Submitted:
03 January 2024
Posted:
03 January 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Template Effects in the Diels-Alder Reaction
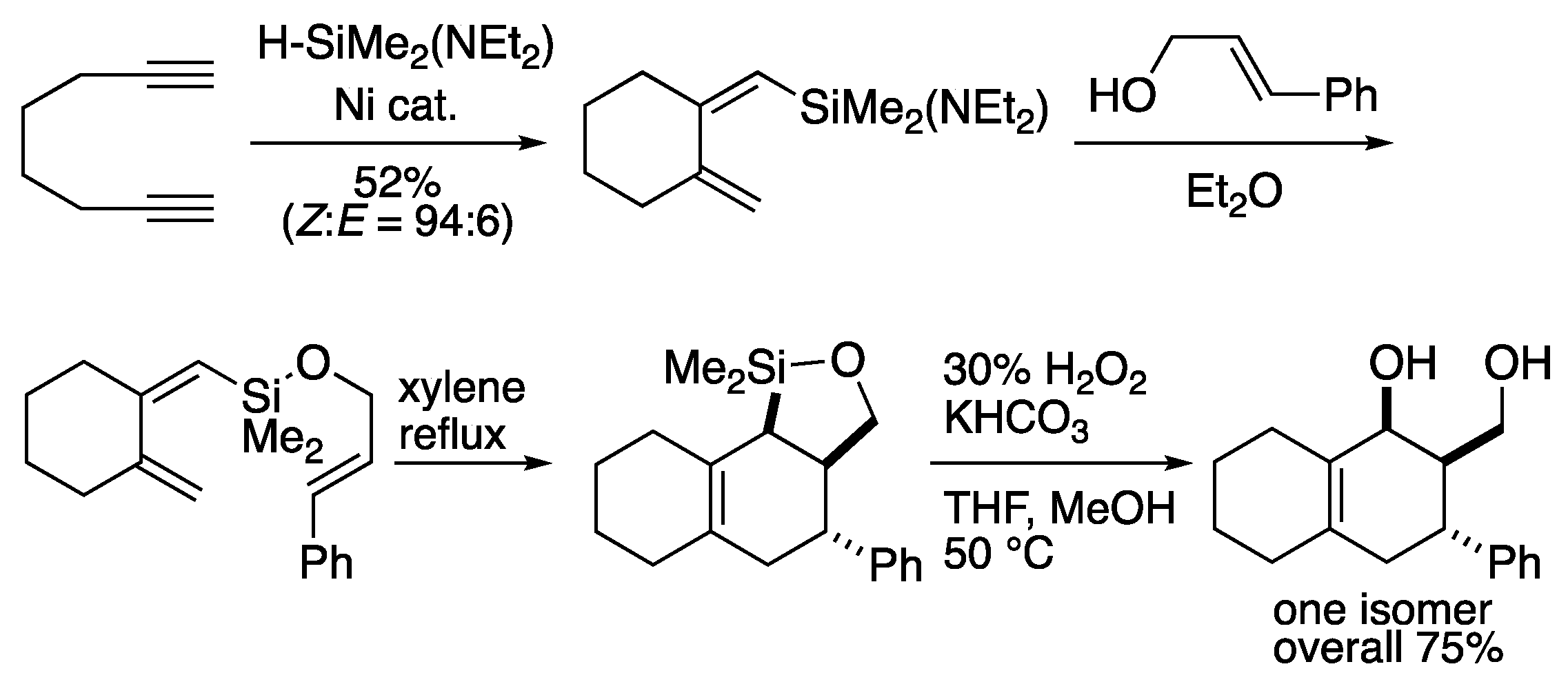
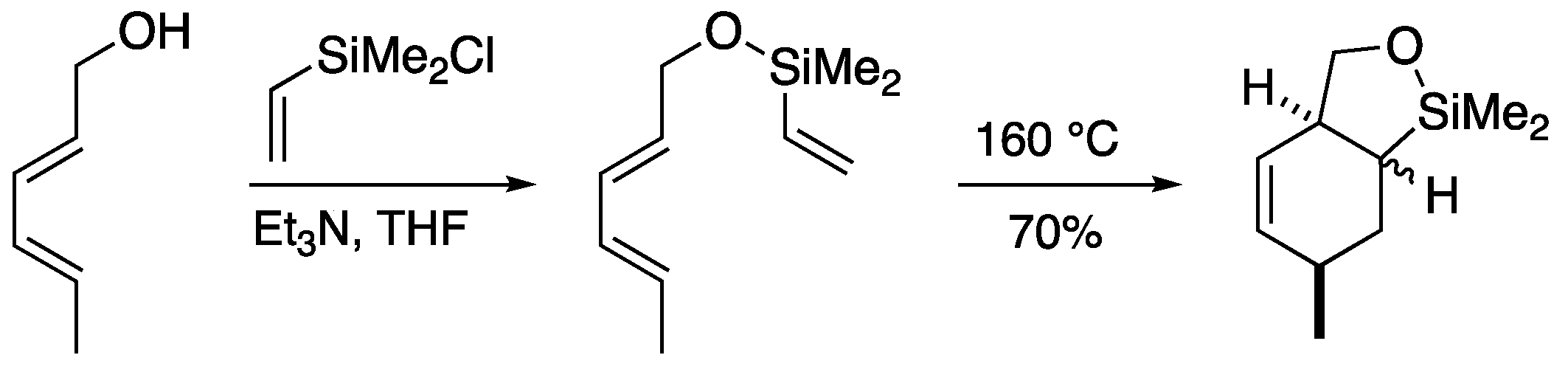
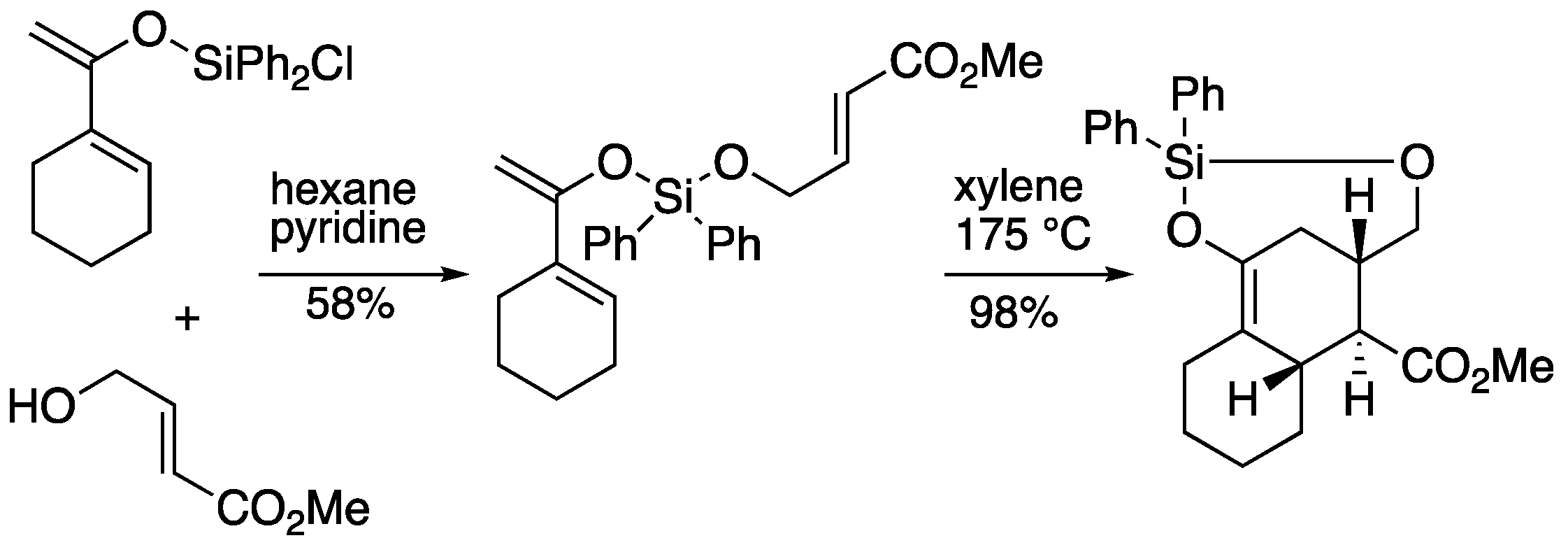
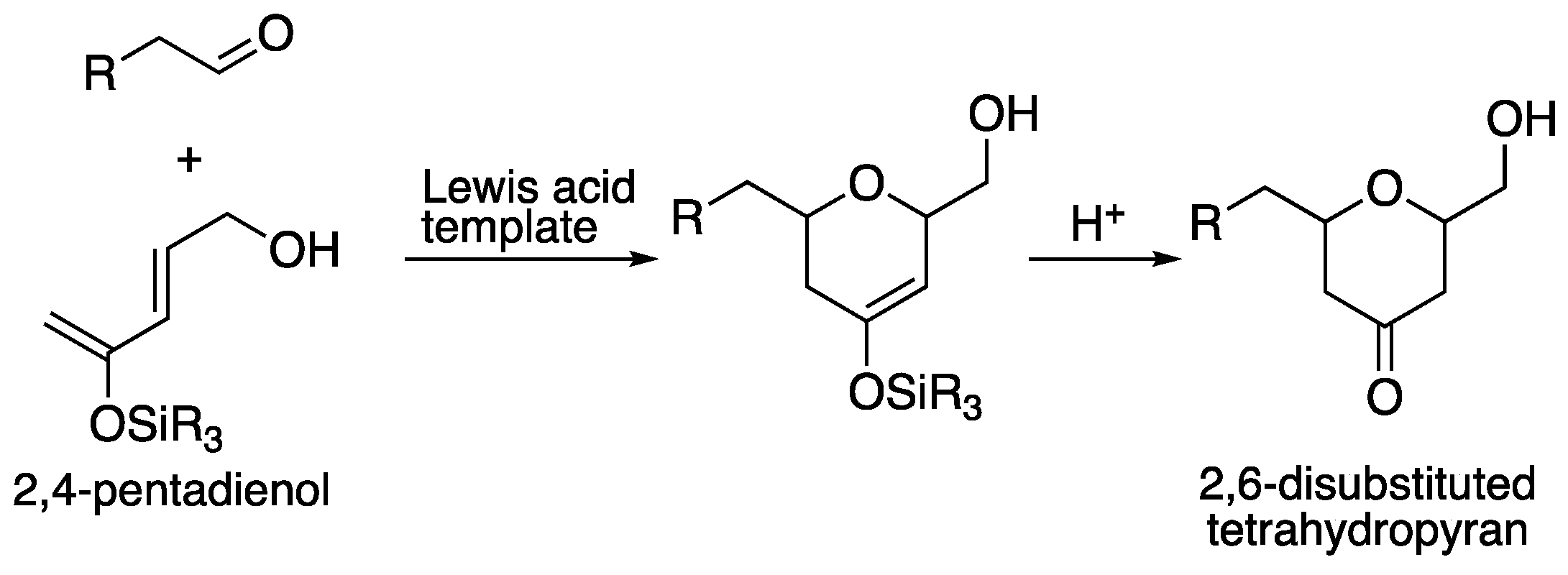
2.1. Silicon-tethered Diels-Alder Reaction
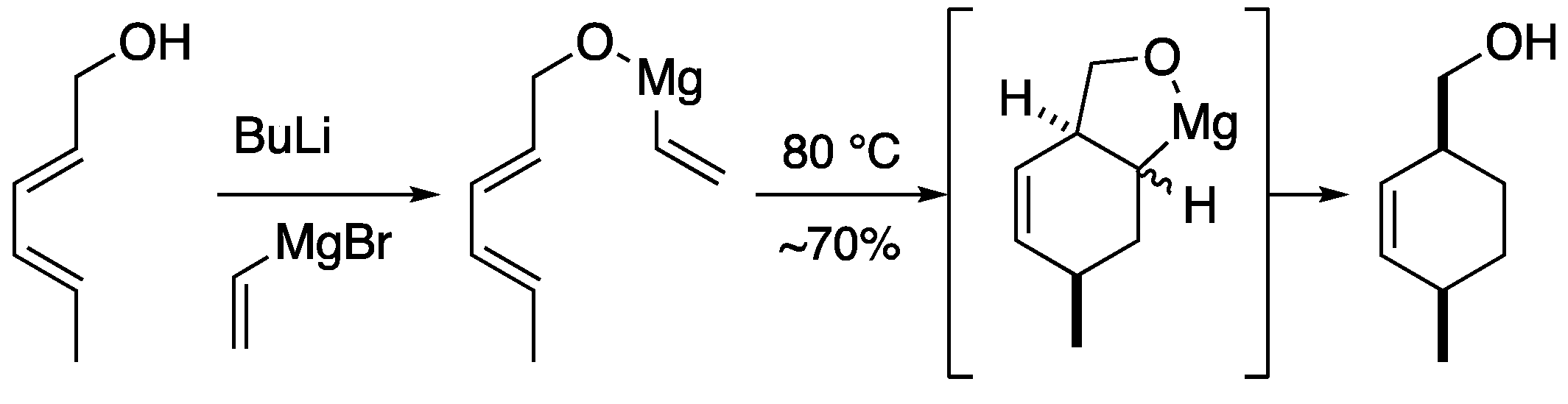
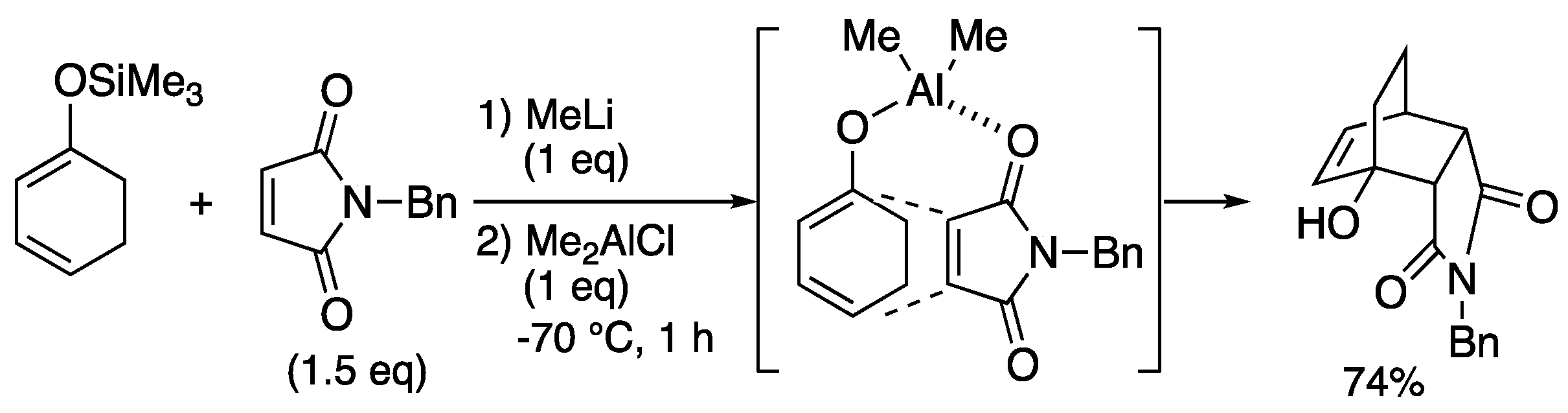
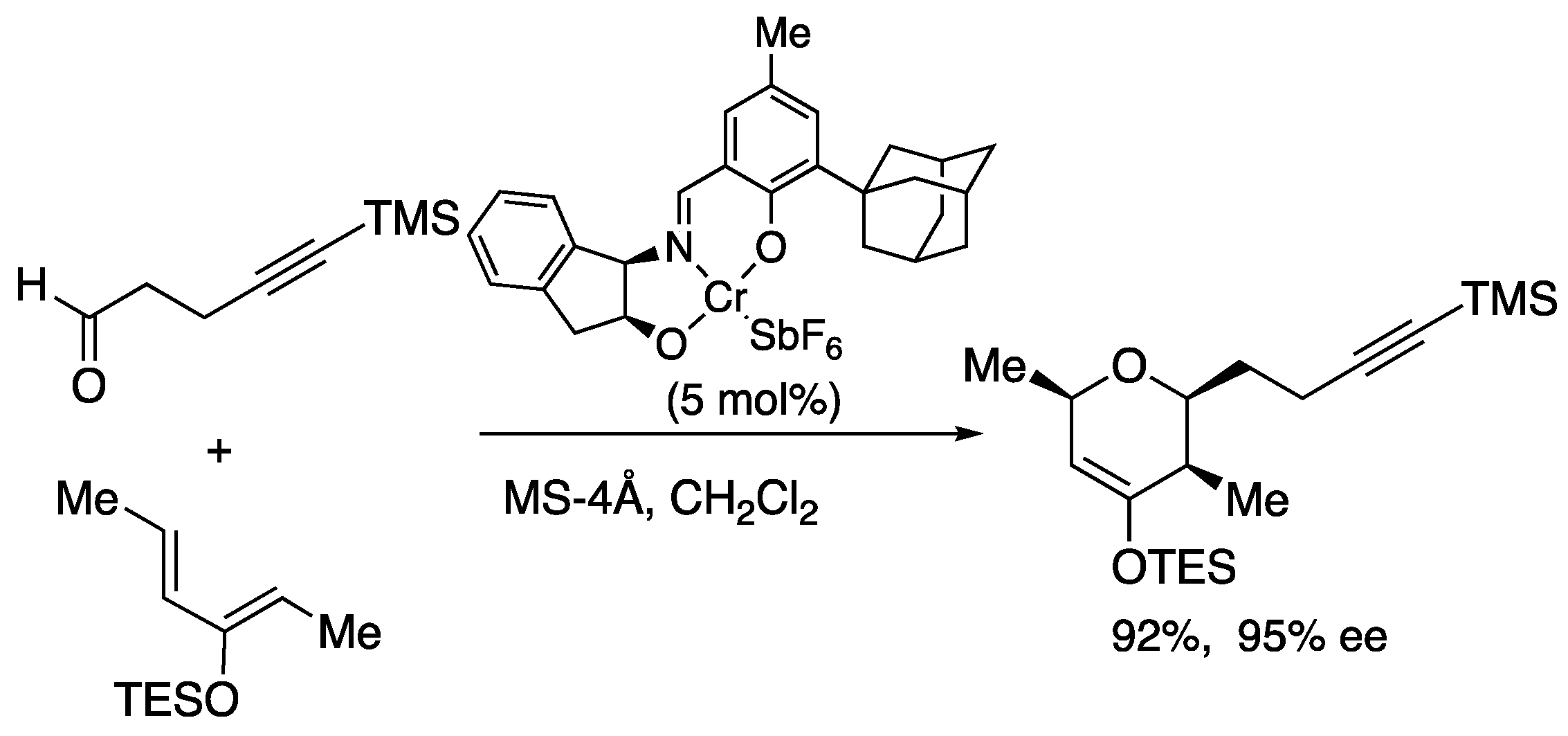
2.2. Magnesium-tethered and aluminum-tethered Diels-Alder Reaction
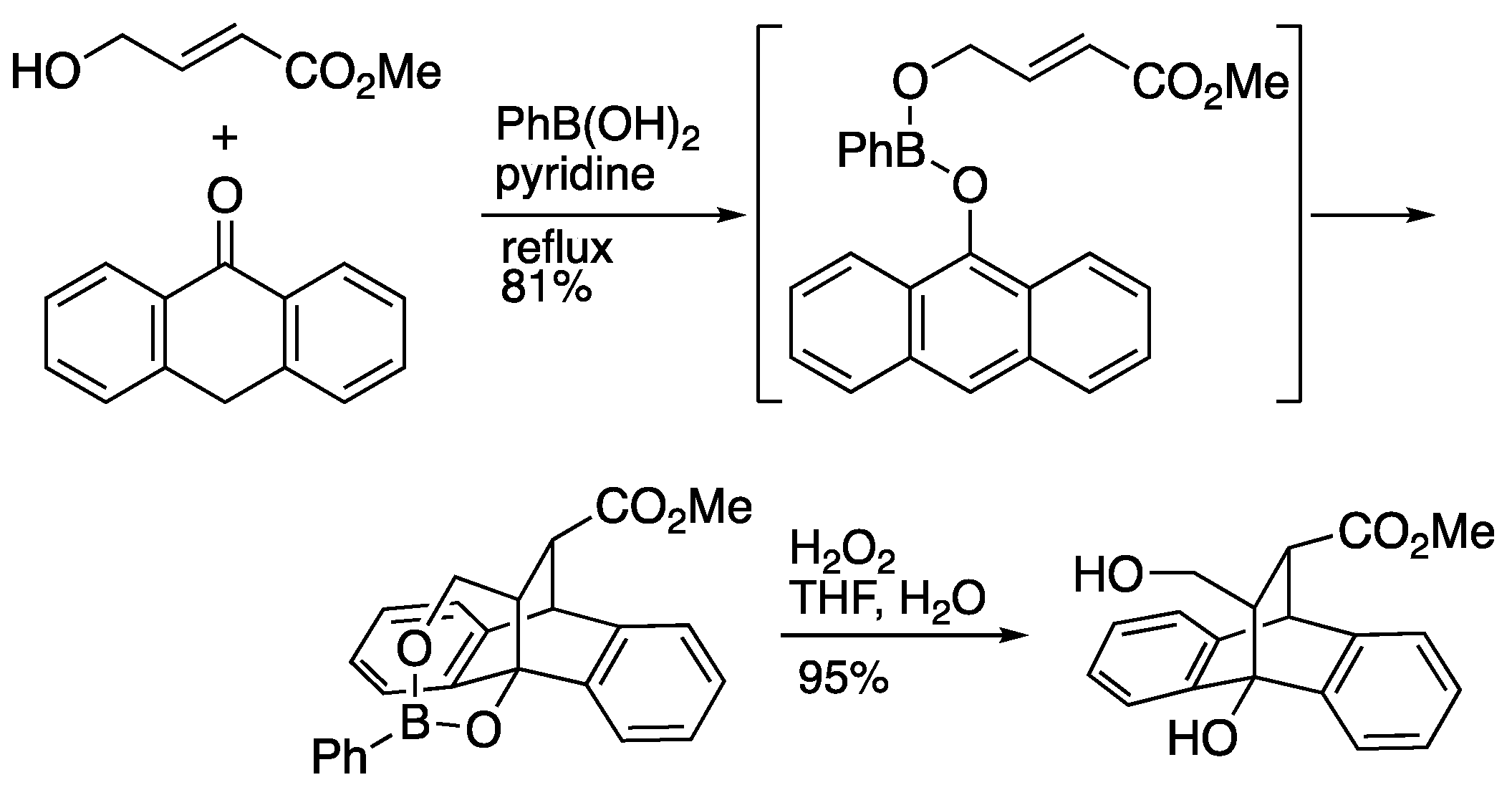
2.3. Boron-tethered Diels-Alder Reaction
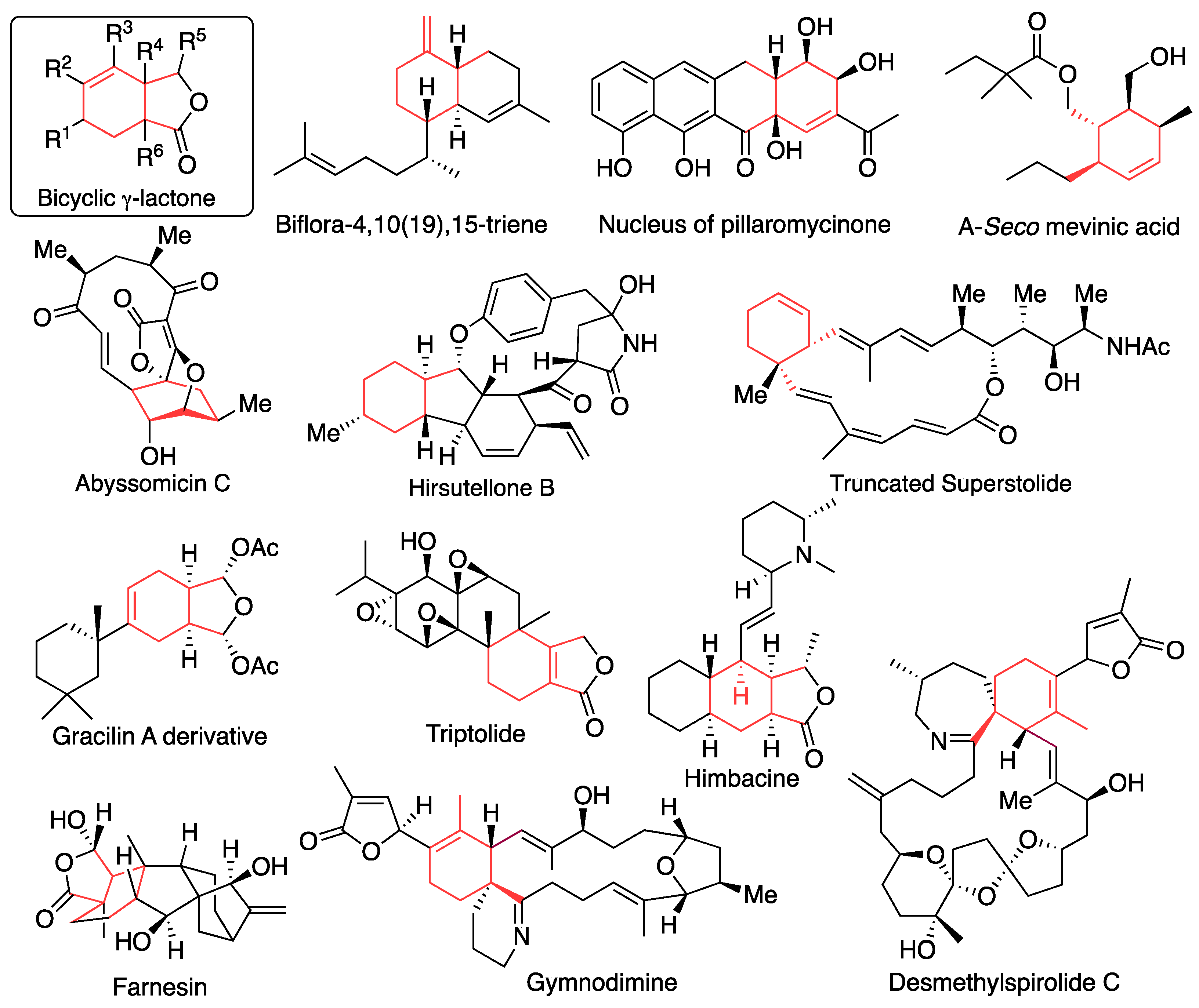
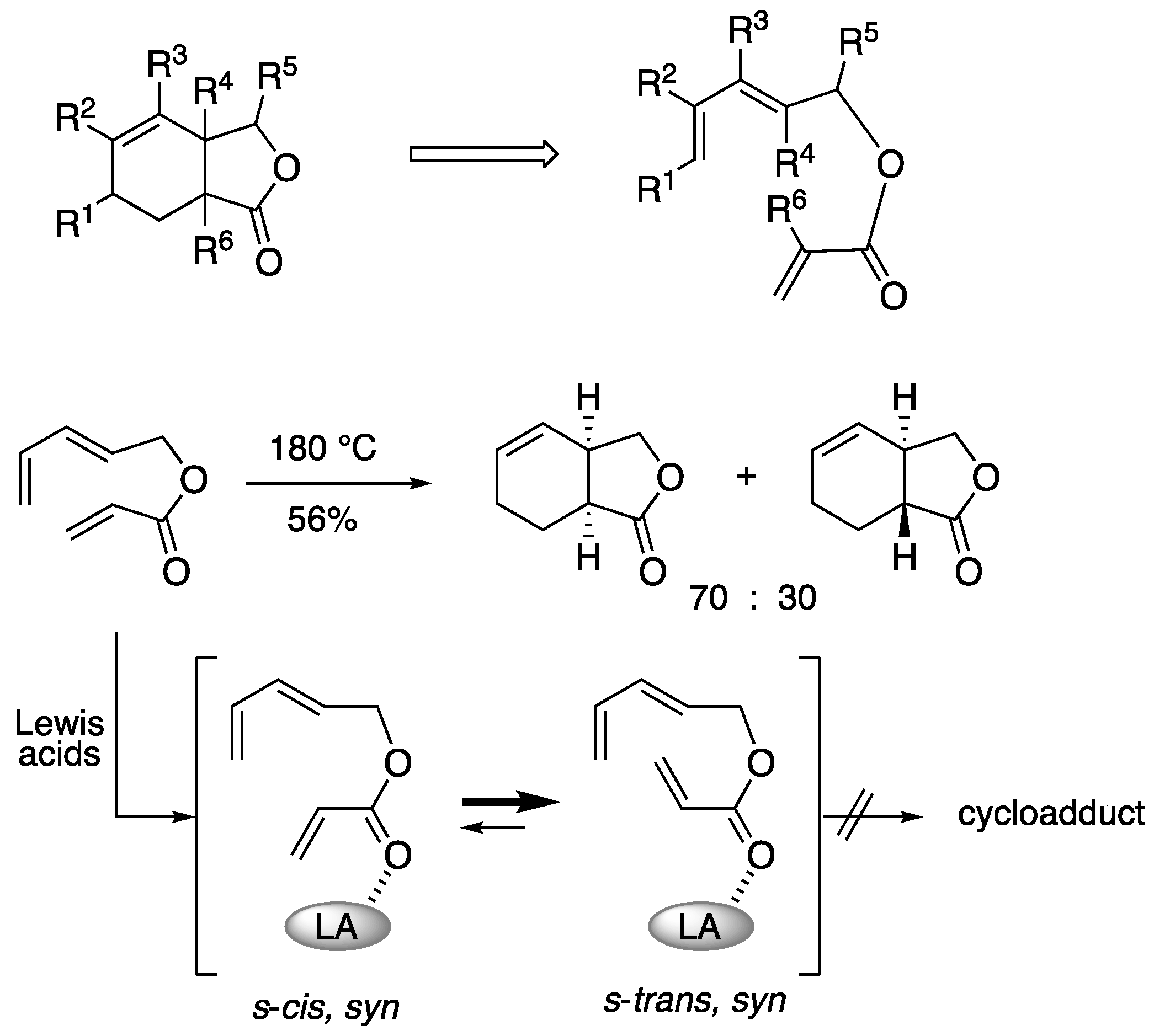
3. Synthesis of bicyclic 𝛾-lactone by intramolecular Diels-Alder reaction
3. Lewis acid template-mediated Diels-Alder reaction.
4. Development of Catalytic Chiral Lewis Acid Template Diels-Alder Reaction
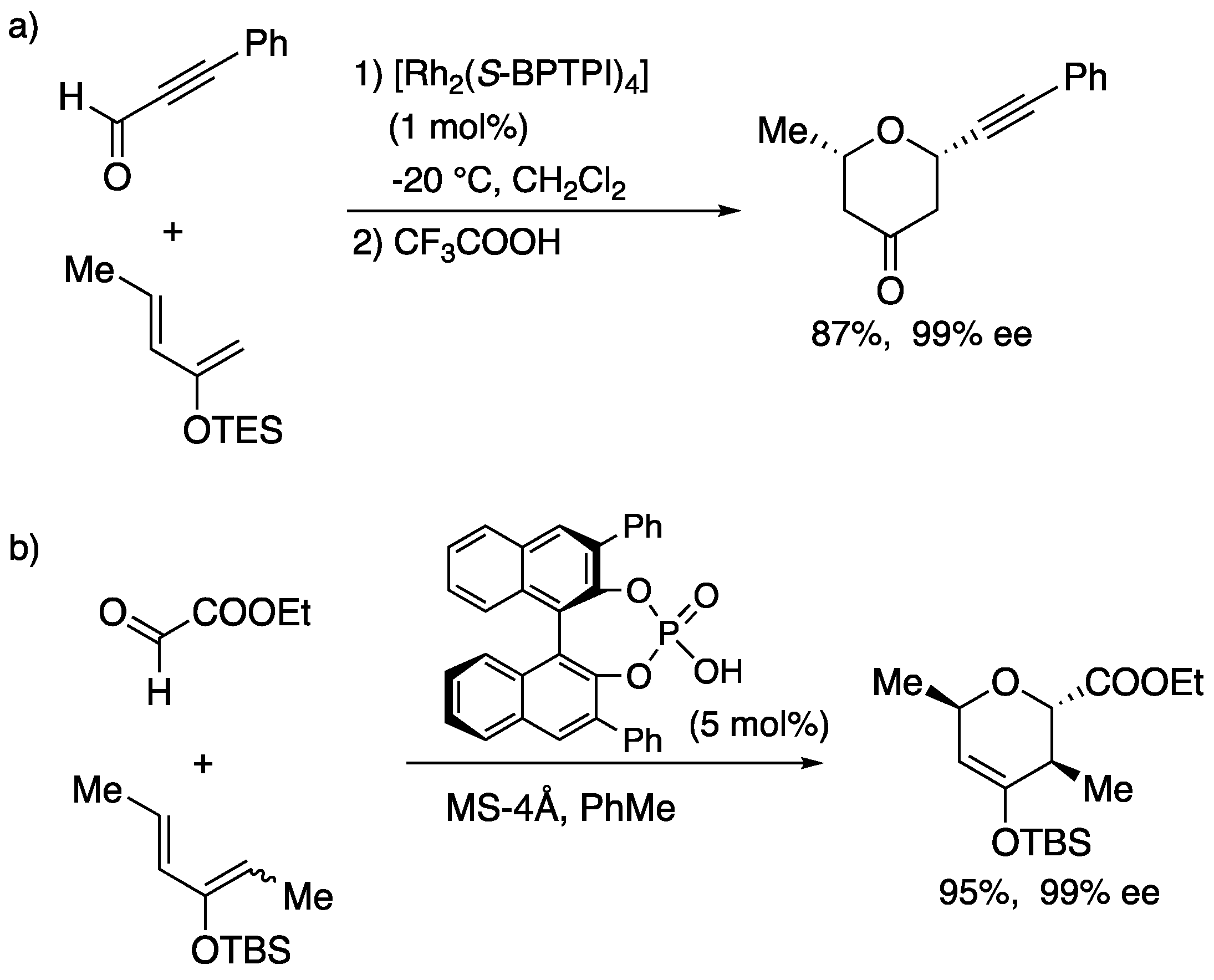
5. Development to the hetero Diels-Alder reaction
6. Application of Lewis acid template-mediated Diels-Alder reaction to biological active natural products
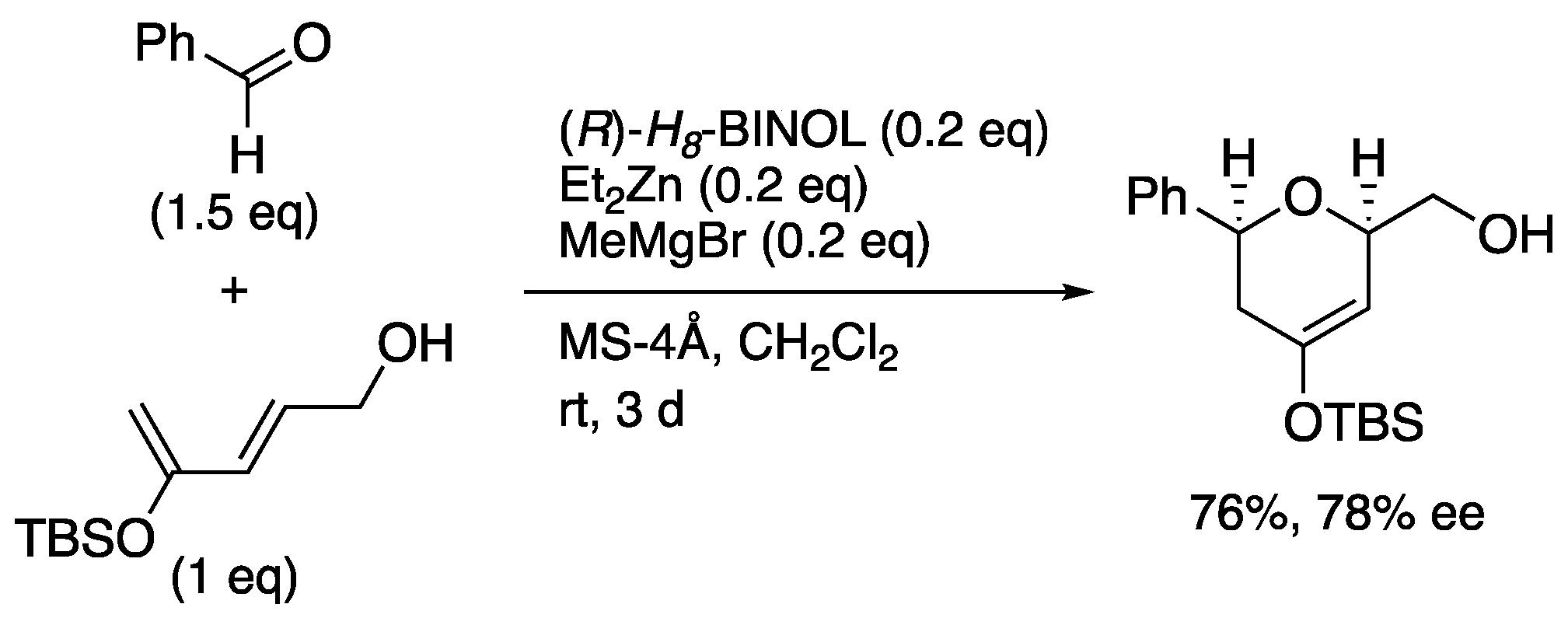
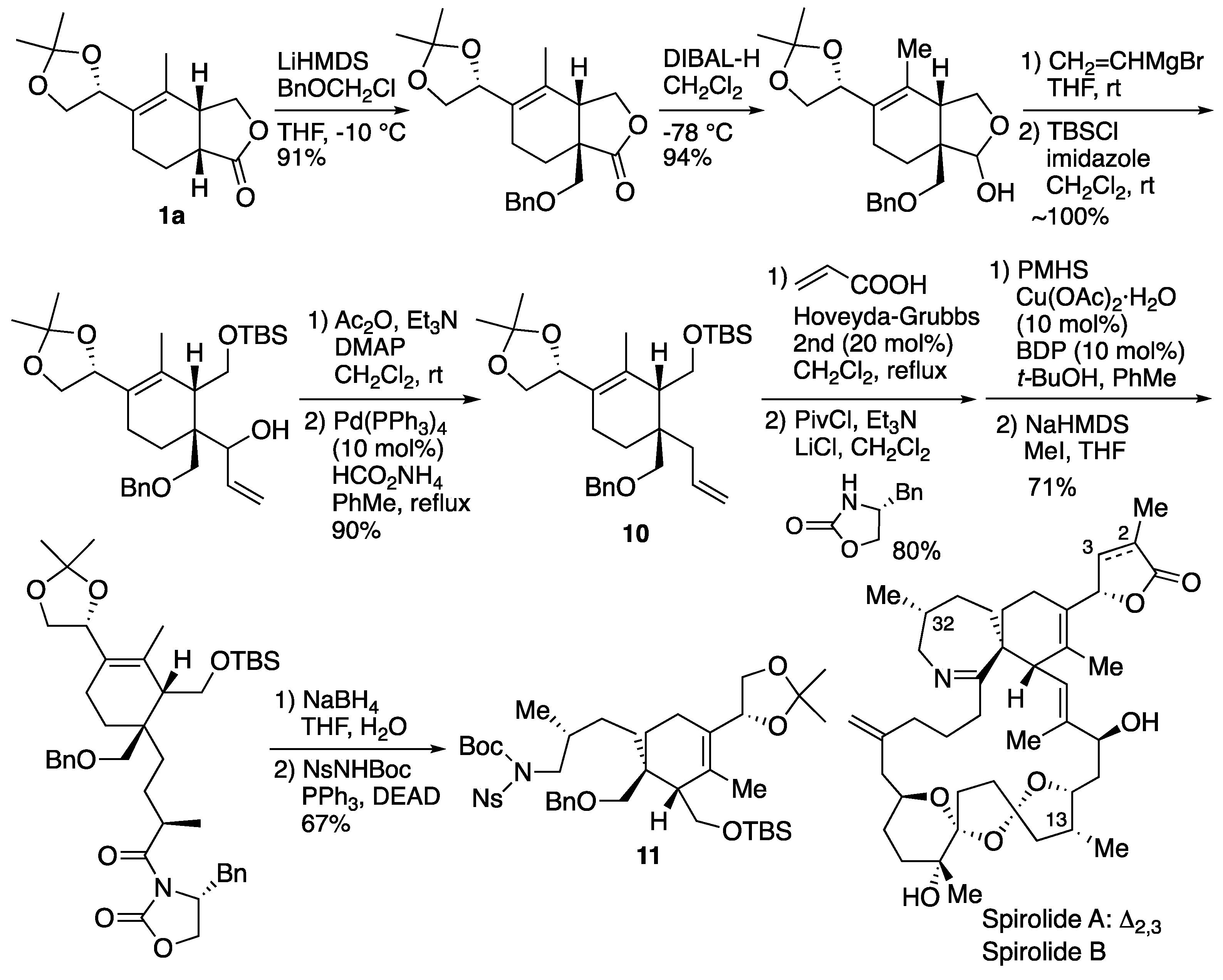
6.2. Ishihara's synthesis of upper segment of spirolide A and B
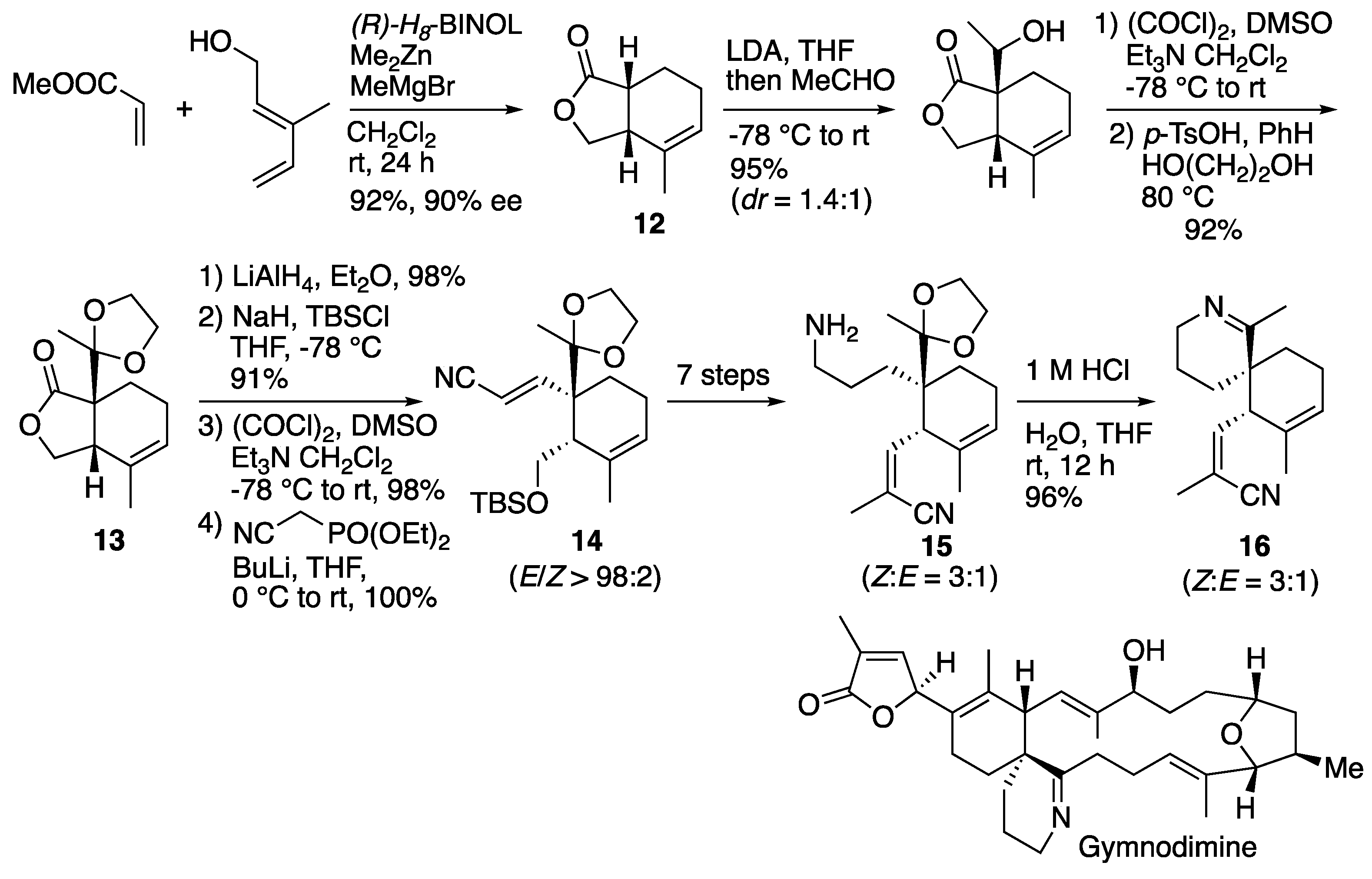
6.2. Landais's synthesis of spirocyclic core of demethylspirolide C and gymnodimine
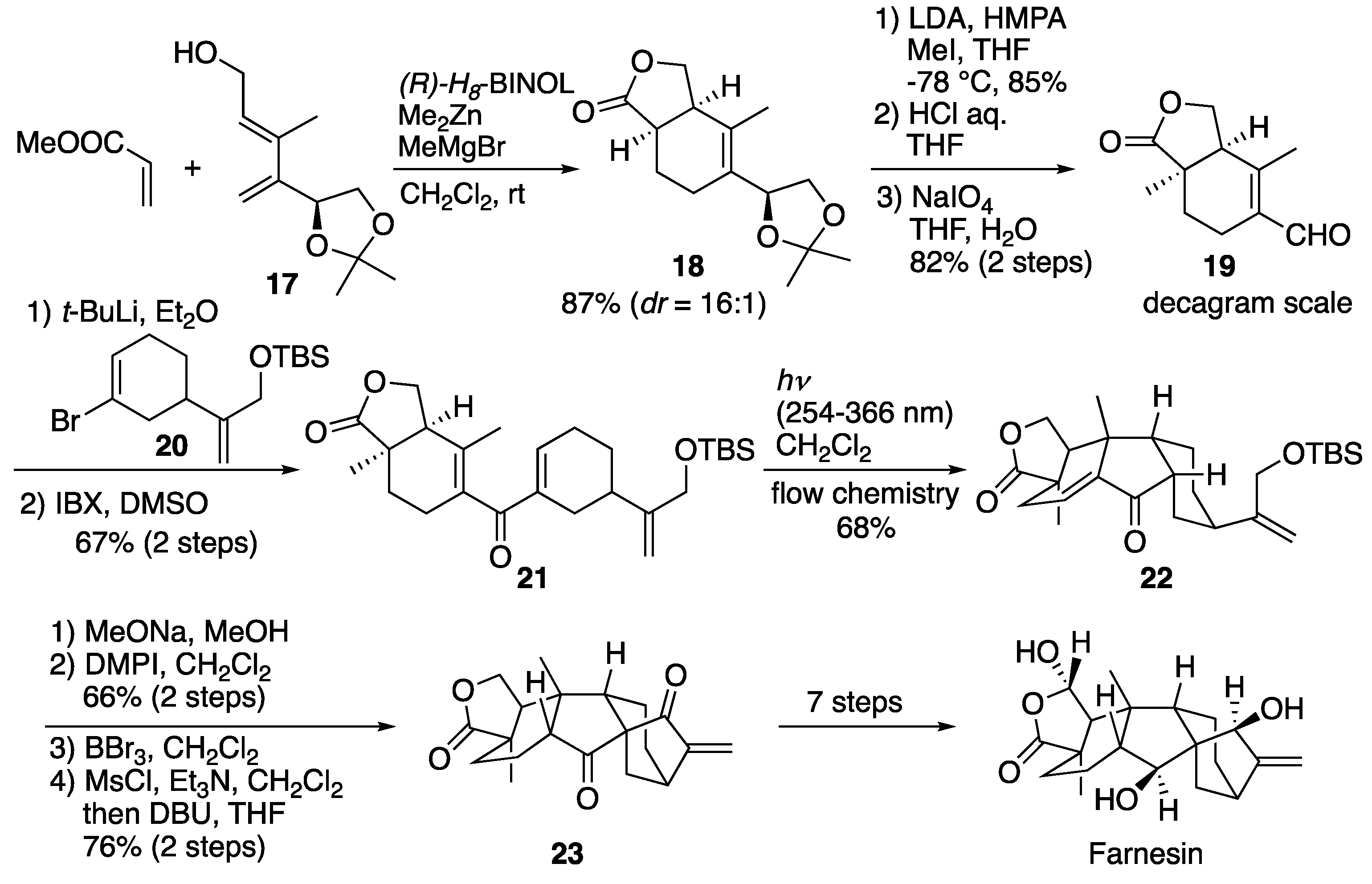
6.3. Gao's total synthesis of farnesin
Conclusion
Acknowledgments
Conflicts of Interest
References
- Newman D. J.; Cragg G. M. Natural Products as Sources of New Drugs over the Nearly Four Decades from 01/1981 to 09/2019. J. Nat. Prod. 2020, 83, 770−803. [CrossRef]
- Atanasov, A. G.; Zotchev, S. B.; Dirsch, V. M. the International Natural Product Sciences Taskforce; Supuran, C. T. Nat. Rev. Drug Discov., 2021, 20, 200–216. [CrossRef]
- Nicolaou, K. C.; Snyder, S. A.; Montagnon T.; Vassilikogiannakis, G. The Diels–Alder Reaction in Total Synthesis. Angew. Chem. Int. Ed. 2002, 41, 1668−1698. [CrossRef]
- Roush, W. R. Intramolecular Diels-Alder Reactions. Comprehensive Organic Synthesis; Trost, B. M., Fleming, I., Paquette, L. A., Eds.; Pergamon: Oxford, 1991; Vol. 5, pp. 513−550.
- Tanner, D.; Ascic, E. Intramolecular Diels-Alder Reactions. Comprehensive Organic Synthesis; 2nd edition; Knochel, P., Molander, G. A. Eds.; Elsevier: Oxford, 2014, Vol. 5, pp. 466−571.
- Craig, D. Stereochemical aspects of the intramolecular Diels–Alder reaction. Chem. Soc. Rev. 1987, 16, 187−238. [CrossRef]
- Fallis, A. G. The intramolecular Diels–Alder reaction: recent advances and synthetic applications. Can. J. Chem. 1984, 62, 183−234. [CrossRef]
- Tamao, K.; Kobayashi, K.; Ito, Y. An intramolecular Diels-Alder reaction of vinylsilanes. J. Am. Chem. Soc., 1989, 111, 6478−6480. [CrossRef]
- Stork G.; Chan, T. Y.; Breault, G. A.; The temporary silicon connection in the control of the regiochemistry of 4 + 2 cycloadditions. J. Am. Chem. Soc., 1992, 114, 7578−7559. [CrossRef]
- Sieburth, S. McN.; Fensterbank, L. An intramolecular Diels-Alder reaction of vinylsilanes. J. Org. Chem., 1992, 57, 5279−5281. [CrossRef]
- Shea, K.; Zandi, K. S.; Staab, A. J.; Carr, R. Disposable tethers in type 2 intramolecular Diels-Alder cycloaddition reactions. Applications in stereochemical control. Tetrahedron Lett., 1990, 31, 5885−5888. [CrossRef]
- Shea, K.; Staab, A. J.; Zandi, K. S. Pericyclic umpolung. Reversal of regioselectivity in the Diels-Alder reaction. Tetrahedron Lett., 1991, 32, 2715−2718. [CrossRef]
- Craig, D.; Reader, J. C. A novel strategy for regio- and stereocontrol in [4 + 2] cycloadditions. Intramolecular diels-alder reaction of a silyl acetal triene. Tetrahedron Lett., 1990, 31, 6585−6588. [CrossRef]
- Gillard, J. W.; Fortin, R.; Grimm, E. L.; Maillard, M.; Tjepkema, M.; Bernstein, M. A.; Glaser, R. The unsymmetrical silaketal as a neutral, removable tether for effecting intramolecular Diels-Alder reactions. Tetrahedron Lett., 1991, 32, 1145−1148. [CrossRef]
- L. Fensterbank, M. Malacria, S. McN. Sieburth, Intramolecular Reactions of Temporarily Silicon-Tethered Molecules. Synthesis, 1997, 813−854. [CrossRef]
- Bols, M.; Skrydstrup, T. Silicon-Tethered Reactions. Chem. Rev., 1995, 95, 1253-1277. [CrossRef]
- G. Stork, T. Y. Chan, Temporary Magnesium and Aluminum Connections in [4 + 2] Cycloadditions. J. Am. Chem. Soc., 1995, 117, 6595−6596. [CrossRef]
- K. Narasaka, S. Shimada, K. Osoda, N. Iwasawa, Phenylboronic Acid as a Template in the Diels-Alder Reaction, Synthesis, 1991, 1171−1172. [CrossRef]
- Grieco, P. A.; Ravi P. Nargund, R. P. Synthetic studies on diterpenes from a termite soldier: Total synthesis of (±)-biflora-4,10(19),15-triene. Tetrahedron Lett. 1986, 27, 4813−4816. [CrossRef]
- White, J. D.; Nolen, E. G., Jr.; Miller, C. H. Stereochemical transcription via the intramolecular Diels-Alder reaction. Enantioselective synthesis of the nucleus of (+)-pillaromycinone. J. Org. Chem. 1986, 51, 1150−1155 . [CrossRef]
- White, J. D.; Demnitz, F. W. J.; Xu, Q.; Martin, W. H. C. Synthesis of an Advanced Intermediate for (+)-Pillaromycinone. Staunton-Weinreb Annulation Revisited. Org. Lett. 2008, 10, 2833−2836. [CrossRef]
- Arseniyadis, S.; Brondi-Alves, R.; Yashunsky,D. V.; Potier, P.; Toupet, L.Formal total synthesis of an A-seco mevinic acid. Tetrahedron, 1997, 53, 1003−1014. [CrossRef]
- Nicolaou, K. C.; Chen, J. S. Abyssomicin C and atrop-Abysomicin C. Classics in Total Synthesis III: Further Targets, Strategies, Methods; Wiley-VCH: Weinheim, 2011; pp. 320−343.
- Nicolaou, K. C.; Harrison, S. T. Total Synthesis of Abyssomicin C and atrop-Abyssomicin C. Angew. Chem., Int. Ed. 2006, 45, 3256−3260. [CrossRef]
- Nicolaou, K. C.; Harrison, S. T. Total Synthesis of Abyssomicin C, Atrop-abyssomicin C, and Abyssomicin D: Implications for Natural Origins of Atrop-abyssomicin C. J. Am. Chem. Soc. 2007, 129, 429−440. [CrossRef]
- Rath, J.-P.; Kinast, S.; Maier, M. E. Synthesis of the Fully Functionalized Core Structure of the Antibiotic Abyssomicin C. Org. Lett. 2005, 7, 3089–3092. [CrossRef]
- Halvorsen, G. T.; Roush, W. R. Stereoselective synthesis of the decahydrofluorene core of the hirsutellones. Tetrahedron Lett. 2011, 52, 2072-2075. [CrossRef]
- Chen, L.; Ahmed, K. B. R.; Huang, P.; Jin, Z. Design, Synthesis, and Biological Evaluation of Truncated Superstolide A. Angew. Chem., Int. Ed. 2013, 52, 3446−3449. [CrossRef]
- Abbasov, M. E., Alvariño, R., Chaheine, C. M. Alonso, E.; Sánchez, J. A.; Conner, M. L.; Alfonso, A.; Jaspars, M.; Botana, L. M. Romo, D. Simplified immunosuppressive and neuroprotective agents based on gracilin A. Nat. Chem. 2019, 11, 342–350. [CrossRef]
- Miller, N. A.; Willis, A. C.; Sherburn, M. S. Formal total synthesis of triptolide. Chem. Commun., 2008, 1226−1228. [CrossRef]
- Wong, L. S.-M.; Sherburn, M. S. IMDA-Radical Cyclization Approach to (+)-Himbacine, Org. Lett. 2003, 5, 3603–3606. [CrossRef]
- Que, Y.; Shao, H.; He, H.; Gao, S. Total Synthesis of Farnesin through an Excited-State Nazarov Reaction. Angew. Chem., Int. Ed. 2020, 59, 7444−7449. [CrossRef]
- Guthertz, A.; Lusseau, J.; Desvergnes, V.; Massip, S. ; Landais Y. An Approach towards the Synthesis of the Spiroimine Fragment of 13-Desmethylspirolide C and Gymnodimine A. Chem. Eur. J. 2019, 25, 1553−1560. [CrossRef]
- Ishihara, J.; Usui, F.; Kurose, T.; Baba, T.; Kawaguchi, Y.; Watanabe, Y.; Hatakeyama, S. Synthetic Studies on Spirolides A and B: Formation of the Upper Carbon Framework Based on a Lewis Acid Template-Catalyzed Diels–Alder Reaction. Chem. Eur. J. 2019, 25, 1543−1552. [CrossRef]
- Cayzer, T. N. ; Wong, L. S.-M.; Turner, P.; Paddon-Row, M. N.; Sherburn, M. S. Optimising Stereoselectivity in Intramolecular Diels-Alder Reactions of Pentadienyl Acrylates: Synthetic and Computational Investigations into the ‘Steric Directing Group’ Approach. Chem. Eur. J. 2002, 8, 739−750. [CrossRef]
- Smith, D. A.; Sakan, K.; Houk, K. N. Stereoselectivities of thermal and Lewis acid catalyzed intramolecular Diels-Alder reactions of internally activated dienophiles to form 5–11 membered rings. Tetrahedron Lett., 1986, 27, 4877−4880. [CrossRef]
- Bienaymé, H. Angew. Chem. Int. Ed. Engl. 1997, 36, 2670−2673. Enantioselective Diels–Alder Cycloaddition by Preorganization on a Chiral Lewis Acid Template . [CrossRef]
- Bienaymé, H.; Longeau, A. Internally Lewis acid-catalyzed Diels-Alder cycloadditions. Tetrahedron, 1997, 53, 9637−9646. [CrossRef]
- Ward, D. E.; Abaee, M. S. Intramolecular Diels−Alder Reaction by Self-Assembly of the Components on a Lewis Acid Template. Org. Lett. 2000, 2, 3937−3940. [CrossRef]
- Zografos, A. L.; Yiotakis, A.; Georgiadis, D. Rapid Access to the Tricyclic Spirotetronic Core of Abyssomicins. Org. Lett. 2005, 7, 4515–4518. [CrossRef]
- D. E. Ward, M. S. Souweha, Catalytic Enantioselective Diels−Alder Reaction by Self-Assembly of the Components on a Lewis Acid Template. Org. Lett., 2005, 7, 3533−3536. [CrossRef]
- Souweha, M. S.; Arab, A. ApSimon, M.; Fallis, A. G. Diene-Transmissive Cycloadditions: Control of Monocycloaddition by Self-Assembly on a Lewis Acid Template. Org. Lett. 2007, 9, 615–618 . [CrossRef]
- Ishihara, J.; Nakadachi, S. Watanabe, Y. Hatakeyama, S. Lewis Acid Template-Catalyzed Asymmetric Diels-Alder Reaction. J. Org. Chem., 2015, 80, 2037−2041. [CrossRef]
- Jørgensen, K. A. Catalytic Enantioselective Cycloaddition Reactions of Carbonyl Compounds. Cycloaddition Reactions in Organic Synthesis; Kobayashi, S., Jørgensen. K. A. Eds.; Wiley-VCH, Weinheim, 2002, pp. 151−185. [CrossRef]
- Ishihara, K.; Sakakura, A. Hetero-Diels-Alder Reactions. Comprehensive Organic Synthesis; 2nd edition; Knochel, P., Molander, G. A. Eds.; Elsevier: Oxford, 2014, Vol. 5, pp. 409−465.
- Dossetter, A. G.; Jamison, T. F.; Jacobsen, E. N. Highly Enantio- and DiastereoselectiveHetero-Diels-Alder Reactions Catalyzed by New Chiral Tridentate Chromium (iii) Catalysts. Angew. Chem., Int. Ed. 1999, 38, 2398−2400. [CrossRef]
- Thompson, C. F.; Jamison, T. F.; Jacobsen, E. N. FR901464: Total Synthesis, Proof of Structure, and Evaluation of Synthetic Analogues. J. Am. Chem. Soc. 2001, 123, 41, 9974–9983. [CrossRef]
- Anada, M.; Washio, T.; Shimada, N.; Kitagaki S.; Nakajima, M.; Shiro, M.; Hashimoto, S. A New Dirhodium(II) Carboxamidate Complex as a Chiral Lewis Acid Catalyst for Enantioselective Hetero-Diels–Alder Reactions. Angew. Chem. Int. Ed., 2004, 43, 2665−2668. [CrossRef]
- Momiyama, N.; Tabuse H.; Terada, M. Chiral Phosphoric Acid-Governed Anti-Diastereoselective and Enantioselective Hetero-Diels−Alder Reaction of Glyoxylate. J. Am. Chem. Soc., 2009, 131, 12882−12883. [CrossRef]
- Ishihara, J.; Ohzono, Y.; Oka, K.; Urayama, Y.; Hatakeyama, S. Bimetallic Lewis Acid Template-Mediated Enantioselective Hetero-Diels-Alder Reactions of 4-Siloxy-2,4-pentadienols. Heterocycles, 2019, 99, 1330−1341. [CrossRef]
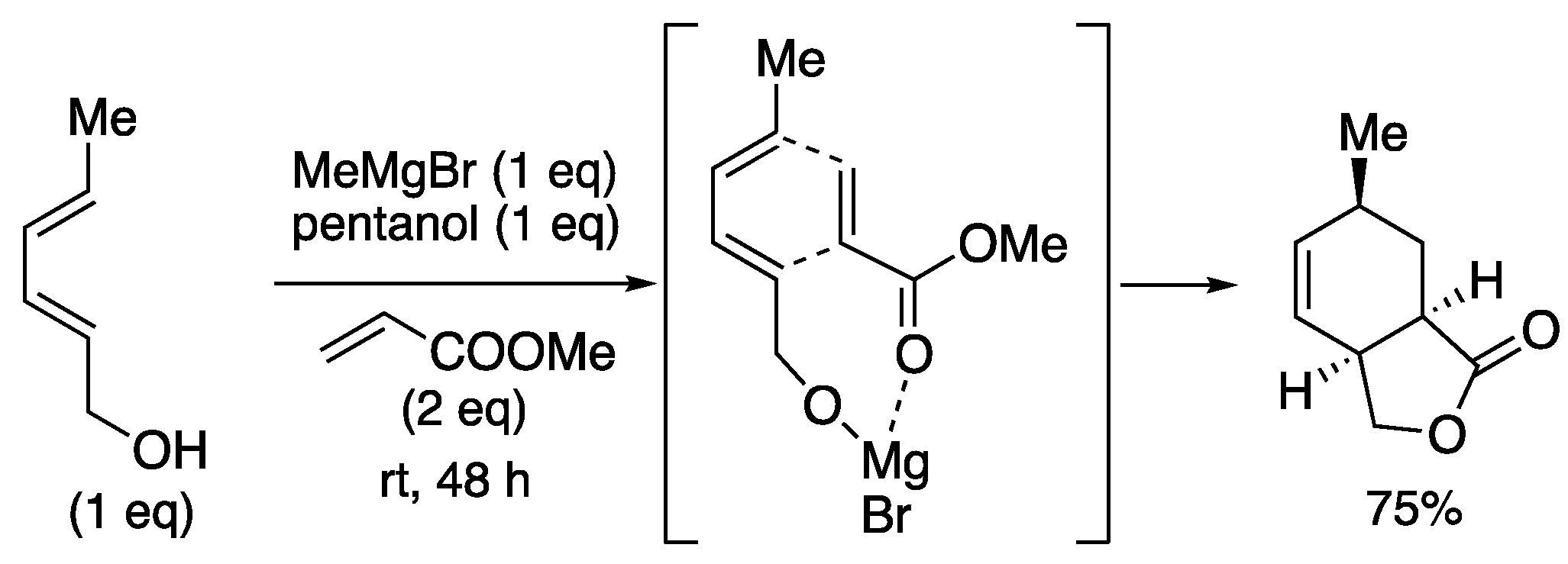
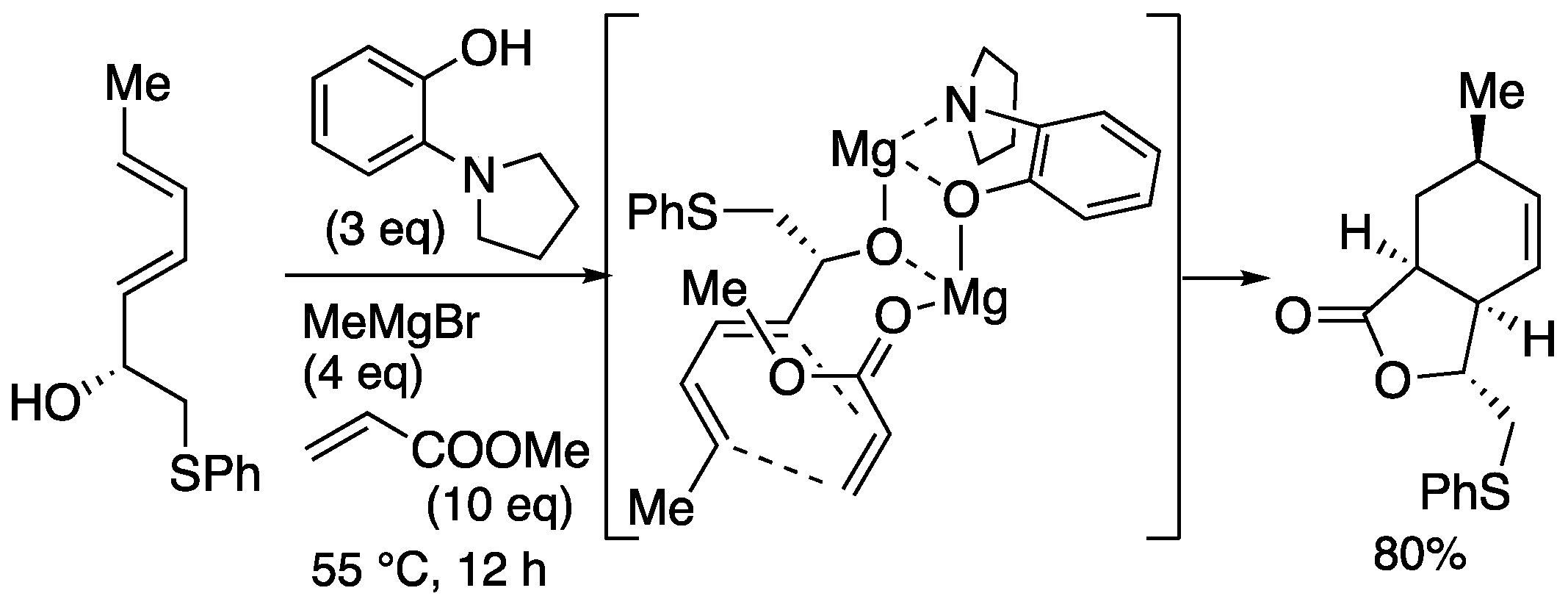
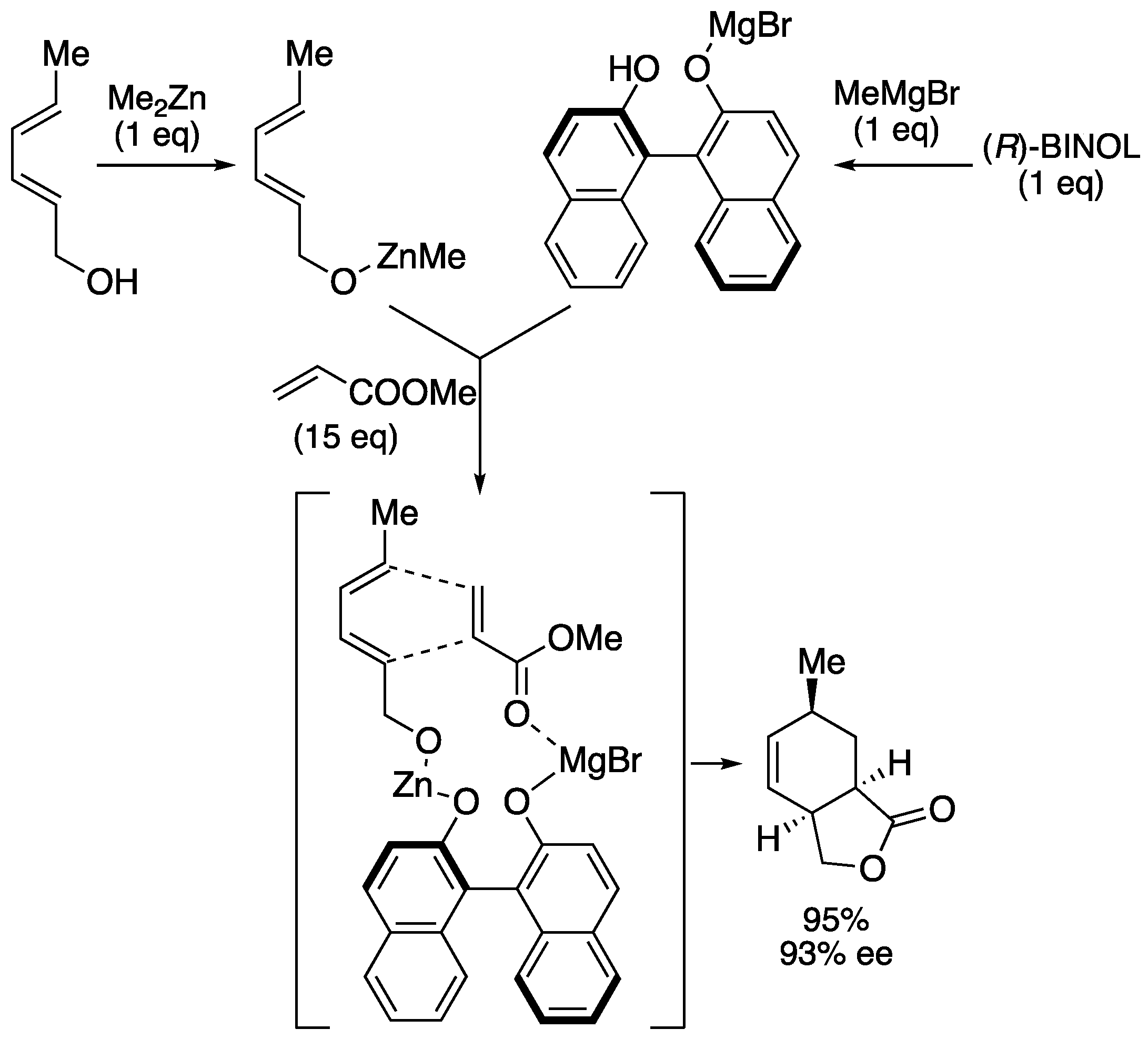
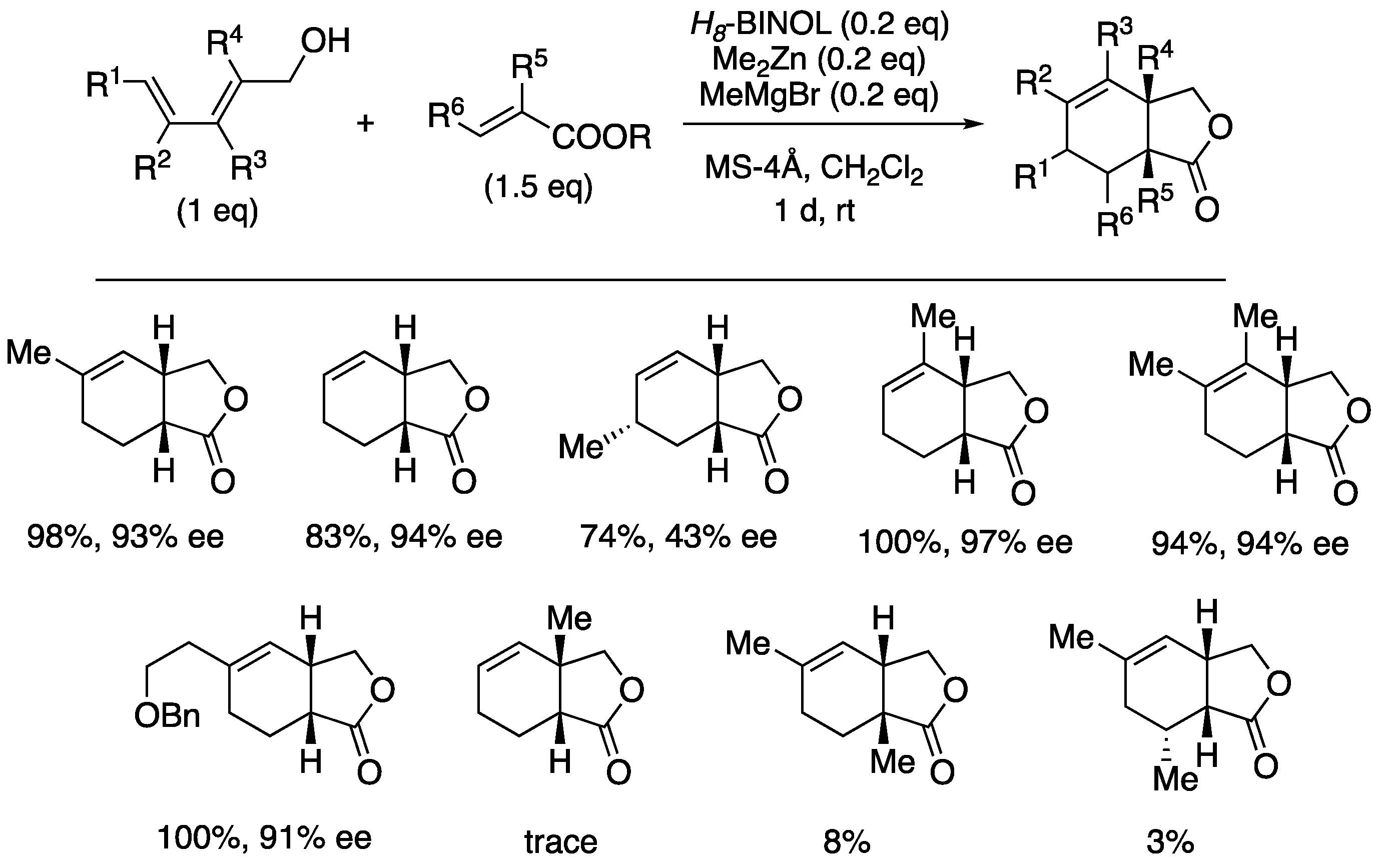
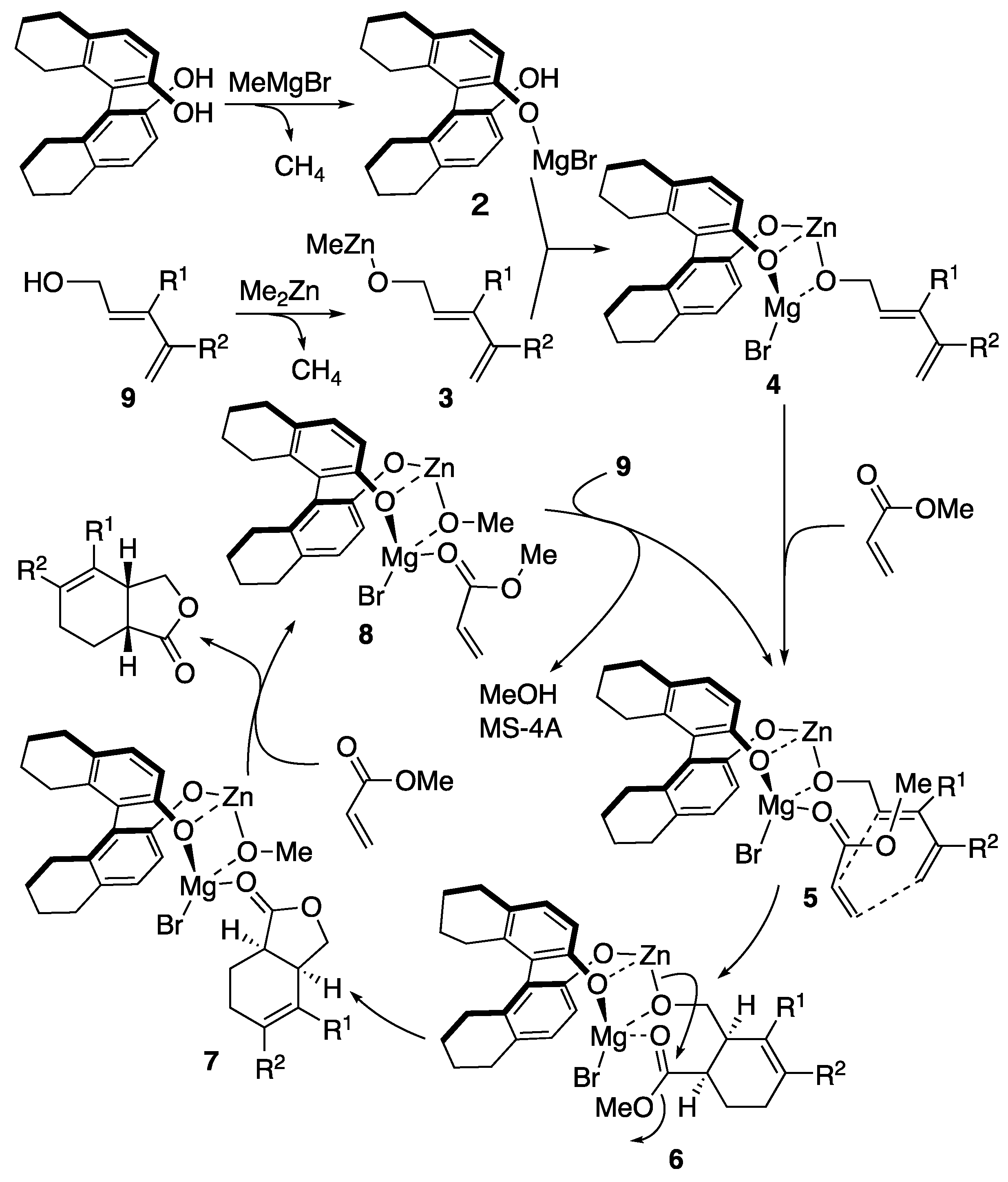
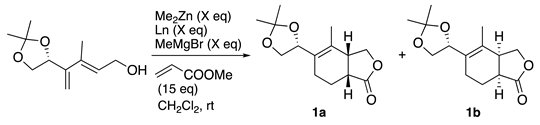
| entry | Ln | X (eq) | time | yield | ratio (1a : 1b) |
| 1 | (R)-BINOL | 1.0 | 3.5 h | 100% | 4.8 : 1 |
| 2 | (S)-BINOL | 1.0 | 24 h | 80% | 1 : 3.7 |
| 3 | (R)-3,3‘-Br2-BINOL | 1.0 | 20 h | 94% | 4.6 : 1 |
| 4 | (R)-H8-BINOL | 1.0 | 24 h | 98% | 9.9 : 1 |
| 5 | (R)-H8-BINOL | 0.5 | 2.5 h | 99% | 10 : 1 |
| 6 | (R)-H8-BINOL | 0.2 | 4 h | 96% | 10 : 1 |
| 7 | (R)-H8-BINOL | 0.1 | 24 h | 98% | 8 : 1 |
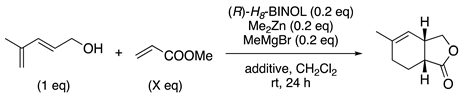
| entry | acrylate ( x eq) | additive | yield | % ee |
| 1 | 15 | - | 83% | 52%ee |
| 2 | 15 | MS 4 Å | 100% | 84%ee |
| 3 | 5 | MS 4 Å | 100% | 88%ee |
| 4 | 2.5 | MS 4 Å | 100% | 92%ee |
| 5 | 1.5 | MS 4 Å | 98% | 93%ee |
| 6 | 1.5 | MS 5 Å | 100% | 95%ee |
| 7 | 1.5 | MS 13 X | NR | - |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




