Submitted:
23 March 2024
Posted:
25 March 2024
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Chemicals and Reagents
2.2. Standards Solutions
2.3. Matrix Calibrations
2.4. Traceability
2.5. Sample Preparation and Extraction
2.6. Instrumentation
2.7. Method Validation
2.7.1. Linearity
2.7.2. Recovery, Trueness and Accuracy
2.7.3. Decision Limit (CCα) and Detection Capability (CCβ)
3. Results and Discussions
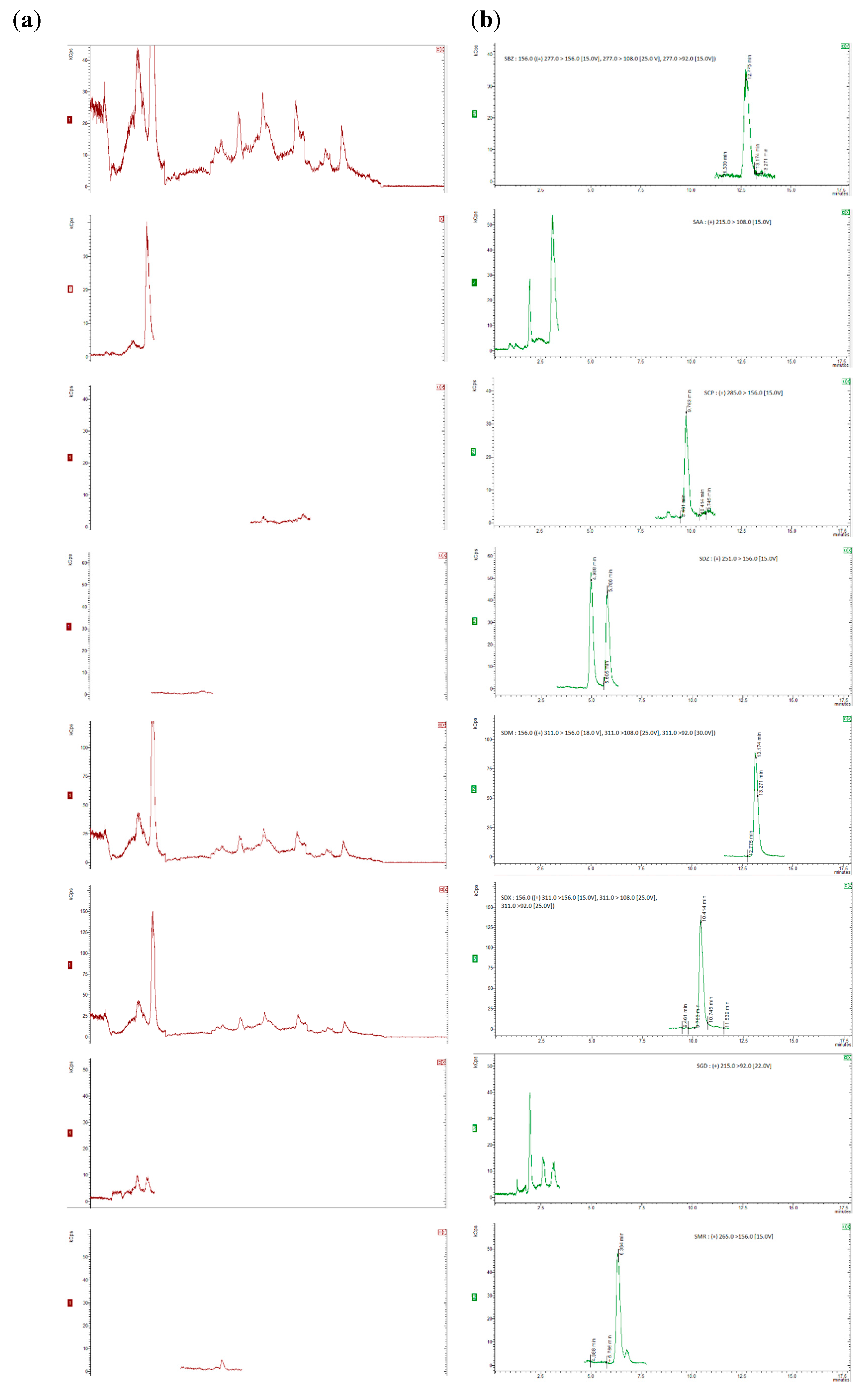
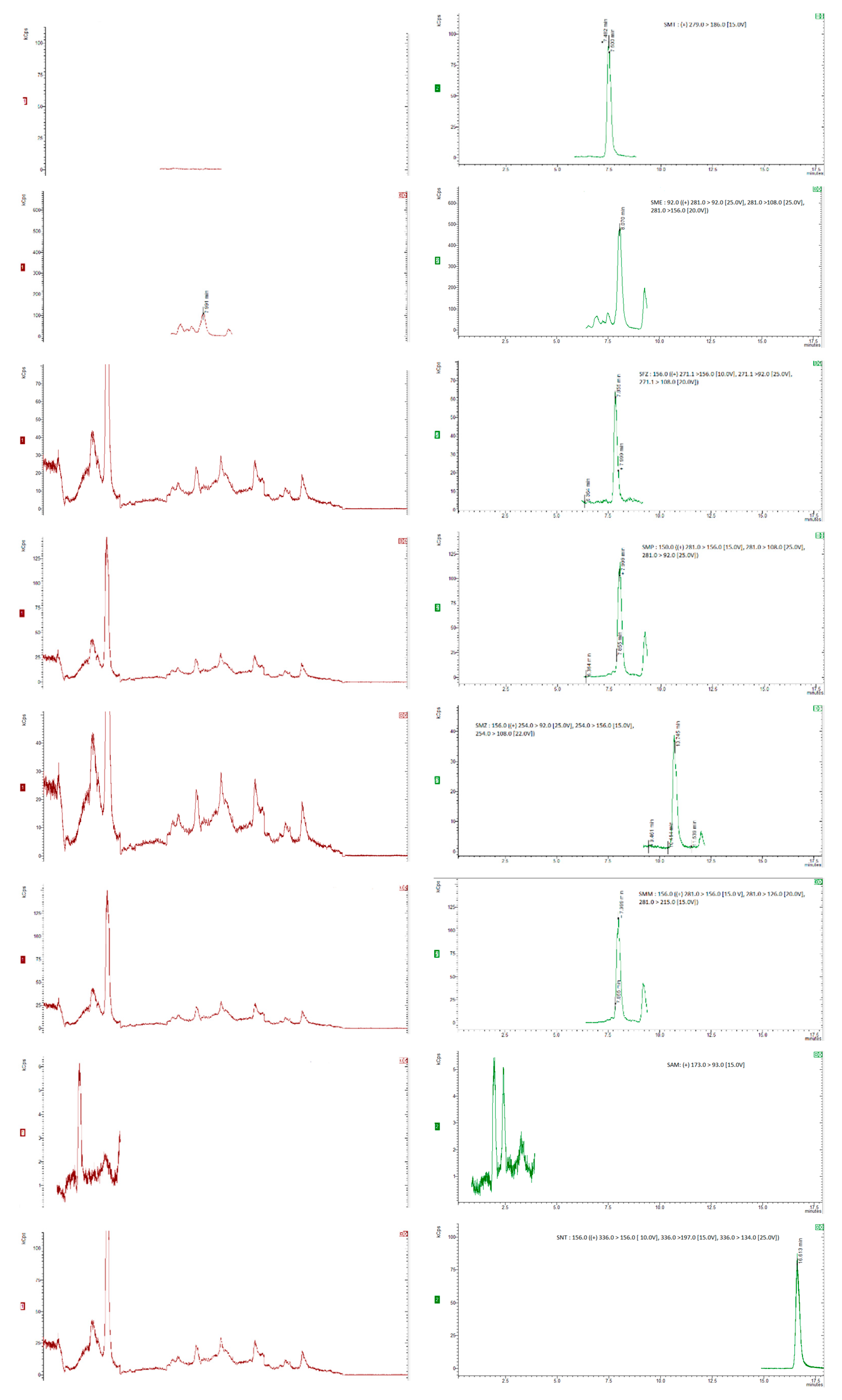
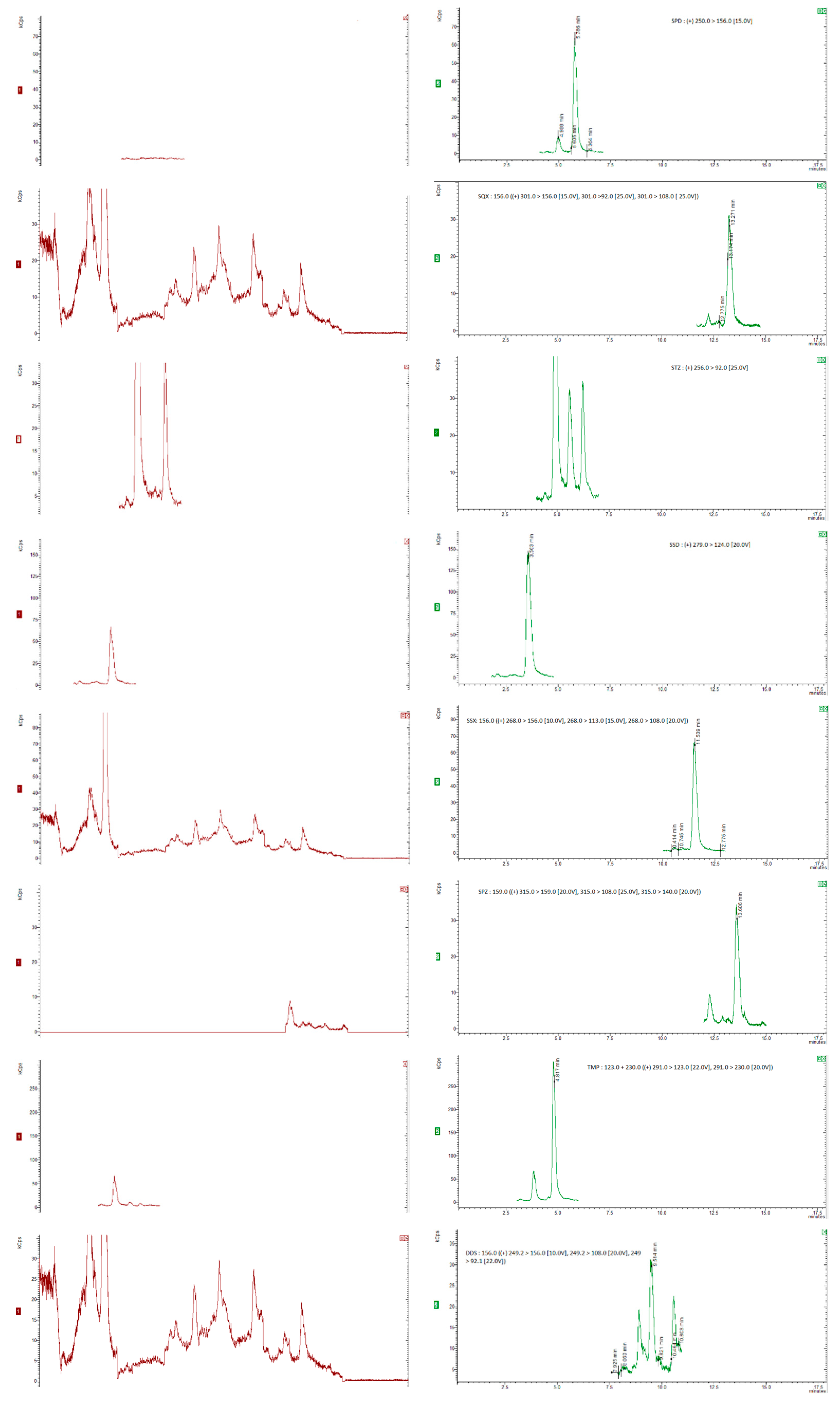
3.1. LC-MS/MS Method Development
3.2. Sample Extraction Procedure
- -
- Sample to be analyzed: 5 g of honey sample mixed with 10 µl of internal standard solutions with concentration of 10 µg/ ml.
- -
- Fortified sample for the determination of recovery: 5 ± 0.5 g honey sample mixed with 250 µl solution of mixed sulfonamides with concentration of 1 µg/ ml, 50 µl solution of dapsone with concentration of 1 µg/ ml, 250 µl solution of trimethoprime with concentration of 1 µg/ ml and 10 µl of internal standard solutions with concentration of 10 µg/ ml.
- -
- Blank sample: contains 5 g of blank honey and 10 µl of internal standard solutions with concentration of 10 µg/ ml.
3.3. Method Validation
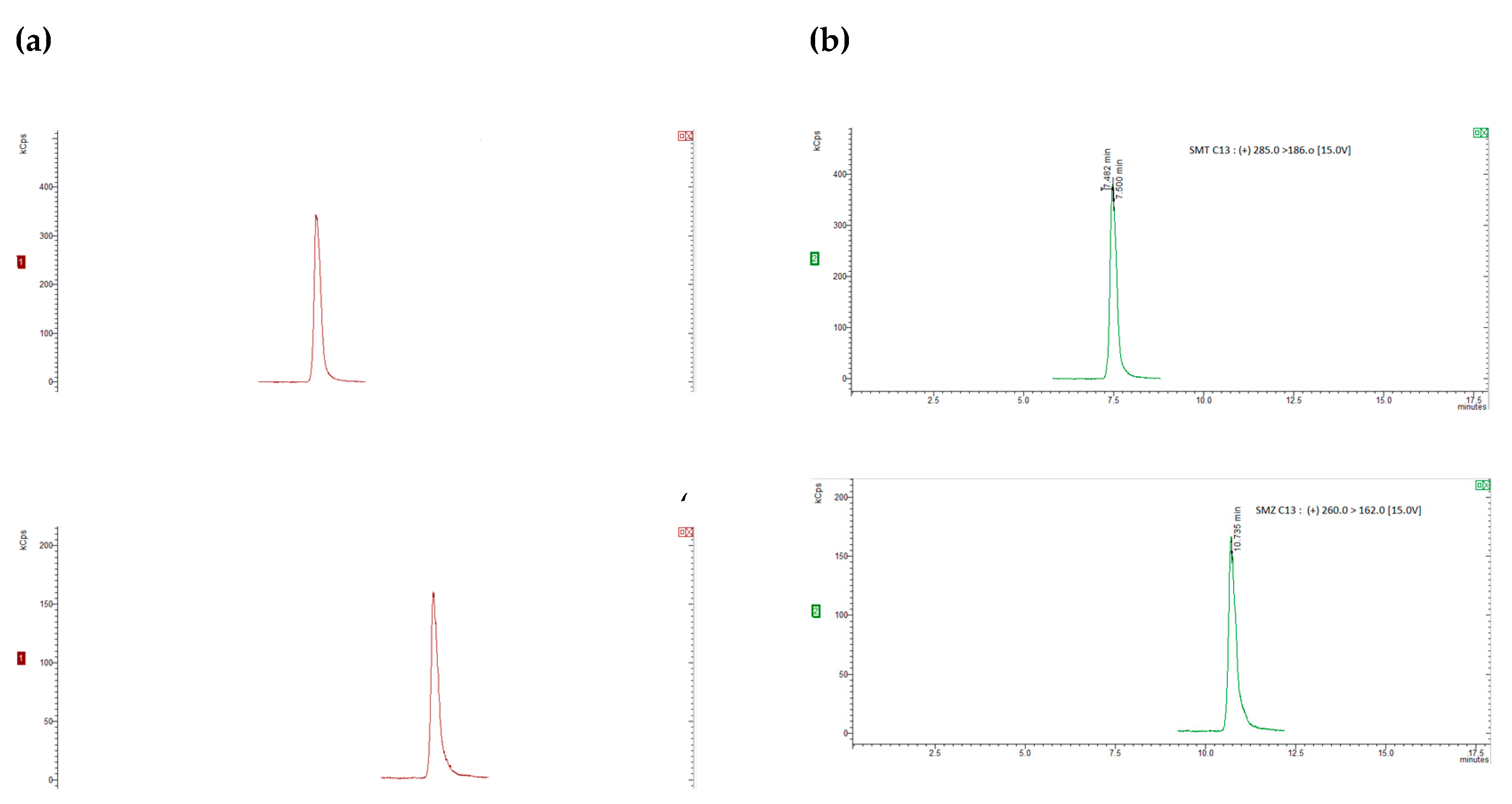
3.3.1. Specificity, Linearity, Sensitivity
S3.3.2. Accuracy
3.3.3. Uncertainty
3.3.4. Decision limits (CCalpha) and detection capabilities (CCbeta)
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Grave, K.; Torren-Edo, J.; Muller, A.; Greko, C.; Moulin, G.; Mackay, D. Variations in the sales and sales patterns of veterinary antimicrobial agents in 25 European countries. J Antimicrob Chemother 2014, 69, 2284–2291. [Google Scholar] [CrossRef]
- Chang, H.; Hu, J.; Asami, M.; Kunikane, S. Simultaneous analysis of 16 sulfonamide and trimethoprim antibiotics in environmental waters by liquid chromatography–electrospray tandem mass spectrometry. Journal of Chromatography A 2008, 1190, 390–393. [Google Scholar] [CrossRef] [PubMed]
- Stolker, A.A.; Brinkman, U.A. Analytical strategies for residue analysis of veterinary drugs and growth-promoting agents in food-producing animals--a review. J Chromatogr A 2005, 1067, 15–53. [Google Scholar] [CrossRef] [PubMed]
- Gentili, A.; Perret, D.; Marchese, S. Liquid chromatography-tandem mass spectrometry for performing confirmatory analysis of veterinary drugs in animal-food products. TrAC Trends in Analytical Chemistry 2005, 24, 704–733. [Google Scholar] [CrossRef]
- Bogdanov, S. Contaminants of bee products. Apidologie 2005, 37. [Google Scholar] [CrossRef]
- Reybroeck, W.; Daeseleire, E.; De Brabander, H.F.; Herman, L. Antimicrobials in beekeeping. Veterinary Microbiology 2012, 158, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Genersch, E.; Evans, J.D.; Fries, I. Honey bee disease overview. Journal of Invertebrate Pathology 2010, 103, S2–S4. [Google Scholar] [CrossRef] [PubMed]
- Dubreil-Chéneau, E.; Pirotais, Y.; Verdon, E.; Hurtaud-Pessel, D. Confirmation of 13 sulfonamides in honey by liquid chromatography–tandem mass spectrometry for monitoring plans: Validation according to European Union Decision 2002/657/EC. Journal of Chromatography A 2014, 1339, 128–136. [Google Scholar] [CrossRef] [PubMed]
- Adams, S.J.; Fussell, R.J.; Dickinson, M.; Wilkins, S.; Sharman, M. Study of the depletion of lincomycin residues in honey extracted from treated honeybee (Apis mellifera L.) colonies and the effect of the shook swarm procedure. Analytica Chimica Acta 2009, 637, 315–320. [Google Scholar] [CrossRef]
- Commission, E. Commission Regulation (EU) No 37/2010 of 22 December 2009 on pharmacologically active substances and their classification regarding maximum residue limits in foodstuffs of animal origin (Text with EEA relevance); Luxembourg, 2010; Vol. L 15, pp. 1–72.
- Commission, E. Council Regulation (EEC) No 2377/90 of 26 June 1990 laying down a Community procedure for the establishment of maximum residue limits of veterinary medicinal products in foodstuffs of animal origin; The Publications Office of the European Union: Luxembourg, 1990; Vol. 224, pp. 1–8. [Google Scholar]
- Commission, E. Regulation (EC) No 470/2009 of the European Parliament and of the Council of 6 May 2009 laying down Community procedures for the establishment of residue limits of pharmacologically active substances in foodstuffs of animal origin, repealing Council Regulation (EEC) No 2377/90 and amending Directive 2001/82/EC of the European Parliament and of the Council and Regulation (EC) No 726/2004 of the European Parliament and of the Council (Text with EEA relevance ); The Publications Office of the European Union: Luxembourg, 2009; Vol. 152, pp. 1–11. [Google Scholar]
- Gaudin, V.; Rault, A.; Verdon, E. Validation of a commercial receptor kit Sulfasensor® Honey for the screening of sulfonamides in honey according to Commission Decision 2002/657/EC. Food Additives & Contaminants: Part A 2012, 29, 942–950. [Google Scholar] [CrossRef]
- Catelani, T.A.; Tóth, I.V.; Lima, J.L.F.C.; Pezza, L.; Pezza, H.R. A simple and rapid screening method for sulfonamides in honey using a flow injection system coupled to a liquid waveguide capillary cell. Talanta 2014, 121, 281–287. [Google Scholar] [CrossRef] [PubMed]
- Granja, R.H.M.M.; Niño, A.M.M.; Rabone, F.; Salerno, A.G. A reliable high-performance liquid chromatography with ultraviolet detection for the determination of sulfonamides in honey. Analytica Chimica Acta 2008, 613, 116–119. [Google Scholar] [CrossRef] [PubMed]
- Zou, Q.-H.; Wang, J.; Wang, X.-F.; Liu, Y.; Han, J.; Hou, F.; Xie, M.-X. Application of Matrix Solid-Phase Dispersion and High-Performance Liquid Chromatography for Determination of Sulfonamides in Honey. Journal of AOAC INTERNATIONAL 2008, 91, 252–258. [Google Scholar] [CrossRef] [PubMed]
- Maudens, K.E.; Zhang, G.-F.; Lambert, W.E. Quantitative analysis of twelve sulfonamides in honey after acidic hydrolysis by high-performance liquid chromatography with post-column derivatization and fluorescence detection. Journal of Chromatography A 2004, 1047, 85–92. [Google Scholar] [CrossRef]
- Tölgyesi, Á.; Berky, R.; Békési, K.; Fekete, S.; Fekete, J.; Sharma, V.K. ANALYSIS OF SULFONAMIDE RESIDUES IN REAL HONEY SAMPLES USING LIQUID CHROMATOGRAPHY WITH FLUORESCENCE AND TANDEM MASS SPECTROMETRY DETECTION. Journal of Liquid Chromatography & Related Technologies 2013, 36, 1105–1125. [Google Scholar] [CrossRef]
- Tsai, W.-H.; Chuang, H.-Y.; Chen, H.-H.; Wu, Y.-W.; Cheng, S.-H.; Huang, T.-C. Application of sugaring-out extraction for the determination of sulfonamides in honey by high-performance liquid chromatography with fluorescence detection. Journal of Chromatography A 2010, 1217, 7812–7815. [Google Scholar] [CrossRef] [PubMed]
- Economou, A.; Petraki, O.; Tsipi, D.; Botitsi, E. Development of a liquid chromatography–tandem mass spectrometry method for the determination of sulfonamides, trimethoprim and dapsone in honey and validation according to Commission Decision 2002/657/EC for banned compounds [corrected]. Talanta 2012, 97, 32–41. [Google Scholar] [CrossRef] [PubMed]
- Verzegnassi, L.; Savoy-Perroud, M.C.; Stadler, R.H. Application of liquid chromatography–electrospray ionization tandem mass spectrometry to the detection of 10 sulfonamides in honey. Journal of Chromatography A 2002, 977, 77–87. [Google Scholar] [CrossRef]
- Tamošiūnas, V.; Padarauskas, A. Comparison of LC and UPLC Coupled to MS–MS for the Determination of Sulfonamides in Egg and Honey. Chromatographia 2008, 67, 783–788. [Google Scholar] [CrossRef]
- Thompson, T.S.; Noot, D.K. Determination of sulfonamides in honey by liquid chromatography–tandem mass spectrometry. Analytica Chimica Acta 2005, 551, 168–176. [Google Scholar] [CrossRef]
- Krivohlavek, A.; Šmit, Z.; Baštinac, M.; Žuntar, I.; Plavšc-Plavšic, F. The determination of sulfonamides in honey by high performance liquid chromatography – mass spectrometry method (LC/MS). Journal of Separation Science 2005, 28, 1434–1439. [Google Scholar] [CrossRef] [PubMed]
- Hammel, Y.-A.; Mohamed, R.; Gremaud, E.; LeBreton, M.-H.; Guy, P.A. Multi-screening approach to monitor and quantify 42 antibiotic residues in honey by liquid chromatography–tandem mass spectrometry. Journal of Chromatography A 2008, 1177, 58–76. [Google Scholar] [CrossRef] [PubMed]
- Lopez, M.I.; Pettis, J.S.; Smith, I.B.; Chu, P.-S. Multiclass Determination and Confirmation of Antibiotic Residues in Honey Using LC-MS/MS. Journal of Agricultural and Food Chemistry 2008, 56, 1553–1559. [Google Scholar] [CrossRef] [PubMed]
- Vidal, J.L.M.; Aguilera-Luiz, M.d.M.; Romero-González, R.; Frenich, A.G. Multiclass Analysis of Antibiotic Residues in Honey by Ultraperformance Liquid Chromatography−Tandem Mass Spectrometry. Journal of Agricultural and Food Chemistry 2009, 57, 1760–1767. [Google Scholar] [CrossRef] [PubMed]
- Sheridan, R.; Policastro, B.; Thomas, S.; Rice, D. Analysis and Occurrence of 14 Sulfonamide Antibacterials and Chloramphenicol in Honey by Solid-Phase Extraction Followed by LC/MS/MS Analysis. Journal of Agricultural and Food Chemistry 2008, 56, 3509–3516. [Google Scholar] [CrossRef] [PubMed]
- Tian, W.; Gao, L.; Zhao, Y.; Peng, W.; Chen, Z. Simultaneous determination of metronidazole, chloramphenicol and 10 sulfonamide residues in honey by LC–MS/MS. Anal. Methods 2013, 5, 1283–1288. [Google Scholar] [CrossRef]
- Commission, E. Council Directive 96/23/EC of 29 April 1996; 1996Vol. L125, pp. 10–32.
- Commission, E. Consolidated text: Commission Decision of 14 August 2002 implementing Council Directive 96/23/EC concerning the performance of analytical methods and the interpretation of results (notified under document number C(2002) 3044) (Text with EEA relevance) (2002/657/EC)Text with EEA relevance. Official Journal 2002, L 221, 8. [Google Scholar]
- Commission, E. SANCO/2004/2726-rev 4-December 2008. GUIDELINES FOR THE IMPLEMENTATION OF DECISION 2002/657/EC 2 Availabe online: https://food.ec.europa.eu/system/files/2016-10/cs_vet-med-residues_cons_2004-2726rev4_en.pdf.
- ISO. ISO-11843-1:1997.Capability of detection. Part1: terms and definitions. International Organization for Standardization (ISO): 1997.
- ISO. ISO-11843-2:2000. Capability of detection.Part 2: methodology in the linear calibration case. International Standard Organization (ISO): 2000.
- Kaufmann, A.; Roth, S.; Ryser, B.; Widmer, M.; Guggisberg, D. Quantitative LC/MS-MS determination of sulfonamides and some other antibiotics in honey. J AOAC Int 2002, 85, 853–860. [Google Scholar] [CrossRef] [PubMed]
- Commission, E. SANCO 2006/3228; 2006.
- Shao, B.; Dong, D.; Wu, Y.; Hu, J.; Meng, J.; Tu, X.; Xu, S. Simultaneous determination of 17 sulfonamide residues in porcine meat, kidney and liver by solid-phase extraction and liquid chromatography–tandem mass spectrometry. Analytica Chimica Acta 2005, 546, 174–181. [Google Scholar] [CrossRef]
- Heller, D.N.; Ngoh, M.A.; Donoghue, D.; Podhorniak, L.; Righter, H.; Thomas, M.H. Identification of incurred sulfonamide residues in eggs: methods for confirmation by liquid chromatography–tandem mass spectrometry and quantitation by liquid chromatography with ultraviolet detection. Journal of Chromatography B 2002, 774, 39–52. [Google Scholar] [CrossRef]
- Zotou, A.; Vasiliadou, C. Selective Determination of Sulfonamide Residues in Honey by SPE-RP-LC with UV Detection. Chromatographia 2006, 64, 307–311. [Google Scholar] [CrossRef]
- Mitrowska, K.; Posyniak, A.; Zmudzki, J. Determination of malachite green and leucomalachite green in carp muscle by liquid chromatography with visible and fluorescence detection. J Chromatogr A 2005, 1089, 187–192. [Google Scholar] [CrossRef] [PubMed]
- Ito, Y.; Oka, H.; Ikai, Y.; Matsumoto, H.; Miyazaki, Y.; Nagase, H. Application of ion-exchange cartridge clean-up in food analysis. V. Simultaneous determination of sulphonamide antibacterials in animal liver and kidney using high-performance liquid chromatography with ultraviolet and mass spectrometric detection. J Chromatogr A 2000, 898, 95–102. [Google Scholar] [CrossRef] [PubMed]
- Commission, E. Commission Decision 2002/657/EC of 12 August 2002; Off. J. Eur. Commun.: 2002; Vol. L 221.
| Compounds | Standard Uncertainty uc (µg/kg) |
Expanded Uncertainty ue (k=2) (µg/kg) |
Final results |
|---|---|---|---|
| Sulfaguanidine | 4,386 | 8,772 | 50 ± 8,772 |
| Sulfacetamide | 4,309 | 8,618 | 50 ± 8,618 |
| Sulfanilamide | 6,155 | 12,310 | 50 ± 12,310 |
| Sulfisomidine | 4,157 | 8,314 | 50 ± 8,314 |
| Sulfadiazine | 3,184 | 6,368 | 50 ± 6,368 |
| Sulfathiazole | 4,030 | 8,060 | 50 ± 8,060 |
| Sulfapyridine | 3,468 | 6,936 | 50 ± 6,936 |
| Sulfamerazine | 3,330 | 6,660 | 50 ± 6,660 |
| Sulfamethazine | 2,773 | 5,546 | 50 ± 5,546 |
| Sulfamethizole | 3,433 | 6,866 | 50 ± 6,866 |
| Sulfamethoxypyridazine | 2,434 | 4,868 | 50 ± 4,868 |
| Sulfamonomethoxine | 3,498 | 6,996 | 50 ± 6,996 |
| Sulfameter | 3,012 | 6,024 | 50 ± 6,024 |
| Sulfachloropyridazine | 3,309 | 6,618 | 50 ± 6,618 |
| Sulfadoxine | 3,613 | 7,226 | 50 ± 7,226 |
| Sulfamethoxazol | 3,167 | 6,334 | 50 ± 6,334 |
| Sulfisoxazole | 3,195 | 6,390 | 50 ± 6,390 |
| Sulfabenzamide | 2,853 | 5,706 | 50 ± 5,706 |
| Sulfadimethoxine | 2,995 | 5,990 | 50 ± 5,990 |
| Sulfaquinoxaline | 3,053 | 6,106 | 50 ± 6,106 |
| Sulfaphenazole | 3,060 | 6,120 | 50 ± 6,120 |
| Sulfanitran | 4,484 | 8,968 | 50 ± 8,968 |
| Trimethoprim | 4,985 | 9,970 | 50 ± 9,970 |
| Dapsone | 0,421 | 0,842 | 5 ± 0,842 |
| Compounds | CCα (µg/kg) | CCβ (µg/kg) |
|---|---|---|
| Sulfaguanidine | 3,50 | 4,33 |
| Sulfacetamide | 2,44 | 3,02 |
| Sulfanilamide | 22,12 | 27,36 |
| Sulfisomidine | 7,78 | 9,62 |
| Sulfadiazine | 2,30 | 2,85 |
| Sulfathiazole | 7,64 | 9,45 |
| Sulfapyridine | 5,58 | 6,90 |
| Sulfamerazine | 4,37 | 5,40 |
| Sulfamethazine | 2,16 | 2,67 |
| Sulfamethizole | 3,53 | 4,37 |
| Sulfamethoxypyridazine | 5,59 | 6,91 |
| Sulfamonomethoxine | 5,94 | 7,35 |
| Sulfameter | 9,36 | 11,57 |
| Sulfachloropyridazine | 7,20 | 8,91 |
| Sulfadoxine | 7,35 | 9,09 |
| Sulfamethoxazol | 7,89 | 9,76 |
| Sulfisoxazole | 7,87 | 9,74 |
| Sulfabenzamide | 7,66 | 9,48 |
| Sulfadimethoxine | 7,61 | 9,42 |
| Sulfaquinoxaline | 8,22 | 10,16 |
| Sulfaphenazole | 8,34 | 10,32 |
| Sulfanitran | 9,52 | 11,77 |
| Trimethoprim | 7,67 | 9,48 |
| Dapsone | 2,45 | 3,03 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




