Submitted:
08 November 2023
Posted:
09 November 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Standards and reagents
2.2. Synthesis and characterization of NADES
2.3. Synthesis of lactulose
2.4. Solubility of LMWC in NADES
2.5. Solubility of binary mixtures of LMWC in NADES
2.6. NADES treatment of the lactose isomerization mixture
2.7. Evaluation of carbohydrate degradation
2.8. HILIC-MS analysis
2.9. Statistical analysis
3. Results and Discussion
3.1. Stability of carbohydrates in NADES
3.2. Solubility of carbohydrates at 25°C
3.3. Solubility of carbohydrates at 45°C in NADES2
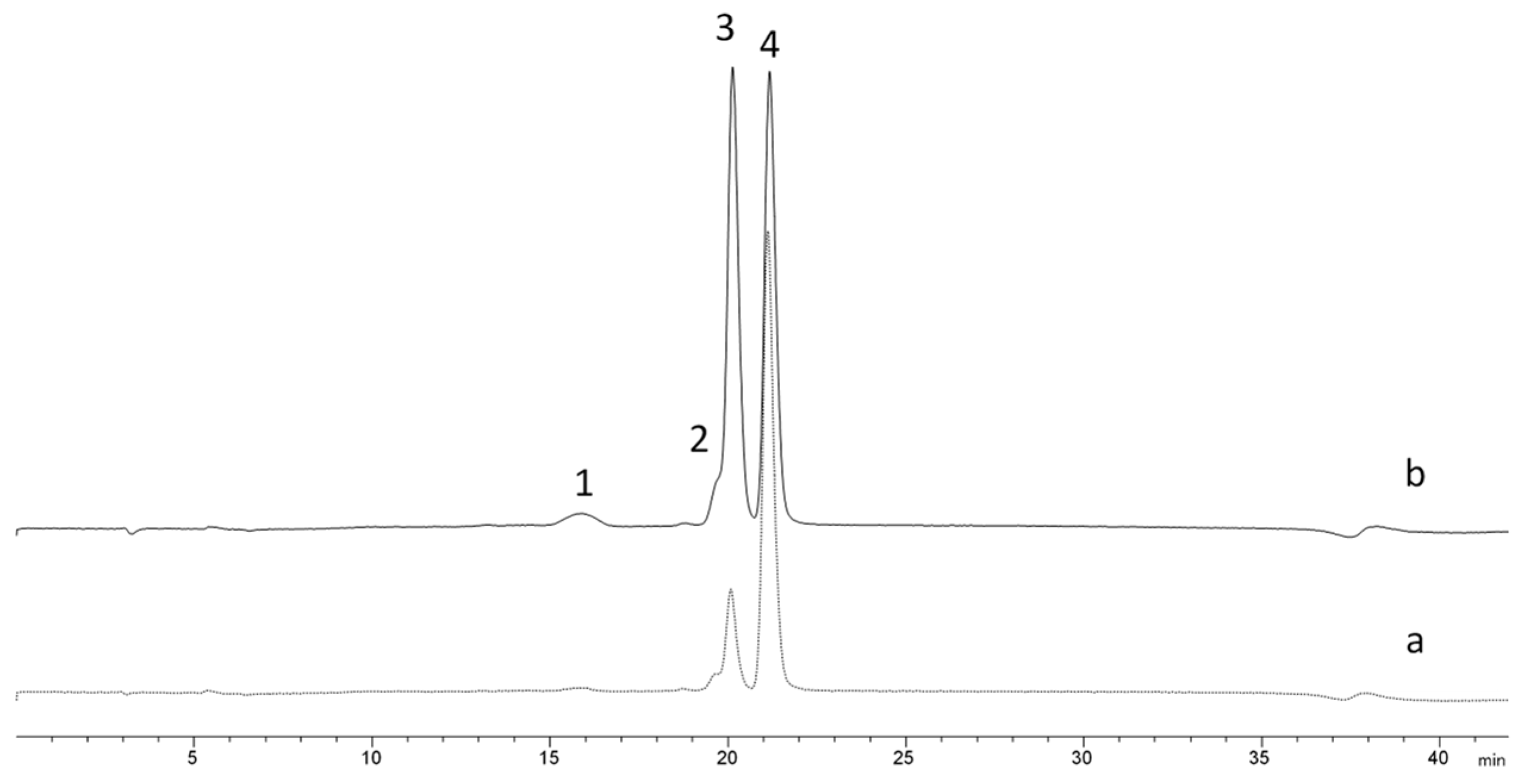
3.4. Fractionation of bioactive ketoses from aldoses in equimolar binary mixtures
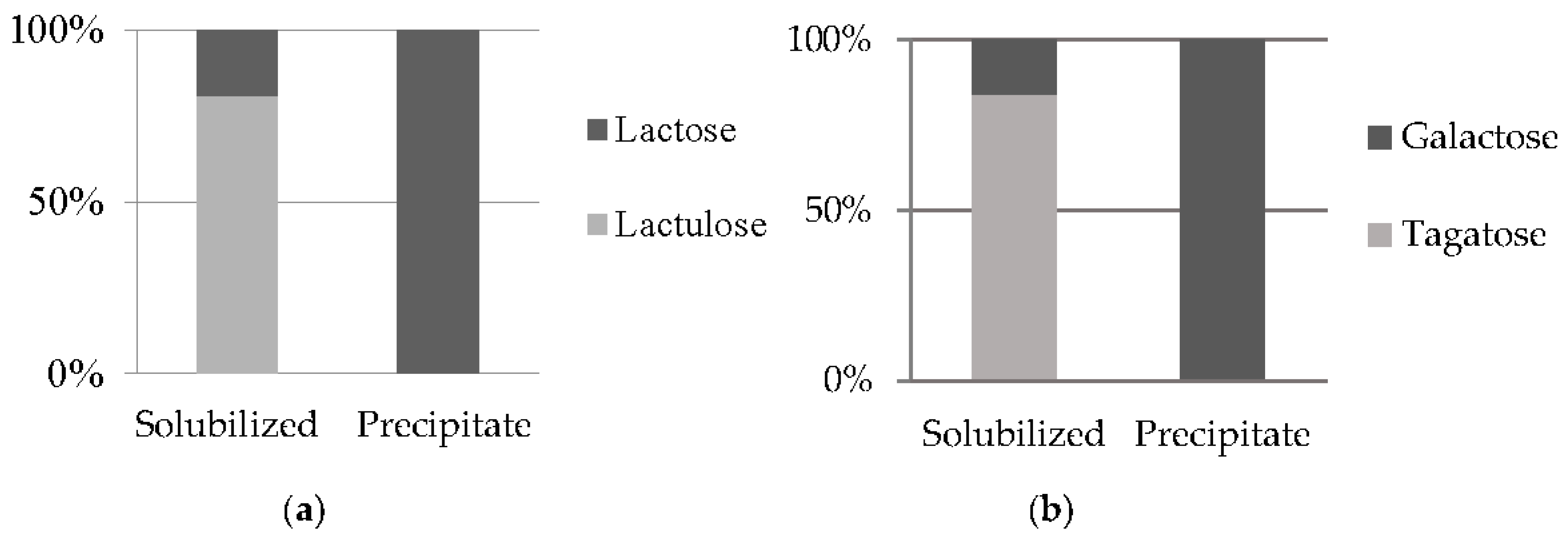
3.5. Fractionation of lactulose from the isomerization reaction mixture
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ruiz-Aceituno, L.; Carrero-Carralero, C.; Ramos, L.; Sanz, M.L. Selective Fractionation of Sugar Alcohols Using Ionic Liquids. Sep. Purif. Technol. 2019, 209, 800–805. [CrossRef]
- Moreno, F.J.; Carrero-Carralero, C.; Hernández-Hernández, O.; Sanz, M.L. Fractionation of Food Bioactive Oligosaccharides. In Food Oligosaccharides; John Wiley & Sons, Ltd: Chichester, UK, 2014; Vol. 21, pp. 255–283 ISBN 9789896540821.
- Hernández, O.; Ruiz-Matute, A.I.; Olano, A.; Moreno, F.J.; Sanz, M.L. Comparison of Fractionation Techniques to Obtain Prebiotic Galactooligosaccharides. Int. Dairy J. 2009, 19, 531–536. [CrossRef]
- Montañés, F.; Olano, A.; Ibáñez, E.; Fornari, T. Modeling Solubilities of Sugars in Alcohols Based on Original Experimental Data. AIChE J. 2007, 53, 2411–2418. [CrossRef]
- Mena-García, A.; Ruiz-Matute, A.I.; Soria, A.C.; Sanz, M.L. Green Techniques for Extraction of Bioactive Carbohydrates. TrAC Trends Anal. Chem. 2019, 119, 115612. [CrossRef]
- Zakrzewska, M.E.; Bogel-Łukasik, E.; Bogel-Łukasik, R. Solubility of Carbohydrates in Ionic Liquids. Energy & Fuels 2010, 24, 737–745. [CrossRef]
- Carneiro, A.P.; Held, C.; Rodríguez, O.; Sadowski, G.; Macedo, E.A. Solubility of Sugars and Sugar Alcohols in Ionic Liquids: Measurement and PC-SAFT Modeling. J. Phys. Chem. B 2013, 117, 9980–9995. [CrossRef]
- Carrero-Carralero, C.; Ruiz-Aceituno, L.; Ramos, L.; Moreno, F.J.; Sanz, M.L. Influence of Chemical Structure on the Solubility of Low Molecular Weight Carbohydrates in Room Temperature Ionic Liquids. Ind. Eng. Chem. Res. 2014, 53, 13843–13850. [CrossRef]
- Carrero-Carralero, C.; Ruiz-Aceituno, L.; Ramos, L.; Sanz, M.L.; Moreno, F.J. Use of Room Temperature Ionic Liquids for the Selective Fractionation of Bioactive Ketoses from Aldoses. Sep. Purif. Technol. 2015, 149, 140–145. [CrossRef]
- Bubalo, M.C.; Radošević, K.; Redovniković, I.R.; Slivac, I.; Srček, V.G. Toxicity Mechanisms of Ionic Liquids. Arch. Ind. Hyg. Toxicol. 2017, 68, 171–179. [CrossRef]
- Benvenutti, L.; Zielinski, A.A.F.; Ferreira, S.R.S. Which Is the Best Food Emerging Solvent: IL, DES or NADES? Trends Food Sci. Technol. 2019, 90, 133–146. [CrossRef]
- Paiva, A.; Craveiro, R.; Aroso, I.; Martins, M.; Reis, R.L.; Duarte, A.R.C. Natural Deep Eutectic Solvents – Solvents for the 21st Century. ACS Sustain. Chem. Eng. 2014, 2, 1063–1071. [CrossRef]
- Liu, Y.; Friesen, J.B.; McAlpine, J.B.; Lankin, D.C.; Chen, S.; Pauli, G.F. Natural Deep Eutectic Solvents: Properties, Applications, and Perspectives. J. Nat. Prod. 2018, 81, 679–690. [CrossRef]
- Paradiso, V.M.; Clemente, A.; Summo, C.; Pasqualone, A.; Caponio, F. Towards Green Analysis of Virgin Olive Oil Phenolic Compounds : Extraction by a Natural Deep Eutectic Solvent and Direct Spectrophotometric Detection. Food Chem. 2016, 212, 43–47. [CrossRef]
- Manuela, P.; Drakula, S.; Cravotto, G.; Verpoorte, R.; Hruškar, M. Biological Activity and Sensory Evaluation of Cocoa By-Products NADES Extracts Used in Food Fortification. Innov. Food Sci. Emerg. Technol. 2020, 66, 102514. [CrossRef]
- Popovic, B.M.; Micic, N.; Potkonjak, A.; Blagojevic, B.; Pavlovic, K.; Milanov, D.; Juric, T. Novel Extraction of Polyphenols from Sour Cherry Pomace Using Natural Deep Eutectic Solvents – Ultrafast Microwave-Assisted NADES Preparation and Extraction. Food Chem. 2022, 366. [CrossRef]
- Procentese, A.; Raganati, F.; Olivieri, G.; Russo, M.E.; Rehmann, L.; Marzocchella, A. Deep Eutectic Solvents Pretreatment of Agro-Industrial Food Waste. Biotechnol. Biofuels 2018, 11, 1–12. [CrossRef]
- Hong, S.; Shen, X.J.; Xue, Z.; Sun, Z.; Yuan, T.Q. Structure-Function Relationships of Deep Eutectic Solvents for Lignin Extraction and Chemical Transformation. Green Chem. 2020, 22, 7219–7232.
- Häkkinen, R.; Abbott, A. Solvation of Carbohydrates in Five Choline Chloride-Based Deep Eutectic Solvents and the Implication for Cellulose Solubility. Green Chem. 2019, 21, 4673–4682. [CrossRef]
- Gómez, A.V.; Tadini, C.C.; Biswas, A.; Buttrum, M.; Kim, S.; Boddu, V.M.; Cheng, H.N. Microwave-Assisted Extraction of Soluble Sugars from Banana Puree with Natural Deep Eutectic Solvents (NADES). LWT 2019, 107, 79–88. [CrossRef]
- Geurink, L.; Tricht, E.; Dudink, J.; Pajic, B.; Sänger – van de Griend, C.E. Four-step Approach to Efficiently Develop Capillary Gel Electrophoresis Methods for Viral Vaccine Protein Analysis. Electrophoresis 2021, 42, 10–18. [CrossRef]
- Montilla, A.; Del Castillo, M.D.; Sanz, M.L.; Olano, A. Egg Shell as Catalyst of Lactose Isomerisation to Lactulose. Food Chem. 2005, 90, 883–890. [CrossRef]
- Meydav, S.; Saguy, I.; Kopelman, I.J. Browning Determination in Citrus Products. J. Agric. Food Chem. 1977, 25, 602–604. [CrossRef]
- Viñas, P.; Campillo, N.; Córdoba, M.H.; Candela, M.E. Simultaneous Liquid Chromatographic Analysis of 5-(Hydroxymethyl)-2-Furaldehyde and Methyl Anthranilate in Honey. Food Chem. 1992, 44, 67–72. [CrossRef]
- Hernández-Hernández, O.; Calvillo, I.; Lebrón-Aguilar, R.; Moreno, F.J.; Sanz, M.L. Hydrophilic Interaction Liquid Chromatography Coupled to Mass Spectrometry for the Characterization of Prebiotic Galactooligosaccharides. J. Chromatogr. A 2012, 1220, 57–67. [CrossRef]
- Panić, M.; Stojković, M.R.; Kraljić, K.; Škevin, D. Ready-to-Use Green Polyphenolic Extracts from Food by-Products. Food Chem. 2019, 283, 628–636. [CrossRef]
- Moreau, C.; Finiels, A.; Vanoye, L. Dehydration of Fructose and Sucrose into 5-Hydroxymethylfurfural in the Presence of 1-H-3-Methyl Imidazolium Chloride Acting Both as Solvent and Catalyst. J. Mol. Catal. A Chem. 2006, 253, 165–169. [CrossRef]
- Wu, M.; Zhou, W.; Pedersen, C.M.; Ma, H.; Qiao, Y.; Guo, X.; Hou, X.; Wang, Y. Isomeric Distribution of Monosaccharides in Deep Eutectic Solvents: NMR Study. J. Mol. Liq. 2018, 255, 244–249. [CrossRef]
- Teong, S.P.; Yi, G.; Zhang, Y. Hydroxymethylfurfural Production from Bioresources: Past, Present and Future. Green Chem. 2014, 16, 2015–2026.
- Rosatella, A.A.; Branco, L.C.; Afonso, C.A.M. Studies on Dissolution of Carbohydrates in Ionic Liquids and Extraction from Aqueous Phase. Green Chem. 2009, 11, 1406–1413. [CrossRef]
- Olsson, C.; Swenson, J. Structural Comparison between Sucrose and Trehalose in Aqueous Solution. J. Phys. Chem. B 2020, 124, 3074–3082. [CrossRef]
- Lu, Y.; Levin, G. V; Donner, T.W. Tagatose, a New Antidiabetic and Obesity Control Drug. Diabetes, Obes. Metab. 2008, 10, 109–134. [CrossRef]
- Nooshkam, M.; Babazadeh, A.; Jooyandeh, H. Lactulose: Properties, Techno-Functional Food Applications, and Food Grade Delivery System. Trends Food Sci. Technol. 2018, 80, 23–34.
- Oh, D.K. Tagatose: Properties, Applications, and Biotechnological Processes. Appl. Microbiol. Biotechnol. 2007, 76, 1–8.
| Hydrogen Bond Acceptor (HBA) | Hydrogen Bond Donnor (HBD) | Molar ratio HBA:HBD | Viscosity (cP) | Water content (%) | |
|---|---|---|---|---|---|
| NADES1 | Choline chloride (ChCl) | 2-Ethylene glycol (EtG) |
1:2 | 35.6 (0.9)* | 0.29 |
| NADES2 | Glycerol (Gly) |
1:2 | 309.2 (0.9) | 0.23 | |
| NADES3 | Ethanedioic acid dihydrate (Eth) |
1:1 | 143.8 (0.7) | 9.96 |
| LMWC | Absorbance at 420 nm (uA) | HMF (% w/w) | ||
|---|---|---|---|---|
| Dilution | PBS | NADES3 | ||
| Tagatose | 1/6 | 0.0467 (0.0003)* | 0.5837 (0.0065) | 6.23 (0.68) |
| Fructose | 1/2 | 0.0491 (0.00017) | 0.4631 (0.0018) | 6.32 (0.16) |
| Glucose | 1/1 | 0.0458 (0.0002) | 0.1226 (0.0003) | 0.02 (0.01) |
| Sucrose | 1/1 | 0.0477 (0.0002) | 0.4752 (0.0002) | 4.00 (0.13) |
| Lactose | 1/1 | 0.0584 (0.0002) | 0.0970 (0.0005) | 0.040 (0.001) |
| Lactulose | 1/1 | 0.0587 (0.0001) | 0.3595 (0.0004) | 2.63 (0.08) |
| LMWCs | NADES1 | NADES2 | ||
|---|---|---|---|---|
| 25°C | 25°C | 45°C | ||
| Monosaccharides | Glucose | 242 (16)*,f | 135.7 (0.2)c | 364 (1)c |
| Galactose | 31.4 (0.4)a | 43 (4)a | 71 (6)b | |
| Fructose | 191 (1)d | 245 (5)d | 806 (16)f | |
| Tagatose | 72 (2)b | 150.0 (0.4)c | 214 (7)b | |
| Disaccharides | Lactose | 147 (5)c | 26.3 (2.9)a | 92 (29)a,b |
| Maltose | 323 (10)g | 280 (14)e | 630 (90)e | |
| Trehalose | 183 (3)d,e | 252 (17)d | 503 (13)d | |
| Sucrose | 152 (3)c,e | 82 (11)b | 368 (15)c | |
| Lactulose | 666 (25)h | 135.1 (0.3)c | 250 (11)b | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




