Submitted:
18 December 2023
Posted:
03 January 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Experimental
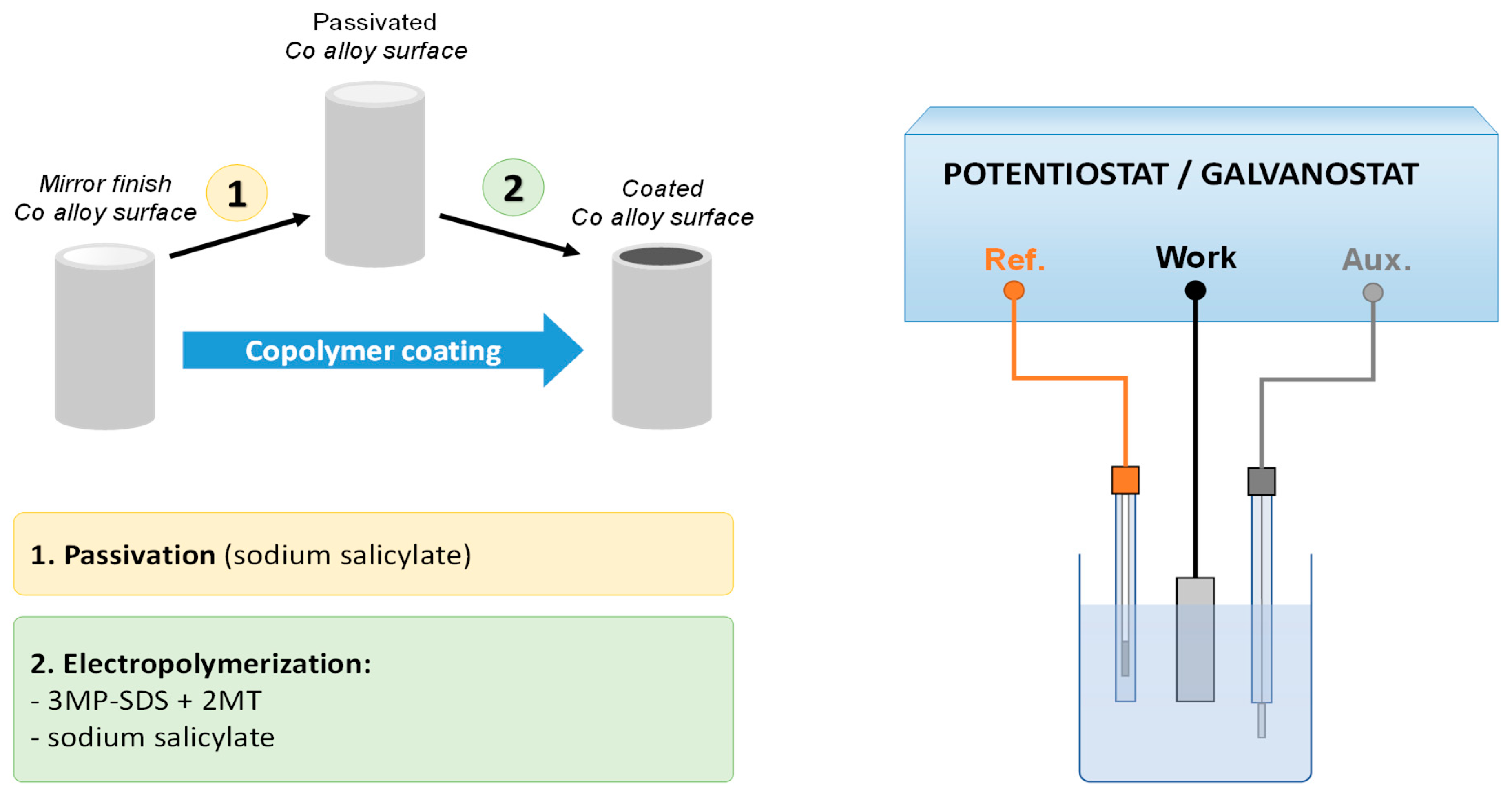
2.1. Materials and Methods
2.2. Instruments
3. Results and Discussion
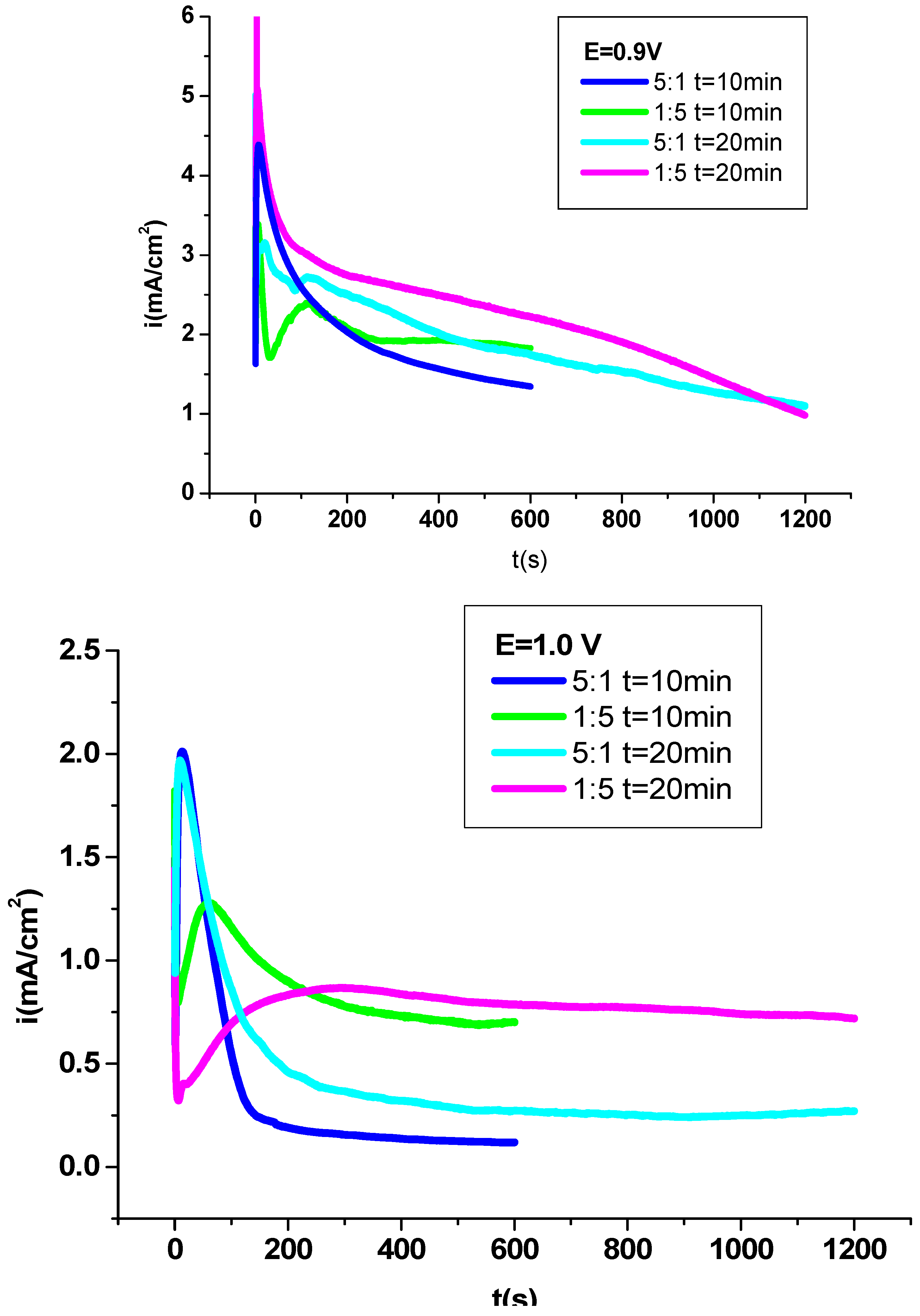
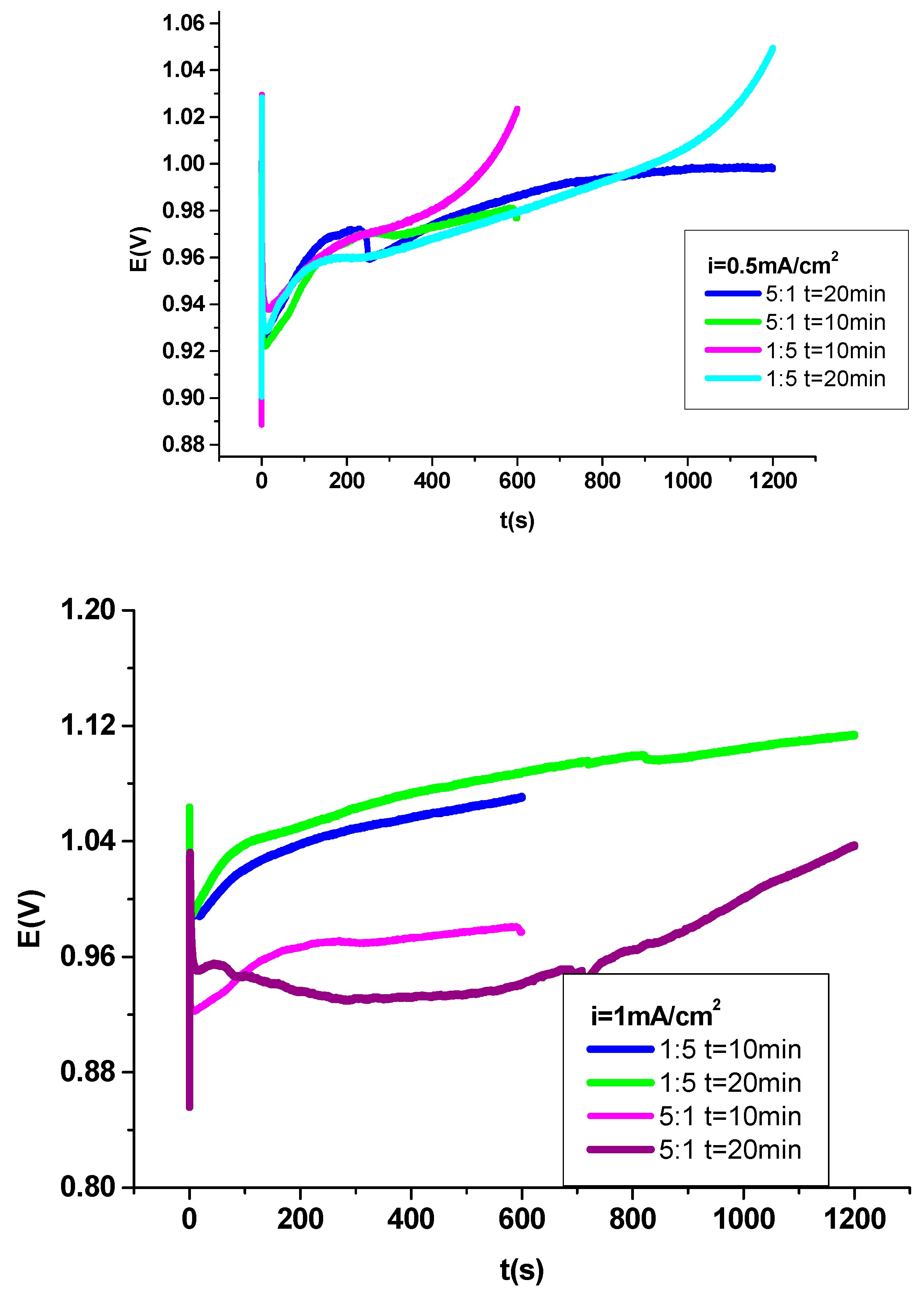
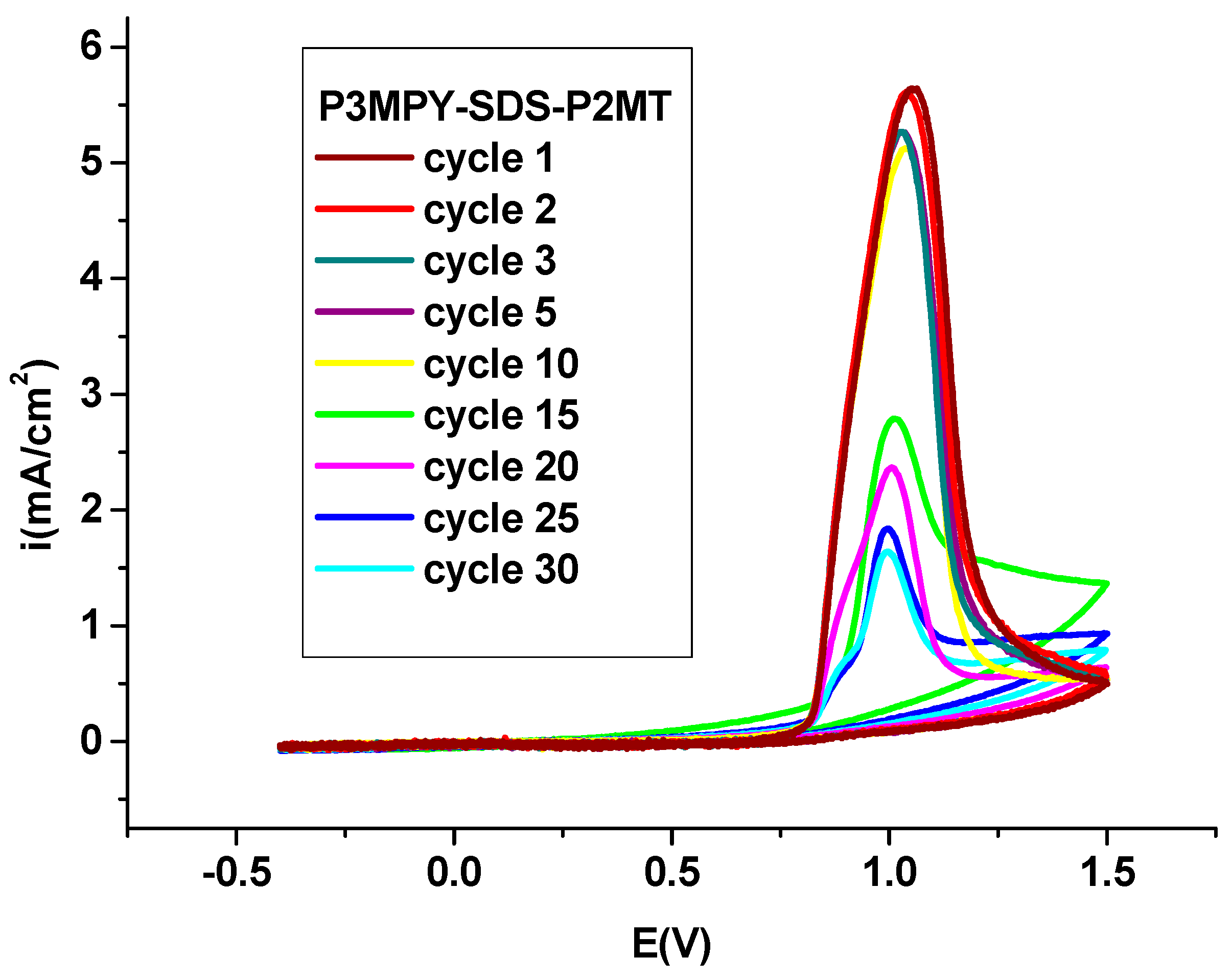
3.1. Electrochemical deposition of P3MPY-SDS/P2MT coating on cobalt alloy
3.2. Electrochemical exploration P3MPY-SDS/P2MT composite coating
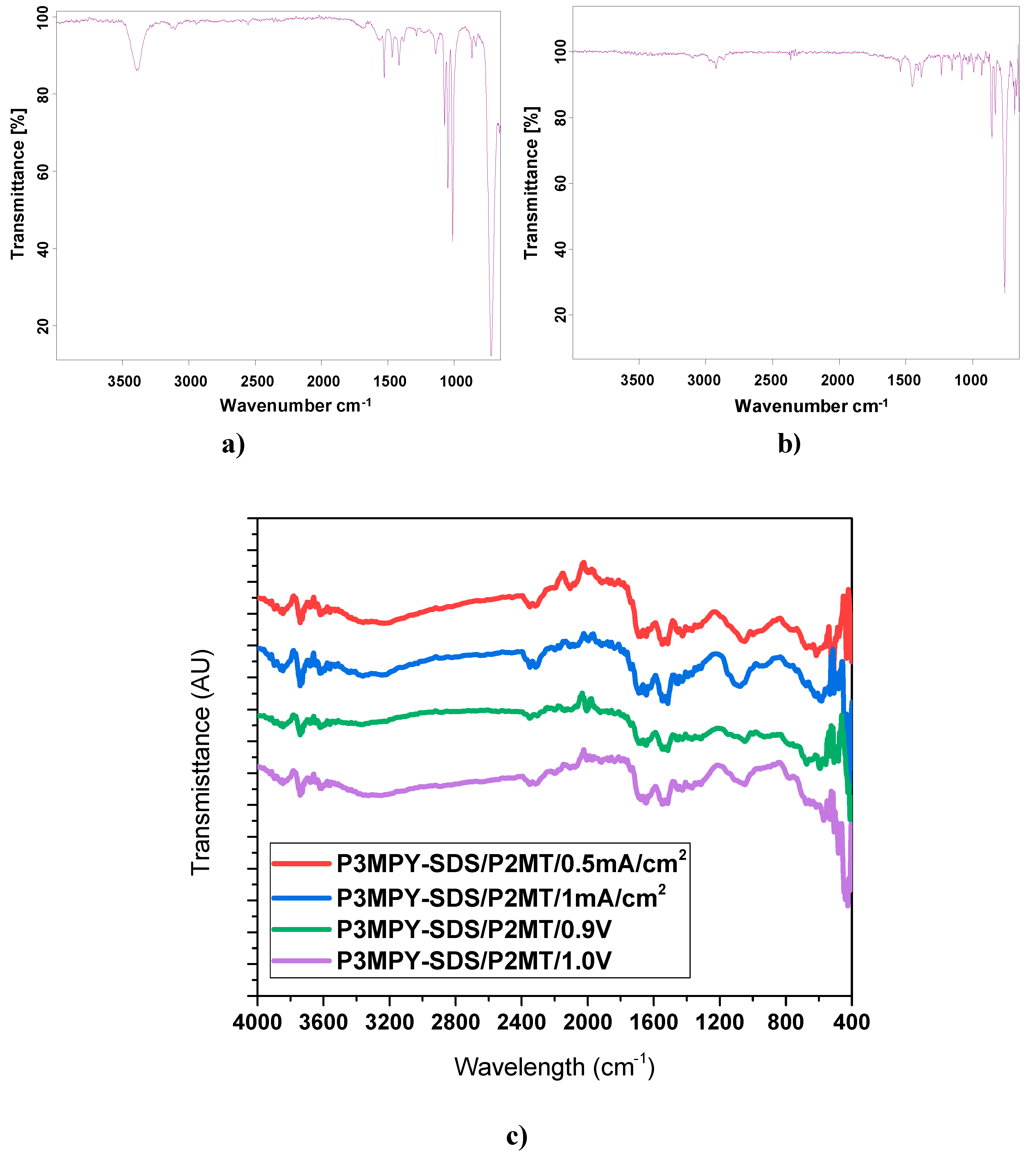
3.3. FT-IR Evaluations
3.4. Electrochemical investigation
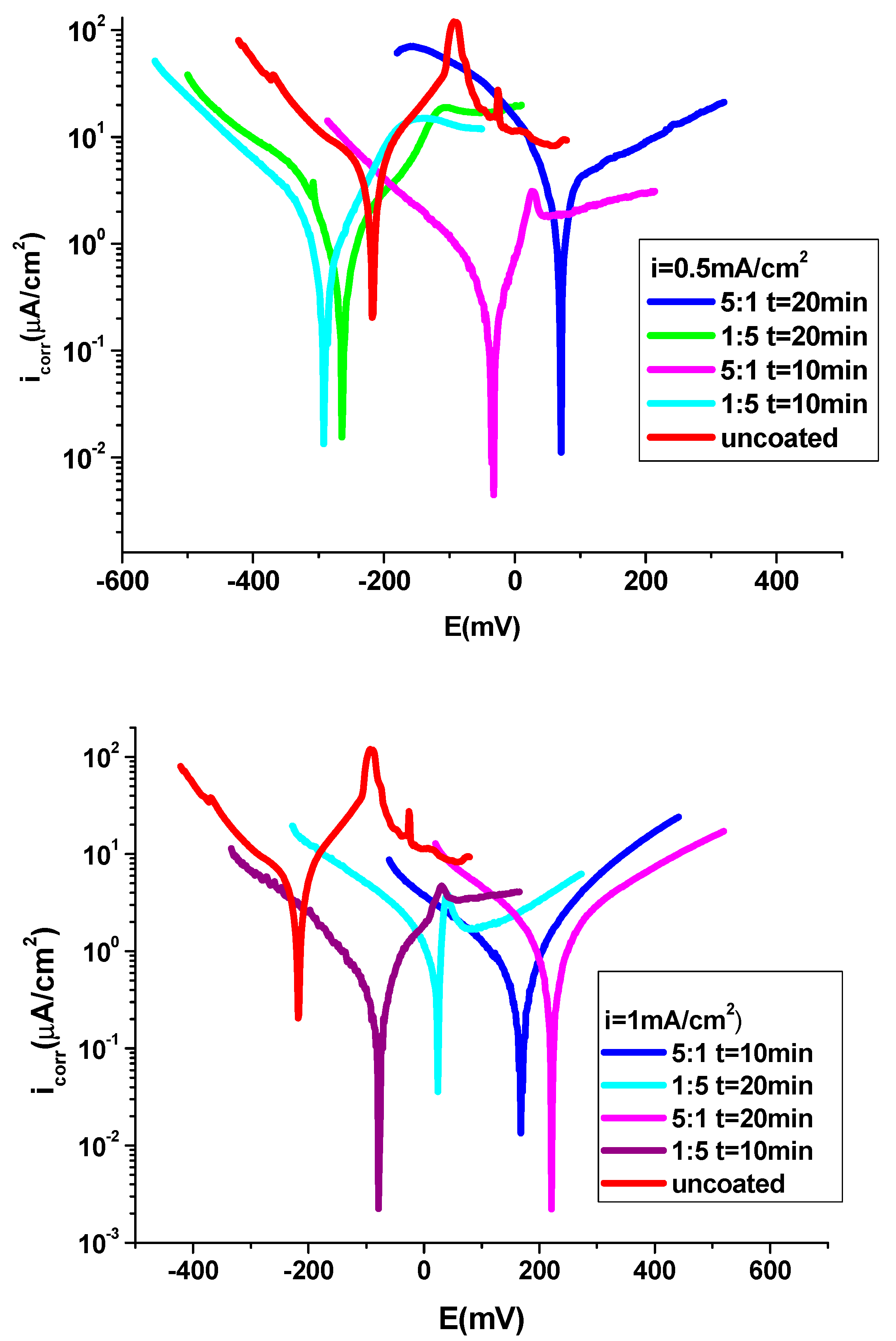
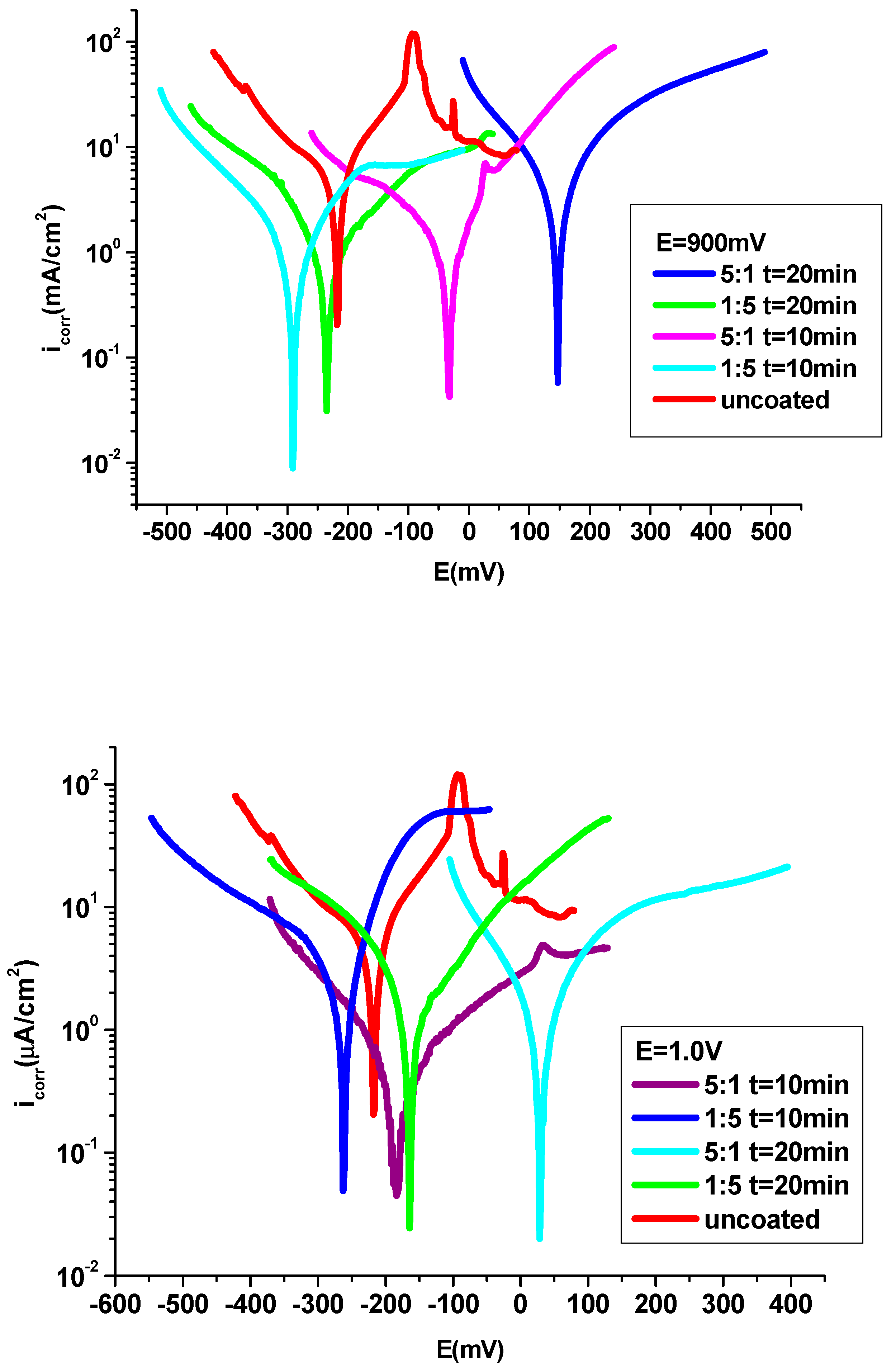
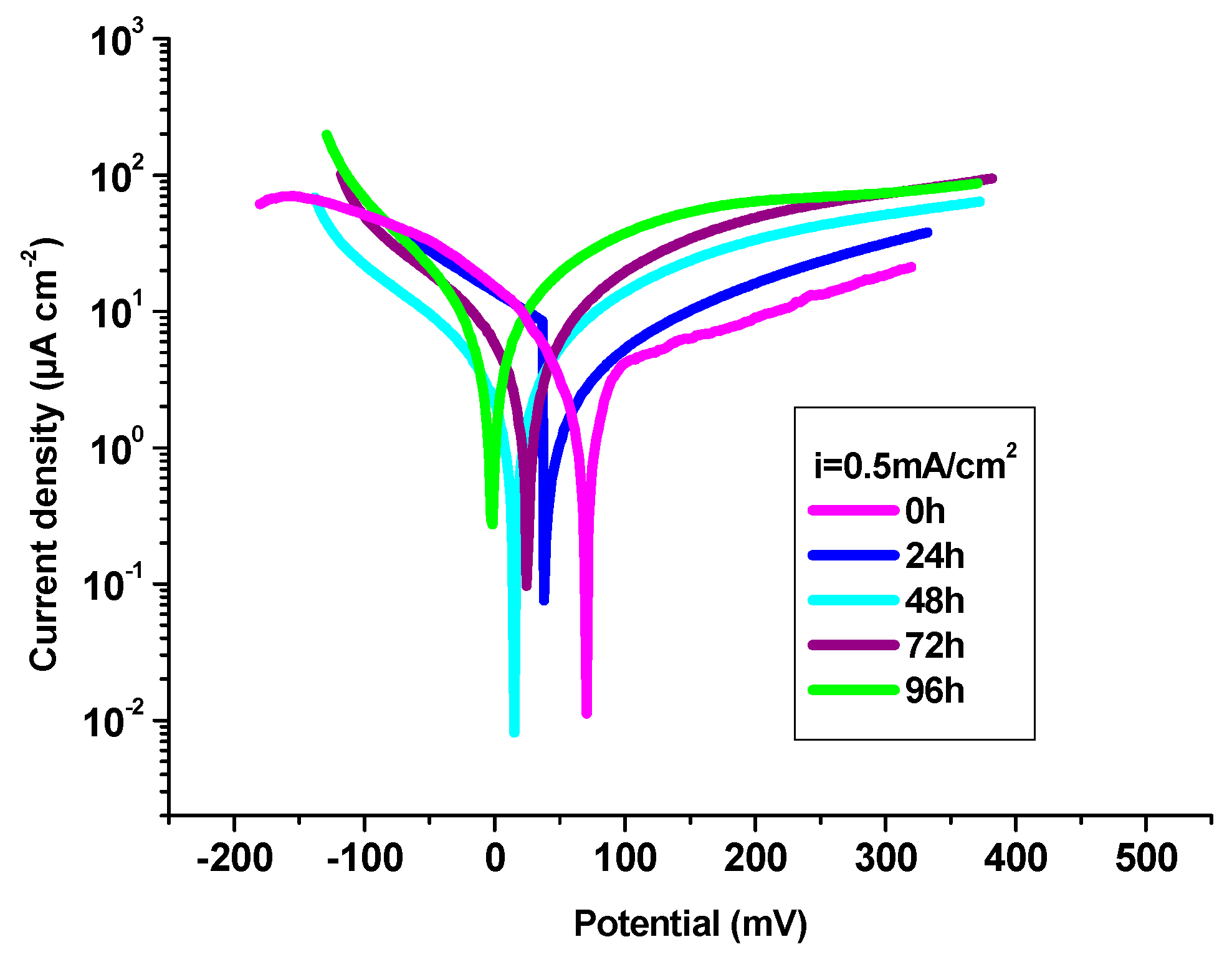
3.4.1. Potentiodynamic polarization technique
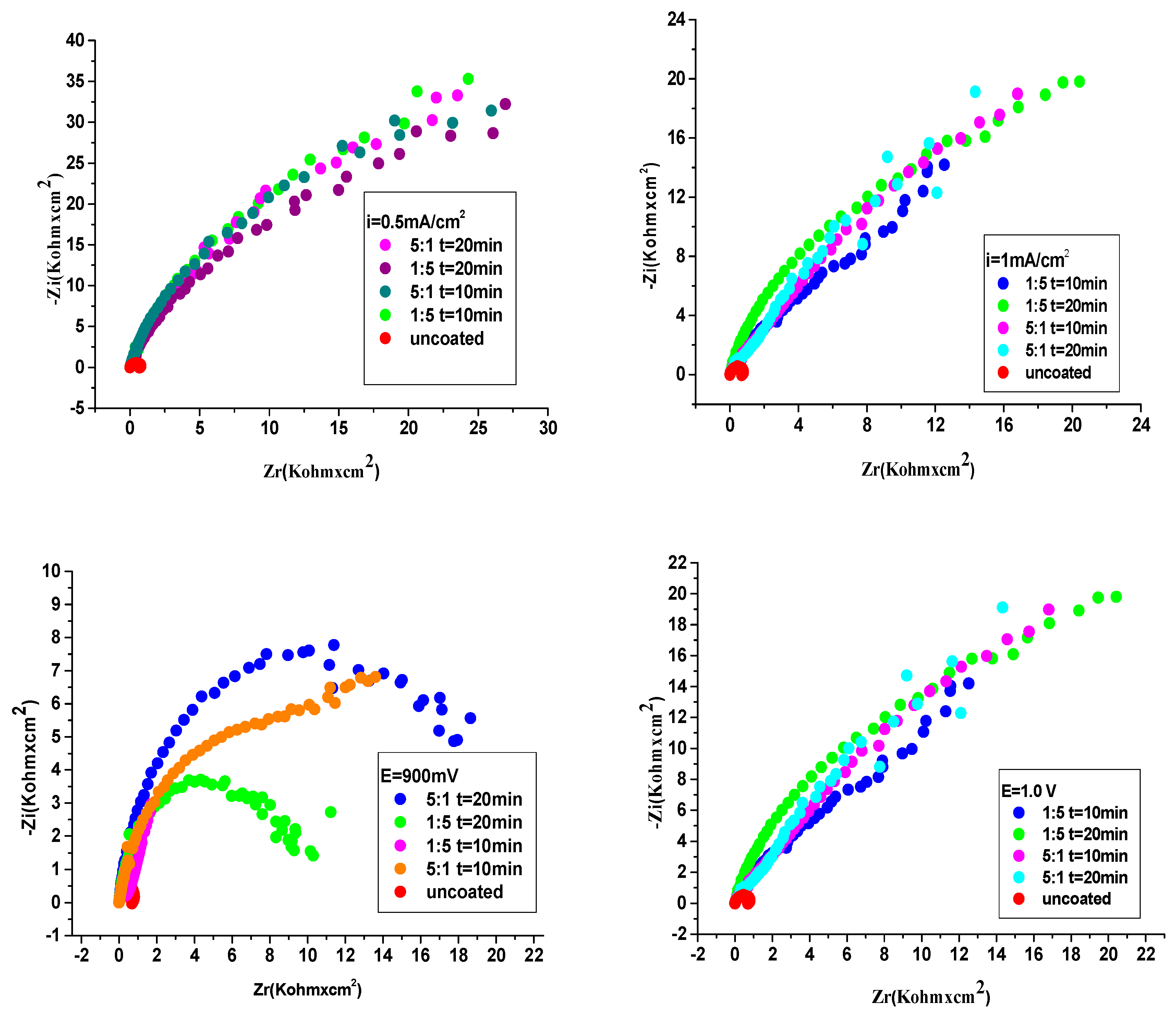
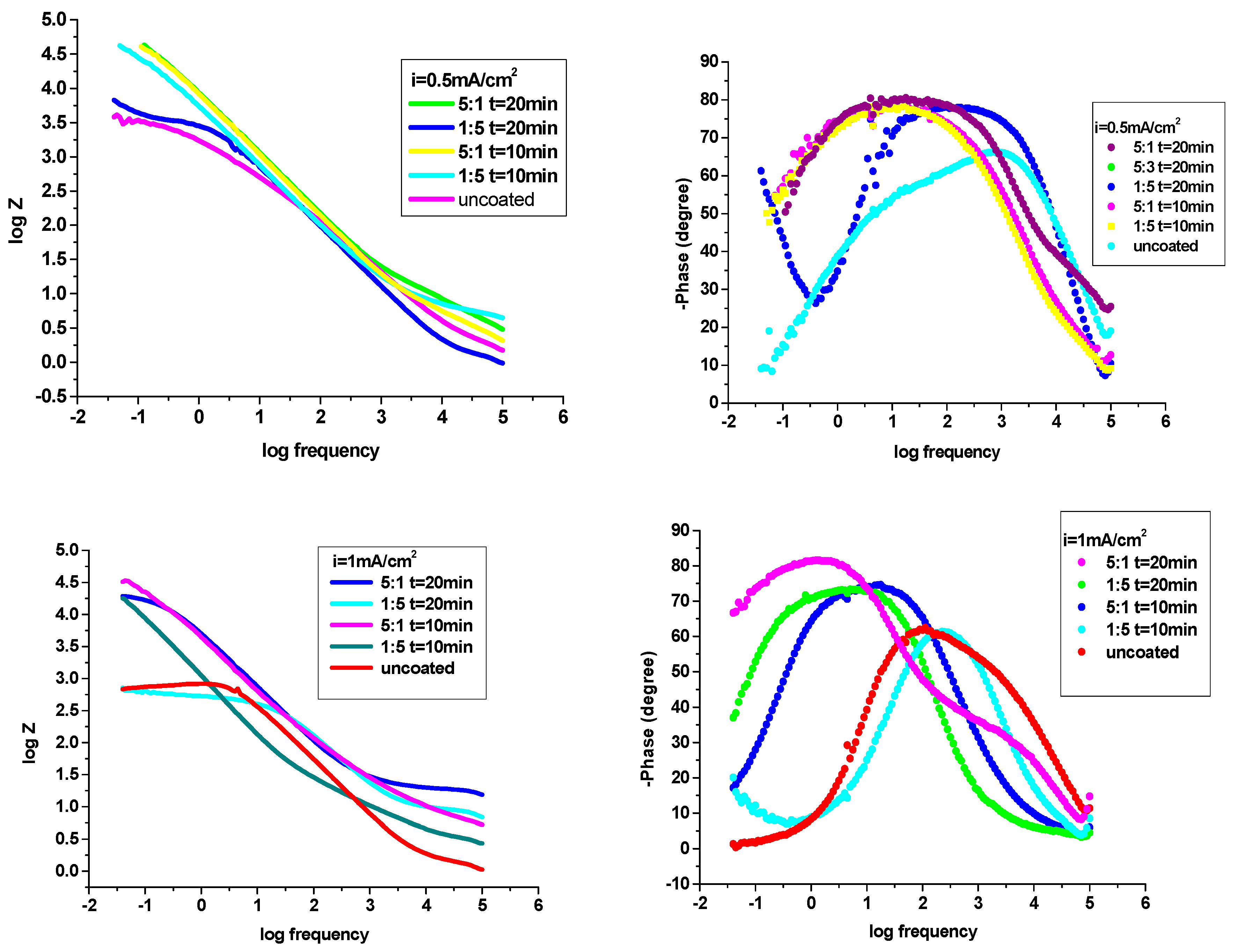
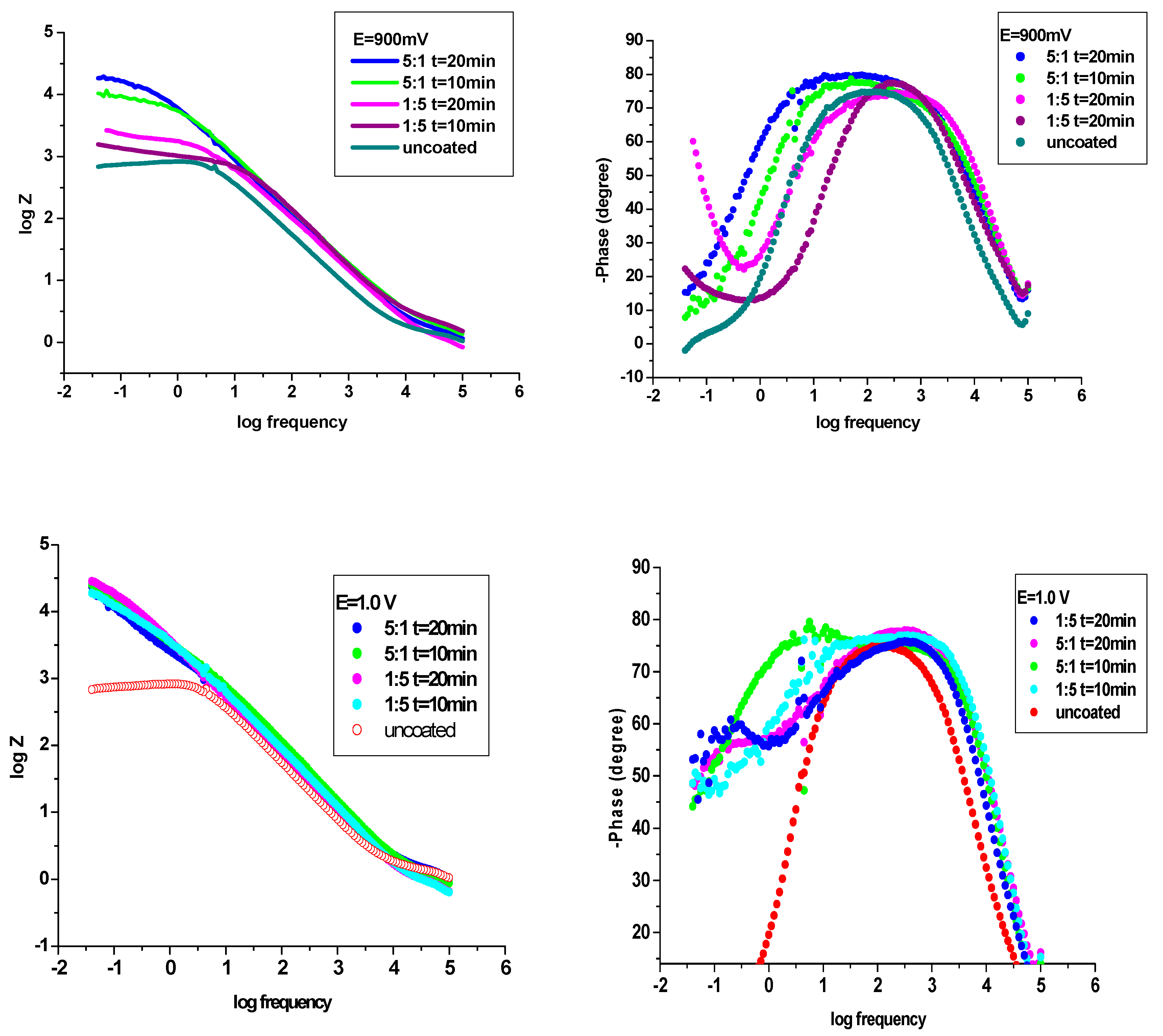
3.4.2. Electrochemical Impedance Spectroscopy (EIS) examinations
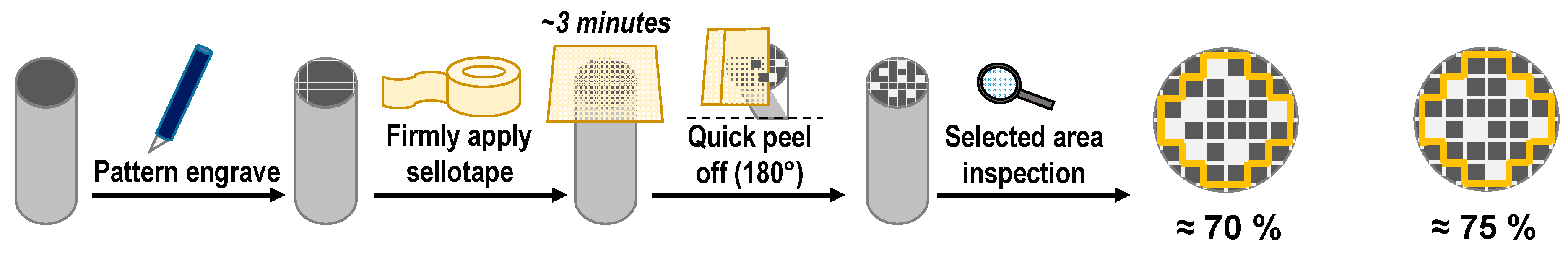
3.5. SEM investigations
4. Conclusions
References
- Wang, T.; Xu, Y.; Liu, Z.; Li, G.; Ren, L. A chitosan/polylactic acid composite coating enhancing the corrosion resistance of the bio-degradable magnesium alloy. Prog. Org. Coat 2023, 178, 107469. [Google Scholar] [CrossRef]
- Yu, H.; Guo, Q.; Wang, C.; Cao, G.; Liu, Y. Preparation and performance of PANI/CNTs composite coating on 316 stainless steel bipolar plates by pulsed electrodeposition. Prog. Org. Coat 2023, 182, 107611. [Google Scholar] [CrossRef]
- Song, L.; Gao, Z.; Sun, Q.; Chu, G.; Shi, H.; Xu, N.; Li, Z.; Hao, N.; Zhang, X.; Wang, M.L. Corrosion protection performance of a coating with 2-aminino-5-mercato-1,3,4-thiadizole-loaded hollow mesoporous silica on copper. Prog. Org. Coat 2023, 175, 107331. [Google Scholar] [CrossRef]
- Liao, F.-Q.; Chen, Y.-C. Siloxane-based epoxy coatings through cationic photopolymerization for corrosion protection. Prog. Org. Coat 2023, 174, 107235. [Google Scholar] [CrossRef]
- Gallegos-Melgar, A.; Serna, S.A.; Lázaro, I.; Gutiérrez-Castañeda, J.; Mercado-Lemus VArcos-Gutierrez, H.H.; Hernández-Hernández, M.; Porcayo-Calderón Jan Mayen, J.; Del Angel Monroy, M. Potentiodynamic Polarization Performance of a Novel Composite Coating System of Al2O3/Chitosan-Sodium Alginate, Applied on an Aluminum AA6063 Alloy for Protection in a Chloride Ions Environment. Coatings 2020, 10, 45. [Google Scholar] [CrossRef]
- Xie, J.; Lu, Z.; Zhou, K.; Li, C.; Ma, J.; Wang, B.; Xu, K.; Cui, H.; Liu, J. Researches on corrosion behaviors of carbon steel/copper alloy couple under organic coating in static and flowing seawater. Progress in Organic Coatings 2022, 166, 106793. [Google Scholar] [CrossRef]
- Zhang, J.; Zhu, A. Study on the synthesis of PANI/CNT nanocomposite and its anticorrosion mechanism in waterborne coatings. Prog. Org. Coat. 2021, 159, 106447. [Google Scholar] [CrossRef]
- Patil, D.; Patil, P.P. Electrodeposition of poly (o-toluidine) on brass from aqueous salicylate solution and its corrosion protection performance. J. Appl. Polym. Sci 2010, 118, 2084–2091. [Google Scholar] [CrossRef]
- Yılmaz, S.M.; Atun, G. Corrosion protection efficiency of the electrochemically synthesized polypyrrole-azo dye composite coating on stainless steel. Prog. Org. Coat. 2022, 169, 106942. [Google Scholar] [CrossRef]
- Branzoi, F.; Branzoi, V. Investigation of some nonionic surfactants as corrosion inhibitors for carbon steel in sulfuric acid medium. Int. J. Electrochem. Sci 2017, 12, 7638–7658. [Google Scholar] [CrossRef]
- Zhang, Y. Strengthening, Corrosion and Protection of High-Temperature Structural Materials. Coatings 2022, 12, 1136. [Google Scholar] [CrossRef]
- Zhai, Y.; Pan, K.; Zhang, E. Anti-Corrosive Coating of Carbon-Steel Assisted by Polymer-Camphor sulfonic Acid Embedded within Graphene. Coatings 2020, 10, 879. [Google Scholar] [CrossRef]
- Shinde, V.; Gaikwad, A.B.; Patil, P.P. Synthesis and characterization of corrosion protective poly(2,5-dimethylaniline) coatings on copper. Applied Surface Science 2006, 253, 1037–1045. [Google Scholar] [CrossRef]
- Branzoi, F.; Branzoi, V. Investigation of Protective Effect of Polymeric Film Coatings on Carbon Steel in Aggressive Solutions. Int. J. Electrochem. Sci 2016, 11, 6564–6579. [Google Scholar] [CrossRef]
- Davoodi, A.; Honarbakhsh, S.; Farzi, G.A. Evaluation of corrosion resistance of polypyrrole/functionalized multi-walled carbon nanotubes composite coatings on 60Cu–40Zn brass alloy. Prog. Org. Coat. 2015, 88, 106–115. [Google Scholar] [CrossRef]
- Liu, A.; Tian, H.; Li, S.; XiaodanJu Yang, H.; Sun, Y.; Wang, L.; Li, W. Bioinspired layered hybrid coatings with greatly enhanced barrier effect and active corrosion protection performance. Prog. Org. Coat. 2021, 152, 106131. [Google Scholar] [CrossRef]
- Ozyılmaz, T.; Colak, N.; Ozyilmaz, G.; Sangun, M.K. Protective properties of polyaniline and poly(aniline-co-o-anisidine) films electrosynthesized on brass A. Prog. Org. Coat. 2007, 60, 24–32. [Google Scholar] [CrossRef]
- Bazzaoui, M.; Martins, J.I.; Bazzaoui, E.A.; Martins, L.; Machnikova, E. Sweet aqueous solution for electrochemical synthesis of polypyrrole part 1B: On copper and its alloys. Electrochim. Acta 2007, 52, 3568–3581. [Google Scholar] [CrossRef]
- González-Tejera, M.J.; García, M.V.; de la Blanca, E.S.; Redondo, M.I.; Raso, M.A.; Carrillo, I. , Electrochemical synthesis of N-methyl and 3-methyl pyrrole perchlorate doped copolymer films. Thin Solid Films 2007, 515, 6805–6811. [Google Scholar] [CrossRef]
- Branzoi, F.; Branzoi, V.; Musina, A. Fabrication and characterisation of conducting composite films based on conducting polymers and functionalised carbon nanotubes. Surface and Interface Anal. 2012, 44, 1076–1081. [Google Scholar] [CrossRef]
- Duran, B.; Bereket, G. Cyclic Voltammetric Synthesis of Poly(N-methyl pyrrole) on Copper and Effects of Polymerization Parameters on Corrosion Performance. Ind. Eng. Chem. Res. 2012, 51, 5246–5255. [Google Scholar] [CrossRef]
- Dararatana, N.; Seidi, F.; Crespy, D. Polymer conjugates for dual functions of reporting and hindering corrosion. Polymer 2020, 194, 122346. [Google Scholar] [CrossRef]
- Luo, Y.; Li, X.; Luo, Z.; Chen, L.; Han, G. Enhanced adhesive and anti-corrosive performances of polymer composite coating for rusted metallic substrates by capillary filling. Prog. Org. Coat 2023, 178, 107467. [Google Scholar] [CrossRef]
- Redondo, M.I.; de la Blanca, E.S.; García, M.V.; González-Tejera, M.J. Poly(N-methylpyrrole) Electrodeposited on copper: Corrosion protection properties. Prog. Org. Coat. 2009, 65, 386–39. [Google Scholar] [CrossRef]
- Ren, S.; Barkey, D. Electrochemically Prepared Poly (3-methylthiophene) Films for Passivation of 430 Stainless Steel. J. Electrochem. Soc. 1992, 139, 4–1021. [Google Scholar] [CrossRef]
- Çakmakci, I.; Duran, B.; Duran, M.; Bereket, G. Experimental and theoretical studies on protective properties of poly (pyrrole-co-N-methyl pyrrole) coatings on copper in chloride media. Corrosion Science 2013, 69, 252–261. [Google Scholar] [CrossRef]
- Zhou, W.; Wu, K.; Zhang, K.; Wang, Z.; Liu, Z.; Hu, S.; Fang, Y.; He, C. Studies on Corrosion Behaviors of Q235 Steel Coated by the Polypyrrole Films Doped with different dopants. Int. J. Electrochem. Sci. 2020, 15, 2594–2603. [Google Scholar] [CrossRef]
- Rui, M.; Aiping Zhu, A. The synthesis and corrosion protection mechanisms of PANI/CNT nanocomposite doped with organic phosphoric acid. Prog.Org.Coat 2021, 153, 106134. [Google Scholar] [CrossRef]
- Marti, M.; Armelin, E.; Iribarren, J.I.; Aleman, C. Soluble polythiophenes as anticorrosive additives for marine epoxy paints. Mater. Corros 2015, 66, 23–30. [Google Scholar] [CrossRef]
- Anaya-Garza, K.; Torres-Huerta, A.M.; Domínguez-Crespo, M.A.; Moreno-Palmerín, J.; Rodríguez-Salazar, A.E. Corrosion resistance improvement of the Ti6Al4V/UHMWPE systems by the assembly of ODPA molecules by dip coating technique. Prog. Org. Coat 2022, 171, 107013. [Google Scholar] [CrossRef]
- Asan, G.; Asan, A.; Çelikkan, H. The effect of 2D-MoS2 doped polypyrrole coatings on brass corrosion. Journal of Molecular Structure 2020, 1203, 127318. [Google Scholar] [CrossRef]
- Guptad, K.; Neupane, S.; Singh, S.; Karki, N.; Yadav, A.P. The effect of electrolytes on the coating of polyaniline on mild steel by electrochemical methods and its corrosion behavior. Prog. Org. Coat 2021, 152, 106127. [Google Scholar] [CrossRef]
- Branzoi, V.; Branzoi, F.; Pilan, L. Characterization of electrodeposited polymeric and composite modified electrodes on cobalt based alloy. Mat. Chem. Phys. 2009, 118, 197–202. [Google Scholar] [CrossRef]
- Yagan, A.; Pekmez, N.Ö.; Yıldız, A. Poly (N-methylaniline) coatings on stainless steel by electropolymerization. Corros. Sci. 2007, 49, 2905–2919. [Google Scholar] [CrossRef]
- Zeybek, B.; Aksun, E. Electrodeposition of poly(N-methylpyrrole) on stainless steel in the presence of sodium dodecylsulfate and its corrosion performance. Prog. Org. Coat. 2015, 81, 1–10. [Google Scholar] [CrossRef]
- Pekmeza, N.Ö.; Cınkıllı, K.; Zeybekba, B. The electrochemical copolymerization of pyrrole and bithiophene on stainless steel in the presence of SDS in aqueous medium and its anticorrosive performance. Prog. Org. Coat. 2014, 77, 1277–1287. [Google Scholar] [CrossRef]
- Redondo, M.I.; de la Blanca, E.S.; García, M.V.; González-Tejera, M.J. Poly(N-methylpyrrole) Electrodeposited on copper: Corrosion protection properties. Prog. Org. Coat. 2009, 65, 386–39. [Google Scholar] [CrossRef]
- Branzoi, F.; Branzoi, V. Enzymatic electrode obtained by immobilizing of urease into a nanocomposite film based on conducting polymers and different additives. Int. J. Polym. Mater. Polym. Biomater 2014, 63, 549–556. [Google Scholar] [CrossRef]
- Su, W.; Iroh, J.O. Electrodeposition mechanism, adhesion and corrosion performance of polypyrrole and poly (N-methylpyrrole) coatings on steel substrates. Synthetic Metals 2000, 114, 225–234. [Google Scholar] [CrossRef]
- Branzoi, F.; Pahom, Z.; Nechifor, G. Corrosion protection of new composite polymer coating for carbon steel in sulfuric acid medium by electrochemical methods. Journal of Adhesion Science and Technology 2018, 2364–2380. [Google Scholar] [CrossRef]
- Gopi, D.; Saraswathy, R.; Kavitha, L.; Kim, D.-K. Electrochemical synthesis of poly(indole-co-thiophene) on low-nickel stainless steel and its anticorrosive performance in 0.5 mol L−1H2SO4. Polym. Int. 2014, 63, 280–289. [Google Scholar] [CrossRef]
- Yagan, A.; Pekmez, O.N.; Yildiz, A. Poly(N-ethylaniline) coatings on 304 stainless steel for corrosion protection in aqueous HCl and NaCl solutions. Electrochim. Acta. 2008, 53, 2474–2482. [Google Scholar] [CrossRef]
- Chou, T.P.; Chandrasekaran, C.; Limmer, S.J.; Seraji, S.; Wu, Y.; Forbess, M.J.; Nguyen, C.; Cao, G.Z. Organic–inorganic hybrid coatings for corrosion protection. Journal of Non-Crystalline Solids 2001, 290, 153–162. [Google Scholar] [CrossRef]
- Fuseini, M.; Zaghloul, M.M.Y. Investigation of Electrophoretic Deposition of PANI Nano fibers as a Manufacturing Technology for corrosion protection. Prog. Org. Coat 2022, 171, 107015. [Google Scholar] [CrossRef]
- Yan, Q.; Pan, W.; Zhong, S.; Guo, R.Z.; Li, X. Effect of solvents on the preparation and corrosion protection of polypyrrole. Prog. Org. Coat 2019, 132, 298–304. [Google Scholar] [CrossRef]
- ARamírez, M.R.; Mieres, F.; Pineda, F.; Grez, P.; Heyser, C. Electrosynthesis of polyindole-carboxylic acids on stainless steel and their corrosion protection at different temperatures in acidic solution. Prog. Org. Coat 2022, 177, 107075. [Google Scholar] [CrossRef]
- Shi, S.; Zhao, Y.; Zhang, Z.; Yu, L. Corrosion protection of a novel SiO2/PANI coating for Q235 carbon steel. Prog. Org. Coat. 2019, 132, 227–234. [Google Scholar] [CrossRef]
- Sun, Y.; Hu, C.; Cui, J.; Shen, S.; Qiu, H.; Li, J. Electrodeposition of polypyrrole coatings doped by benzenesulfonic acid-modified graphene oxide on metallic bipolar plates. Prog. Org. Coat 2022, 170, 106995. [Google Scholar] [CrossRef]
- Menkuer, M.; Ozkazanc, H. Anticorrosive polypyrrole/zirconium-oxide composite film prepared in oxalic acid and dodecylbenzene sulfonic acid mix electrolyte. Prog. Org. Coat. 2020, 147, 105815. [Google Scholar] [CrossRef]
- Branzoi, F.; Mihai, M.A.; Petrescu, S. Corrosion Protection Efficacy of the Electrodeposit of Poly (N-Methyl Pyrrole-Tween20/3-Methylthiophene) Coatings on Carbon Steel in Acid Medium. Coatings 2022, 12, 1062. [Google Scholar] [CrossRef]
- Pekmeza, N.Ö.; Cinkillia, K.; Zeybekba, B. The electrochemical copolymerization of pyrrole and bithiophene on stainless steel in the presence of SDS in aqueous medium and its anticorrosive performance. Prog. Org. Coat. 2014, 77, 1277–1287. [Google Scholar] [CrossRef]
- Vernickaite, E.; Tsyntsaru, N.; Cesiulis, E.H. Electrodeposition and corrosion behaviour of nanostructured cobalt–tungsten alloys coatings. Transactions of the IMF 2016, 94, 313–321. [Google Scholar] [CrossRef]
- Hong, J.H.; Yeoh, F.Y. Mechanical properties and corrosion resistance of cobalt-chrome alloy fabricated using additive manufacturing. Materials Today Proceedings 2020, 29, 196–201. [Google Scholar] [CrossRef]
- Ma, T.; Tan, B.; Guo, L.; Wang, W.; Li, W.; Ji, J.; Yan, M.; Kaya, S. Experimental and theoretical investigation on the inhibition performance of disulfide derivatives on cobalt corrosion in alkaline medium. Journal of Molecular Liquids 2021, 341, 116907. [Google Scholar] [CrossRef]
- Hong, J.H.; Yeoh, F.Y. Mechanical properties and corrosion resistance of cobalt-chrome alloy fabricated using additive manufacturing. Materials Today Proceedings 2020, 29, 196–201. [Google Scholar] [CrossRef]
- Nadolski, M.; Golański, G.; Klimas, J.; Szota, M.; Szymański, J. Microstructure and functional properties of prosthetic cobalt alloys COCRW. Institute of Metallurgy and Materials Science of Polish Academy of Sciences 2015, 3. [Google Scholar] [CrossRef]
- Güven, N.C.; Ozkazanc, H. Corrosion protection behavior of poly(N-methylpyrrole)/boron nitride composite film on aluminum-1050. Progress in Organic Coatings 2022, 164, 106696. [Google Scholar] [CrossRef]
- Hernández-Martínez, D.; León-Silva, U.; Nicho, M.E. Corrosion protection of steel by poly(3-hexylthiophene) polymer blends. Anti-Corrosion Methods and Materials 2015, 62, 229–240. [Google Scholar] [CrossRef]
- Branzoi, F.; Branzoi, V.; Musina, A. Coatings based on conducting polymers and functionalized carbon nanotubes obtained by electropolymerization. Prog. Org. Coat. 2013, 76, 632–638. [Google Scholar] [CrossRef]
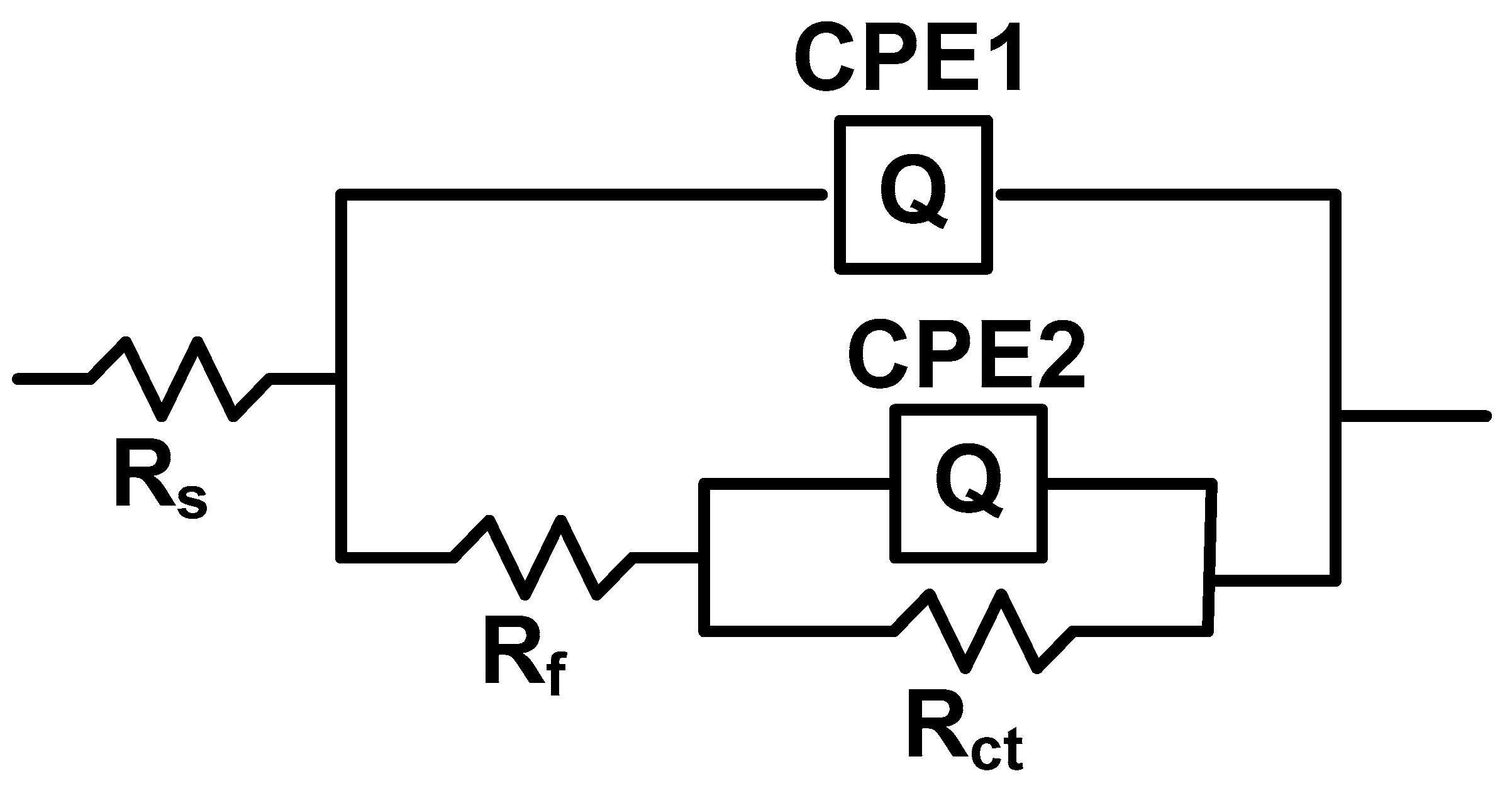


| The system P3MPY-SDS/P2MT /CoCrW | Ecorr (mV) |
icorr (µA/cm2 |
Rp kΩcm2 |
Rmpy | Pmm/year | Kg (g/m2h) |
ba (mV/ decade |
-bc (mV/ decade |
E (%) | %P |
| CoCrW + 1 M HCl | -230 | 21 | 0.390 | 9.91 | 0.255 | 0.234 | 86 | -90 | - | |
| P3MPY-SDS/P2MT 1mA/cm2 1:5 molar ratio, t=10min | -146 | 0.67 | 41 | 0.316 | 0.008 | 0.0074 | 108 | -102 | 95 | 0.0009 |
| P3MPY-SDS/P2MT 1mA/cm2 5:1 molar ratio, t=10min | 160 | 0.39 | 49 | 0.180 | 0.0045 | 0.0042 | 80 | -117 | 98 | 0.00003 |
| P3MPY-SDS/P2MT 1mA/cm2 1:5 molar ratio, t=20min | 57 | 0.46 | 44 | 0.217 | 0.0055 | 0.0051 | 91 | -93 | 97 | 0.00004 |
| P3MPY-SDS/P2MT 1mA/cm2 5:1 molar ratio, t=20min | 180 | 0.33 | 59 | 0.155 | 0.0039 | 0.0036 | 119 | -102 | 98 | 0.00003 |
| P3MPY-SDS/P2MT 0.5mA/cm2 1:5 molar ratio, t=10min | -232 | 0.63 | 37 | 0.297 | 0.0075 | 0.0070 | 94 | -111 | 96 | 0.0001 |
| P3MPY-SDS/P2MT 0.5mA/cm2 5:1 molar ratio, t=10min | -60 | 0.36 | 41 | 0.169 | 0.0042 | 0.0040 | 121 | -87 | 98 | 0.00009 |
| P3MPY-SDS/P2MT 0.5mA/cm2 1:5 molar ratio, t=20min | -236 | 0.43 | 46 | 0.202 | 0.0051 | 0.0047 | 85 | -88 | 97 | 0.0059 |
| P3MPY-SDS/P2MT 0.5mA/cm2 5:1 molar ratio, t=20min | 80 | 0.29 | 60 | 0.136 | 0.0034 | 0.0032 | 118 | -94 | 98.5 | 0.00001 |
| The system P3MPY-SDS/P2MT /CoCrW | Ecorr (mV) |
icorr (µA/cm2 |
Rp kΩcm2 |
Rmpy | Pmm/year | Kg (g/m2h) |
ba (mV/ decade |
-bc (mV/ decade |
E (%) | %P |
| CoCrW + 1 M HCl | -230 | 21 | 0.390 | 9.91 | 0.255 | 0.234 | 86 | -90 | - | |
| P3MPY-SDS/P2MT 0.9V 1:5 molar ratio, t=10min | -242 | 0.97 | 36 | 0.451 | 0.011 | 0.010 | 109 | -78 | 94 | 0.0007 |
| P3MPY-SDS/P2MT 0.9V 5:1 molar ratio, t=10min | -65 | 0.49 | 41 | 0.231 | 0.005 | 0.0045 | 107 | -88 | 97 | 0.000095 |
| P3MPY-SDS/P2MT 0.9V 1:5 molar ratio, t=20min | -225 | 0.56 | 39 | 0.264 | 0.007 | 0.0066 | 83 | -81 | 97 | 0.000016 |
| P3MPY-SDS/P2MT 0.9V 5:1 molar ratio, t=20min | 80 | 0.37 | 44 | 0.174 | 0.0044 | 0.0041 | 77 | -88 | 98 | 0.00063 |
| P3MPY-SDS/P2MT 1.0V 1:5 molar ratio, t=10min | -220 | 0.73 | 27 | 0.344 | 0.0088 | 0.0081 | 111 | -91 | 95 | 0.0058 |
| P3MPY-SDS/P2MT 1.0V 5:1 molar ratio, t=10min | -130 | 0.47 | 39 | 0.22 | 0.0056 | 0.0052 | 76 | -78 | 97 | 0.000054 |
| P3MPY-SDS/P2MT 1.0V 1:5 molar ratio, t=20min | -143 | 0.63 | 37 | 0.30 | 0.007 | 0.0069 | 69 | 79 | 97 | 0.0015 |
| P3MPY-SDS/P2MT 1.0V 5:1 molar ratio, t=20min | 62 | 0.35 | 47 | 0.165 | 0.0041 | 0.0039 | 78 | 82 | 98 | 0.000033 |
| The system P3MPY-SDS/P2MT /CoCrW | Ecorr (mV) |
icorr (µA/cm2 |
Rmpy | Pmm/year | Kg (g/m2h) |
ba (mV/ decade |
-bc (mV/ decade |
E (%) |
| P3MPY-SDS/P2MT 0.5mA/cm2 5:1 molar ratio, t=10 min 0h | 70 | 0.46 | 0.169 | 0.0042 | 0.0040 | 121 | -87 | 98 |
| P3MPY-SDS/P2MT 0.5mA/cm2 5:1 molar ratio, t=10 min 24h | 38 | 1.16 | 0.547 | 0.0138 | 0.0129 | 74 | -68 | 95 |
| P3MPY-SDS/P2MT 0.5mA/cm2 5:1 molar ratio, t=10min 48h | 15 | 2.43 | 1.146 | 0.029 | 0.027 | 99 | -86 | 88 |
| P3MPY-SDS/P2MT 0.5mA/cm2 5:1 molar ratio, t=10min 72h | 24 | 3.29 | 1.469 | 0.037 | 0.034 | 93 | -88 | 84 |
| P3MPY-SDS/P2MT 0.5mA/cm2 5:1 molar ratio, t=10min 96h | -2 | 4.96 | 2.341 | 0.059 | 0.055 | 95 | -91 | 77 |
| The system P3MPY-SDS/P2MT/cobalt alloy | Rs ohm⋅cm2 | Q-Yo S⋅s-n⋅cm-2 |
Q-n | Rf ohm⋅cm2 | Q-Yo S⋅s-n⋅cm-2 |
Q-n | Rct ohm⋅cm2 |
χ | |
| CoCrW + 1 M HCl | 1.30 | 4.69E-5 | 0.89 | 13 | 1.378E-5 | 0.86 | 809 | 8.25e-03 | |
| P3MPY-SDS/P2MT 1mA/cm2 1:5 molar ratio, t=10min | 2.53 | 1.13E-5 | 0.93 | 64 | 5.14E-5 | 0.57 | 7.576E4 | 7.523e-04 | |
| P3MPY-SDS/P2MT 1mA/cm2 5:1 molar ratio, t=10min | 2.66 | 1.91E-5 | 0.98 | 64.73 | 6.73E-5 | 0.84 | 8.921E4 | 3.733e-03 | |
| P3MPY-SDS/P2MT 1mA/cm2 1:5 molar ratio, t=20min | 4.34 | 6.38E-4 | 0.61 | 49 | 4.96E-5 | 0.83 | 4.956E4 | 1.647e-03 | |
| P3MPY-SDS/P2MT 1mA/cm2 5:1 molar ratio, t=20min | 2.63 | 7.43E-4 | 0.63 | 65 | 1.66E-4 | 0.93 | 4.976E4 | 3.935e-04 | |
| P3MPY-SDS/P2MT 0.5mA/cm2 1:5 molar ratio, t=10min | 1.76 | 3.58E-5 | 0.79 | 94.8 | 2.26E-5 | 0.89 | 8.018E4 | 8.016e-04 | |
| P3MPY-SDS/P2MT 0.5mA/cm2 5:1 molar ratio, t=10min | 3.22 | 1.16E-4 | 0.61 | 94.5 | 3.48E-5 | 0.84 | 6.963E4 | 6.653e-04 | |
| P3MPY-SDS/P2MT 0.5mA/cm2 1:5 molar ratio, t=20min | 5.912 | 3.32E-4 | 0.64 | 65.2 | 4.94E-5 | 0.89 | 7.846E4 | 1.021e-03 | |
| PNMPY-1SSD/P2MT 0.5mA/cm2 5:1 molar ratio, t=20min | 2.789 | 1.02E-5 | 0.84 | 174 | 1.24E-5 | 0.91 | 9.228E4 | 7.428e-04 | |
| The system P3MPY-SDS/P2MT/cobalt alloy | Rs ohm⋅cm2 | Q-Yo S⋅s-n⋅cm-2 |
Q-n | Rf ohm⋅cm2 |
Q-Yo S⋅s-n⋅cm-2 |
Q-n | Rct ohm⋅cm2 |
χ |
| CoCrW + 1 M HCl | 1.30 | 4.69E-5 | 0.89 | 13 | 1.378E-5 | 0.86 | 809 | 8.25e-03 |
| P3MPY-SDS/P2MT 0.9V 1:5 molar ratio, t=10min | 0.79 | 3.88E-5 | 0.88 | 362 | 7.35E-4 | 0.74 | 1.71E4 | 5.10e-03 |
| P3MPY-SDS/P2MT 0.91V 5:1 molar ratio, t=10min | 0.68 | 3.52E-5 | 0.61 | 64 | 2.81E-5 | 0.89 | 2.15E4 | 1.87e-03 |
| P3MPY-SDS/P2MT 0.9V 1:5 molar ratio, t=20min | 1.06 | 4.33E-5 | 0.84 | 304 | 2.576E-5 | 0.89 | 8639 | 1.16e-03 |
| P3MPY-SDS/P2MT 0.9V 5:1 molar ratio, t=20min | 1.44 | 2.85E-5 | 0.61 | 3805 | 5.456E-5 | 0.88 | 2.77E5 | 1.86e-03 |
| P3MPY-SDS/P2MT 1.0V 1:5 molar ratio, t=10min | 0.681 | 7.16E-5 | 0.61 | 88.5 | 3.305E-5 | 0.89 | 8.53E5 | 3.88e-03 |
| P3MPY-SDS/P2MT 1.0V 5:1 molar ratio, t=10min | 1.03 | 2.79E-5 | 0.89 | 2180 | 6.789E-5 | 0.63 | 1.1E5 | 1.89e-03 |
| P3MPY-SDS/P2MT 1.0V 1:5 molar ratio, t=20min | 0.806 | 3.72E-5 | 0.89 | 237 | 1.752E-5 | 0.78 | 3.539E4 | 6.12e-03 |
| P3MPY-SDS/P2MT 1.0V 5:1 molar ratio, t=20min | 2.51 | 3.76E-5 | 0.87 | 1778 | 9.026E-5 | 0.66 | 7.24E5 | 2.59e-03 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




