics in the assessment of majority agreement among multiple observers. Biometrics 33:363-74.
- Valdes-Fernandez BN, Duconge J, Espino AM, Ruano G. 2021. Personalized health and the coronavirus vaccines-Do individual genetics matter? Bioessays 43:e2100087. Epub 2021 Jul 26. [CrossRef]
- Isho B, Abe KT, Zuo M, Jamal AJ, Rathod B, Wang JH, Li Z, Chao G, Rojas OL, Bang YM, Pu A, Christie-Holmes N, Gervais C, Ceccarelli D, Samavarchi-Tehrani P, Guvenc F, Budylowski P, Li A, Paterson A, Yue FY, Marin LM, Caldwell L, Wrana JL, Colwill K, Sicheri F, Mubareka S, Gray-Owen SD, Drews SJ, Siqueira WL, Barrios-Rodiles M, Ostrowski M, Rini JM, Durocher Y, McGeer AJ, Gommerman JL, Gingras AC. 2020. Persistence of serum and saliva antibody responses to SARS-CoV-2 spike antigens in COVID-19 patients. Sci Immunol 5(52): eabe5511. [CrossRef]
- Chia WN, Tan CW, Foo R, Kang AEZ, Peng Y, Sivalingam V, Tiu C, Ong XM, Zhu F, Young BE, Chen MI, Tan YJ, Lye DC, Anderson DE, Wang LF. 2020. Serological differentiation between COVID-19 and SARS infections. Emerg Microbes Infect 9:1497-1505. [CrossRef]
- Oliveira BA, Oliveira LC, Sabino EC, Okay TS. 2020. SARS-CoV-2 and the COVID-19 disease: a mini review on diagnostic methods. Rev Inst Med Trop Sao Paulo 62:e44. eCollection 2020. [CrossRef]
- Karger AB, Brien JD, Christen JM, Dhakal S, Kemp TJ, Klein SL, Pinto LA, Premkumar L, Roback JD, Binder RA, Boehme KW, Boppana S, Cordon-Cardo C, Crawford JM, Daiss JL, Dupuis AP, 2nd, Espino AM, Firpo-Betancourt A, Forconi C, Forrest JC, Girardin RC, Granger DA, Granger SW, Haddad NS, Heaney CD, Hunt DT, Kennedy JL, King CL, Krammer F, Kruczynski K, LaBaer J, Lee FE, Lee WT, Liu SL, Lozanski G, Lucas T, Mendu DR, Moormann AM, Murugan V, Okoye NC, Pantoja P, Payne AF, Park J, Pinninti S, Pinto AK, Pisanic N, Qiu J, Sariol CA, Simon V, Song L, et al. 2022. The Serological Sciences Network (SeroNet) for COVID-19: Depth and Breadth of Serology Assays and Plans for Assay Harmonization. mSphere 7:e0019322. Epub 2022 Jun 15. [CrossRef]
- Luo H, Jia T, Chen J, Zeng S, Qiu Z, Wu S, Li X, Lei Y, Wang X, Wu W, Zhang R, Zou X, Feng T, Ding R, Zhang Y, Chen YQ, Sun C, Wang T, Fang S, Shu Y. 2021. The Characterization of Disease Severity Associated IgG Subclasses Response in COVID-19 Patients. Front Immunol 12:632814. eCollection 2021. [CrossRef]
- Korobova ZR, Zueva EV, Arsentieva NA, Batsunov OK, Liubimova NE, Khamitova IV, Kuznetsova RN, Rubinstein AA, Savin TV, Stanevich OV, Kulikov AN, Pevtsov DE, Totolian AA. 2022. Changes in Anti-SARS-CoV-2 IgG Subclasses over Time and in Association with Disease Severity. Viruses 14(5):941. [CrossRef]
- Rispens T, Huijbers MG. 2023. The unique properties of IgG4 and its roles in health and disease. [CrossRef]
- Santiago GA, Flores B, Gonzalez GL, Charriez KN, Huertas LC, Volkman HR, Van Belleghem SM, Rivera-Amill V, Adams LE, Marzan M, Hernandez L, Cardona I, O'Neill E, Paz-Bailey G, Papa R, Munoz-Jordan JL. 2022. Genomic surveillance of SARS-CoV-2 in Puerto Rico enabled early detection and tracking of variants. Commun Med (Lond) 2:100. eCollection 2022. [CrossRef]
- Arevalo-Rodriguez I B-GD, Simancas-Racines D, Zambrano-Achig P, del Campo R, Ciapponi A, Sued O, Martinez-Garcia L, Rutjes A, Low N, Perez-Molina JA, Zamora J. 2020. FALSE-NEGATIVE RESULTS OF INITIAL RT-PCR ASSAYS FOR COVID-19: A SYSTEMATIC REVIEW. [CrossRef]
- Chan JF, Yip CC, To KK, Tang TH, Wong SC, Leung KH, Fung AY, Ng AC, Zou Z, Tsoi HW, Choi GK, Tam AR, Cheng VC, Chan KH, Tsang OT, Yuen KY. 2020. Improved Molecular Diagnosis of COVID-19 by the Novel, Highly Sensitive and Specific COVID-19-RdRp/Hel Real-Time Reverse Transcription-PCR Assay Validated In Vitro and with Clinical Specimens. J Clin Microbiol 58(5): e00310-20. Print 2020 Apr 23. [CrossRef]
- Loeffelholz MJ, Tang YW. 2020. Laboratory diagnosis of emerging human coronavirus infections - the state of the art. Emerg Microbes Infect 9:747-756. [CrossRef]
- Tang YW, Schmitz JE, Persing DH, Stratton CW. 2020. Laboratory Diagnosis of COVID-19: Current Issues and Challenges. J Clin Microbiol 58(6):e00512-20. Print 2020 May 26. [CrossRef]
- Giovanetti M, Benedetti F, Campisi G, Ciccozzi A, Fabris S, Ceccarelli G, Tambone V, Caruso A, Angeletti S, Zella D, Ciccozzi M. 2021. Evolution patterns of SARS-CoV-2: Snapshot on its genome variants. Biochem Biophys Res Commun 538:88-91. Epub 2020 Nov 6. [CrossRef]
- Roy, U. 2022. Comparative structural analyses of selected spike protein-RBD mutations in SARS-CoV-2 lineages. Immunol Res 70:143-151. Epub 2021 Nov 16. [CrossRef]
- Chakraborty C, Bhattacharya M, Sharma AR, Mallik B. 2022. Omicron (B.1.1.529) - A new heavily mutated variant: Mapped location and probable properties of its mutations with an emphasis on S-glycoprotein. Int J Biol Macromol 219:980-997. Epub 2022 Aug 8. [CrossRef]
- Verbeke R, Lentacker I, De Smedt SC, Dewitte H. 2021. The dawn of mRNA vaccines: The COVID-19 case. J Control Release 333:511-520. Epub 2021 Mar 30. [CrossRef]
- De Greef J, Scohy A, Zech F, Aboubakar F, Pilette C, Gerard L, Pothen L, Yildiz H, Belkhir L, Yombi JC. 2021. Determinants of IgG antibodies kinetics after severe and critical COVID-19. J Med Virol 93:5416-5424. Epub 2021 May 12. [CrossRef]
- Wei J, Stoesser N, Matthews PC, Ayoubkhani D, Studley R, Bell I, Bell JI, Newton JN, Farrar J, Diamond I, Rourke E, Howarth A, Marsden BD, Hoosdally S, Jones EY, Stuart DI, Crook DW, Peto TEA, Pouwels KB, Eyre DW, Walker AS, team C-IS. 2021. Antibody responses to SARS-CoV-2 vaccines in 45,965 adults from the general population of the United Kingdom. Nat Microbiol 6:1140-1149. Epub 2021 Jul 21. [CrossRef]
- Choe PG, Hong J, Park J, Chang E, Kang CK, Kim NJ, Lee CH, Park WB, Oh MD. 2022. Persistent Antibody Responses Up to 18 Months After Mild Severe Acute Respiratory Syndrome Coronavirus 2 Infection. J Infect Dis 226:1224-1230. [CrossRef]
- Altarawneh HN, Chemaitelly H, Hasan MR, Ayoub HH, Qassim S, AlMukdad S, Coyle P, Yassine HM, Al-Khatib HA, Benslimane FM, Al-Kanaani Z, Al-Kuwari E, Jeremijenko A, Kaleeckal AH, Latif AN, Shaik RM, Abdul-Rahim HF, Nasrallah GK, Al-Kuwari MG, Butt AA, Al-Romaihi HE, Al-Thani MH, Al-Khal A, Bertollini R, Tang P, Abu-Raddad LJ. 2022. Protection against the Omicron Variant from Previous SARS-CoV-2 Infection. N Engl J Med 386:1288-1290. Epub 2022 Feb 9. [CrossRef]
- Accorsi EK, Britton A, Fleming-Dutra KE, Smith ZR, Shang N, Derado G, Miller J, Schrag SJ, Verani JR. 2022. Association Between 3 Doses of mRNA COVID-19 Vaccine and Symptomatic Infection Caused by the SARS-CoV-2 Omicron and Delta Variants. JAMA 327:639-651. [CrossRef]
- Hoffmann M, Kruger N, Schulz S, Cossmann A, Rocha C, Kempf A, Nehlmeier I, Graichen L, Moldenhauer AS, Winkler MS, Lier M, Dopfer-Jablonka A, Jack HM, Behrens GMN, Pohlmann S. 2022. The Omicron variant is highly resistant against antibody-mediated neutralization: Implications for control of the COVID-19 pandemic. Cell 185:447-456 e11. Epub 2021 Dec 24. [CrossRef]
- Subramanian SV, Kumar A. 2021. Increases in COVID-19 are unrelated to levels of vaccination across 68 countries and 2947 counties in the United States. Eur J Epidemiol 36:1237-1240. Epub 2021 Sep 30. [CrossRef]
- Maslinska M, Dmowska-Chalaba J, Jakubaszek M. 2021. The Role of IgG4 in Autoimmunity and Rheumatic Diseases. Front Immunol 12:787422. eCollection 2021. [CrossRef]
- Irrgang P, Gerling, J., Kocher, K., Lapuente D., Steininger P., Habenicht, K., Wytopil, M., Beileke, S., Schafer S., Zhong J. 2022. Class switch towards non-inflammatory, spike-specific IgG4 antibodies after repeated SARS-CoV-2 mRNA vaccination. Sci Immunol 8:eade2798. Epub 2023 Jan 27. [CrossRef]
- Fernandes-Siqueira LO, Sousa BG, Cleto CE, Wermelinger LS, Caetano BLL, Pacheco AR, Costa SM, Almeida FCL, Ferreira GC, Salmon D, Alves AMB, Da Poian AT. 2022. IgA quantification as a good predictor of the neutralizing antibodies levels after vaccination against SARS-CoV-2. J Clin Virol Plus 2:100121. Epub 2022 Nov 4. [CrossRef]
- Hale M, Netland J, Chen Y, Thouvenel CD, Smith KN, Rich LM, Vanderwall ER, Miranda MC, Eggenberger J, Hao L, Watson MJ, Mundorff CC, Rodda LB, King NP, Guttman M, Gale M, Abraham J, Debley JS, Pepper M, Rawlings DJ. 2022. IgM antibodies derived from memory B cells are potent cross-variant neutralizers of SARS-CoV-2. J Exp Med 219 (9):e20220849. Epub 2022 Aug 8. [CrossRef]
- Kim KO, Jang BI. 2022. Management of inflammatory bowel disease in the COVID-19 era. Intest Res 20(1):3-10. Epub 2021 Feb 3. [CrossRef]
- Alexander JL, Kennedy NA, Ibraheim H, Anandabaskaran S, Saifuddin A, Castro Seoane R, Liu Z, Nice R, Bewshea C, D'Mello A, Constable L, Jones GR, Balarajah S, Fiorentino F, Sebastian S, Irving PM, Hicks LC, Williams HRT, Kent AJ, Linger R, Parkes M, Kok K, Patel KV, Teare JP, Altmann DM, Boyton RJ, Goodhand JR, Hart AL, Lees CW, Ahmad T, Powell N, investigators VIPs. 2022. COVID-19 vaccine-induced antibody responses in immunosuppressed patients with inflammatory bowel disease (VIP): a multicentre, prospective, case-control study. Lancet Gastroenterol Hepatol 7(4):342-352. Epub 2022 Feb 4. [CrossRef]
- Aalberse RC, Schuurman J. 2002. IgG4 breaking the rules. Immunology 105(1):9-19. [CrossRef]
- Aalberse RC, Stapel SO, Schuurman J, Rispens T. 2009. Immunoglobulin G4: an odd antibody. Clin Exp Allergy 39(4):469-77. [CrossRef]
- Huijbers MG, Plomp JJ, van der Maarel SM, Verschuuren JJ. 2018. IgG4-mediated autoimmune diseases: a niche of antibody-mediated disorders. Ann N Y Acad Sci 1413(1):92-103. Epub 2018 Jan 28. [CrossRef]
- Karagiannis P, Gilbert AE, Josephs DH, Ali N, Dodev T, Saul L, Correa I, Roberts L, Beddowes E, Koers A, Hobbs C, Ferreira S, Geh JL, Healy C, Harries M, Acland KM, Blower PJ, Mitchell T, Fear DJ, Spicer JF, Lacy KE, Nestle FO, Karagiannis SN. 2013. IgG4 subclass antibodies impair antitumor immunity in melanoma. J Clin Invest 123(4):1457-74. [CrossRef]
- Karagiannis P, Gilbert AE, Nestle FO, Karagiannis SN. 2013. IgG4 antibodies and cancer-associated inflammation: Insights into a novel mechanism of immune escape. Oncoimmunology 2:e24889. Epub 2013 May 7. [CrossRef]
- Sato Y, Kojima M, Takata K, Morito T, Asaoku H, Takeuchi T, Mizobuchi K, Fujihara M, Kuraoka K, Nakai T, Ichimura K, Tanaka T, Tamura M, Nishikawa Y, Yoshino T. 2009. Systemic IgG4-related lymphadenopathy: a clinical and pathologic comparison to multicentric Castleman's disease. Mod Pathol 22(4):589-99. Epub 2009 Mar 6. [CrossRef]
- Zen Y, Inoue D, Kitao A, Onodera M, Abo H, Miyayama S, Gabata T, Matsui O, Nakanuma Y. 2009. IgG4-related lung and pleural disease: a clinicopathologic study of 21 cases. Am J Surg Pathol 33(12):1886-93. [CrossRef]
- Stone JH, Khosroshahi A, Hilgenberg A, Spooner A, Isselbacher EM, Stone JR. 2009. IgG4-related systemic disease and lymphoplasmacytic aortitis. Arthritis Rheum 60(10):3139-45. [CrossRef]
- Billeskov R, Beikzadeh B, Berzofsky JA. 2019. The effect of antigen dose on T cell-targeting vaccine outcome. Hum Vaccin Immunother 15(2):407-411. Epub 2018 Oct 25. [CrossRef]
- Deaths by Vaccination Status in England. Office for National Statistics. Available online: https://wwwonsgovuk/peoplepopulationandcommunity/birthsdeathsandmarriages/deaths/datasets/deathsbyvaccinationstatusengland.
- Aarstad JK, O.A. 2023. Is There a Link between the 2021 COVID-19 Vaccination Uptake in Europe and 2022 Excess All-Cause Mortality? Asian Pac J Health Sci 10:25-31.
- Uversky VN, Redwan EM, Makis W, Rubio-Casillas A. 2023. IgG4 Antibodies Induced by Repeated Vaccination May Generate Immune Tolerance to the SARS-CoV-2 Spike Protein. Vaccines (Basel) 11 (5):991. [CrossRef]
- Shuai H, Chan JF, Hu B, Chai Y, Yuen TT, Yin F, Huang X, Yoon C, Hu JC, Liu H, Shi J, Liu Y, Zhu T, Zhang J, Hou Y, Wang Y, Lu L, Cai JP, Zhang AJ, Zhou J, Yuan S, Brindley MA, Zhang BZ, Huang JD, To KK, Yuen KY, Chu H. 2022. Attenuated replication and pathogenicity of SARS-CoV-2 B.1.1.529 Omicron. Nature 603(7902):693-699. Epub 2022 Jan 21. [CrossRef]
- Wolter N, Jassat W, Walaza S, Welch R, Moultrie H, Groome M, Amoako DG, Everatt J, Bhiman JN, Scheepers C, Tebeila N, Chiwandire N, du Plessis M, Govender N, Ismail A, Glass A, Mlisana K, Stevens W, Treurnicht FK, Makatini Z, Hsiao NY, Parboosing R, Wadula J, Hussey H, Davies MA, Boulle A, von Gottberg A, Cohen C. 2022. Early assessment of the clinical severity of the SARS-CoV-2 omicron variant in South Africa: a data linkage study. Lancet 399(10323):437-446. Epub 2022 Jan 19. [CrossRef]
| COVID-19 Convalescent No-Vaccinated (Cohort-1) | |
|---|---|
|
04/26/2020 to 06/05/2020 |
|
85 (31 sera and 54 plasma) |
| Days since RT-qPCR confirmation test | |
|
0 to 139 days |
|
35.5 days |
|
26 22 days 32 37.5 days 11 84 days 16 |
| Pre-Pandemic Samples (Cohort-5) | |
| Healthy subjects | |
|
2012 |
|
78 |
| Other respiratory / viral infections | |
|
2018-2019 |
|
47 |
| Cohort-2: Serial samples from previously SARS-CoV-2 infected subjects that received two doses of mRNA vaccine (Pfizer-BioNTech or Moderna-1273) | |
|---|---|
| Number of subjects | 12 |
|
7 female and 5 males |
|
10/27/2020 to 09/20/2021 |
|
36 |
| Sample 1 (Baseline) | Days after infection |
|
|
|
|
| Sample 2 | Days after the 2nd dose |
|
15 to 32 days |
|
21.5 |
| Sample 3 | Days after the 2nd dose |
|
74 to 169 days |
|
96 |
| Cohort-3a: Serial samples from no previous SARS-CoV-2 infected subjects that received multiple Pfizer-BioNTech vaccinations. | |
|
4 |
|
3 female and 1 male |
|
08/10/2020 to 08/10/2023 |
|
26 |
| Sample 1 (baseline) | Previous 1st dose |
| Sample 2 | Days after the 2nd dose |
|
19 to 35 days |
|
20 |
| Sample 3 | Days after the 3rd dose |
|
31 to 43 days |
|
31 |
| Sample 4 | Days after the 3rd dose |
|
180 to 420 days |
|
255 |
| Sample 5 | Days after bivalent vaccine |
|
30 days |
| Sample 6 | Days after bivalent vaccine |
|
90-180 days |
| Median | 180 |
| Cohort-3b: Single sample from no previous SARS-CoV-2 infection multiple Pfizer-BioNTech vaccinations, collected ~ 2 years after the last dose. | |
|
8 |
|
4 female and 4 males |
|
8 |
|
08/03/2023 to 10/23/2023 |
|
09/30/2021 to 12/28/2021 |
| Sample 1 | Days after the 3rd dose |
|
351 to 723 days |
|
634 |
| Cohort-4: Serial samples from no-previous SARS-CoV-2 infected subjects with inflammatory bowel disease (IBD) that received three doses of mRNA vaccine (Pfizer). | |
|
6 |
|
3 female and 3 males. |
|
04/14/2021 to 07/22/2022 |
|
24 |
| Sample 1 (baseline) | Days after 2nd dose |
|
15 to 28 days |
|
17 |
| Sample 2 | Days after 3rd dose |
|
60 |
| Sample 3 | Days after 3rd dose |
|
180 |
| Antibody class/subclass | Neutralization Test | |||
|---|---|---|---|---|
| Total IgG | WT | Alpha | Delta | Omicron |
|
98.87 | 75.29 | 69.41 | 3.529 |
|
0.661 | 0.056 | 0.049 | 0.00057 |
|
Substantial agreement | Slight agreement | Slight agreement | No agreement |
| IgG1 | ||||
|
76.47 | 72.94 | 57.64 | 24.70 |
|
0.050 | 0.268 | -0.038 | -0.016 |
|
Slight agreement | Fair agreement | No agreement | No agreement |
| IgG2 | ||||
|
9.41 | 31.76 | 34.88 | -- |
|
0.002 | 0.032 | -0.014 | No computed |
|
No agreement | Slight agreement | No agreement | -- |
| IgG3 | ||||
|
30.58 | 48.23 | 50.58 | 69.23 |
|
0.018 | 0.151 | 0.147 | 0.006 |
|
Slight agreement | Slight agreement | Slight agreement | No agreement |
| IgG4 | ||||
|
14.11 | 30.588 | 35.29 | -- |
|
0.0064 | 0.011 | -0.026 | No computed |
|
No agreement | Slight agreement | No agreement | -- |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




