Submitted:
27 November 2023
Posted:
29 November 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Results
2.1. Detection of autoantibodies in serum samples of patients
2.2. Characterization of the exome data
2.3. Analysis of genetic variants
2.4. Overview of HLA system risk and protective alleles/haplotypes in studied families and controls.
3. Discussion
4. Materials and Methods
4.1. Patients
4.2. Clinical samples and genomic DNA isolation
4.3. Detection of autoantibodies in serum samples by ELISA kits and microarray-based assay
4.4. Exome Sequencing and Genetic Examination
4.5. High-Resolution HLA Typing
- -
- quality threshold for reads (low quality reads were trimmed or discarded);
- -
- lowest absolute and relative coverage for each position;
- -
- the highest number of differences (insertions, substitutions, deletions) from the group average for each read;
- -
- maximum relative position error - the number of differences (insertions, substitutions, deletions) from the consensus sequence in each position should not exceed the specified threshold;
- -
- the highest average error per read for a group;
- -
- the lowest number of reads in groups for each exon (I-class 2,3,4 exons, II-class-2,3 exons);
- -
- the allelic imbalance should not exceed a given threshold; the ratio of the read number for the exons from each allele and the sum of these ratios;
- -
- the presence of phantom (cross-mapping) and chimeric sequences;
- -
- the percentage of combined, clustered, and used for typing reads computed for each sample.
4.6. Sanger Sequencing
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Kochi, Y. Genetics of autoimmune diseases: perspectives from genome-wide association studies. Int. Immunol. 2016, 28, 155-161. [CrossRef]
- Anaya, J.M.; Rojas-Villarraga, A.; García-Carrasco, M. The autoimmune tautology: From polyautoimmunity and familial autoimmunity to the autoimmune genes. Autoimmune Dis. 2012, 2012, 297193. [CrossRef]
- Rojas, M.; Ramírez-Santana, C.; Acosta-Ampudia, Y.; Monsalve, D.M.; Rodriguez-Jimenez, M.; Zapata, E.; Naranjo-Pulido, A.; Suárez-Avellaneda, A.; Ríos-Serna, L.J.; Prieto, C.; et al. New insights into the taxonomy of autoimmune diseases based on polyautoimmunity. J. Autoimmun. 2022, 126, 102780. [CrossRef]
- Charmandari, E.; Nicolaides, N.C.; Chrousos, G.P. Adrenal insufficiency. Lancet 2014, 383, 2152-2167. [CrossRef]
- Ruggeri, R.M.; Giuffrida, G.; Campennì, A. Autoimmune endocrine diseases. Minerva Endocrinol. 2018, 43, 305-322. [CrossRef]
- Su, M.A.; Anderson, M.S. Monogenic autoimmune diseases: insights into self-tolerance. Pediatr. Res. 2009, 65, 20R-25R. [CrossRef]
- Betterle, C.; Dal Pra, C.; Mantero, F.; Zanchetta, R. Autoimmune adrenal insufficiency and autoimmune polyendocrine syndromes: autoantibodies, autoantigens, and their applicability in diagnosis and disease prediction. Endocr. Rev. 2002, 23, 327-364. [CrossRef]
- Betterle, C.; Furmaniak, J.; Sabbadin, C.; Scaroni, C.; Presotto, F. Type 3 autoimmune polyglandular syndrome (APS-3) or type 3 multiple autoimmune syndrome (MAS-3): an expanding galaxy. J. Endocrinol. Invest. 2023, 46, 643-665. [CrossRef]
- Vargas-uricoechea, H.; Nogueira, J.P.; Pinz, V.; Schwarzstein, D. The Usefulness of Thyroid Antibodies in the Diagnostic Approach to Autoimmune Thyroid Disease. Antibodies 2023, 12. [CrossRef]
- Torii, S. Expression and function of IA-2 family proteins, unique neuroendocrine-specific protein-tyrosine phosphatases. Endocr. J. 2009, 56, 639-648. [CrossRef]
- Yi, L.; Swensen, A.C.; Qian, W.J. Serum biomarkers for diagnosis and prediction of type 1 diabetes. Transl. Res. 2018, 201, 13-25. [CrossRef]
- Øksnes, M.; Husebye, E.S. Approach to the patient: Diagnosis of primary adrenal insufficiency in adults. J. Clin. Endocrinol. Metab. 2023, dgad402. [CrossRef]
- Meager, A.; Visvalingam, K.; Peterson, P.; Möll, K.; Murumägi, A.; Krohn, K.; Eskelin, P.; Perheentupa, J.; Husebye, E.; Kadota, Y.; et al. Anti-interferon autoantibodies in autoimmune polyendocrinopathy syndrome type 1. PLoS Med. 2006, 3, 1152–1164. [CrossRef]
- Kisand, K.; Bøe Wolff, A.S.; Podkrajšek, K.T.; Tserel, L.; Link, M.; Kisand, K. V.; Ersvaer, E.; Perheentupa, J.; Erichsen, M.M.; Bratanic, N.; et al. Chronic mucocutaneous candidiasis in APECED or thymoma patients correlates with autoimmunity to Th17-associated cytokines. J. Exp. Med. 2010, 207, 299–308. [CrossRef]
- Chung, S.A.; Criswell, L,A. PTPN22: Its role in SLE and autoimmunity. Autoimmunity 2007, 40, 582–590. [CrossRef]
- Fichna, M.; Małecki, P.P.; Żurawek, M.; Furman, K.; Gębarski, B.; Fichna, P.; Ruchała, M. Genetic variants and risk of endocrine autoimmunity in relatives of patients with Addison's disease. Endocr. Connect. 2023, 12, e230008. [CrossRef]
- Gough, S.C.; Simmonds, M.J. The HLA Region and Autoimmune Disease: Associations and Mechanisms of Action. Curr. Genomics 2007, 8, 453-65. [CrossRef]
- Shin, D.H.; Baek, I.C.; Kim, H.J.; Choi. E.J.; Ahn, M.; Jung, M.H.; Suh, B.K.; Cho, W.K.; Kim, T.G. HLA alleles, especially amino-acid signatures of HLA-DPB1, might contribute to the molecular pathogenesis of early-onset autoimmune thyroid disease. PLoS One 2019, 14, e0216941. [CrossRef]
- Gambelunghe, G.; Falorni, A.; Ghaderi, M.; Laureti, S.; Tortoioli, C.; Santeusanio, F.; Brunetti, P.; Sanjeevi, C.B. Microsatellite polymorphism of the MHC class I chain-related (MIC-A and MIC-B) genes marks the risk for autoimmune Addison's disease. J. Clin. Endocrinol. Metab. 1999, 84, 3701-3707. [CrossRef]
- Savvateeva, E.N.; Yukina, M.Y.; Nuralieva, N.F.; Filippova, M.A.; Gryadunov, D.A., Troshina, E.A. Multiplex Autoantibody Detection in Patients with Autoimmune Polyglandular Syndromes. Int. J. Mol. Sci. 2021, 22, 5502. [CrossRef]
- Nuralieva, N.; Yukina, M.; Sozaeva, L.; Donnikov, M.; Kovalenko, L.; Troshina, E.; Orlova, E.; Gryadunov, D.; Savvateeva, E.; Dedov, I. Diagnostic Accuracy of Methods for Detection of Antibodies against Type I Interferons in Patients with Endocrine Disorders. J. Pers. Med. 2022, 12, 1948. [CrossRef]
- Frommer, L.; Kahaly, G.J. Autoimmune Polyendocrinopathy. J. Clin. Endocrinol. Metab. 2019, 104, 4769-4782. [CrossRef]
- Eriksson, D.; Røyrvik, E.C.; Aranda-Guillén, M.; Berger, A.H.; Landegren, N.; Artaza, H.; Hallgren, Å.; Grytaas, M.A.; Ström, S.; Bratland, E. et al. GWAS for autoimmune Addison's disease identifies multiple risk loci and highlights AIRE in disease susceptibility. Nat. Commun. 2021, 12, 959. [CrossRef]
- Köhler, S.; Gargano, M.; Matentzoglu, N.; Carmody, L.C.; Lewis-Smith, D.; Vasilevsky, N.A.; Danis, D.; Balagura, G.; Baynam, G.; Brower, A.M.; et al. The Human Phenotype Ontology in 2021. Nucleic Acids Res. 2021, 8, D1207-D1217. [CrossRef]
- Pereira, J.S.; da Silva, J.G.; Tomaz, R.A.; Pinto, A.E.; Bugalho, M.J.; Leite, V.; Cavaco, B.M. Identification of a novel germline FOXE1 variant in patients with familial non-medullary thyroid carcinoma (FNMTC). Endocrine 2015, 49, 204-214. [CrossRef]
- Hwangbo, Y.; Park, Y.J. Genome-Wide Association Studies of Autoimmune Thyroid Diseases, Thyroid Function, and Thyroid Cancer. Endocrinol. Metab. (Seoul). 2018, 33, 175-184. [CrossRef]
- Lim, G.; Widiapradja, A.; Levick, S.P.; McKelvey, K.J.; Liao, X.H.; Refetoff, S.; Bullock, M.; Clifton-Bligh, R.J. Foxe1 Deletion in the Adult Mouse Is Associated With Increased Thyroidal Mast Cells and Hypothyroidism. Endocrinology 2022 163, bqac158. [CrossRef]
- Richards, S.; Aziz, N.; Bale, S.; Bick, D.; Das, S.; Gastier-Foster, J.; Grody, W.W.; Hegde, M.; Lyon, E.; Spector, E.; et al. Standards and guidelines for the interpretation of sequence variants: a joint consensus recommendation of the American College of Medical Genetics and Genomics and the Association for Molecular Pathology. Genet Med. 2015, 17, 405-424. [CrossRef]
- Yukina, M.Y.; Larina, A.A.; Vasilyev, E.V.; Troshina, E.A.; Dimitrova, D.A. Search for Genetic Predictors of Adult Autoimmune Polyendocrine Syndrome in Monozygotic Twins. Clin. Med. Insights Endocrinol. Diabetes 2021, 14, 11795514211009796. [CrossRef]
- Tanaka, N.; Abe-Dohmae, S.; Iwamoto, N.; Fitzgerald, M.L.; Yokoyama, S. Helical apolipoproteins of high-density lipoprotein enhance phagocytosis by stabilizing ATP-binding cassette transporter A7. J. Lipid Res. 2010, 51, 2591-2599. [CrossRef]
- Yamazaki, K.; Yoshino, Y.; Kawabe, K.; Ibuki, T.; Ochi, S.; Mori, Y.; Ozaki, Y.; Numata, S.; Iga, J.I.; Ohmori, T.; et al. ABCA7 Gene Expression and Genetic Association Study in Schizophrenia. Neuropsychiatr. Dis. Treat. 2020, 16, 441-446. [CrossRef]
- Oftedal, B.E.; Berger, A.H.; Bruserud, Ø.; Goldfarb, Y.; Sulen, A.; Breivik, L.; Hellesen, A.; Ben-Dor, S.; Haffner-Krausz, R.; Knappskog, P.M.; et al. A partial form of AIRE deficiency underlies a mild form of autoimmune polyendocrine syndrome type 1. J. Clin. Invest. 2023, 133, e169704. [CrossRef]
- Yukina, M.Yu.; Nuralieva, N.F.; Troshina, E.A. Analysis of the prevalence and incidence of adrenal insufficiency in the world. Ateroscleroz 2022, 18, 426-429. [CrossRef]
- Ueda, H.; Howson, J.M.; Esposito, L.; Heward, J.; Snook, H.; Chamberlain, G.; Rainbow, D.B.; Hunter, K.M.; Smith, A.N.; Di Genova, G.; et al. Association of the T-cell regulatory gene CTLA4 with susceptibility to autoimmune disease. Nature 2003, 423, 506-511. [CrossRef]
- Maine, C.J.; Hamilton-Williams, E.E.; Cheung, J.; Stanford, S.M.; Bottini, N.; Wicker, L.S.; Sherman, L.A. PTPN22 alters the development of regulatory T cells in the thymus. J. Immunol. 2012, 188, 5267-5275. [CrossRef]
- Park, Y.J.; Yoo, S.A.; Kim, M.; Kim, W.U. The Role of Calcium-Calcineurin-NFAT Signaling Pathway in Health and Autoimmune Diseases. Front Immunol. 2020, 11, 195. [CrossRef]
- Napier, C.; Mitchell, A.L.; Gan, E.; Wilson, I.; Pearce, S.H. Role of the X-linked gene GPR174 in autoimmune Addison's disease. J. Clin. Endocrinol. Metab. 2015, 100, E187-E190. [CrossRef]
- Agliardi, C.; Guerini, F.R.; Bolognesi, E.; Zanzottera, M.; Clerici, M. VDR Gene Single Nucleotide Polymorphisms and Autoimmunity: A Narrative Review. Biology (Basel) 2023, 12, 916. [CrossRef]
- Fan, H.H.; Cui, L.; Jiang, X.X.; Song, Y.D.; Liu, S.S.; Wu, K.Y.; Dong, H.J.; Mao, M.; Ovlyakulov, B.; Wu, H.M.; et al. Autoimmune Disease Associated CLEC16A Variants Convey Risk of Parkinson's Disease in Han Chinese. Front. Genet. 2022, 13, 856493. [CrossRef]
- Richard-Miceli, C.; Criswell, L.A. Emerging patterns of genetic overlap across autoimmune disorders. Genome Med. 2012, 4, 6. [CrossRef]
- Nitschke, Y.; Baujat, G.; Botschen, U.; Wittkampf, T.; du Moulin, M.; Stella, J.; Le Merrer, M.; Guest, G.; Lambot, K.; Tazarourte-Pinturier, M.F.; et al. Generalized arterial calcification of infancy and pseudoxanthoma elasticum can be caused by mutations in either ENPP1 or ABCC6. Am. J. Hum. Genet. 2012, 90, 25-39. [CrossRef]
- Farh, K.K.; Marson, A.; Zhu, J.; Kleinewietfeld, M.; Housley, W.J.; Beik, S.; Shoresh, N.; Whitton, H.; Ryan, R.J.; Shishkin, A.A.; et al. Genetic and epigenetic fine mapping of causal autoimmune disease variants. Nature 2015, 518, 337-343. [CrossRef]
- Caliskan, M.; Brown, C.D.; Maranville, J.C. A catalog of GWAS fine-mapping efforts in autoimmune disease. Am. J. Hum. Genet. 2021, 108, 549-563. [CrossRef]
- Belova, V.; Pavlova, A.; Afasizhev, R.; Moskalenko, V.; Korzhanova, M.; Krivoy, A.; Cheranev, V.; Nikashin, B.; Bulusheva, I.; Rebrikov, D.; et al. System Analysis of the Sequencing Quality of Human Whole Exome Samples on BGI NGS Platform. Sci. Rep. 2022, 12, 609. [CrossRef]
- Andrews, S. FastQC: A Quality Control Tool for High Throughput Sequence Data; Babraham Institute: Cambridge, UK, 2017.
- Bushnell, B. BBMap: A Fast, Accurate, Splice-Aware Aligner. 2014. Available online: https://github.com/BioInfoTools/BBMap (accessed on 1 November 2023).
- Li, H.; Durbin, H. Fast and accurate short read alignment with Burrows–Wheeler transform. Bioinformatics 2009, 25, 1754–176. [CrossRef]
- Li, H.; Handsaker, B.; Wysoker, A.; Fennell, T.; Ruan, J.; Homer, N.; Marth, G.; Abecasis, G.; Durbin, R.; 1000 Genome Project Data Processing Subgroup. The Sequence Alignment/Map Format and SAMtools. Bioinformatics 2009, 25, 2078–2079. [CrossRef]
- Broad Institute. Picard Toolkit. 2014. Available online: https://broadinstitute.github.io/picard/ (accessed on 1 November 2023).
- Li, H. A Statistical Framework for SNP Calling, Mutation Discovery, Association Mapping and Population Genetical Parameter Estimation from Sequencing Data. Bioinformatics 2011, 27, 2987–2993. [CrossRef]
- Tan, A.; Abecasis, G.R.; Kang, H.M. Unified Representation of Genetic Variants. Bioinformatics 2015, 31, 2202–2204. [CrossRef]
- Li, Q.; Wang, K. InterVar: Clinical Interpretation of Genetic Variants by the 2015 ACMG-AMP Guidelines. Am. J. Hum. Genet. 2017, 100, 267–280. [CrossRef]
- Buianova, A.A.; Proskura, M.V.; Cheranev, V.V.; Belova, V.A.; Shmitko, A.O.; Pavlova, A.S.; Vasiliadis, I.A.; Suchalko, O.N.; Rebrikov, D.V.; Petrosyan, E.K.; et al. Candidate Genes for IgA Nephropathy in Pediatric Patients: Exome-Wide Association Study. Int. J. Mol. Sci. 2023, 24, 15984. [CrossRef]
- Purcell, S.; Chang, C. PLINK v. 1.90b6.24. Available online https://github.com/chrchang/plink-ng/tree/master/1.9 (accessed on 20 June 2023).
- Zhou, Y.; Zhou, B.; Pache, L.; Chang, M.; Khodabakhshi, A.H.; Tanaseichuk, O.; Benner, C.; Chanda, S.K. Metascape Provides a Biologist-Oriented Resource for the Analysis of Systems-Level Datasets. Nat. Commun. 2019, 10, 1523. [CrossRef]
- Barker, D.J.; Maccari, G.; Georgiou, X.; Cooper, M.A.; Flicek, P.; Robinson, J.; Marsh, S.G.E. The IPD-IMGT/HLA Database. Nucleic Acids Res. 2023, 51, D1053-D1060. [CrossRef]
- Excoffier, L.; Lischer, H.E. Arlequin suite ver 3.5: a new series of programs to perform population genetics analyses under Linux and Windows. Mol Ecol Resour. 2010, 10, 564-567. [CrossRef]
- Okonechnikov, K.; Golosova, O.; Fursov, M.; UGENE team. Unipro UGENE: a unified bioinformatics toolkit. Bioinformatics 2012, 28, 1166-1167. [CrossRef]
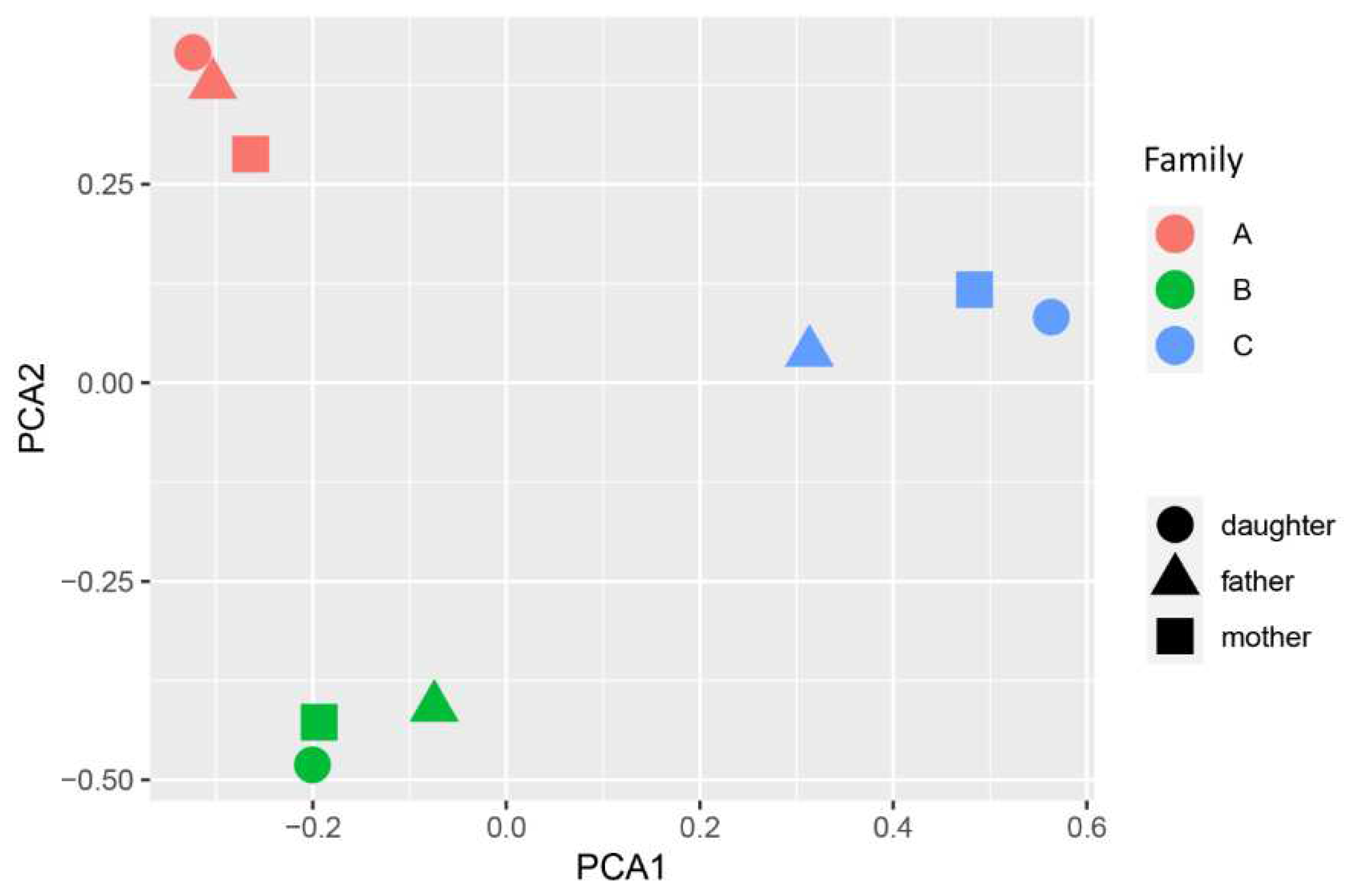
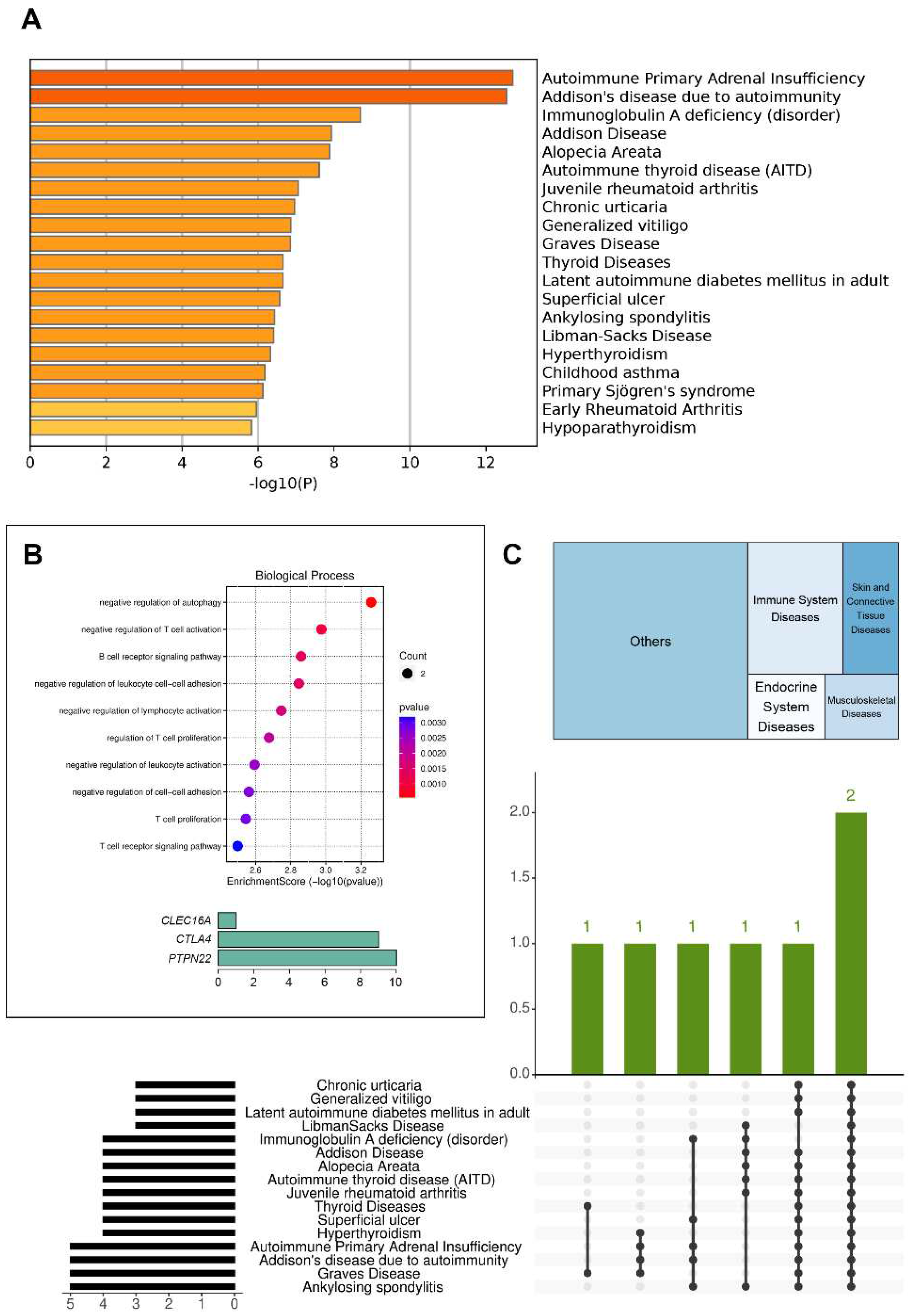
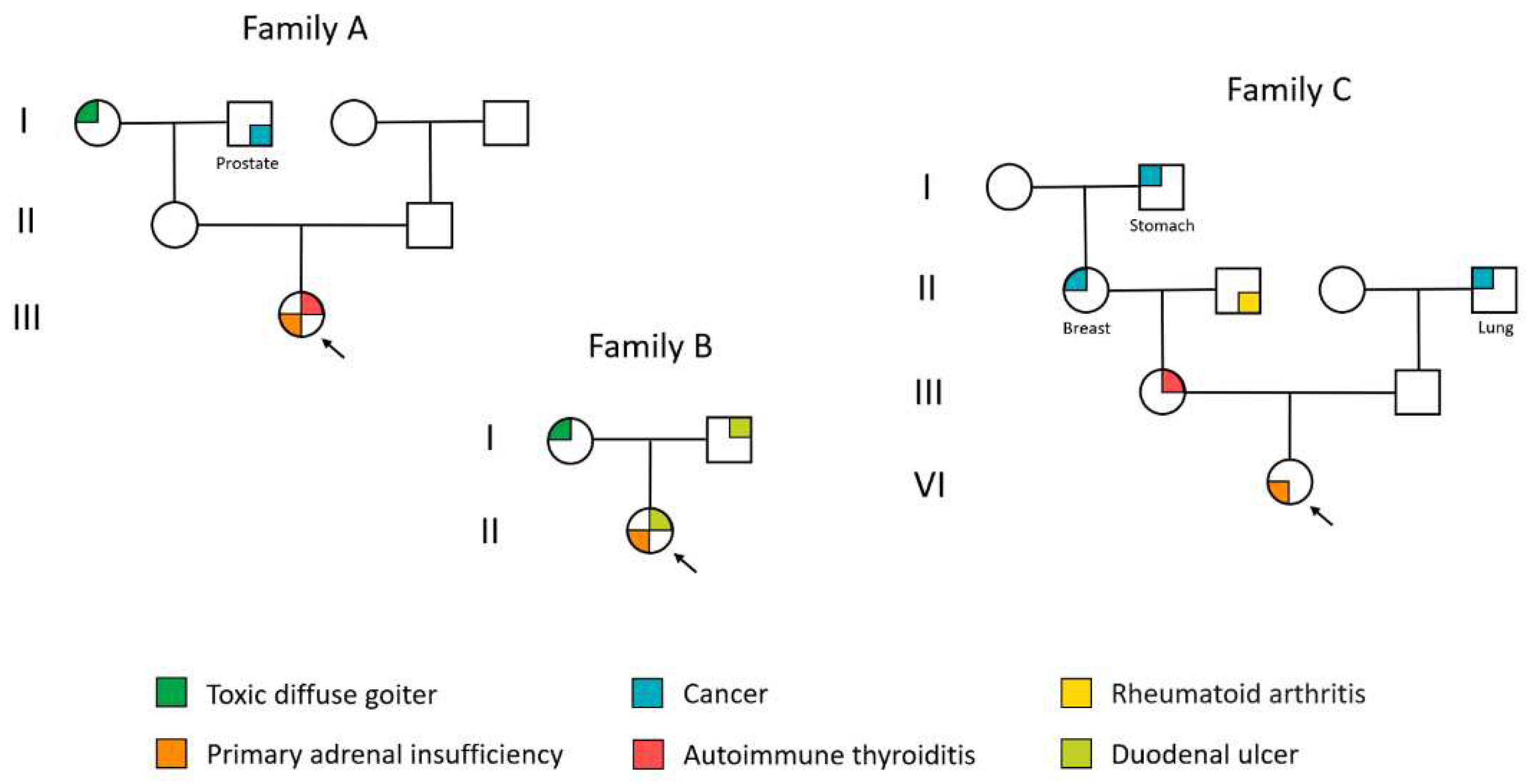
| Autoantibodies | Patient A | Patient B | Patient C |
| Anti-21OH ELISA/Microarray |
+/+ | +/+ | +/+ |
| Anti-TPO ELISA/Microarray |
+/+ | +/+ | -/- |
| Anti-Tg ELISA/Microarray |
-/- | -/- | -/- |
| Anti-IA2 ELISA/Microarray |
+/- | -/- | -/- |
| Anti-ICA ELISA/Microarray |
-/- | -/- | -/- |
| Anti-GAD ELISA/Microarray |
-/- | -/- | -/- |
| Anti-Zn8 ELISA |
- | - | - |
| Anti-IAA ELISA |
- | - | + |
| Anti-ATP4 ELISA |
- | - | - |
| Anti-GIF ELISA |
- | - | - |
| Anti-GLD ELISA |
not tested | not tested | - |
| Anti-TGM2 ELISA |
not tested | not tested | - |
| Anti-IFN-ω Microarray |
- | - | - |
| Anti-IFN-α Microarray |
- | - | - |
| Anti-IL-22 Microarray |
- | - | - |
| Metrics | Mean | Min | Max | |
|---|---|---|---|---|
| Single reads per sample | 102,044,084 | 83,932,144 | 154,800,134 | |
| Estimated library size | 162,054,959 | 106,209,935 | 226,432,803 | |
| Duplicates | 14.89 | 8.70 | 20.00 | |
| On-target bases | 88.1% | 87.2% | 88.7% | |
| Mean target coverage | 108.93 | 90.30 | 159.50 | |
| Median target coverage | 102.33 | 78.00 | 154.00 | |
| Width 10× | 95.80% | 93.10% | 97.00% | |
| Width 20× | 93.82% | 90.20% | 96.40% | |
| Width 30× | 91.14% | 87.80% | 95.70% |
| Variant | Patient A |
Patient B |
Patient C |
P-value for TDT |
|---|---|---|---|---|
|
CTLA4 (NM_005214.5):c.49A>G (p.Thr17Ala) rs231775 |
+/+ | +/- | +/- | 0.0455 |
|
PTPN22 (NM_015967.7):c.1858T>C (p.Trp620Arg) rs2476601 |
+/+ | +/+ | +/+ | NA* |
|
NFATC1 (NM_001278669.2):c.2251T>G (p.Cys751Gly) rs754093 |
+/- | +/- | -/- | 0.5637 |
|
GPR174 (NM_032553.3):c.484T>C (p.Ser162Pro) rs3827440 |
+/- | +/- | +/+ | 0.3173 |
|
VDR (NM_000376.3):c.1025-49G>T rs7975232 |
-/- | +/- | +/+ | 0.3173 |
|
VDR (NM_000376.3):c.1056T>C (p.Ile352=) rs731236 |
-/- | +/- | +/- | 1 |
| Family | Member | HLA-DRB1* | HLA-DQA1* | HLA-DQB1* | |||
|---|---|---|---|---|---|---|---|
| A | Mother | 04:04:01G | 12:01:01G | 03:01:01G | 05:01:01G | 03:01:01G | 03:02:01G |
| Father | 03:01:01G | 11:01:01G | 05:01:01G | 05:01:01G | 02:01:01G | 03:01:01G | |
| Daughter | 03:01:01G | 12:01:01G | 05:01:01G | 05:01:01G | 02:01:01G | 03:01:01G | |
| B | Mother | 04:03:01G | 11:01:01G | 03:01:01G | 05:01:01G | 03:01:01G | 03:02:01G |
| Father | 03:01:01G | 15:02:01G | 01:03:01G | 05:01:01G | 02:01:01G | 06:01:01G | |
| Daughter | 03:01:01G | 04:03:01G | 03:01:01G | 05:01:01G | 02:01:01G | 03:02:01G | |
| C | Mother | 03:01:01G | 13:01:01G | 01:03:01G | 05:01:01G | 02:01:01G | 06:03:01G |
| Father | 04:03:01G | 15:01:01G | 01:02:01G | 03:01:01G | 03:02:01G | 06:02:01G | |
| Daughter | 03:01:01G | 04:03:01G | 03:01:01G | 05:01:01G | 02:01:01G | 03:02:01G | |
| Analysis | Indicator, reference range | Patient A | Patient B | Patient C |
|---|---|---|---|---|
| Biochemical blood test | Ca total, 2.15-2.55 mmol/L; Ca ionized, 1.03-1.29 mmol/L |
Ca*, 2.29 mmol/L | Ca*, 2.4 mmol/L | Ca*, 2.51 mmol/L |
| R, 0.74-1.52 mmol/L | 1.46 mmol/L | 1.28 mmol/L | 1.36 mmol/L | |
| glucose, 3.1-6.1 mmol/L | 4.64 mmol/L | 4.63 mmol/L | 4.68 mmol/L | |
| ALT, 0-55.0 U/L | 10 U/L | 16 U/L | 10 U/L | |
| AST, 5.0-34.0 U/L | 15 U/L | 15 U/L | 14 U/L | |
| creatinine, 50-98 µmol/L | 61.6 μmol/L | 66.5 μmol/L | 84.3 μmol/L | |
| vitamin B12, 191-663 pg/mL | 187 pg/mL | 764 pg/mL | - | |
| Thyrotropine | 0.25-3.5 mIU/L | 1.755 mIU/L (while undergoing levothyroxine sodium replacement therapy) | 1.163 mIU/L | 1.264 mIU/L |
| LH, FSH, estradiol | LH, 2.6-12.1 U/L | 7.71 U/L | - | 7.43 U/L |
| FSH, 1.9-11.7 U/L | 3.79 U/L | - | 3.84 U/L | |
| estradiol, 97-592 pmol/L | 150.43 pmol/L | - | 166.09 pmol/L | |
| Aldosterone, renin | aldosterone, 69.8-1085.8 pmol/L | - | 78.3 pmol/L | - |
| renin, 2.8-39.9 mU/L | 33.21 mU/L | > 500 mU/L | 24 mU/L | |
| ACTH, cortisol |
ACTH, 7.2-63.3 pg/mL | - | >2000 pg/mL | - |
| cortisol during insulin hypoglycemia test, more than 500 nmol/L | - | peak cortisol level during insulin hypoglycemia test 126.7 nmol/L | - | |
| Glycated hemoglobin | up to 6% | 5.6% | 5% | 5.3% |
| Insulin, C-peptide | 2.6-24.9 µU/mL | 12.74 µU/mL | 11.75 µU/mL | - |
| 1.1-4.4 ng/mL | 2.98 ng/mL | 2.11 ng/mL | 2.6 ng/mL |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




