Submitted:
01 January 2024
Posted:
05 January 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction

1.1. Effect of ASR on Durability of Concrete

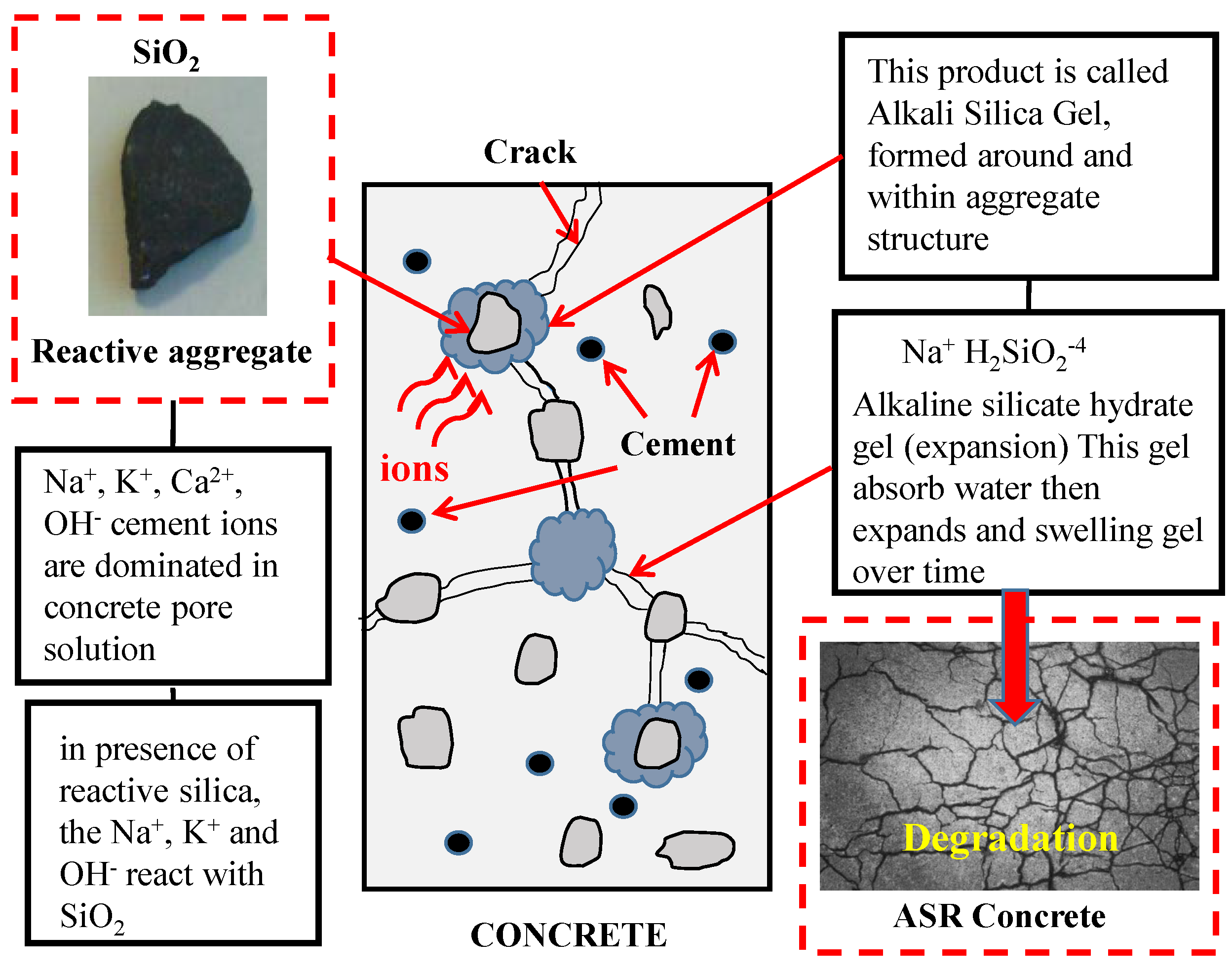
1.2.1. Reactive Aggregates
1.2.2. Alkalinity of cementations materials
2. Materials and Methods
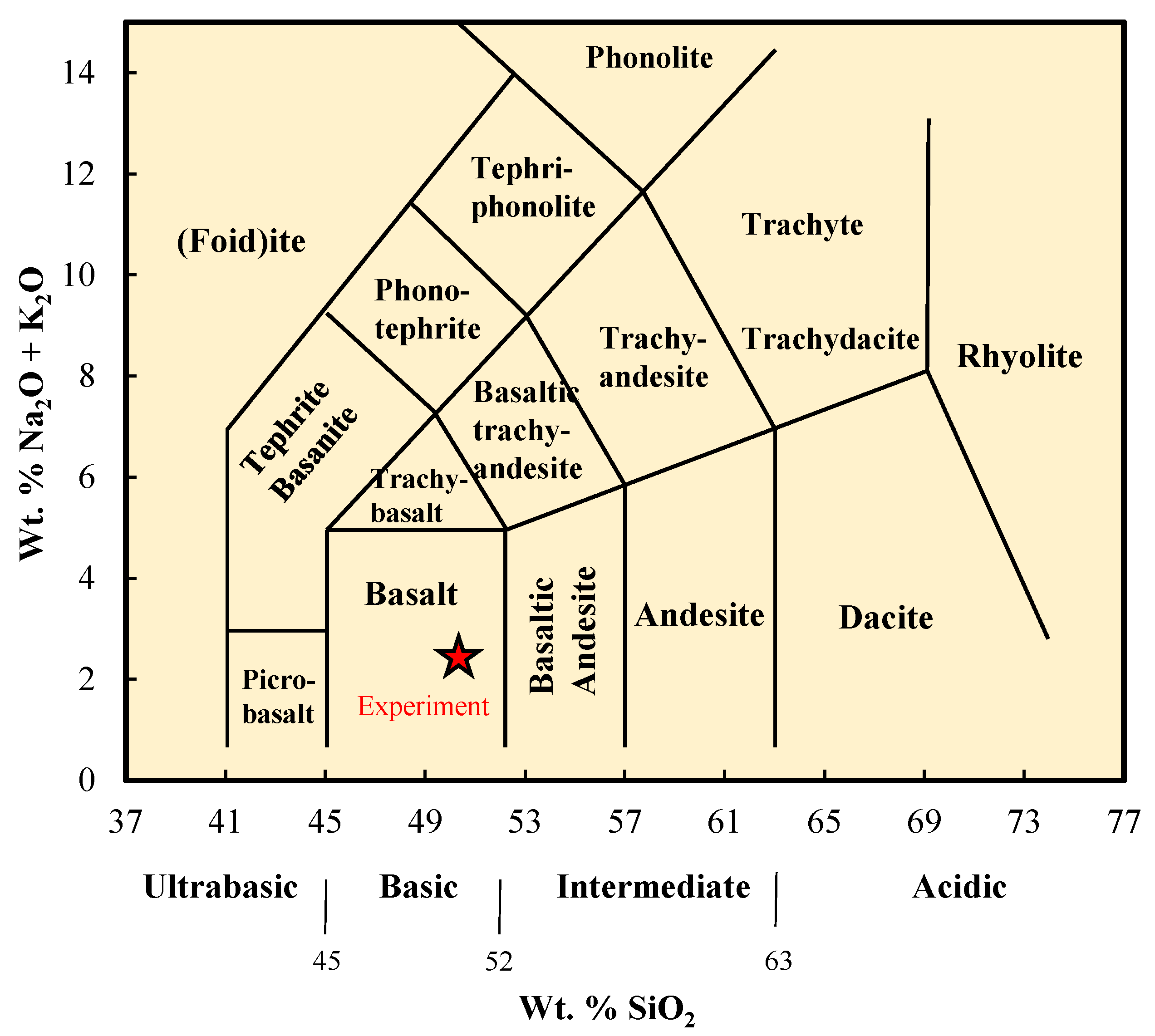
2.1. Materials
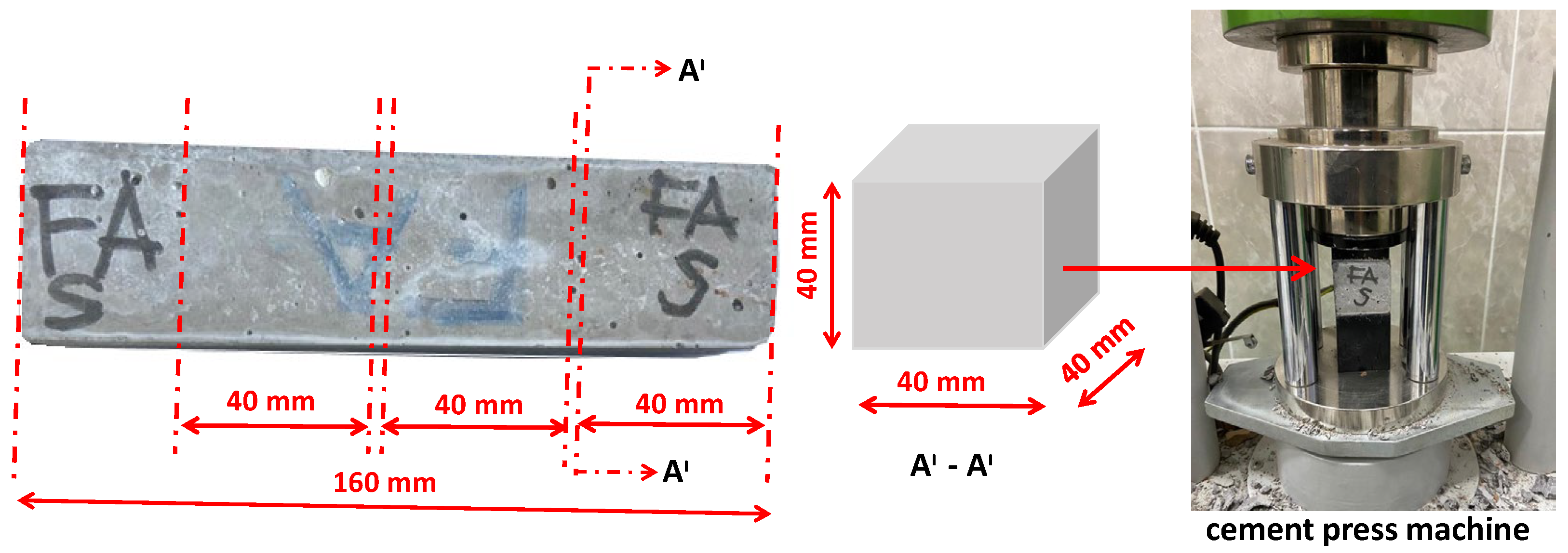
2.2. Preparation of Specimens
| Specimens | Sieves | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| No:4 | No:8 | No:16 | No:30 | No:50 | ||||||
| %10 | %25 | %25 | %25 | %15 | ||||||
| Aggregate | C1 | FA2 | W3 | W/B4 | C/A5 | |||||
| Gram | ||||||||||
| PL | 99 | 247.5 | 247.5 | 247.5 | 148.5 | 440 | - | 206.8 | 0.47 | 2.25 |
| BBC | 99 | 247.5 | 247.5 | 247.5 | 148.5 | 440 | - | 206.8 | 0.47 | 2.25 |
| BBC20FA | 99 | 247.5 | 247.5 | 247.5 | 148.5 | 352 | 88 | 206.8 | 0.47 | 2.25 |
| 1C=Cement; 2FA= Fly Ash;3 W= Water; 4W/B= Water- Binding ratio; 5C/A= Cement- Aggregate ratio | ||||||||||
2.3. Mechanical Properties
2.4. Mineralogical and Chemical Characterisation
3. Results and Discussion
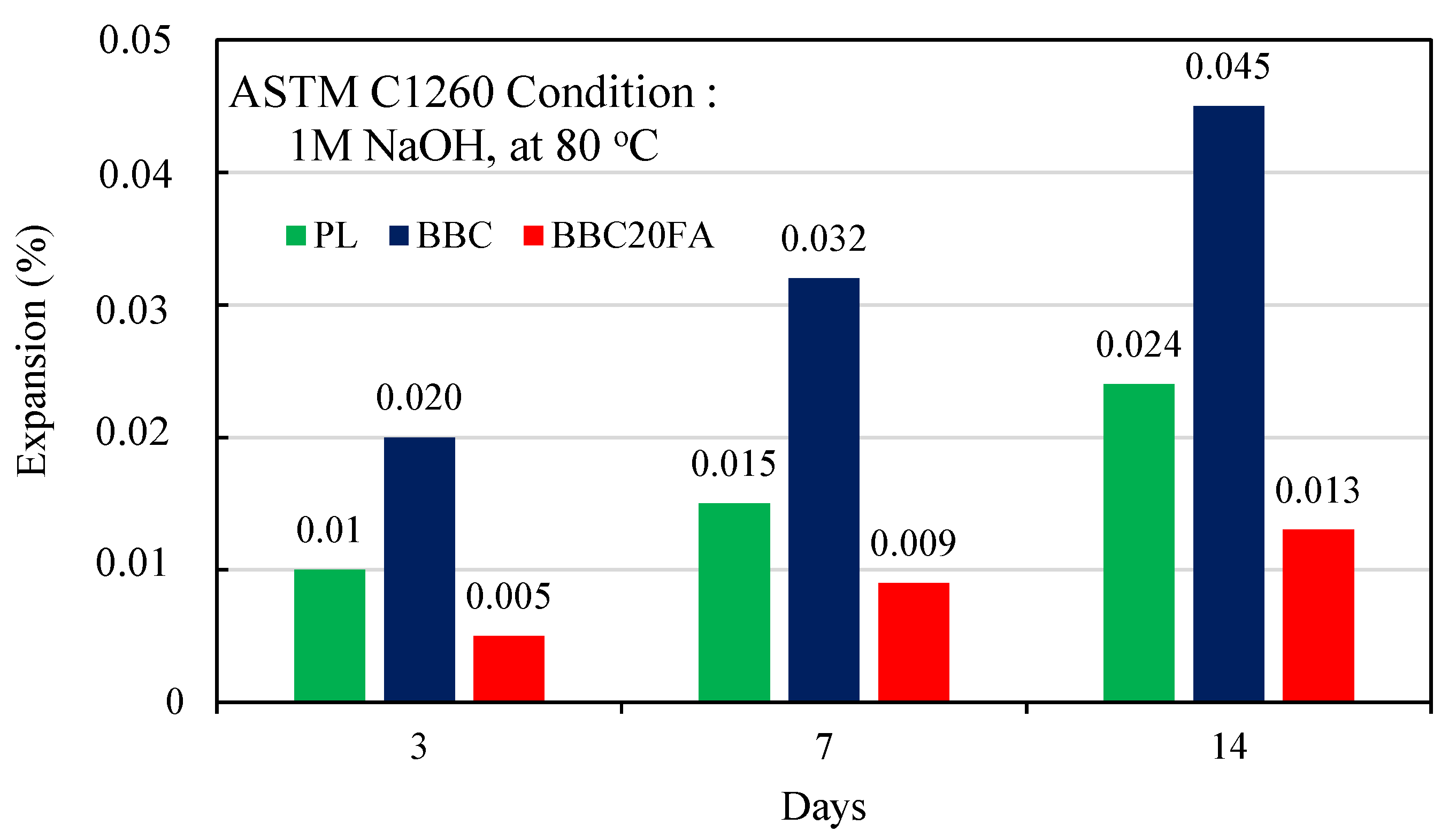
3.1. Accelerated Mortar Bar Testing (AMBT) ASTM C1260
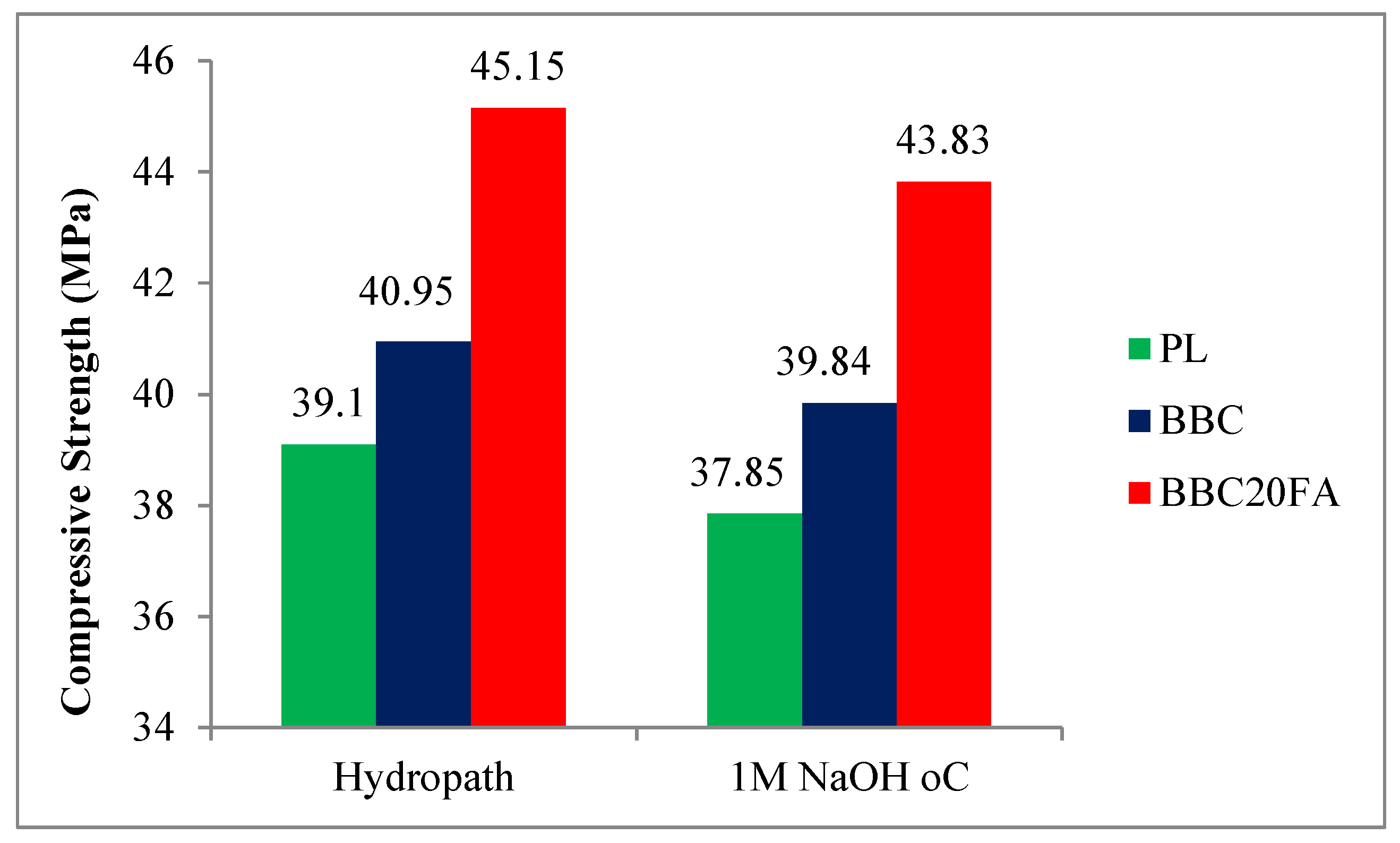
3.2. Compressive Strength
| Hydropath | 1M NaOH 80 oC | Strength loss (CSL),% | |
|---|---|---|---|
| PL | 39.10 MPa | 37.85 MPa | -3.19 |
| BBC | 40.95 MPa | 39.84 MPa | -2.79 |
| BBC20FA | 45.15 MPa | 43.83 MPa | -3.01 |
| FAE* | +10.25% | +10.01% | |
| FAE* = Effect of Fly Ash admixture on compressive strength of concrete | |||
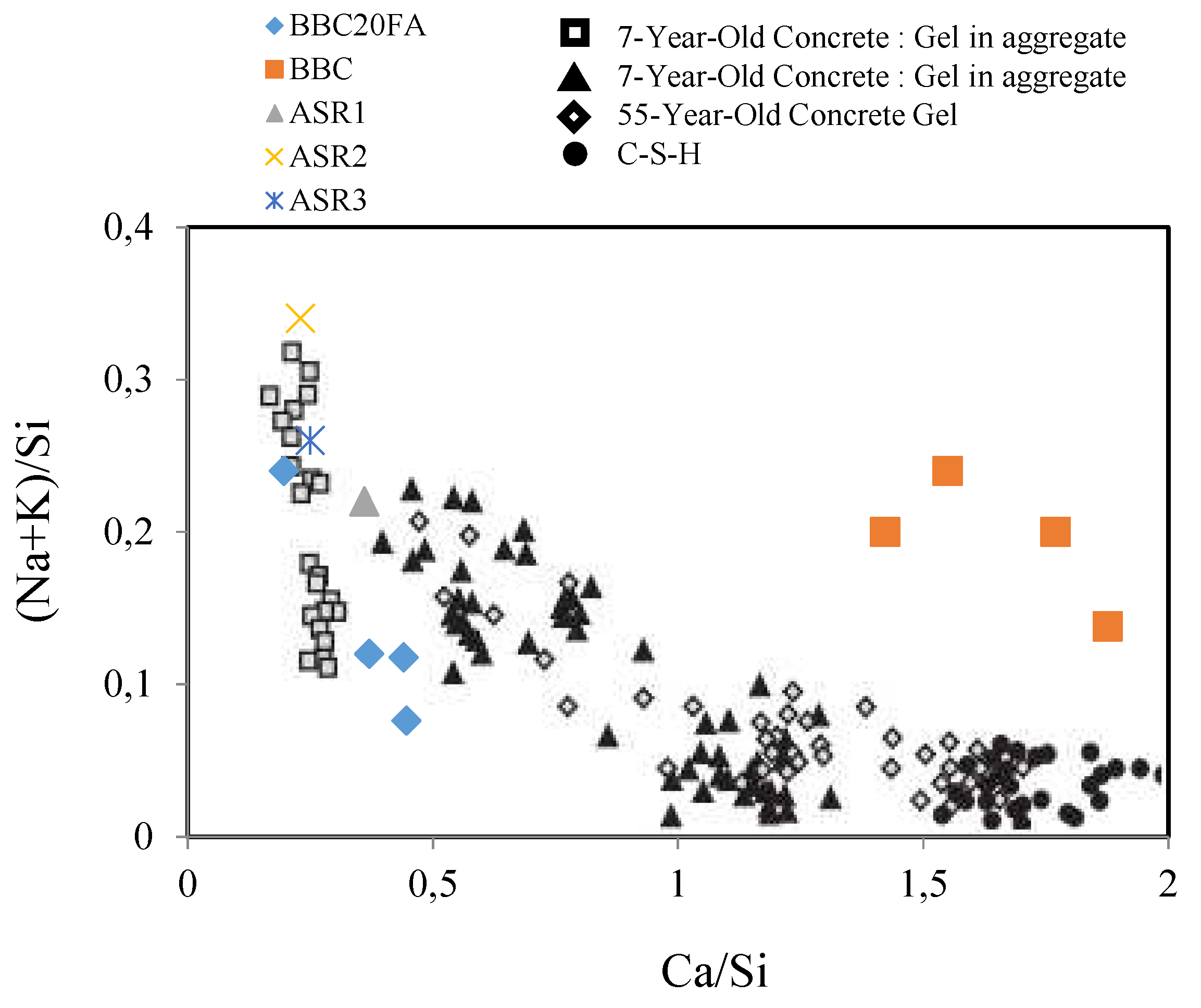
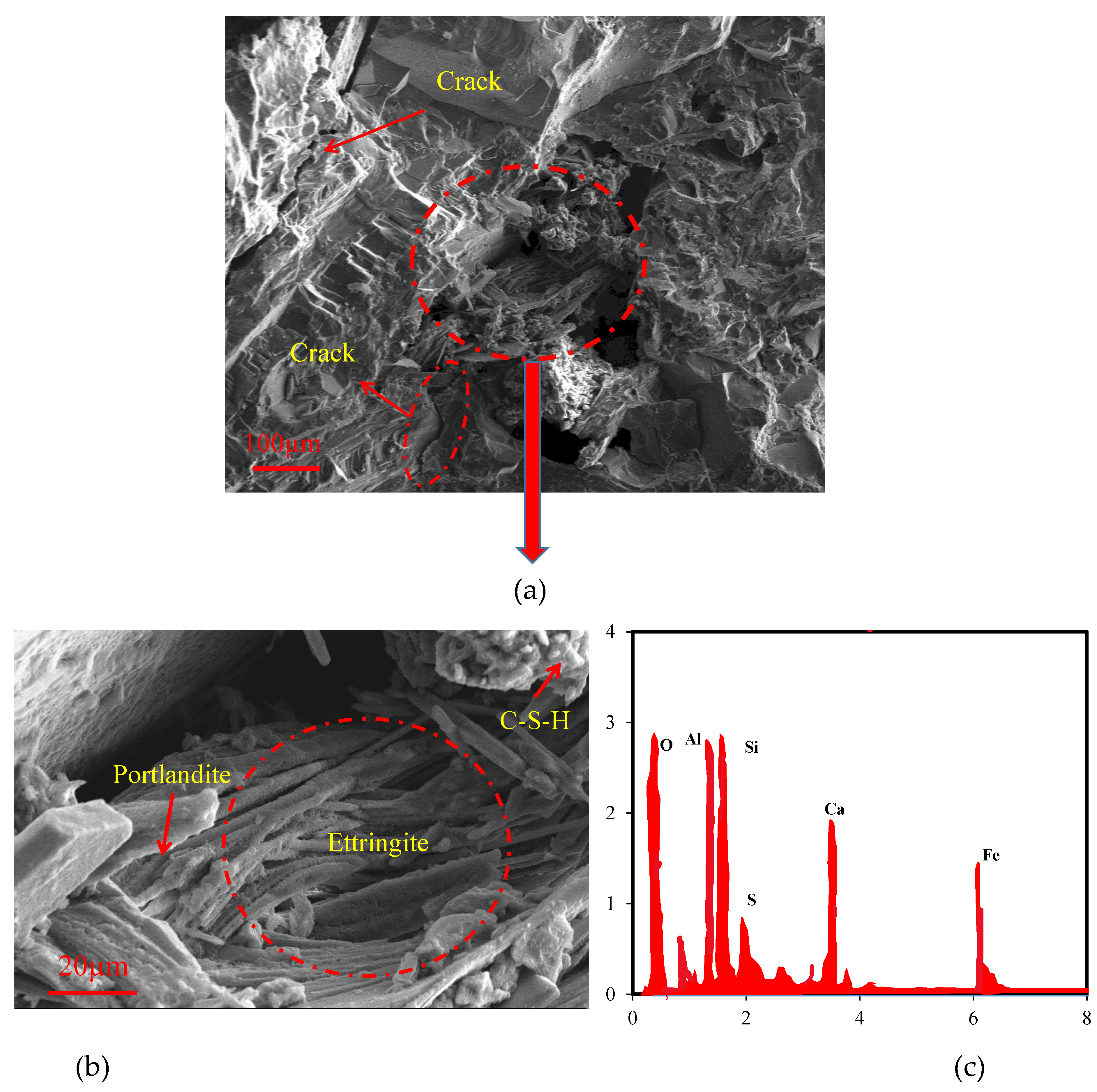
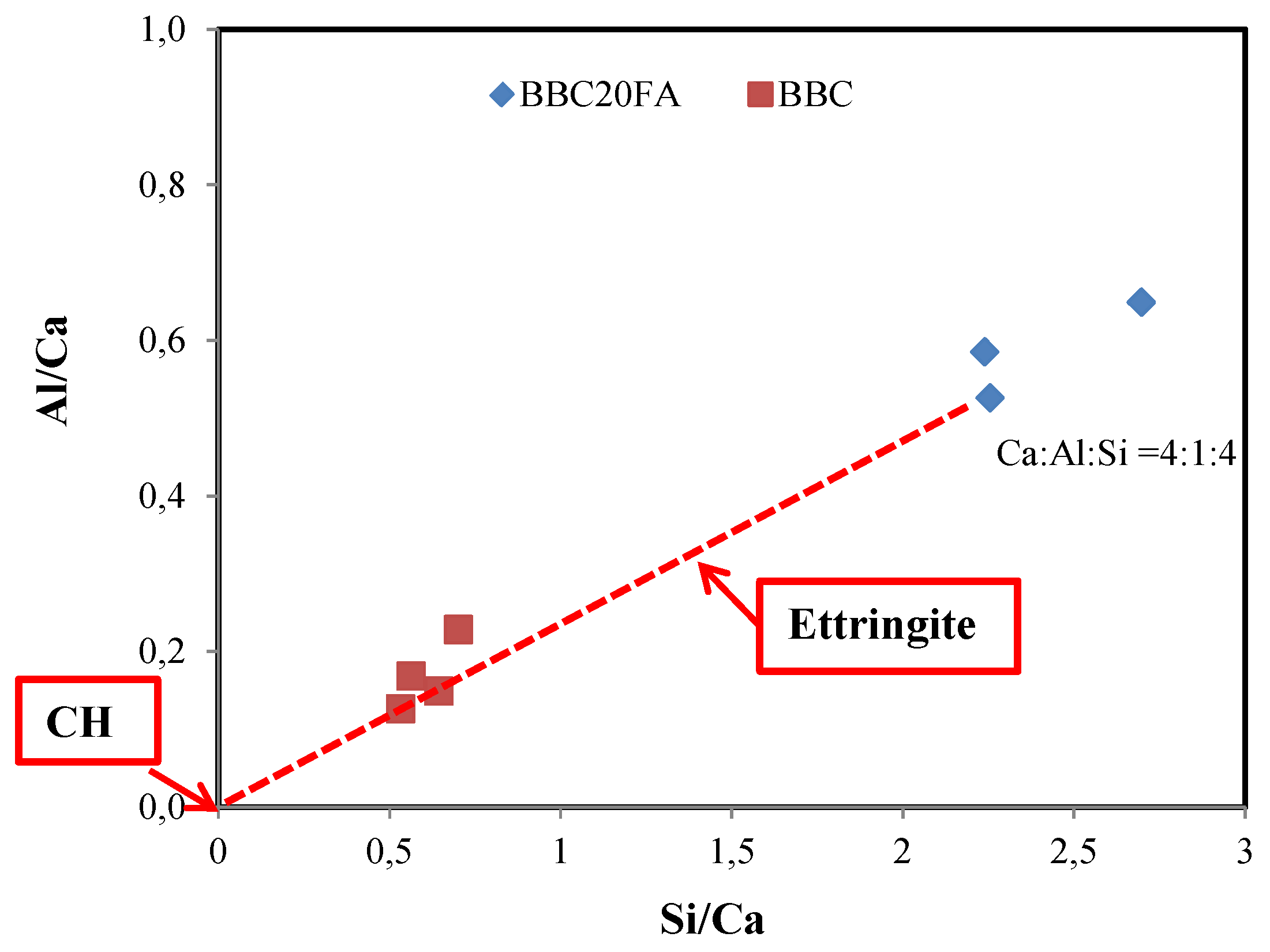
3.3. SEM-EDX analysis
| Specimens | Ca | Si | Al | Na | K | Fe | Ti | Mg | O | |||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Atom % | ||||||||||||
| BBC | 19.5 | 11.0 | 3.3 | 1.9 | 0.4 | 1.8 | - | 0.3 | 46.3 | 0.20 | 1.77 | 4.95 |
| BBC20FA | 6.8 | 15.3 | 8.0 | 1.5 | 0.3 | 0.3 | .08 | .04 | 67.0 | 0.12 | 0.44 | 4.62 |
| ASR1 | 5.3 | 20.9 | 0.4 | 1.4 | 3.2 | 0.1 | - | - | 68.6 | 0.22 | 0.36 | 0.46 |
| ASR2 | 4.5 | 19.4 | - | 2.8 | 3.7 | 0.5 | - | - | 69.0 | 0.34 | 0.23 | 0.76 |
| ASR3 | 4.5 | 19.9 | - | 0.8 | 3.6 | - | - | - | 71.2 | 0.26 | 0.25 | 0.61 |
| 1,2,3ASR: ASR concrete (Leeman et.al. 2011) | ||||||||||||
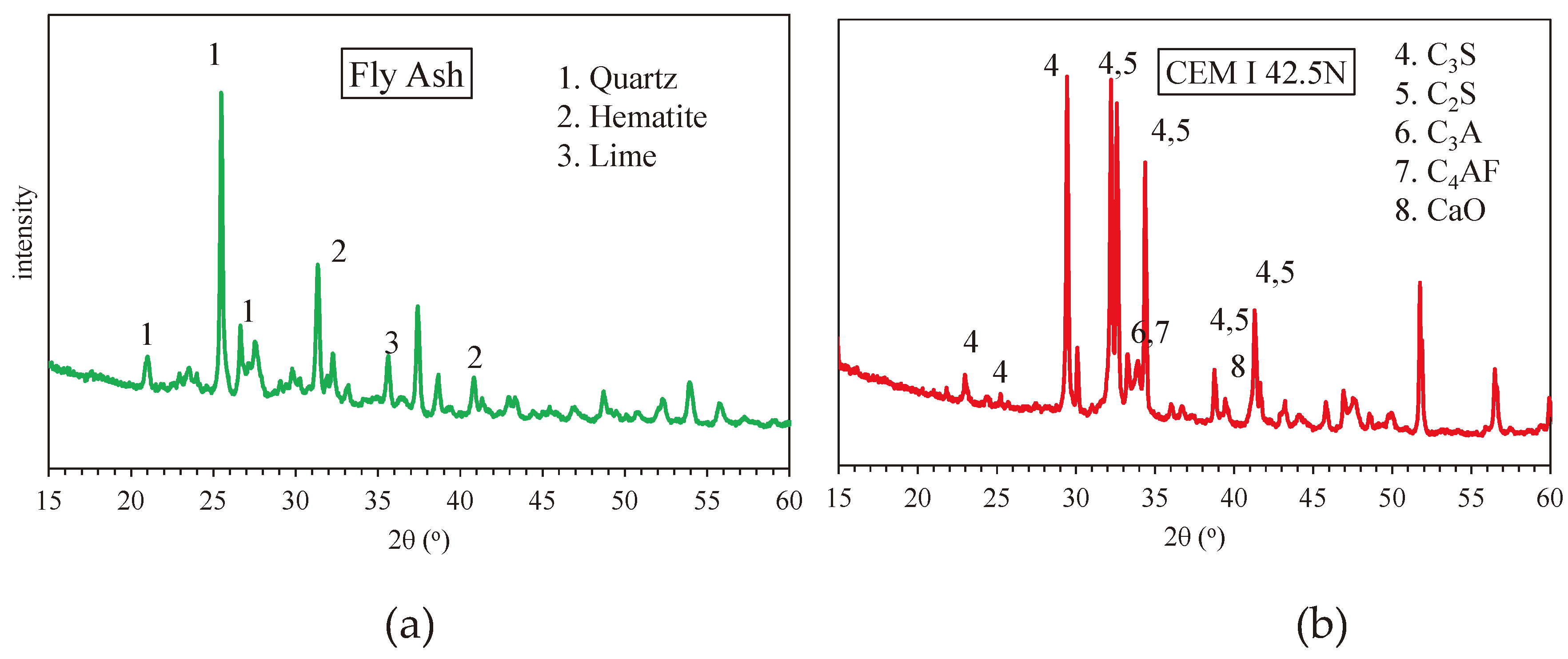
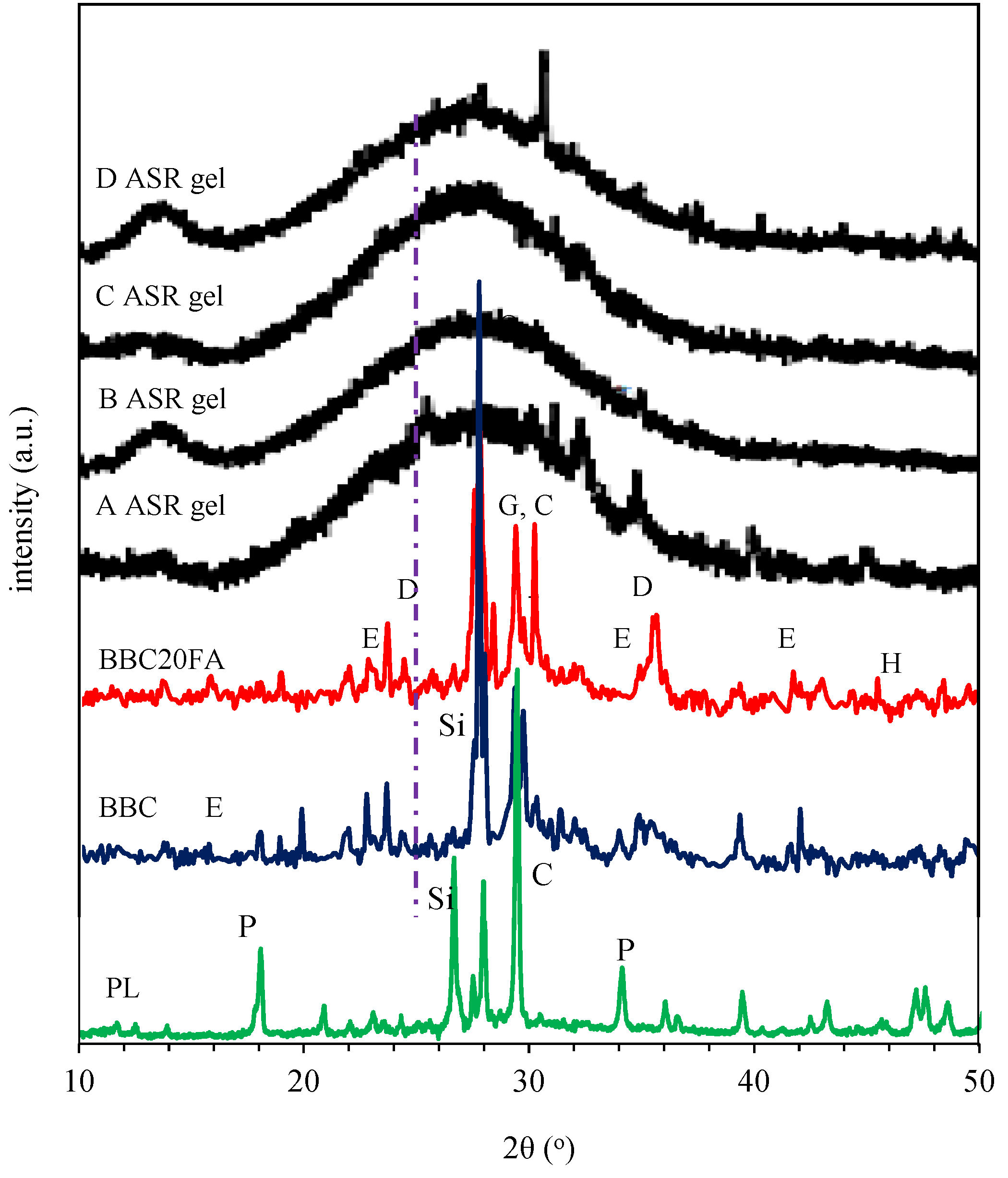
3.2.3. X-Ray Diffraction (XRD)
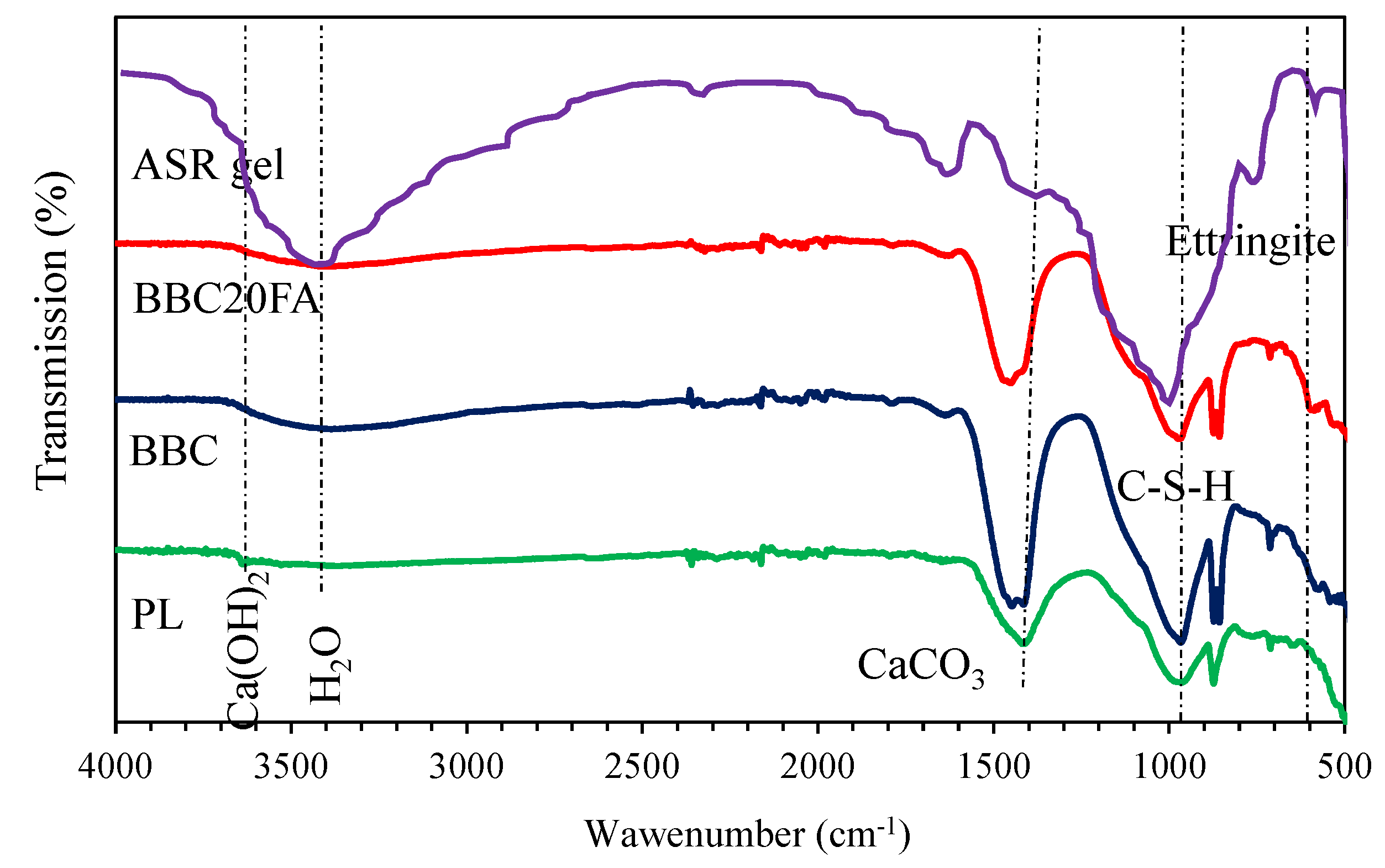
3.2.4. FT-IR
| Wave number range (cm-1) | Assignment | Compound formation | References |
|---|---|---|---|
| 530-558 | Si-O out of plane bending | Ettringite | [71,72,73] |
| 882-890 | CO32- | Carbonates | [73] |
| 969-1004 | Si-O stretching and vibration | C-S-H | [71,73,74,75] |
| 1520-1524 | CO32- | Calcium carbonate | [74,75] |
| 1636-1646 | H-O-H | - | [73,76] |
| 1640-1650 | C-H bending | Chemically bonded water | [73,76] |
| 3200-3400 | O-H | H2O | [74,77] |
| 3618-3627 | O-H | Portlandite | [72,74] |
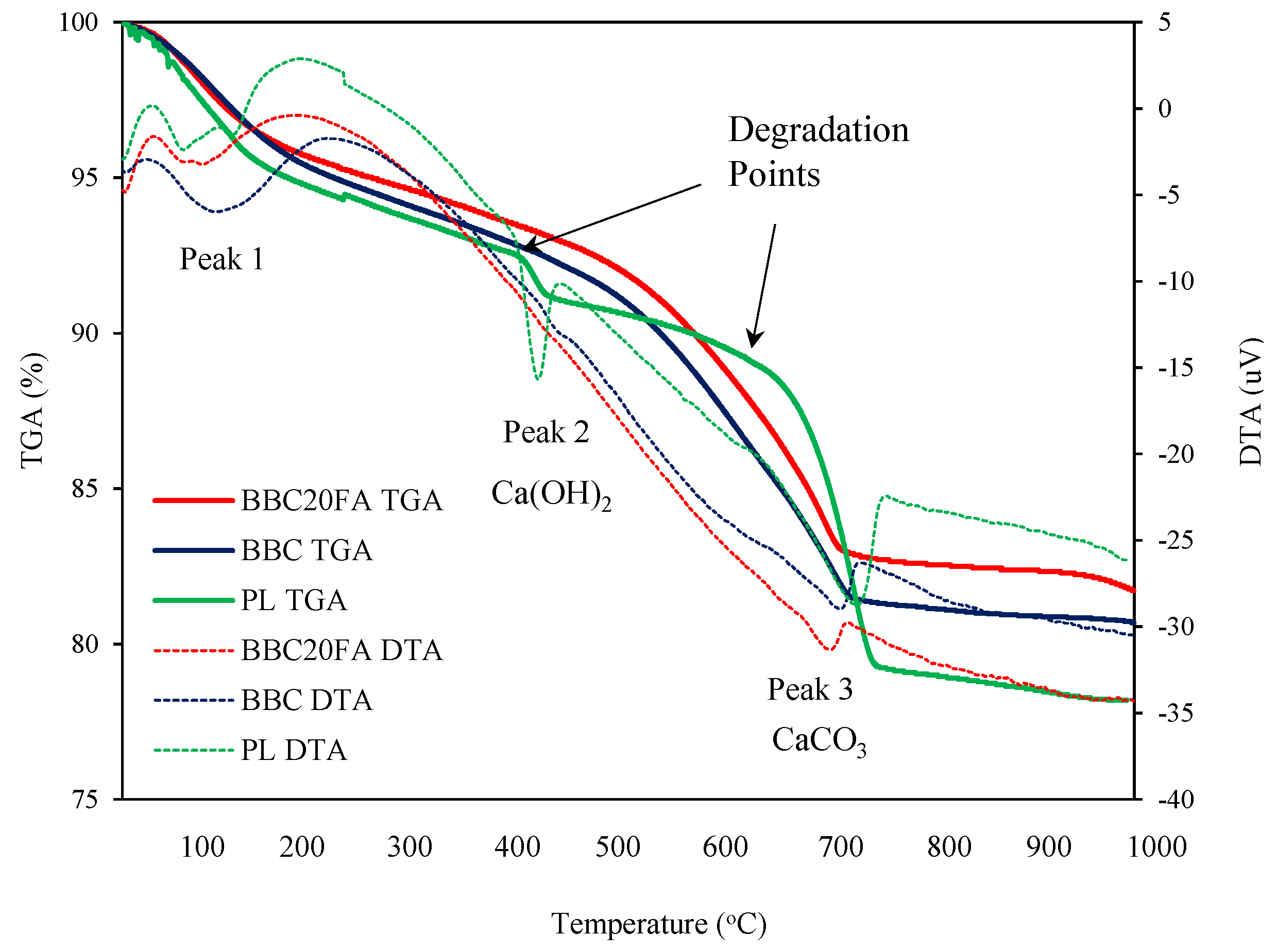
3.2.5. Thermogravimetric and Differential Thermal Analysis (TGA/ DTA)
5. Conclusions
References
- Y.E. Erdoğan, Materials of Construction, M.E.T.U. Press, Ankara, 2002.
- B. Baradan, Yapı Malzemesi II, D.E.Ü. Müh. Fak. Yay, İzmir, 2004.
- Ö. Andiç-Çakır, Investigation of Test Methods on Alkali Aggregate Reaction, Ege University Institute of Science, 2007.
- R. Kambiz, Alkali-silica reaction in concrete a review, in: In Proceedings of the 2013 Ready Concrete Congress, İstanbul, 2013: pp. 289–311.
- T. Katayama, A Crıtıcal Revıew of Carbonate Rock Reactıons-Is Theır Reactıvıty Useful Or Harmful ?, 1992.
- T. Katayama, Alkali Aggregate Reaction In The Vicinity Of Izmir, Western Turkey, Alkali Aggregate Reaction In Concrete, in: Proceedings of 11th International Conference, Canada, 2000.
- F. Rajabipour, E. F. Rajabipour, E. Giannini, C. Dunant, J.H. Ideker, M.D.A. Thomas, Alkali-silica reaction: Current understanding of the reaction mechanisms and the knowledge gaps, Cem Concr Res. 76 (2015) 130–146. [CrossRef]
- T. Ichikawa, M. T. Ichikawa, M. Miura, Modified model of alkali-silica reaction, Cem Concr Res. 37 (2007) 1291–1297. [CrossRef]
- Karasin, M. Hadzima-Nyarko, E. Işık, M. Doğruyol, I.B. Karasin, S. Czarnecki, The Effect of Basalt Aggregates and Mineral Admixtures on the Mechanical Properties of Concrete Exposed to Sulphate Attacks, Materials. 15 (2022). [CrossRef]
- Leemann, P. Lura, E-modulus of the alkali-silica-reaction product determined by micro-indentation, Constr Build Mater. 44 (2013) 221–227. [CrossRef]
- T. Katayama, Petrographic Study of the Alkali-aggregate Reactions in Concrete, Grad. Sch. Sci. Univ. Tokyo, Dep. Earth Planet. Sci,. (2012).
- Z. Shi, A. Z. Shi, A. Leemann, D. Rentsch, B. Lothenbach, Synthesis of alkali-silica reaction product structurally identical to that formed in field concrete, Mater Des. 190 (2020). [CrossRef]
- C.E. Tambelli, J.F. C.E. Tambelli, J.F. Schneider, N.P. Hasparyk, P.J.M. Monteiro, Study of the structure of alkali-silica reaction gel by high-resolution NMR spectroscopy, J Non Cryst Solids. 352 (2006) 3429–3436. [CrossRef]
- M Imaoka,. H. Hasegawa, I.Yasui. X-ray dıffractıon study of the structure of sılıcate glasses. Iı: alkalı dısılıcate glasses., (1983).
- T.E. Stanton, Influence of cement and aggregate on concrete expansion, Eng. NewsRecord. 1, 1940.
- Mohammadi, E. Ghiasvand, M. Nili, Relation between mechanical properties of concrete and alkali-silica reaction (ASR); a review, Constr Build Mater. 258 (2020). [CrossRef]
- J. Duchesne, M. A. J. Duchesne, M. A. Bérubé, The effectiveness of supplementary cementing materials in suppressing expansion due to ASR: another look at the reaction mechanisms part 2: pore solution chemistry., 24(2), 221-230, Cem Concr Res. 24 (1994) 221–230.
- B Fournier, M. A Bérubé, K. J.F olliard, M. Thomas, Report on the diagnosis, prognosis, and mitigation of Alkali-Silica Reaction (ASR) in transportation structures (No. FHWA-HIF-09-004)., 2010.
- M.A. Bérubé, C. M.A. Bérubé, C. Tremblay, B. Fournier, M.D. Thomas, D.B. Stokes, Influence of lithium-based products proposed for counteracting ASR on the chemistry of pore solution and cement hydrates, Cem Concr Res. 34 (2004) 1645–1660. [CrossRef]
- S. Poyet, A. S. Poyet, A. Sellier, B. Capra, G. Foray, J.M. Torrenti, H. Cognon, E. Bourdarot, Chemical modelling of Alkali Silica reaction: Influence of the reactive aggregate size distribution, Materials and Structures/Materiaux et Constructions. 40 (2007) 229–239. [CrossRef]
- S.A. Mohammad Shahidul Islam, A Critical Assessment to the Performance of Alkali-Silica Reaction (ASR) in Concrete, Can Chem Trans. (2013) 253–266. [CrossRef]
- M. Kawamura, H. M. Kawamura, H. Fuwa, Effects of lithium salts on ASR gel composition and expansion of mortars, Cem Concr Res. 33 (2003) 913–919. [CrossRef]
- T. Kurihara, K Katawaki, Effects of moisture control and inhibition on alkali silica reaction, in: 8th ICAAR, International Conference on Alkali Aggregate Reactions, Kyoto, 1989: pp. 629–634.
- D. W Hobbs, J. D. W Hobbs, J. Bensted, P. Barnes, Alkali–Silica Reaction in Concrete, in: Structure and Performance of Cement, 2002: pp. 265–281.
- F. Gaboriaud, D. F. Gaboriaud, D. Chaumont, A. Nonat, A. Craeivich, Study of the Influence of Alkaline Ions (Li, Na and K) on the Structure of the Silicate Entities in Silico Alkaline Sol and on the Formation of the Silico-Calco-Alkaline Gel, 1998.
- F Gaboriaud, A. , Nonat, D. Chaumont, A.Craievich, Aggregation and gel formation in basic silico-calco-alkaline solutions studied: a SAXS, SANS, and ELS study,, J Phys Chem B. (1999) 5775–5781.
- S. Diamond, ASR — another look at mechanisms, Proc., in: 8th International Conf. on Alkali–Aggregate Reaction (ICAAR), Kyoto, 1989: pp. 83–94.
- E.O. Fanijo, J.T. E.O. Fanijo, J.T. Kolawole, A. Almakrab, Alkali-silica reaction (ASR) in concrete structures: Mechanisms, effects and evaluation test methods adopted in the United States, Case Studies in Construction Materials. 15 (2021). [CrossRef]
- Baradan, H. Yazici, H. Ün, Betonarme Yapılarda Kalıcılık(Durabilite)., D.E.Ü. Müh. Fak. Yayın, 2002.
- Tremblay, M.A. Bérubé, B. Fournier, M.D. Thomas, K.J. Folliard, Experimental investigation of the mechanisms by which LiNO3 is effective against ASR, Cem Concr Res. 40 (2010) 583–597. [CrossRef]
- R.W. le Maitre, A classification of Igneous Rocks and Glossary of Terms, Blackwell Scientific Publ, Oxford, UK, 1989.
- P. Hagelia, I. P. Hagelia, I. Fernandes, ON THE AAR SUSCEPTIBILITY OF GRANITIC AND QUARTZITIC AGGREGATES IN VIEW OF PETROGRAPHIC CHARACTERISTICS AND ACCELERATED TESTING, n.d.
- M.H. Shehata, M.D.A. M.H. Shehata, M.D.A. Thomas, R.F. Bleszynski, The effects of fly ash composition on the chemistry of pore solution in hydrated cement pastes, 1999.
- P. Kumar Mehta, P.J. P. Kumar Mehta, P.J. M Monteiro, Concrete: Microstructure, Properties, and Materials, Fourth Edition, n.d.
- BS EN 196-6, BS EN 196-6:2018 Methods of testing cement is classified in these ICS categories: 91.100.10 Cement. Gypsum. Lime. Mortar, 2018.
- ASTM 204, Standard Test Methods for Fineness of Hydraulic Cement by Air-Permeability Apparatus, (2013).
- Standard, B. 8007: Design of concrete structures for retaining aqueous liquids. BSI, London, UK., 1987.
- ASTM (American Society for Testing and Materials) Standards (2006a) Standard test method for the resistance to degradation of small-size coarse aggregates by abrasion and impact in the Los Angeles Machine, C131–03, v. 04.02, USA, 2006.
- ASTM Standard Standard Test Method for Tensile Strength of Monolithic Advanced Ceramics at Ambient Temperatures, C 1273-05. Annual Book of ASTM Standards, Vol. 15.01, American Society of Testing and Materials, Philadelphia, PA., 2005.
- B. 85 (2006) 2537–2544. [CrossRef]
- R.T. Chancey, P. R.T. Chancey, P. Stutzman, M.C.G. Juenger, D.W. Fowler, Comprehensive phase characterization of crystalline and amorphous phases of a Class F fly ash, Cem Concr Res. 40 (2010) 146–156. [CrossRef]
- X. 6 (2013) 1485–1495. [CrossRef]
- P. Duxson, A. P. Duxson, A. Fernández-Jiménez, J.L. Provis, G.C. Lukey, A. Palomo, J.S.J. Van Deventer, Geopolymer technology: The current state of the art, J Mater Sci. 42 (2007) 2917–2933. [CrossRef]
- M.L. Granizo, S. M.L. Granizo, S. Alonso, M.T. Blanco-Varela, A. Palomo, Alkaline activation of metakaolin: Effect of calcium hydroxide in the products of reaction, in: Journal of the American Ceramic Society, American Ceramic Society, 2002: pp. 225–231. [CrossRef]
- C.K. Yip, J.S.J. C.K. Yip, J.S.J. Van Deventer, Microanalysis of calcium silicate hydrate gel formed within a geopolymeric binder, n.d.
- S.-D. Wang, K.L. S.-D. Wang, K.L. Scrivener, P.L. Pratt, 0008-8846(94) 00046-8 FACTORS AFFECTING THE STRENGTH OF ALKALI-ACTIVATED SLAG, 1994.
- ASTM C 1260, “Standart Test Method for Potential Alkali Reactivity of Aggregates (Mortar-Bar Method)”, Annual Book of ASTM Standarts,, Philadelphia, USA., n.d.
- B. Middendorf, J.J. B. Middendorf, J.J. Hughes, K. Callebaut, G. Baronio, I. Papayianni, Investigative methods for the characterisation of historic mortars - Part 1: Mineralogical characterisation, Materials and Structures/Materiaux et Constructions. 38 (2005) 761–769. [CrossRef]
- Leemann, G. le Saout, F. Winnefeld, D. Rentsch, B. Lothenbach, Alkali-Silica reaction: The Influence of calcium on silica dissolution and the formation of reaction products, Journal of the American Ceramic Society. 94 (2011) 1243–1249. [CrossRef]
- M.l Thomas, The role of calcium hydroxide in alkali recycling in concrete, in: J. Skalny, J. Gebauer, I. Odler (Eds.), Materials Science of Concrete Special Volume on Calcium Hydroxide in Concrete, American Ceramic Society, Westerville, OH 2001, pp. 269–280., In Materials Science of Concrete Special Volume on Calcium Hydroxide in Concrete. (2001) 269–280.
- S. Urhan, Alkali silica and pozzolanic reactions in concrete. Part 1: interpretation of published results and an hypothesis concerning the mechanism, Cement Concrete Research. 17 (1987) 141–152.
- R.K. Iler, Effect of Adsorbed Alumina on the Solubility of Amorphous Silica in Water, n.d.
- S.-Y. Hong, F.P. S.-Y. Hong, F.P. Glasser, Alkali sorption by C-S-H and C-A-S-H gels Part II. Role of alumina, n.d.
- K. 37 (2007) 1193–1205. [CrossRef]
- T. Chappex, K.L. T. Chappex, K.L. Scrivener, The effect of aluminum in solution on the dissolution of amorphous silica and its relation to cementitious systems, Journal of the American Ceramic Society. 96 (2013) 592–597. [CrossRef]
- T. Chappex, K. T. Chappex, K. Scrivener, Alkali fixation of C-S-H in blended cement pastes and its relation to alkali silica reaction, Cem Concr Res. 42 (2012) 1049–1054. [CrossRef]
- K. 37 (2007) 1193–1205. [CrossRef]
- García Lodeiro, A. Fernández-Jimenez, A. Palomo, D.E. Macphee, Effect on fresh C-S-H gels of the simultaneous addition of alkali and aluminium, Cem Concr Res. 40 (2010) 27–32. [CrossRef]
- MTA, General Directorate of Mineral Research And Exploration, Ankara, Turkey, n.d.
- M M Smedskjaer; M Jensen; Y-Z Yue, Theoretical calculation and measurement of the hardness of diopside, Journal of the American Ceramic Society. [CrossRef]
- L.N. Warr, IMA–CNMNC approved mineral symbols, Mineral Mag. 85 (2021) 291–320. [CrossRef]
- Puchta, R. , A brighter beryllium, Nature Chemistry,Https://Doi.Org/10.1038/Nchem.1033. 3(5), 416. (2011).
- W. A. Deer, R. A. W. A. Deer, R. A. Howie, J. Zussman, An Introduction to the Rock Forming Minerals. ISBN 0-582-44210-9., 1966.
- Hamoudi, L. Khouchaf, C. Depecker, B. Revel, L. Montagne, P. Cordier, Microstructural evolution of amorphous silica following alkali-silica reaction, J Non Cryst Solids. 354 (2008) 5074–5078. [CrossRef]
- M. Imaoka, H. M. Imaoka, H. Hasegawa, I.Yasui,, X-ray diffraction study of the structure of silicate glasses. II. alkali disilicate glasses., Physics and Chemistry of Glasses. (1983).
- J. Serra, P. J. Serra, P. González, S. Liste, C. Serra, S. Chiussi, B. León, M. Pérez-Amor, H.O. Ylänen, M. Hupa, FTIR and XPS studies of bioactive silica based glasses, J Non Cryst Solids. 332 (2003) 20–27. [CrossRef]
- H. Takadama, H.M. H. Takadama, H.M. Kim, T. Kokubo, T. Nakamura, Mechanism of biomineralization of apatite on a sodium silicate glass: TEM-EDX study in vitro, Chemistry of Materials. 13 (2001) 1108–1113. [CrossRef]
- T. Uchino, T. T. Uchino, T. Sakka, K. Hotta, M. Lwasaki, Attenuated Total Reflectance Fourier-Transform Infrared Spectra of a Hydrated Sodium Silicate Glass, 1989.
- F.X., and D.J.Kiemle. Webster, Spectrometric identification of organic compounds.., John Wiley & Sons, 2005.
- S. Ricol, E. S. Ricol, E. Vernaz, P. Barboux, Synthesis of Gels in the System Na20-ZrO2-SiO2, Kluwer Academic Publishers, 1997.
- Y. Ping, R.J. Y. Ping, R.J. Kirkpatrick, P. Brent, P.F. McMillan, X. Cong, Structure of calcium silicate hydrate (C-S-H): Near-, mid-, and far-infrared spectroscopy, Journal of the American Ceramic Society. 82 (1999) 742–748. [CrossRef]
- M. Singh, S. M. Singh, S. Waghmare, S. Vinodh Kumar, Characterization of lime plasters used in 16th century Mughal monument, J Archaeol Sci. 42 (2014) 430–434. [CrossRef]
- R. Choudhary, R. R. Choudhary, R. Gupta, R. Nagar, Impact on fresh, mechanical, and microstructural properties of high strength self-compacting concrete by marble cutting slurry waste, fly ash, and silica fume, Constr Build Mater. 239 (2020). [CrossRef]
- M. Chollet, M. M. Chollet, M. Horgnies, Analyses of the surfaces of concrete by Raman and FT-IR spectroscopies: Comparative study of hardened samples after demoulding and after organic post-treatment, Surface and Interface Analysis. 43 (2011) 714–725. [CrossRef]
- M. Horgnies, P. M. Horgnies, P. Willieme, O. Gabet, Influence of the surface properties of concrete on the adhesion of coating: Characterization of the interface by peel test and FT-IR spectroscopy, Prog Org Coat. 72 (2011) 360–379. [CrossRef]
- P.A. Bhat, N.C. P.A. Bhat, N.C. Debnath, Theoretical and experimental study of structures and properties of cement paste: The nanostructural aspects of CSH, Journal of Physics and Chemistry of Solids. 72 (2011) 920–933. [CrossRef]
- M., Horgnies, J. J., Chen, C. Bouillon, Overview about the use of Fourier transform infrared spectroscopy to study cementitious materials., 77th ed., WIT Trans. Eng. Sci, 2013.
- Z. P Bažant, M. F. Z. P Bažant, M. F. Kaplan, Z.P.Bazant, Concrete at high temperatures: material properties and mathematical models., 1996.
- Noumowe, Effet de hautes températures (20-600 C) sur le béton: cas particulier du béton a hautes performances (Doctoral dissertation,, INSA)., 1995.
- G. Platret, Suivi de l’hydratation du ciment et de l’évolution des phases solides dans les bétons par analyse thermique, caractéristiques microstructurales et propriétés relativesa la durabilité des bétons., 2002.
- B. Zhang, Effects of moisture evaporation (weight loss) on fracture properties of high performance concrete subjected to high temperatures, Fire Saf J. 46 (2011) 543–549. [CrossRef]
- Savva, P. Manita, K.K. Sideris, Influence of elevated temperatures on the mechanical properties of blended cement concretes prepared with limestone and siliceous aggregates, Cem Concr Compos. 27 (2005) 239–248. [CrossRef]
- J. Moon, S. J. Moon, S. Bae, K. Celik, S. Yoon, K.H. Kim, K.S. Kim, P.J.M. Monteiro, Characterization of natural pozzolan-based geopolymeric binders, Cem Concr Compos. 53 (2014) 97–104. [CrossRef]
- K. Kupwade-Patil, S.D. K. Kupwade-Patil, S.D. Palkovic, A. Bumajdad, C. Soriano, O. Büyüköztürk, Use of silica fume and natural volcanic ash as a replacement to Portland cement: Micro and pore structural investigation using NMR, XRD, FTIR and X-ray microtomography, Constr Build Mater. 158 (2018) 574–590. [CrossRef]
- K. Annaba, Y. K. Annaba, Y. El Mendili, H. Stout, A. Ech-chebab, B. Ouaki, M. Cherkaoui, C. Florence, Mechanical, electrochemical (EIS), and microstructural characterization of reinforced concrete incorporating natural volcanic pozzolan, Case Studies in Construction Materials. 19 (2023). [CrossRef]
- Karaşin, M. Doğruyol, An experimental study on strength and durability for utilization of fly ash in concrete mix, Advances in Materials Science and Engineering. 2014 ( 2014. [CrossRef]
- 2023 Kahramanmaraş depremleri- Vikipedi, (n.d.). https://tr.wikipedia.org/wiki/2023_Kahramanmara%C5%9F_depremleri (accessed , 2023)., (2023). 3 December.




| SiO2 | Al2O3 | Fe2O3 | CaO | CaCO3 | MgO | SO3 | Na2O | K2O | LOI1 | Na2Oeq* | |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Cement | 18.12 | 5.21 | 3.03 | 62.06 | - | 2.70 | 3.21 | 0.09 | 0.96 | 3.98 | 0.72 |
| FA | 21.65 | 33.55 | 8.76 | 27.50 | 4.50 | 0.05 | 0.84 | 1.76 | 1.29 | 2.00 | |
| Basalt | 50.63 | 10.51 | 6.49 | - | 21.55 | 3.32 | - | 0.45 | 1.63 | 2.08 | 2.08 |
| 1LOI: Loss on ignition; Na2Oeq* = Na2O + 0.658 K2O | |||||||||||
| 3CaO.SiO2(C3S) | 2CaO.SiO2(C2S) | 3CaO.Al2O3(C3A) | 4CaO.Al2O3. Fe2O3 (C4AF) |
| 66.40 | 1.87 | 8.68 | 9.22 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




