Submitted:
10 October 2023
Posted:
16 October 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Ethics
2.2. Diet preparation
2.2. Experimental design
2.3. Growth performance and conversion efficiency of the BSF larvae
2.4. Diet ingredients and larval composition analysis
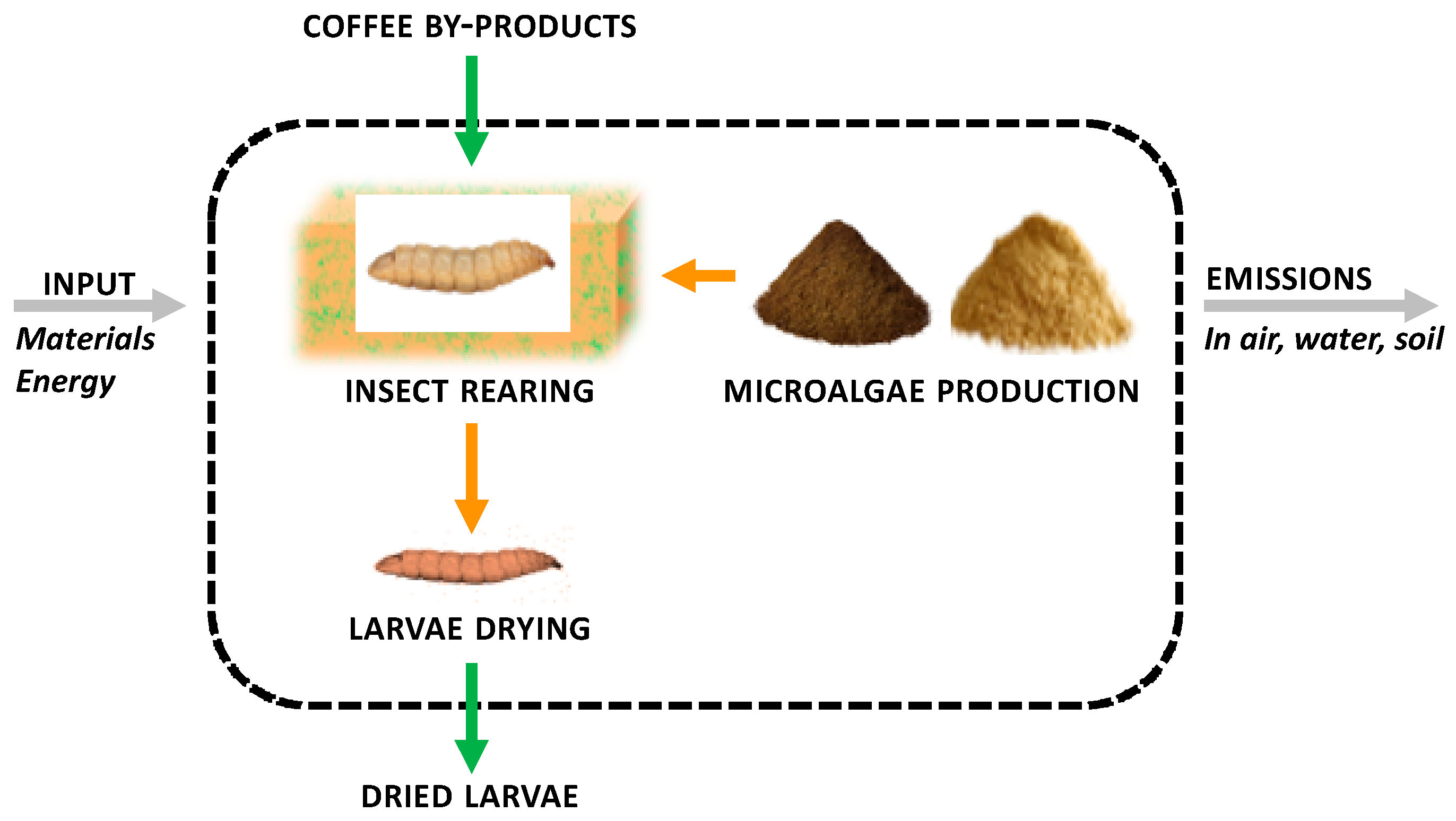
2.5. Life Cycle Assessment
2.5.1. Goal and Scope
2.5.2. Life Cycle Inventory
2.5.3. Life Cycle Impact Assessment
2.6. Statistical analysis
3. Results
3.1. Growth performance and conversion efficiency of the BSF larvae
3.2. Diet ingredient and larva composition analysis
3.2.1. Diet composition analysis
3.2.2. Insect composition analysis
3.3. Life Cycle Assessment
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Tucker, C.M. Coffee Culture: Local Experiences, Global Connections, 2nd ed.; Taylor & Francis: Abingdon, UK, 2017; ISBN 9781317392255. [Google Scholar]
- ICO. Historical data on the global coffee trade. 2020. Available online: http://www.ico.org/new_historical.asp?section= Statistics (accessed on 17 November 2020).
- Alves, R.C.; Rodrigues, F.; Antónia Nunes, M.; Vinha, A.F.; Oliveira, M.B.P.P. State of the art in coffee processing by-products. In Handbook of Coffee Processing By-Products; Elsevier, 2017; pp. 1–26 ISBN 978-0-12-811290-8.
- Mussatto, S.I.; Machado, E.M.S.; Martins, S.; Teixeira, J.A. Production, composition, and application of coffee and its industrial residues. Food Bioprocess Technol. 2011, 4, 661–672. [Google Scholar] [CrossRef]
- Tores De La Cruz, S.; Iriondo-DeHond, A.; Herrera, T.; Lopez-Tofiño, Y.; Galvez-Robleño, C.; Prodanov, M.; Velazquez-Escobar, F.; Abalo, R.; Castillo, M.D.D. An assessment of the bioactivity of coffee silverskin melanoidins. Foods 2019, 8, 68. [Google Scholar] [CrossRef]
- Ballesteros, L.F.; Teixeira, J.A.; Mussatto, S.I. Chemical, functional, and structural properties of spent coffee grounds and coffee silverskin. Food Bioprocess Technol. 2014, 7, 3493–3503. [Google Scholar] [CrossRef]
- Sung, S.H.; Chang, Y.; Han, J. Development of polylactic acid nanocomposite films reinforced with cellulose nanocrystals derived from coffee silverskin. Carbohydrate Polymers 2017, 169, 495–503. [Google Scholar] [CrossRef]
- Hijosa-Valsero, M.; Garita-Cambronero, J.; Paniagua-García, A.I.; Díez-Antolínez, R. Biobutanol production from coffee silverskin. Microb. Cell Fact. 2018, 17, 154. [Google Scholar] [CrossRef]
- Bessada, S.M.F.; Alves, R.C.; Costa, A.S.G.; Nunes, M.A.; Oliveira, M.B.P.P. Coffea canephora silverskin from different geographical origins: a comparative study. Science of The Total Environment 2018, 645, 1021–1028. [Google Scholar] [CrossRef]
- Iriondo-DeHond, A.; Iriondo-DeHond, M.; Del Castillo, M.D. Applications of compounds from coffee processing by-products. Biomolecules 2020, 10, 1219. [Google Scholar] [CrossRef]
- Diener, S.; Zurbrügg, C.; Tockner, K. Conversion of organic material by black soldier fly larvae: establishing optimal feeding rates. Waste Manag. Res. 2009, 27, 603–610. [Google Scholar] [CrossRef]
- Oonincx, D.G.A.B.; Van Broekhoven, S.; Van Huis, A.; Van Loon, J.J.A. Feed conversion, survival and development, and composition of four insect species on diets composed of food by-products. PLoS ONE 2015, 10, e0144601. [Google Scholar] [CrossRef]
- Parra Paz, A.S.; Carrejo, N.S.; Gómez Rodríguez, C.H. Effects of larval density and feeding rates on the bioconversion of vegetable waste using black soldier fly larvae Hermetia Illucens (L.), (Diptera: Stratiomyidae). Waste Biomass Valor. 2015, 6, 1059–1065. [Google Scholar] [CrossRef]
- Spranghers, T.; Ottoboni, M.; Klootwijk, C.; Ovyn, A.; Deboosere, S.; De Meulenaer, B.; Michiels, J.; Eeckhout, M.; De Clercq, P.; De Smet, S. Nutritional composition of black soldier fly (hermetia illucens) prepupae reared on different organic waste substrates: nutritional composition of black soldier fly. J. Sci. Food Agric. 2017, 97, 2594–2600. [Google Scholar] [CrossRef]
- Jucker, C.; Erba, D.; Leonardi, M.G.; Lupi, D.; Savoldelli, S. Assessment of vegetable and fruit substrates as potential rearing media for Hermetia Illucens (Diptera: Stratiomyidae) larvae. Environmental Entomology 2017, 46, 1415–1423. [Google Scholar] [CrossRef] [PubMed]
- Salomone, R.; Saija, G.; Mondello, G.; Giannetto, A.; Fasulo, S.; Savastano, D. Environmental impact of food waste bioconversion by insects: Application of life cycle assessment to process Using Hermetia Illucens. Journal of Cleaner Production 2017, 140, 890–905. [Google Scholar] [CrossRef]
- Meneguz, M.; Schiavone, A.; Gai, F.; Dama, A.; Lussiana, C.; Renna, M.; Gasco, L. Effect of rearing substrate on growth performance, waste reduction efficiency and chemical composition of black soldier fly (Hermetia Illucens) Larvae: J. Sci. Food Agric. 2018, 98, 5776–5784. [Google Scholar] [CrossRef]
- Kawasaki, K.; Hashimoto, Y.; Hori, A.; Kawasaki, T.; Hirayasu, H.; Iwase, S.; Hashizume, A.; Ido, A.; Miura, C.; Miura, T.; et al. Evaluation of black soldier fly (Hermetia illucens) larvae and pre-pupae raised on household organic waste, as potential ingredients for poultry feed. Animals 2019, 9, 98. [Google Scholar] [CrossRef]
- Bava, L.; Jucker, C.; Gislon, G.; Lupi, D.; Savoldelli, S.; Zucali, M.; Colombini, S. Rearing of Hermetia illucens on different organic by-products: influence on growth, waste reduction, and environmental impact. Animals 2019, 9, 289. [Google Scholar] [CrossRef]
- Scala, A.; Cammack, J.A.; Salvia, R.; Scieuzo, C.; Franco, A.; Bufo, S.A.; Tomberlin, J.K.; Falabella, P. Rearing substrate impacts growth and macronutrient composition of Hermetia illucens (L.) (diptera: stratiomyidae) larvae produced at an industrial scale. Sci. Rep. 2020, 10, 19448. [Google Scholar] [CrossRef]
- Hadj Saadoun, J.; Montevecchi, G.; Zanasi, L.; Bortolini, S.; Macavei, L.I.; Masino, F.; Maistrello, L.; Antonelli, A. Lipid profile and growth of black soldier flies (Hermetia illucens, Stratiomyidae) reared on by-products from different food chains. J. Sci. Food Agric. 2020, 100, 3648–3657. [Google Scholar] [CrossRef]
- Van Huis, A.; Oonincx, D.G.A.B.; Rojo, S.; Tomberlin, J.K. Insects as feed: house fly or black soldier fly? Journal of Insects as Food and Feed 2020, 6, 221–229. [Google Scholar] [CrossRef]
- Gasco, L.; Biancarosa, I.; Liland, N.S. From Waste to Feed: A Review of recent knowledge on insects as producers of protein and fat for animal feeds. Current Opinion in Green and Sustainable Chemistry 2020, 23, 67–79. [Google Scholar] [CrossRef]
- Barbi, S.; Macavei, L.I.; Fuso, A.; Luparelli, A.V.; Caligiani, A.; Ferrari, A.M.; Maistrello, L.; Montorsi, M. Valorization of seasonal agri-food leftovers through insects. Science of The Total Environment 2020, 709, 136209. [Google Scholar] [CrossRef]
- Zarantoniello, M.; Randazzo, B.; Nozzi, V.; Truzzi, C.; Giorgini, E.; Cardinaletti, G.; Freddi, L.; Ratti, S.; Girolametti, F.; Osimani, A.; et al. Physiological responses of Siberian sturgeon (Acipenser baerii) juveniles fed on full-fat insect-based diet in an aquaponic system. Sci. Rep. 2021, 11, 1057. [Google Scholar] [CrossRef]
- Poveda, J. Insect frass in the development of sustainable agriculture. A review. Agron. Sustain. Dev. 2021, 41, 5. [Google Scholar] [CrossRef]
- Müller, A.; Wolf, D.; Gutzeit, H.O. The black soldier fly, Hermetia illucens—a promising source for sustainable production of proteins, lipids and bioactive substances. Zeitschrift für Naturforschung C 2017, 72, 351–363. [Google Scholar] [CrossRef]
- Randazzo, B.; Zarantoniello, M.; Cardinaletti, G.; Cerri, R.; Giorgini, E.; Belloni, A.; Contò, M.; Tibaldi, E.; Olivotto, I. Hermetia illucens and poultry by-product meals as alternatives to plant protein sources in gilthead seabream (Sparus aurata) diet: a multidisciplinary study on fish gut status. Animals 2021, 11, 677. [Google Scholar] [CrossRef]
- Makkar, H.P.S.; Tran, G.; Heuzé, V.; Ankers, P. State-of-the-art on use of insects as animal feed. Animal Feed Science and Technology 2014, 197, 1–33. [Google Scholar] [CrossRef]
- Barragan-Fonseca, K.B.; Dicke, M.; Van Loon, J.J.A. Nutritional value of the black soldier fly (Hermetia illucens L.) and its suitability as animal feed – A review. Journal of Insects as Food and Feed 2017, 3, 105–120. [Google Scholar] [CrossRef]
- Vargas, A.; Randazzo, B.; Riolo, P.; Truzzi, C.; Gioacchini, G.; Giorgini, E.; Loreto, N.; Ruschioni, S.; Zarantoniello, M.; Antonucci, M.; et al. Rearing zebrafish on black soldier fly (Hermetia illucens): biometric, histological, spectroscopic, biochemical, and molecular implications. Zebrafish 2018, 15, 404–419. [Google Scholar] [CrossRef]
- Zarantoniello, M.; Bruni, L.; Randazzo, B.; Vargas, A.; Gioacchini, G.; Truzzi, C.; Annibaldi, A.; Riolo, P.; Parisi, G.; Cardinaletti, G.; et al. Partial dietary inclusion of hermetia illucens (black soldier fly) full-fat prepupae in zebrafish feed: biometric, histological, biochemical, and molecular implications. Zebrafish 2018, 15, 519–532. [Google Scholar] [CrossRef]
- Cardinaletti, G.; Randazzo, B.; Messina, M.; Zarantoniello, M.; Giorgini, E.; Zimbelli, A.; Bruni, L.; Parisi, G.; Olivotto, I.; Tulli, F. Effects of graded dietary inclusion level of full-fat Hermetia illucens prepupae meal in practical diets for rainbow trout (Oncorhynchus mykiss). Animals 2019, 9, 251. [Google Scholar] [CrossRef]
- Giannetto, A.; Oliva, S.; Riolo, K.; Savastano, D.; Parrino, V.; Cappello, T.; Maisano, M.; Fasulo, S.; Mauceri, A. Waste valorization via Hermetia illucens to produce protein-rich biomass for feed: insight into the critical nutrient taurine. Animals 2020, 10, 1710. [Google Scholar] [CrossRef]
- St-Hilaire, S.; Cranfill, K.; McGuire, M.A.; Mosley, E.E.; Tomberlin, J.K.; Newton, L.; Sealey, W.; Sheppard, C.; Irving, S. Fish offal recycling by the black soldier fly produces a foodstuff high in omega-3 fatty acids. J. World Aquaculture Soc. 2007, 38, 309–313. [Google Scholar] [CrossRef]
- Liland, N.S.; Biancarosa, I.; Araujo, P.; Biemans, D.; Bruckner, C.G.; Waagbø, R.; Torstensen, B.E.; Lock, E.-J. Modulation of nutrient composition of black soldier fly (Hermetia illucens) larvae by feeding seaweed-enriched media. PLoS ONE 2017, 12, e0183188. [Google Scholar] [CrossRef]
- Pinotti, L.; Giromini, C.; Ottoboni, M.; Tretola, M.; Marchis, D. Review: Insects and former foodstuffs for upgrading food waste biomasses/streams to feed ingredients for farm animals. Animals 2019, 13, 1365–1375. [Google Scholar] [CrossRef]
- Hoc, B.; Genva, M.; Fauconnier, M.-L.; Lognay, G.; Francis, F.; Caparros Megido, R. About lipid metabolism in Hermetia illucens (L. 1758): on the origin of fatty acids in prepupae. Sci. Rep. 2020, 10, 11916. [Google Scholar] [CrossRef]
- Zarantoniello, M.; Zimbelli, A.; Randazzo, B.; Compagni, M.D.; Truzzi, C.; Antonucci, M.; Riolo, P.; Loreto, N.; Osimani, A.; Milanović, V.; et al. Black soldier fly (Hermetia Illucens) reared on roasted coffee by-product and Schizochytrium sp. as a sustainable terrestrial ingredient for aquafeeds production. Aquaculture 2020, 518, 734659. [Google Scholar] [CrossRef]
- Brown, J.H.; Gillooly, J.F.; Allen, A.P.; Savage, V.M.; West, G.B. Toward a metabolic theory of ecology. Ecology 2004, 85, 1771–1789. [Google Scholar] [CrossRef]
- Nguyen, T.T.X.; Tomberlin, J.K.; Vanlaerhoven, S. Influence of resources on Hermetia illucens (Diptera: Stratiomyidae) larval development. jnl. med. entom. 2013, 50, 898–906. [Google Scholar] [CrossRef]
- Cammack, J.; Tomberlin, J. The impact of diet protein and carbohydrate on select life-history traits of the black soldier fly Hermetia illucens (L.) (diptera: stratiomyidae). Insects 2017, 8, 56. [Google Scholar] [CrossRef]
- Gligorescu, A.; Toft, S.; Hauggaard-Nielsen, H.; Axelsen, J.A.; Nielsen, S.A. Development, metabolism and nutrient composition of black soldier fly larvae (Hermetia illucens; Diptera: Stratiomyidae) in relation to temperature and diet. Journal of Insects as Food and Feed 2018, 4, 123–133. [Google Scholar] [CrossRef]
- Smetana, S.; Palanisamy, M.; Mathys, A.; Heinz, V. Sustainability of insect use for feed and food: life cycle assessment perspective. Journal of Cleaner Production 2016, 137, 741–751. [Google Scholar] [CrossRef]
- Smetana, S.; Schmitt, E.; Mathys, A. Sustainable use of Hermetia illucens insect biomass for feed and food: attributional and consequential life cycle assessment. Resources, Conservation and Recycling 2019, 144, 285–296. [Google Scholar] [CrossRef]
- Maiolo, S.; Parisi, G.; Biondi, N.; Lunelli, F.; Tibaldi, E.; Pastres, R. Fishmeal partial substitution within aquafeed formulations: life cycle assessment of four alternative protein sources. Int. J. Life Cycle Assess. 2020, 25, 1455–1471. [Google Scholar] [CrossRef]
- Zarantoniello, M.; Randazzo, B.; Secci, G.; Notarstefano, V.; Giorgini, E.; Lock, E.J.; Parisi, G.; Olivotto, I. Application of laboratory methods for understanding fish responses to black soldier fly (Hermetia illucens) based diets. Journal of Insects as Food and Feed 2022, 8, 1173–1195. [Google Scholar] [CrossRef]
- Gambelli, D.; Naspetti, S.; Zander, K.; Zanoli, R. Organic aquaculture: economic, market and consumer aspects. In Organic Aquaculture; Lembo, G., Mente, E., Eds.; Springer International Publishing: Cham, 2019; ISBN 978-3-030-05602-5. [Google Scholar]
- Brown, M.R.; Farmer, C.L. Riboflavin content of six species of microalgae used in mariculture. J. Appl. Phycol. 1994, 6, 61–65. [Google Scholar] [CrossRef]
- Nakahara, T.; Yokochi, T.; Higashihara, T.; Tanaka, S.; Yaguchi, T.; Honda, D. Production of docosahexaenoic and docosapentaenoic acids by Schizochytrium sp. isolated from yap islands. J. Am. Oil. Chem. Soc. 1996, 73, 1421–1426. [Google Scholar] [CrossRef]
- Ren, L.-J.; Ji, X.-J.; Huang, H.; Qu, L.; Feng, Y.; Tong, Q.-Q.; Ouyang, P.-K. Development of a stepwise aeration control strategy for efficient docosahexaenoic acid production by Schizochytrium sp. Appl. Microbiol. Biotechnol. 2010, 87, 1649–1656. [Google Scholar] [CrossRef] [PubMed]
- Fitzer, S.C.; Plancq, J.; Floyd, C.J.; Kemp, F.M.; Toney, J.L. Increased pCO2 changes the lipid production in important aquacultural feedstock algae Isochrysis galbana, but not in Tetraselmis suecica. Aquaculture and Fisheries 2019, 4, 142–148. [Google Scholar] [CrossRef]
- Bruni, L.; Randazzo, B.; Cardinaletti, G.; Zarantoniello, M.; Mina, F.; Secci, G.; Tulli, F.; Olivotto, I.; Parisi, G. Dietary inclusion of full-fat Hermetia illucens prepupae meal in practical diets for rainbow trout (Oncorhynchus mykiss): lipid metabolism and fillet quality investigations. Aquaculture 2020, 529, 735678. [Google Scholar] [CrossRef]
- Truzzi, C.; Giorgini, E.; Annibaldi, A.; Antonucci, M.; Illuminati, S.; Scarponi, G.; Riolo, P.; Isidoro, N.; Conti, C.; Zarantoniello, M.; et al. Fatty Acids Profile of Black Soldier Fly (Hermetia Illucens): Influence of feeding substrate based on coffee-waste silverskin enriched with microalgae. Animal Feed Science and Technology 2020, 259, 114309. [Google Scholar] [CrossRef]
- May, B. M. The occurrence in New Zealand and the life-history of the soldier fly Hermetia illucens (L.)(Diptera: Stratiomyidae). Nzj. Sci. 1961, 55-65, 55–65. [Google Scholar]
- Waldbauer, G.P. The consumption and utilization of food by insects. In Advances in Insect Physiology; Elsevier, 1968; Vol. 5, pp. 229–288 ISBN 978-0-12-024205-4.
- Le Féon, S.; Thévenot, A.; Maillard, F.; Macombe, C.; Forteau, L.; Aubin, J. Life cycle assessment of fish fed with insect meal: case study of mealworm inclusion in trout feed, in France. Aquaculture 2019, 500, 82–91. [Google Scholar] [CrossRef]
- Ermolaev, E.; Lalander, C.; Vinnerås, B. Greenhouse gas emissions from small-scale fly larvae composting with Hermetia illucens. Waste Management 2019, 96, 65–74. [Google Scholar] [CrossRef]
- Mertenat, A.; Diener, S.; Zurbrügg, C. Black soldier fly biowaste treatment—assessment of global warming potential. Waste Management 2019, 84, 173–181. [Google Scholar] [CrossRef] [PubMed]
- Hanserud, O.S.; Cherubini, F.; Øgaard, A.F.; Müller, D.B.; Brattebø, H. Choice of mineral fertilizer substitution principle strongly influences LCA environmental benefits of nutrient cycling in the agri-food system. Science of The Total Environment 2018, 615, 219–227. [Google Scholar] [CrossRef] [PubMed]
- Tschirner, M.; Simon, A. Influence of different growing substrates and processing on the nutrient composition of black soldier fly larvae destined for animal feed. Journal of Insects as Food and Feed 2015, 1, 249–259. [Google Scholar] [CrossRef]
- Liu, Z.; Minor, M.; Morel, P.C.H.; Najar-Rodriguez, A.J. Bioconversion of three organic wastes by black soldier fly (Diptera: Stratiomyidae) larvae. Environmental Entomology 2018, 47, 1609–1617. [Google Scholar] [CrossRef] [PubMed]
- Sheppard, D. C.; Newton, G. L.; Thompson, S. A.; Savage, S. A value added manure management system using the black soldier fly. Bioresource technology.
- Craig Sheppard, D.; Larry Newton, G.; Thompson, S.A.; Savage, S. A value added manure management system using the black soldier fly. Bioresource Technology 1994, 50, 275–279. [Google Scholar] [CrossRef]
- Tomberlin, J.K.; Sheppard, D.C.; Joyce, J.A. Selected life-history traits of black soldier flies (Diptera: Stratiomyidae) reared on three artificial diets. Ann. Entomol. Soc. Am. 2002, 95, 379–386. [Google Scholar] [CrossRef]
- Moreau, J.; Benrey, B.; Thiery, D. Assessing larval food quality for phytophagous insects: are the facts as simple as they appear? Funct. Ecology. 2006, 20, 592–600. [Google Scholar] [CrossRef]
- Newton, G.L.; Sheppard, D.C.; Watson, D.W.; Burtle, G.J.; Dove, C.R.; Tomberlin, J.K.; Thelen, E.E. The black soldier fly, Hermetia illucens, as a manure management/ resource recovery tool. Symposium on the State of the Science of Animal Manure and Waste Management. , San Antonio, TX. (http://www.cals.ncsu.edu/waste_mgt/natlcenter/sanantonio/proceedings. 5–7 January.
- Gobbi, P.; Martínez-Sánchez, A.; Rojo, S. The effects of larval diet on adult life-history traits of the black soldier fly, Hermetia illucens (Diptera: Stratiomyidae). Eur. J. Entomol. 2013, 110, 461–468. [Google Scholar] [CrossRef]
- Myers, H.M.; Tomberlin, J.K.; Lambert, B.D.; Kattes, D. Development of black soldier fly (diptera: stratiomyidae) larvae fed dairy manure. Environ. Entomol. 2008, 37(1), 11–5. [Google Scholar] [CrossRef] [PubMed]
- Barragan-Fonseca, K.B.; Dicke, M.; Van Loon, J.J.A. Influence of larval density and dietary nutrient concentration on performance, body protein, and fat contents of black soldier fly larvae (Hermetia Illucens). Entomol. Exp. Appl. 2018, 166, 761–770. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, T.T.X.; Tomberlin, J.K.; Vanlaerhoven, S. Ability of black soldier fly (Diptera: Stratiomyidae) larvae to recycle food waste. Environmental Entomology 2015, 44, 406–410. [Google Scholar] [CrossRef] [PubMed]
- Tomberlin, J.K.; Adler, P.H.; Myers, H.M. Development of the Black Soldier Fly (Diptera: Stratiomyidae) in Relation to Temperature. Environ. Entomol. 2009, 38, 930–934. [Google Scholar] [CrossRef] [PubMed]
- Chia, S.Y.; Tanga, C.M.; Osuga, I.M.; Mohamed, S.A.; Khamis, F.M.; Salifu, D.; Sevgan, S.; Fiaboe, K.K.M.; Niassy, S.; Van Loon, J.J.A.; et al. Effects of waste stream combinations from brewing industry on performance of black soldier fly, Hermetia Illucens (Diptera: Stratiomyidae). PeerJ 2018, 6, e5885. [Google Scholar] [CrossRef] [PubMed]
- Permana, A.D.; Ramadhani Eka Putra, J.E.N. Growth of black soldier fly (Hermetia Illucens) larvae fed on spent coffee ground. IOP Conf. Ser.: Earth Environ. Sci. 2018, 187, 012070. [Google Scholar] [CrossRef]
- Ujvari, B.; Wallman, J.F.; Madsen, T.; Whelan, M.; Hulbert, A.J. Experimental studies of blowfly (Calliphora stygia) longevity: a little dietary fat is beneficial but too much is detrimental. Comparative Biochemistry and Physiology Part A: Molecular & Integrative Physiology 2009, 154, 383–388. [Google Scholar] [CrossRef]
- Wilkinson, J.M. Re-defining Efficiency of feed use by livestock. Animals 2011, 5, 1014–1022. [Google Scholar] [CrossRef]
- de Vries, M.; De Boer, I.J.M. Comparing environmental impacts for livestock products: A review of life cycle assessments. Livestock Science 2010, 128, 1–11. [Google Scholar] [CrossRef]
- Scriber, J.M.; Slansky, F. The nutritional ecology of immature insects. Annu. Rev. Entomol. 1981, 26, 183–211. [Google Scholar] [CrossRef]
- Rehman, K.U.; Rehman, A.; Cai, M.; Zheng, L.; Xiao, X.; Somroo, A.A.; Wang, H.; Li, W.; Yu, Z.; Zhang, J. Conversion of mixtures of dairy manure and soybean curd residue by black soldier fly larvae (Hermetia Illucens L.). Journal of Cleaner Production 2017, 154, 366–373. [Google Scholar] [CrossRef]
- Friend, W.G. Nutritional requirements of phytophagous insects. Annu. Rev. Entomol. 1958, 3, 57–74. [Google Scholar] [CrossRef]
- Lundy, M.E.; Parrella, M.P. Crickets are not a free lunch: protein capture from scalable organic side-streams via high-density populations of Acheta domesticus. PLoS ONE 2015, 10, e0118785. [Google Scholar] [CrossRef] [PubMed]
- Leong, S.Y.; Kutty, S.R.M.; Malakahmad, A.; Tan, C.K. Feasibility study of biodiesel production using lipids of Hermetia illucens larva fed with organic waste. Waste Management 2016, 47, 84–90. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Muros, M.-J.; Barroso, F.G.; Manzano-Agugliaro, F. Insect meal as renewable source of food for animal feeding: A review. Journal of Cleaner Production 2014, 65, 16–27. [Google Scholar] [CrossRef]
- Pérez-López, P.; De Vree, J.H.; Feijoo, G.; Bosma, R.; Barbosa, M.J.; Moreira, M.T.; Wijffels, R.H.; Van Boxtel, A.J.B.; Kleinegris, D.M.M. Comparative life cycle assessment of real pilot reactors for microalgae cultivation in different seasons. Applied Energy 2017, 205, 1151–1164. [Google Scholar] [CrossRef]
- Ye, J.; Zhou, Q.; Zhang, X.; Hu, Q. Microalgal dewatering using a polyamide thin film composite forward osmosis membrane and fouling mitigation. Algal Research 2018, 31, 421–429. [Google Scholar] [CrossRef]
- Henriksson, P.J.G.; Dickson, M.; Allah, A.N.; Al-Kenawy, D.; Phillips, M. Benchmarking the environmental performance of best management practice and genetic improvements in egyptian aquaculture using life cycle assessment. Aquaculture 2017, 468, 53–59. [Google Scholar] [CrossRef]
| Diets | CB (%) | Schizochytrium limacinum (%) | Isochrysis galbana (%) |
|---|---|---|---|
| 5S | 95 | 5 | - |
| 10S | 90 | 10 | - |
| 20S | 80 | 20 | - |
| 25S | 75 | 25 | - |
| 5I | 95 | - | 5 |
| 10I | 90 | - | 10 |
| 20I | 80 | - | 20 |
| 25I | 75 | - | 25 |
| C | 100 | - | - |
| Diets | Prepupal weight (mg) | Survival rate (%) | Development time (days) | Growth Rate (mg/day) |
|---|---|---|---|---|
| 5S | 143.0 ± 12.4cB | 88.2 ± 6.1aA | 20 ± 0cC | 7.0 ± 0.6cC |
| 10S | 161.3 ± 10.7bcAB | 91.0 ± 5.4aA | 19 ± 0bB | 8.4 ± 0.6bB |
| 20S | 173.9 ± 9.6abA | 87.8 ± 3.1aA | 19 ± 0bB | 9.0 ± 0.5bB |
| 25S | 180.2 ± 11.5aA | 84.5 ± 12.6aA | 18 ± 0aA | 9.9 ± 0.7aA |
| 5I | 107.6 ± 5.1dB | 84.3 ± 3.1aA | 20 ± 0cC | 5.3 ± 0.3eC |
| 10I | 143.1 ± 5.2cA | 82.0 ± 7.2aA | 20 ± 0cC | 7.0 ± 0.3cA |
| 20I | 113.1 ± 11.8dB | 85.2 ± 5.6aA | 19 ± 0bB | 5.8 ± 0.6dB |
| 25I | 132.3 ± 11.2cA | 84.3 ± 6.5aA | 18 ± 0aA | 7.2 ± 0.6cA |
| C | 72.4 ± 7.7eCC | 81.0 ± 8.8aAA | 39 ± 0dDD | 1.8 ± 0.2fDD |
| Diets | WRI (%/day) | FCR |
|---|---|---|
| 5S | 1.5 ± 0.1dC | 5.2 ± 0.3aA |
| 10S | 2.6 ± 0.2aA | 7.21 ± 0.7cB |
| 20S | 2.1 ± 0.3bcB | 5.6 ± 0.8abA |
| 25S | 2.0 ± 0.2cB | 5.0 ± 0.3aA |
| 5I | 1.2 ± 0.1fCD | 5.8 ± 0.3bB |
| 10I | 1.4 ± 0.1eB | 5.1 ± 0.5aA |
| 20I | 2.2 ± 0.1bA | 9.5 ± 0.9dC |
| 25I | 1.3 ± 0.1efBC | 4.5 ± 0.5aA |
| C | 1.2 ± 0.1gDD | 34.2 ± 6.4eCD |
| Diet | Crude protein | Lipid | Ash |
|---|---|---|---|
| 5S | 21.42 ± 1.10 | 0.75 ± 0.06 | 8.62 ± 0.13 |
| 10S | 22.42 ± 3.34 | 0.81 ± 0.04 | 8.71 ± 0.05 |
| 20S | 28.06 ± 1.15 | 0.82 ± 0.05 | 8.84 ± 0.01 |
| 25S | 31.07 ± 2.19 | 0.93 ± 0.03 | 8.91 ± 0.75 |
| 5I | 23.52 ± 2.26 | 1.66 ± 0.10 | 8.80 ± 0.25 |
| 10I | 24.79 ± 3.18 | 2.11 ± 0.06 | 8.65 ± 0.16 |
| 20I | 25.63 ± 2.41 | 2.67 ± 0.10 | 9.22 ± 0.10 |
| 25I | 27.00 ± 3.22 | 3.56 ± 0.05 | 9.79 ± 0.48 |
| C | 20.96 ± 1.32 | 0.71 ± 0.07 | 8.71 ± 0.22 |
| Diet | Crude protein | Lipid | Ash |
|---|---|---|---|
| 5S | 14.86±0.42cDC | 4.05±0.14cC | 7.13±0.13abB |
| 10S | 15.59±0.36bCB | 4.88±0.11bB | 6.34±0.16cbC |
| 20S | 16.85±0.21bBA | 7.74±0.12aA | 4.70±0.26deD |
| 25S | 18.29±0.23aA | 7.93±0.35aA | 4.39±0.13eD |
| 5I | 14.86±0.28cBC | 4.28±0.26cbB | 7.68±0.01abA |
| 10I | 15.59±0.30bAB | 4.00±0.06cB | 6.99±0.77abcAB |
| 20I | 16.85±0.39bA | 3.81±0.03cdB | 5.81±0.29cdCD |
| 25I | 18.29±0.22aA | 4.95±0.07bA | 4.35±0.44eD |
| C | 13.47±0.53dD | 3.18±0.08dD | 8.12±0.20aA |
| Impact category | Unit | 5S | 10S | 20S | 25S |
|---|---|---|---|---|---|
| Global warming | kg CO2 eq | 8,03E+00 | 1,85E+01 | 2,73E+01 | 3,11E+01 |
| Freshwater eutrophication | kg P eq | 3,74E-03 | 7,96E-03 | 1,15E-02 | 1,30E-02 |
| Land use | m2a crop eq | 4,09E-01 | 8,89E-01 | 1,29E+00 | 1,46E+00 |
| Mineral resource scarcity | kg Cu eq | 2,72E-02 | 6,58E-02 | 9,83E-02 | 1,12E-01 |
| Fossil resource scarcity | kg oil eq | 1,97E+00 | 4,63E+00 | 6,87E+00 | 7,83E+00 |
| Water consumption | m3 | 1,75E-01 | 4,05E-01 | 5,98E-01 | 6,80E-01 |
| Impact category | Unit | 5I | 10I | 20I | 25I |
|---|---|---|---|---|---|
| Global warming | kg CO2 eq | 8,78E+00 | 1,37E+01 | 4,48E+01 | 2,77E+01 |
| Freshwater eutrophication | kg P eq | 4,04E-03 | 6,00E-03 | 1,85E-02 | 1,16E-02 |
| Land use | m2a crop eq | 4,43E-01 | 6,66E-01 | 2,09E+00 | 1,31E+00 |
| Mineral resource scarcity | kg Cu eq | 2,99E-02 | 4,79E-02 | 1,63E-01 | 9,97E-02 |
| Fossil resource scarcity | kg oil eq | 2,16E+00 | 3,39E+00 | 1,13E+01 | 6,97E+00 |
| Water consumption | m3 | 1,92E-01 | 2,99E-01 | 9,80E-01 | 6,06E-01 |
| Impact category | Unit | 5S | 10S | 20S | 25S |
|---|---|---|---|---|---|
| Global warming | kg CO2 eq | 1,65E+01 | 3,63E+01 | 5,45E+01 | 6,10E+01 |
| Freshwater eutrophication | kg P eq | 7,69E-03 | 1,56E-02 | 2,30E-02 | 2,55E-02 |
| Land use | m2a crop eq | 8,41E-01 | 1,75E+00 | 2,57E+00 | 2,86E+00 |
| Mineral resource scarcity | kg Cu eq | 5,59E-02 | 1,29E-01 | 1,96E-01 | 2,20E-01 |
| Fossil resource scarcity | kg oil eq | 4,05E+00 | 9,09E+00 | 1,37E+01 | 1,54E+01 |
| Water consumption | m3 | 3,60E-01 | 7,95E-01 | 1,19E+00 | 1,33E+00 |
| Impact category | Unit | 5I | 10I | 20I | 25I |
|---|---|---|---|---|---|
| Global warming | kg CO2 eq | 1,84E+01 | 2,76E+01 | 8,20E+01 | 4,56E+01 |
| Freshwater eutrophication | kg P eq | 8,46E-03 | 1,21E-02 | 3,39E-02 | 1,91E-02 |
| Land use | m2a crop eq | 9,28E-01 | 1,34E+00 | 3,83E+00 | 2,15E+00 |
| Mineral resource scarcity | kg Cu eq | 6,26E-02 | 9,65E-02 | 2,98E-01 | 1,64E-01 |
| Fossil resource scarcity | kg oil eq | 4,52E+00 | 6,83E+00 | 2,07E+01 | 1,15E+01 |
| Water consumption | m3 | 4,02E-01 | 6,02E-01 | 1,79E+00 | 9,97E-01 |
| Impact category | Unit | 5S | 10S | 20S | 25S |
|---|---|---|---|---|---|
| Global warming | kg CO2 eq | 6,06E+01 | 1,16E+02 | 1,19E+02 | 1,41E+02 |
| Freshwater eutrophication | kg P eq | 2,82E-02 | 4,99E-02 | 5,00E-02 | 5,88E-02 |
| Land use | m2a crop eq | 3,09E+00 | 5,58E+00 | 5,61E+00 | 6,61E+00 |
| Mineral resource scarcity | kg Cu eq | 2,05E-01 | 4,13E-01 | 4,27E-01 | 5,07E-01 |
| Fossil resource scarcity | kg oil eq | 1,49E+01 | 2,91E+01 | 2,98E+01 | 3,54E+01 |
| Water consumption | m3 | 1,32E+00 | 2,54E+00 | 2,60E+00 | 3,08E+00 |
| Impact category | Unit | 5I | 10I | 20I | 25I |
|---|---|---|---|---|---|
| Global warming | kg CO2 eq | 6,38E+01 | 1,08E+02 | 3,63E+02 | 1,68E+02 |
| Freshwater eutrophication | kg P eq | 2,94E-02 | 4,71E-02 | 1,50E-01 | 7,05E-02 |
| Land use | m2a crop eq | 3,22E+00 | 5,23E+00 | 1,69E+01 | 7,96E+00 |
| Mineral resource scarcity | kg Cu eq | 2,17E-01 | 3,76E-01 | 1,32E+00 | 6,06E-01 |
| Fossil resource scarcity | kg oil eq | 1,57E+01 | 2,66E+01 | 9,15E+01 | 4,24E+01 |
| Water consumption | m3 | 1,40E+00 | 2,35E+00 | 7,94E+00 | 3,68E+00 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).




