Submitted:
08 January 2024
Posted:
09 January 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Process Parameter Development
2.2. Microstructural Characterization, Corrosion and Mechanical Tests
3. Results
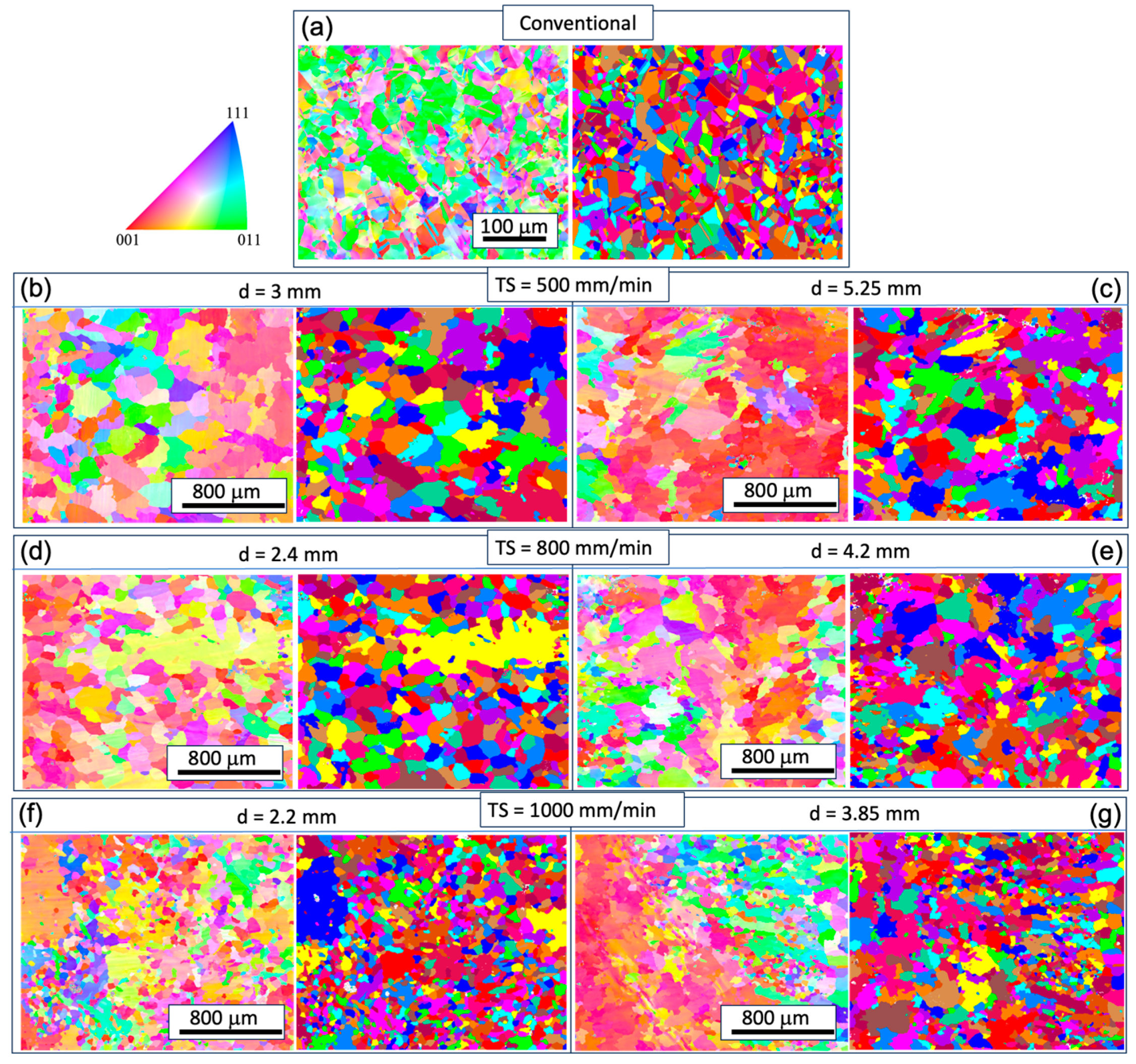
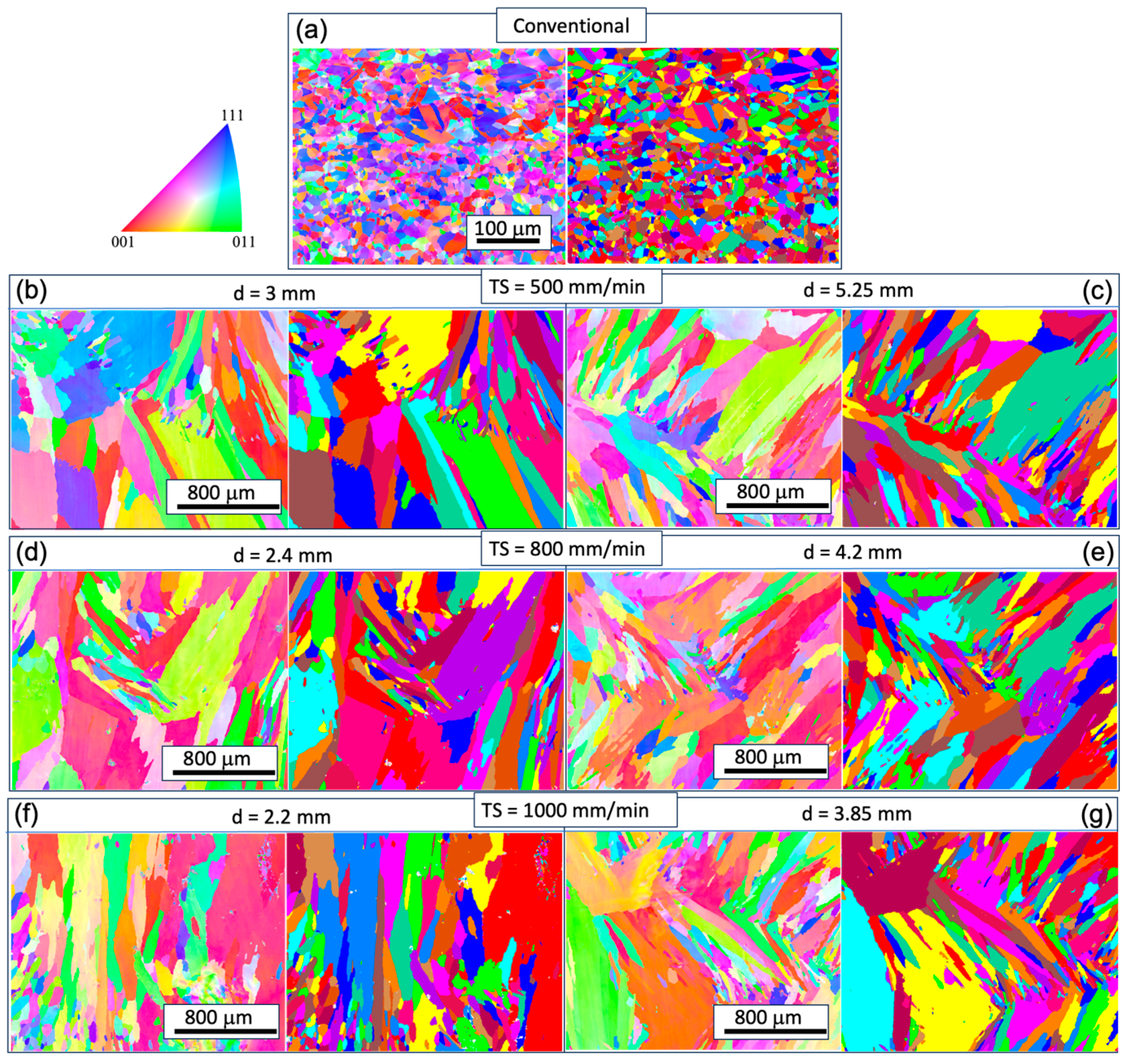
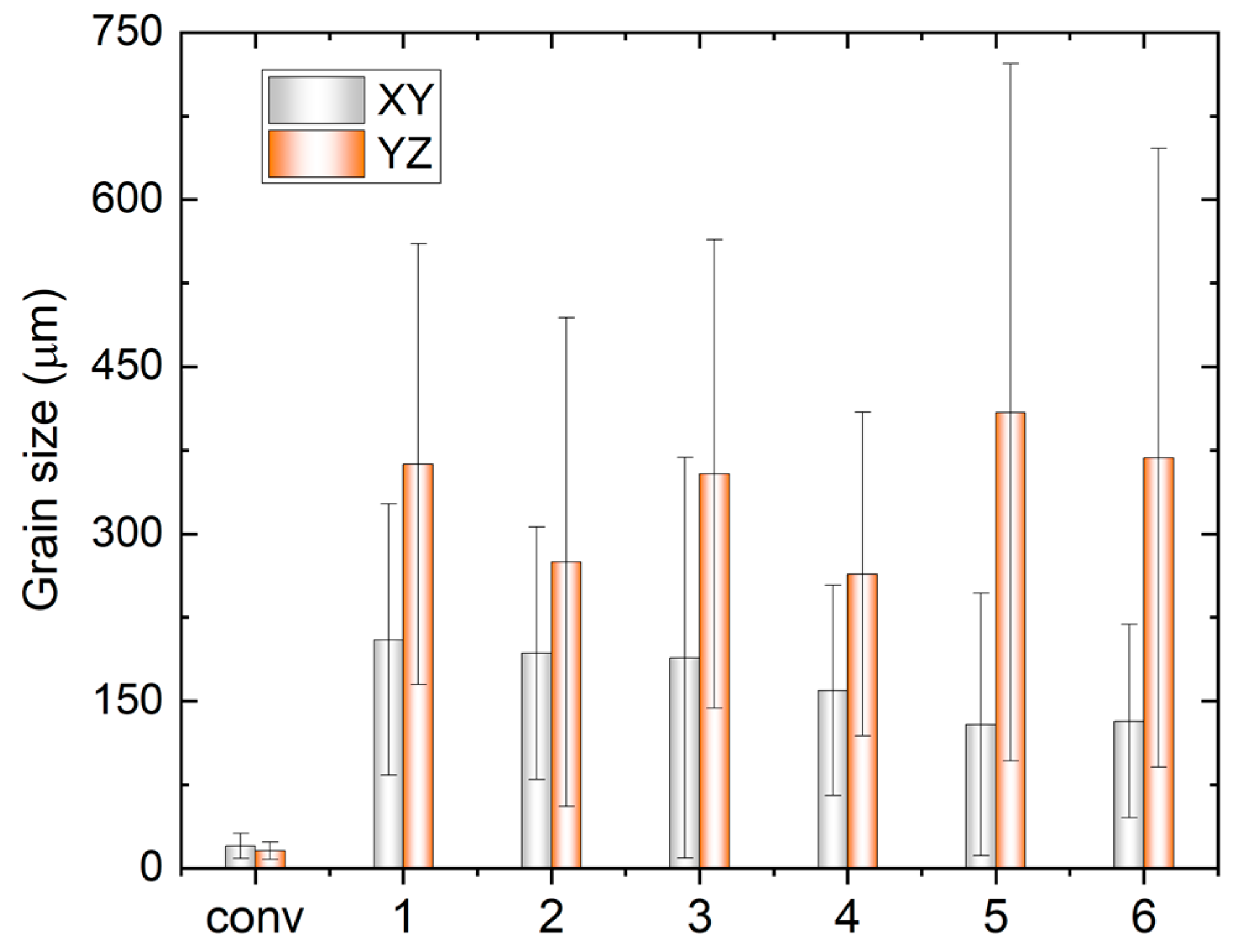
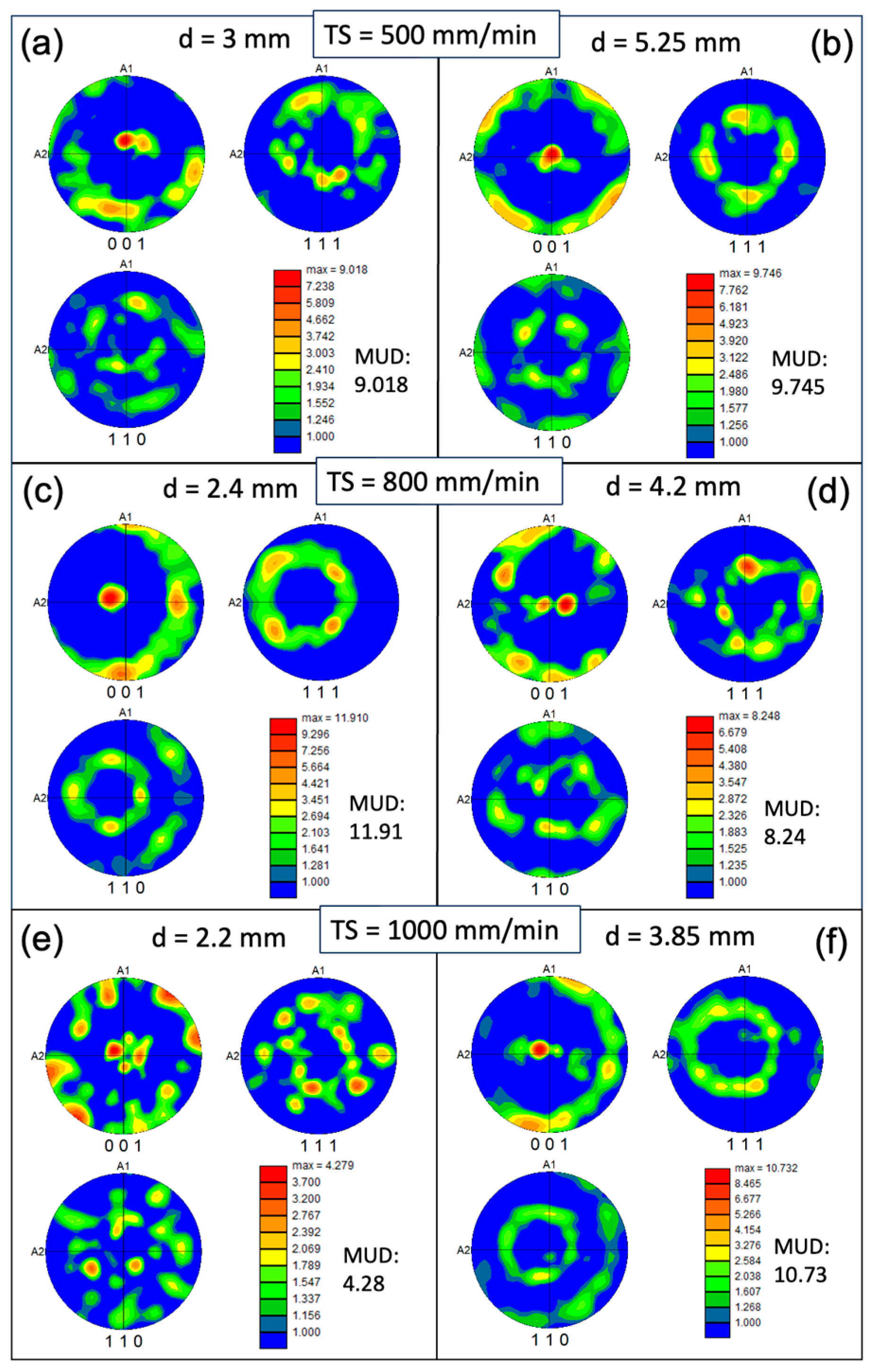
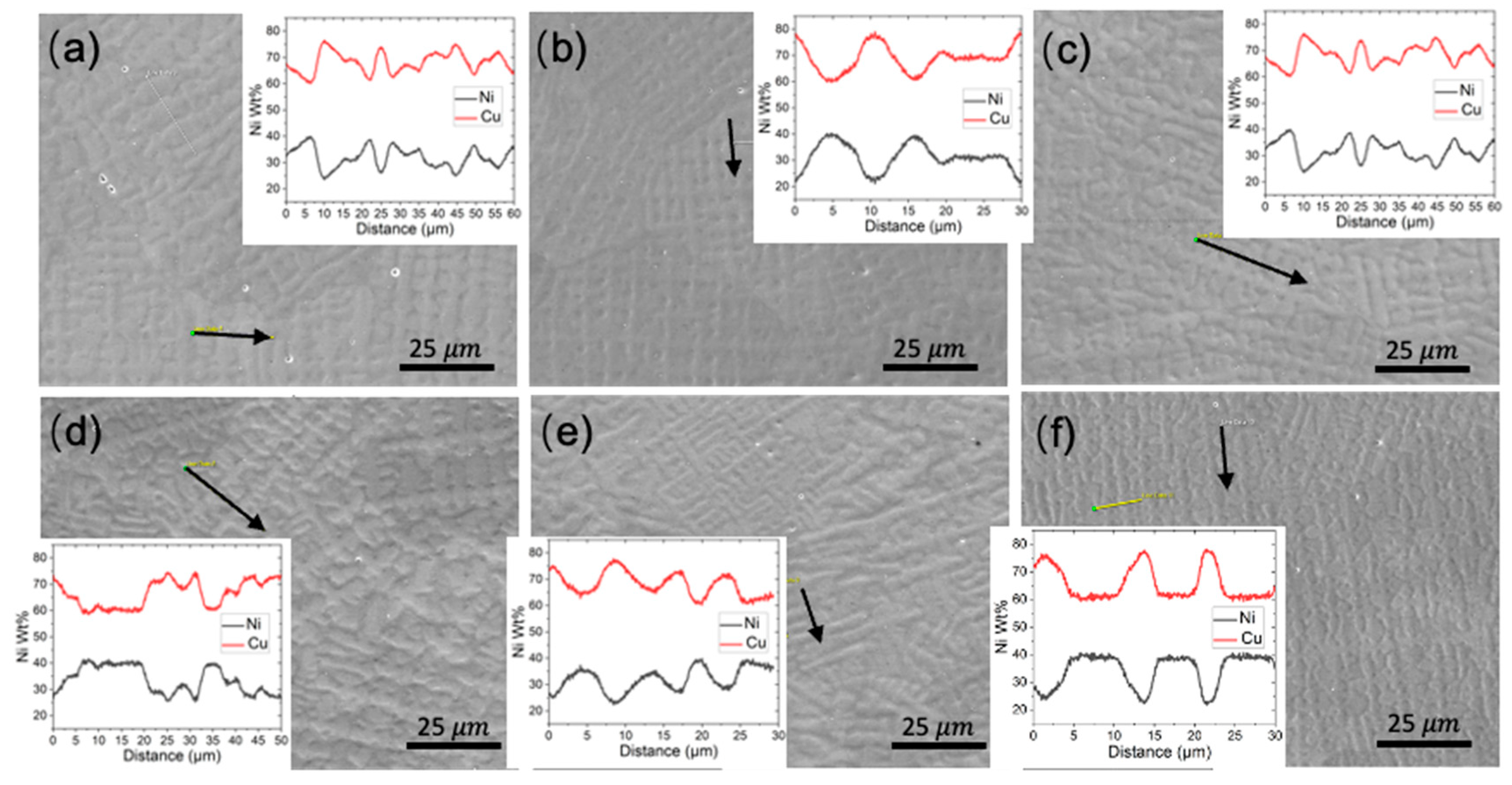
3.1. Microstructural Analysis
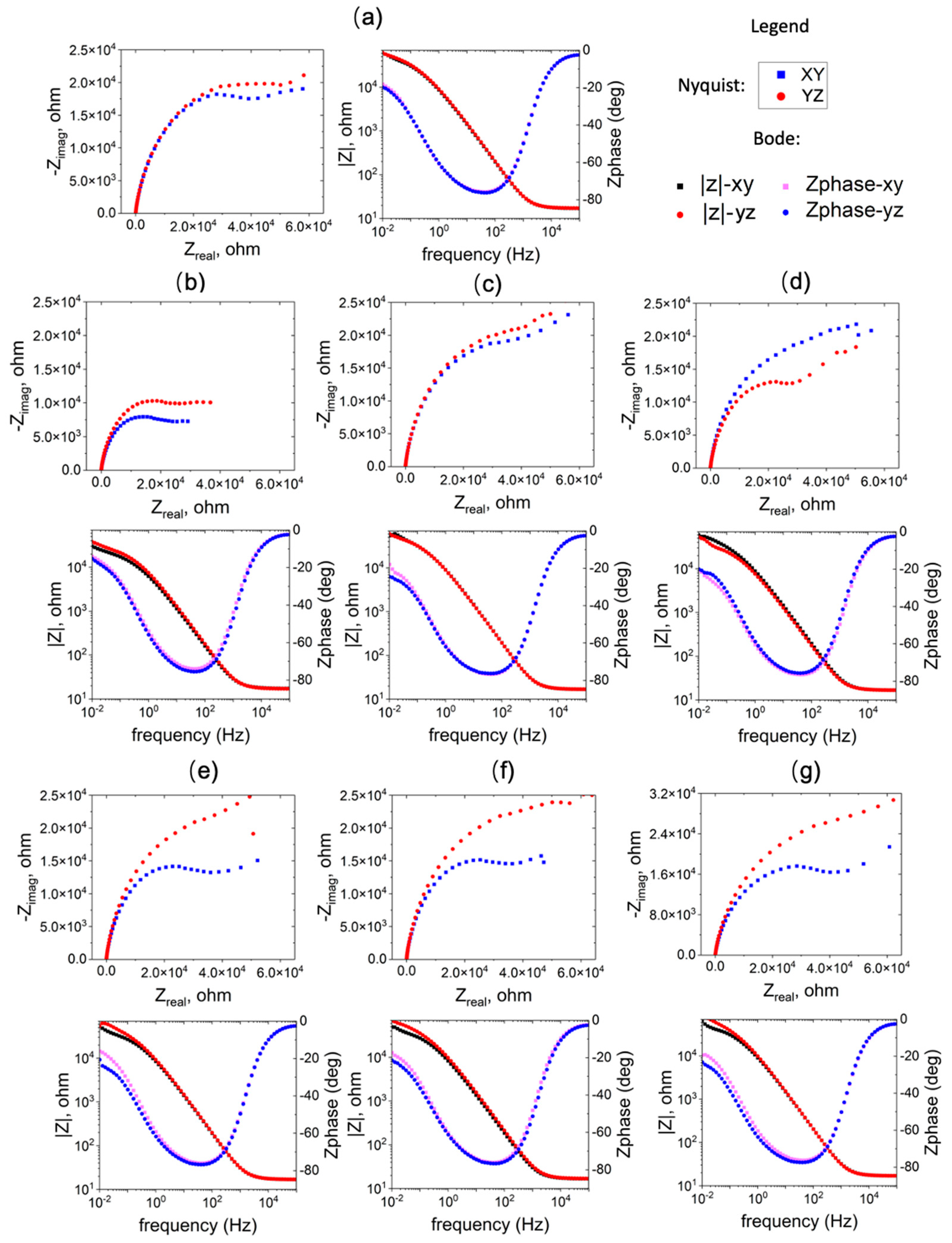
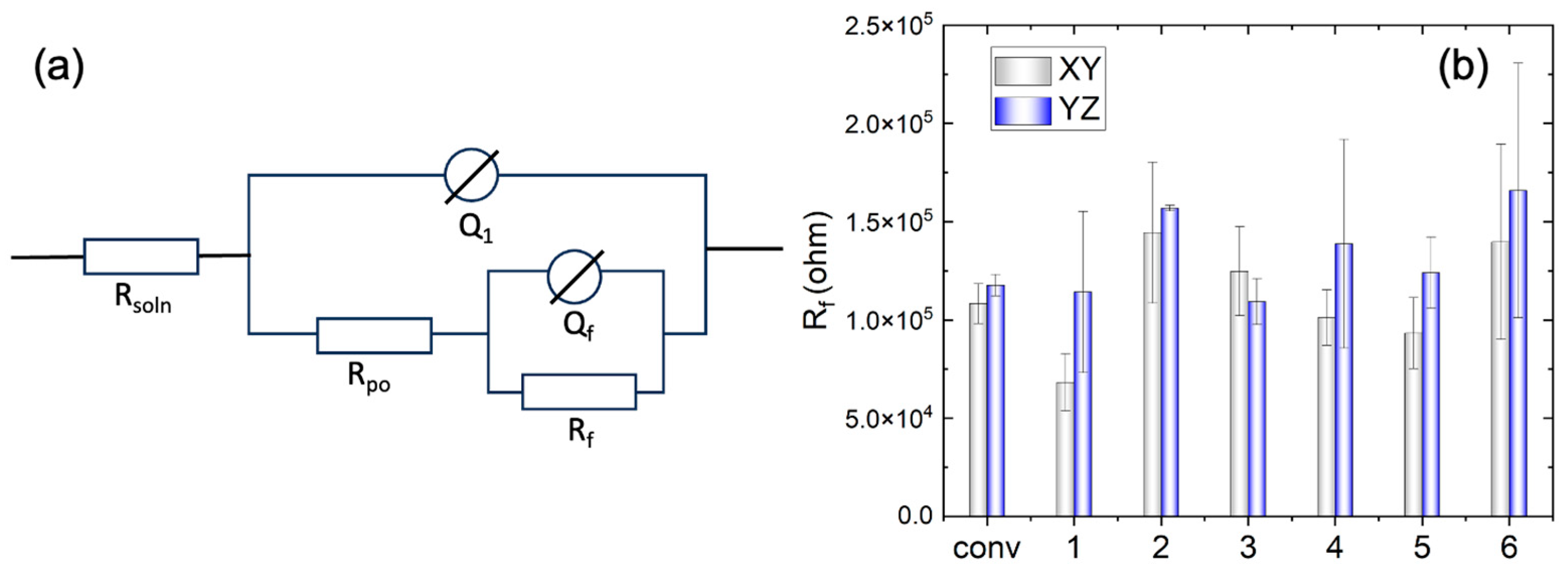
3.2. Electrochemical Impedance Spectroscopy
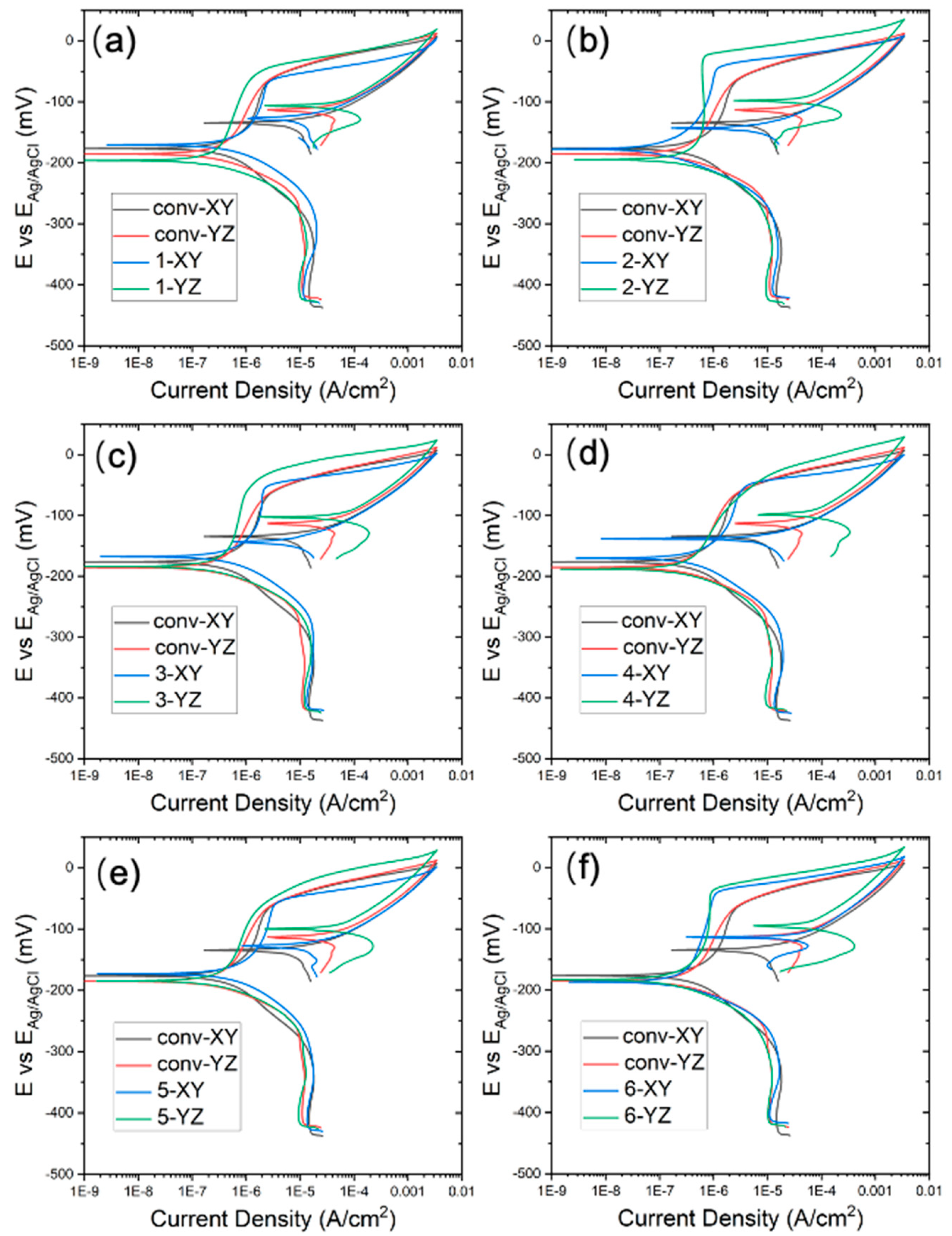
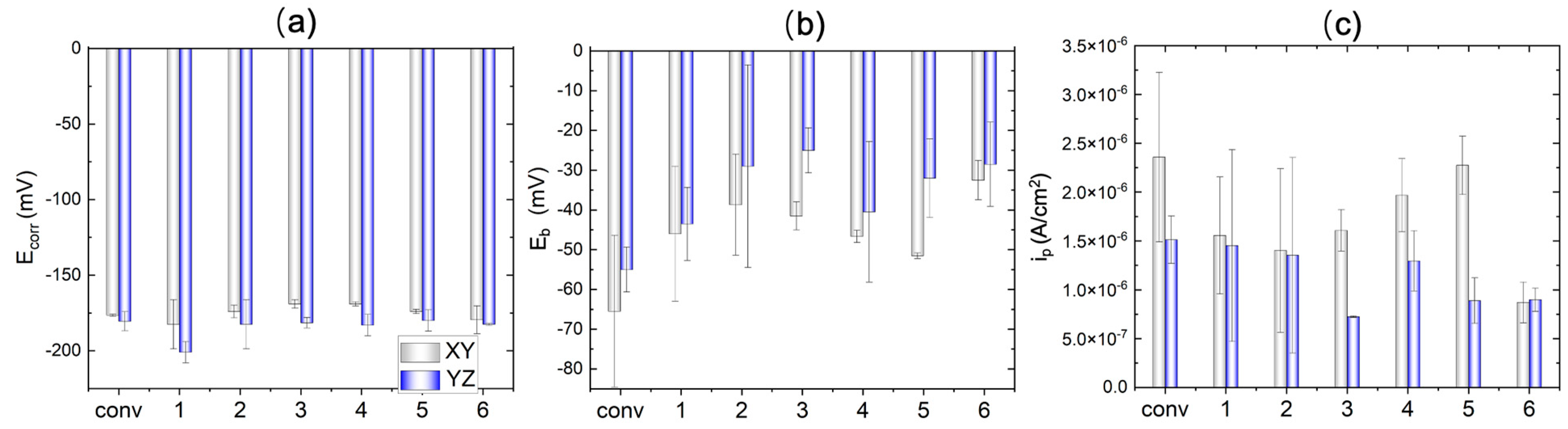
3.3. ElectroPolarization Test
4. Discussion
5. Conclusions
- The as-fabricated microstructures are characterized by columnar grains with prominent dendrites. The longitudinal direction of the columnar grain deviates from the build direction. Strong chemical segregation has been observed in the form of Ni element depletion in the inter-dendritic area.
- With increasing TS, the aspect ratio of columnar grains increases, resulting in a reduced size of the XY cross-section and an increased length of the YZ cross-section. The grain size also decreases with increasing track distance. A close-to-{001} texture has developed on 30% to 40% of the area on the XY plane. The grain size of the conventional sample is one order of magnitude smaller compared with that of the WAAM ones, and exhibits random orientation.
- The corrosion performance is evaluated in terms of film resistance at OCP, the passive current density and breakdown potential during polarization. Two WAAM samples, produced under a TS of 800 mm/min and track distance of 5.25 mm as well as a TS of 1000 mm/min and track distance of 3.85 mm, have the best corrosion performance in the 3.5% NaCl solution.
- Anisotropy in the corrosion performance has also been observed. The YZ plane exhibits higher film resistance at OCP, a higher breakdown potential, and lower passive current density, compared to the XY plane, indicating better corrosion resistance.
- At the grain/subgrain level, the conventional Cu-Ni demonstrates crystallography-dependent corrosion resistance, whereas the WAAM Cu-Ni exhibits preferential corrosion attack at the inter-dendritic area due to the uneven distribution of Ni element. The reason behind the apparent corrosion enhancement in the aforementioned WAAM Cu-Ni samples compared to its conventional counterpart need further analysis, which will be carried out in our study in the near future.
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Zhu, X.; Lei, T. Characteristics and formation of corrosion product films of 70Cu–30Ni alloy in seawater. Corrosion Science 2002, 44, 67–79. [Google Scholar] [CrossRef]
- Wang, Y.Z.; Beccaria, A.M.; Poggi, G. The effect of temperature on the corrosion behaviour of a 70/30 Cu-Ni commercial alloy in seawater. Corrosion Science 1994, 36, 1277–1288. [Google Scholar] [CrossRef]
- Chauhan, P.K.; Gadiyar, H.S. An XPS study of the corrosion of Cu-10 Ni alloy in unpolluted and polluted sea-water; the effect of FeSO4 addition. Corrosion Science 1985, 25, 55–68. [Google Scholar] [CrossRef]
- Mansfeld, F.; Little, B. Microbiologically influenced corrosion of copper-based materials exposed to natural seawater. Electrochimica Acta 1992, 37, 2291–2297. [Google Scholar] [CrossRef]
- Mao, X.; Fang, F.; Jiang, J.; Tan, R. Effect of rare earths on corrosion resistance of Cu-30Ni alloys in simulated seawater. Journal of Rare Earths 2009, 27, 1037–1041. [Google Scholar] [CrossRef]
- Martin, J.R.; Heidersbach, R.H.; Lenard, D.R. Dealloying of cupronickels in stagnant seawater in Proceedings of NACE International, Houston, TX, USA, 1999.
- Lee, W.-H.; Chung, K.C. Investigation of a copper–nickel alloy resistor using co-electrodeposition. Journal of Applied Electrochemistry 2020, 50, 535–547. [Google Scholar] [CrossRef]
- Armstrong, M.; Mehrabi, H.; Naveed, N. An overview of modern metal additive manufacturing technology. Journal of Manufacturing Processes 2022, 84, 1001–1029. [Google Scholar] [CrossRef]
- Durai Murugan, P.; Vijayananth, S.; Natarajan, M.P.; Jayabalakrishnan, D.; Arul, K.; Jayaseelan, V.; Elanchezhian, J. A current state of metal additive manufacturing methods: A review. Materials Today: Proceedings 2022, 59, 1277–1283. [Google Scholar] [CrossRef]
- Shah, A.; Aliyev, R.; Zeidler, H.; Krinke, S. A Review of the Recent Developments and Challenges in Wire Arc Additive Manufacturing (WAAM) Process. Journal of Manufacturing and Materials Processing 2023, 7. [Google Scholar] [CrossRef]
- Kumar, N.; Bhavsar, H.; Mahesh, P.V.S.; Srivastava, A.K.; Bora, B.J.; Saxena, A.; Dixit, A.R. Wire Arc Additive Manufacturing – A revolutionary method in additive manufacturing. Materials Chemistry and Physics 2022, 285, 126144. [Google Scholar] [CrossRef]
- Rodrigues, T.A.; Duarte, V.; Miranda, R.M.; Santos, T.G.; Oliveira, J.P. Current Status and Perspectives on Wire and Arc Additive Manufacturing (WAAM). Materials 2019, 12. [Google Scholar] [CrossRef]
- Guo, C.; Kang, T.; Wu, S.; Ying, M.; Liu, W.M.; Chen, F. Microstructure, mechanical, and corrosion resistance of copper nickel alloy fabricated by wire-arc additive manufacturing. MRS Communications 2021, 11, 910–916. [Google Scholar] [CrossRef]
- Miao, Y.; Li, C.; Zhao, Y.; Wu, Y.; Liu, J.; Wang, Z.; Zhang, B. Material Properties of Functionally Gradient Copper-Nickel Alloy Fabricated by Wire Arc Additive Manufacturing Based on Bypass-Current Paw. 2023. [Google Scholar] [CrossRef]
- Hunt, J.D.; Lu, S.Z. Numerical modeling of cellular/dendritic array growth: spacing and structure predictions. Metallurgical and Materials Transactions A 1996, 27, 611–623. [Google Scholar] [CrossRef]
- Song, J.; Jimenez, X.A.; Russell, C.; To, A.C.; Fu, Y. Unusually high room and elevated-temperature tensile properties observed in direct aged wire-arc directed energy deposited Inconel 718. Scientific Reports 2023, 13, 19235. [Google Scholar] [CrossRef] [PubMed]
- Hsu, C.H.; Mansfeld, F. Technical Note: Concerning the Conversion of the Constant Phase Element Parameter Y0 into a Capacitance. Corrosion 2001, 57, 747–748. [Google Scholar] [CrossRef]
- Mansfeld, F. Electrochemical impedance spectroscopy (EIS) as a new tool for investigating methods of corrosion protection. Electrochimica Acta 1990, 35, 1533–1544. [Google Scholar] [CrossRef]
- Geng, R.; Cheng, Y.; Chao, L.; Wei, Z.; Ma, N. Microstructure and Solute Concentration Analysis of Epitaxial Growth during Wire and Arc Additive Manufacturing of Aluminum Alloy. Crystals 2023, 13. [Google Scholar] [CrossRef]
- EFIRD, K.D. Potential-pH Diagrams for 90-10 and 70-30 Cu-Ni in Sea Water. Corrosion 2013, 31, 77–83. [Google Scholar] [CrossRef]
- Ma, A.L.; Jiang, S.L.; Zheng, Y.G.; Ke, W. Corrosion product film formed on the 90/10 copper–nickel tube in natural seawater: Composition/structure and formation mechanism. Corrosion Science 2015, 91, 245–261. [Google Scholar] [CrossRef]
- Wu, L.; Ma, A.; Zhang, L.; Li, G.; Hu, L.; Wang, Z.; Zheng, Y. Erosion–Corrosion Behavior of 90/10 and 70/30 Copper–Nickel Tubes in 1 wt% NaCl Solution. Metals 2023, 13. [Google Scholar] [CrossRef]
- Tuthill, A.H. Guidelines for the Use of Copper Alloys in Seawater. NiDI Publication 1988, 12003. [Google Scholar]
- Schleich, W. Application of Copper-Nickel Alloy UNS C70600 for Seawater Service in Proceedings of CORROSION 2005, Houston, Texas, 2005.




| Specimen | WFS | TS | Track Distance | Interpass Temp (°C) |
|---|---|---|---|---|
| 1 | 8 m/min | 500 mm/min | 3 mm | 150~200 |
| 2 | 5.25 mm | |||
| 3 | 800 mm/min | 2.4 mm | ||
| 4 | 4.2 mm | |||
| 5 | 1000 mm/min | 2.2 mm | ||
| 6 | 3.85 mm |
| Specimen | Rf × 105 (Ω) | Qf × 10-5 (F) | nf | df (μm) | ||||
| XY | YZ | XY | YZ | XY | YZ | XY | YZ | |
| Wrought | 1.08 | 1.18 | 2.44 | 2.37 | 0.34 | 0.32 | 0.13 | 0.095 |
| 1 | 0.68 | 1.14 | 2.84 | 2.83 | 0.340 | 0.29 | 0.20 | 0.041 |
| 2 | 1.44 | 1.57 | 2.13 | 2.29 | 0.29 | 0.31 | 0.063 | 0.051 |
| 3 | 1.25 | 1.09 | 2.54 | 2.98 | 0.29 | 0.31 | 0.046 | 0.049 |
| 4 | 1.01 | 1.39 | 2.36 | 2.34 | 0.31 | 0.30 | 0.12 | 0.058 |
| 5 | 0.93 | 1.24 | 2.43 | 2.04 | 0.32 | 0.33 | 0.14 | 0.14 |
| 6 | 1.40 | 1.66 | 2.68 | 2.47 | 0.27 | 0.28 | 0.021 | 0.023 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




