Submitted:
30 December 2023
Posted:
04 January 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Microtubule cytoskeleton
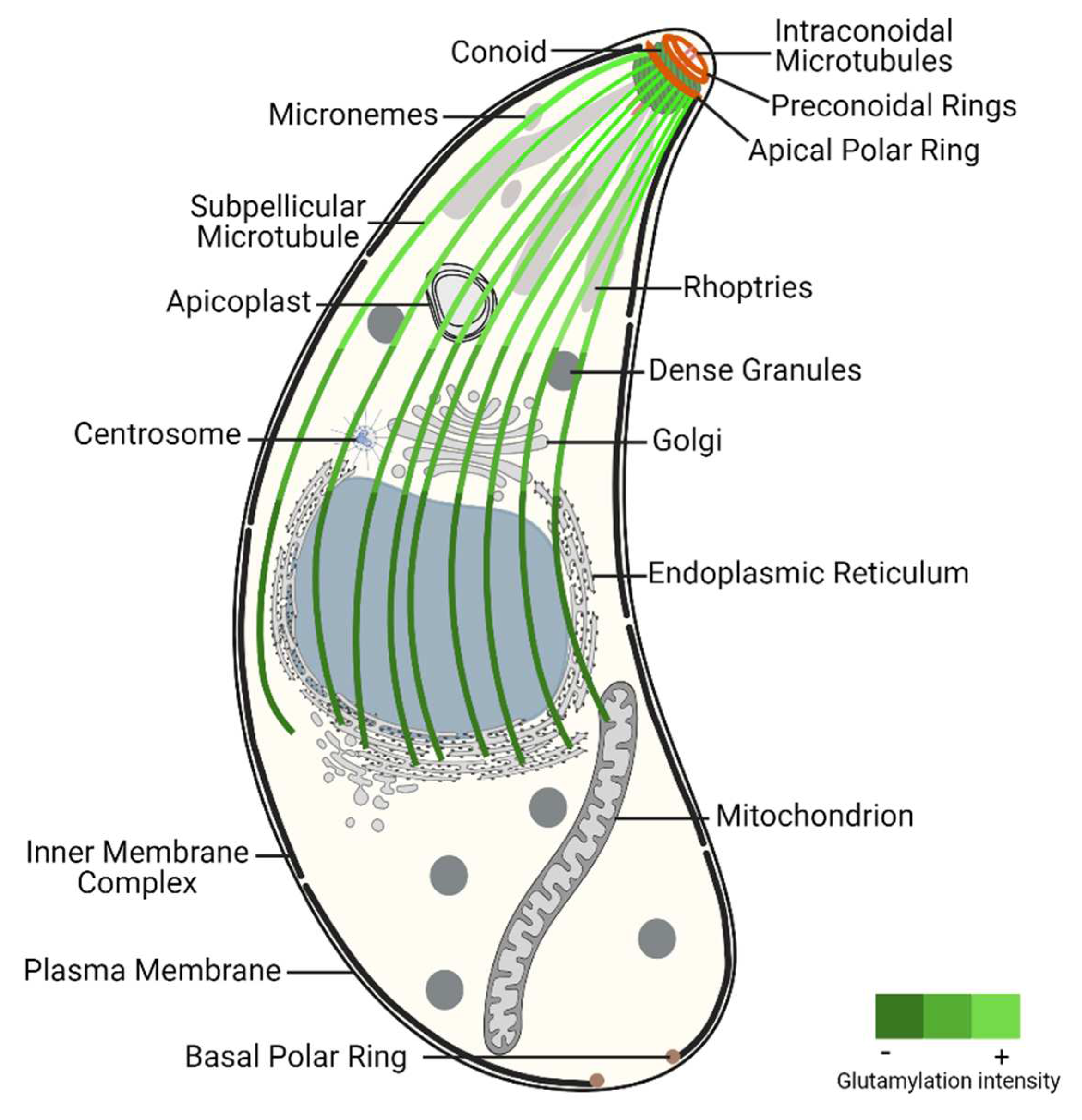
3. The specialized microtubule structures of Toxoplasma gondii
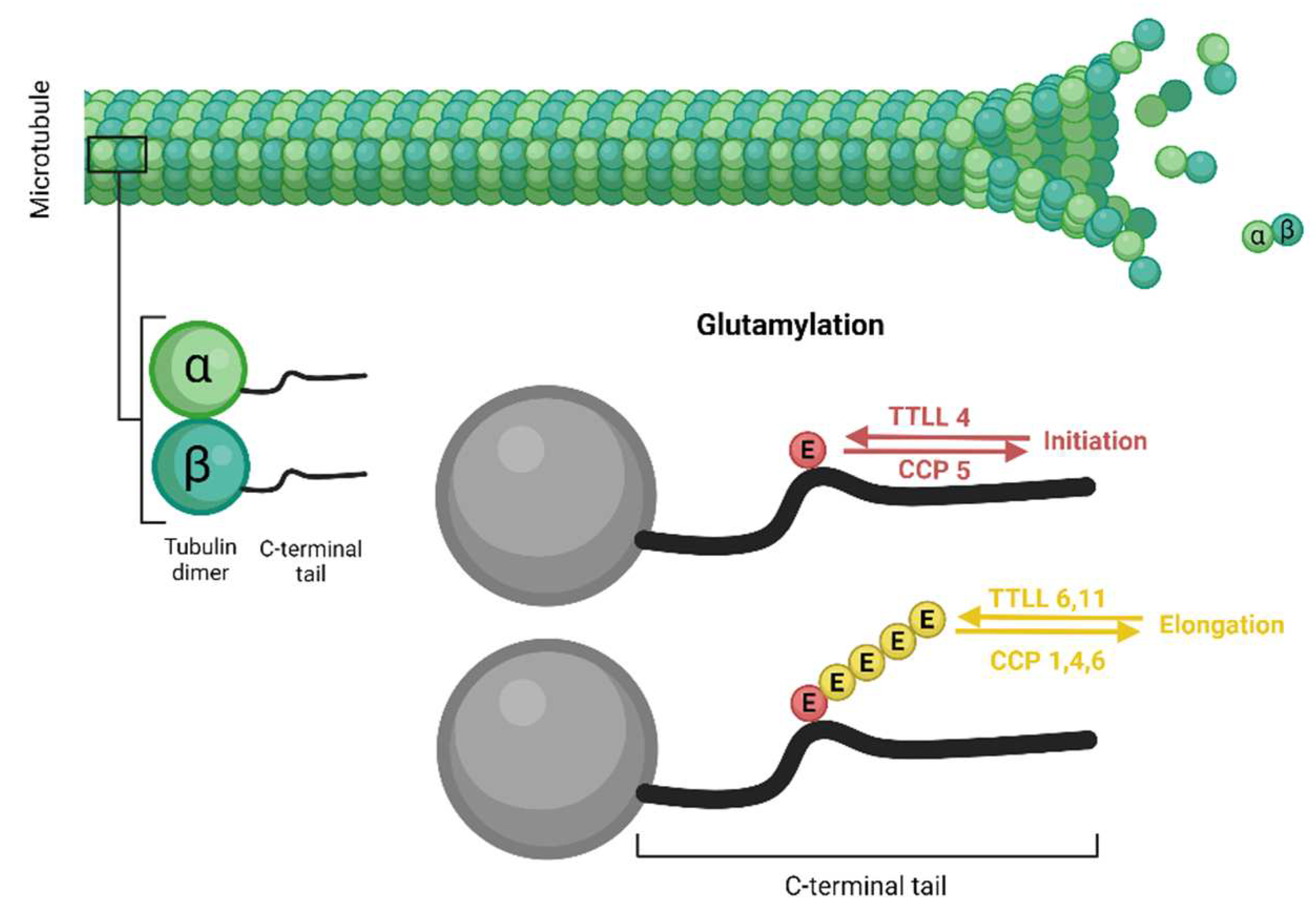
4. T. gondii tubulin post-translational modifications
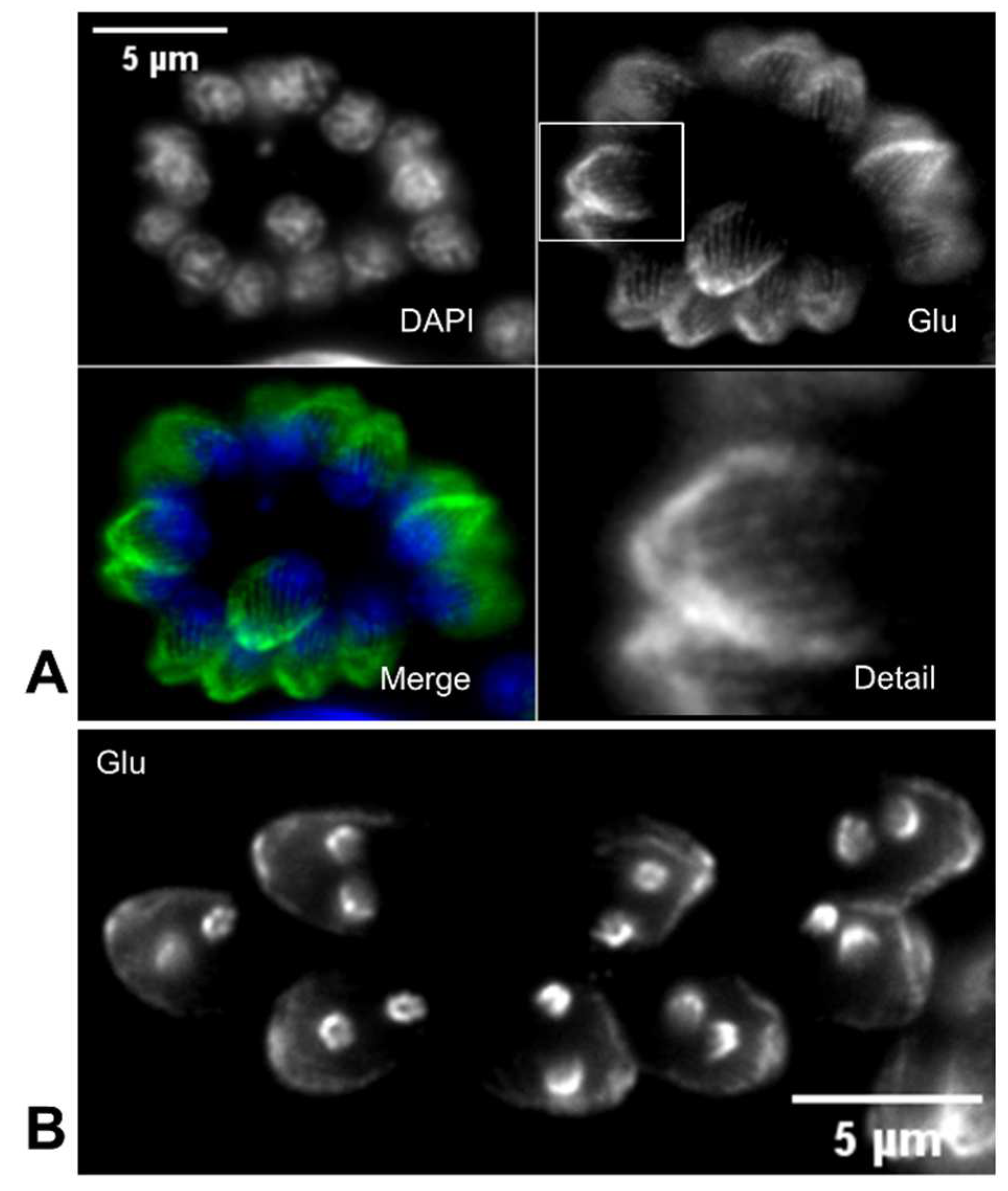
5. T. gondii tubulin glutamylation
6. Concluding remarks
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Levine, N.D. Progress in Taxonomy of the Apicomplexan Protozoa. J. Protozool. 1988, 35, 518–20. [CrossRef]
- [WHO] World Malaria Report 2020: 20 Years of Global Progress and Challenges; Geneva, 2020.
- Blake, D.P.; Tomley, F.M. Securing Poultry Production from the Ever-Present Eimeria Challenge. Trends Parasitol. 2014, 30, 12–19. [CrossRef]
- Chartier, C.; Paraud, C. Coccidiosis Due to Eimeria in Sheep and Goats, a Review. Small Rumin. Res. 2012, 103, 84–92. [CrossRef]
- Tzipori, S.; Ward, H. Cryptosporidiosis: Biology, Pathogenesis and Disease. Microbes Infect. 2002, 4, 1047–1058. [CrossRef]
- Álvarez-García, G.; García-Lunar, P.; Gutiérrez-Expósito, D.; Shkap, V.; Ortega-Mora, L.M. Dynamics of Besnoitia Besnoiti Infection in Cattle. Parasitology 2014, 141, 1419–1435. [CrossRef]
- Jacob, S.S.; Sengupta, P.P.; Paramanandham, K.; Suresh, K.P.; Chamuah, J.K.; Rudramurthy, G.R.; Roy, P. Bovine Babesiosis: An Insight into the Global Perspective on the Disease Distribution by Systematic Review and Meta-Analysis. Vet. Parasitol. 2020, 283, 109136. [CrossRef]
- Mans, B.J.; Pienaar, R.; Latif, A.A. A Review of Theileria Diagnostics and Epidemiology. Int. J. Parasitol. Parasites Wildl. 2015, 4, 104–118. [CrossRef]
- Delgado, I.L.S.; Zúquete, S.; Santos, D.; Basto, A.P.; Leitão, A.; Nolasco, S. The Apicomplexan Parasite Toxoplasma Gondii. Encyclopedia 2022, 2, 189–211. [CrossRef]
- Khan, K.; Khan, W. Congenital Toxoplasmosis: An Overview of the Neurological and Ocular Manifestations. Parasitol. Int. 2018, 67, 715–721. [CrossRef]
- Wang, Z.-D.; Liu, H.-H.; Ma, Z.-X.; Ma, H.-Y.; Li, Z.-Y.; Yang, Z.-B.; Zhu, X.-Q.; Xu, B.; Wei, F.; Liu, Q. Toxoplasma Gondii Infection in Immunocompromised Patients: A Systematic Review and Meta-Analysis. Front. Microbiol. 2017, 8. [CrossRef]
- Slifko, T.R.; Smith, H. V; Rose, J.B. Emerging Parasite Zoonoses Associated with Water and Food. Int. J. Parasitol. 2000, 30, 1379–1393. [CrossRef]
- Frénal, K.; Dubremetz, J.-F.; Lebrun, M.; Soldati-Favre, D. Gliding Motility Powers Invasion and Egress in Apicomplexa. Nat. Rev. Microbiol. 2017, 15, 645–660. [CrossRef]
- Hu, K.; Johnson, J.; Florens, L.; Fraunholz, M.; Suravajjala, S.; DiLullo, C.; Yates, J.; Roos, D.S.; Murray, J.M. Cytoskeletal Components of an Invasion Machine—The Apical Complex of Toxoplasma Gondii. PLoS Pathog. 2006, 2, e13. [CrossRef]
- Findeisen, P.; Mühlhausen, S.; Dempewolf, S.; Hertzog, J.; Zietlow, A.; Carlomagno, T.; Kollmar, M. Six Subgroups and Extensive Recent Duplications Characterize the Evolution of the Eukaryotic Tubulin Protein Family. Genome Biol. Evol. 2014, 6, 2274–88. [CrossRef]
- Jaglin, X.H.; Poirier, K.; Saillour, Y.; Buhler, E.; Tian, G.; Bahi-Buisson, N.; Fallet-Bianco, C.; Phan-Dinh-Tuy, F.; Kong, X.P.; Bomont, P.; et al. Mutations in the Beta-Tubulin Gene TUBB2B Result in Asymmetrical Polymicrogyria. Nat. Genet. 2009, 41, 746–52. [CrossRef]
- Tischfield, M.A.; Baris, H.N.; Wu, C.; Rudolph, G.; Van Maldergem, L.; He, W.; Chan, W.-M.; Andrews, C.; Demer, J.L.; Robertson, R.L.; et al. Human TUBB3 Mutations Perturb Microtubule Dynamics, Kinesin Interactions, and Axon Guidance. Cell 2010, 140, 74–87. [CrossRef]
- Tischfield, M.A.; Engle, E.C. Distinct Alpha- and Beta-Tubulin Isotypes Are Required for the Positioning, Differentiation and Survival of Neurons: New Support for the “multi-Tubulin” Hypothesis. Biosci. Rep. 2010, 30, 319–30. [CrossRef]
- Neff, N.F.; Thomas, J.H.; Grisafi, P.; Botstein, D. Isolation of the Beta-Tubulin Gene from Yeast and Demonstration of Its Essential Function in Vivo. Cell 1983, 33, 211–9. [CrossRef]
- Schatz, P.J.; Pillus, L.; Grisafi, P.; Solomon, F.; Botstein, D. Two Functional Alpha-Tubulin Genes of the Yeast Saccharomyces Cerevisiae Encode Divergent Proteins. Mol. Cell. Biol. 1986, 6, 3711–21. [CrossRef]
- Barahona, I.; Soares, H.; Cyrne, L.; Penque, D.; Denoulet, P.; Rodrigues-Pousada, C. Sequence of One Alpha- and Two Beta-Tubulin Genes of Tetrahymena Pyriformis. Structural and Functional Relationships with Other Eukaryotic Tubulin Genes. J. Mol. Biol. 1988, 202, 365–82. [CrossRef]
- Gaertig, J.; Thatcher, T.H.; McGrath, K.E.; Callahan, R.C.; Gorovsky, M.A. Perspectives on Tubulin Isotype Function and Evolution Based on the Observation That Tetrahymena Thermophila Microtubules Contain a Single Alpha- and Beta-Tubulin. Cell Motil. Cytoskeleton 1993, 25, 243–53. [CrossRef]
- Vainberg, I.E.; Lewis, S.A.; Rommelaere, H.; Ampe, C.; Vandekerckhove, J.; Klein, H.L.; Cowan, N.J. Prefoldin, a Chaperone That Delivers Unfolded Proteins to Cytosolic Chaperonin. Cell 1998, 93, 863–73. [CrossRef]
- Hansen, W.J.; Cowan, N.J.; Welch, W.J. Prefoldin-Nascent Chain Complexes in the Folding of Cytoskeletal Proteins. J. Cell Biol. 1999, 145, 265–77. [CrossRef]
- Gestaut, D.; Zhao, Y.; Park, J.; Ma, B.; Leitner, A.; Collier, M.; Pintilie, G.; Roh, S.-H.; Chiu, W.; Frydman, J. Structural Visualization of the Tubulin Folding Pathway Directed by Human Chaperonin TRiC/CCT. Cell 2022, 185, 4770-4787.e20. [CrossRef]
- Liu, C.; Jin, M.; Wang, S.; Han, W.; Zhao, Q.; Wang, Y.; Xu, C.; Diao, L.; Yin, Y.; Peng, C.; et al. Pathway and Mechanism of Tubulin Folding Mediated by TRiC/CCT along Its ATPase Cycle Revealed Using Cryo-EM. Commun. Biol. 2023, 6, 531. [CrossRef]
- Serna, M.; Zabala, J.C. Tubulin Folding and Degradation. In Encyclopedia of Life Sciences; Wiley, 2016; pp. 1–9.
- Gonçalves, J.; Tavares, A.; Carvalhal, S.; Soares, H. Revisiting the Tubulin Folding Pathway: New Roles in Centrosomes and Cilia. Biomol. Concepts 2010, 1, 423–34. [CrossRef]
- Nolasco, S.; Bellido, J.; Serna, M.; Carmona, B.; Soares, H.; Zabala, J.C. Colchicine Blocks Tubulin Heterodimer Recycling by Tubulin Cofactors TBCA, TBCB, and TBCE. Front. cell Dev. Biol. 2021, 9, 656273. [CrossRef]
- Francis, J.W.; Goswami, D.; Novick, S.J.; Pascal, B.D.; Weikum, E.R.; Ortlund, E.A.; Griffin, P.R.; Kahn, R.A. Nucleotide Binding to ARL2 in the TBCD∙ARL2∙β-Tubulin Complex Drives Conformational Changes in β-Tubulin. J. Mol. Biol. 2017, 429, 3696–3716. [CrossRef]
- Francis, J.W.; Newman, L.E.; Cunningham, L.A.; Kahn, R.A. A Trimer Consisting of the Tubulin-Specific Chaperone D (TBCD), Regulatory GTPase ARL2, and β-Tubulin Is Required for Maintaining the Microtubule Network. J. Biol. Chem. 2017, 292, 4336–4349. [CrossRef]
- Spiegelman, B.M.; Penningroth, S.M.; Kirschner, M.W. Turnover of Tubulin and the N Site GTP in Chinese Hamster Ovary Cells. Cell 1977, 12, 587–600. [CrossRef]
- Kristensson, M.A. The Game of Tubulins. Cells 2021, 10. [CrossRef]
- Chabin-Brion, K.; Marceiller, J.; Perez, F.; Settegrana, C.; Drechou, A.; Durand, G.; Poüs, C. The Golgi Complex Is a Microtubule-Organizing Organelle. Mol. Biol. Cell 2001, 12, 2047–60. [CrossRef]
- Yukawa, M.; Ikebe, C.; Toda, T. The Msd1-Wdr8-Pkl1 Complex Anchors Microtubule Minus Ends to Fission Yeast Spindle Pole Bodies. J. Cell Biol. 2015, 209, 549–62. [CrossRef]
- Sanchez, A.D.; Feldman, J.L. Microtubule-Organizing Centers: From the Centrosome to Non-Centrosomal Sites. Curr. Opin. Cell Biol. 2017, 44, 93–101. [CrossRef]
- Jaspersen, S.L. Anatomy of the Fungal Microtubule Organizing Center, the Spindle Pole Body. Curr. Opin. Struct. Biol. 2021, 66, 22–31. [CrossRef]
- Wloga, D.; Gaertig, J. Post-Translational Modifications of Microtubules. J. Cell Sci. 2010, 123, 3447–55. [CrossRef]
- Janke, C.; Montagnac, G. Causes and Consequences of Microtubule Acetylation. Curr. Biol. 2017, 27, R1287–R1292. [CrossRef]
- Xiao, H.; El Bissati, K.; Verdier-Pinard, P.; Burd, B.; Zhang, H.; Kim, K.; Fiser, A.; Angeletti, R.H.; Weiss, L.M. Post-Translational Modifications to Toxoplasma Gondii Alpha- and Beta-Tubulins Include Novel C-Terminal Methylation. J. Proteome Res. 2010, 9, 359–72. [CrossRef]
- Kimura, Y.; Tsutsumi, K.; Konno, A.; Ikegami, K.; Hameed, S.; Kaneko, T.; Kaplan, O.I.; Teramoto, T.; Fujiwara, M.; Ishihara, T.; et al. Environmental Responsiveness of Tubulin Glutamylation in Sensory Cilia Is Regulated by the P38 MAPK Pathway. Sci. Rep. 2018, 8, 8392. [CrossRef]
- Power, K.M.; Akella, J.S.; Gu, A.; Walsh, J.D.; Bellotti, S.; Morash, M.; Zhang, W.; Ramadan, Y.H.; Ross, N.; Golden, A.; et al. Mutation of NEKL-4/NEK10 and TTLL Genes Suppress Neuronal Ciliary Degeneration Caused by Loss of CCPP-1 Deglutamylase Function. PLoS Genet. 2020, 16, e1009052. [CrossRef]
- Leander, B.S.; Keeling, P.J. Morphostasis in Alveolate Evolution. Trends Ecol. Evol. 2003, 18, 395–402. [CrossRef]
- Leander, B.S.; Kuvardina, O.N.; Aleshin, V. V; Mylnikov, A.P.; Keeling, P.J. Molecular Phylogeny and Surface Morphology of Colpodella Edax (Alveolata): Insights into the Phagotrophic Ancestry of Apicomplexans. J. Eukaryot. Microbiol. 2003, 50, 334–40. [CrossRef]
- Goodenough, U.; Roth, R.; Kariyawasam, T.; He, A.; Lee, J.-H. Epiplasts: Membrane Skeletons and Epiplastin Proteins in Euglenids, Glaucophytes, Cryptophytes, Ciliates, Dinoflagellates, and Apicomplexans. MBio 2018, 9. [CrossRef]
- Suvorova, E.S.; Francia, M.; Striepen, B.; White, M.W. A Novel Bipartite Centrosome Coordinates the Apicomplexan Cell Cycle. PLoS Biol. 2015, 13, e1002093. [CrossRef]
- Leung, J.M.; He, Y.; Zhang, F.; Hwang, Y.-C.; Nagayasu, E.; Liu, J.; Murray, J.M.; Hu, K. Stability and Function of a Putative Microtubule-Organizing Center in the Human Parasite Toxoplasma Gondii. Mol. Biol. Cell 2017, 28, 1361–1378. [CrossRef]
- Russell, D.G.; Burns, R.G. The Polar Ring of Coccidian Sporozoites: A Unique Microtubule-Organizing Centre. J. Cell Sci. 1984, 65, 193–207. [CrossRef]
- Tran, J.Q.; de Leon, J.C.; Li, C.; Huynh, M.-H.; Beatty, W.; Morrissette, N.S. RNG1 Is a Late Marker of the Apical Polar Ring in Toxoplasma Gondii. Cytoskeleton (Hoboken). 2010, 67, 586–98. [CrossRef]
- Gould, S.B.; Kraft, L.G.K.; van Dooren, G.G.; Goodman, C.D.; Ford, K.L.; Cassin, A.M.; Bacic, A.; McFadden, G.I.; Waller, R.F. Ciliate Pellicular Proteome Identifies Novel Protein Families with Characteristic Repeat Motifs That Are Common to Alveolates. Mol. Biol. Evol. 2011, 28, 1319–31. [CrossRef]
- Katris, N.J.; van Dooren, G.G.; McMillan, P.J.; Hanssen, E.; Tilley, L.; Waller, R.F. The Apical Complex Provides a Regulated Gateway for Secretion of Invasion Factors in Toxoplasma. PLoS Pathog. 2014, 10, e1004074. [CrossRef]
- Morrissette, N.S.; Sibley, L.D. Cytoskeleton of Apicomplexan Parasites. Microbiol. Mol. Biol. Rev. 2002, 66, 21–38; table of contents. [CrossRef]
- Morrissette, N.S.; Murray, J.M.; Roos, D.S. Subpellicular Microtubules Associate with an Intramembranous Particle Lattice in the Protozoan Parasite Toxoplasma Gondii. J. Cell Sci. 1997, 110 ( Pt 1), 35–42. [CrossRef]
- Tran, J.Q.; Li, C.; Chyan, A.; Chung, L.; Morrissette, N.S. SPM1 Stabilizes Subpellicular Microtubules in Toxoplasma Gondii. Eukaryot. Cell 2012, 11, 206–216. [CrossRef]
- Mondragon, R.; Frixione, E. Ca(2+)-Dependence of Conoid Extrusion in Toxoplasma Gondii Tachyzoites. J. Eukaryot. Microbiol. 1996, 43, 120–7. [CrossRef]
- Hu, K.; Roos, D.S.; Murray, J.M. A Novel Polymer of Tubulin Forms the Conoid of Toxoplasma Gondii. J. Cell Biol. 2002, 156, 1039–50. [CrossRef]
- Nichols, B.A.; Chiappino, M.L. Cytoskeleton of Toxoplasma Gondii. J. Protozool. 1987, 34, 217–26. [CrossRef]
- Morrissette, N.; Gubbels, M.-J. The Toxoplasma Cytoskeleton: Structures, Proteins, and Processes. Toxoplasma gondii 2020, 743–788. [CrossRef]
- Mageswaran, S.K.; Guérin, A.; Theveny, L.M.; Chen, W.D.; Martinez, M.; Lebrun, M.; Striepen, B.; Chang, Y.-W. In Situ Ultrastructures of Two Evolutionarily Distant Apicomplexan Rhoptry Secretion Systems. Nat. Commun. 2021, 12, 4983. [CrossRef]
- Francia, M.E.; Dubremetz, J.-F.; Morrissette, N.S. Basal Body Structure and Composition in the Apicomplexans Toxoplasma and Plasmodium. Cilia 2015, 5, 3. [CrossRef]
- Naumov, A.; Kratzer, S.; Ting, L.-M.; Kim, K.; Suvorova, E.S.; White, M.W. The Toxoplasma Centrocone Houses Cell Cycle Regulatory Factors. MBio 2017, 8. [CrossRef]
- Farrell, M.; Gubbels, M.-J. The Toxoplasma Gondii Kinetochore Is Required for Centrosome Association with the Centrocone (Spindle Pole). Cell. Microbiol. 2014, 16, 78–94. [CrossRef]
- Chen, C.-T.; Kelly, M.; Leon, J. de; Nwagbara, B.; Ebbert, P.; Ferguson, D.J.P.; Lowery, L.A.; Morrissette, N.; Gubbels, M.-J. Compartmentalized Toxoplasma EB1 Bundles Spindle Microtubules to Secure Accurate Chromosome Segregation. Mol. Biol. Cell 2015, 26, 4562–76. [CrossRef]
- Hu, K. Organizational Changes of the Daughter Basal Complex during the Parasite Replication of Toxoplasma Gondii. PLoS Pathog. 2008, 4, e10. [CrossRef]
- Gubbels, M.-J.; White, M.; Szatanek, T. The Cell Cycle and Toxoplasma Gondii Cell Division: Tightly Knit or Loosely Stitched? Int. J. Parasitol. 2008, 38, 1343–58. [CrossRef]
- Blader, I.J.; Coleman, B.I.; Chen, C.-T.; Gubbels, M.-J. Lytic Cycle of Toxoplasma Gondii: 15 Years Later. Annu. Rev. Microbiol. 2015, 69, 463–85. [CrossRef]
- Nagel, S.D.; Boothroyd, J.C. The Alpha- and Beta-Tubulins of Toxoplasma Gondii Are Encoded by Single Copy Genes Containing Multiple Introns. Mol. Biochem. Parasitol. 1988, 29, 261–73. [CrossRef]
- Morrissette, N. Targeting Toxoplasma Tubules: Tubulin, Microtubules, and Associated Proteins in a Human Pathogen. Eukaryot. Cell 2015, 14, 2–12. [CrossRef]
- Morrissette, N.; Abbaali, I.; Ramakrishnan, C.; Hehl, A.B. The Tubulin Superfamily in Apicomplexan Parasites. Microorganisms 2023, 11, 706. [CrossRef]
- Sidik, S.M.; Huet, D.; Ganesan, S.M.; Huynh, M.-H.; Wang, T.; Nasamu, A.S.; Thiru, P.; Saeij, J.P.J.; Carruthers, V.B.; Niles, J.C.; et al. A Genome-Wide CRISPR Screen in Toxoplasma Identifies Essential Apicomplexan Genes. Cell 2016, 166, 1423-1435.e12. [CrossRef]
- Nogales, E.; Wolf, S.G.; Downing, K.H. Structure of the Alpha Beta Tubulin Dimer by Electron Crystallography. Nature 1998, 391, 199–203. [CrossRef]
- Wang, Z.; Sheetz, M.P. The C-Terminus of Tubulin Increases Cytoplasmic Dynein and Kinesin Processivity. Biophys. J. 2000, 78, 1955–64. [CrossRef]
- Varberg, J.M.; Padgett, L.R.; Arrizabalaga, G.; Sullivan, W.J. TgATAT-Mediated α-Tubulin Acetylation Is Required for Division of the Protozoan Parasite Toxoplasma Gondii. mSphere 2016, 1. [CrossRef]
- Spiliotis, E.T.; Hunt, S.J.; Hu, Q.; Kinoshita, M.; Nelson, W.J. Epithelial Polarity Requires Septin Coupling of Vesicle Transport to Polyglutamylated Microtubules. J. Cell Biol. 2008, 180, 295–303. [CrossRef]
- Plessmann, U.; Reiter-Owona, I.; Lechtreck, K.-F. Posttranslational Modifications of Alpha-Tubulin of Toxoplasma Gondii. Parasitol. Res. 2004, 94, 386–9. [CrossRef]
- Tosetti, N.; Dos Santos Pacheco, N.; Bertiaux, E.; Maco, B.; Bournonville, L.; Hamel, V.; Guichard, P.; Soldati-Favre, D. Essential Function of the Alveolin Network in the Subpellicular Microtubules and Conoid Assembly in Toxoplasma Gondii. Elife 2020, 9. [CrossRef]
- de Leon, J.C.; Scheumann, N.; Beatty, W.; Beck, J.R.; Tran, J.Q.; Yau, C.; Bradley, P.J.; Gull, K.; Wickstead, B.; Morrissette, N.S. A SAS-6-like Protein Suggests That the Toxoplasma Conoid Complex Evolved from Flagellar Components. Eukaryot. Cell 2013, 12, 1009–19. [CrossRef]
- Kubo, T.; Yanagisawa, H.; Yagi, T.; Hirono, M.; Kamiya, R. Tubulin Polyglutamylation Regulates Axonemal Motility by Modulating Activities of Inner-Arm Dyneins. Curr. Biol. 2010, 20, 441–5. [CrossRef]
- Suryavanshi, S.; Eddé, B.; Fox, L.A.; Guerrero, S.; Hard, R.; Hennessey, T.; Kabi, A.; Malison, D.; Pennock, D.; Sale, W.S.; et al. Tubulin Glutamylation Regulates Ciliary Motility by Altering Inner Dynein Arm Activity. Curr. Biol. 2010, 20, 435–40. [CrossRef]
- Lacroix, B.; van Dijk, J.; Gold, N.D.; Guizetti, J.; Aldrian-Herrada, G.; Rogowski, K.; Gerlich, D.W.; Janke, C. Tubulin Polyglutamylation Stimulates Spastin-Mediated Microtubule Severing. J. Cell Biol. 2010, 189, 945–54. [CrossRef]
- Rogowski, K.; van Dijk, J.; Magiera, M.M.; Bosc, C.; Deloulme, J.-C.; Bosson, A.; Peris, L.; Gold, N.D.; Lacroix, B.; Bosch Grau, M.; et al. A Family of Protein-Deglutamylating Enzymes Associated with Neurodegeneration. Cell 2010, 143, 564–78. [CrossRef]
- van Dijk, J.; Rogowski, K.; Miro, J.; Lacroix, B.; Eddé, B.; Janke, C. A Targeted Multienzyme Mechanism for Selective Microtubule Polyglutamylation. Mol. Cell 2007, 26, 437–48. [CrossRef]
- Berezniuk, I.; Vu, H.T.; Lyons, P.J.; Sironi, J.J.; Xiao, H.; Burd, B.; Setou, M.; Angeletti, R.H.; Ikegami, K.; Fricker, L.D. Cytosolic Carboxypeptidase 1 Is Involved in Processing α- and β-Tubulin. J. Biol. Chem. 2012, 287, 6503–17. [CrossRef]
- Kimura, Y.; Kurabe, N.; Ikegami, K.; Tsutsumi, K.; Konishi, Y.; Kaplan, O.I.; Kunitomo, H.; Iino, Y.; Blacque, O.E.; Setou, M. Identification of Tubulin Deglutamylase among Caenorhabditis Elegans and Mammalian Cytosolic Carboxypeptidases (CCPs). J. Biol. Chem. 2010, 285, 22936–41. [CrossRef]
- Xia, P.; Ye, B.; Wang, S.; Zhu, X.; Du, Y.; Xiong, Z.; Tian, Y.; Fan, Z. Glutamylation of the DNA Sensor CGAS Regulates Its Binding and Synthase Activity in Antiviral Immunity. Nat. Immunol. 2016, 17, 369–378. [CrossRef]
- Ye, B.; Li, C.; Yang, Z.; Wang, Y.; Hao, J.; Wang, L.; Li, Y.; Du, Y.; Hao, L.; Liu, B.; et al. Cytosolic Carboxypeptidase CCP6 Is Required for Megakaryopoiesis by Modulating Mad2 Polyglutamylation. J. Exp. Med. 2014, 211, 2439–2454. [CrossRef]
- Janke, C.; Rogowski, K.; Wloga, D.; Regnard, C.; Kajava, A. V; Strub, J.-M.; Temurak, N.; van Dijk, J.; Boucher, D.; van Dorsselaer, A.; et al. Tubulin Polyglutamylase Enzymes Are Members of the TTL Domain Protein Family. Science 2005, 308, 1758–62. [CrossRef]
- Sharma, N.; Bryant, J.; Wloga, D.; Donaldson, R.; Davis, R.C.; Jerka-Dziadosz, M.; Gaertig, J. Katanin Regulates Dynamics of Microtubules and Biogenesis of Motile Cilia. J. Cell Biol. 2007, 178, 1065–79. [CrossRef]
- Roll-Mecak, A.; McNally, F.J. Microtubule-Severing Enzymes. Curr. Opin. Cell Biol. 2010, 22, 96–103. [CrossRef]
| ToxoDB ID | Ortholog | Fitness score1 | Protein evidence2 | |
|---|---|---|---|---|
| Designation | Uniprot reference | |||
| TGME49_278930 | TTLL1 | O95922 | non-essential | no |
| TGME49_228410 | TTLL2 | Q9BWV7 | non-essential | no |
| TGME49_230670 | TTLL6 | Q8N841 | essential | no |
| TGME49_244500 | TTLL11 | Q8NHH1 | essential | yes |
| TGME49_307760 | TTLL11 | Q8NHH1 | non-essential | yes |
| TGME49_265780 | CCP1 | Q9UPW5 | essential | yes |
| TGME49_315680 | Spastin | Q9UBP0 | essential | yes |
| TGME49_244590 | Katanin p60 subunit | O75449 | non-essential | no |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




