Submitted:
05 January 2024
Posted:
08 January 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Study design
2.2. ECL immunoassay for anti-HCV
2.3. CLIA immunoassay for anti-HCV
2.4. Recombinant immunoblot assay for anti-HCV
2.5. Statistical analisys
3. Results
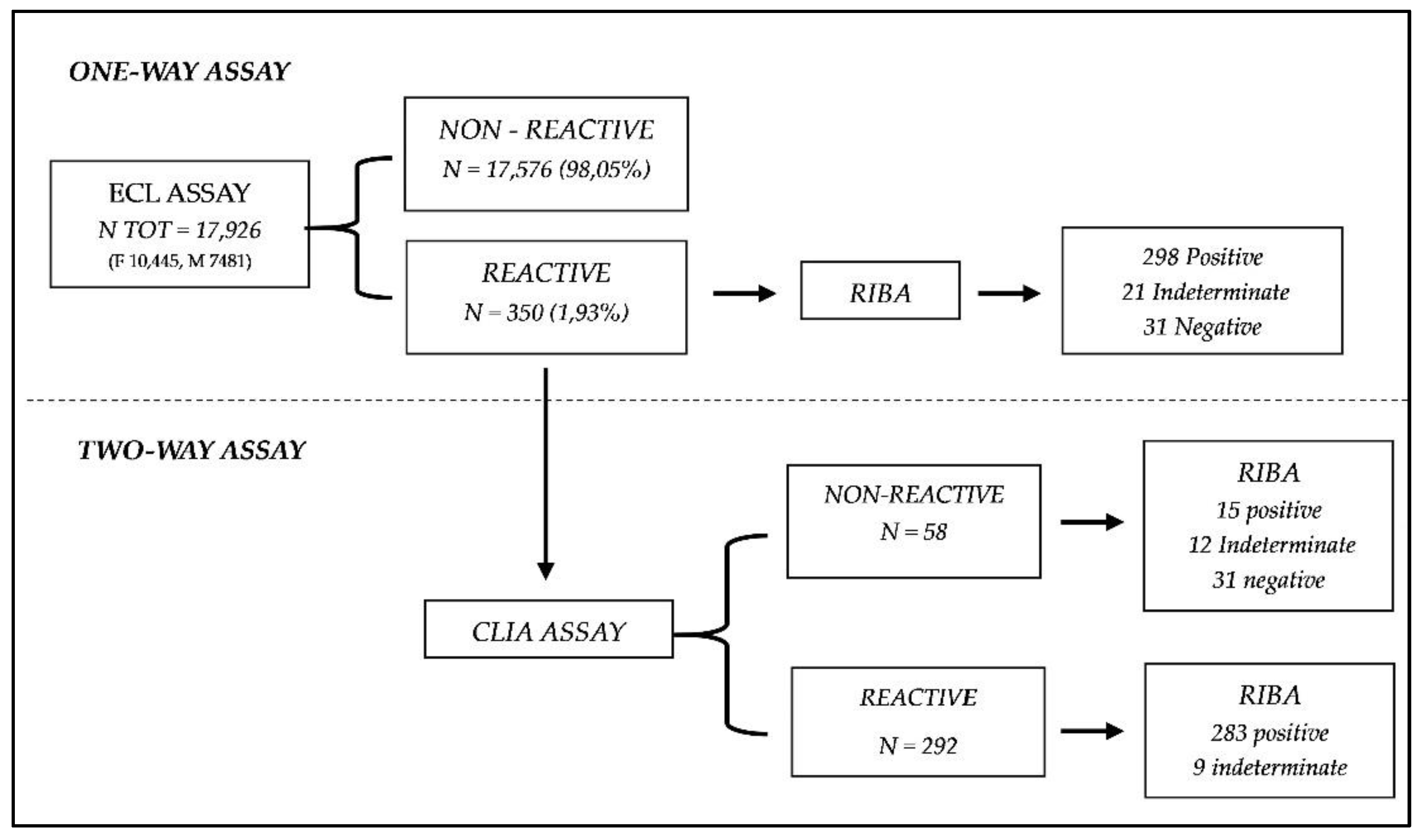
3.1. One-assay serological testing strategy using ECL screening
3.2. Two-assay serological testing strategy using CLIA testing, after ECL screening
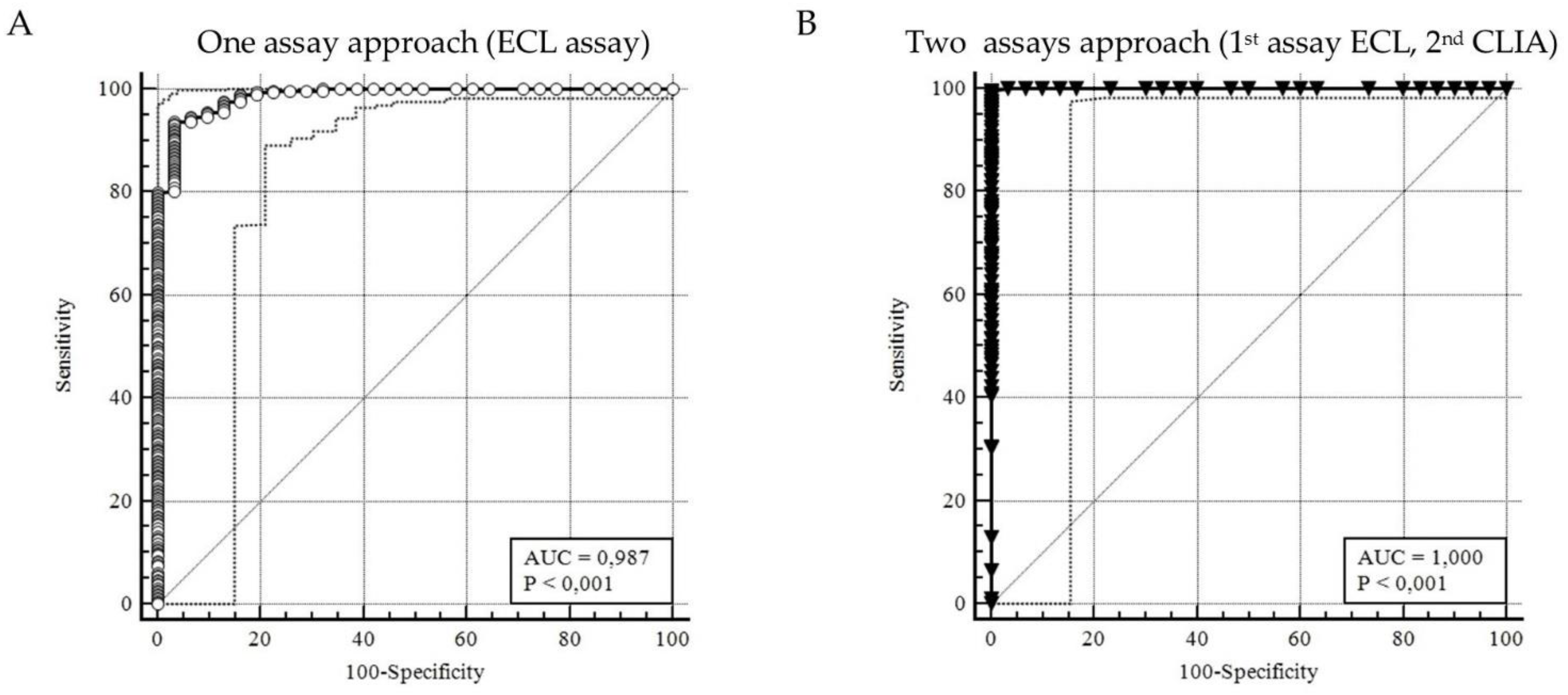
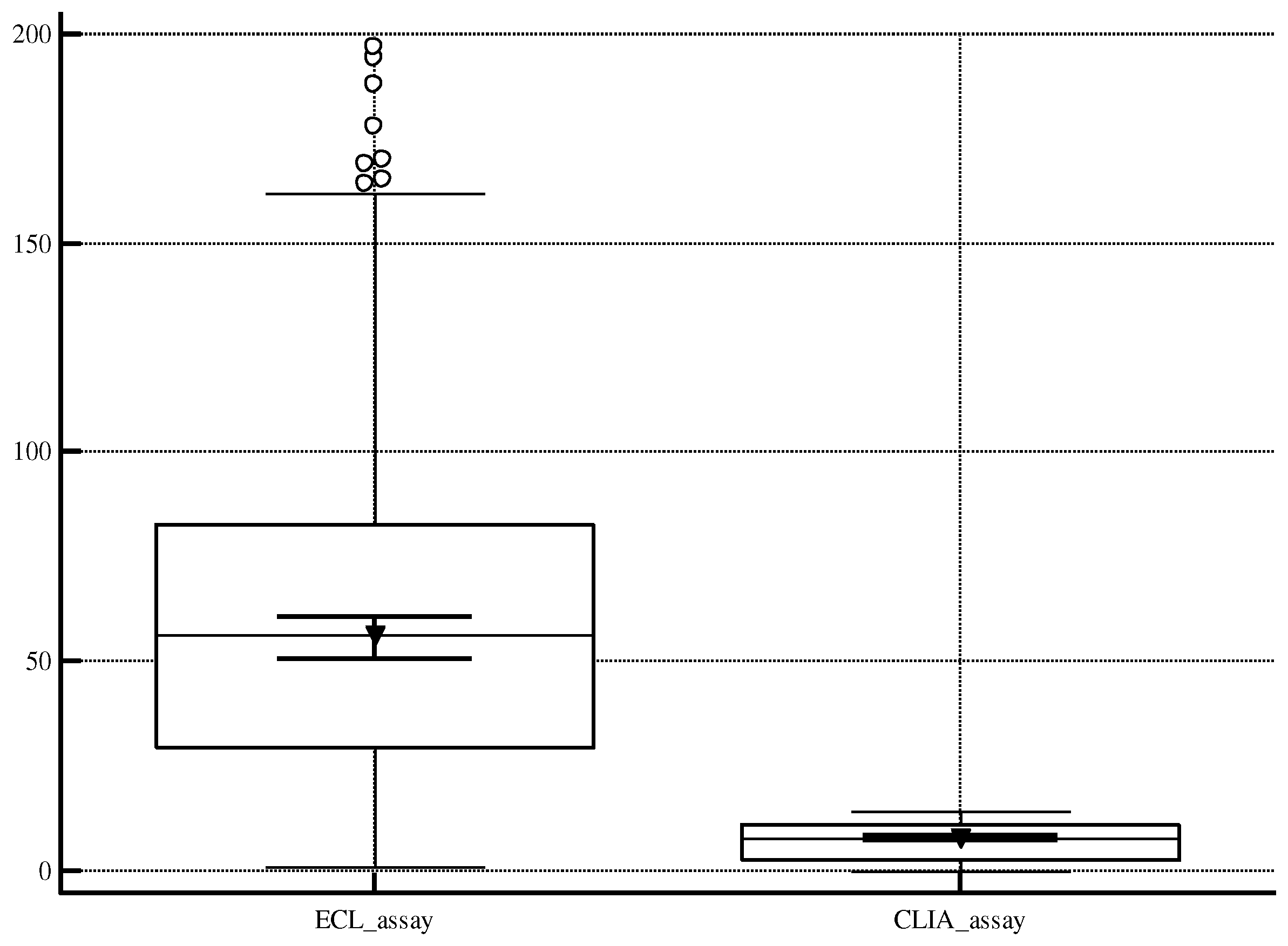
3.3. ROC analyzing ECL screening and CLIA retesting
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Beste, L.A.; Leipertz, S.L.; Green, P.K.; Dominitz, J.A.; Ross, D.; Ioannou, G.N. Trends in Burden of Cirrhosis and Hepatocellular Carcinoma by Underlying Liver Disease in US Veterans, 2001-2013. Gastroenterology 2015, 149, 1471–1482.e5, quiz e17-8. [Google Scholar] [CrossRef]
- Kulik, L.; El-Serag, H.B. Epidemiology and Management of Hepatocellular Carcinoma. Gastroenterology 2019, 156, 477–491.e1. [Google Scholar] [CrossRef]
- GBD 2013 Mortality and Causes of Death Collaborators Global, Regional, and National Age-Sex Specific All-Cause and Cause-Specific Mortality for 240 Causes of Death, 1990-2013: A Systematic Analysis for the Global Burden of Disease Study 2013. Lancet 2015, 385, 117–171. [CrossRef]
- Morozov, V.A.; Lagaye, S. Hepatitis C Virus: Morphogenesis, Infection and Therapy. World J Hepatol 2018, 10, 186–212. [Google Scholar] [CrossRef]
- Manns, M.P.; Buti, M.; Gane, E.; Pawlotsky, J.-M.; Razavi, H.; Terrault, N.; Younossi, Z. Hepatitis C Virus Infection. Nat Rev Dis Primers 2017, 3, 17006. [Google Scholar] [CrossRef]
- Grebely, J.; Prins, M.; Hellard, M.; Cox, A.L.; Osburn, W.O.; Lauer, G.; Page, K.; Lloyd, A.R.; Dore, G.J. International Collaboration of Incident HIV and Hepatitis C in Injecting Cohorts (InC3) Hepatitis C Virus Clearance, Reinfection, and Persistence, with Insights from Studies of Injecting Drug Users: Towards a Vaccine. Lancet Infect Dis 2012, 12, 408–414. [Google Scholar] [CrossRef]
- Blackard, J.T.; Shata, M.T.; Shire, N.J.; Sherman, K.E. Acute Hepatitis C Virus Infection: A Chronic Problem. Hepatology 2008, 47, 321–331. [Google Scholar] [CrossRef]
- Haflidadottir, S.; Jonasson, J.G.; Norland, H.; Einarsdottir, S.O.; Kleiner, D.E.; Lund, S.H.; Björnsson, E.S. Long-Term Follow-up and Liver-Related Death Rate in Patients with Non-Alcoholic and Alcoholic Related Fatty Liver Disease. BMC Gastroenterol 2014, 14, 166. [Google Scholar] [CrossRef]
- Alter, M.J. Epidemiology of Hepatitis C Virus Infection. World J Gastroenterol 2007, 13, 2436–2441. [Google Scholar] [CrossRef] [PubMed]
- Global HIV, H. and Sti.P. (HHS); Guidelines Review Committee Guidelines on Hepatitis B and C Testing; Geneva Switzerland, 2017.
- Rossetti, B.; Bai, F.; Tavelli, A.; Galli, M.; Antinori, A.; Castelli, F.; Pellizzer, G.; Cozzi-Lepri, A.; Bonora, S.; Monforte, A. d’Arminio; et al. Evolution of the Prevalence of Hepatitis C Virus Infection and Hepatitis C Virus Genotype Distribution in Human Immunodeficiency Virus-Infected Patients in Italy between 1997 and 2015. Clin Microbiol Infect 2018, 24, 422–427. [Google Scholar] [CrossRef] [PubMed]
- Kondili, L.A.; Aghemo, A.; Andreoni, M.; Galli, M.; Rossi, A.; Babudieri, S.; Nava, F.; Leonardi, C.; Mennini, F.S.; Gardini, I.; et al. Milestones to Reach Hepatitis C Virus (HCV) Elimination in Italy: From Free-of-Charge Screening to Regional Roadmaps for an HCV-Free Nation. Dig Liver Dis 2022, 54, 237–242. [Google Scholar] [CrossRef] [PubMed]
- Agenzia Italiana del Farmaco (AIFA) Aggiornamento Epatite C; Rome, 2019.
- Martin, N.K.; Skaathun, B.; Vickerman, P.; Stuart, D. Modeling Combination HCV Prevention among HIV-Infected Men Who Have Sex With Men and People Who Inject Drugs. AIDS Rev 2017, 19, 97–104. [Google Scholar] [CrossRef] [PubMed]
- Matsuura, T.; Ohfuji, S.; Enomoto, M.; Tamori, A.; Kubo, S.; Kioka, K.; Kawada, N.; Fukushima, W. Risk Factors for Hepatocellular Carcinoma in Treated Chronic Hepatitis C Patients–Relationship to Smoking and Alcohol. JGH Open 2020, 4, 867–875. [Google Scholar] [CrossRef] [PubMed]
- Schiff, E.R.; Ozden, N. Hepatitis C and Alcohol. Alcohol Res Health 2003, 27, 232–239. [Google Scholar] [CrossRef] [PubMed]
- Maruyama, H.; Shiina, S. Pathogenesis of Hepatitis C Virus-Related Hepatocellular Carcinoma: Evidence from Recent Studies. J Public Health Emerg 2021, 5, 30–30. [Google Scholar] [CrossRef]
- Roudot-Thoraval, F. Epidemiology of Hepatitis C Virus Infection. Clin Res Hepatol Gastroenterol 2021, 45, 101596. [Google Scholar] [CrossRef] [PubMed]
- Easterbrook, P.J. WHO Guidelines Development Group Who to Test and How to Test for Chronic Hepatitis C Infection - 2016 WHO Testing Guidance for Low- and Middle-Income Countries. J Hepatol 2016, 65, S46–S66. [Google Scholar] [CrossRef] [PubMed]
- Freiman, J.M.; Tran, T.M.; Schumacher, S.G.; White, L.F.; Ongarello, S.; Cohn, J.; Easterbrook, P.J.; Linas, B.P.; Denkinger, C.M. Hepatitis C Core Antigen Testing for Diagnosis of Hepatitis C Virus Infection: A Systematic Review and Meta-Analysis. Ann Intern Med 2016, 165, 345–355. [Google Scholar] [CrossRef]
- Richter, S.S. Laboratory Assays for Diagnosis and Management of Hepatitis C Virus Infection. J Clin Microbiol 2002, 40, 4407–4412. [Google Scholar] [CrossRef]
- Zachary, P.; Ullmann, M.; Djeddi, S.; Meyer, N.; Wendling, M.-J.; Schvoerer, E.; Stoll-Keller, F.; Gut, J.-P. Evaluation of Three Commercially Available Hepatitis C Virus Antibody Detection Assays under the Conditions of a Clinical Virology Laboratory. Journal of Clinical Virology 2005, 34, 207–210. [Google Scholar] [CrossRef]
- Lai, K.K.Y.; Jin, M.; Yuan, S.; Larson, M.F.; Dominitz, J.A.; Bankson, D.D. Improved Reflexive Testing Algorithm for Hepatitis C Infection Using Signal-to-Cutoff Ratios of a Hepatitis C Virus Antibody Assay. Clin Chem 2011, 57, 1050–1056. [Google Scholar] [CrossRef]
- Oethinger, M.; Mayo, D.R.; Falcone, J.; Barua, P.K.; Griffith, B.P. Efficiency of the Ortho VITROS Assay for Detection of Hepatitis C Virus-Specific Antibodies Increased by Elimination of Supplemental Testing of Samples with Very Low Sample-to-Cutoff Ratios. J Clin Microbiol 2005, 43, 2477–2480. [Google Scholar] [CrossRef]
- Choi, M.S.; Lee, K.; Hong, Y.J.; Song, E.Y.; Kim, D.S.; Song, J. The Role of the Signal-to-Cutoff Ratio in Automated Anti-HCV Chemiluminescent Immunoassays by Referring to the Nucleic Acid Amplification Test and the Recombinant Immunoblot Assay. Ann Lab Med 2018, 38, 466–472. [Google Scholar] [CrossRef]
- Nahm, F.S. Receiver Operating Characteristic Curve: Overview and Practical Use for Clinicians. Korean J Anesthesiol 2022, 75, 25–36. [Google Scholar] [CrossRef]
- Petrosillo, N.; Colucci, A.; Luzi, A.M.; Gallo, P.; Palmieri, F. ; National Focal Point (NFP) italiano [Italian Public and Non-Governmental Organisations Related to HIV/AIDS Infection and Migrant Populations]. Ann Ist Super Sanita 2004, 40, 433–439. [Google Scholar]
- Nicolai, E.; Sarubbi, S.; Pelagalli, M.; Basile, V.; Terrinoni, A.; Minieri, M.; Cennamo, O.; Grelli, S.; Bernardini, S.; Pieri, M. Performance Evaluation of the New Chemiluminescence Immunoassay CL-1200i for HBV, HIV Panels. Diseases 2023, 11. [Google Scholar] [CrossRef]
- Workowski, K.A.; Bachmann, L.H.; Chan, P.A.; Johnston, C.M.; Muzny, C.A.; Park, I.; Reno, H.; Zenilman, J.M.; Bolan, G.A. Sexually Transmitted Infections Treatment Guidelines, 2021. MMWR. Recommendations and Reports 2021, 70, 1–187. [Google Scholar] [CrossRef]
- World Health Organization (WHO) Guidelines on Hepatitis B and C Testing: Policy Brief. World Health Organization; Geneva Switzerland, 2016.
- Yoo, S.J.; Wang, L.L.; Ning, H.-C.; Tao, C.M.; Hirankarn, N.; Kuakarn, S.; Yang, R.; Han, T.H.; Chan, R.C.; Hussain, B.M.; et al. Evaluation of the Elecsys(®) Anti-HCV II Assay for Routine Hepatitis C Virus Screening of Different Asian Pacific Populations and Detection of Early Infection. J Clin Virol 2015, 64, 20–27. [Google Scholar] [CrossRef]
- Maugard, C.; Relave, J.; Klinkicht, M.; Fabra, C. Clinical Performance Evaluation of Elecsys HIV Duo, Anti-HCV II, HBsAg II, Anti-HBc II, and Syphilis Assays for Routine Screening of First-Time Blood Donor Samples at a French Blood Donation Center. Transfus Clin Biol 2022, 29, 79–83. [Google Scholar] [CrossRef]
- Huang, Y.; Pan, H.; Gao, Q.; Lv, P.; Xu, X.; Zhao, Z. The Role of a Two-Assay Serological Testing Strategy for Anti-HCV Screening in Low-Prevalence Populations. Sci Rep 2021, 11, 8689. [Google Scholar] [CrossRef]
- Geue, C.; Wu, O.; Xin, Y.; Heggie, R.; Hutchinson, S.; Martin, N.K.; Fenwick, E.; Goldberg, D.; Consortium; ECDC. Cost-Effectiveness of HBV and HCV Screening Strategies--A Systematic Review of Existing Modelling Techniques. PLoS One 2015, 10, e0145022. [CrossRef]
| Classification | Confirmed positive by RIBA | Confirmed negative by RIBA | Indeterminate for RIBA | PPV | NPV | χ2 test | p value |
|---|---|---|---|---|---|---|---|
| One assay | 298 | 31 | 21 | 90.6% | ND | 34.51 | < 0.001 |
| Two assays | 283 | 31 | 12 | 95.00% | 77.50% | 7.59 | < 0.01 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




