Submitted:
01 January 2024
Posted:
03 January 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
2.1. Influence of Irrigation Regimes and Microbial Inoculants on Soil Chemical Properties
| pH | EC (mS cm-1) | P Olsen (mg kg-1) | TN (%) | OC (%) | |||
| Pre-Plant | 7.55 ± 0.20 | 4.71 ± 0.04 | 42.84 ± 0.61 | 0.32 ± 0.01 | 3.11 ± 0.05 | ||
| Atriplex halimus | Non-inoculated | Control | 6.72 ± 0.04a | 4.35 ± 0.02b | 40.86 ± 3.63b | 0.51 ± 0.02a | 4.70 ± 0.02a |
| Moderate stress | 6.62 ± 0.05b | 4.34 ± 0.01b | 40.17 ± 5.35b | 0.42 ± 0.03b | 4.68 ± 0.11a | ||
| Severe stress | 6.75 ± 0.03a | 4.42 ± 0.02a | 52.68 ± 3.06a | 0.38 ± 0.04b | 3.68 ± 0.09b | ||
| **F=8.055 | **F=13.796 | *F=8.604 | *F=13.069 | ***F=134.088 | |||
| Inoculated | Control | 6.57 ± 0.02a | 4.21 ± 0.04b | 51.30 ± 5.31a | 0.54 ± 0.01a | 4.71 ± 0.04a | |
| Moderate stress | 6.54 ± 0.05a | 4.32 ± 0.05a | 55.30 ± 5.85a | 0.53 ± 0.04a | 4.37 ± 0.58a | ||
| Severe stress | 6.51 ± 0.02a | 3.90 ± 0.04c | 55.31 ± 6.09a | 0.38 ± 0.43b | 4.68 ± 0.11a | ||
| NSF=2.167 | ***F=65.068 | NSF=0.483 | *F=15.932 | NSF=1.503 | |||
| ***F(I)=72.532 | ***F(I)=180.549 | **F(I)=15.793 | **F(I)=7.506 | NSF(I)=0.015 | |||
| *F(R)=5.277 | ***F(R)=36.093 | *F(R)=4.152 | ***F(R)=25.434 | *F(R)=10.111 | |||
| *F(IxR)=6.004 | ***F(IxR)=78.615 | NSF(IxR)=2.370 | *F(IxR)=3.945 | NSF(IxR)=1.393 | |||
| pH | EC (mS cm-1) | P Olsen (mg kg-1) | TN (%) | OC (%) | |||
| Penisetum setaceum | Non-inoculated | Control | 6.66 ± 0.05a | 4.21 ± 0.04b | 47.97 ± 1.35a | 0.42 ± 0.02a | 3.64 ± 0.02a |
| Moderate stress | 6.72 ± 0.03a | 4.32 ± 0.05a | 48.63 ± 0.15a | 0.45 ± 0.07a | 3.60 ± 0.03a | ||
| Severe stress | 6.72 ± 0.04a | 3.90 ± 0.04c | 45.31 ± 2.89a | 0.34 ± 0.04b | 3.71 ± 0.04a | ||
| NSF=1.596 | ***F=65.068 | NSF=2.707 | *F=8.179 | NSF=3.201 | |||
| Inoculated | Control | 7.25 ± 0.01a | 4.32 ± 0.02b | 60.86 ± 4.38a | 0.50 ± 0.07b,c | 4.20 ± 0.02b | |
| Moderate stress | 6.51 ± 0.05b | 4.34 ± 0.04b | 63.51 ± 1.35a | 0.54 ± 0.02b | 4.20 ± 0.02b | ||
| Severe stress | 6.46 ± 0.30b | 4.42 ± 0.02a | 41.68 ± 3.50b | 0.56 ± 0.03a,b | 4.36 ± 0.03a | ||
| *F=18.268 | **F=13.794 | ***F=39.607 | *F=6.581 | *F=8.934 | |||
| NSF(I)=0.379 | ***F(I)=180.549 | ***F(I)=41.102 | ***F(I)=85.323 | ***F(I)=611.163 | |||
| ***F(R)=14.706 | **F(R)=36.093 | ***F(R)=39.541 | *F(R)=4.316 | *F(R)=10.866 | |||
| ***F(IxR)=19.829 | ***F(IxR)=78.615 | ***F(IxR)=21.873 | **F(IxR)=11.155 | NSF(IxR)=0.891 | |||
| pH | EC (mS cm-1) | P Olsen (mg kg-1) | TN (%) | OC (%) | |||
| Peganum harmala | Non-inoculated | Control | 7.12 ± 0.02a | 3.67 ± 0.02c | 37.42 ± 2.50a | 0.42 ± 0.04a | 3.81 ± 0.01a |
| Moderate stress | 6.44 ± 0.02b | 4.35 ± 0.04b | 40.29 ± 1.87a | 0.43 ± 0.01a | 3.77 ± 0.02a | ||
| Severe stress | 6.35 ± 0.03c | 4.46 ± 0.02a | 38.21 ± 1.56a | 0.45 ± 0.03a | 3.65 ± 0.04b | ||
| ***F=814.085 | ***F=510.735 | NSF=1.611 | NSF=2.192 | **F=15.624 | |||
| Inoculated | Control | 7.10 ± 0.02a | 4.23 ± 0.01a | 54.15 ± 1.78c | 0.53 ± 0.07a | 4.67 ± 0.05a | |
| Moderate stress | 6.31 ± 0.02b | 4.23 ± 0.03a | 57.42 ± 0.94b | 0.53 ± 0.08a | 4.67 ± 0.01a | ||
| Severe stress | 6.32 ± 0.01b | 3.87 ± 0.07a | 60.29 ± 1.87a | 0.51 ± 0.02a | 4.43 ± 0.02b | ||
| ***F=1773.414 | ***F=60.310 | **F=11.178 | NSF=2.294 | *F=17.448 | |||
| ***F(I)=119.102 | *F(I)=6.258 | ***F(I)=472.278 | ***F(I)=152.581 | ***F(I)=1293.901 | |||
| ***F(R)=2258.193 | ***F(R)=110.180 | *F(R)=6.516 | NSF(R)=0.070 | ***F(R)=31.168 | |||
| NSF(IxR)=2.261 | ***F(IxR)=309.705 | *F(IxR)=4.021 | *F(IxR)=4.395 | NSF(IxR)=2.806 | |||
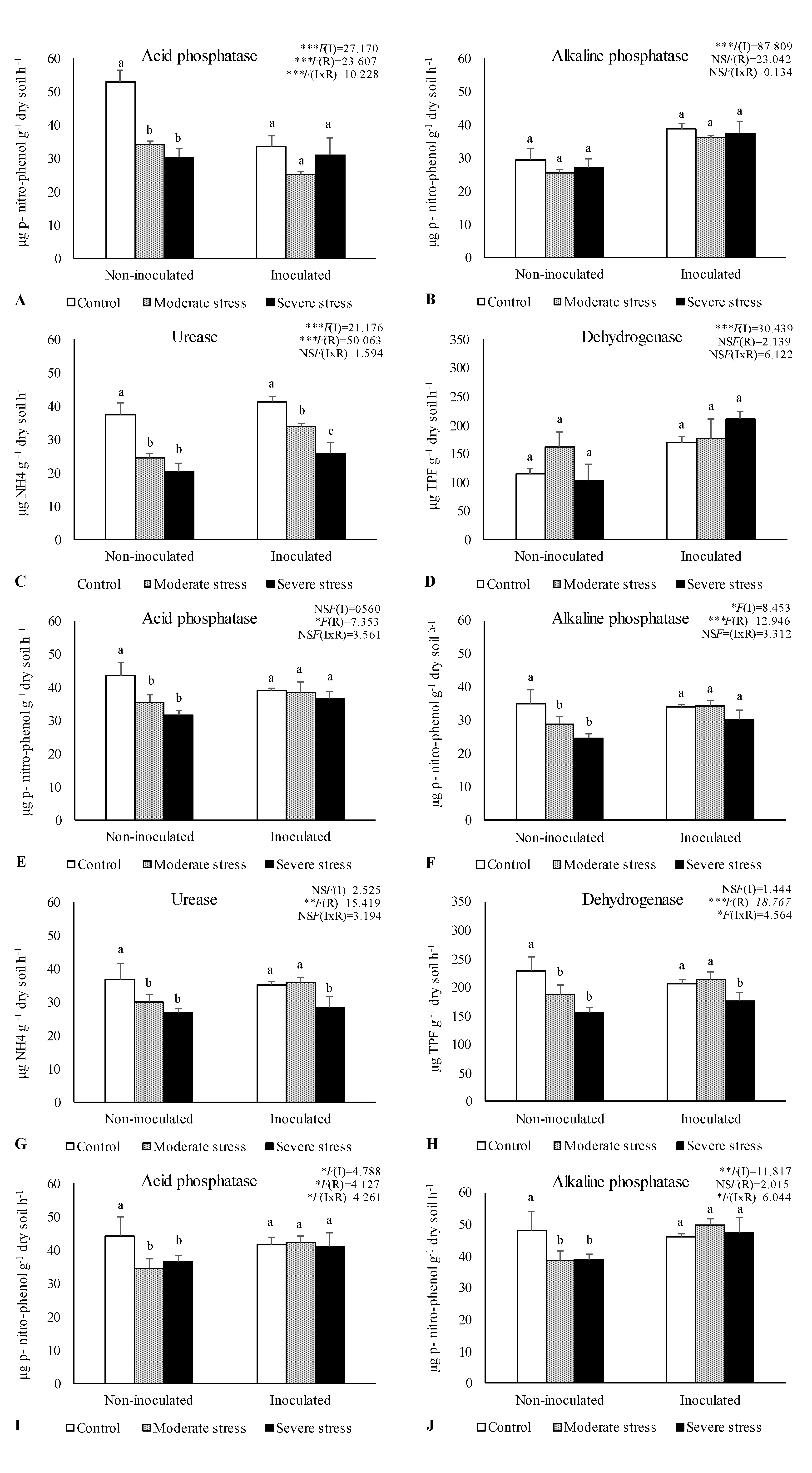
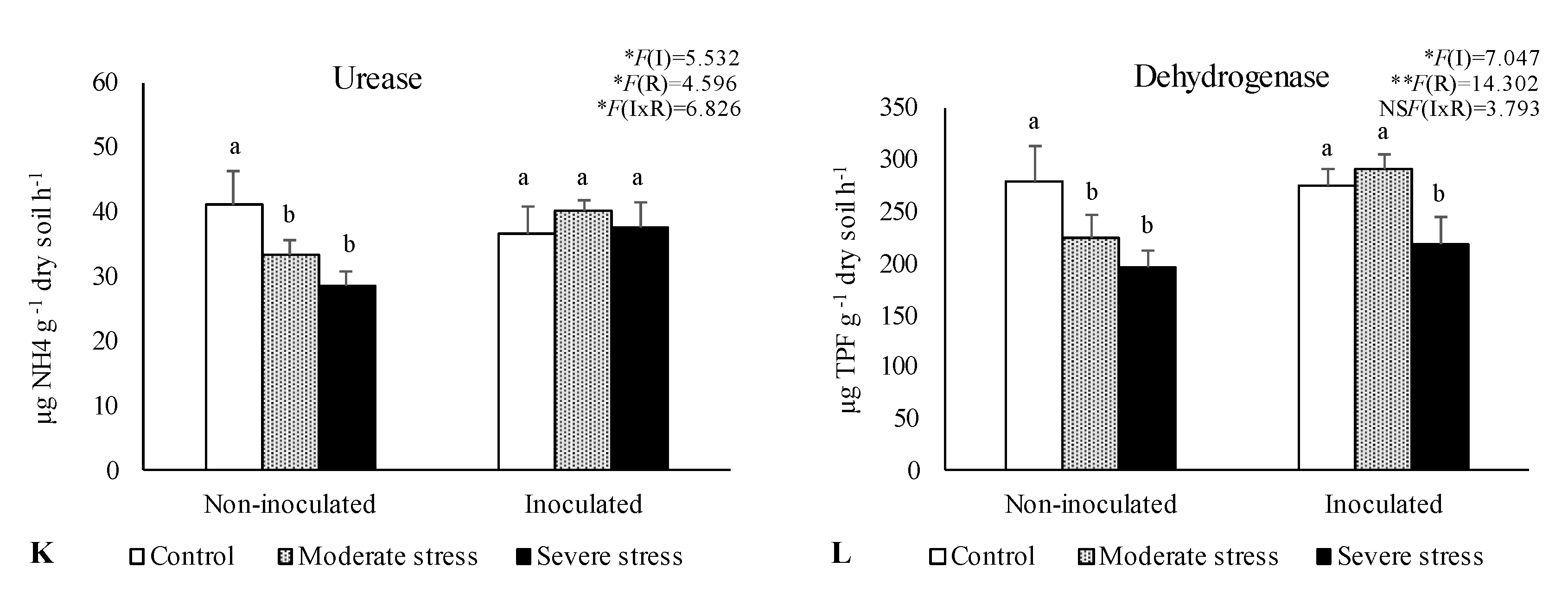
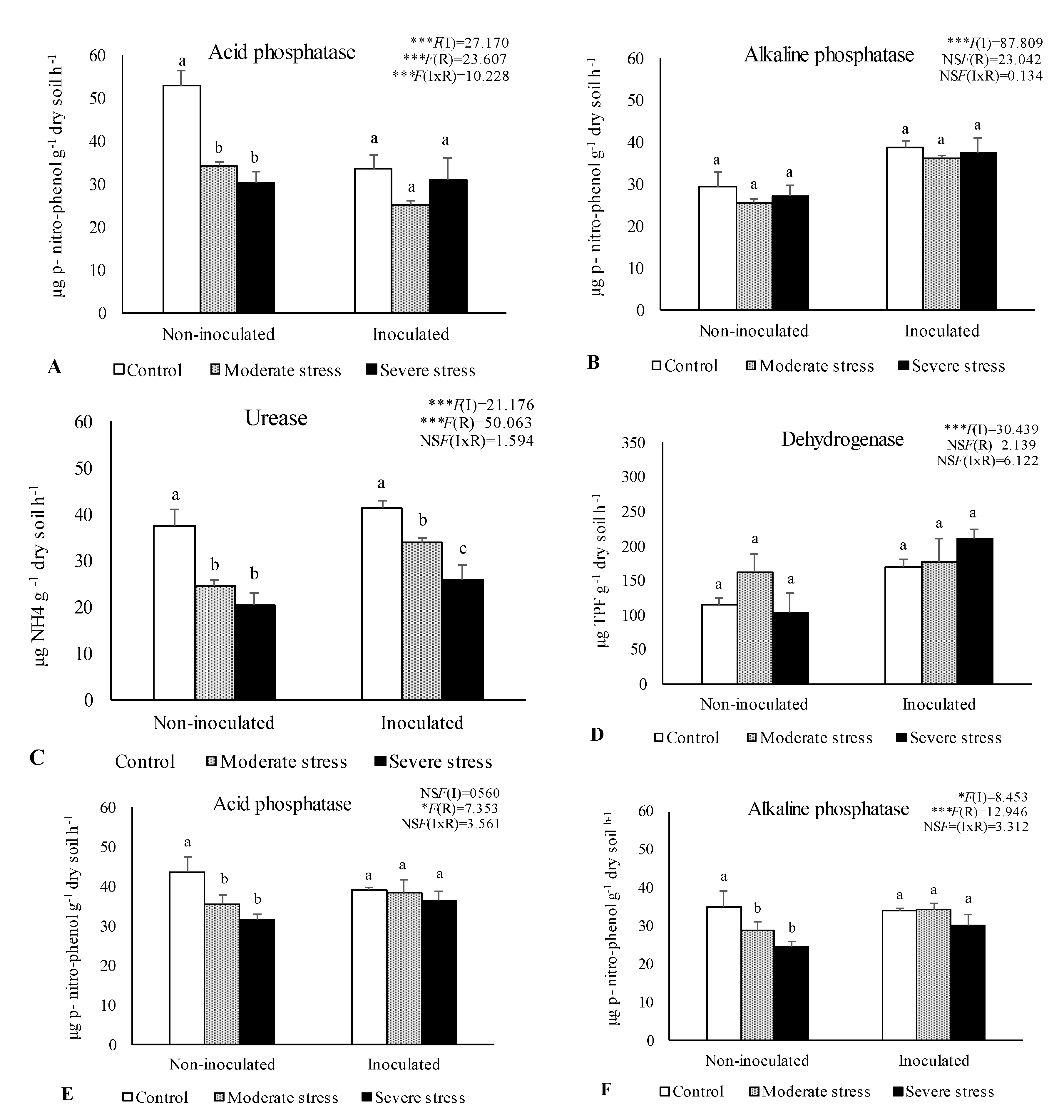
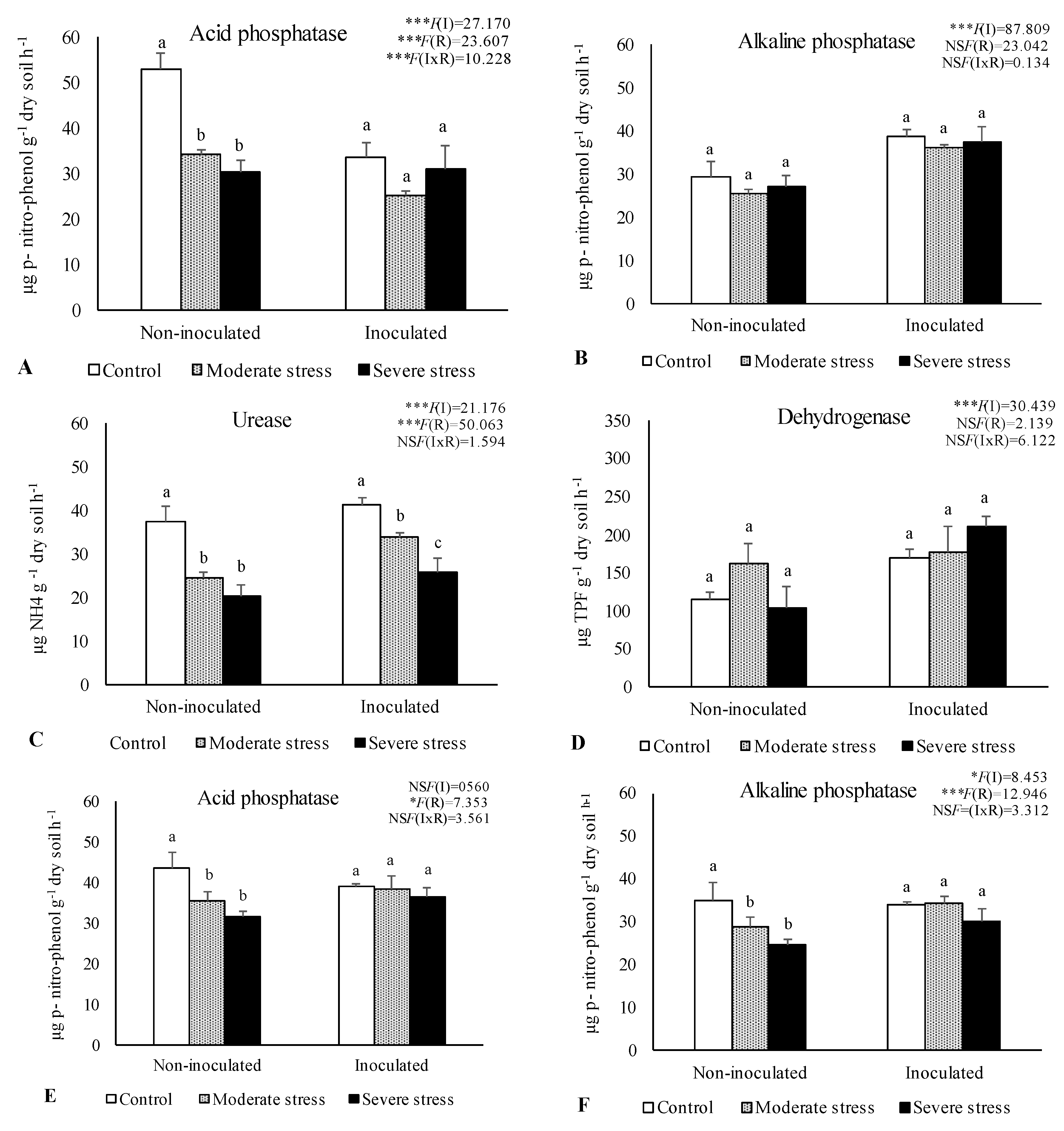
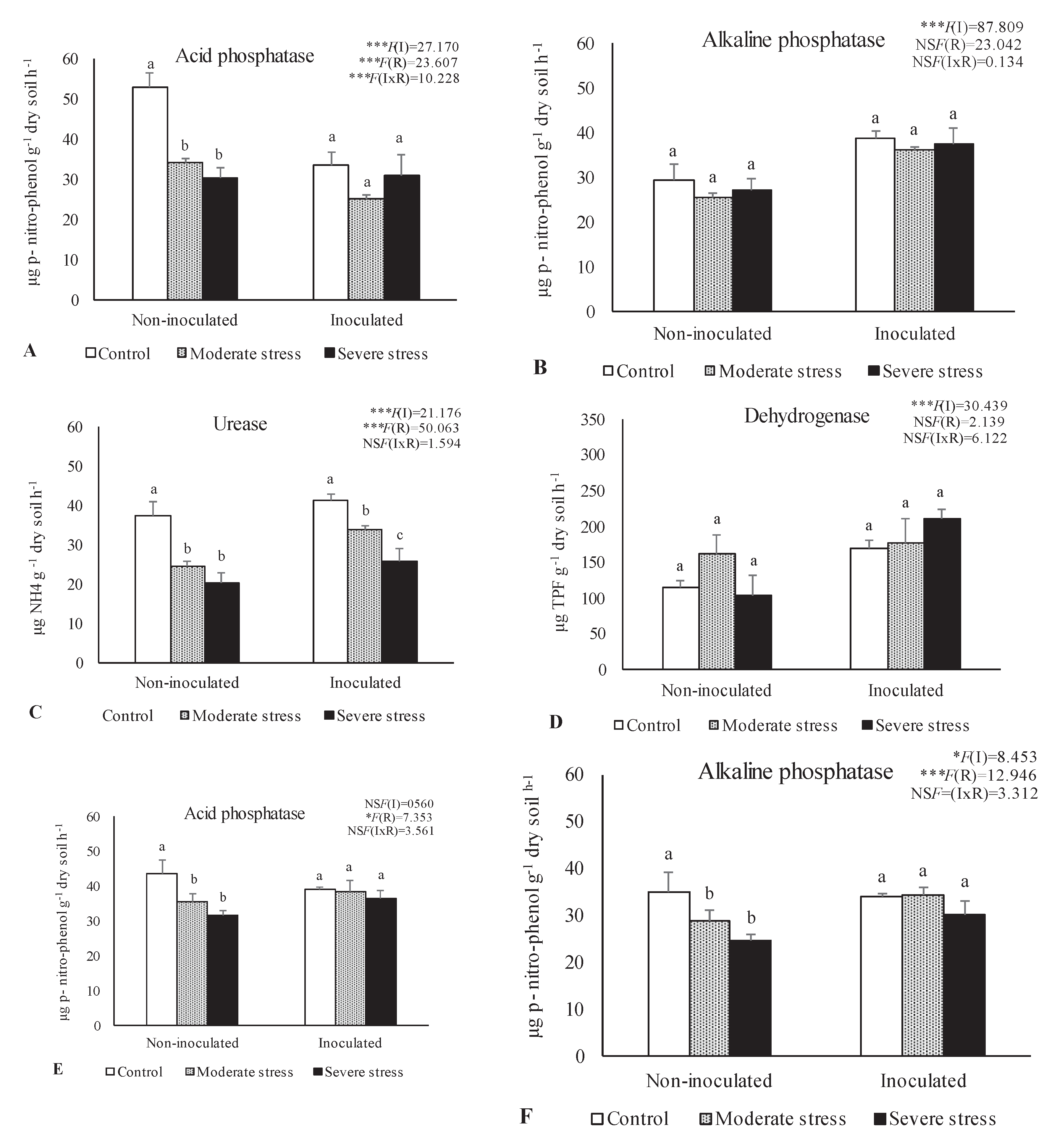
2.2. Influence of Irrigation Regimes and Microbial Inoculants on Soil Microbial Activity and Structure
| Bacteria (x107 CFU g-1 dry soil) |
Fungi (x104 CFU g-1 dry soil) | Actinobacteria (x103 CFU g-1 dry soil) | ||||
|---|---|---|---|---|---|---|
| Atriplex halimus | Non-inoculated | Control | 10.79 ± 0.01a | 2.33 ± 0.51b | 28.72 ± 0.01a | |
| Moderate stress | 10.95 ± 0.01a | 5.00 ± 0.60a | 24.05 ± 0.02b | |||
| Severe stress | 9.053 ± 0.02b | 4.53 ± 0.11a | 20.08 ± 0.04c | |||
| ***F=119.961 | ***F=39.114 | *F=9.769 | ||||
| Inoculated | Control | 11.32 ± 0.01a | 5.40 ± 0.30a | 34.72 ± 0.01a | ||
| Moderate stress | 11.34 ± 0.03a | 4.93 ± 0.57a | 33.33 ± 0.03a | |||
| Severe stress | 10.33 ± 0.02b | 3.20 ± 0.40b | 22.72 ± 0.02b | |||
| ***F=62.214 | *F=18.510 | ***F=33.444 | ||||
| ***F(I)=105.585 | *F(I)=7.440 | ***F(I)=33.136 | ||||
| ***F(R)=182.036 | ***F(R)=12.964 | ***F(R)=34.682 | ||||
| ***F(IxR)=16.730 | ***F(IxR)=41.226 | NSF(IxR)=3.409 | ||||
|
Bacteria (x107 CFU g-1 dry soil) |
Fungi (x104 CFU g-1 dry soil) | Actinobacteria (x103 CFU g-1 dry soil) | ||||
| Penisetum setaceum | Non-inoculated | Control | 11.48 ± 0.48a | 3.47 ± 0.23a | 22.05 ± 0.20a | |
| Moderate stress | 10.68 ± 0.40b | 2.40 ± 0.01b | 25.34 ± 0.41b | |||
| Severe stress | 9.413 ± 0.12c | 1.80 ± 0.20c | 14.72 ± 0.20c | |||
| ***F=36.628 | ***F=68.714 | ***F=25.125 | ||||
| Inoculated | Control | 12.63 ± 0.18a | 4.13 ± 0.46a | 25.32 ± 0.23a | ||
| Moderate stress | 12.29 ± 0.13b | 4.73 ± 0.23a | 27.36 ± 0.23a | |||
| Severe stress | 10.67 ± 0.23c | 2.67 ± 0.42b | 18.08 ± 0.11b | |||
| ***F=98.443 | *F=21.194 | *F=8.150 | ||||
| ***F(I)=113.483 | ***F(I)=78.233 | *F(I)=6.036 | ||||
| ***F(R)=105.897 | ***F(R)=44.860 | ***F(R)=25.857 | ||||
| NSF(IxR)=1.303 | ***F(IxR)=13.000 | NSF(IxR)=0.143 | ||||
|
Bacteria (x107 CFU g-1 dry soil) |
Fungi (x104 CFU g-1 dry soil) | Actinobacteria (x103 CFU g-1 dry soil) | ||||
| Peganum harmala | Non-inoculated | Control | 99.10 ± 0.32a | 4.80 ± 0.69a | 31.33 ± 0.11a | |
| Moderate stress | 94.51 ± 0.61b | 3.73 ± 0.23b | 32.02 ± 0.20a | |||
| Severe stress | 88.57 ± 0.14c | 2.40 ± 0.40c | 14.73 ± 0.23b | |||
| ***F=19.804 | ***F=18.769 | ***F=81.375 | ||||
| Inoculated | Control | 106.21 ± 0.72a | 5.27 ± 0.30a | 46.75 ± 0.30a | ||
| Moderate stress | 105.03 ± 0.11b | 4.47 ± 0.11b | 40.08 ± 0.34b | |||
| Severe stress | 103.24 ± 0.30c | 3.67 ± 0.11c | 32.03 ± 0.20c | |||
| ***F=15.250 | ***F=48.000 | ***F=19.158 | ||||
| ***F(I)=212.253 | ***F(I)=22.443 | ***F(I)=137.815 | ||||
| ***F(R)=29.521 | ***F(R)=44.328 | ***F(R)=69.148 | ||||
| ***F(IxR)=9.23 | ***F(IxR)=1.836 | *F(IxR)=6.037 | ||||
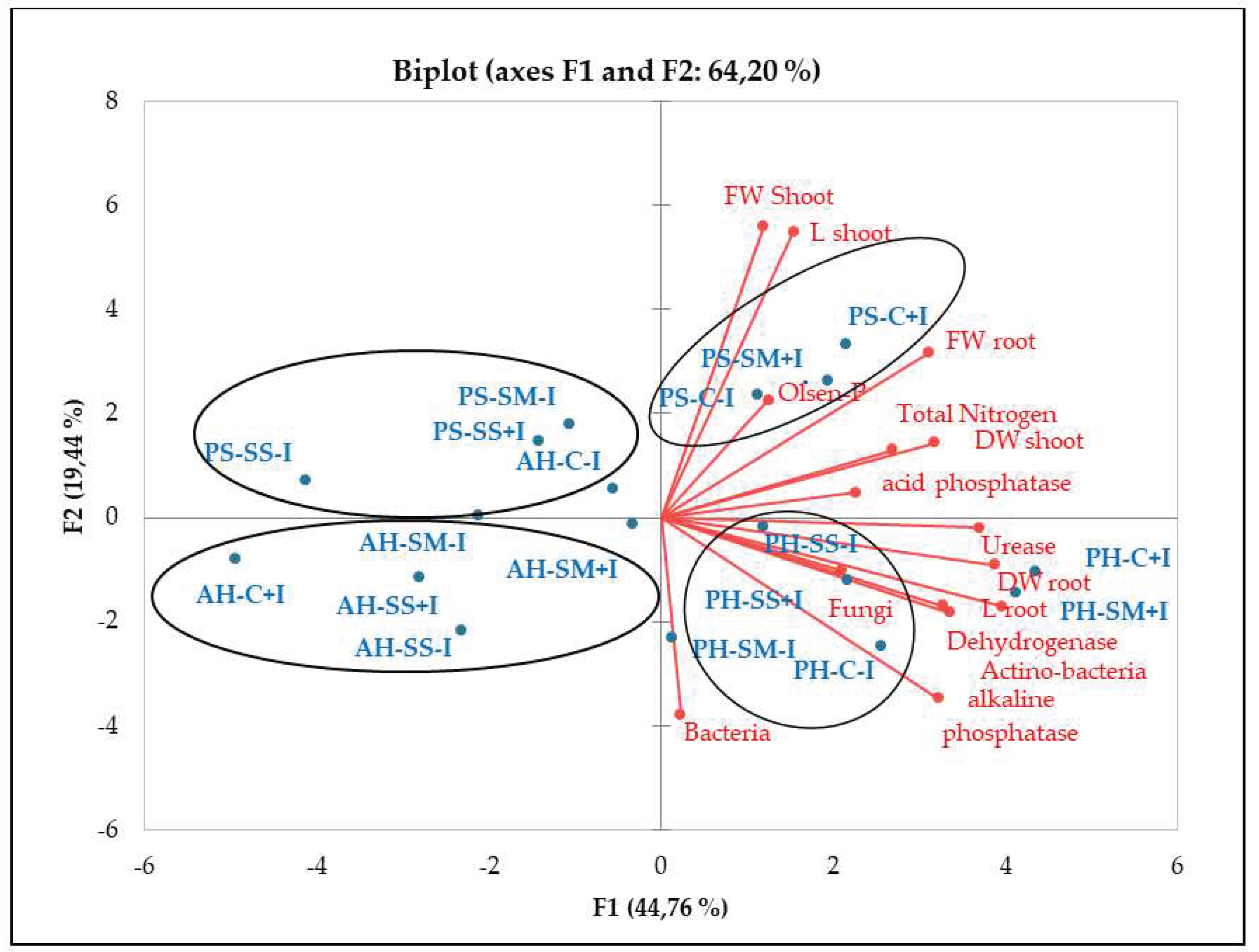
2.3. Influence of Irrigation Regimes and Microbial Inoculants on Plant Growth
3. Discussion
3.1. Improvement of Mine Tailings’ Chemical and Microbiological Properties by the Combined Use of Organo-Mineral Amendments and Bacterial Inoculants
3.2. Plant Growth in Amended Mine Tailings under Water Stress Conditions
4. Materials and Methods
4.1. Plant Species
4.2. PGPR Consortium, Seed Sterilization and Seedling Growth
4.3. Sampling and Characterization of Mine Tailings, Topsoil, and Organo-Mineral Amendments
| Parameters | Mine Tailings | Topsoil | Marble Sludge | Sheep Manure |
|---|---|---|---|---|
| pH H2O | 2.36 ± 0.01 | 8.02 ± 0.07 | 10.33 ± 0.24 | 8.36 ± 0.11 |
| pH KCl | 2.19 ± 0.12 | 7.88 ± 0.13 | 8.89 ± 0.11 | 7.92 ± 0.21 |
| EC (mS cm-1) | 7.86 | 2.44 | 0.198 | 11.47 |
| Organic Carbon (%) | Nd* | 1.13 ± 0.08 | 1.2 ± 0.05 | 40 ± 0.11 |
| Total N (%) | Nd* | 0.17 ± 0.06 | 0.14 ± 0.16 | 1.28 ± 0.05 |
| Olsen-P (mg kg-1) | Nd* | 43.42 ± 0.04 | 20 ± 1.23 | 1047 ± 11.24 |
| CaCO3 (%) | - | 16.89 ± 0.14 | 57.42 ± 0.25 | - |
| Texture | Loam | Sandy Loam | - | - |
| Clay (%) | 7.47 | 5.58 | - | - |
| Silt (%) | 37.37 | 39.08 | - | - |
| Sand (%) | 50.47 | 52.89 | - | - |
| Moisture content (%) | 41.92 | 40.72 | 40.62 | 40.49 |
4.4. Greenhouse Pot Experiment
4.4.1. Soil Physicochemical Analysis
4.4.2. Soil Enzymatic Activity
4.4.3. Enumeration of Culturable Microorganisms
4.4.4. Plant Biometric Parameters
4.4.5. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Franzaring, Franzaring, J., S. Ancora, L. Paoli, A. H. Fongoh, P. Büttner, A. Fangmeier, S. Schlosser, and F. Monaci. "Phytotoxicity of polymetallic mine wastes from southern Tuscany and Saxony." Ecotoxicology and Environmental Safety 162, 2018: 505-513. [CrossRef]
- Akcil, Ata, Ceren Erust, Sevda Ozdemiroglu, Viviana Fonti, and Francesca Beolchini. "A review of approaches and techniques used in aquatic contaminated sediments: metal removal and stabilization by chemical and biotechnological processes." Journal of Cleaner Production 86, 2015: 24-36. [CrossRef]
- Shackira, A. M., and Jos T. Puthur. "Phytostabilization of heavy metals: Understanding of principles and practices." Plant-metal interactions (2019): 263-282. [CrossRef]
- Tang, Chunfang, Yonghua Chen, Qianni Zhang, Jianbin Li, Fuyun Zhang, and Zhiming Liu. "Effects of peat on plant growth and lead and zinc phytostabilization from lead-zinc mine tailing in southern China: Screening plant species resisting and accumulating metals." Ecotoxicology and environmental safety 176, 2019: 42-49. [CrossRef]
- Testiati E, Parinet J, Massiani C, Laffont-Schwob I, Rabier J, Pfeifer HR & Prudent P. Trace metal and metalloid contamination levels in soils and in two native plant species of a former industrial site: evaluation of the phytostabilization potential. J. Hazard. Mater. 2013: 248: 131-141. [CrossRef]
- Dary M, Chamber-Pérez MA, Palomares AJ, Pajuelo E. “In situ” phytostabilisation of heavy metal polluted soils using Lupinusluteus inoculated with metal resistant plant growth promoting rhizobacteria. J. Hazard. Mater. 2010, 177: 323-330. [CrossRef]
- Clemente,R., Walker, D.J., Pardo, T., Martínez-Fernández, D. & Bernal, M.P. The use of a halophytic plant species and organic amendments for the remediation of a trace elements-contaminated soil under semi-arid conditions. J. Hazard. Mater. 2012, 223–224, 63– 71. [CrossRef]
- Ma, Ying, Mani Rajkumar, Chang Zhang, and Helena Freitas. "Inoculation of Brassica oxyrrhina with plant growth promoting bacteria for the improvement of heavy metal phytoremediation under drought conditions." Journal of hazardous materials 320 2016: 36-44. [CrossRef]
- Visser, A., Kroes, J., van Vliet, M. T., Blenkinsop, S., Fowler, H. J., & Broers, H. P. Climate change impacts on the leaching of a heavy metal contamination in a small lowland catchment. Journal of contaminant hydrology, 2012, 127(1-4), 47-64. [CrossRef]
- Jha, S., & Srivastava, R. Impact of drought on vegetation carbon storage in arid and semi-arid regions. Remote Sensing Applications: Soci and Envi, 2018, 11, 22-29. [CrossRef]
- Párraga-Aguado, I., González-Alcaraz, M.N., Álvarez-Rogel, J., Conesa, 593 H.M. Assessment of the employment of halophyte plant species for the phytomanagement of mine tailings in semiarid areas. Ecol. Eng. 2014, 71, 598–604. [CrossRef]
- Boularbah A, Schwartz C, Bitton G, Aboudrar W, Ouhammou A & Morel JL. Heavy metal contamination from mining sites in South Morocco: 2. Assessment of metal accumulation and toxicity in plants. Chem. 2006b 63: 811-817. [CrossRef]
- Boularbah A, Schwartz C, Bitton G & Morel JL. Heavy metal contamination from mining sites in South Morocco: 1. Use of a biotest to assess metal toxicity of tailings and soils. Chem. 2006a, 63: 802-810. [CrossRef]
- Mendez MO, Maier RM. Phytostabilization of mine tailings in arid and semiarid environments–an emerging remediation technology. Environ Health Perspect. 2008, 116:278–283.
- Maestri E, Marmiroli M, Visioli G, Marmiroli N. Metal tolerance and hyperaccumulation: costs and trade-offs between traits and environment. Environ Exp Biol. 2010, 68:1–1. [CrossRef]
- Zou T, Li T, Zhang X, Yu H & Huang H. Lead accumulation and phytostabilization potential of dominant plant species growing in a lead–zinc mine tailing. Environ. Earth Sci.2012, 65: 621-630. [CrossRef]
- Ali H, Khan E & Sajad MA. Phytoremediation of heavy metals: concepts and applications.c).Chemo. 2013, 91:869–881. [CrossRef]
- Meeinkuirt, W., Pokethitiyook, P., Kruatrachue, M., Tanhan, P., Chaiyarat, R. Phytostabilization of a Pb-contaminated mine tailing by various tree species in potand field trial experiments. Int. J. Phytoremediation. 2012, 14, 925–938. 2012; 14, 925–938. [CrossRef]
- Wasilkowski, D., Nowak, A., Płaza, G., & Mrozik, A. Effects of pulp and Na bentonite amendments on the mobility of trace elements, soil enzymes activity and microbial parameters under ex situ aided phytostabilization. Plos one. 2017, 12(1), e0169688. [CrossRef]
- Zhou, Hu, Chong Chen, Daozhong Wang, Emmanuel Arthur, Zhongbin Zhang, Zichun Guo, Xinhua Peng, and Sacha J. Mooney. "Effect of long-term organic amendments on the full-range soil water retention characteristics of a Vertisol." Soil and Tillage Research 202, 2020: 104663. [CrossRef]
- Kang, Sung-Won, and Kwang-Ho Ahn. "The Influence of Organic Matter Origin on the Chlorine Bulk Decay Coefficient in Reclaimed Water." Water 14, no. 5. 2022: 765. [CrossRef]
- Taban, M., and S. A. R. Movahedi Naeini. "Effect of aquasorb and organic compost amendments on soil water retention and evaporation with different evaporation potentials and soil textures." Communications in soil science and plant analysis 37, no. 13-14. 2006: 2031-2055. [CrossRef]
- Ju, W., Liu, L., Fang, L., Cui, Y., Duan, C., Wu, H. Impact of co-inoculation with plant-growth-promoting rhizobacteria and rhizobium on the biochemical responses of alfalfa-soil system in copper contaminated soil. Ecotoxicol. Environ. Saf. 2019. 167, 218–226. [CrossRef]
- Zainab, N., Din, B. U., Javed, M. T., Afridi, M. S., Mukhtar, T., Kamran, M. A. and Chaudhary, H. J. Deciphering metal toxicity responses of flax (Linum usitatissimum L.) with exopolysaccharide and ACC-deaminase producing bacteria in industrially contaminated soils. Plant physiology and biochemistry. 2020. 152, 90-99. [CrossRef]
- Benidire L, Pereira S, Aboudrar W, Hafidi M, Castro P, Boularbah A. Remediation of metal-contaminated mine tailings by the application of organic and mineral amendments. J Soils Sediments. 2022. 1-14. [CrossRef]
- Rani R, Kumar V, Gupta P, Chandra A. Potential use of Solanum lycopersicum and plant growth promoting rhizobacterial (PGPR) strains for the phytoremediation of endosulfan stressed soil. Chemosphere. 2021. 279: 130589. [CrossRef]
- Madline, A., Benidire, L., & Boularbah, A. Alleviation of salinity and metal stress using plant growth-promoting rhizobacteria isolated from semiarid Moroccan copper-mine soils. Environmental Science and Pollution Research. 2021. 28(47), 67185-67202. [CrossRef]
- Sandhya VS KZ, Ali SZ, Grover M, Reddy G & Venkateswarlu B. Effect of plant growth promoting Pseudomonas spp. on compatible solutes, antioxidant status and plant growth of maize under drought stress. Plant Growth Regul. 62: 21- 30. [CrossRef]
- Wang, X., Tang, C., Severi, J., Butterly, C. R., and Baldock, J. A. Rhizosphere priming effect on soil organic carbon decomposition under plant species differing in soil acidification and root exudation. New Phytologist. 2016. 211(3), 864-873. [CrossRef]
- Gispert, M., Emran, M., Pardini, G., Doni, S., Ceccanti, B. The impact of land management and abandonment on soil enzymatic activity, glomalin content and aggregate stability. Geoderma. 2013. 202–203, 51–61. [CrossRef]
- Yadav, B. K., Akhtar, M., and Panwar, J. Rhizospheric plant-microbe interactions: key factors to soil fertility and plant nutrition. In Plant microbes symbiosis: applied facets. 2015. (pp. 127-145). Springer, New Delhi. [CrossRef]
- Bowsher, A. W., Ali, R., Harding, S. A., Tsai, C. J., and Donovan, L. A. Evolutionary divergences in root exudate composition among ecologically-contrasting Helianthus species. Plos one. 2016. 11(1), e0148280. [CrossRef]
- Gamage, S. S. W., Masakorala, K., Brown, M. T., and Gamage, S. M. K. W. Comparative phytoremediation potentials of Impatiens balsamina L. and Crotalaria retusa L. for soil contaminated with used lubricating oil. Environmental Advances. 2021. 5, 100095. [CrossRef]
- Chaparro, J. M., Sheflin, A. M., Manter, D. K., and Vivanco, J. M. Manipulating the soil microbiome to increase soil health and plant fertility. Biology and Fertility of Soils. 2012. 48(5), 489-499. [CrossRef]
- Kim, Jae Hwan, So-Jeong Kim, and In-Hyun Nam. "Effect of treating acid sulfate soils with phosphate solubilizing bacteria on germination and growth of tomato (Lycopersicon esculentum L.)." International Journal of Environmental Research and Public Health 18, no. 17. 2021. 8919. [CrossRef]
- Altaf, M. Functional diversity of nitrogen-fixing plant growth-promoting Rhizobacteria: The story so far. In Soil Nitrogen Ecology. 2021. (pp. 327-348). Springer, Cham. [CrossRef]
- Zeng, Q., Ding, X., Wang, J., Han, X., Iqbal, H., and Bilal, M. Insight into soil nitrogen and phosphorus availability and agricultural sustainability by plant growth-promoting rhizobacteria. Environmental Science and Pollution Research. 2022. 1-18. [CrossRef]
- Pramanik, K., Mitra, S., Sarkar, A., and Maiti, T. K. Alleviation of phytotoxic effects of cadmium on rice seedlings by cadmium resistant PGPR strain Enterobacter aerogenes MCC 3092. Journal of hazardous materials. 2018. 351, 317-329. [CrossRef]
- Benidire L, Madline A, Pereira, SIA, Castro, PML, Boularbah A. Synergistic effect of organo-mineral amendments and plant growth-promoting rhizobacteria (PGPR) on the establishment of vegetation cover and amelioration of mine tailings. Chemosphere. 2020. 262: 127803. [CrossRef]
- Daraz, U., Li, Y., Sun, Q., Zhang, M., and Ahmad, I. Inoculation of Bacillus spp. modulate the soil bacterial communities and available nutrients in the rhizosphere of vetiver plant irrigated with acid mine drainage. Chemosphere. 2021. 263, 128345. [CrossRef]
- Madejón, P., Navarro-Fernández, C. M., Madejón, E., López-García, Á., and Marañón, T. Plant response to mycorrhizal inoculation and amendments on a contaminated soil. Science of the Total Environment. 2021. 789, 147943. [CrossRef]
- Tirry, N., Kouchou, A., Laghmari, G., Lemjereb, M., Hnadi, H., Amrani, K. and El Ghachtouli, N. (2021). Improved salinity tolerance of Medicago sativa and soil enzyme activities by PGPR. Biocatalysis and Agricultural Biotechnology. 2021. 31, 101914. [CrossRef]
- Dey, G., Banerjee, P., Maity, J. P., Sharma, R. K., Gnanachandrasamy, G., Huang, Y. H., and Chen, C. Y. Heavy metals distribution and ecological risk assessment including arsenic resistant PGPR in tidal mangrove ecosystem. Marine Pollution Bulletin. 2022. 181, 113905. [CrossRef]
- Sardans, J.; Peñuelas, J. Drought decreases soil enzyme activity in a Mediterranean Quercus ilex L. forest. Soil Biol. Biochem. 2005, 37, 455–461. [Google Scholar] [CrossRef]
- Pohlon, E.; Fandino, A.O. & Marxsen, J. Bacterial community composition and extracellular enzyme activity in temperate streambed sediment during drying and rewetting. PLoS ONE. 2013. 8, e83365. [CrossRef]
- Siebielec, S., Siebielec, G.; Klimkowicz-Pawlas, A.; Gał ˛azka, A.; Grz ˛adziel, J.; Stuczy ´nski, T. Impact of water stress on microbial community and activity in sandy and loamy soils. Agronomy. 2020, 10, 1429. [CrossRef]
- Schreckinger, J.; Mutz, M.; Mendoza-Lera, C.; Frossard, A. Attributes of drying define the structure and functioning of microbial communities in temperate riverbed sediment. Front. Microbiol. 2021, 12, 676615. [Google Scholar] [CrossRef]
- Sardans, J., and Peñuelas, J. Soil enzyme activity in a Mediterranean forest after six years of drought. Soil Science Society of America Journal. 2010. 74(3), 838-851. [CrossRef]
- Gao, W., Reed, S. C., Munson, S. M., Rui, Y., Fan, W., Zheng, Z., and Hao, Y. Responses of soil extracellular enzyme activities and bacterial community composition to seasonal stages of drought in a semiarid grassland. Geoderma. 2021. 401, 115327. [CrossRef]
- Chaudhary, D. R., Rathore, A. P., and Sharma, S. Effect of halotolerant plant growth promoting rhizobacteria inoculation on soil microbial community structure and nutrients. Applied Soil Ecology. 2022. 150, 103461. [CrossRef]
- Zhu, L., Wang, X., Chen, F., Li, C., and Wu, L. Effects of the successive planting of Eucalyptus urophylla on soil bacterial and fungal community structure, diversity, microbial biomass, and enzyme activity. Land degradation and development. 2019. 30(6), 636-646. [CrossRef]
- Lin, H., Liu, C., Li, B., and Dong, Y. Trifolium repens L. regulated phytoremediation of heavy metal contaminated soil by promoting soil enzyme activities and beneficial rhizosphere associated microorganisms. Journal of Hazardous Materials. 2021. 402, 123829. [CrossRef]
- Piromyou, P., Noisangiam, R., Uchiyama, H., Tittabutr, P., Boonkerd, N., and Teaumroong, N. Indigenous microbial community structure in rhizosphere of Chinese kale as affected by plant growth-promoting rhizobacteria inoculation. Pedosphere, 2013. 23(5), 577-592. [CrossRef]
- Ilyas, N., Mumtaz, K., Akhtar, N., Yasmin, H., Sayyed, R. Z., Khan, W., and Ali, Z. Exopolysaccharides producing bacteria for the amelioration of drought stress in wheat. Sustainability, 2020. 12(21), 8876. [CrossRef]
- Kumar, A., Singh, S., Gaurav, A. K., Srivastava, S., and Verma, J. P. Plant growth-promoting bacteria: biological tools for the mitigation of salinity stress in plants. Frontiers in Microbiology, 2020. 11, 1216. [CrossRef]
- Shi, G., Xia, S., Ye, J., Huang, Y., Liu, C., and Zhang, Z. PEG-simulated drought stress decreases cadmium accumulation in castor bean by altering root morphology. Environmental and Experimental Botany, 2015. 111, 127-134. [CrossRef]
- Wang, J. Q., Li, H., Liu, Q., and Xiang, D. (2019). Effects of drought stress on root development and physiological characteristics of sweet potato at seedling stage. Ying Yong Sheng tai xue bao= The Journal of Applied Ecology, 2019. 30(9), 3155-3163. [CrossRef]
- Xiao, X. X. "The physiological and biochemical response of longan (Dimocarpus longan Lour.) to aluminum stress and rectification of aluminum toxicity." Fujian Journal of Agricultural Science 17, 2002: 182-185.
- Orrego, F., Ortíz-Calderón, C., Lutts, S., and Ginocchio, R. Effect of single and combined Cu, NaCl and water stresses on three Atriplex species with phytostabilization potential. South African Journal of Botany, 2020. 131, 161-168. [CrossRef]
- Nedjimi B, Beladel B, Guit B. Biodiversity of halophytic vegetation in chott Zehrez lake of Djelfa (Algeria). Am J Plant Sci3. 2012. 1513–1660. [CrossRef]
- Geng, M., Xu, M., Xiao, H., Wang, H., He, L., Zhao, Z., and Yu, M. Protective role of mucilage against Al toxicity to root apex of pea (Pisum sativum). Acta physiologiae plantarum. 2012. 34(4), 1261-1266. https://link.springer.com/article/10.1007/s11738-011-0919-1.
- Liao, M., and Xie, X. M. Cadmium release in contaminated soils due to organic acids. Pedosphere, 2004. 14(2), 223-228.
- Bali, A. S., Sidhu, G. P. S., & Kumar, V. Root exudates ameliorate cadmium tolerance in plants: a review. Environmental Chemistry Letters, 2020. 18(4), 1243-1275. [CrossRef]
- Yamaguchi, M., &Sharp, R. E. Complexity and coordination of root growth at low water potentials: recent advances from transcriptomic and proteomic analyses. Plant, Cell and Environment, 2010. 33(4), 590-603. [CrossRef]
- Naseem, H., Ahsan, M., Shahid, M. A., and Khan, N. Exopolysaccharides producing rhizobacteria and their role in plant growth and drought tolerance. Journal of basic microbiology, 2018. 58(12), 1009-1022. [CrossRef]
- Abdelkrim, S., Jebara, S. H., Saadani, O., Abid, G., Taamalli, W., Zemni, H., and Jebara, M. In situ effects of Lathyrus sativus-PGPR to remediate and restore quality and fertility of Pb and Cd polluted soils. Ecotoxicology and environmental safety. 2020. 192, 110260. [CrossRef]
- Morcillo, R. J., and Manzanera, M. The effects of plant-associated bacterial exopolysaccharides on plant abiotic stress tolerance. Metabolites, 2021. 11(6), 337. [CrossRef]
- Ahmed, B., Shahid, M., Syed, A., Rajput, V. D., Elgorban, A. M., Minkina, T., and Lee, J. Drought tolerant Enterobacter sp./Leclercia adecarboxylata secretes indole-3-acetic acid and other biomolecules and enhances the biological attributes of Vigna radiata (L.) R. Wilczek in water deficit conditions. Biology, 2021. 10(11), 1149. [CrossRef]
- Hou, X., Teng, W., Hu, Y., Yang, Z., Li, C., Scullion, J., and Zheng, R. Potential phytoremediation of soil cadmium and zinc by diverse ornamental and energy grasses. BioResources, 2020. 15(1), 616-640. [CrossRef]
- Benidire L, Pereira, SIA, Castro, PML, Boularbah, A. Assessment of plant growth-promoting bacterial populations in the rhizosphere of metallophytes from the Kettara mine, Marrakech. Environ Sci Pollut Res, 2016. 23: 21751-21765. [CrossRef]
- El Hamiani O, El Khalil H, Sirguey C, Ouhammou A, Bitton G, Schwartz C, Boularbah A. Metal concentrations in plants from mining areas in South Morocco: health risks assessment of consumption of edible and aromatic plants. CLEAN–Soil Air Water, 2015. 43: 399-407. [CrossRef]
- Olsen SR, Sommers LE. Phosphorus. In: Page AL, Miller RH, Keeney DR (eds.), Methods of Soil Analysis Part 2, Second Edition. Agron Soc Am, 1982. Madison, pp. 403e430.
- Aubert G (ed). Methodes d’analyses des sols. C.R.D.P., 1978. Marseille, p. 360.
- Tabatabai MA, Bremner JM. Use of p-nitrophenyl phosphate for assay of soil phosphatase activity. Soil Biol Biochem, 1969. 1: 301e307. [CrossRef]
- Alef K, Nannipieri P. Methods in applied soil microbiology and biochemistry. Inst Ecol Chem Geochem, 1995. Univ Bayreuth, Germany, 569p.
- Kandeler E, Gerber H. Short-term assay of soil urease activity using colorimetric determination of ammonium. Biol Fertil Soils, 1988. 6: 68-72. [CrossRef]
- Zhu, L., Wang, X., Chen, F., Li, C., and Wu, L. Effects of the successive planting of Eucalyptus urophylla on soil bacterial and fungal community structure, diversity, microbial biomass, and enzyme activity. Land degradation and development, 2019. 30(6), 636-646. [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




