Submitted:
06 December 2023
Posted:
08 December 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
| Authors | TiO2 (%) in the raw material | Processes |
|---|---|---|
| Chao and Senkler (1991) | 74,5 | Upgrade: chloride leaching, alkaline leaching, chloride leaching |
| Chao et al. (1993) | 62,9 | Upgrade: a reducing roasting, magnetic separation, pressure chloride leaching |
| Freitas et al. (2007) | 53,8 | Upgrade: calcination, reduction in H2, magnetic separation, chloride leaching |
| Freitas and Gracioso (1985) | 70,0 | Chloride leaching, alkaline digestion, sulphuric digestion, hydrolysis- precipitation |
| Jha and Tathavadkar (2005) | 57,8 | Alkaline digestion, leaching with water, |
| Patent: Mineração Vale do Paranaíba (1980) | 78,1 | Upgrade: Acid digestion under pressure, chloride leaching, magnetic separation |
| Paixão and Mendonça (1979) | 75,6 | Upgrade: roasting, magnetic separation, HCl chloride leaching, NaOH neutralization |
| Smith Jr and Castro Sheldon (2007) | 52,9 | Upgrade: leaching in autoclave with H2SO4 e FeSO4, drying with NaCl addition, calcination, chloride leaching |
| Trindade e Teixeira (1988) | 76,5 | Upgrade: chloride leaching in 4 stages |
2. Materials and experimental procedure
2.1. Material
2.2. Chemical and mineralogical characterization
2.3. Sulphuric digestion
3. Results and discussion
3.1. Chemical and mineralogical characterization of the anatase concentrate
3.2. Sulphuric acid digestion
3.2.1. Effect of Temperature on sulphuric digestion of the concentrate
3.2.2. Effect of the anatase:sulphuric acid ratio in the digestion
3.2.3. Effect of time in sulphuric digestion
3.2.4. Effect of using dilute sulphuric acid as a leaching agent
4. Conclusion
Acknowledgments
References
- Aguiar, E.M.M.M. Recuperação de titânio e vanádio de fonte secundária. 2021. Dissertation (Master’s in Chemical Engineering)—USP (Escola Politécnica da Universidade de São Paulo), São Paulo, 2021. [CrossRef]
- Barnard, K.R.; Mcdonald, R.G.; Pownceby, M.I.; Sparrow, G.J.; Zhang, W. Processing anatase ores for pigment production. Hydrometallurgy, v. 185, p. 226-237, 2019. [CrossRef]
- Baltar, C.A.M.; Sampaio, J.A.; Andrade, M.C.; Pinto, D.C. Minerais de Titânio. In: LUZ, A.B.; LINS, F.A.F. (org.). Rochas & Minerais Industriais. 2 ed.Rio de Janeiro: CETEM/MCT, v. 1 c.37, p. 841-864, 2009.
- Bekker, J.H.; Dutton, D.F., 2004. Recovery of titanium dioxide from titanium oxide bearing materials like steelmaking slags. PCT Patent US2004/0136899 A1. Date of filing: December 12, 2000; Date of publication: July 15, 2004.
- Chao, N.T.; G.H.; Senkler, G.J. 1991. Method for purifying TiO2 ore. US Patent 5011666; Date of filing: July 28, 1988. Date of publication: April 30, 1991.
- Chao, T., Kremer, W.L., Fonseca Mourão, M.J., Jardim Paixão, J.M., 1993. Process for purifying anatase TiO2 ore. PCT Patent WO93/22465. Date of filing: May 01, 1992; Date of publication: November 11, 1993.
- Chaves, N. 1978. Processo de utilização de concentrado de anatásio como matéria prima para fabricação de pigmento de titânio pelo processo sulfato. PI 7605001. Date of filing: July 30, 1976; Date of publication: February 14, 1978.
- Demol, J.;Ho, E.; Senanayake, G. Sulfuric acid baking and leaching of rare earth elements, thorium and phosphate from a monazite concentrate: Effect of bake temperature from 200 to 800 °C. Hydrometallurgy, v. 179, p. 254-267, 2018. [CrossRef]
- Freitas, L.R.; Brocchi, E.A. Digestão Sulfúrica de Materiais à base de Titânio. In: ENCONTRO NACIONAL DE TRATAMENTO DE MINÉRIOS E METALURGIA EXTRATIVA, 16, 1995, p. 17–30.
- Freitas, L.R.; Gracioso, J.E. Abertura do anatásio por sulfatação. In: ENCONTRO NACIONAL DE TRATAMENTO DE MINÉRIOS E HIDROMETALURGIA, 11, 1985, Natal, v. II, p. 96- 108.
- Freitas, L.R., Horta, R.M., Tude, J.A.L., 2007. Process for enrichment of anatase mechanical concentrates in order to obtain synthetic rutile with low contents of rare earths and radioactive elements. PCT Patent WO2007/048210 A1. Date of filing: May 03, 2007; Date of publication: November 17, 2008.
- Gao, L.; Rao, B.; Daí, H.; Xie, H.; Wang, P.; Ma, F. Kinetics of sulphuric acid leaching of titanium from refractory anatase under atmospheric pressure. Physicochemical Problems of Mineral Processing, v. 55, n. 2, p. 467–478, 2019. [CrossRef]
- Gazquez, M.J.,; Bolívar, J.P.; Garcia-Tenorio, R.; Vaca, F. A review of the production cycle of Titanium Dioxide Pigment. Materials Sciences and Applications, 2014, 5, 441–458. [CrossRef]
- Gontijo, V.L.; Teixeira, L.A.V.; Ciminelli, V.S.T. The reactivity of iron oxides and hydroxide during low-temperature sulphation. Hydrometallurgy, v. 197, 105452, 2020. [CrossRef]
- Jablonski, M.; Tylutka, S. The influence of initial concentration of sulfuric acid on the degree of leaching of the main elements of ilmenite raw materials. Journal of Thermal Analysis and Calorimetry, v. 124, p. 355- 361, 2016. [CrossRef]
- Jha, A.; Tathavadkar, V.D., 2005. Process for the recovery of titanium dioxide from titanium- containing compositions. PCT Patent WO2005/028369 A1. Date of filing: September 18, 2003; Date of publication: May 19, 2005.
- Lakshmanan, V.I.; Bhowmick, A.; Halim, M.A. Titanium Dioxide: Production, Properties and Applications. In: BROWN, J. Titanium dioxide: chemical properties, applications, and environmental effects. New York: Nova Science Publishers, Inc., 2014, cap. 5, 75-130.
- Lane, G.R.; Martin, C.;Pirard, E. Techniques and applications for predictive metallurgy and ore characterization using optical image analysis. Minerals Engineering, v. 21, p. 568-577, 2008. [CrossRef]
- Mineração Vale Do Paranaíba, S.A., 1980. A method for obtaining higher TiO2 grade anatase concentrates from lower TiO2 grade anatase concentrates. GB Patent 1568333. Date of filing: March 09, 1977; Date of publication: May 29, 1980.
- Paixão, J.M.J., Mendonça, P.A.F., 1979. Process for concentration of titanium containing anatase ore. US Patent 4176159. Date of filing: June 19, 1978; Date of publication: November 27, 1979.
- Queiroz Filho, A.A.; Amorim Neto, A.A. 2019. Titânio. In: Agência Nacional de Mineração, Sumário Mineral 2017. Brasília: ANM, vol 37, 167-169. ISSN: 0101-2053. Available online: https://www.gov.br/anm/pt-br/centrais-de-conteudo/publicacoes/serie-estatisticas-e-economia-mineral/sumario-mineral/sumariomineral_2017.
- Smith Jr., E.M., De Castro Sheldon, A., 2007. Titaniferous ore beneficiation. PCT Patent WO2007/046975 A2. Date of filing: October 18, 2005; Date of publication: April 26, 2007.
- Tian, C. Internal influences of hydrolysis conditions on rutile TiO2 pigment. [CrossRef]
- production via short sulfate process. Materials Research Bulletin, v. 103, p. 83-88, 2018.
- Trindade, R.B.E.; Teixeira, L.A. Beneficiamento de concentrado de titânio (anatásio) por lixiviação oxidante de impurezas. In: ENCONTRO NACIONAL DE TRATAMENTO DE MINÉRIOS E HIDROMETALURGIA, 12, 1988, São Paulo, p. 823- 836.
- Sukla, L.B.; Panda, S.C.; Jena, P.K. Recovery of cobalt, nickel and copper from converter slag through roasting with ammonium sulphate and sulphuric acid. Hydrometallurgy, v. 16, p. 153-165, 1986. [CrossRef]
- Xue, T.; Wang, L.; Qi, T.; Chu, J.; Qu, J.; Liu, C. Decomposition kinectics of titanium slag in sodium hydroxide system. Hydrometallurgy, v. 95, p. 22- 27, 2009. [CrossRef]
- Wang, Y.; Li, J.; Wang, L.; Xue, T., Qi, T. Preparation of Rutile Titanium Dioxide White Pigment via Doping and Calcination of Metatitanic Acid Obtained by the NaOH Molten Salt Method. Industrial & Engineering Chemistry Research, v. 49, p. 7693-7696, 2010. [CrossRef]
- ZHANG, W.; ZHU, Z.; CHENG, C.Y. A literature review of titanium metallurgical processes. Hydrometallurgy, v. 108, 177-188, 2011. [CrossRef]


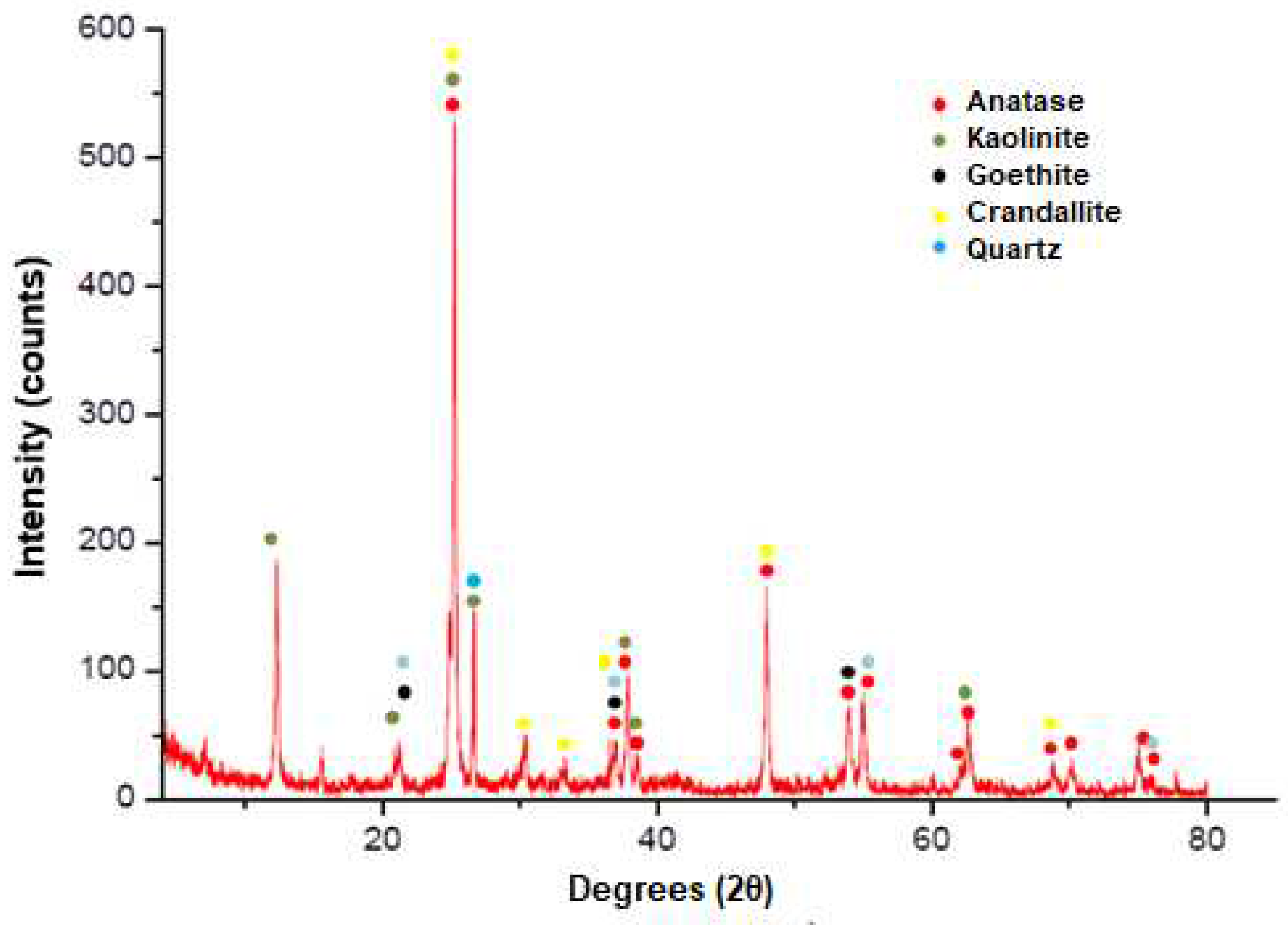
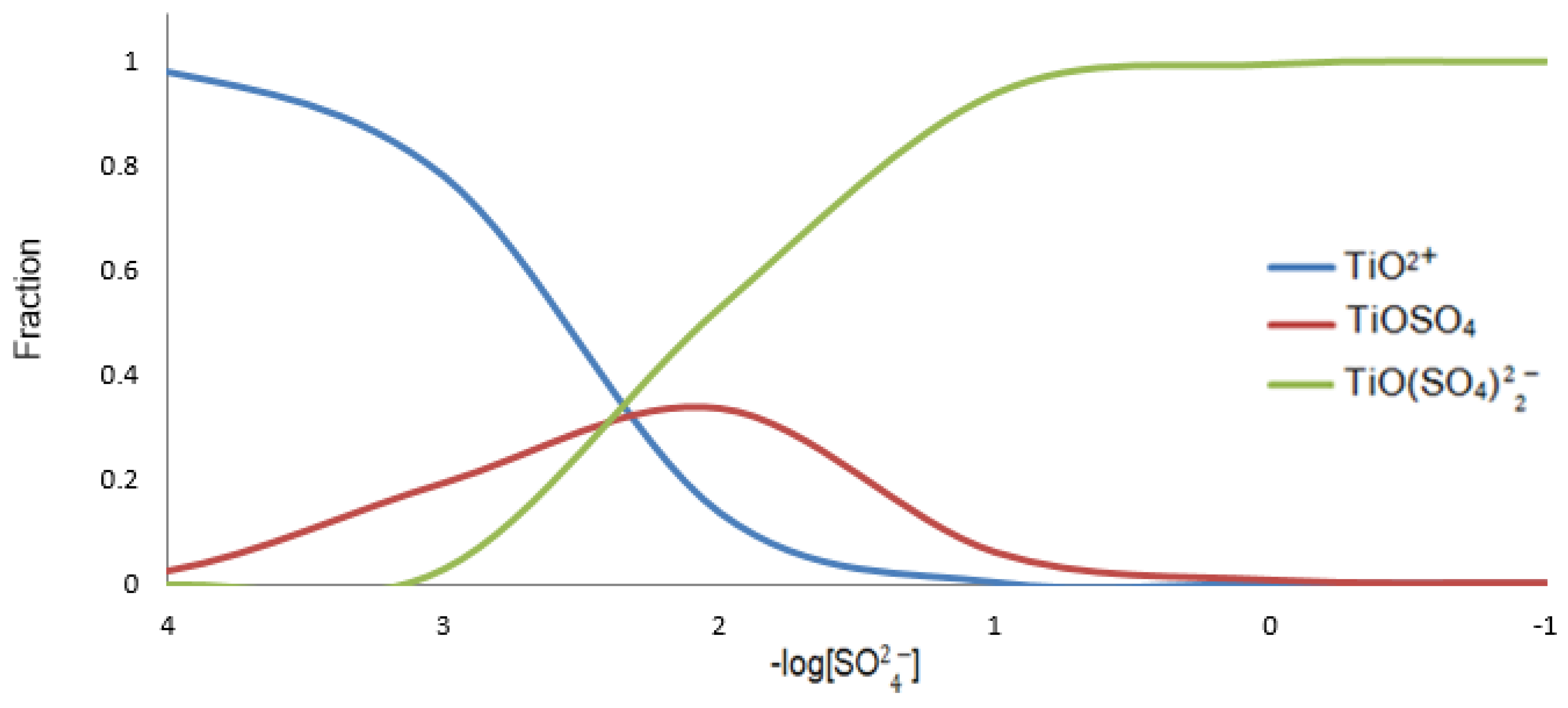
| Elements | Anatase concentrate |
|---|---|
| TiO2 | 56.5 |
| Fe2O3 | 15.0 |
| P2O5 | 4.11 |
| SiO2 | 6.01 |
| Al2O3 | 5.34 |
| CaO | 1.61 |
| ZrO2 | 0.59 |
| Nb2O5 | 0.62 |
| REE | 2.19 |
| Temperature (°C) | TiO2 (%) | Fe2O3 (%) | P2O5 (%) | ZrO2 (%) | Nb2O5 (%) | Mass reduction (%) | |
|---|---|---|---|---|---|---|---|
| 190 | Residue | 57.2 | 8.14 | 1.60 | 0.828 | 1.08 | 68.9 |
| Metallurgical recovery | 68.6 | 83.1 | 87.9 | 56.3 | 45.9 | ||
| 200 | Residue | 50.9 | 7.52 | 2.59 | 0.506 | 0.954 | 63.4 |
| Metallurgical recovery | 67.0 | 81.6 | 76.9 | 68.6 | 43.4 | ||
| 210 | Residue | 47.8 | 8.24 | 2.71 | 0.629 | 1.04 | 70.9 |
| Metallurgical recovery | 75.4 | 84.1 | 80.8 | 68.9 | 51.0 | ||
| 220 | Residue | 35.8 | 7.87 | 4.40 | 0.460 | 0.657 | 78.0 |
| Metallurgical recovery | 86.0 | 88.4 | 76.5 | 82.9 | 76.4 |
| Ratio Concentrate: H2SO4 | TiO2 (%) | Fe2O3 (%) | P2O5 (%) | ZrO2 (%) | Nb2O5 (%) | Mass reduction (%) | |
|---|---|---|---|---|---|---|---|
| 1:1.3 | Residue | 51.9 | 7.62 | 2.32 | 0.461 | 0.997 | 69.0 |
| Metallurgical recovery (%) | 71.6 | 84.5 | 82.2 | 76.1 | 50.4 | ||
| 1:2 | Residue | 35.8 | 7.87 | 4.40 | 0.460 | 0.657 | 78.0 |
| Metallurgical recovery (%) | 86.0 | 88.4 | 76.5 | 82.9 | 76.4 |
| Sulphuric digestion time (h) | TiO2 (%) | Fe2O3 (%) | P2O5 (%) | ZrO2 (%) | Nb2O5 (%) | Mass reduction (%) | |
|---|---|---|---|---|---|---|---|
| 3 | Residue | 39.3 | 8.72 | 3.41 | 0.524 | 0.964 | 87.3 |
| Metallurgical recovery (%) | 82.3 | 85.2 | 78.9 | 77.4 | 60.3 | ||
| 4 | Residue | 35.0 | 7.61 | 3.85 | 0.388 | 0.658 | 78.0 |
| Metallurgical recovery (%) | 86.3 | 88.8 | 79.4 | 85.5 | 76.4 | ||
| 5 | Residue | 44.0 | 7.43 | 3.17 | 0.437 | 0.823 | 72.0 |
| Metallurgical recovery (%) | 78.2 | 86.1 | 78.4 | 79.2 | 62.7 |
| Leaching agent | TiO2 (%) | Fe2O3 (%) | P2O5 (%) | ZrO2 (%) | Nb2O5 (%) | Mass reduction (%) | |
|---|---|---|---|---|---|---|---|
| H2SO4 5% | Residue | 40.7 | 6.44 | 3.54 | 0.483 | 0.87 | 75.3 |
| Metallurgical recovery (%) | 81.2 | 88.9 | 77.7 | 78.42 | 63.2 | ||
| Milli-Q water | Residue | 35.8 | 7.87 | 4.40 | 0.460 | 0.657 | 78.0 |
| Metallurgical recovery (%) | 86.0 | 88.4 | 76.5 | 82.9 | 76.4 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




