Submitted:
24 October 2023
Posted:
25 October 2023
You are already at the latest version
Abstract
Keywords:
1. The Significance of Bacteriophage Φ6
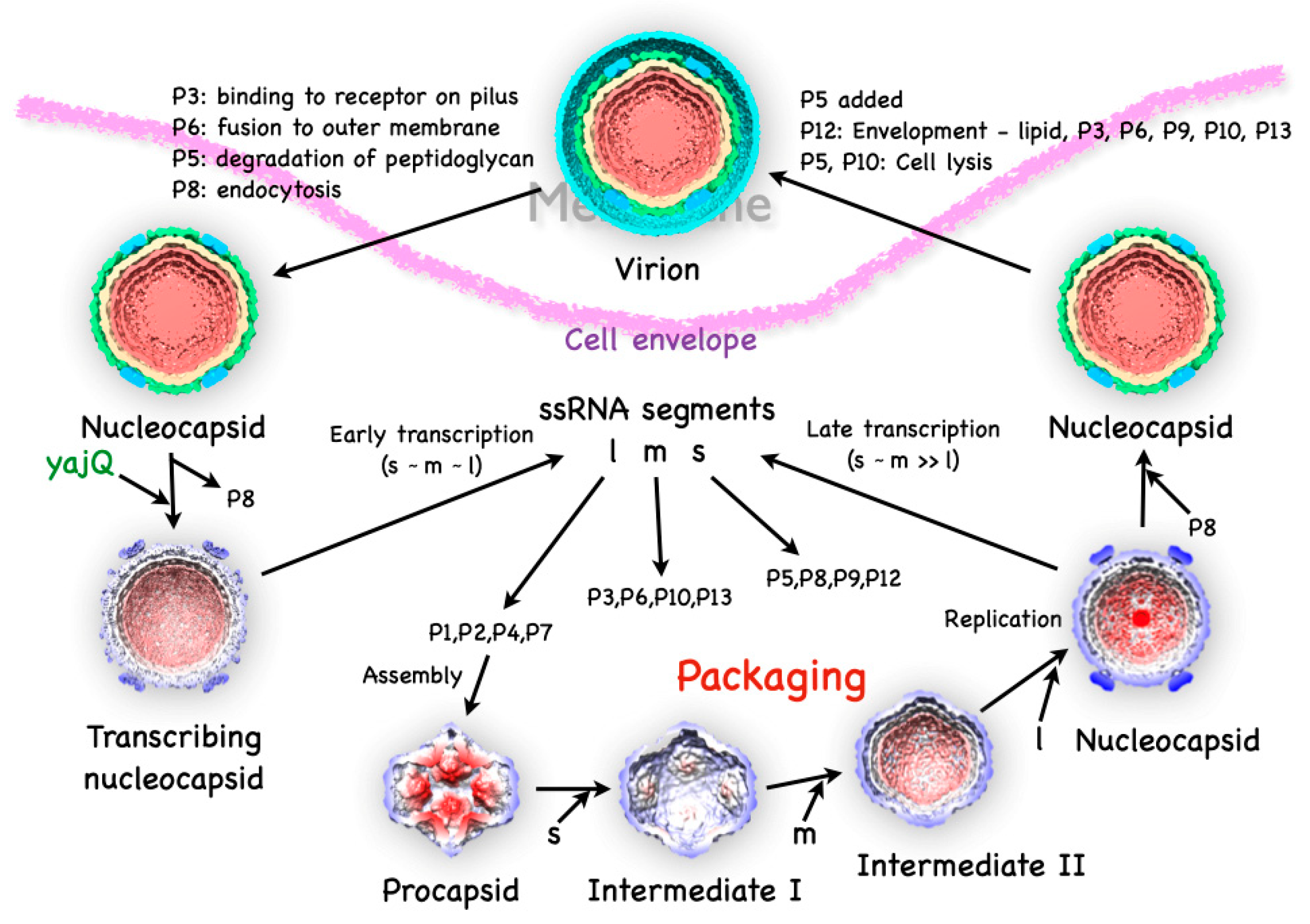
2. The Life Cycle of Bacteriophage Φ6
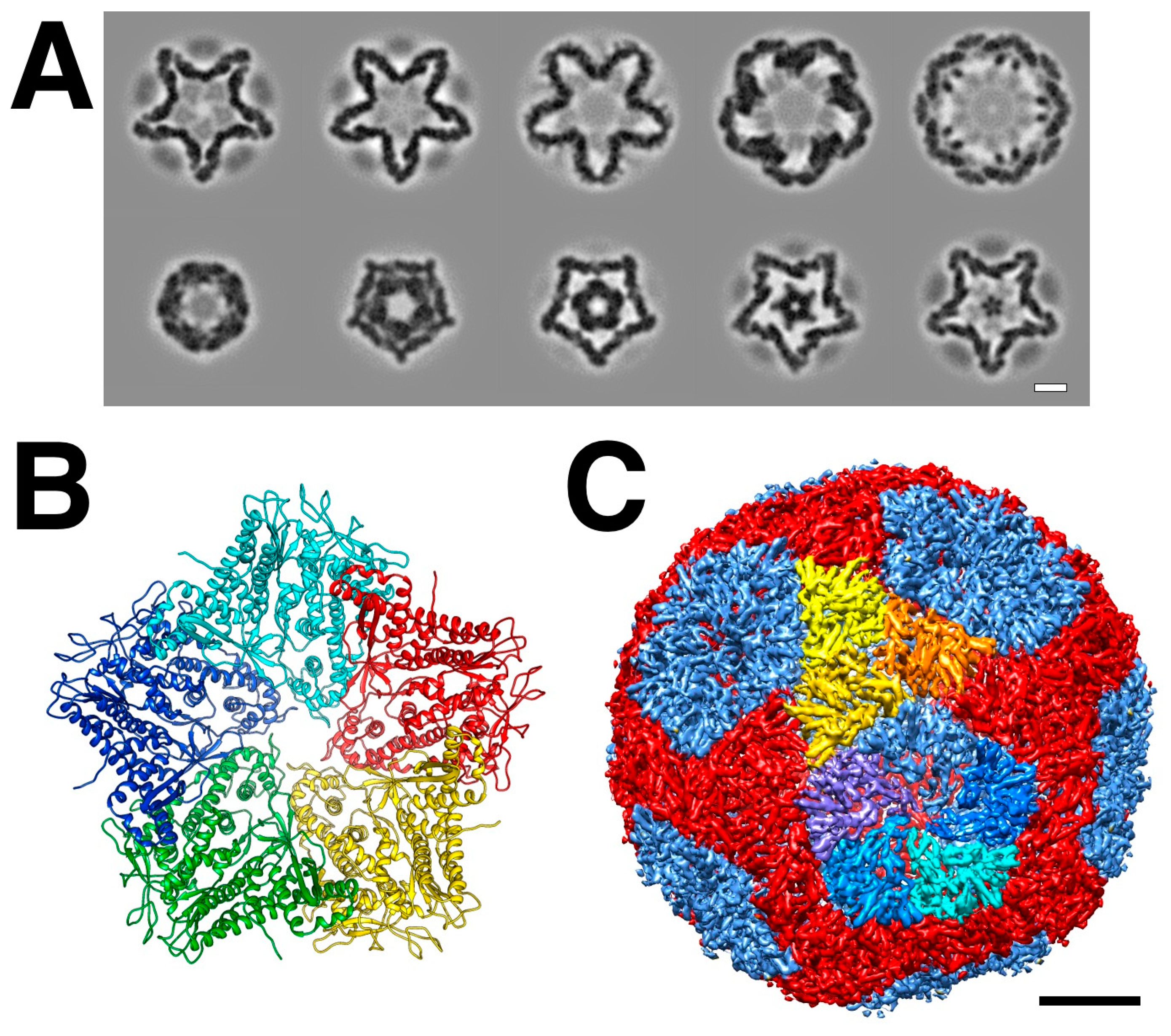
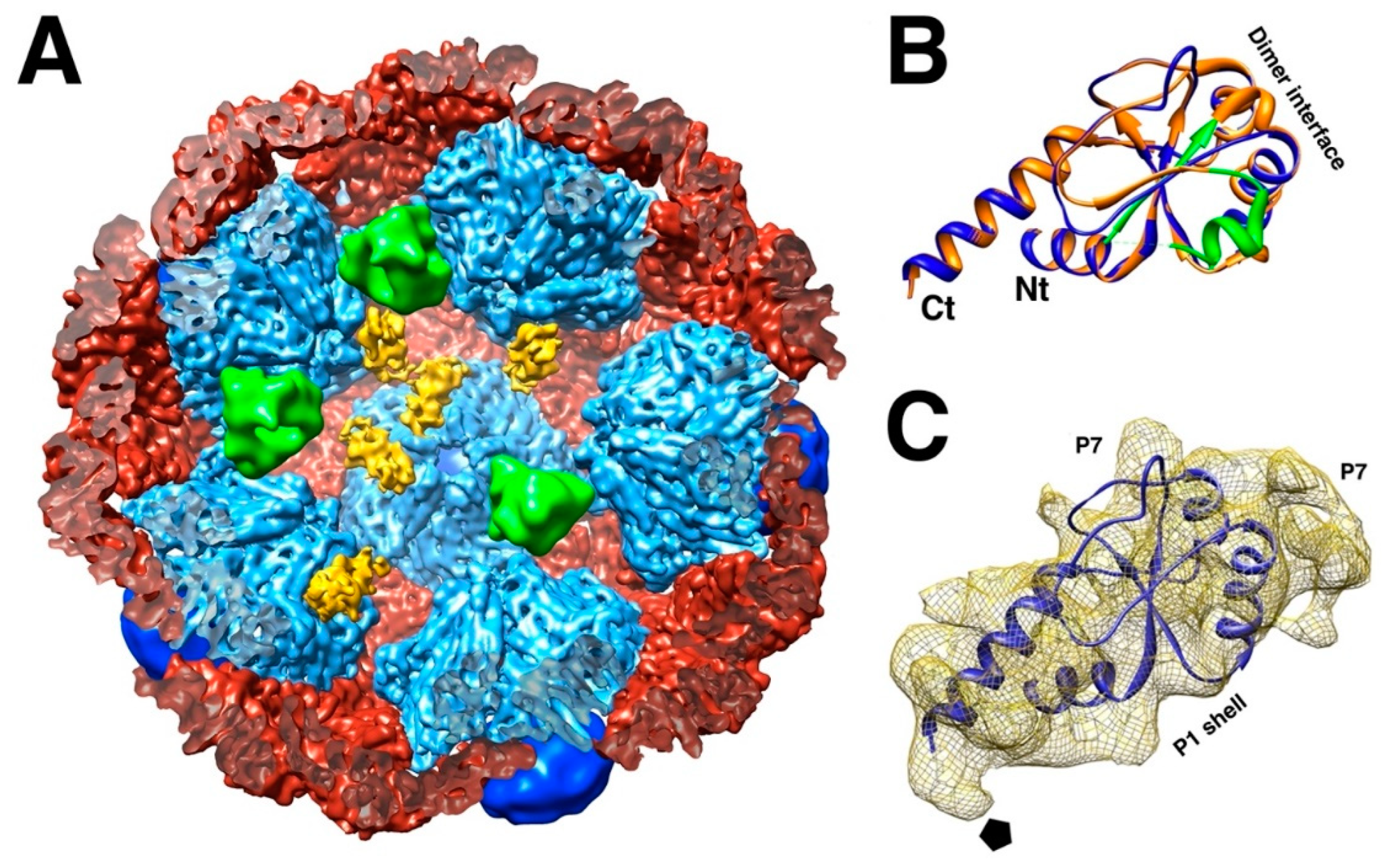
3. The Initial Assembly Product, the Procapsid
3.1. The P1 Shell
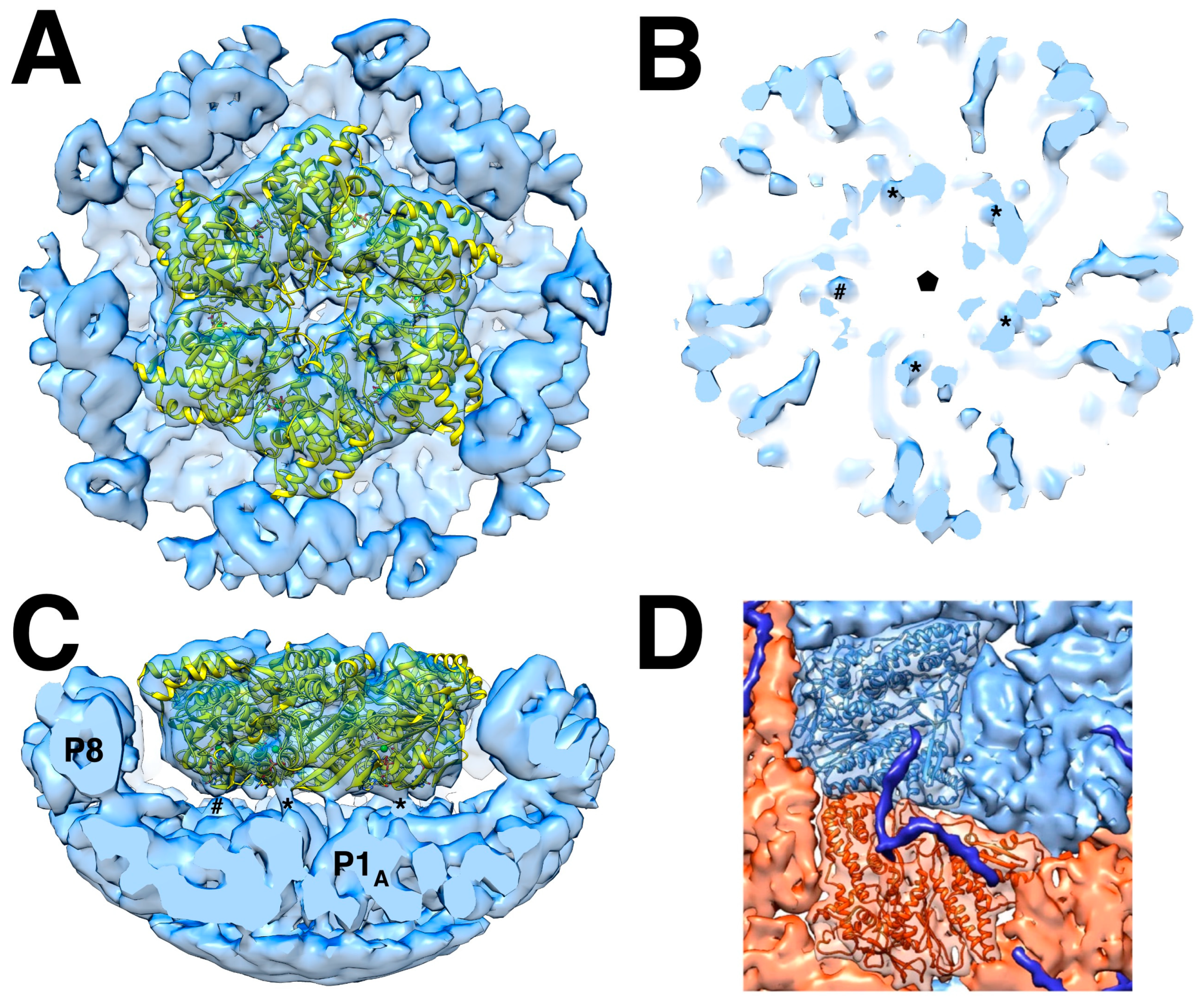
3.2. The Packaging Motor, Protein P4
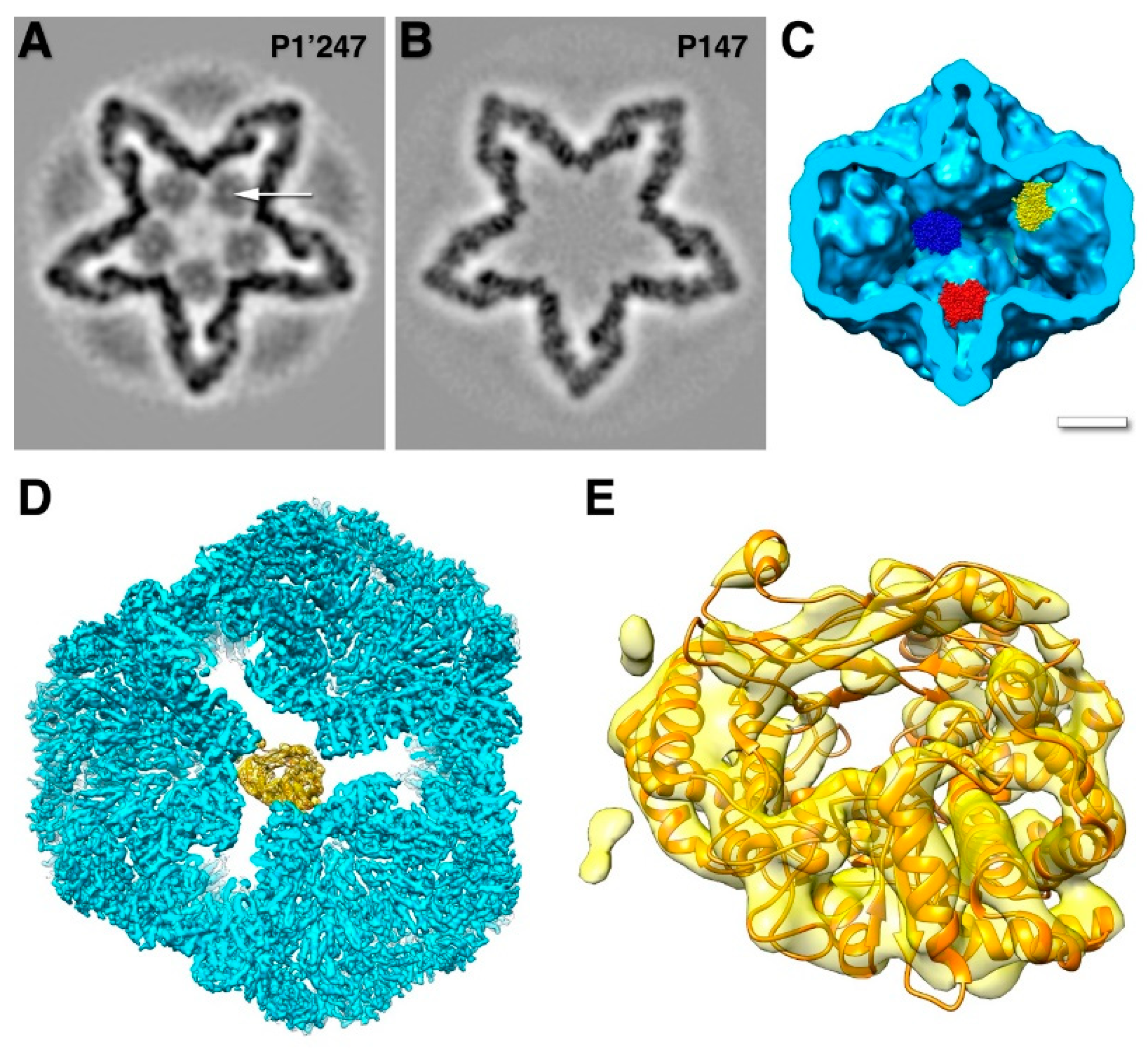
3.3. The RNA-Dependent RNA Polymerase, Protein P2
3.4. The Packaging Enhancer, Protein P7
3.5. Procapsid Assembly
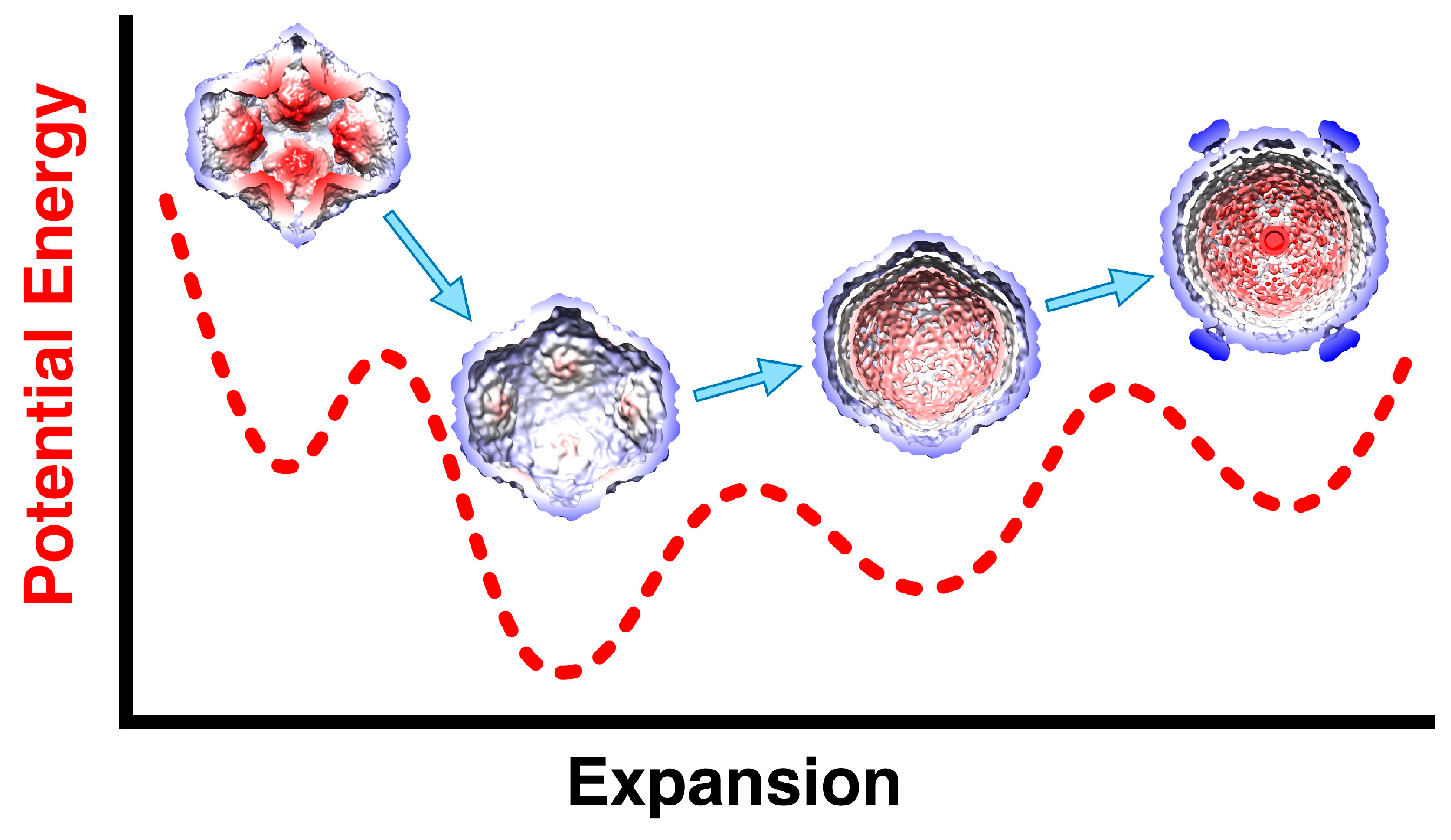
4. Packaging of the ssRNA Segments and Capsid Expansion
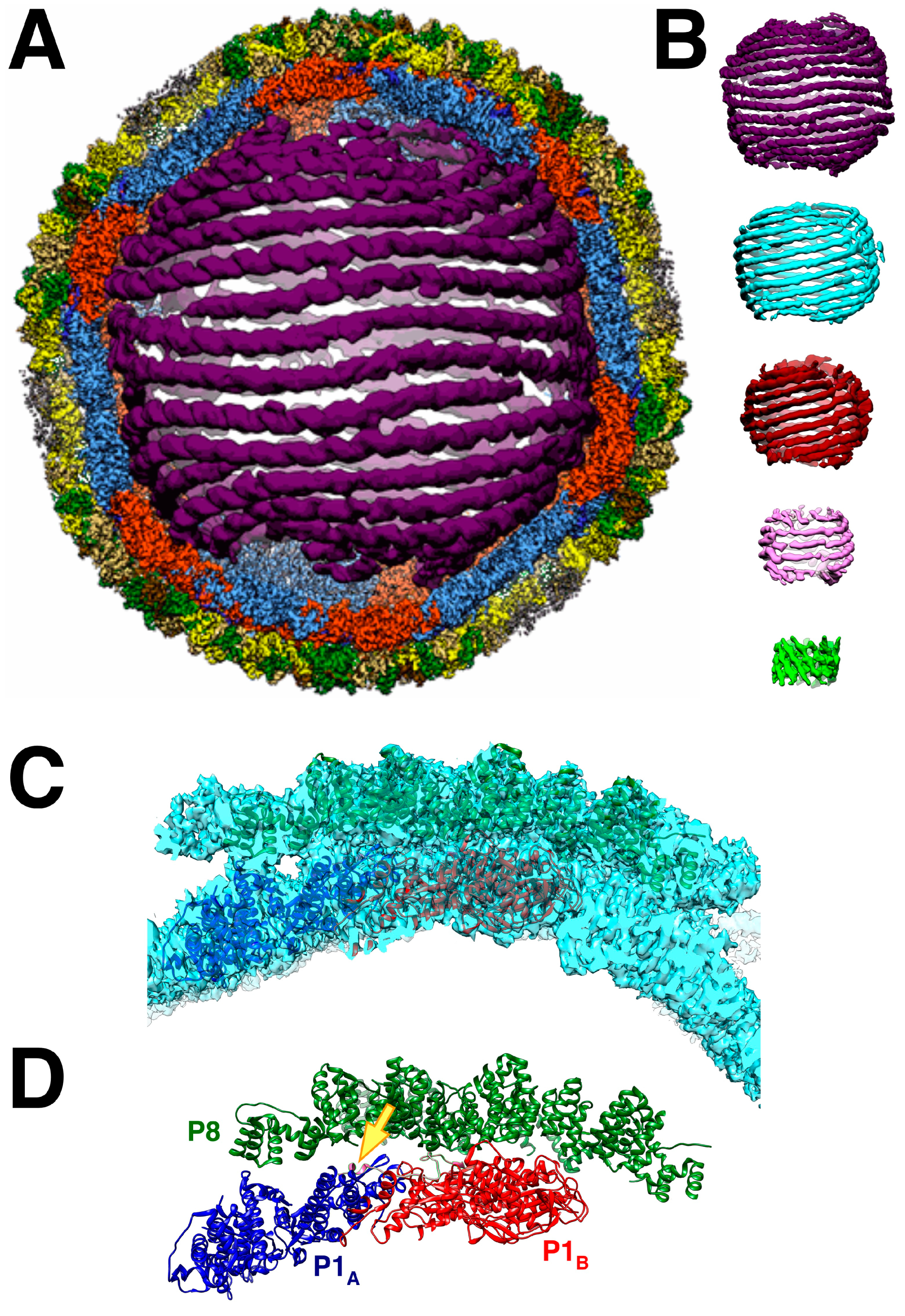
5. Completing the Virion
5.1. The dsRNA Packaging of the Nucleocapsid
5.2. The Intermediate Layer Composed of P4 and P8
5.3. The Envelope
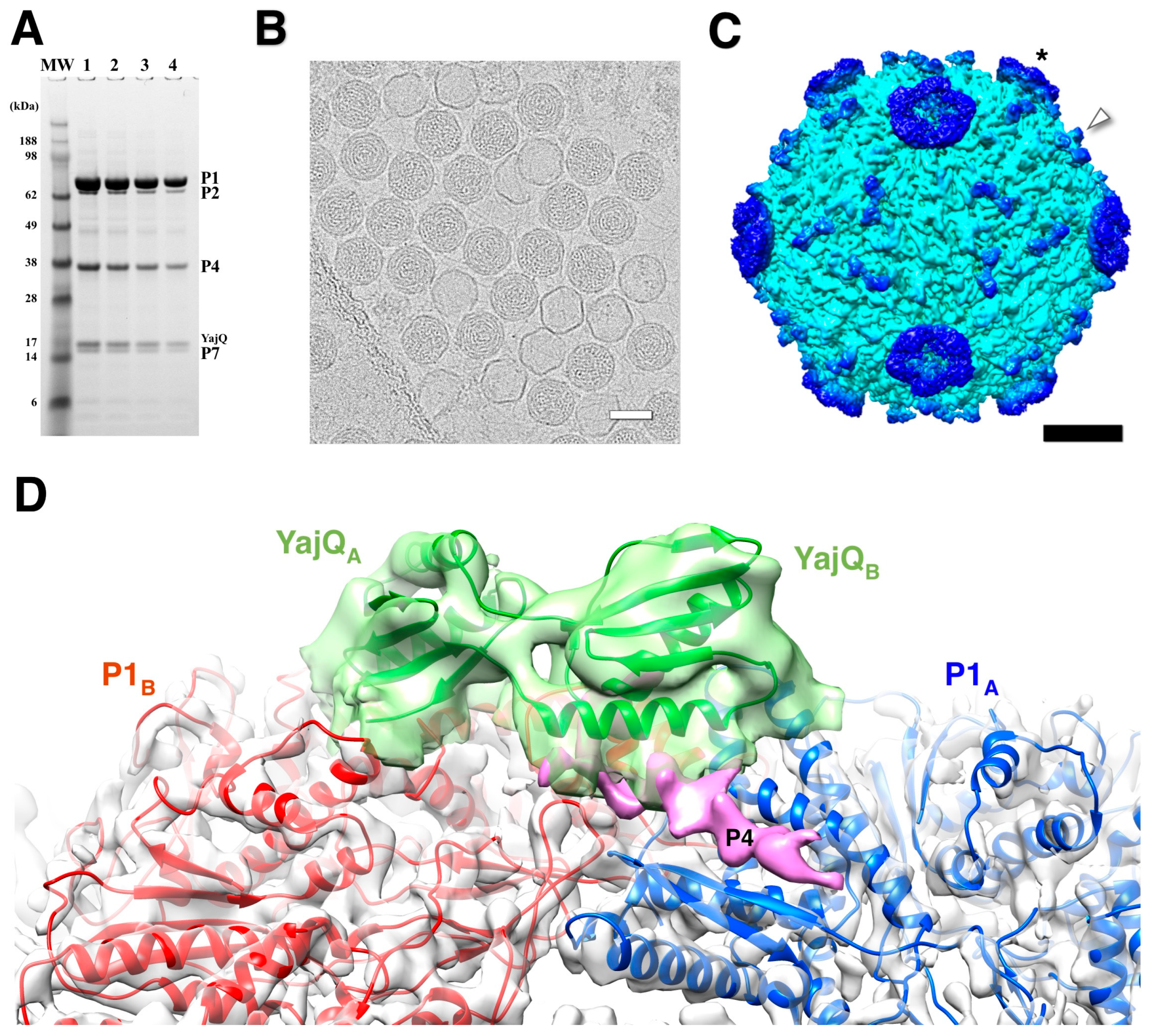
6. Coopting the Host Factor YajQ for Transcription Regulation
7. The Broader Impact of the Studies on the Structure and Transformations of Bacteriophage Φ6
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Vidaver, A.K.; Koski, R.K.; Van Etten, J.L. Bacteriophage phi6: a Lipid-Containing Virus of Pseudomonas phaseolicola. J Virol 1973, 11, 799-805. [CrossRef]
- Borodavka, A.; Desselberger, U.; Patton, J.T. Genome packaging in multi-segmented dsRNA viruses: distinct mechanisms with similar outcomes. Curr Opin Virol 2018, 33, 106-112. [CrossRef]
- Serrano-Aroca, A. Antiviral Characterization of Advanced Materials: Use of Bacteriophage Phi 6 as Surrogate of Enveloped Viruses Such as SARS-CoV-2. Int J Mol Sci 2022, 23. [CrossRef]
- Chen, Y.G.; Hur, S. Cellular origins of dsRNA, their recognition and consequences. Nat Rev Mol Cell Biol 2022, 23, 286-301. [CrossRef]
- Pinheiro, L.A.M.; Pereira, C.; Barreal, M.E.; Gallego, P.P.; Balcao, V.M.; Almeida, A. Use of phage varphi6 to inactivate Pseudomonas syringae pv. actinidiae in kiwifruit plants: in vitro and ex vivo experiments. Appl Microbiol Biotechnol 2020, 104, 1319-1330. [CrossRef]
- Pinheiro, L.A.M.; Pereira, C.; Frazao, C.; Balcao, V.M.; Almeida, A. Efficiency of Phage phi6 for Biocontrol of Pseudomonas syringae pv. syringae: An in Vitro Preliminary Study. Microorganisms 2019, 7. [CrossRef]
- de Carvalho, N.A.; Stachler, E.N.; Cimabue, N.; Bibby, K. Evaluation of Phi6 Persistence and Suitability as an Enveloped Virus Surrogate. Environmental Science & Technology 2017, 51, 8692-8700. [CrossRef]
- Frilander, M.; Poranen, M.; Bamford, D.H. The large genome segment of dsRNA bacteriophage phi6 is the key regulator in the in vitro minus and plus strand synthesis. RNA 1995, 1, 510-518.
- Cuppels, D.A.; Van Etten, J.L.; Burbank, D.E.; Lane, L.C.; Vidaver, A.K. In vitro translation of the three bacteriophage phi 6 RNAs. J. Virol. 1980, 35, 249-251. [CrossRef]
- Bamford, D.H.; Romantschuk, M.; Somerharju, P.J. Membrane fusion in prokaryotes: bacteriophage phi 6 membrane fuses with the Pseudomonas syringae outer membrane. EMBO J 1987, 6, 1467-1473. [CrossRef]
- Mindich, L.; Lehman, J. Cell wall lysin as a component of the bacteriophage phi 6 virion. J Virol 1979, 30, 489-496. [CrossRef]
- Caldentey, J.; Bamford, D.H. The lytic enzyme of the Pseudomonas phage phi 6. Purification and biochemical characterization. Biochim Biophys Acta 1992, 1159, 44-50. [CrossRef]
- Olkkonen, V.M.; Ojala, P.M.; Bamford, D.H. Generation of infectious nucleocapsids by in vitro assembly of the shell protein on to the polymerase complex of the dsRNA bacteriophage phi 6. J Mol Biol 1991, 218, 569-581. [CrossRef]
- Qiao, X.; Sun, Y.; Qiao, J.; Mindich, L. The role of host protein YajQ in the temporal control of transcription in bacteriophage Phi6. Proc Natl Acad Sci U S A 2008, 105, 15956-15960. [CrossRef]
- Mindich, L.; Davidoff-Abelson, R. The characterization of a 120 S particle formed during phi 6 infection. Virology 1980, 103, 386-391. [CrossRef]
- Gottlieb, P.; Strassman, J.; Bamford, D.H.; Mindich, L. Production of a polyhedral particle in Escherichia coli from a cDNA copy of the large genomic segment of bacteriophage phi 6. J Virol 1988, 62, 181-187. [CrossRef]
- Qiao, X.; Casini, G.; Qiao, J.; Mindich, L. In vitro packaging of individual genomic segments of bacteriophage phi 6 RNA: serial dependence relationships. J Virol 1995, 69, 2926-2931. [CrossRef]
- Johnson, M.D., 3rd; Mindich, L. Plasmid-directed assembly of the lipid-containing membrane of bacteriophage phi 6. J Bacteriol 1994, 176, 4124-4132. [CrossRef]
- Johnson, M.D., 3rd; Mindich, L. Isolation and characterization of nonsense mutations in gene 10 of bacteriophage phi 6. J Virol 1994, 68, 2331-2338. [CrossRef]
- Day, L.A.; Mindich, L. The molecular weight of bacteriophage phi 6 and its nucleocapsid. Virology 1980, 103, 376-385. [CrossRef]
- Mantynen, S.; Sundberg, L.R.; Poranen, M.M. Recognition of six additional cystoviruses: Pseudomonas virus phi6 is no longer the sole species of the family Cystoviridae. Arch Virol 2018, 163, 1117-1124. [CrossRef]
- Makeyev, E.V.; Bamford, D.H. Replicase activity of purified recombinant protein P2 of double-stranded RNA bacteriophage phi6. The EMBO journal 2000, 19, 124-133. [CrossRef]
- Gottlieb, P.; Strassman, J.; Mindich, L. Protein P4 of the bacteriophage phi 6 procapsid has a nucleoside triphosphate-binding site with associated nucleoside triphosphate phosphohydrolase activity. J Virol 1992, 66, 6220-6222. [CrossRef]
- Paatero, A.O.; Syvaoja, J.E.; Bamford, D.H. Double-stranded RNA bacteriophage phi 6 protein P4 is an unspecific nucleoside triphosphatase activated by calcium ions. J Virol 1995, 69, 6729-6734. [CrossRef]
- Juuti, J.T.; Bamford, D.H. RNA binding, packaging and polymerase activities of the different incomplete polymerase complex particles of dsRNA bacteriophage phi 6. J Mol Biol 1995, 249, 545-554. [CrossRef]
- Juuti, J.T.; Bamford, D.H. Protein P7 of phage phi6 RNA polymerase complex, acquiring of RNA packaging activity by in vitro assembly of the purified protein onto deficient particles. J Mol Biol 1997, 266, 891-900. [CrossRef]
- Olkkonen, V.M.; Bamford, D.H. The nucleocapsid of the lipid-containing double-stranded RNA bacteriophage phi 6 contains a protein skeleton consisting of a single polypeptide species. J Virol 1987, 61, 2362-2367. [CrossRef]
- Butcher, S.J.; Dokland, T.; Ojala, P.M.; Bamford, D.H.; Fuller, S.D. Intermediates in the assembly pathway of the double-stranded RNA virus phi6. Embo J 1997, 16, 4477-4487. [CrossRef]
- Huiskonen, J.T.; de Haas, F.; Bubeck, D.; Bamford, D.H.; Fuller, S.D.; Butcher, S.J. Structure of the bacteriophage phi6 nucleocapsid suggests a mechanism for sequential RNA packaging. Structure 2006, 14, 1039-1048.
- Sen, A.; Heymann, J.B.; Cheng, N.; Qiao, J.; Mindich, L.; Steven, A.C. Initial location of the RNA-dependent RNA polymerase in the bacteriophage Phi6 procapsid determined by cryo-electron microscopy. J Biol Chem 2008, 283, 12227-12231. [CrossRef]
- Nemecek, D.; Heymann, J.B.; Qiao, J.; Mindich, L.; Steven, A.C. Cryo-electron tomography of bacteriophage φ6 procapsids shows random occupancy of the binding sites for RNA polymerase and packaging NTPase. J. Struct. Biol. 2010, 171, 389-396. [CrossRef]
- Katz, G.; Wei, H.; Alimova, A.; Katz, A.; Morgan, D.G.; Gottlieb, P. Protein P7 of the cystovirus phi6 is located at the three-fold axis of the unexpanded procapsid. PloS one 2012, 7, e47489. [CrossRef]
- Nemecek, D.; Qiao, J.; Mindich, L.; Steven, A.C.; Heymann, J.B. Packaging accessory protein P7 and polymerase P2 have mutually occluding binding sites inside the bacteriophage φ6 procapsid. J. Virol. 2012, 86, 11616-11624. [CrossRef]
- Luque, D.; Mata, C.P.; Suzuki, N.; Ghabrial, S.A.; Caston, J.R. Capsid Structure of dsRNA Fungal Viruses. Viruses 2018, 10. [CrossRef]
- Nemecek, D.; Boura, E.; Wu, W.; Cheng, N.; Plevka, P.; Qiao, J.; Mindich, L.; Heymann, J.B.; Hurley, J.H.; Steven, A.C. Subunit folds and maturation pathway of a dsRNA virus capsid. Structure 2013, 21, 1374-1383. [CrossRef]
- Jaalinoja, H.T.; Huiskonen, J.T.; Butcher, S.J. Electron cryomicroscopy comparison of the architectures of the enveloped bacteriophages phi6 and phi8. Structure 2007, 15, 157-167.
- Caspar, D.L.; Klug, A. Physical principles in the construction of regular viruses. Cold Spring Harb Symp Quant Biol 1962, 27, 1-24. [CrossRef]
- Juuti, J.T.; Bamford, D.H.; Tuma, R.; Thomas, G.J., Jr. Structure and NTPase activity of the RNA-translocating protein (P4) of bacteriophage phi 6. J Mol Biol 1998, 279, 347-359. [CrossRef]
- El Omari, K.; Meier, C.; Kainov, D.; Sutton, G.; Grimes, J.M.; Poranen, M.M.; Bamford, D.H.; Tuma, R.; Stuart, D.I.; Mancini, E.J. Tracking in atomic detail the functional specializations in viral RecA helicases that occur during evolution. Nucleic Acids Res 2013, 41, 9396-9410. [CrossRef]
- de Haas, F.; Paatero, A.O.; Mindich, L.; Bamford, D.H.; Fuller, S.D. A symmetry mismatch at the site of RNA packaging in the polymerase complex of dsRNA bacteriophage phi6. J Mol Biol 1999, 294, 357-372. [CrossRef]
- Sun, X.; Bamford, D.H.; Poranen, M.M. Probing, by self-assembly, the number of potential binding sites for minor protein subunits in the procapsid of double-stranded RNA bacteriophage Phi6. J. Virol. 2012, 86, 12208-12216. [CrossRef]
- Huiskonen, J.T.; Jaalinoja, H.T.; Briggs, J.A.; Fuller, S.D.; Butcher, S.J. Structure of a hexameric RNA packaging motor in a viral polymerase complex. J Struct Biol 2007, 158, 156-164. [CrossRef]
- Nemecek, D.; Cheng, N.; Qiao, J.; Mindich, L.; Steven, A.C.; Heymann, J.B. Stepwise expansion of the bacteriophage φ6 procapsid: possible packaging intermediates. J. Mol. Biol. 2011, 414, 260-271. [CrossRef]
- Futerman, E. φ6 polymerase conformation; RNA packaging portal stability. City College of New York, CUNY, CUNY Academic Works, 2012.
- Ilca, S.L.; Kotecha, A.; Sun, X.; Poranen, M.M.; Stuart, D.I.; Huiskonen, J.T. Localized reconstruction of subunits from electron cryomicroscopy images of macromolecular complexes. Nat Commun 2015, 6, 8843. [CrossRef]
- Sun, Z.; El Omari, K.; Sun, X.; Ilca, S.L.; Kotecha, A.; Stuart, D.I.; Poranen, M.M.; Huiskonen, J.T. Double-stranded RNA virus outer shell assembly by bona fide domain-swapping. Nat Commun 2017, 8, 14814. [CrossRef]
- Paatero, A.O.; Mindich, L.; Bamford, D.H. Mutational analysis of the role of nucleoside triphosphatase P4 in the assembly of the RNA polymerase complex of bacteriophage phi6. J Virol 1998, 72, 10058-10065. [CrossRef]
- Sinclair, J.F.; Mindich, L. RNA synthesis during infection with bacteriophage phi6. Virology 1976, 75, 209-217. [CrossRef]
- Gottlieb, P.; Strassman, J.; Qiao, X.Y.; Frucht, A.; Mindich, L. In vitro replication, packaging, and transcription of the segmented double-stranded RNA genome of bacteriophage phi 6: studies with procapsids assembled from plasmid-encoded proteins. J Bacteriol 1990, 172, 5774-5782. [CrossRef]
- Butcher, S.J.; Grimes, J.M.; Makeyev, E.V.; Bamford, D.H.; Stuart, D.I. A mechanism for initiating RNA-dependent RNA polymerization. Nature 2001, 410, 235-240. [CrossRef]
- Sarin, L.P.; Wright, S.; Chen, Q.; Degerth, L.H.; Stuart, D.I.; Grimes, J.M.; Bamford, D.H.; Poranen, M.M. The C-terminal priming domain is strongly associated with the main body of bacteriophage varphi6 RNA-dependent RNA polymerase. Virology 2012, 432, 184-193. [CrossRef]
- Poranen, M.M.; Koivunen, M.R.; Bamford, D.H. Nontemplated terminal nucleotidyltransferase activity of double-stranded RNA bacteriophage phi6 RNA-dependent RNA polymerase. J Virol 2008, 82, 9254-9264, . [CrossRef]
- Poranen, M.M.; Salgado, P.S.; Koivunen, M.R.; Wright, S.; Bamford, D.H.; Stuart, D.I.; Grimes, J.M. Structural explanation for the role of Mn2+ in the activity of phi6 RNA-dependent RNA polymerase. Nucleic Acids Res 2008, 36, 6633-6644. [CrossRef]
- Yang, H.; Makeyev, E.V.; Bamford, D.H. Comparison of polymerase subunits from double-stranded RNA bacteriophages. J. Virol. 2001, 75, 11088-11095. [CrossRef]
- Wright, S.; Poranen, M.M.; Bamford, D.H.; Stuart, D.I.; Grimes, J.M. Noncatalytic ions direct the RNA-dependent RNA polymerase of bacterial double-stranded RNA virus varphi6 from de novo initiation to elongation. J. Virol. 2012, 86, 2837-2849. [CrossRef]
- Qiao, J.; Mindich, L. The template specificity of bacteriophage Phi6 RNA polymerase. J Virol 2013, 87, 10190-10194. [CrossRef]
- Sun, X.; Ilca, S.L.; Huiskonen, J.T.; Poranen, M.M. Dual Role of a Viral Polymerase in Viral Genome Replication and Particle Self-Assembly. mBio 2018, 9. [CrossRef]
- Poranen, M.M.; Paatero, A.O.; Tuma, R.; Bamford, D.H. Self-assembly of a viral molecular machine from purified protein and RNA constituents. Mol Cell 2001, 7, 845-854. [CrossRef]
- Eryilmaz, E.; Benach, J.; Su, M.; Seetharaman, J.; Dutta, K.; Wei, H.; Gottlieb, P.; Hunt, J.F.; Ghose, R. Structure and dynamics of the P7 protein from the bacteriophage phi 12. J Mol Biol 2008, 382, 402-422. [CrossRef]
- Wei, H.; Cheng, R.H.; Berriman, J.; Rice, W.J.; Stokes, D.L.; Katz, A.; Morgan, D.G.; Gottlieb, P. Three-dimensional structure of the enveloped bacteriophage phi12: an incomplete T = 13 lattice is superposed on an enclosed T = 1 shell. PLoS One 2009, 4, e6850. [CrossRef]
- Ikonen, T.; Kainov, D.; Timmins, P.; Serimaa, R.; Tuma, R. Locating the minor components of double-stranded RNA bacteriophage 6 by neutron scattering. J. Appl. Crystall. 2003, 36, 525-529. [CrossRef]
- El Omari, K.; Sutton, G.; Ravantti, J.J.; Zhang, H.; Walter, T.S.; Grimes, J.M.; Bamford, D.H.; Stuart, D.I.; Mancini, E.J. Plate tectonics of virus shell assembly and reorganization in phage phi8, a distant relative of mammalian reoviruses. Structure 2013, 21, 1384-1395. [CrossRef]
- Dokland, T. Scaffolding proteins and their role in viral assembly. Cell Mol Life Sci 1999, 56, 580-603. [CrossRef]
- Trask, S.D.; McDonald, S.M.; Patton, J.T. Structural insights into the coupling of virion assembly and rotavirus replication. Nature reviews. Microbiology 2012, 10, 165-177. [CrossRef]
- Frilander, M.; Bamford, D.H. In vitro packaging of the single-stranded RNA genomic precursors of the segmented double-stranded RNA bacteriophage phi 6: the three segments modulate each other's packaging efficiency. J Mol Biol 1995, 246, 418-428. [CrossRef]
- Pirttimaa, M.J.; Paatero, A.O.; Frilander, M.J.; Bamford, D.H. Nonspecific nucleoside triphosphatase P4 of double-stranded RNA bacteriophage phi6 is required for single-stranded RNA packaging and transcription. J Virol 2002, 76, 10122-10127. [CrossRef]
- Ilca, S.L.; Sun, X.; El Omari, K.; Kotecha, A.; de Haas, F.; DiMaio, F.; Grimes, J.M.; Stuart, D.I.; Poranen, M.M.; Huiskonen, J.T. Multiple liquid crystalline geometries of highly compacted nucleic acid in a dsRNA virus. Nature 2019, 570, 252-256. [CrossRef]
- Liu, H.; Cheng, L. Cryo-EM shows the polymerase structures and a nonspooled genome within a dsRNA virus. Science 2015, 349, 1347-1350. [CrossRef]
- Zhang, X.; Ding, K.; Yu, X.; Chang, W.; Sun, J.; Zhou, Z.H. In situ structures of the segmented genome and RNA polymerase complex inside a dsRNA virus. Nature 2015, 527, 531-534. [CrossRef]
- Strauss, S.; Acker, J.; Papa, G.; Desiro, D.; Schueder, F.; Borodavka, A.; Jungmann, R. Principles of RNA recruitment to viral ribonucleoprotein condensates in a segmented dsRNA virus. Elife 2023, 12. [CrossRef]
- Tuma, R.; Bamford, J.K.; Bamford, D.H.; Thomas, G.J., Jr. Assembly dynamics of the nucleocapsid shell subunit (P8) of bacteriophage phi6. Biochemistry 1999, 38, 15025-15033. [CrossRef]
- Cvirkaite-Krupovic, V.; Poranen, M.M.; Bamford, D.H. Phospholipids act as secondary receptor during the entry of the enveloped, double-stranded RNA bacteriophage phi6. J Gen Virol 2010, 91, 2116-2120. [CrossRef]
- Romantschuk, M.; Olkkonen, V.M.; Bamford, D.H. The nucleocapsid of bacteriophage phi 6 penetrates the host cytoplasmic membrane. Embo J 1988, 7, 1821-1829. [CrossRef]
- Laurinavicius, S.; Kakela, R.; Bamford, D.H.; Somerharju, P. The origin of phospholipids of the enveloped bacteriophage phi6. Virology 2004, 326, 182-190. [CrossRef]
- Sinclair, J.F.; Tzagoloff, A.; Levine, D.; Mindich, L. Proteins of bacteriophage phi6. J Virol 1975, 16, 685-695. [CrossRef]
- Van Etten, J.; Lane, L.; Gonzalez, C.; Partridge, J.; Vidaver, A. Comparative properties of bacteriophage phi6 and phi6 nucleocapsid. J Virol 1976, 18, 652-658. [CrossRef]
- Stitt, B.L.; Mindich, L. The structure of bacteriophage phi 6: protease digestion of phi 6 virions. Virology 1983, 127, 459-462. [CrossRef]
- Stitt, B.L.; Mindich, L. Morphogenesis of bacteriophage phi 6: a presumptive viral membrane precursor. Virology 1983, 127, 446-458. [CrossRef]
- Lyytinen, O.L.; Starkova, D.; Poranen, M.M. Microbial production of lipid-protein vesicles using enveloped bacteriophage phi6. Microb Cell Fact 2019, 18, 29. [CrossRef]
- Kenney, J.M.; Hantula, J.; Fuller, S.D.; Mindich, L.; Ojala, P.M.; Bamford, D.H. Bacteriophage phi 6 envelope elucidated by chemical cross-linking, immunodetection, and cryoelectron microscopy. Virology 1992, 190, 635-644. [CrossRef]
- Leo-Macias, A.; Katz, G.; Wei, H.; Alimova, A.; Katz, A.; Rice, W.J.; Diaz-Avalos, R.; Hu, G.B.; Stokes, D.L.; Gottlieb, P. Toroidal surface complexes of bacteriophage varphi12 are responsible for host-cell attachment. Virology 2011, 414, 103-109. [CrossRef]
- Emori, Y.; Iba, H.; Okada, Y. Semi-conservative transcription of double-stranded RNA catalyzed by bacteriophage phi 6 RNA polymerase. J Biochem 1980, 88, 1569-1575.
- Coplin, D.L.; Van Etten, J.L.; Koski, R.K.; Vidaver, A.K. Intermediates in the biosynthesis of double-stranded ribonucleic acids of bacteriophage phi 6. Proc Natl Acad Sci U S A 1975, 72, 849-853. [CrossRef]
- Emori, Y.; Iba, H.; Okada, Y. Transcriptional regulation of three double-stranded RNA segments of bacteriophage phi 6 in vitro. J Virol 1983, 46, 196-203. [CrossRef]
- Ojala, P.M.; Bamford, D.H. In vitro transcription of the double-stranded RNA bacteriophage phi 6 is influenced by purine NTPs and calcium. Virology 1995, 207, 400-408. [CrossRef]
- Miron, S.; Borza, T.; Saveanu, C.; Gilles, A.M.; Barzu, O.; Craescu, C.T. 1H, 13C and 15N resonance assignment of YajQ, a protein of unknown structure and function from Escherichia coli. J Biomol NMR 2001, 20, 287-288. [CrossRef]
- Qiao, J.; Qiao, X.; Sun, Y.; Mindich, L. Role of host protein glutaredoxin 3 in the control of transcription during bacteriophage Phi2954 infection. Proc Natl Acad Sci U S A 2010, 107, 6000-6004. [CrossRef]
- Qiao, X.; Sun, Y.; Qiao, J.; Mindich, L. Interaction of a host protein with core complexes of bacteriophage phi6 to control transcription. J Virol 2010, 84, 4821-4825. [CrossRef]
- Heymann, J.B.; Nemecek, D.; Huang, R.; Cheng, N.; Qiao, J.; Mindich, L.; Steven, A.C. A Polymerase-Activating Host Factor, YajQ, Bound to the Bacteriophage ϕ6 Capsid. Microscopy and Microanalysis 2016, 22, 1110-1111. [CrossRef]
- Teplyakov, A.; Obmolova, G.; Bir, N.; Reddy, P.; Howard, A.J.; Gilliland, G.L. Crystal structure of the YajQ protein from Haemophilus influenzae reveals a tandem of RNP-like domains. J Struct Funct Genomics 2003, 4, 1-9. [CrossRef]
- McDonald, S.M.; Nelson, M.I.; Turner, P.E.; Patton, J.T. Reassortment in segmented RNA viruses: mechanisms and outcomes. Nat Rev Microbiol 2016, 14, 448-460. [CrossRef]
- Mindich, L. Precise packaging of the three genomic segments of the double-stranded-RNA bacteriophage phi6. Microbiol Mol Biol Rev 1999, 63, 149-160. [CrossRef]
- Rao, V.B.; Feiss, M. Mechanisms of DNA Packaging by Large Double-Stranded DNA Viruses. Annu Rev Virol 2015, 2, 351-378. [CrossRef]
- Haralampiev, I.; Prisner, S.; Nitzan, M.; Schade, M.; Jolmes, F.; Schreiber, M.; Loidolt-Kruger, M.; Jongen, K.; Chamiolo, J.; Nilson, N.; et al. Selective flexible packaging pathways of the segmented genome of influenza A virus. Nat Commun 2020, 11, 4355. [CrossRef]
- Ye, L.; Ambi, U.B.; Olguin-Nava, M.; Gribling-Burrer, A.S.; Ahmad, S.; Bohn, P.; Weber, M.M.; Smyth, R.P. RNA Structures and Their Role in Selective Genome Packaging. Viruses 2021, 13. [CrossRef]
- Selivanovitch, E.; Douglas, T. Virus capsid assembly across different length scales inspire the development of virus-based biomaterials. Curr Opin Virol 2019, 36, 38-46. [CrossRef]
- Perlmutter, J.D.; Hagan, M.F. Mechanisms of virus assembly. Annu Rev Phys Chem 2015, 66, 217-239. [CrossRef]
| 1 | The segments are denoted in lower case for single stranded RNA and in upper case for double stranded RNA. See 8. Frilander, M.; Poranen, M.; Bamford, D.H. The large genome segment of dsRNA bacteriophage phi6 is the key regulator in the in vitro minus and plus strand synthesis. RNA 1995, 1, 510-518. for examples. |
| 2 | The term nucleocapsid is used with or without the P8 layer. |
| 3 | An example of "quasi-equivalence" of the subunits of the capsid 37. Caspar, D.L.; Klug, A. Physical principles in the construction of regular viruses. Cold Spring Harb Symp Quant Biol 1962, 27, 1-24.. |
| Composition | Method* | P2 | P4 | P7 | Reference |
| P1247 | C | 8 | 5# | - | [31] |
| D | 7 | 25 | 10 | [33] | |
| S | 4 | 29 | 12 | [33] | |
| D | 10 | 34 | [32] | ||
| P124 | D | 6 | 31 | [33] | |
| S | 4 | 40 | [33] | ||
| D | 6 | [32] | |||
| P147 | D | 33 | 3 | [33] | |
| S | 35 | 12 | [33] | ||
| D | 11 | [32] | |||
| Maximum | 20 | 72 | 60 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




