Submitted:
09 January 2024
Posted:
10 January 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
2.1. Patients’ Characteristics
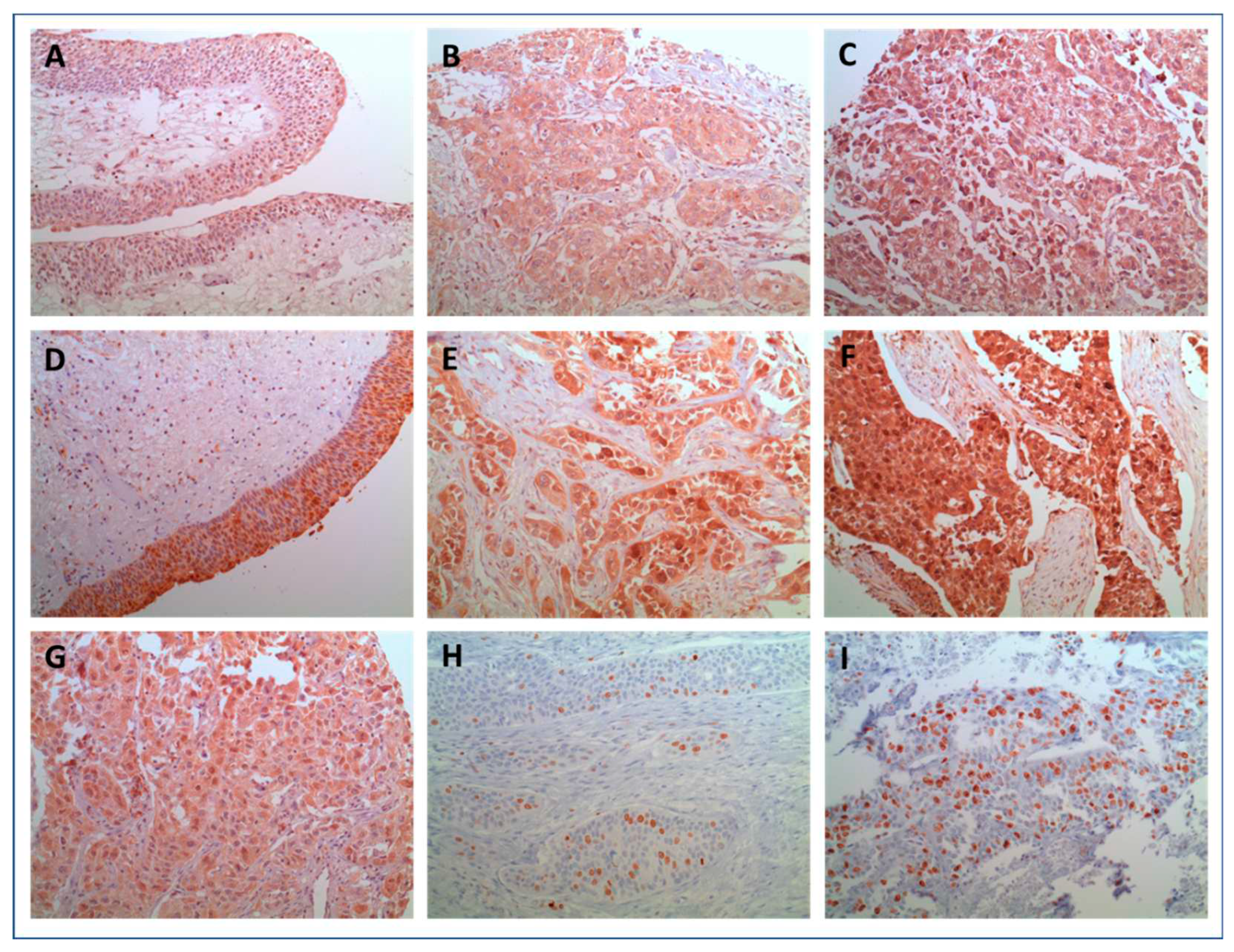
2.2. Expression Characteristics of IL-1β and IL1RA
2.3. Correlation of IL-1 Axis with histopathologic Parameters
2.4. Combined Scores and Clinical Data
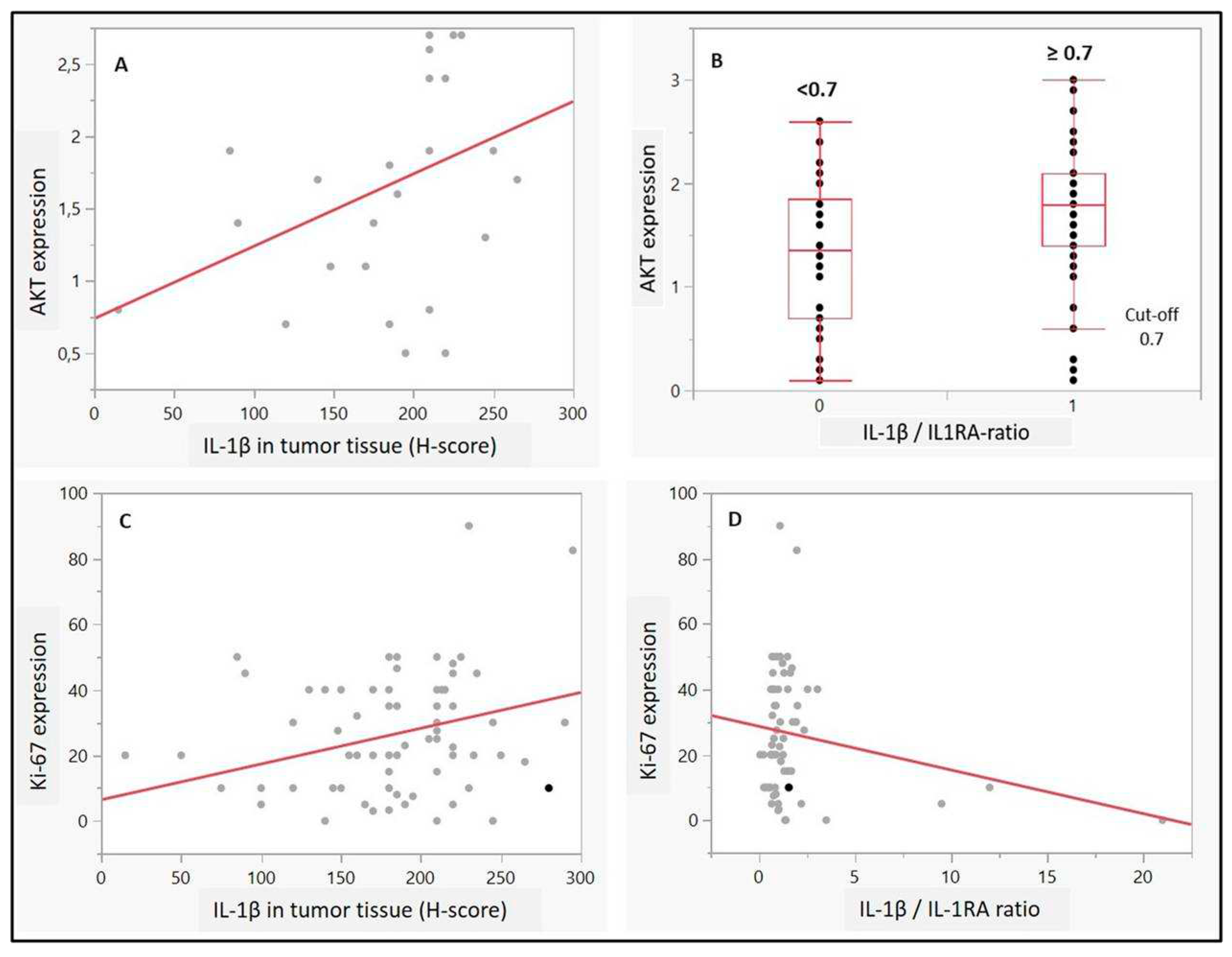
2.5. Correlation of IL-1 Axis with Ki-67 and AKT Expression
2.6. Survival Analysis
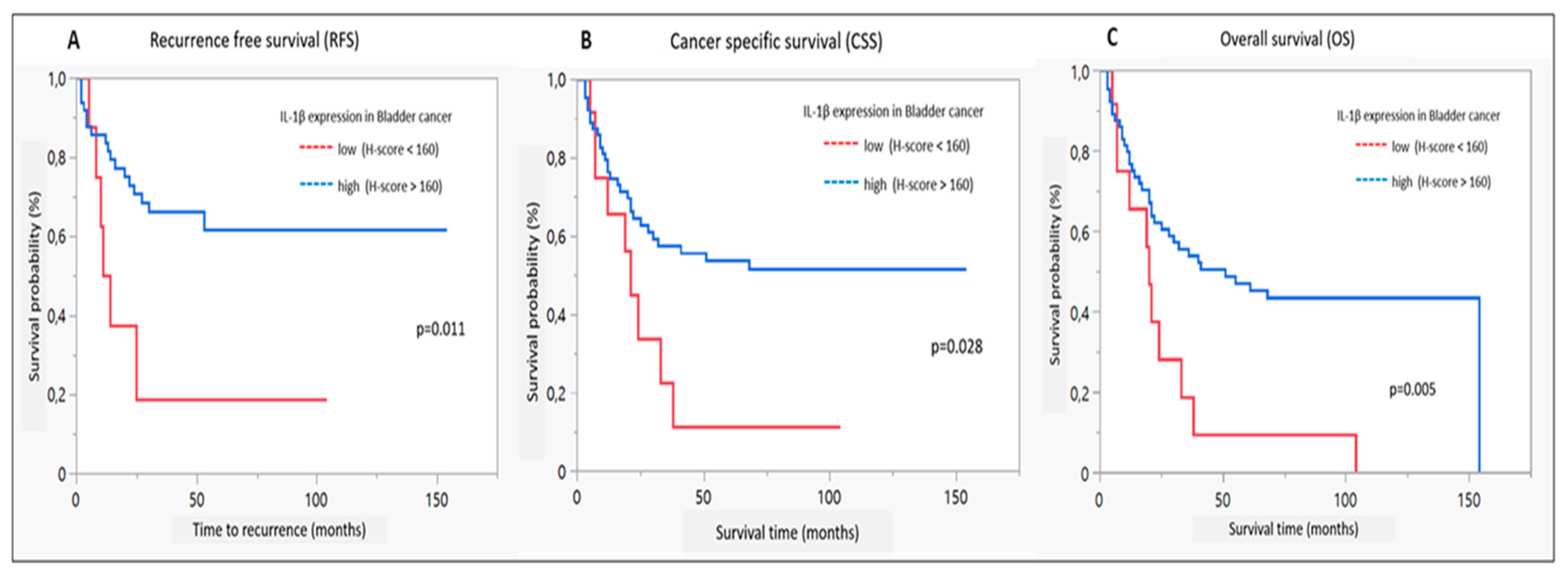
2.7. Uni- and Multivariate Cox Proportional Hazard Analysis
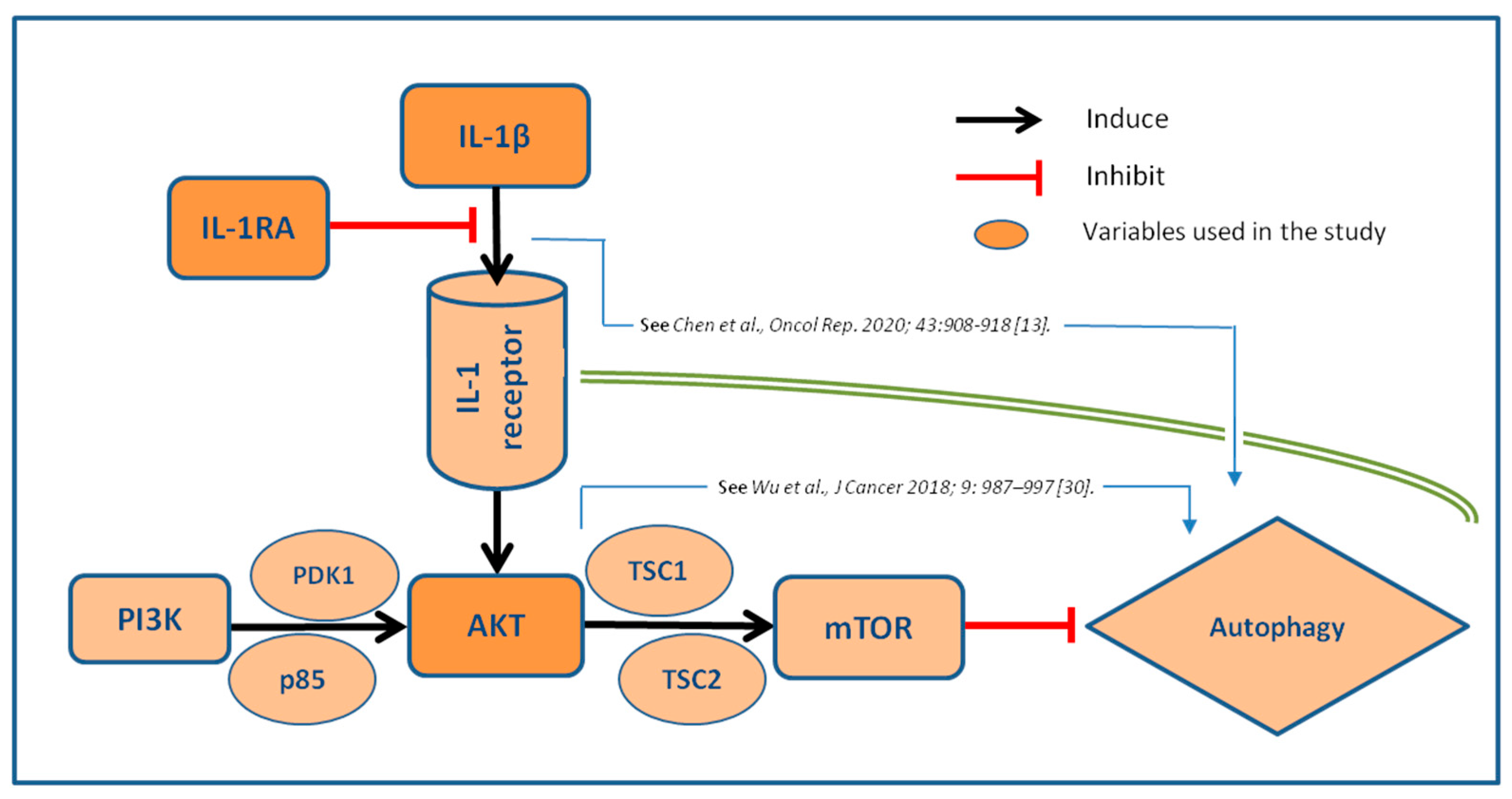
3. Discussion
4. Materials and Methods
4.1. Patient Cohorts
4.2. Tissue Microarray (TMA) and Immunohistochemical (IHC) Staining
4.3. Immunohistochemical Protocol
4.4. Patient Follow-Up
4.5. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Wang JZ ZW, Han J, Yang X, Zhou R, Lu HC, Yu H, Yuan WB, Li PC, Tao J, Lu, Q WJ, Yang H. The role of the HIF-1α/ALYREF/PKM2 axis in glycolysis and tumorigenesis of bladder cancer. Cancer Commun (Lond). 2021;41(7):560-75. [CrossRef]
- Huang W LY, Zhang C, Zha H, Zhou X, Fu B, Guo J, Wang G. IGF2BP3 facilitates cell proliferation and tumorigenesis via modulation of JAK/STAT signalling pathway in human bladder cancer. J Cell Mol Med. 2020;24(23):13949-60. [CrossRef]
- Rausch S HJ, Teepe K, Kuehs U, Aufderklamm S, Bier S, Mischinger J, Gakis G, Stenzl A, Schwentner C, Todenhöfer T. Muscle-invasive bladder cancer is characterized by overexpression of thymidine kinase 1. Urol Oncol. 2015;33(10):426.e21-.e29. [CrossRef]
- Xia H GD, Zou W. Autophagy in tumour immunity and therapy. Nat Rev Cancer. 2021;21(5):281-97. [CrossRef]
- Vetterlein MW RJ, Gild P, Marks P, Soave A, Doh O, Isbarn H,, Höppner W WW, Shariat SF, Brausi M, Büscheck F, Sauter G, Fisch M, Rink, M. Impact of the Ki-67 labeling index and p53 expression status on disease-free survival in pT1 urothelial carcinoma of the bladder. Transl Androl Urol. 2017;6(6):1018-26. [CrossRef]
- Liu GH ZQ, Ye YL, Wang HB, Hu LJ, Qin ZK, Zeng MS, Zeng BH. Expression of beclin 1 in bladder cancer and its clinical significance. Int J Biol Markers. 2013;28(1):56-62. [CrossRef]
- Kim H LS, Lee IK, Min SC, Sung HH, Jeong BC, Lee J, Park SH. Synergistic Effects of Combination Therapy with AKT and mTOR Inhibitors on Bladder Cancer Cells. Int J Mol Sci. 2020;21(8):2825. [CrossRef]
- Matsumoto H MM, Shimizu K, Fujii N, Kobayashi K, Inoue R, Yamamoto, Y NK, Matsuyama H. Risk stratification using Bmi-1 and Snail expression is a useful prognostic tool for patients with upper tract urothelial carcinoma. Int J Urol. 2016;23(12):1030-7. [CrossRef]
- Wang F LJ, Chen X, Zheng X, Qu N, Zhang B, Xia C. IL-1β receptor antagonist (IL-1Ra) combined with autophagy inducer (TAT-Beclin1) is an effective alternative for attenuating extracellular matrix degradation in rat and human osteoarthritis chondrocytes. Arthritis Res Ther. 2019;21(1):171. [CrossRef]
- de Luca A SS, Casagrande A, Iannitti R, Conway KL, Gresnigt MS,, Begun J PT, Joosten LA, van der Meer JW, Chamilos G, Netea MG, Xavier, RJ DC, Romani L, van de Veerdonk FL. IL-1 receptor blockade restores autophagy and reduces inflammation in chronic granulomatous disease in mice and in humans. Proc Natl Acad Sci U S A. 2014;111(9):3526-31. [CrossRef]
- Crişan TO PT, van de Veerdonk FL, Farcaş MF, Stoffels M, Kullberg, BJ vdMJ, Joosten LA, Netea MG. Inflammasome-independent modulation of cytokine response by autophagy in human cells. PLoS One. 2011;6(4):e18666. [CrossRef]
- Humphrey PA MH, Cubilla AL, Ulbright TM, Reuter VE. The 2016 WHO Classification of Tumours of the Urinary System and Male Genital Organs-Part B: Prostate and Bladder Tumours. Eur Urol. 2016;70(1):106-19. [CrossRef]
- Chen Y YZ, Deng B, Wu D, Quan Y, Min Z. nterleukin 1β/1RA axis in colorectal cancer regulates tumor invasion, proliferation and apoptosis via autophagy. Oncol Rep. 2020;43(3):908-18. [CrossRef]
- John A GC, Bolenz C, Vidal-Y-Sy S, Bauer AT, Schneider SW, Gorzelanny, Chen Y YZ, Deng B, Wu D, Quan Y, Min Z. Bladder cancer-derived interleukin-1 converts the vascular endothelium into a pro-inflammatory and pro-coagulatory surface. BMC Cancer. 2020;2(20):1178. [CrossRef]
- Yao SJ MH, Liu GM, Gao Y, Wang W. Increased IL-1α expression is correlated with bladder cancer malignant progression. Arch Med Sci. 2020;19(1):160-70. [CrossRef]
- Lykkegaard Andersen N BA, Lelkaitis G, Nielsen S, Friis Lippert M,, M V. Virtual Double Staining: A Digital Approach to Immunohistochemical Quantification of Estrogen Receptor Protein in Breast Carcinoma Specimens. Appl Immunohistochem Mol Morphol. 2018;26(9):620-6. [CrossRef]
- Le Page C KI, Alam-Fahmy M, Mes-Masson AM, Saad F. Expression and localisation of Akt-1, Akt-2 and Akt-3 correlate with clinical outcome of prostate cancer patients. Br J Cancer. 2006;94(12):1906-12. [CrossRef]
- Aman NA DB, Koffi KD, Koui BS, Traore ZC, Kouyate M, Toure I, Effi, AB. Immunohistochemical Evaluation of Ki-67 and Comparison with Clinicopathologic Factors in Breast Carcinomas. Asian Pac J Cancer Prev. 2019;20(1):73-9. [CrossRef]
- Bertz S OW, Denzinger S, Wieland WF, Burger M, Stöhr R, Link S,, Hofstädter F HA. Combination of CK20 and Ki-67 immunostaining analysis predicts recurrence, progression, and cancer-specific survival in pT1 urothelial bladder cancer. Eur Urol. 2014;65(1):218-26. [CrossRef]
- Liu Y YT, Wei YW. What is the difference between overall survival, recurrence-free survival and time-to-recurrence? Br J Surg. 2020;107(12):e634.
- Semilia M HJ, Pavone C, Bischoff T, Kühs U, Gakis G, Bedke J,, Stenzl A SC, Todenhöfer T. Expression patterns and prognostic role of transketolase-like 1 in muscle-invasive bladder cancer. World J Urol. 2015;33(10):1403-9. [CrossRef]
- Wilson F JN, Choudhury A. Biomarkers in muscle invasive bladder cancer. Adv Clin Chem. 2022;107:265-97.
- Audenet F AK, Sfakianos JP. The evolution of bladder cancer genomics: What have we learned and how can we use it? Urol Oncol. 2018;36(7):313-20. [CrossRef]
- Rébé C GF. Interleukin-1β and Cancer. Cancers (Basel). 2020;12(7):1791. [CrossRef]
- Schneider L LJ, Zhang C, Azoitei A, Meessen S, Zheng X, Cremer C,, Gorzelanny C K-GS, Brunner C, Wezel F, Bolenz C, Gunes C, John A. The Role of Interleukin-1-Receptor-Antagonist in Bladder Cancer Cell Migration and Invasion. Int J Mol Sci. 2021;22(11):5875. [CrossRef]
- Waugh J PC. Anakinra: a review of its use in the management of rheumatoid arthritis. BioDrugs. 2005;19(3):189-202. [CrossRef]
- Lust JA LM, Zeldenrust SR, Dispenzieri A, Gertz MA, Witzig TE, Kumar S,, Hayman SR RS, Buadi FK, Geyer SM, Campbell ME, Kyle RA, Rajkumar SV,, Greipp PR KM, Xiong Y, Moon-Tasson LL, Donovan KA. Induction of a chronic disease state in patients with smoldering or indolent multiple myeloma by targeting interleukin 1{beta}-induced interleukin 6 production and the myeloma proliferative component. Mayo Clin Proc. 2009;84(2):114-22. [CrossRef]
- Minoli M KM, Thalmann GN, Kruithof-de Julio M, Seiler R. Evolution of Urothelial Bladder Cancer in the Context of Molecular Classifications. Int J Mol Sci. 2020;21(16):5670. [CrossRef]
- Iula L KI, Sabbione F, Fuentes F, Guzman M, Galletti JG, Gerber, PP OM, Geffner JR, Jancic CC, Trevani AS. Autophagy Mediates Interleukin-1β Secretion in Human Neutrophils. Front Immunol. 2018;9:269. [CrossRef]
- Wu N ZY, Xu X, Zhu Y, Song Y, Pang L, Chen Z. The anti-tumor effects of dual PI3K/mTOR inhibitor BEZ235 and histone deacetylase inhibitor Trichostatin A on inducing autophagy in esophageal squamous cell carcinoma. J Cancer. 2018;9(6):987-97. [CrossRef]
| Patients characteristics | Group I (n=102) | Group II (n=92) | P | ||
| N | % | n | % | ||
| Gender | 0.56 | ||||
| - female | 24 | 23.53 | 25 | 27.18 | |
| - male | 78 | 76.47 | 67 | 72.82 | |
| Age | Median 69 | Range (59-74) | Median 65 | Range (55-72) | 0.19 |
| Pathological stage | 0.55 | ||||
| -T1 | 0 | 0.00 | 9 | 9.78 | |
| -T2 | 31 | 30.39 | 21 | 22.82 | |
| -T3 | 47 | 46.08 | 42 | 45.66 | |
| -T4 | 24 | 23.53 | 18 | 19.57 | |
| -only CIS | 0 | 0.00 | 2 | 2.17 | |
| Lymph Node status | 0.50 | ||||
| -N0 | 58 | 56.86 | 57 | 61.95 | |
| -N+ | 44 | 43.14 | 35 | 38.05 | |
| Distant metastasis | 0.59 | ||||
| -M0 | 89 | 87.23 | 82 | 89.13 | |
| -M1 | 9 | 8.83 | 10 | 10.87 | |
| -Mx | 4 | 3.93 | 0 | 0.00 | |
| Pathological grade | 0.94 | ||||
| -G1 | 0 | 0.00 | 1 | 1.08 | |
| -G2 | 24 | 23.53 | 20 | 21.74 | |
| -G3 | 78 | 76.47 | 71 | 77.18 | |
| Resection margin status | 0.74 | ||||
| -R0 | 84 | 82.35 | 74 | 80.43 | |
| -R1 | 15 | 14.70 | 14 | 15.22 | |
| -R2 | 3 | 2.95 | 2 | 2.18 | |
| -Rx | 0 | 0.00 | 2 | 2.17 | |
| Carcinoma in situ (CIS) | 0.57 | ||||
| - No | 68 | 66.66 | 61 | 66.31 | |
| - Yes | 29 | 28.44 | 31 | 33.69 | |
| -CISx | 5 | 4.90 | 0 | 0.00 | |
| Histopathology variables | IL-1β expression (H-score) |
IL-1RA expression (H-score) |
IL1-β/IL-1RA ratio (cut-off 0.7) |
| Tumor grade | Medians | ||
| G2 | 188 | 250*** | 0.7 |
| G3 | 193* | 185 | 1.1*** |
| Tumor stadium pT |
|||
| T2 | 205 | 178 | 1.0 |
| T3 | 190 | 200 | 1.0 |
| T4 | 190 | 200 | 0.9 |
| Lymphatic invasion | |||
| No | 180 | 194 | 1.0 |
| yes | 210* | 200 | 1.0 |
| Vascular invasion | |||
| no | 183 | 190 | 1.0 |
| yes | 210* | 203 | 1.0 |
| Nodal invasion | |||
| N0 | 185 | 194 | 1.0 |
| N1 | 185 | 200 | 0.9 |
| N2 | 210 | 205 | 1.1 |
| N3 | 198 | 175 | 1.0 |
| Metastatic disease | |||
| M0 | 185 | 194 | 1.0 |
| M1 | 210 | 200 | 1.0 |
| Asterisk depicts statistically significant correlation between expression pattern of IL-1 axis and corresponding histological variable (* = <0.05; *** = < 0.005) | |||
| Variable | Ki-67+ and IL-1β+ | Ki-67+ (15%) or IL-1β+ | Ki-67+ and IL1RA+ | Ki-67+ (15%) or IL-1RA+ |
| Pathological stage >T2 vs. T2 |
0.61 | 0.67 | 0.81 | 0.06 |
| Nodal status N+ vs. N0 |
0.97 | 0.25 | 0.39 | 0.54 |
| Metastasis status M+ vs. M0 |
0.28 | 0.40 | 0.46 | 0.56 |
| Grade ≥G3 vs. < G3 |
1.00 | 0.02 | 0.52 | 0.57 |
| Lymphatic invasion L+ vs. L0 |
0.93 | 0.51 | 0.30 | 0.70 |
| Vascular invasion V+ vs. V0 |
0.02 | 0.92 | 0.33 | 0.90 |
| Variable | Univariate analysis | Multivariate analysis |
| Recurrence (RFS) | P HR 95% CI | P HR 95% CI |
| Pathological stage >T2 vs. T2 |
0.0099 3.25 1.33-7.95 | 0.1339 2.14 0.79-5.80 |
| Nodal status N+ vs. N0 |
0.0879 1.88 0.91-3.88 | - |
| Metastasis status M+ vs. M0 |
- | - |
| Grade ≥G3 vs. < G3 |
0.0531 1.30 0.60-2.83 | - |
| IL-1β Low vs.high tumor expression (cut-off 160) |
0.0172 3.16 1.23-8.16 | 0.0265 2.97 1.14-7.77 |
| Age, year | 0.2397 1.02 0.99-1.06 | - |
| Variable | Univariate analysis | Multivariate analysis |
| CSS | P HR 95% CI | P HR 95% CI |
| Pathological stage >T2 vs. T2 |
0.0107 2.53 1.24-5.17 | 0.3407 1.51 0.65-3.49 |
| Nodal status N+ vs. N0 |
0.0309 1.96 1.06-3.62 | 0.3170 1.48 0.69-3.17 |
| Metastasis status M+ vs. M0 |
0.0015 3.56 1.63-7.79 | 0.0504 2.58 1.00-6.65 |
| Grade ≥G3 vs. < G3 |
0.0069 3.62 1.42-9.22 | 0.1247 2.38 0.79-7.18 |
| IL-1β Low vs.high tumor expression (cut-off 160) |
0.0343 2.27 1.06-4.84 | 0.0460 2.35 1.02-5.42 |
| Age, year | 0.0047 1.05 1.01-1.08 | 0.3421 1.02 0.98-1.06 |
| Variable | Univariate analysis | Multivariate analysis |
| OS | P HR 95% CI | P HR 95% CI |
| Pathological stage >T2 vs. T2 |
0.0020 2.79 1.45-5.33 | 0.1240 1.85 0.85-4.03 |
| Nodal status N+ vs. N0 |
0.0258 1.85 1.08-3.17 | 0.1909 1.60 0.79-3.24 |
| Metastasis status M+ vs. M0 |
0.0036 3.13 1.45-6.73 | 0.1148 2.12 0.83-5.37 |
| Grade ≥G3 vs. < G3 |
0.0173 2.31 1.16-4.60 | 0.1611 1.94 0.77-4.88 |
| IL-1β Low vs.high tumor expression (cut-off 160) |
0.0082 2.54 1.27-5.06 | 0.0249 2.40 1.12-5.18 |
| Age, year | 0.0031 1.05 1.02-1.08 | 0.1274 1.03 0.99-1.07 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




