Submitted:
09 January 2024
Posted:
10 January 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. CYP enzymes
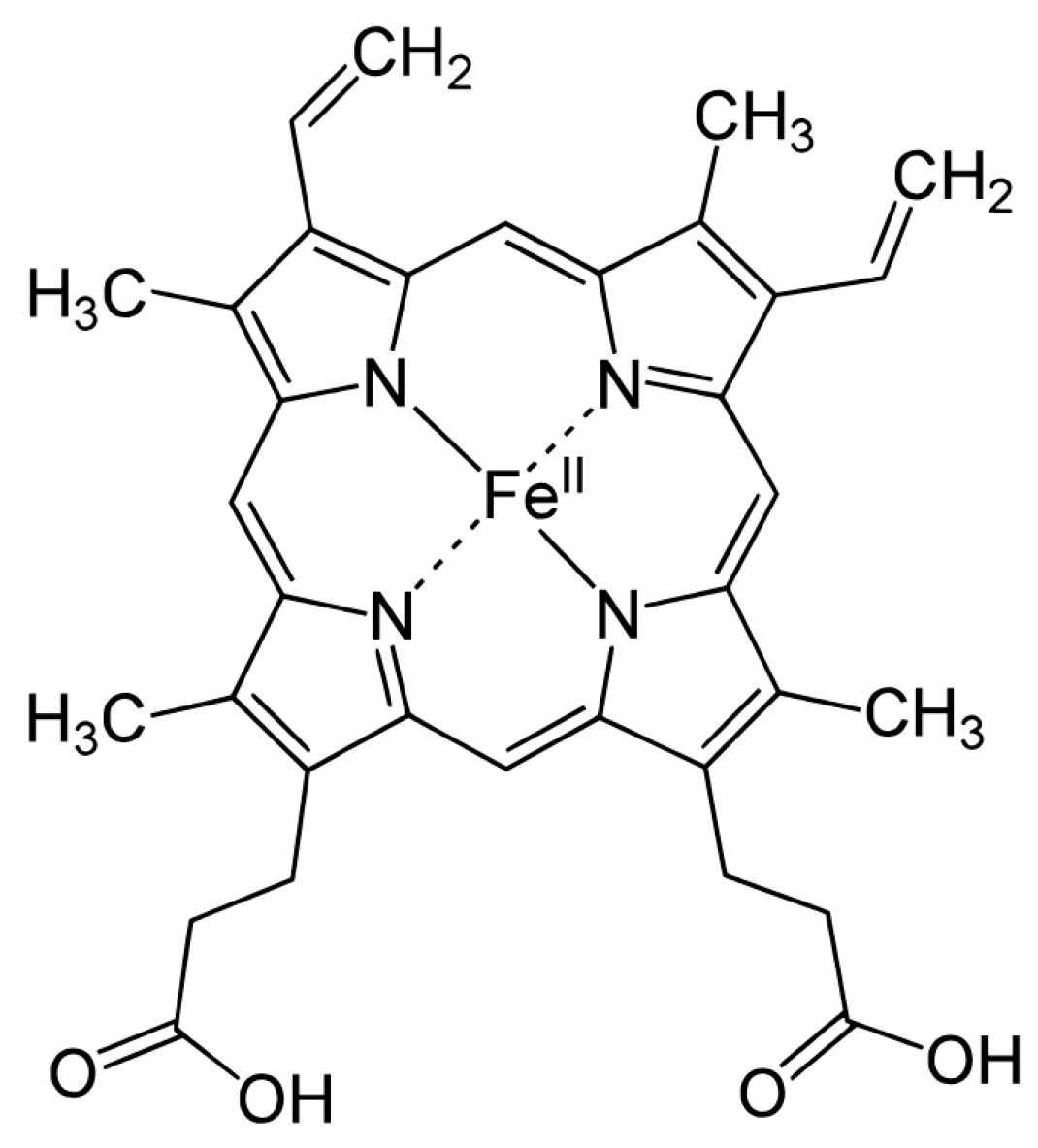
2.1. CYP3A4 enzyme
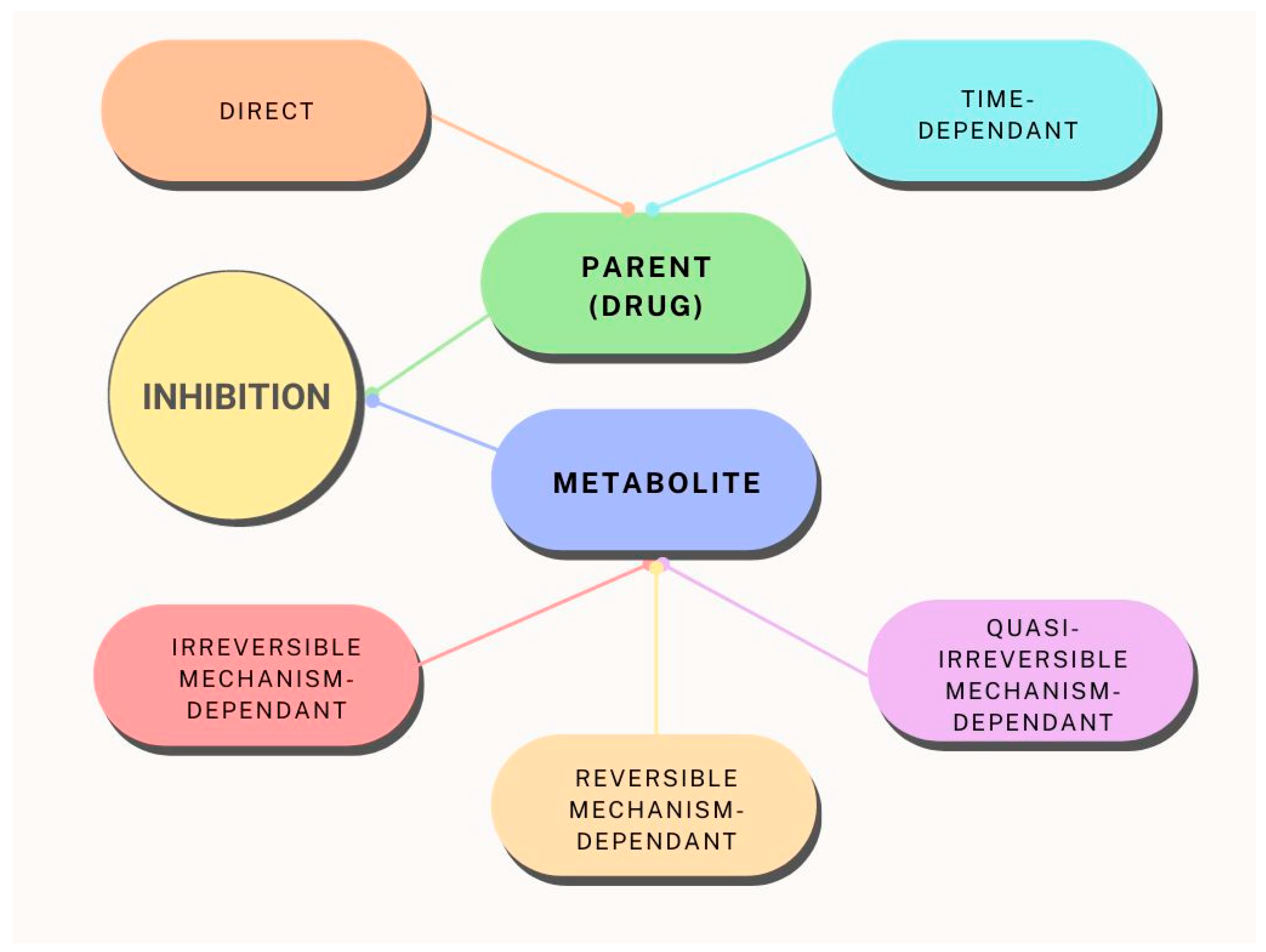
2.2. CYP3A4 inhibitors
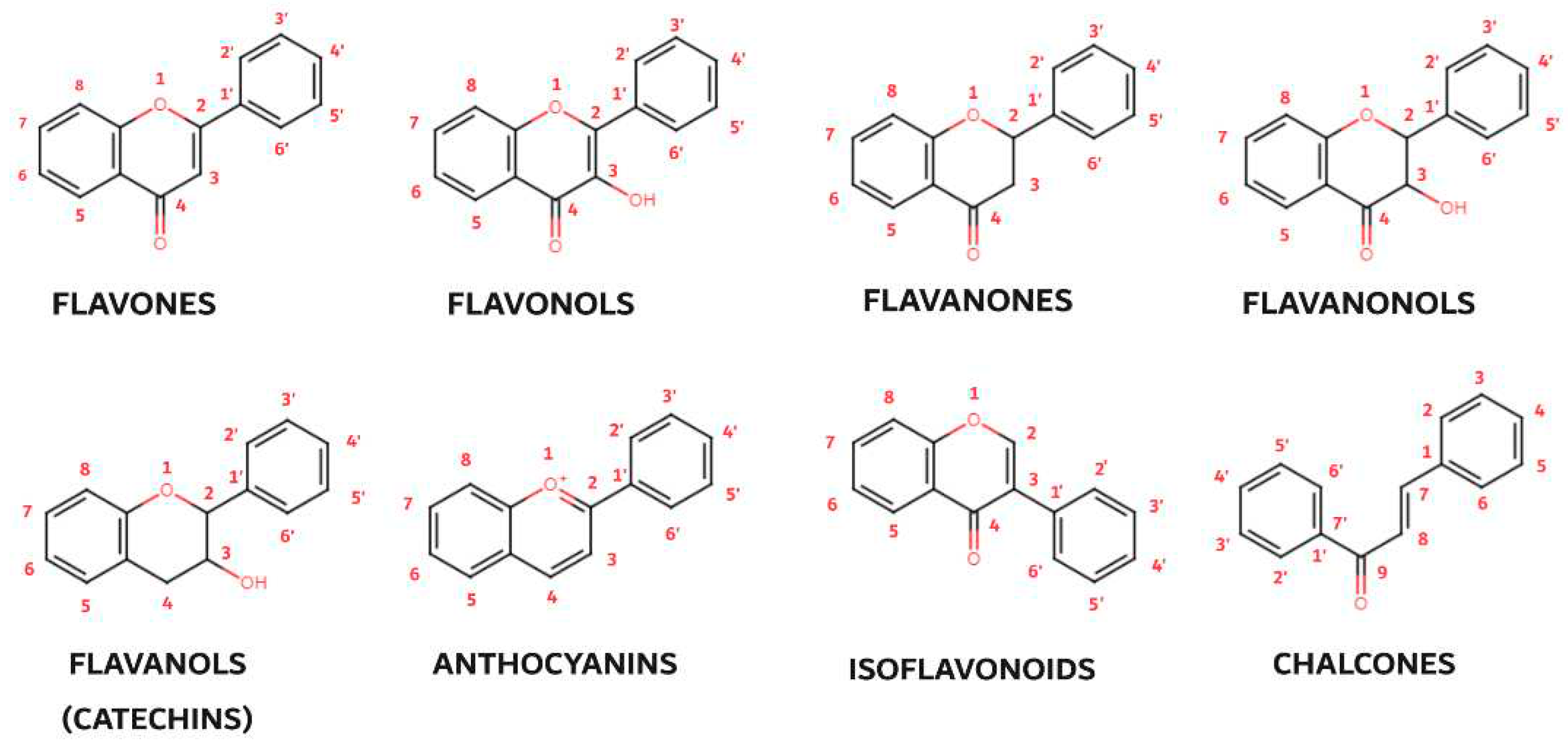
3. Flavonoids
3.1. Flavones
3.2. Flavonols
3.3. Flavanones
3.4. Flavanonols, Flavanols/Catechins, Isoflavonoids and Chalcones
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bailey, D.G.; Dresser, G.K. Natural products and adverse drug interactions. CMAJ 2004, 170(10), 1531–2. [Google Scholar] [CrossRef]
- Ruschitzka, F.; Meier, P.J.; Turina, M.; Luscher, T.T.; Noll, G. Acute heart transplant rejection due to Saint John's wort. Lancet 2000, 355, 548–9. [Google Scholar] [CrossRef]
- Piscitelli, S.C.; Burstein, A.H.; Chaitt, D.; Alfaro, R.M.; Falloon, J. Indinavir concentrations and St John's wort. Lancet 2000, 355, 547–8. [Google Scholar] [CrossRef] [PubMed]
- Kondža, M.; Mandić, M.; Ivančić, I.; Vladimir-Knežević, S.; Brizić, I. Artemisia annua L. Extracts Irreversibly Inhibit the Activity of CYP2B6 and CYP3A4 Enzymes. Biomedicines 2023, 11(1), 232. [Google Scholar] [CrossRef]
- Choudhury, A.; Singh, P.A.; Bajwa, N.; Dash, S.; Bisht, P. Pharmacovigilance of herbal medicines: Concerns and future prospects. J Ethnopharmacol 2023, 309, 116383. [Google Scholar] [CrossRef] [PubMed]
- Górniak, I.; Bartoszewski, R.; Króliczewski, J. Comprehensive review of antimicrobial activities of plant flavonoids. Phytochem. Rev 2019, 18, 241–272. [Google Scholar] [CrossRef]
- Biharee, A.; Sharma, A.; Kumar, A.; Jaitak, V. Antimicrobial flavonoids as a potential substitute for overcoming antimicrobial resistance. Fitoterapia 2020, 146, 104720. [Google Scholar] [CrossRef]
- Donadio, G.; Mensitieri, F.; Santoro, V.; Parisi, V.; Bellone, M.; De Tommasi, N.; Izzo, V.; Piaz, F.D. Interactions with Microbial Proteins Driving the Antibacterial Activity of Flavonoids. Pharmaceutics 2021, 13, 660. [Google Scholar] [CrossRef] [PubMed]
- Song, M.; Liu, Y.; Li, T.; Liu, X.; Hao, Z.; Ding, S.; Panichayupakaranant, P.; Zhu, K.; Shen, J. Plant Natural Flavonoids Against Multidrug Resistant Pathogens. Adv Sci 2021, 8, 2100749. [Google Scholar] [CrossRef] [PubMed]
- Ruddock, P.S.; Charland, M.; Ramirez, S.; López, A.; Towers, G.H.N.; Arnason, J.T.; Liao, M.; Dillon, J.A.R. Antimicrobial Activity of Flavonoids from Piper lanceaefolium and Other Colombian Medicinal Plants Against Antibiotic Susceptible and Resistant Strains of Neisseria gonorrhoeae. Sex Transm Dis 2011, 38, 82–88. [Google Scholar] [CrossRef] [PubMed]
- Bibi, Z. Role of cytochrome P450 in drug interactions. Nutr Metab 2008, 5, 27. [Google Scholar] [CrossRef] [PubMed]
- Manikandan, P.; Nagini, S. Cytochrome P450 Structure, Function and Clinical Significance: A Review. Curr Drug Targets 2018, 19(1), 38–54. [Google Scholar] [CrossRef] [PubMed]
- Coleman, T.; Podgorski, M.N.; Doyle, M.L.; Scaffidi-Muta, J.M.; Campbell, E.C.; Bruning, J.B.; De Voss, J.J.; Bell, S.G. Cytochrome P450-catalyzed oxidation of halogen-containing substrates. J Inorg Biochem 2023, 244, 112234. [Google Scholar] [CrossRef]
- Rendić, S.; Guengerich, F.P. Survey of Human Oxidoreductases and Cytochrome P450 Enzymes Involved in the Metabolism of Xenobiotic and Natural Chemicals. Chem Res Toxicol 2015, 28, 38–42. [Google Scholar] [CrossRef] [PubMed]
- Zhao, M.; Ma, J.; Li, M.; Zhang, Y.; Jiang, B.; Zhao, X.; Huai, C.; Shen, L.; Zhang, N.; He, L.; Qin, S. Cytochrome P450 Enzymes and Drug Metabolism in Humans. Int J Mol Sci 2021, 22(23), 12808. [Google Scholar] [CrossRef]
- Gao, L.; Tu, Y.; Wegman, P.; Wingren, S.; Eriksson, L.A. A mechanistic hypothesis for the cytochrome P450-catalyzed cis-trans isomerization of 4-hydroxytamoxifen: an unusual redox reaction. J Chem Inf Model 2011, 51(9), 2293–301. [Google Scholar] [CrossRef] [PubMed]
- Medić Šarić, M.; Rendić, S. Cytochrome P450 Enzymes. In Metabolizam lijekova i odabranih ksenobiotika, 1st ed.; Medić-Šarić, M., Ed.; Medicinska naklada: Zagreb, Croatia, 2013; p. 136. [Google Scholar]
- Bojić, M. Predklinička ispitivanja inhibicijskog i interakcijskog potencijala novih lijekova na razini citokroma P450. Farm Glas 2015, 71, 229–242. [Google Scholar]
- Danielson, P.B. The cytochrome P450 superfamily: Biochemistry, evolution and drug metabolism in humans. Curr Drug Metab 2002, 3, 561–597. [Google Scholar] [CrossRef]
- Klyushova, L.S.; Perepechaeva, M.L.; Grishanova, A.Y. The Role of CYP3A in Health and Disease. Biomedicines 2022, 10, 2686. [Google Scholar] [CrossRef]
- Stipp, M.C.; Acco, A. Involvement of cytochrome P450 enzymes in inflammation and cancer: A review. Cancer Chemother Pharmacol 2021, 87, 295–309. [Google Scholar] [CrossRef]
- Burlaka, V.S.; Burlaka, A.A. Cytochrome P450 content in primary tumors and liver metastases of patients with metastatic colorectal cancer. Exp Oncol 2020, 42, 1–3. [Google Scholar] [CrossRef] [PubMed]
- Barros-Oliveira, M.D.C.; Costa-Silva, D.R.; Dos Santos, A.R.; Pereira, R.O.; Soares-Júnior, J.M.; Silva, B.B.D. Influence of CYP19A1 gene expression levels in women with breast cancer: A systematic review of the literature. Clinics 2021, 76, e2846. [Google Scholar] [CrossRef] [PubMed]
- Dutour, R.; Poirier, D. Inhibitors of cytochrome P450 (CYP) 1B1. Eur J Med Chem 2017, 135, 296–306. [Google Scholar] [CrossRef] [PubMed]
- Karkhanis, A.; Hong, Y.; Chan, E.C.Y. Inhibition and inactivation of human CYP2J2: Implications in cardiac pathophysiology and opportunities in cancer therapy. Biochem Pharmacol 2017, 135, 12–21. [Google Scholar] [CrossRef] [PubMed]
- Karlgren, M.; Ingelman-Sundberg, M. Tumor-specific expression of CYP2W1: Its potential as a drug target in cancer therapy. Expert Opin Ther Targets 2007, 11, 61–67. [Google Scholar] [CrossRef] [PubMed]
- Finta, C.; Zaphiropoulos, P. The human cytochromeP450 3A locus. Gene evaluation by capture of downstream exons. Gene 2000, 260, 13–23. [Google Scholar] [CrossRef] [PubMed]
- Johnson, T.N.; Tucker, G.T.; Rostami-Hodjegan, A. Development of CYP2D6 and CYP3A4 in the first year of life. CPT 2008, 83(5), 670–1. [Google Scholar] [CrossRef] [PubMed]
- Kudzi, W.; Dodoo, A.N.; Mills, J.J. Genetic polymorphisms in MDR1, CYP3A4 and CYP3A5 genes in a Ghanaian population: a plausible explanation for altered metabolism of ivermectin in humans? BMC Med Genet 2010, 11, 111. [Google Scholar] [CrossRef] [PubMed]
- Shapiro, L.E.; Shear, N.H. Drug interactions: Proteins, pumps, and P-450s. J Am Acad Dermatol 2002, 7, 467–84. [Google Scholar] [CrossRef]
- Rendić, S.; Guengerich, F.P. Survey of Human Oxidoreductases and Cytochrome P450 Enzymes Involved in the Metabolism of Xenobiotic and Natural Chemicals. Chem Res Toxicol 2015, 28, 38–42. [Google Scholar] [CrossRef] [PubMed]
- Zanger, U.M.; Schwab, M. Cytochrome P450 enzymes in drug metabolism: regulation of gene expression, enzyme activities, and impact of genetic variation. Pharmacol Ther 2013, 138(1), 103–41. [Google Scholar] [CrossRef] [PubMed]
- Hirota, T.; Fujita, Y.; Ieiri, I. An updated review of pharmacokinetic drug interactions and pharmacogenetics of statins. Expert Opin Drug Metab Toxicol 2020, 16(9), 809–822. [Google Scholar] [CrossRef] [PubMed]
- Hunt, C.M.; Westerkam, W.R.; Stave, G.M. Effect of age and gender on the activity of human hepatic CYP3A4. Biochem Pharmacol 1992, 44, 275–28. [Google Scholar] [CrossRef] [PubMed]
- Kramlinger, V.M.; Rojas, M.A.; Kanamori, T.; Guengerich, F.P. Cytochrome P450 3A Enzymes Catalyze the O-6-Demethylation of Thebaine, a Key Step in Endogenous Mammalian Morphine Biosynthesis. JBC 2015, 290, 20200–20210. [Google Scholar] [CrossRef] [PubMed]
- Schmiedlin-Ren, P.; Edwards, D.J.; Fitzsimmons, M.E.; He, K.; Lown, K.S.; Woster, P.M.; Rahman, A.; Thummel, K.E.; Fisher, J.M.; Hollenberg, P.F.; Watkins, P.B. Mechanisms of enhanced oral availability of CYP3A4 substrates by grapefruit constituents. Decreased enterocyte CYP3A4 concentration and mechanism-based inactivation by furanocoumarins. Drug Metab Disp 1997, 25, 1228–33. [Google Scholar]
- Chu, V.; Einolf, H.J.; Evers, R.; Kumar, G.; Moore, D.; Ripp, S.; Silva, J.; Sinha, V.; Sinz, M.; Skerjanec, A. In vitro and in vivo induction of cytochrome p450: a survey of the current practices and recommendations: a pharmaceutical research and manufacturers of america perspective. Drug Metab Dispos 2009, 37, 1339–54. [Google Scholar] [CrossRef] [PubMed]
- Gibson, G.G.; Plant, N.J.; Swales, K.E.; Ayrton, A.; El-Sankary, W. Receptor-dependent transcriptional activation of cytochrome P4503A genes: induction mechanisms, species differences and interindividual variation in man. Xenobiotica 2002, 32, 165–206. [Google Scholar] [CrossRef]
- Wang, K.; Chen, S.; Xie, W.; Wan, Y.J. Retinoids induce cytochrome P450 3A4 through RXR/VDR-mediated pathway. Biochem Pharmacol 2008, 75(11), 2204–13. [Google Scholar] [CrossRef]
- Zhang, J.G.; Ho, T.; Callendrello, A.L.; Crespi, C.L.; Stresser, D.M. A multi-endpoint evaluation of cytochrome P450 1A2, 2B6 and 3A4 induction response in human hepatocyte cultures after treatment with β-naphthoflavone, phenobarbital and rifampicin. Drug Metab Lett 2010, 4, 185–194. [Google Scholar] [CrossRef]
- Pascussi, J.M.; Robert, A.; Nguyen, M.; Walrant-Debray, O.; Garabedian, M. Possible involvement of pregnane X receptor-enhanced CYP24 expression in drug-induced osteomalacia. J Clin Invest 2005, 115, 177–186. [Google Scholar] [CrossRef]
- Gibbons, J.A.; de Vries, M.; Krauwinkel, W.; Ohtsu, Y.; Noukens, J.; van der Walt, J.S.; Mol, R.; Mordenti, J.; Ouatas, T. Pharmacokinetic Drug Interaction Studies with Enzalutamide. Clin Pharmacokinet 2015, 54(10), 1057–69. [Google Scholar] [CrossRef] [PubMed]
- Johannessen, S.I.; Landmark, C.J. Antiepileptic drug interactions - principles and clinical implications. Curr Neupharmacol 2010, 8(3), 254–67. [Google Scholar] [CrossRef] [PubMed]
- Fuhr, L.M.; Marok, F.Z.; Hanke, N.; Selzer, D.; Lehr, T. Pharmacokinetics of the CYP3A4 and CYP2B6 Inducer Carbamazepine and Its Drug-Drug Interaction Potential: A Physiologically Based Pharmacokinetic Modeling Approach. Pharmaceutics 2021, 13(2), 270. [Google Scholar] [CrossRef] [PubMed]
- Nallani, S.C.; Glauser, T.A.; Hariparsad, N.; Setchell, K.; Buckley, D.J.; Buckley, A.R.; Desai, P.B. Dose-dependent induction of cytochrome P450 (CYP) 3A4 and activation of pregnane X receptor by topiramate. Epilepsia 2003, 44(12), 1521–8. [Google Scholar] [CrossRef] [PubMed]
- Aquinos, B.M.; García Arabehety, J.; Canteros, T.M.; de Miguel, V.; Scibona, P.; Fainstein-Day, P. Adrenal crisis associated with modafinil use. Medicina 2021, 81(5), 846–849. [Google Scholar] [PubMed]
- Han, E.H.; Kim, H.G.; Choi, J.H.; Jang, Y.J.; Lee, S.S.; Kwon, K.I.; Kim, E.; Noh, K.; Jeong, T.C.; Hwang, Y.P.; Chung, Y.C.; Kang, W.; Jeong, H.G. Capsaicin induces CYP3A4 expression via pregnane X receptor and CCAAT/enhancer-binding protein β activation. Mol Nutr Food Res 2012, 56(5), 797–809. [Google Scholar] [CrossRef] [PubMed]
- Roby, C.A.; Anderson, G.D.; Kantor, E.; Dryer, D.A.; Burstein, A.H. St John's Wort: effect on CYP3A4 activity. Clin Pharmacol Ther 2000, 67(5), 451–7. [Google Scholar] [CrossRef] [PubMed]
- Beunk, L.; Nijenhuis, M.; Soree, B.; de Boer-Veger, N.J.; Buunk, A.M.; Guchelaar, H.J.; Houwink, E.J.F.; Risselada, A.; Rongen, G.A.P.J.M.; van Schaik, R.H.N.; Swen, J.J.; Touw, D.; van Westrhenen, R.; Deneer, V.H.M.; van der Weide, J. Dutch Pharmacogenetics Working Group (DPWG) guideline for the gene-drug interaction between CYP2D6, CYP3A4 and CYP1A2 and antipsychotics. Eur J Hum Genet 2023. Advance online publication. [Google Scholar] [CrossRef]
- Wen, J.; Chen, Y.; Zhao, M.; Hu, W.; Xiao, Y.W. Effects of clarithromycin on the pharmacokinetics of tacrolimus and expression of CYP3A4 and P-glycoprotein in rats. Fundam Clin Pharmacol 2023, 37(4), 843–848. [Google Scholar] [CrossRef] [PubMed]
- Kumaraswami, K.; Katkam, S.K.; Aggarwal, A.; Sharma, A.; Manthri, R.; Kutala, V.K.; Rajasekhar, L. Epistatic interactions among CYP2C19*2, CYP3A4 and GSTP1 on the cyclophosphamide therapy in lupus nephritis patients. Pharmacogenomics 2017, 18(15), 1401–1411. [Google Scholar] [CrossRef] [PubMed]
- Zhou, S.F. Drugs behave as substrates, inhibitors and inducers of human cytochrome P450 3A4. Curr Drug Metab 2008, 9(4), 310–22. [Google Scholar] [CrossRef]
- Bagdasaryan, A.A.; Chubarev, V.N.; Smolyarchuk, E.A.; Drozdov, V.N.; Krasnyuk, I.I.; Liu, J.; Fan, R.; Tse, E.; Shikh, E.V.; Sukocheva, O.A. Pharmacogenetics of Drug Metabolism: The Role of Gene Polymorphism in the Regulation of Doxorubicin Safety and Efficacy. Cancers 2022, 14(21), 5436. [Google Scholar] [CrossRef]
- Bansal, S.; Zamarripa, C.A.; Spindle, T.R.; Weerts, E.M.; Thummel, K.E.; Vandrey, R.; Paine, M.F.; Unadkat, J.D. Evaluation of Cytochrome P450-Mediated Cannabinoid-Drug Interactions in Healthy Adult Participants. Clin Pharmacol Ther 2023, 114(3), 693–703. [Google Scholar] [CrossRef] [PubMed]
- Bilbao-Meseguer, I.; Jose, B.S.; Lopez-Gimenez, L.R.; Gil, M.A.; Serrano, L.; Castaño, M.; Sautua, S.; Basagoiti, A.D.; Belaustegui, A.; Baza, B.; Baskaran, Z.; Bustinza, A. Drug interactions with sunitinib. J Oncol Pharm Pract 2015, 21(1), 52–66. [Google Scholar] [CrossRef] [PubMed]
- Ramos, K.N.; Gregornik, D.; Ramos, K.S. Pharmacogenomics insights into precision pediatric oncology. Curr Opin Pediatr 2021, 33(6), 564–569. [Google Scholar] [CrossRef] [PubMed]
- Enting, R.H.; Hoetelmans, R.M. Antiretroviral drugs and the central nervous system. AIDS. 1998, 12, 1941–55. [Google Scholar] [CrossRef] [PubMed]
- Shen, G.L.; Liang, A.H.; Zhao, Y.; Cao, C.Y.; Liu, T.; Li, C.Y.; Odd, G.N. Interaction between four herb compounds and a western drug by CYP3A4 enzyme metabolism in vitro. China J Chin Mater Med. 2009, 34, 1705–1711. [Google Scholar]
- Qin, M.N.; Liu, R.; Liu, G.F.; Dong, F. Effects of Breviscapines Injections on CYP Activities in Rat Liver Microsomes in vitro. China Pharm 2012, 15, 147–150. [Google Scholar]
- Greenblatt, D.J.; Zhao, Y.; Venkatakrishnan, K.; Duan, S.X.; Harmatz, J.S.; Parent, S.J.; Court, M.H.; von Moltke, L.L. Mechanism of cytochrome P450-3A inhibition by ketoconazole. J Pharm Pharmacol 2011, 63(2), 214–21. [Google Scholar] [CrossRef] [PubMed]
- Deodhar, M.; Al Rihani, S.B.; Arwood, M.J.; Darakjian, L.; Dow, P.; Turgeon, J.; Michaud, V. Mechanisms of CYP450 Inhibition: Understanding Drug-Drug Interactions Due to Mechanism-Based Inhibition in Clinical Practice. Pharmaceutics. 2020, 12(9), 846. [Google Scholar] [CrossRef] [PubMed]
- A Guide to In Vitro CYP Inhibition Studies: Elements of Study Design and Important Considerations in Data Analysis. Available online: https://bioivt.com/resources/articles-publications/a-guide-to-in-vitro-cyp-inhibition-studies-elements-of-study-design-and-important-considerations-in-data-analysis (accessed on 7 December 2023).
- Fowler, S.; Zhang, H. In vitro evaluation of reversible and irreversible cytochrome P450 inhibition: current status on methodologies and their utility for predicting drug-drug interactions. AAPS J 2008, 10(2), 410–24. [Google Scholar] [CrossRef] [PubMed]
- Bojić, M.; Barbero, L.; Dolgos, H.; Freisleben, A.; Galleman, D.; Riva, S.; Guengerich, F.P. Time- and NADPH-dependent inhibition of cytochrome P450 3A4 by the cyclopentapeptide cilengitide: significance of the guanidine group and accompanying spectral changes. Drug Metabol Disp 2014, 4, 1438–1446. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.Y.; Lee, S.Y.; Oh, S.J.; Lee, K.H.; Jung, Y.S.; Kim, S.K. Assesment of drug-drug interactions caused by metabolism-dependent cytochrome P450 inhibition. Chem Biol Interact 2012, 198, 49–56. [Google Scholar] [CrossRef] [PubMed]
- Polasek, T. M.; Elliot, D. J.; Lewis, B. C.; Miners, J. O. An evaluation of potential mechanism-based inactivation of human drug metabolizing cytochromes P450 by monoamine oxidase inhibitors, including isoniazid. Br J Pharmacol Exp Ther 2000, 311(3), 996–1007. [Google Scholar] [CrossRef] [PubMed]
- Jones, D. R.; Ekins, S.; Li, L.; Hall, S. D. Computational approaches that predict metabolic intermediate complex formation with CYP3A4 (+b5). Drug Metab Dispos 2007, 35(9), 1466–1475. [Google Scholar] [CrossRef] [PubMed]
- Hulskotte, E.G.; Feng, H.P.; Xuan, F.; Gupta, S.; van Zutven, M.G.; O'Mara, E.; Wagner, J.A.; Butterton, J.R. Pharmacokinetic evaluation of the interaction between hepatitis C virus protease inhibitor boceprevir and 3-hydroxy-3-methylglutaryl coenzyme A reductase inhibitors atorvastatin and pravastatin. Antimicrob Agents Chemother 2013, 57(6), 2582–8. [Google Scholar] [CrossRef] [PubMed]
- Tinel, M.; Descatoire, V.; Larrey, D.; Loeper, J.; Labbe, G.; Letteron, P.; Pessayre, D. Effects of clarithromycin on cytochrome P-450. Comparison with other macrolides. J Pharmacol Exp Ther 1989, 250(2), 746–751. [Google Scholar] [PubMed]
- Masubuchi, Y.; Ose, A.; Horie, T. Diclofenac-Induced Inactivation of CYP3A4 and Its Stimulation by Quinidine. Drug Metab Dispos, 2002; 30, 10, 1143–1148. [Google Scholar] [CrossRef]
- Hanioka, N.; Ozawa, S.; Jinno, H.; Tanaka-Kagawa, T.; Nishimura, T. ando, M.; Sawada, J. I. Interaction of irinotecan (CPT-11) and its active metabolite 7-ethyl-10-hydroxycamptothecin (SN-38) with human cytochrome P450 enzymes. Drug Metab Dispos 2002, 30(4), 391–396. [Google Scholar] [CrossRef] [PubMed]
- Guengerich, F. P. Mechanism-based inactivation of human liver microsomal cytochrome P-450 IIIA4 by gestodene. Chem Res Toxicol 1990, 3(4), 363–371. [Google Scholar] [CrossRef]
- Iwata, H.; Tezuka, Y.; Kadota, S.; Hiratsuka, A.; Watabe, T. Mechanism-based inactivation of human liver microsomal CYP3A4 by rutaecarpine and limonin from Evodia fruit extract. Drug Metab Pharmacokinet 2005, 20(1), 34–45. [Google Scholar] [CrossRef]
- Watanabe, A.; Nakamura, K.; Okudaira, N.; Okazaki, O.; Sudo, K. I. Risk assessment for drug-drug interaction caused by metabolism-based inhibition of CYP3A using automated in vitro assay systems and its application in the early drug discovery process. Drug Metab Dispos 2007, 35(7), 1232–1238. [Google Scholar] [CrossRef]
- von Moltke, L. L.; Durol, A. L.; Duan, S. X.; Greenblatt, D. J. Potent mechanism-based inhibition of human CYP3A in vitro by amprenavir and ritonavir: comparison with ketoconazole. Eur J Clin Pharmacol 2000, 56(3), 259–261. [Google Scholar] [CrossRef] [PubMed]
- Ernest, C. S.; Hall, S. D.; Jones, D. R. Mechanism-based inactivation of CYP3A by HIV protease inhibitors. J Pharmacol Exp Ther 2005, 312(2), 583–591. [Google Scholar] [CrossRef]
- Guengerich, F.P. Analysis and Characterization of Enzymes and Nucleic Acids Relevant to Toxicology. In Hayes' Principles and Methods of Toxicology, 6th ed.; Hayes, A.W., Kruger, C.L., Eds.; CRC Press: Boca Raton, FL, USA, 2014; pp. 1906–1964. [Google Scholar]
- 78. Shen, N.; Wang, T.; Gan, Q.; Liu, S.; Wang, L.; Jin, B. Plant flavonoids: Classification, distribution, biosynthesis, and antioxidant activity. Food Chem 2022, 383, 132531. [Google Scholar] [CrossRef]
- Panche, A.; Diwan, A.; Chandra, S. Flavonoids: An overview. J Nutr Sci 2016, 5, e47. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Feng, Y.; Yu, S.; Fan, Z.; Li, X.; Li, J.; Yin, H. The Flavonoid Biosynthesis Network in Plants. Int J Mol Sci 2021, 22(23), 12824. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.Q. What about the progress in the synthesis of flavonoid from 2020? Eur J Med Chem 2022, 243, 114671. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Cheng, F.B.; Wu, X.R.; Zhu, W.; Liao, J.W.; Jiang, Y.; Zhang, C.; Niu, W.Y.; Yu, Y.; Duan, H.Q.; Qin, N. Flavonoid derivatives synthesis and anti-diabetic activities. Bioorg Chem 2020, 95, 103501. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Zhao, H.; Chen, L.; Lin, J.; Wang, Z.; Pan, J.; Yang, F.; Ni, X.; Wang, Y.; Wang, Y.; Li, R.; Pi, E.; Wang, S. Multifaceted roles of WRKY transcription factors in abiotic stress and flavonoid biosynthesis. Front Plant Sci 2023, 14, 1303667. [Google Scholar] [CrossRef] [PubMed]
- Mierziak, J.; Kostyn, K.; Kulma, A. Flavonoids as important molecules of plant interactions with the environment. Molecules 2014, 19(10), 16240–65. [Google Scholar] [CrossRef] [PubMed]
- Mathesius, U. Flavonoid Functions in Plants and Their Interactions with Other Organisms. Plants 2018, 7(2), 30. [Google Scholar] [CrossRef]
- Ramaroson, M.-L.; Koutouan, C.; Helesbeux, J.-J.; Le Clerc, V.; Hamama, L.; Geoffriau, E.; Briard, M. Role of Phenylpropanoids and Flavonoids in Plant Resistance to Pests and Diseases. Molecules 2022, 27, 8371. [Google Scholar] [CrossRef]
- Brunetti, C.; Di Ferdinando, M.; Fini, A.; Pollastri, S.; Tattini, M. Flavonoids as antioxidants and developmental regulators: relative significance in plants and humans. Int J Mol Sci 2013, 14(2), 3540–55. [Google Scholar] [CrossRef]
- Thilakarathna, S.; Rupasinghe, H. Flavonoid Bioavailability and Attempts for Bioavailability Enhancement. Nutrients 2013, 5, 3367–3387. [Google Scholar] [CrossRef]
- Aleksandra Kozłowska, D.S.-W. Flavonoids-food sources and health benefits. Rocz Panstw Zakl Hig 2014, 65, 65. [Google Scholar]
- Iwashina, T. Flavonoid properties of five families newly incorporated into the order Caryophyllales (Review). Bull Natl Mus Nat Sci 2013, 39, 25–51. [Google Scholar]
- Camero, C.M.; Germanò, M.P.; Rapisarda, A.; D’Angelo, V.; Amira, S.; Benchikh, F.; Braca, A.; De Leo, M. Anti-angiogenic activity of iridoids from Galium tunetanum. Rev Bras de Farmacogn 2018, 28, 374–377. [Google Scholar] [CrossRef]
- Zhao, L.; Yuan, X.; Wang, J.; Feng, Y.; Ji, F.; Li, Z.; Bian, J. A review on flavones targeting serine/threonine protein kinases for potential anticancer drugs. Bioorganic Med Chem 2019, 27, 677–685. [Google Scholar] [CrossRef] [PubMed]
- Xue, Z.; Wang, J.; Chen, Z.; Ma, Q.; Guo, Q.; Gao, X.; Chen, H. Antioxidant, antihypertensive, and anticancer activities of the flavonoid fractions from green, oolong, and black tea infusion waste. J Food Biochem 2018, 42, e12690. [Google Scholar] [CrossRef]
- Patel, K.; Kumar, V.; Rahman, M.; Verma, A.; Patel, D.K. New insights into the medicinal importance, physiological functions and bioanalytical aspects of an important bioactive compound of foods ‘Hyperin’: Health benefits of the past, the present, the future. Beni-Suef Univ J Basic Appl Sc. 2018, 7, 31–42. [Google Scholar] [CrossRef]
- Gupta, T.; Kataria, R.; Sardana, S. A Comprehensive Review on Current Perspectives of Flavonoids as Antimicrobial Agent. Curr Top Med Chem 2022, 22(6), 425–434. [Google Scholar] [CrossRef] [PubMed]
- Faggio, C.; Sureda, A.; Morabito, S.; Sanches-Silva, A.; Mocan, A.; Nabavi, S.F.; Nabavi, S.M. Flavonoids and platelet aggregation: A brief review. Eur J Pharmacol 2017, 807, 91–101. [Google Scholar] [CrossRef] [PubMed]
- Xie, J.; Xiong, J.; Ding, L.-S.; Chen, L.; Zhou, H.; Liu, L.; Zhang, Z.-F.; Hu, X.-M.; Luo, P.; Qing, L.-S. A efficient method to identify cardioprotective components of Astragali Radix using a combination of molecularly imprinted polymers-based knockout extract and activity evaluation. J Chromatogr A 2018, 1576, 10–18. [Google Scholar] [CrossRef] [PubMed]
- Al-Khayri, J.M.; Sahana, G.R.; Nagella, P.; Joseph, B.V.; Alessa, F.M.; Al-Mssallem, M.Q. Flavonoids as Potential Anti-Inflammatory Molecules: A Review. Molecules 2022, 27(9), 2901. [Google Scholar] [CrossRef] [PubMed]
- Quintal Martínez, J.P.; Segura Campos, M.R. Flavonoids as a therapeutical option for the treatment of thrombotic complications associated with COVID-19. Phytother Res 2023, 37(3), 1092–1114. [Google Scholar] [CrossRef] [PubMed]
- An Overview of Global Flavonoid Intake and its Food Sources. Available online: https://www.intechopen.com/chapters/54289 (accessed on 18 November 2023).
- Hollman, P.C.; Katan, M.B. Dietary flavonoids: intake, health effects and bioavailability. Food Chem Toxicol 1999, 37(9-10), 937–42. [Google Scholar] [CrossRef] [PubMed]
- Zamora-Ros, R.; Knaze, V.; Luján-Barroso, L.; Romieu, I.; Scalbert, A.; Slimani, N.; Hjartåker, A.; Engeset, D.; Skeie, G.; Overvad, K. Differences in dietary intakes, food sources and determinants of total flavonoids between mediterranean and non-mediterranean countries participating in the European Prospective Investigation into Cancer and Nutrition (EPIC) study. Br J Nutr 2013, 109(8), 1498–507. [Google Scholar] [CrossRef]
- Chun, O.K.; Chung, S.J.; Song, W.O. Estimated dietary flavonoid intake and major food sources of US adults. J Nutr 2007, 137(5), 1244–52. [Google Scholar] [CrossRef]
- Kent, K.; Charlton, K.E.; Russell, J.; Mitchell, P.; Flood, V.M. Estimation of flavonoid intake in older Australians: Secondary Data Analysis of the Blue Mountains Eye Study. J Nutr Gerontol Geriatr 2015, 34(4), 388–98. [Google Scholar] [CrossRef]
- Zhang, Z.; He, L.; Liu, Y.; Liu, J.; Su, Y.; Chen, Y. Association between dietary intake of flavonoid and bone mineral density in middle aged and elderly Chinese women and men. Osteoporos Int 2014, 25(10), 2417–25. [Google Scholar] [CrossRef] [PubMed]
- Jun, S.; Shin, S.; Joung, H. Estimation of dietary flavonoid intake and major food sources of Korean adults. Br J Nutr 2016, 115(3), 480–9. [Google Scholar] [CrossRef]
- Chen, T.H.; Liu, J.C.; Chang, J.J.; Tsai, M.F.; Hsieh, M.H.; Chan, P. The in vitro inhibitory effect of flavonoid astilbin on 3-hydroxy-3-methylglutaryl coenzyme A reductase on Vero cells. Zhon Yi Xue Za Zhi 2001, 64(7), 382–7. [Google Scholar] [CrossRef]
- Schewe, T.; Kühn, H.; Sies, H. Flavonoids of cocoa inhibit recombinant human 5-lipoxygenase. J Nutr 2002, 132(7), 1825–9. [Google Scholar] [CrossRef]
- Ribeiro, D.; Freitas, M.; Tomé, S.M.; Silva, A.M.; Laufer, S.; Lima, J.L.; Fernandes, E. Flavonoids inhibit COX-1 and COX-2 enzymes and cytokine/chemokine production in human whole blood. Inflammation 2015, 38(2), 858–70. [Google Scholar] [CrossRef] [PubMed]
- Ferriola, P.C.; Cody, V.; Middleton, E., Jr. Protein kinase C inhibition by plant flavonoids. Kinetic mechanisms and structure-activity relationships. Biochem Pharmacol 1989, 38(10), 1617–24. [Google Scholar] [CrossRef] [PubMed]
- Kondža, M.; Bojić, M.; Tomić, I.; Maleš, Ž.; Rezić, V.; Ćavar, I. Characterization of the CYP3A4 Enzyme Inhibition Potential of Selected Flavonoids. Molecules 2021, 26(10), 3018. [Google Scholar] [CrossRef] [PubMed]
- Kondža, M.; Rimac, H.; Maleš, Ž.; Turčić, P.; Ćavar, I.; Bojić, M. Inhibitory Effect of Acacetin, Apigenin, Chrysin and Pinocembrin on Human Cytochrome P450 3A4. Croat Chem Acta 2020, 93(1), 33–39. [Google Scholar] [CrossRef]
- Cho, Y.A.; Choi, J.S.; Burm, J.P. Effects of the antioxidant baicalein on the pharmacokinetics of nimodipine in rats: a possible role of P-glycoprotein and CYP3A4 inhibition by baicalein. Pharmacol Rep 2011, 63(4), 1066–73. [Google Scholar] [CrossRef] [PubMed]
- Shimada, T.; Tanaka, K.; Takenaka, S.; Murayama, N.; Martin, M.V.; Foroozesh, M.K; Yamazaki, H.; Guengerich, F.P.; Komori, M. Structure-function relationships of inhibition of human cytochromes P450 1A1, 1A2, 1B1, 2C9, and 3A4 by 33 flavonoid derivatives. Chem Res Toxicol 2010, 23(12), 1921–35. [Google Scholar] [CrossRef] [PubMed]
- Scott, L.M.; Durant, P.; Leone-Kabler, S.; Wood, C.E.; Register, T.C.; Townsend, A.; Cline, J.M. Effects of prior oral contraceptive use and soy isoflavonoids on estrogen-metabolizing cytochrome P450 enzymes. J Steroid Biochem Mol Biol 2008, 112(4-5), 179–85. [Google Scholar] [CrossRef] [PubMed]
- Šarić Mustapić, D.; Debeljak, Ž.; Maleš, Ž.; Bojić, M. The Inhibitory Effect of Flavonoid Aglycones on the Metabolic Activity of CYP3A4 Enzyme. Molecules 2018, 23(10), 2553. [Google Scholar] [CrossRef] [PubMed]
- Niwa, T.; Toyota, M.; Kawasaki, H.; Ishii, R.; Sasaki, S. Comparison of the Stimulatory and Inhibitory Effects of Steroid Hormones and α-Naphthoflavone on Steroid Hormone Hydroxylation Catalyzed by Human Cytochrome P450 3A Subfamilies. Biol Pharm Bull 2021, 44(4), 579–584. [Google Scholar] [CrossRef] [PubMed]
- Brahmi, Z.; Niwa, H.; Yamasato, M.; Shigeto, S.; Kusakari, Y.; Sugaya, K.; Onose, J.; Abe, N. Effective cytochrome P450 (CYP) inhibitor isolated from thyme (Thymus saturoides) purchased from a Japanese market. Biosci Biotechnol Biochem 2011, 75(11), 2237–9. [Google Scholar] [CrossRef] [PubMed]
- Meng, M.; Li, X.; Zhang, X.; Sun, B. Baicalein inhibits the pharmacokinetics of simvastatin in rats via regulating the activity of CYP3A4. Pharm Biol 2021, 59(1), 880–883. [Google Scholar] [CrossRef] [PubMed]
- Ho, P.C.; Saville, D.J.; Wanwimolruk, S. Inhibition of human CYP3A4 activity by grapefruit flavonoids, furanocoumarins and related compounds. J Pharm Pharm Sci 2001, 4(3), 217–27. [Google Scholar]
- Li, Y.; Qin, J.; Wu, H.; Xu, Y.; Zhang, L.; Su, K.; Cui, Y.; Wang, H. In vitro inhibitory effect of lysionotin on the activity of cytochrome P450 enzymes. Pharm Biol 2020, 58(1), 695–700. [Google Scholar] [CrossRef]
- Han, Y.L.; Li, D.; Yang, Q.J.; Zhou, Z.Y.; Liu, L.Y.; Li, B.; Lu, J.; Guo, C. In vitro inhibitory effects of scutellarin on six human/rat cytochrome P450 enzymes and P-glycoprotein. Molecules 2014, 19(5), 5748–60. [Google Scholar] [CrossRef] [PubMed]
- Berginc, K.; Milisav, I.; Kristl, A. Garlic flavonoids and organosulfur compounds: impact on the hepatic pharmacokinetics of saquinavir and darunavir. Drug Metab Pharmacokinet 2010, 25(6), 521–30. [Google Scholar] [CrossRef] [PubMed]
- Wei, S.; Ji, H.; Yang, B.; Ma, L.; Bei, Z.; Li, X.; Dang, H.; Yang, X.; Liu, C.; Wu, X.; Chen, J. Impact of chrysosplenetin on the pharmacokinetics and anti-malarial efficacy of artemisinin against Plasmodium berghei as well as in vitro CYP450 enzymatic activities in rat liver microsome. Malar J 2015, 14, 432. [Google Scholar] [CrossRef]
- Zhang, N.; Liu, J.; Chen, Z.; Dou, W. In vitro inhibitory effects of kaempferitrin on human liver cytochrome P450 enzymes. Pharm Biol 2019, 57(1), 571–576. [Google Scholar] [CrossRef]
- Liu, L.; Sun, S.; Rui, H.; Li, X. In vitro inhibitory effects of dihydromyricetin on human liver cytochrome P450 enzymes. Pharm Biol 2017, 55(1), 1868–1874. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.J.; Xia, Z.Y.; Wang, J.M.; Song, X.T.; Wie, J.F.; Kang, W.Y. Effects of Flavonoids in Lysimachia clethroides Duby on the Activities of Cytochrome P450 CYP2E1 and CYP3A4 in Rat Liver Microsomes. Molecules 2016, 21(6), 738. [Google Scholar] [CrossRef] [PubMed]
- Pilipenko, N.; Ropstad, E.; Halsne, R.; Zamaratskaia, G. Effect of Naringenin, Quercetin, and Sesamin on Xenobiotica-Metabolizing CYP1A and CYP3A in Mice Offspring after Maternal Exposure to Persistent Organic Pollutants. Biomed Res Int 2017, 2017, 8472312. [Google Scholar] [CrossRef]
- Alrushaid, S.; Zhao, Y.; Sayre, C.L.; Maayah, Z.H.; Forrest, M.L.; Senadheera, S.N.; Chaboyer, K.; Anderson, H.D.; El-Kadi, A.O.S.; Davies, N.M. Mechanistically elucidating the in vitro safety and efficacy of a novel doxorubicin derivative. Drug Deliv Transl Res 2017, 7(4), 582–597. [Google Scholar] [CrossRef] [PubMed]
- Ekstrand, B.; Rasmussen, M.K.; Woll, F.; Zlabek, V.; Zamaratskaia, G. In vitro gender-dependent inhibition of porcine cytochrome p450 activity by selected flavonoids and phenolic acids. Biomed Res Int 2015, 2015, 387918. [Google Scholar] [CrossRef]
- Yim, D.; Kim, M.J.; Shin, Y.; Lee, S.J.; Shin, J.G.; Kim, D.H. Inhibition of Cytochrome P450 Activities by Sophora flavescens Extract and Its Prenylated Flavonoids in Human Liver Microsomes. Evid Based Complement Alternat Med 2019, 2019, 2673769. [Google Scholar] [CrossRef]
- Song, H.; Wei, C.; Yang, W.; Niu, Z.; Gong, M.; Hu, H.; Wang, H. Alpinetin suppresses CYP3A4, 2C9, and 2E1 activity in vitro. Pharm Biol 2022, 60(1), 1032–1037. [Google Scholar] [CrossRef]
- Lu, W.J.; Ferlito, V.; Xu, C.; Flockhart, D.A.; Caccamese, S. Enantiomers of naringenin as pleiotropic, stereoselective inhibitors of cytochrome P450 isoforms. Chirality 2011, 23(10), 891–6. [Google Scholar] [CrossRef] [PubMed]
- Satoh, T.; Fujisawa, H.; Nakamura, A.; Takahashi, N.; Watanabe, K. Inhibitory Effects of Eight Green Tea Catechins on Cytochrome P450 1A2, 2C9, 2D6, and 3A4 Activities. J Pharm Pharm Sci 2016, 19(2), 188–97. [Google Scholar] [CrossRef]
- Albassam, A.A.; Markowitz, J.S. An Appraisal of Drug-Drug Interactions with Green Tea (Camellia sinensis). Planta Med 2017, 83(6), 496–508. [Google Scholar] [CrossRef]
- Misaka, S.; Kawabe, K.; Onoue, S.; Werba, J.P.; Giroli, M.; Tamaki, S.; Kan, T.; Kimura, J.; Watanabe, H.; Yamada, S. Effects of green tea catechins on cytochrome P450 2B6, 2C8, 2C19, 2D6 and 3A activities in human liver and intestinal microsomes. Drug Metab Pharmacokinet 2013, 28(3), 244–9. [Google Scholar] [CrossRef] [PubMed]
- Monostory, K.; Vereczkey, L.; Lévai, F.; Szatmári, I. Ipriflavone as an inhibitor of human cytochrome P450 enzymes. Br J Pharmacol 1998, 123(4), 605–10. [Google Scholar] [CrossRef] [PubMed]
- Im, Y.; Kim, Y.W.; Song, I.S.; Joo, J.; Shin, J.H.; Wu, Z.; Lee, H.S.; Park, K.H.; Liu, K.H. Effect of TSHAC on human cytochrome P450 activity, and transport mediated by P-glycoprotein. J Microbiol Biotechnol 2012, 22(12), 1659–64. [Google Scholar] [CrossRef] [PubMed]
- Li, G.; Simmler, C.; Chen, L.; Nikolić, D.; Chen, S.N.; Pauli, G.F.; van Breemen, R.B. Cytochrome P450 inhibition by three licorice species and fourteen licorice constituents. Eur J Pharm Sci 2017, 109, 182–190. [Google Scholar] [CrossRef]
| classification | name | amino acid homogeneity |
|---|---|---|
| superfamily | CYP | - |
| family | CYP3 | ≥40% |
| subfamily | CYP3A | ≥55% |
| enzyme | CYP3A4 | ≥98% |
| drug | reversible inhibitor | mechanism of inhibition | reference |
|---|---|---|---|
| amiodarone | yes | protein | 66 |
| amlodipine | yes | heme | 67 |
| boceprevir | no | heme | 68 |
| clarithromycin | no | heme | 69 |
| diclofenac | yes | protein | 70 |
| diltiazem | yes | heme | 65 |
| irinotecan | no | protein | 71 |
| levonorgestrel | yes | heme & protein | 72 |
| oleandomycin | no | heme | 73 |
| prazosin | yes | protein | 74 |
| ritonavir | yes | heme | 74 |
| sertralin | yes | protein | 75 |
| saquinavir | yes | protein | 76 |
| flavonoid | substrate | material | IC50 (μM) | potency [58] | substituents | REF | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| R3 | R5 | R6 | R7 | R8 | R2’ | R3’ | R4’ | R5’ | ||||||
| 2’-methoxy-5,7-dihydroxyflavone | MDM | CYP3A4 | 2 | intermediate | - | OH | - | OH | - | OCH3 | - | - | - | 114 |
| 3’4’-dimethoxy-5,7-dihydroxyflavone | MDM | CYP3A4 | 6.5 | intermediate | - | OH | - | OH | - | - | OCH3 | OCH3 | - | 114 |
| 3’-methoxy-5,7-dihydroxyflavone | MDM | CYP3A4 | 16 | intermediate | - | OH | - | OH | - | - | OCH3 | - | - | 114 |
| 3-hydroxyflavone | TST | HLM | 66.9 ± 4.0 | weak | OH | - | - | - | - | - | - | - | - | 115 |
| 5-hydroxyflavone | TST | HLM | 103.82 ± 0.98 | weak | - | OH | - | - | - | - | - | - | - | 115 |
| 7-hydroxyflavone | TST | HLM | 66.2 ± 5.8 | weak | - | - | - | OH | - | - | - | - | - | 115 |
| acacetin | NFD | CYP3A4 | 7.5 ± 2.7 | intermediate | - | OH | - | OH | - | - | - | OCH3 | - | 111 |
| acacetin | TST | CYP3A4 | 10.9 ± 0.3 | intermediate | 111 | |||||||||
| acacetin | MDM | CYP3A4 | 6.5 | intermediate | 114 | |||||||||
| acacetin | TST | CYP3A4 | 10.9 ± 0.3 | intermediate | 112 | |||||||||
| acacetin | TST | HLM | 6.25 ± 0.96 | intermediate | 115 | |||||||||
| apigenin | NFD | CYP3A4 | 8.4 ± 1.1 | intermediate | - | OH | - | OH | - | - | - | OH | - | 111 |
| apigenin | TST | CYP3A4 | 11.4 ± 0.4 | intermediate | 111 | |||||||||
| apigenin | MDM | CYP3A4 | 2.3 ± 0.3 | intermediate | 114 | |||||||||
| apigenin | TST | CYP3A4 | 11.4 ± 0.4 | intermediate | 112 | |||||||||
| apigenin | TST | CYP3A4 | 30.8 ± 7.6 | intermediate | 118 | |||||||||
| apigenin | TST | HLM | 1.47 ± 0.24 | intermediate | 115 | |||||||||
| baicalein | BTC | BAC | 9.2 | intermediate | - | OH | OH | OH | - | - | - | - | - | 113 |
| baicalein | TST | RLM | 12.03 | intermediate | 119 | |||||||||
| baicalein | TST | HLM | 9.60 ± 1.18 | intermediate | 115 | |||||||||
| chrysin | NFD | CYP3A4 | 2.5 ± 0.6 | intermediate | - | OH | - | OH | - | - | - | - | - | 111 |
| chrysin | TST | CYP3A4 | 0.6 ± 0.5 | strong | 111 | |||||||||
| chrysin | QIN | HLM | 70 | weak | 120 | |||||||||
| chrysin | MDM | CYP3A4 | 7.4 ± 1.1 | intermediate | 114 | |||||||||
| chrysin | TST | CYP3A4 | 0.6 ± 0.5 | strong | 112 | |||||||||
| chrysin | TST | CYP3A4 | 94.7 ± 30.9 | weak | 118 | |||||||||
| chrysin | TST | HLM | 3.76 ± 1.13 | intermediate | 115 | |||||||||
| daidzein | TST | HLM | >100 | weak | - | - | OH | - | - | - | - | OH | - | 115 |
| continued | ||||||||||||||
| diosmetin | TST | HLM | 58.6 ± 26.5 | intermediate | - | OH | - | OH | - | - | OH | OCH3 | - | 115 |
| flavone | QIN | HLM | 224 | weak | - | - | - | - | - | - | - | - | - | 120 |
| flavone | TST | HLM | >100 | weak | 115 | |||||||||
| luteolin | TST | CYP3A4 | 57.7 ± 16.1 | weak | - | OH | - | OH | - | - | OH | OH | - | 118 |
| luteolin | TST | HLM | 4.62 ± 1.26 | intermediate | 115 | |||||||||
| lysionotin | TST | HLM | 13.85 | intermediate | - | OH | OCH3 | OH | OCH3 | - | - | OCH3 | - | 121 |
| nobiletin | TST | CYP3A4 | 20.6 ± 5.2 | intermediate | - | OCH3 | OCH3 | OCH3 | OCH3 | - | - | OCH3 | OCH3 | 118 |
| scutellarin | MDM | HLM | >100 | weak | - | OH | OH | O-GRD | - | - | - | OH | - | 122 |
| scutellarin | MDM | RLM | >100 | weak | 122 | |||||||||
| α-naphthoflavone 2’-propargyl ether | MDM | CYP3A4 | 64 | weak | - | - | - | benz. R8 | benz. R7 | PGE | - | - | - | 114 |
| α-naphthoflavone 4’-propargyl ether | MDM | CYP3A4 | 55 | weak | - | - | - | benz. R8 | benz. R7 | - | - | PGE | - | 114 |
| α-naphtoflavone | MDM | CYP3A4 | 18 | intermediate | - | - | - | benz. R8 | benz. R7 | - | - | - | - | 114 |
| flavonoid | substrate | material | conc. (μM) | CYP3A4 residual activity (%) | substituents | REF | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| R3 | R5 | R6 | R7 | R8 | R2’ | R3’ | R4’ | R5’ | ||||||
| acacetin | TST | BAC | 100 | 5 ± 4 | - | OH | - | OH | - | - | - | OCH3 | - | 116 |
| apigenin | TST | BAC | 100 | 24 ± 3 | - | OH | - | OH | - | - | - | OH | - | 116 |
| apigenin | QIN | HLM | 100 | 63.6 | 120 | |||||||||
| baicalein | BTC | BAC | 10 | 50 | - | OH | OH | OH | - | - | - | - | - | 113 |
| baicalein | TST | RLM | 100 | 20 | 119 | |||||||||
| chrysin | TST | BAC | 100 | 17 ± 3 | - | OH | - | OH | - | - | - | - | - | 116 |
| chrysin | QIN | HLM | 200 | 44.6 | 120 | |||||||||
| chrysin dimethylether | TST | BAC | 100 | 61 ± 21 | - | OCH3 | - | OCH3 | - | - | - | - | - | 116 |
| flavone | QIN | HLM | 100 | 87.7 | - | - | - | - | - | - | - | - | - | 120 |
| flavone | QIN | HLM | 200 | 60.4 | 120 | |||||||||
| lysionotin | TST | HLM | 100 | 13.67 | - | OH | OCH3 | OH | OCH3 | - | - | OCH3 | - | 121 |
| nobiletin | SAQM1 | HLM | 1 | 83.7 ± 4.1 | - | OCH3 | OCH3 | OCH3 | OCH3 | - | - | OCH3 | OCH3 | 123 |
| nobiletin | SAQM3 | HLM | 1 | 85.2 ± 4.6 | 123 | |||||||||
| rutin | SAQM1 | HLM | 1 | 81.7 ± 1.4 | O-G-R | OH | - | OH | - | - | OH | OH | - | 123 |
| rutin | SAQM3 | HLM | 1 | 83.0 ± 3.7 | 123 | |||||||||
| tangeretin | TST | BAC | 1 | 42 ± 3 | - | OCH3 | OCH3 | OCH3 | OCH3 | - | - | OCH3 | - | 116 |
| tangeretin | SAQM1 | HLM | 1 | 89.0 ± 2.0 | 123 | |||||||||
| tangeretin | SAQM3 | HLM | 1 | 85.3 ± 6.1 | 123 | |||||||||
| α-naphtoflavone | TST | HLM | 100 | 6.8 | - | - | - | benz. R8 | benz. R7 | - | - | - | - | 117 |
| flavonoid | substrate | material | IC50 (μM) | potency [58] | substituents | REF | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| R3 | R5 | R6 | R7 | R2’ | R3’ | R4’ | R5’ | R5’ | ||||||
| chrysosplenetin | MDM | RLM | 3.38 | intermediate | - | OH | OCH3 | OCH3 | - | OCH3 | - | - | - | 124 |
| dihydromyricetin | TST | HLM | 14.75 | intermediate | - | OH | - | OH | - | OH | OH | OH | - | 126 |
| fisetin | QIN | HLM | 44 | intermediate | - | - | - | OH | - | - | OH | OH | - | 120 |
| fisetin | TST | CYP3A4 | 40.7 ± 7.4 | intermediate | 118 | |||||||||
| galangin | QIN | HLM | 117 | weak | - | OH | - | OH | - | - | - | - | - | 120 |
| galangin | MDM | CYP3A4 | 2.3 | intermediate | 114 | |||||||||
| kaempferitrin | TST | HLM | 13.87 | intermediate | R | OH | - | O-R | - | - | OH | - | - | 125 |
| kaempferol | QIN | HLM | 223 | weak | - | OH | - | OH | - | - | OH | - | - | 120 |
| kaempferol | TST | CYP3A4 | 32.65 ± 1.32 | intermediate | 127 | |||||||||
| kaempferol | TST | CYP3A4 | 18.3 ± 5.3 | intermediate | 118 | |||||||||
| kaempferol | TST | HLM | 6.51 ± 1.01 | intermediate | 115 | |||||||||
| morin | QIN | HLM | 75.2 ± 8.7 | weak | - | OH | - | OH | OH | - | OH | - | - | 115 |
| myricetin | TST | HLM | 10.7 ± 2.2 | intermediate | - | OH | - | OH | - | OH | OH | OH | - | 115 |
| quercetin | TST | CYP3A4 | 28.0 ± 5.2 | intermediate | - | OH | - | OH | - | OH | OH | - | - | 127 |
| quercetin | TST | CYP3A4 | 28.0 ± 5.2 | intermediate | 118 | |||||||||
| quercetin | MDM | RLM | 27.1 | intermediate | 128 | |||||||||
| quercetin | TST | HLM | 5.74 ± 1.16 | intermediate | 115 | |||||||||
| flavonoid | substrate | material | conc. (μM) | CYP3A4 residual activity (%) | substituents | REF | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| R3 | R5 | R6 | R7 | R8 | R2’ | R3’ | R4’ | R5’ | ||||||
| astragalin | TST | CYP3A4 | 5 | 93.35 | G | OH | - | OH | - | - | - | OH | - | 127 |
| chrysosplenetin | MDM | RLM | 50 | 18.27 ± 14.55 | - | OH | OCH3 | OCH3 | - | OCH3 | - | - | - | 124 |
| dihydromyricetin | TST | HLM | 100 | 18 | - | OH | - | OH | - | OH | OH | OH | - | 126 |
| doxorubicine-quercetine conjugate | QIN | CYP3A4 | 100 | 50 | - | OH | - | OH | - | - | OH | D | - | 129 |
| fisetin | QIN | HLM | 100 | 27.5 | - | - | - | OH | - | - | OH | OH | - | 120 |
| galangin | QIN | HLM | 100 | 45.5 | - | OH | - | OH | - | - | - | - | - | 120 |
| isoquercetin | TST | CYP3A4 | 5 | 83.32 | G | OH | - | OH | - | - | OH | OH | - | 127 |
| isorahmetin | TST | BAC | 100 | 73 ± 6 | - | OH | - | OH | - | - | OCH3 | OH | - | 116 |
| isorahmetin | BTC | PLM | 128 | 60 | 130 | |||||||||
| kaempferitrin | TST | HLM | 100 | 18 | R | OH | - | O-R | - | - | OH | - | - | 125 |
| kaempferol | QIN | HLM | 100 | 58.6 | - | OH | - | OH | - | - | OH | - | - | 120 |
| kaempferol | TST | CYP3A4 | 5 | 73.8 | 127 | |||||||||
| morin | QIN | HLM | 188 | 65.6 | - | OH | - | OH | OH | - | OH | - | - | 120 |
| myricetin | QIN | HLM | 100 | 6.4 | - | OH | - | OH | - | OH | OH | OH | - | 120 |
| myricetin | BTC | PLM | 128 | 60 | 130 | |||||||||
| quercetin | QIN | HLM | 100 | 35.1 | - | OH | - | OH | - | OH | OH | - | - | 120 |
| quercetin | TST | CYP3A4 | 5 | 66.82 | 127 | |||||||||
| quercetin | SAQ | HLM | 1 | 90.6 ± 0.5 | 123 | |||||||||
| flavonoid | substrate | material | IC50 (μM) | potency [58] | substituents | REF | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| R3 | R5 | R6 | R7 | R8 | R2’ | R3’ | R4’ | R5’ | R6’ | ||||||
| (R)-naringenin | TST | HLM | 9.7 | intermediate | - | OH | - | OH | - | - | - | OH | - | - | 133 |
| (S)-naringenin | TST | HLM | 21.4 | intermediate | - | OH | - | OH | - | - | - | OH | - | - | 133 |
| 7-Hydroxyflavanone | TST | HLM | 53.5 ± 7.2 | weak | - | - | - | OH | - | - | - | - | - | 115 | |
| alpinetin | TST | HLM | 8.23 | intermediate | - | OCH3 | - | OH | - | - | - | - | - | 132 | |
| eriodictyol | TST | HLM | 65.2 ± 8.0 | weak | - | OH | - | OH | - | - | OH | OH | - | - | 115 |
| flavanone | TST | HLM | 101 ± 15 | weak | - | - | - | - | - | - | - | - | - | - | 115 |
| hesperetin | QIN | HLM | 163 | weak | - | OH | - | OH | - | - | - | OCH3 | OH | - | 120 |
| hesperetin | TST | HLM | 61.8 ± 1.3 | weak | 115 | ||||||||||
| hesperidin | QIN | HLM | 601 | weak | - | OH | - | O-G-R | - | - | OH | OCH3 | - | - | 120 |
| kushenol A | MDM | HLM | >50 | weak | - | OH | - | OH | MPH | - | - | - | - | OH | 131 |
| kushenol C | MDM | HLM | 3.95 | intermediate | - | OH | - | OH | MPH | OH | - | OH | - | - | 131 |
| kushebol I | MDM | HLM | 0.57 | strong | OH | OCH3 | - | OH | MPH | OH | - | OH | - | - | 131 |
| kushenol M | MDM | HLM | 1.29 | intermediate | OH | OH | MP | OH | MPH | OH | - | OH | - | - | 131 |
| leachianone A | MDM | HLM | 0.69 | strong | - | OH | OH | MPH | - | - | OH | - | OCH3 | 131 | |
| naringenin | MDM | RLM | 40.3 | intermediate | - | OH | - | OH | - | - | - | OH | - | - | 128 |
| naringenin | QIN | HLM | 139 | weak | 120 | ||||||||||
| naringenin | TST | HLM | 17.6 | intermediate | 133 | ||||||||||
| naringenin | TST | HLM | 6.50 ± 1.54 | intermediate | 115 | ||||||||||
| naringin | QIN | HLM | 1349 | weak | - | OH | O-R-G | - | - | - | OH | - | - | 120 | |
| neohesperidin | QIN | HLM | 280 | weak | - | OH | O-R-G | - | - | OH | OCH3 | - | - | 120 | |
| pinocembrin | NIF | CYP3A4 | 4.3 ± 1.1 | intermediate | - | OH | - | OH | - | - | - | - | - | - | 111 |
| pinocembrin | TST | CYP3A4 | 5.0 ± 0.6 | intermediate | 111 | ||||||||||
| pinocembrin | TST | CYP3A4 | 5.0 ± 0.6 | intermediate | 112 | ||||||||||
| pinocembrin | TST | HLM | 28.6 ± 6.1 | intermediate | 115 | ||||||||||
| sophoraflavone G | MDM | HLM | 0.78 | strong | - | OH | - | O-G | - | - | - | OH | - | - | 131 |
| flavonoid | substrate | material | conc. (μM) | CYP3A4 residual activity (%) | substituents | REF | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| R3 | R5 | R6 | R7 | R8 | R2’ | R3’ | R4’ | R5’ | R6’ | ||||||
| alpinetin | TST | HLM | 100 | 20 | - | OCH3 | - | OH | - | - | - | - | - | 132 | |
| hesperetin | QIN | HLM | 100 | 64.5 | - | OH | - | OH | - | - | - | OCH3 | OH | - | 120 |
| hesperidin | QIN | HLM | 100 | 79.6 | - | OH | - | O-G-R | - | - | OH | OCH3 | - | - | 120 |
| naringenin | QIN | HLM | 100 | 60.9 | - | OH | - | OH | - | - | - | OH | - | - | 120 |
| naringenin | SAQ | HLM | 1 | 77.0 ± 1.1 | 123 | ||||||||||
| naringin | QIN | HLM | 100 | 51.1 | - | OH | O-R-G | - | - | - | OH | - | - | 120 | |
| naringin | SAQ | HLM | 1 | 76.7 ± 0.3 | 123 | ||||||||||
| neohesperidin | QIN | HLM | 200 | 61.7 | - | OH | O-R-G | - | - | OH | OCH3 | - | - | 120 | |
| pinocembrin | TST | BAC | 100 | 50 ± 15 | - | OH | - | OH | - | - | - | - | - | - | 116 |
| prunin | QIN | HLM | 100 | 73.9 | OH | O-G | OH | 120 | |||||||
| flavonoid | substrate | material | IC50 (μM) | potency [58] | substituents | REF | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| R2 | R3 | R4 | R5 | R6 | R7 | R8 | R3’ | R4’ | R5’ | ||||||
| FLAVANONOLS | |||||||||||||||
| taxifolin | TST | HLM | 68.9 ± 2.3 | weak | - | - | - | OH | - | OH | - | - | - | - | 115 |
| FLAVANOLS (CATECHINS) | |||||||||||||||
| (-)-catechin-3-O-gallate | MDM | HLM | 71.5 ± 7.51 | weak | - | GA | - | OH | - | OH | - | - | OH | OH | 134 |
| (-)-epigallocatechin-3-gallate | MDM | BAC | 41 | intermediate | - | GA | - | OH | - | OH | - | OH | OH | OH | 135 |
| (-)-epigallocatechin-3-gallate | MDM | HLM | 23 | intermediate | 135 | ||||||||||
| (-)-epigallocatechin-3-gallate | MDM | HLM | 23.3 | intermediate | 136 | ||||||||||
| (-)-epigallocatechin-3-O-gallate | MDM | HLM | 23.7 ± 1.54 | intermediate | - | O-GA | - | OH | - | OH | - | OH | OH | OH | 134 |
| (-)-epigallocatechin-3-O-gallate | MDM | HLM | 40.8 ± 6.93 | intermediate | 134 | ||||||||||
| ISOFLAVONOIDS | |||||||||||||||
| 7-hydroxy-isoflavone | NIF | HLM | 129.5 ± 12.49 | weak | - | - | - | - | - | OH | - | - | - | - | 137 |
| 8-methoxycirsilineol | TST | CYP3A4 | 1.71 ± 0.3 | intermediate | - | - | OH | OCH3 | OCH3 | OCH3 | OH | - | - | OCH3 | 118 |
| genistein | TST | HLM | >100 | weak | - | - | - | OH | - | OH | - | - | OH | - | 115 |
| CHALCONES | |||||||||||||||
| TSAHC | MDM | HLM | >20 | intermediate | - | - | OH | - | - | - | - | - | TSA | - | 138 |
| licochalcone A | MDM | HLM | 47.4 | intermediate | OCH3 | OH | MBE | OH | 139 | ||||||
| flavonoid | substrate | material | conc. (μM) | CYP3A4 residual activity (%) | substituents | REF | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| R2 | R3 | R4 | R5 | R6 | R7 | R8 | R3’ | R4’ | R5’ | ||||||
| FLAVANOLS (CATECHINS) | |||||||||||||||
| (-)-epicatechin | IRT | HLM | 100 | 53 | - | OH | - | OH | - | OH | - | - | OH | OH | 135 |
| (-)-epigallocatechin-3-gallate | IRT | HLM | 100 | 53 | - | GA | - | OH | - | OH | - | OH | OH | OH | 135 |
| ISOFLAVONOIDS | |||||||||||||||
| 7-hydroxy-isoflavone | NIF | HLM | 100 | 75 | - | - | - | - | - | OH | - | - | - | - | 137 |
| CHALCONES | |||||||||||||||
| N9 | MDM | HLM | 100 | 75 | - | OCH3 | - | - | OCH3 | - | - | pyr.R4’ | pyr.R3’ | - | 138 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




