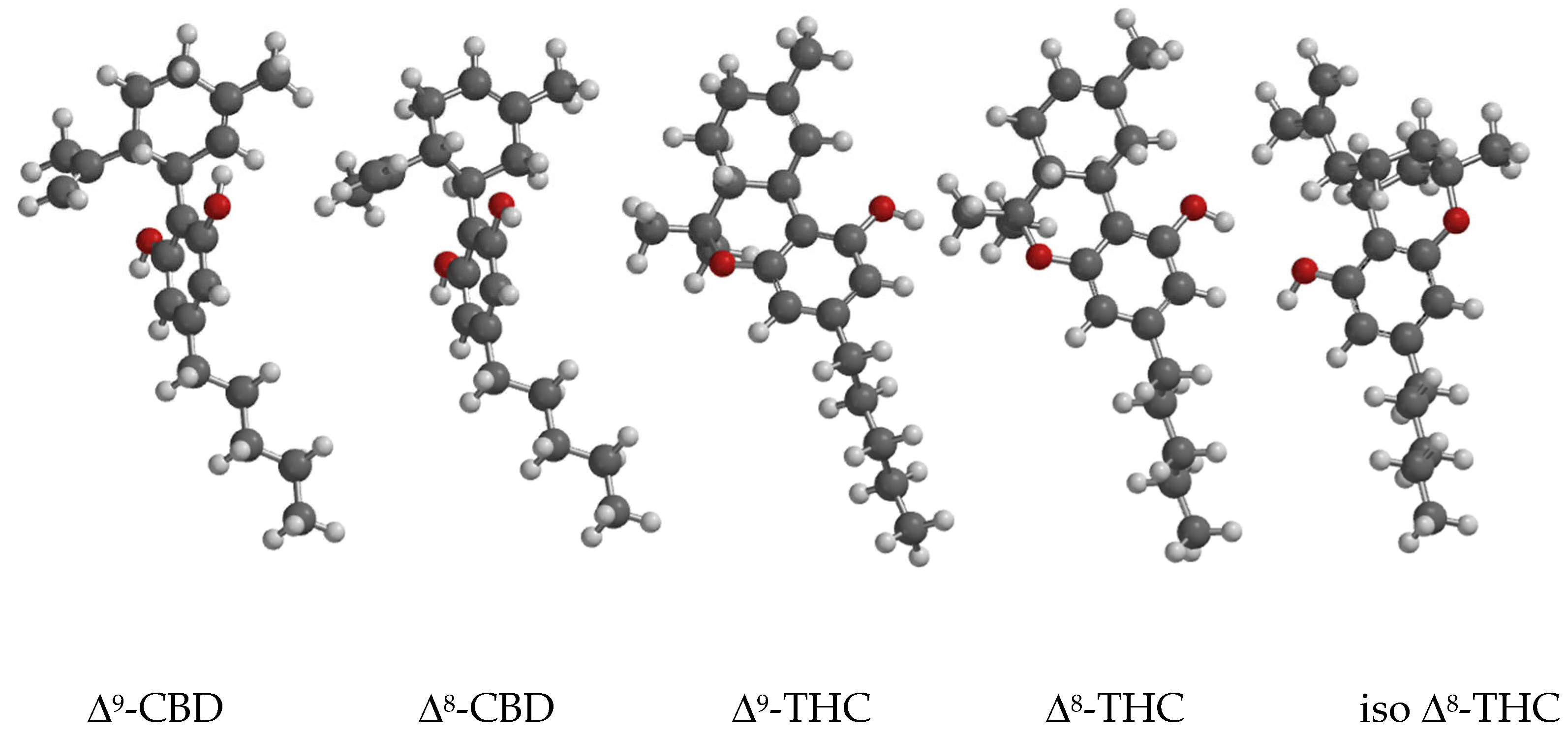
3.1.1. Cannabinoid Structures
To avoid confusion, the structures of all relevant cannabinoids are shown in
Figure 1 and shortly discussed below.
The structures of Δ
9-CBD, Δ
8-CBD , Δ
9-THC, and Δ
8-THC have been discussed before [
19] and originate from CD calculations as described in 2. Materials and Methods. The same approach was adopted for iso Δ
8-THC, using a model wherein the C
5-chain was reduced to a methyl group too. Like Δ
9-THC and Δ
8-THC, the conformational freedom of iso Δ
8-THC is restricted by the ring closure of the phenolic group to the Δ
8-C of the (former) cyclohexenyl substituent and shows 13 conformers only, all of them with the newly formed cyclohexane ring in a chair position with three axial substituents: the C-C(Ar)-, the C-O(Ar)-, and the isopropenyl substituent. Only the C-CH
3 at the Δ
8-position is in an equatorial position. The main differences between the conformers are in the position of the remaining phenolic group and the orientation of the isopropenyl substituent. The best conformer accounts for 84.6% in the Boltzmann weights, and the second-best conformer, showing a phenol group pointing to the isopropenyl cyclohexane substituent, accounts for 6.6 % in the Boltzmann weights.
3.1.1. The Conversion of Δ9-CBD to Δ8-THC, with pTSA as Brønsted Acid Catalyst
The conversion of Δ
9-CBD to Δ
8-THC, with pTSA as Brønsted acid catalyst, requires two steps: a ring closure and an isomerization of the double bond from the Δ
9 to the Δ
8-position. Ring closure reactions catalyzed by pTSA are quite rare. A recent example can be found in the pTSA condensation polymerization of dicarboxylic acids and polyols like sorbitol, where it appears as a side reaction on a secondary carbon atom [
20]. Ring closure by a phenol on a tertiary carbon seems possible only because of the intramolecular presence of the phenolic group, the absence of more suitable nucleophiles, and activation of the alkene by pTSA. These conditions are fulfilled by the reaction conditions described above: dry pTSA as a catalyst in refluxing toluene.
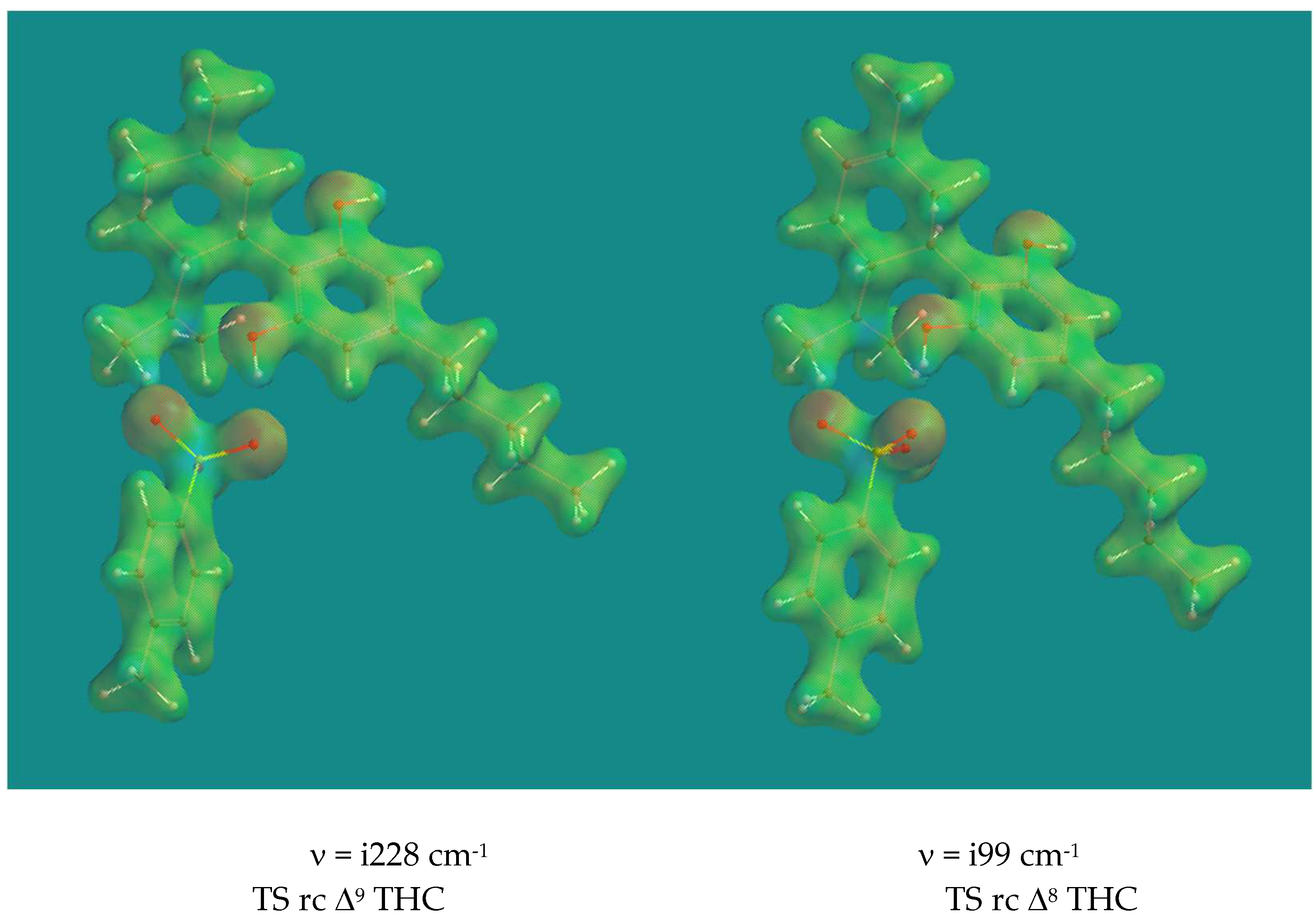
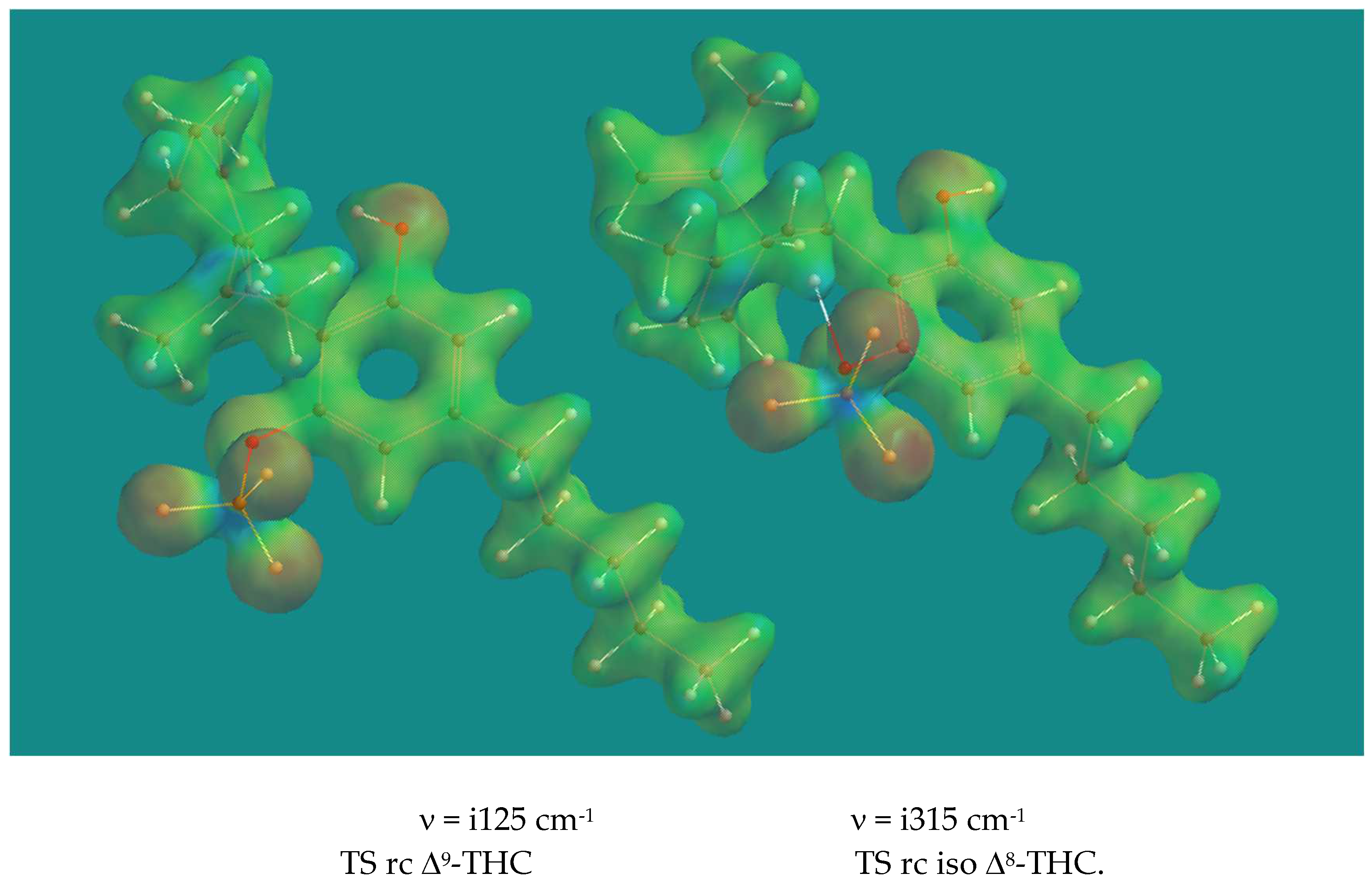
Figure 2 shows the Transition States of the ring closure of Δ
9-CBD to Δ
9-THC and Δ
8-CBD to Δ
8-THC catalyzed by pTSA.
The two Transition States look very similar. There is almost complete proton transfer from pTSA to the CH2 of the isopropenyl group: the CH2-HOSO2Ar-pCH3 distance is 1.237 Å and 1.212 Å respectively. This is also visible from the reduced electron density plot. The phenolic O-atom is poised to form a C-O bond with the tertiary C of the isopropenyl group despite the relative long distance between them: 2.758 Å and 3.204 Å respectively. The electrostatic potentials on the reduced electron density surface of the tertiary C’s are +1594 and +1544 kJ/mol respectively, a clear indication of their carbocation character. Animation of both imaginary frequencies show the right movement of all atoms involved. The H-atom of the phenol group is H-bridged to an O-atom of pTSA with distances of 1.723 Å and 1.746 Å respectively. The activation barriers are 70.5 and 57.5 kJ/mol respectively, the latter a reflection of the relative stability of Δ9-CBD compared to Δ8-CBD, which is 11.8 kJ/mol in favor of Δ9-CBD.
For the isomerization reaction two mechanisms were considered:
- a)
a concerted process with simultaneous proton transfers from pTSA to D9-C and D7-C to pTSA, and
- b)
a two-step process, starting with proton transfer from pTSA to D9-C, followed by proton transfer from D7-C to pTSA.
These mechanistic proposals for acid catalyzed isomerization are not new but go back to 1932 [
21,
22] and evidence has been obtained for both cases, depending on specific reaction conditions and catalysts.
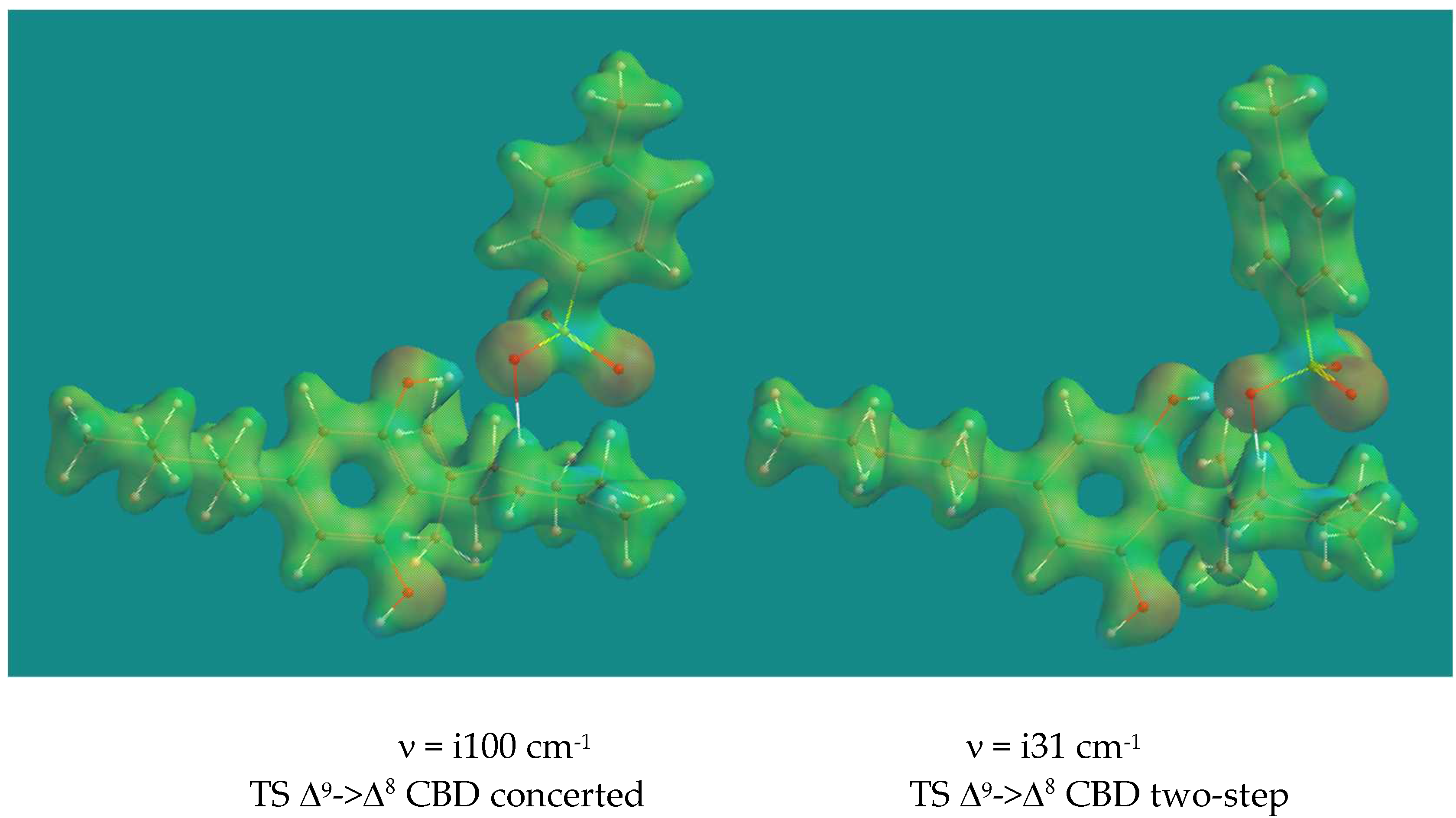
Figure 3 shows the Transition States of the concerted and the two-step process of the isomerization of Δ
9-CBD to Δ
8-CBD with pTSA.
On first sight the two Transition States look very similar, however appearances are deceiving. In the concerted process proton transfer from pCH3ArSO3H to Δ9-C is almost complete with an SO-H distance of 1.911 Å and a Δ9 C-H distance of 1.126 Å, while the SO-H Δ7 C distance is 1.720 Å and a Δ7 C-H distance is 1.169 Å. In the two-step process, proton transfer from pCH3ArSO3H to Δ9-C is less complete with an SO-H distance of 1.621 Å and a Δ9 C-H distance of 1.199 Å. Note that in this case the SO-H Δ7 C distance is 2.175 Å and a Δ7 C-H distance is 1.112 Å, also quite different from the concerted case. Once more the imaginary frequencies are low, the one for the two-step process even very low, but show the correct movement in both cases. The activation barriers are 75.3 and 67.2 kJ/mol respectively. The two-step process is clearly favored over the concerted process. The starting complex for both processes is the same. The activation barriers for the reverse process, the isomerization of Δ8->Δ9 CBD, show significantly higher activation barriers of 93.4 and 81.8 kJ/mol. The reason for that is the higher stabilization enthalpy of the starting complex caused by the H-bridge of the phenol group of Δ8 CBD to an O=S of pTSA, compared to the H-bridge of that phenol group of Δ9 CBD to an HO-S of pTSA.
For the isomerization of Δ9 THC and Δ8 THC again the two previous mentioned options were considered. However, it turned out that for the concerted option only a true Transition State could be established, despite many attempts to locate a Transition State for the two-step process. These attempts either led to the Transition State of the concerted process or to non converged structures which can be described best as intimate ion pairs, consisting of the Δ8 THC cation and the pTSA anion with no good imaginary frequency left, and a total energy of 1-3 kJ/mol higher only than the energy of the Transition State of the concerted process. In the concerted process proton transfer from pCH3ArSO3H to Δ9-C is almost complete with an SO-H distance of 1.836 Å and a Δ9 C-H distance of 1.146 Å, while the SO-H Δ7 C distance is 1.755 Å and a Δ7 C-H distance is 1.162 Å. The imaginary frequency is i152 cm-1 and its animation shows the correct movement of the proton transfers and the corresponding skeletal adaptation. The activation barriers for the TS Δ9->Δ8 THC and Δ8->Δ9 THC are 95.7 and 103.9 kJ/mol, reflecting the relative stabilities of Δ9 and Δ8 THC.
3.1.2. The Conversion of Δ9-CBD to Δ8-THC, with BF3·Et2O as Lewis Acid Catalyst
Though BF
3·Et
2O is a well know catalyst, it is difficult to find examples wherein ring closure of an O-nucleophile or alkene isomerization are described. Closest to the actual BF
3·Et
2O catalyzed ring closure are the inverse reactions, the B(C
6F
5)
3 catalyzed ring opening of an 2,2-disubstituted oxetane to a homoallylic alcohol, and the BF
3·Et
2O catalyzed decomposition of a t-butyldimethylsilyl ether of a tertiary alcohol [
23,
24]. The latter is very close to the previously described Example 2 with respect to the reaction conditions applied. In 2022 an example of an alkene isomerization with B(C
6F
5)
3 was published, including a mechanistic description [
25]. They found experimental and computational evidence that the isomerization of 2-propenyl benzene occurs via a direct hydride abstraction by B(C
6F
5)
3 leading to a mixed allylic-benzylic carbenium ion and a 1,2-hydride shift to the terminal alkene- B(C
6F
5)
3 complex. They used the M06-2X DFT functional with an extended basis set and solvent model. Unfortunately, the computational data are not available in their Supplementary Material. Attempts to locate similar Transition States with BF
3, using either the M06-2X or the B3LYP functional, were not successful.
As the electron deficient BF
3 wants to interact with electron rich systems, the complexation of BF
3 with the alkene and phenol groups of Δ
9 CBD was investigated to get an impression of the complexation energy. Results are listed in
Table 1. As a reference BF
3·Et
2O is listed too.
From
Table 1 it is clear that complexation of BF
3 with a phenol group is favored over complexation with an alkene like the 2-propenyl substituent on the cyclohexene ring, or ethers like in BF
3·Et
2O and BF
3· Δ
9 THC. It was realized that complexation of the Lewis acid BF
3 with a phenol transforms the weakly acidic phenol into a strong Brønsted acid.
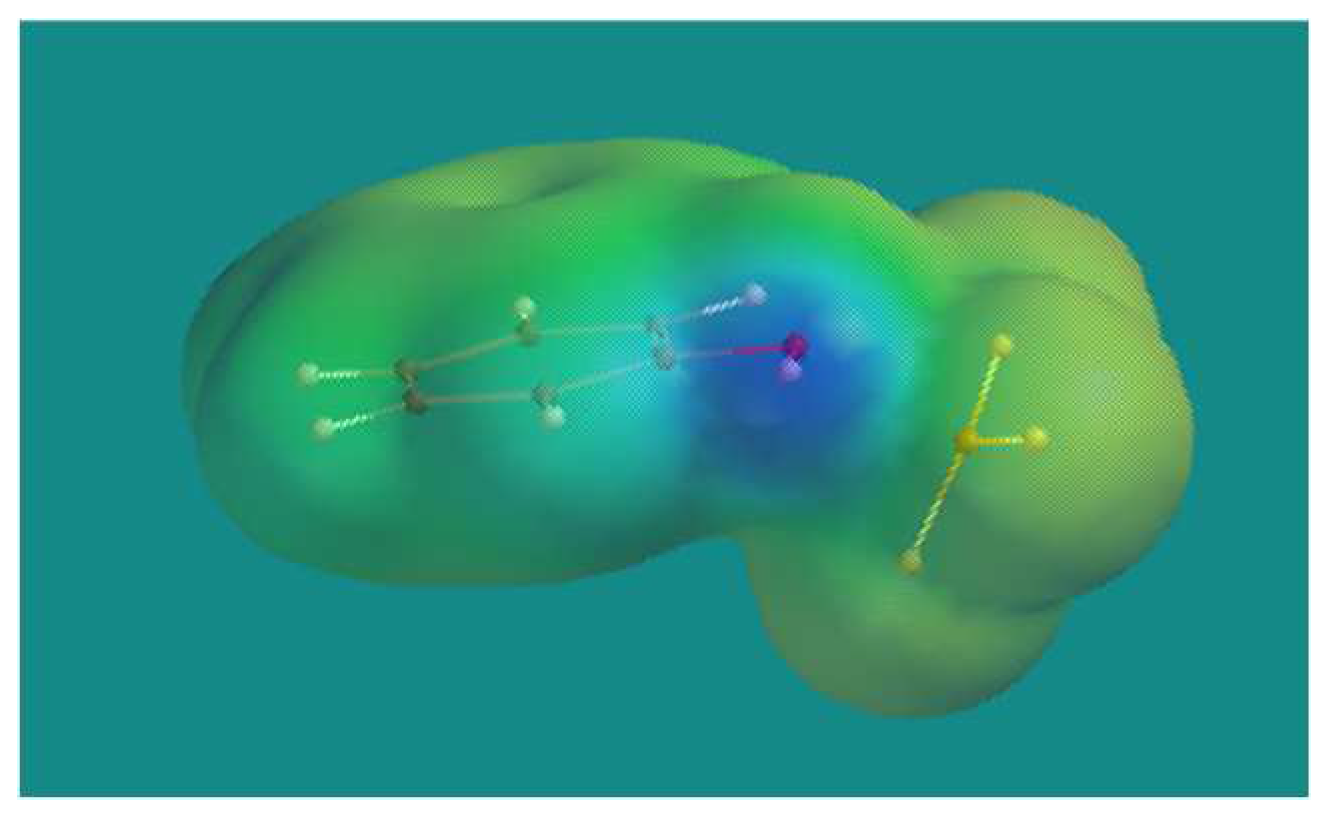
Figure 4 shows the BF
3·phenol complex. There is a clear blue spot at the surface of the phenolic group, indicative for a positive electrostatic potential. The Maximum Electrostatic Potential (MEP) shows a value of +314.7 kJ/mol. Using an earlier derived linear relation [
26] between MEP and pK
a yielded -2.17 as an estimate for the pK
a of the BF
3·phenol complex which is more acidic than pTSA (MEP = +261.6 kJ/mol); pK
a = -1.34) and slightly less acidic than H
2SO
4 (MEP = +327.0 kJ/mol; pK
a = -2.49).
Thus, reactions starting from a BF
3-phenolic cannabinoid complex were investigated.
Figure 5 shows the Transition States of the ring closure to Δ
9-THC and iso Δ
8-THC.
Both Transition States show nearly complete proton transfer from the phenol to the CH
2 of the 2-propenyl substituent and the Δ
9-C of the cyclohexenyl substituent respectively. Their O-H, H
2C-H and O-H, Δ
9-C distances are: 1.850 Å, 1.142 Å and 1.743 Å, 1.157 Å and their activation barriers are 80.9 and 82.1 kJ/mol respectively. The animation of their imaginary frequencies clearly shows the proton transfer process and some skeletal adaptation to the formation of the tertiary carbenium ion. No movement of the phenolic O to the tertiary is visible, and no sign of double bond isomerization is visible. Therefore, so called Energy Profiles (EP) were constructed, starting from the Transition States, and leading to either the starting BF
3·Δ
9-CBD complexes or the BF
3·Δ
9-THC, BF
3· iso Δ
8-THC, or the double bond isomerized products. In an EP a constraint is applied and next this constraint is varied in regular small steps from the starting to the final situation in a series of full geometry optimizations. A plot of the (relative) energy versus the steps provides an impression of the feasibility of such a pathway.
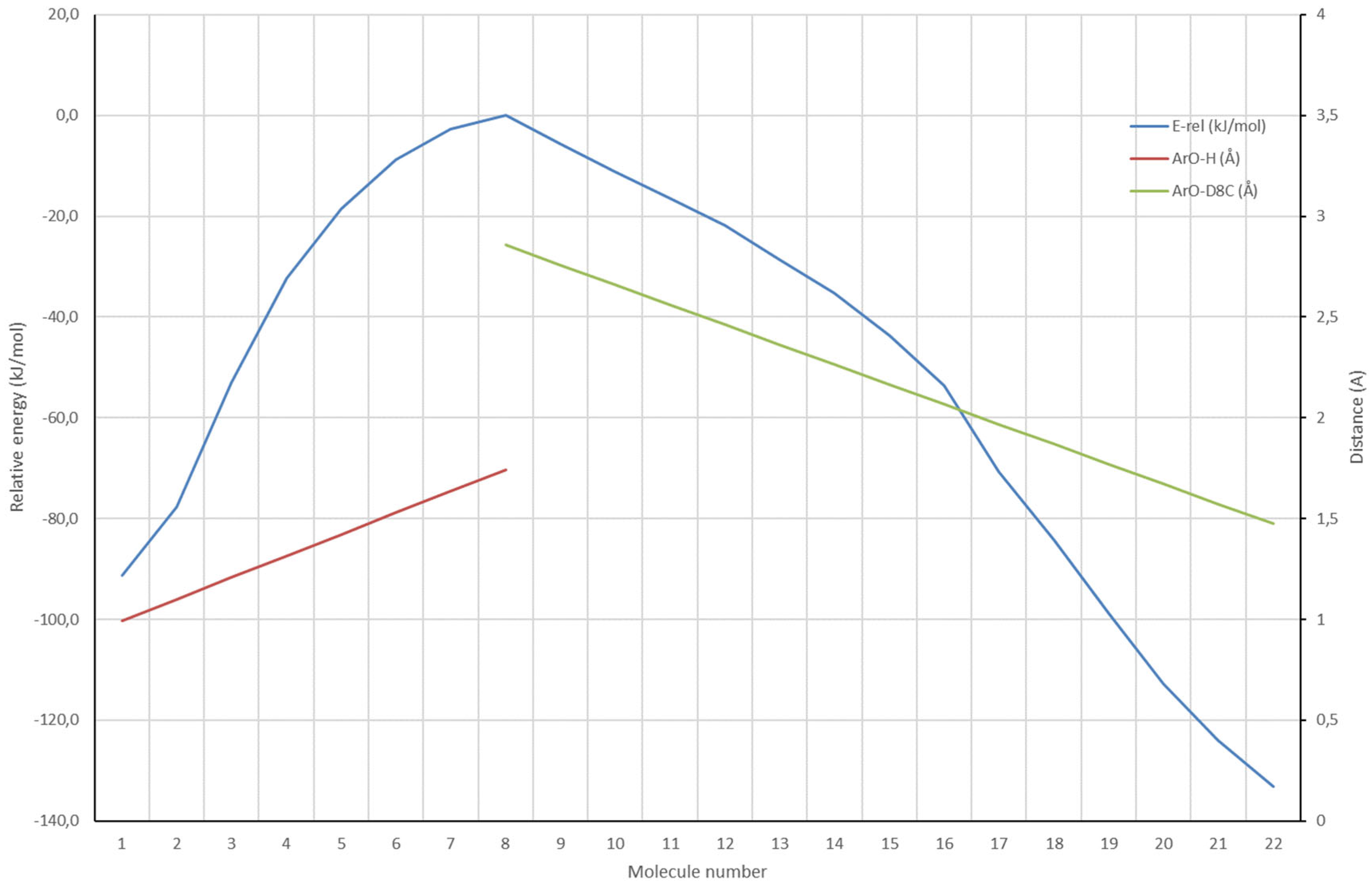
Figure 6 shows the case for the formation of iso Δ
8-THC, starting from BF
3·Δ
9-CBD.
In
Figure 6 two EPs are combined which both start from the TS ring closure iso Δ
8-THC as shown in
Figure 5. The first EP is the reverse of the proton transfer from BF
3·Δ
9-CBD to the Δ
9-C of the cyclohexane ring. This EP ranges from molecule 8-1, and the ArO-H distance changes in steps of ~0.1 Å from 1.743 Å to 0.993 Å, the ArO-H equilibrium distance (red line). The second EP is the movement of the phenolic-O to the Δ
8-C of the cyclohexane ring. This EP ranges from molecule 8-22, and the ArO- Δ
8-C distance changes in steps of ~0.1 Å from 2.857 Å to 1.475 Å, the ArO- Δ
8-C equilibrium distance (green line). The energy curve (blue) does not show additional local maxima or minima. The ΔE from the TS ring closure iso Δ
8-THC to iso Δ
8-THC is -133.2 kJ/mol. A very similar EP was determined showing the formation of Δ
8-CBD, the isomerized double bond product, from the TS ring closure iso Δ
8-THC. That energy curve does not show additional local maxima or minima too. However, the ΔE from the TS ring closure iso Δ
8-THC to iso Δ
8-CBD is -92.0 kJ/mol. So, in principle fully reversible isomerization is very well possible but at sufficiently high reaction rates, the thermodynamic product, iso Δ
8-THC, will be formed exclusively.
3.1.3. Kinetic Models: Comparison of Computational and Experimental Results
Table 2 gives an overview of all activation barriers related to the conversion of Δ
9-CBD to Δ
8-THC, with pTSA and BF
3·Et
2O.
As discussed in 2. Materials and Methods, reaction rates were calculated using:
k = k0*[catalyst]/ [substrate]*e-Ha/RT (s-1)
Thus, k represents a pseudo first order rate constant. As all Transition State structures contain the catalyst, pTSA or BF
3, the pseudo first order rate constant was corrected for the [catalyst]/ [substrate] ratio, as this the maximum amount of substrate that can react in time. The activation barriers for pTSA catalyzed ring closure do lead to pseudo first order rate constants which are far too high. It took some time before it was realized that pTSA in an apolar solvent under dry conditions actually is predominantly present as a dimer with a ΔH = -77.5 kJ/mol. The corresponding equilibrium constant K= 3.31*10
+10, and the fraction pTSA-monomer is 5.50*10
-6 only. Correction for this low amount of monomeric pTSA led to rate constants k
c, listed in the last column of
Table 2.
For the case of catalysis with pTSA a kinetic model was developed using the k
c values for ring closure Δ
9 THC pTSA, ring closure Δ
8-THC pTSA and two-step isomerization Δ
9->Δ
8-CBD pTSA only. The concerted isomerization of Δ
9-CBD is not operative as discussed above, and all other values for k
c are too low to play a role. Using the exact values of
Table 2, a yield of 73.7% Δ
8-THC at a total conversion of Δ
9-CBD of 99.9% was predicted. It seems that overall selectivity to Δ
8-THC is slightly too low, while the overall conversion is slightly too high. Probably more important is the apparent absence of Δ
8-CBD in the reaction mixture after less than 15 minutes, because the presence of Δ
8-CBD was not observed experimentally. Furthermore, the high selectivity to Δ
8-THC instead of Δ
9-THC is explained by the lower activation barriers for two-step isomerization from Δ
9-CBD to Δ
8-CBD and ring closure to Δ
8-THC. Adaptations of +2 kJ/mol) in activation barriers of ring closure of Δ
9 THC and ring closure of Δ
8-THC, lead to an almost perfect fit. The fitted selectivity is 84% compared to 86% experimentally. Alternatively, the ΔH of dimerization of pTSA could be adapted, yielding a very similar result. It should be realized that such small adaptations are within the error limit of the calculations.
For the case of catalysis with BF
3·Et
2O a kinetic model was developed using the k values for ring closure Δ
9 THC BF
3·Et
2O, and ring closure iso Δ
8-THC BF
3·Et
2O. Using the exact values of
Table 2, a yield of 61.9 % Δ
9-THC and 36.5 % iso Δ
8-THC at a total conversion of Δ
9-CBD of 98.5% was predicted. It seems that overall selectivity to Δ
9-THC is slightly too low, while the overall conversion is slightly too high. An adaptation from 80.9 to 81.5 kJ/mol of the activation barrier for ring closure to Δ
9 THC BF
3·Et
2O and an adaptation from 82.1 to 83.5 kJ/mol of the activation barrier for ring closure to iso Δ
8-THC BF
3·Et
2O leads to a perfect fit with 66.6 % Δ
9-THC and 27.6 % iso Δ
8-THC and a total conversion of 94.2%.