Submitted:
07 October 2023
Posted:
10 October 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
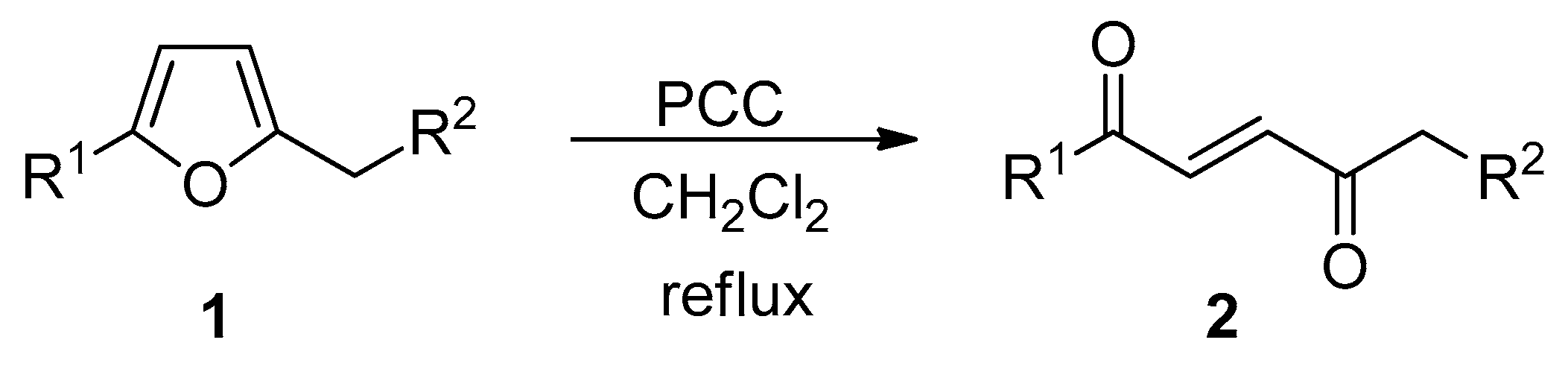
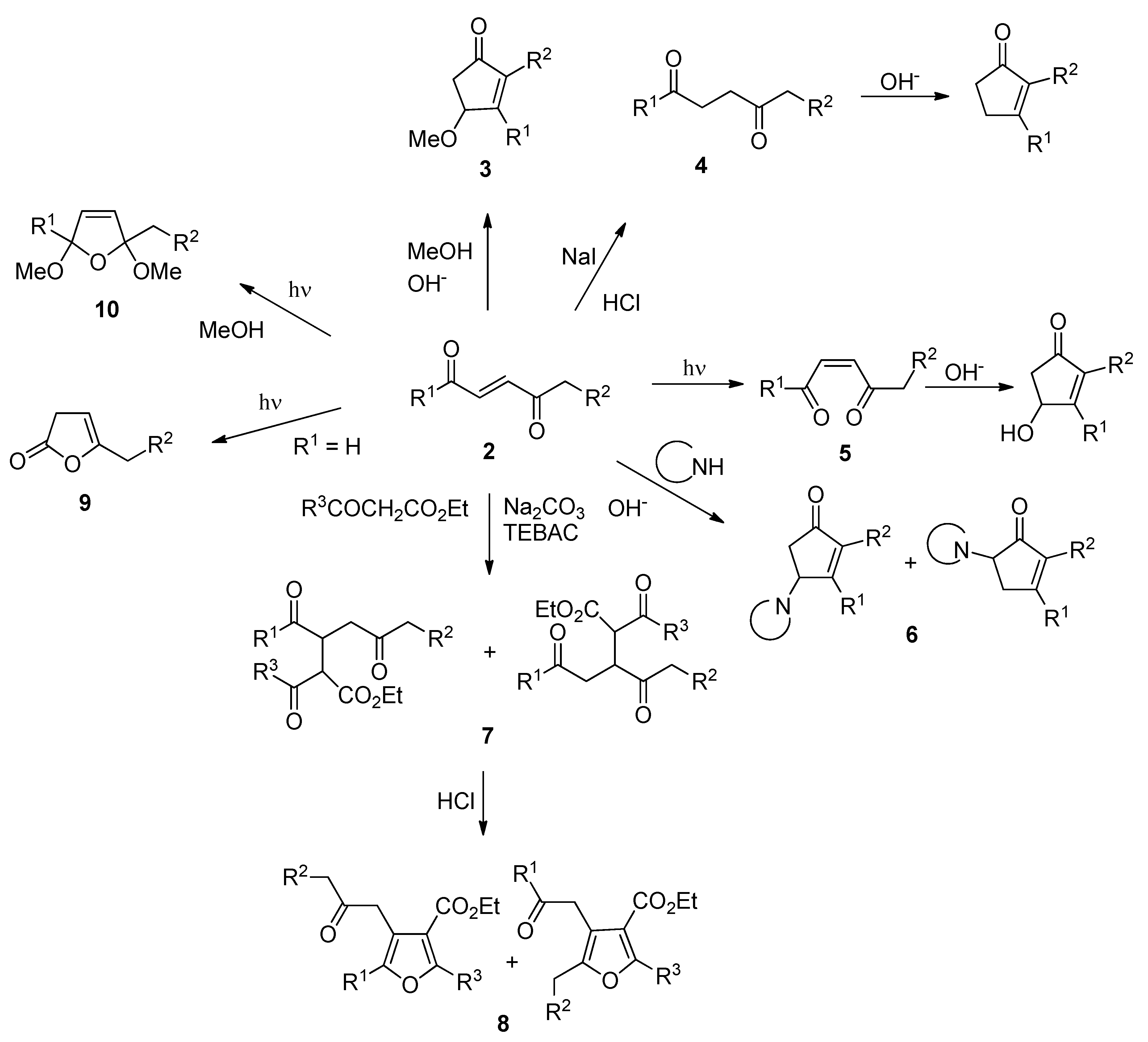
2.1. Pyridinium Chlorochromate
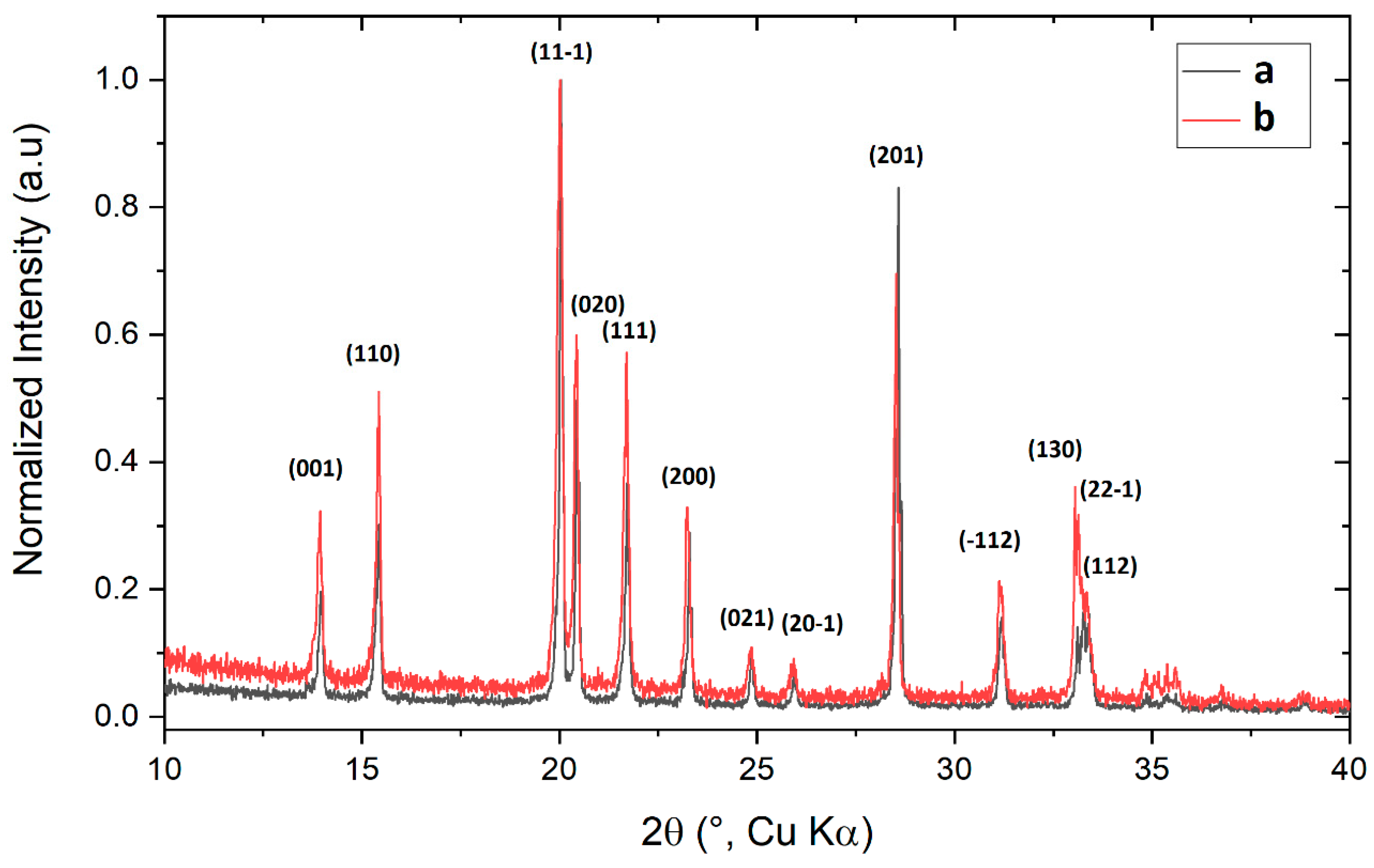
2.2. XRD Spectra
2.3. XPS Spectra
2.4. Curve-fitting procedure
2.5. DFT Calculation
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Piancatelli, G. : Scettri, A.; D'Auria M. Oxidative Ring Opening of Furan Derivatives to α,β-Unsaturated γ-Dicarbonyl Compounds, Useful Intermediates for 3-Oxocyclopentenes Synthesis. Tetrahedron 1980, 36, 661–663. [Google Scholar] [CrossRef]
- Piancatelli, G.; Scettri, A.; D'Auria, M. Pyridinium Chlorochromate: a Versatile Oxidant in Organic Synthesis. Synthesis 1982, 245.-258. [CrossRef]
- Piancatelli, G.; D'Auria, M.; D'Onofrio, F. Synthesis of 1,4-Dicarbonyl Compounds and Cyclopentenones from Furans. Synthesis 1994, 867–889. [Google Scholar] [CrossRef]
- D'Auria, M.; Piancatelli, G.; Scettri, A. A Mild and Selective Reduction of Enedicarbonyl Compounds. Synthesis 1980, 245–247. [Google Scholar] [CrossRef]
- Piancatelli, G.; D’Auria, M.; Scettri, A. Process for the synthesis of 3-keto-cyclopentene-5-oxy derivatives having insecticide activity, US Pat. 441 3145, 1981. [Google Scholar]
- D'Ascoli, R.; D'Auria, M.; De Mico, A.; Piancatelli, G.; Scettri, A. A Rapid and Efficient Route to 4- and 5-Amino-3-oxocyclopentene Derivatives. J. Org. Chem. 1980, 45, 4500–4502. [Google Scholar] [CrossRef]
- Antonioletti, R.; D'Auria, M.; Piancatelli, G.; Scettri, A. Michael Addition to trans-Enedicarbonyl Compounds: a Facile Route to 2,3,4,5-Tetra-substituted Furans. J. Chem. Soc. Perkin Trans. I 1981, 2398–2400. [Google Scholar] [CrossRef]
- D'Auria, M.; De Mico, A.; Piancatelli, G.; Scettri, A. A Facile Route to 5-Alkyl-2(3H)-furanones by Photoisomerisation of Enedicarbonyl Compounds. Tetrahedron 1982, 38, 1661–1666. [Google Scholar] [CrossRef]
- Antonioletti, R.; D'Auria, M.; Piancatelli, G.; Scettri, A. Photochemical Synthesis of 2,5-Dialkoxy- and 2,5-Diacetoxy-dihydrofurans from trans-Enedicarbonyl Compounds. Tetrahedron Lett. 1982, 2981–2984. [Google Scholar] [CrossRef]
- Corey, E. J.; Suggs, J. W. Pyridinium chlorochromate. An efficient reagent for oxidation of primary and secondary alcohols to carbonyl compounds. Tetrahedron Lett. 1975, 2647–2650. [Google Scholar] [CrossRef]
- Piancatelli, G.; Scettri, A.; D'Auria, M. The Oxidation of Furan Derivatives with Pyridinium Chlorochromate: a Novel Synthesis of 6-Hydroxy-2H-pyran-3(6H)-ones. Tetrahedron Lett. 1977, 2199–2200. [Google Scholar] [CrossRef]
- Piancatelli, G.; Scettri, A.; D'Auria, M. Pyridinium Chlorochromate in the Organic Synthesis: a Convenient Oxidation of Enol-ethers to Esters and Lactones. Tetrahedron Lett. 1977, 3483–3484. [Google Scholar] [CrossRef]
- Piancatelli, G.; Scettri, A.; D'Auria, M. Pyridinium Chlorochromate in the Organic Synthesis: a Convenient Oxidation of 5-Bromo-2-furan-derivatives into γ-Hydroxy-butenolides. Tetrahedron Lett. 1979, 1507–1508. [Google Scholar] [CrossRef]
- D'Auria, M.; D'Onofrio, F.; Piancatelli, G.; Scettri, A. Studies on Reactivity of Pyridinium Chlorochromate-Iodine System: an Efficient Method for Converting Enolsilyl Ethers into α-Iodo Ketones. Synth. Commun. 1982, 1127–1138. [Google Scholar] [CrossRef]
- Antonioletti, R.; D'Auria, M.; De Mico, A.; Piancatelli, G.; Scettri, A. The Oxidative C-C Cleavage of Phenyloxiranes by Pyridinium Chlorochromate. Synthesis 1983, 890–891. [Google Scholar] [CrossRef]
- Antonioletti, R.; D'Auria, M.; De Mico, A.; Piancatelli, G.; Scettri, A. Pyridinium Chlorochromate in Organic Synthesis. A Convenient Preparation of 4-Oxo-2-alkenethioic S-Esters. Synthesis 1984, 280-281. [CrossRef]
- Bonadies, F.; Di Fabio, R.; Bonini, C. Use of pyridinium chlorochromate as methylene oxidant in 5,6-dihydropyrans: a practical one-step preparation of the anhydromevalonolactone. J. Org. Chem. 1984, 49, 1647–1649. [Google Scholar] [CrossRef]
- Bonadies, F.; Bonini, C. Oxidation of Active Methylene Compounds by Pyridinium Chlorochromate. Synth. Commun. 1988, 18, 1573–1580. [Google Scholar] [CrossRef]
- Castle, J. E.; Chapman-Kpodo, H.; Proctor, A.; Salvi A., M. Curve-fitting in XPS using extrinsic and intrinsic background structure. J. Electron Spectrosc. Relat. Phenom. 1999, 106, 65–80. [Google Scholar] [CrossRef]
- Castle, J. E.; Salvi, A. M. Chemical state information from the near-peak region of the X-ray photoelectron background. J. Electron Spectrosc. Relat. Phenom. 2001, 114–116, 1103–1113. [CrossRef]
- Gaussian 09, Revision A.1, M. J. Frisch, G. W. Trucks, H. B. Schlegel, G. E. Scuseria, M. A. Robb, J. R. Cheeseman, G. Scalmani, V. Barone, B. Mennucci, G. A. Petersson, H. Nakatsuji, M. Caricato, X. Li, H. P. Hratchian, A. F. Izmaylov, J. Bloino, G. Zheng, J. L. Sonnenberg, M. Hada, M. Ehara, K. Toyota, R. Fukuda, J. Hasegawa, M. Ishida, T. Nakajima, Y. Honda, O. Kitao, H. Nakai, T. Vreven, J. A. Montgomery, Jr., J. E. Peralta, F. Ogliaro, M. Bearpark, J. J. Heyd, E. Brothers, K. N. Kudin, V. N. Staroverov, R. Kobayashi, J. Normand, K. Raghavachari, A. Rendell, J. C. Burant, S. S. Iyengar, J. Tomasi, M. Cossi, N. Rega, J. M. Millam, M. Klene, J. E. Knox, J. B. Cross, V. Bakken, C. Adamo, J. Jaramillo, R. Gomperts, R. E. Stratmann, O. Yazyev, A. J. Austin, R. Cammi, C. Pomelli, J. W. Ochterski, R. L. Martin, K. Morokuma, V. G. Zakrzewski, G. A. Voth, P. Salvador, J. J. Dannenberg, S. Dapprich, A. D. Daniels, O. Farkas, J. B. Foresman, J. V. Ortiz, J. Cioslowski, and D. J. Fox, Gaussian, Inc., Wallingford CT, 2009.
- Parr, R. G.; Yang, W. Density Functional Theory of Atoms and Molecules, Oxford University Press: Oxford, UK, 1989.
- Becke, A. D. Molecular excitation energies to high-lying bound states from time-dependent density-functional response theory: Characterization and correction of the time-dependent local density approximation ionization threshold. J. Chem. Phys. 1993, 98, 5648–5652. [Google Scholar] [CrossRef]
- Małuszyńka, H.; Czarnecki, P.; Czarnecka, A.; Pajak, Z. Structures and phase transitions in a new ferro-electric – pyridinium chlorochromate – studied by X-ray diffraction, DSC and dielectric methods. Acta Cryst. B 2012, B68, 128–136. [Google Scholar] [CrossRef]
- Suvitha, A.; Sathyanarayanamoorthi, V.; Murugakoothan, P. Growth, spectroscopy properties and DFT based PCM calculations of guanidinium chlorochromate. Spectrochim. Acta A 2013, 110, 255–261. [Google Scholar] [CrossRef]
- Desimoni, E.; Malitesta, C.; Zambonin, G.; Rivière, J. C. An x-ray photoelectron spectroscopic study of some chromium–oxygen systems. Surf. Interface Anal. 1988, 13, 173–179. [Google Scholar] [CrossRef]
- Salvi, A. M.; Castle, J. E.; Watts, J. F.; Desimoni, E. Peak fitting of the chromium 2p XPS spectrum. Appl. Surface Sci. 1995, 90, 333–341. [Google Scholar] [CrossRef]
- Aronniemi, M.; Sainio, J.; Lahtinen, J. Chemical state quantification of iron and chromium oxides using XPS: the effect of the background subtraction method. Surface Sci. 2005, 578, 108–123. [Google Scholar] [CrossRef]
- Wessjohann, L. A.; Scheid, G. Recent advances in Chromium(II)- and Chromium(III)-madiated organic synthesis. Synthesis 1999, 1–36. [Google Scholar] [CrossRef]
- Bousquet, C.; Gilheany, D. G. Chromium catalysed asymmetric alkene epoxidation. greater selectivity for an E-alkene versus its Z-isomer. Tetrahedron Lett. 1995, 36, 7739–7742. [Google Scholar] [CrossRef]
- Imanishi, H.; Katsuki, T. Unusual solvent-effect in stereochemistry of asymmetric epoxidation using a (salen)chromium(III) complex as a catalyst. Tetrahedron Lett. 1997, 38, 251–254. [Google Scholar] [CrossRef]
- Chatterjee, D.; Basak, S.; Muzart, J. Asymmetric epoxidation of alkenes with aqueous t-BuOOH catalyzed by novel chiral complexes of chromium(III) containing tridentate Schiff-base ligands. J. Mol. Catal. A: Chem. 2007, 271, 270–276. [Google Scholar] [CrossRef]
- Ikeda, H.; Nishi, K.; Tsurugi, H.; Mashima, K. Chromium-catalyzed cyclopropanation of alkenes with bromoform in the presence of 2,3,5,6-tetramethyl-1,4-bis(trimethylsilyl)-1,4-dihydropyrazine. Chem. Sci. 2020, 11, 3604–3609. [Google Scholar] [CrossRef]

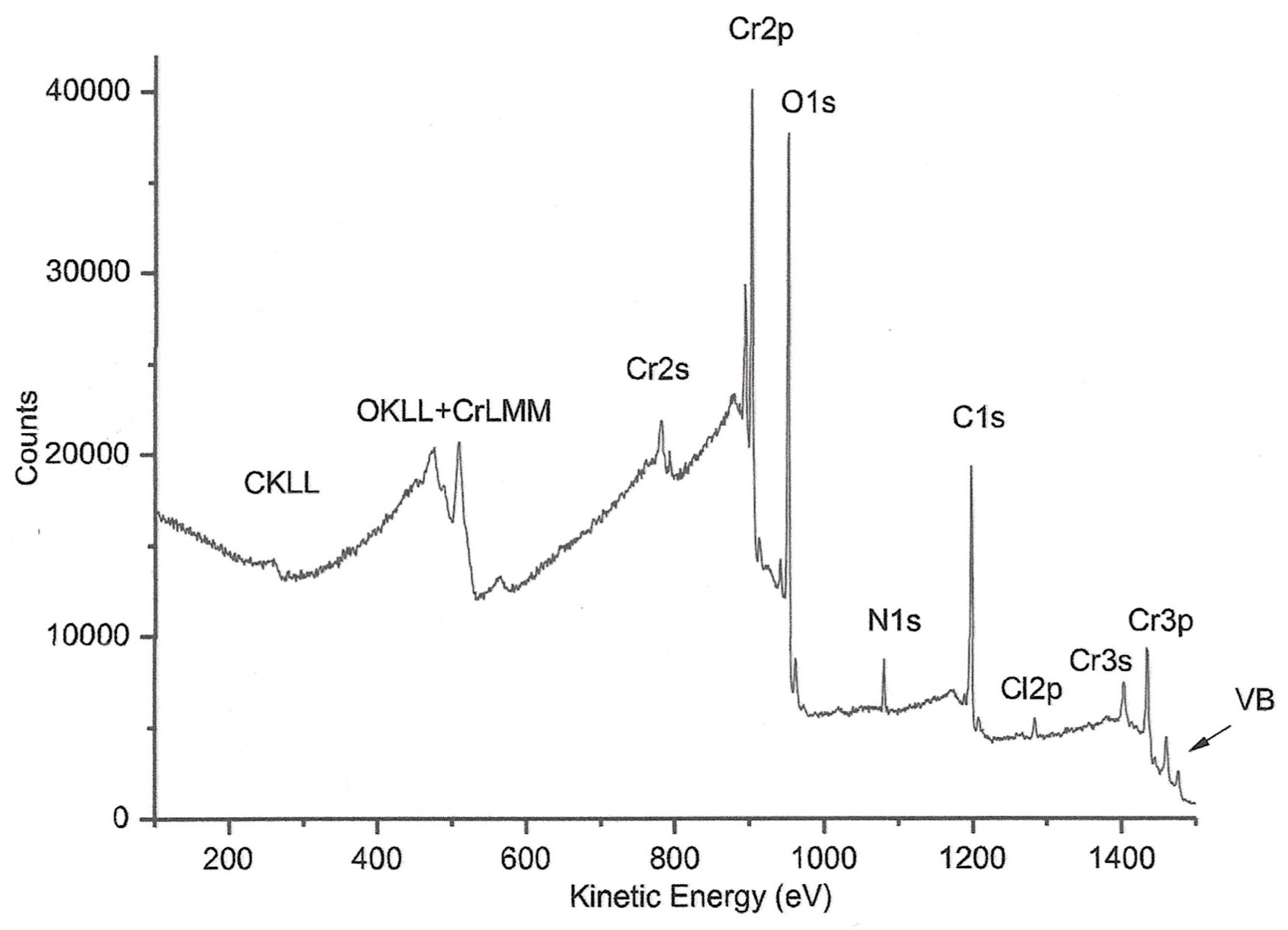
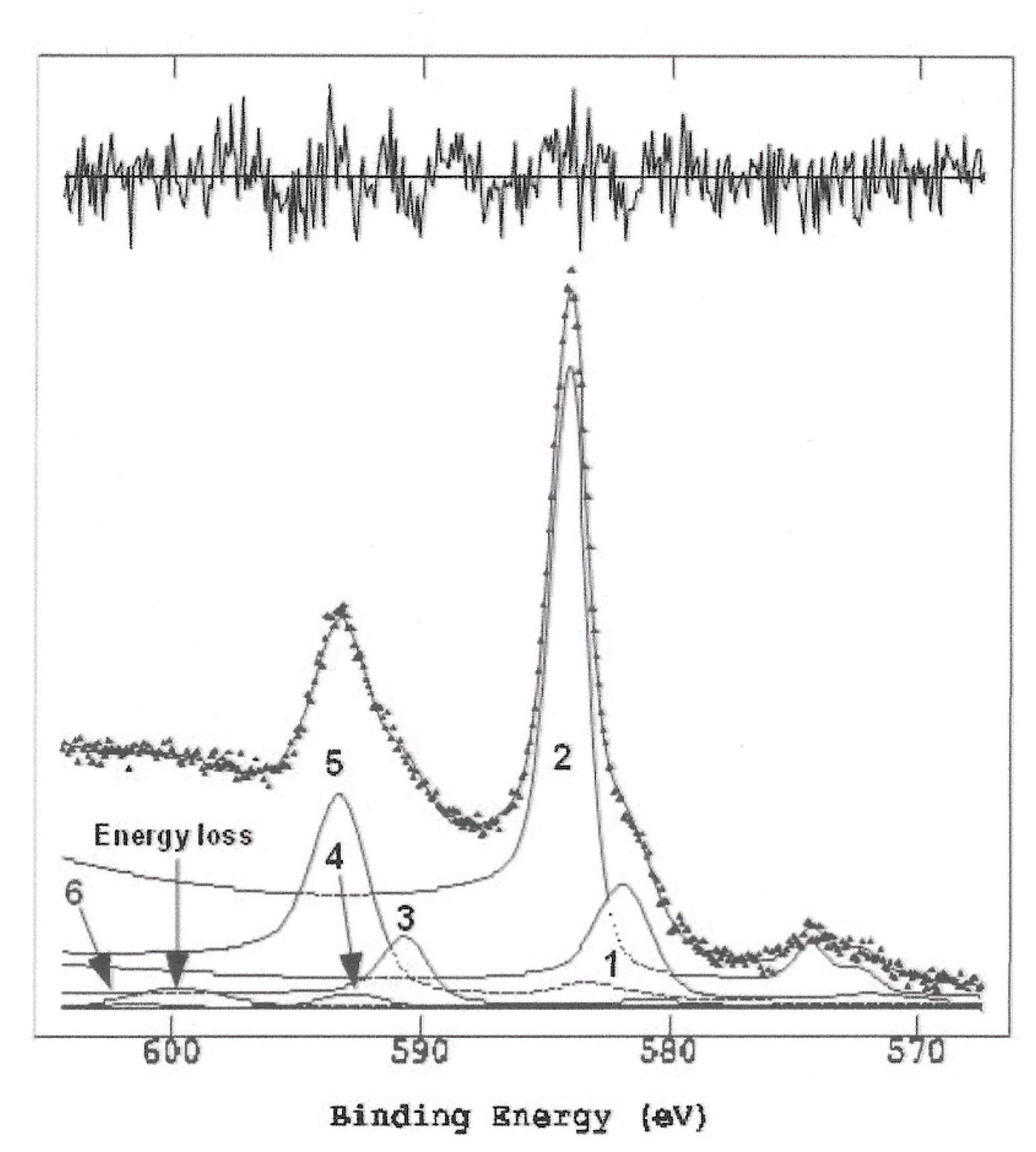
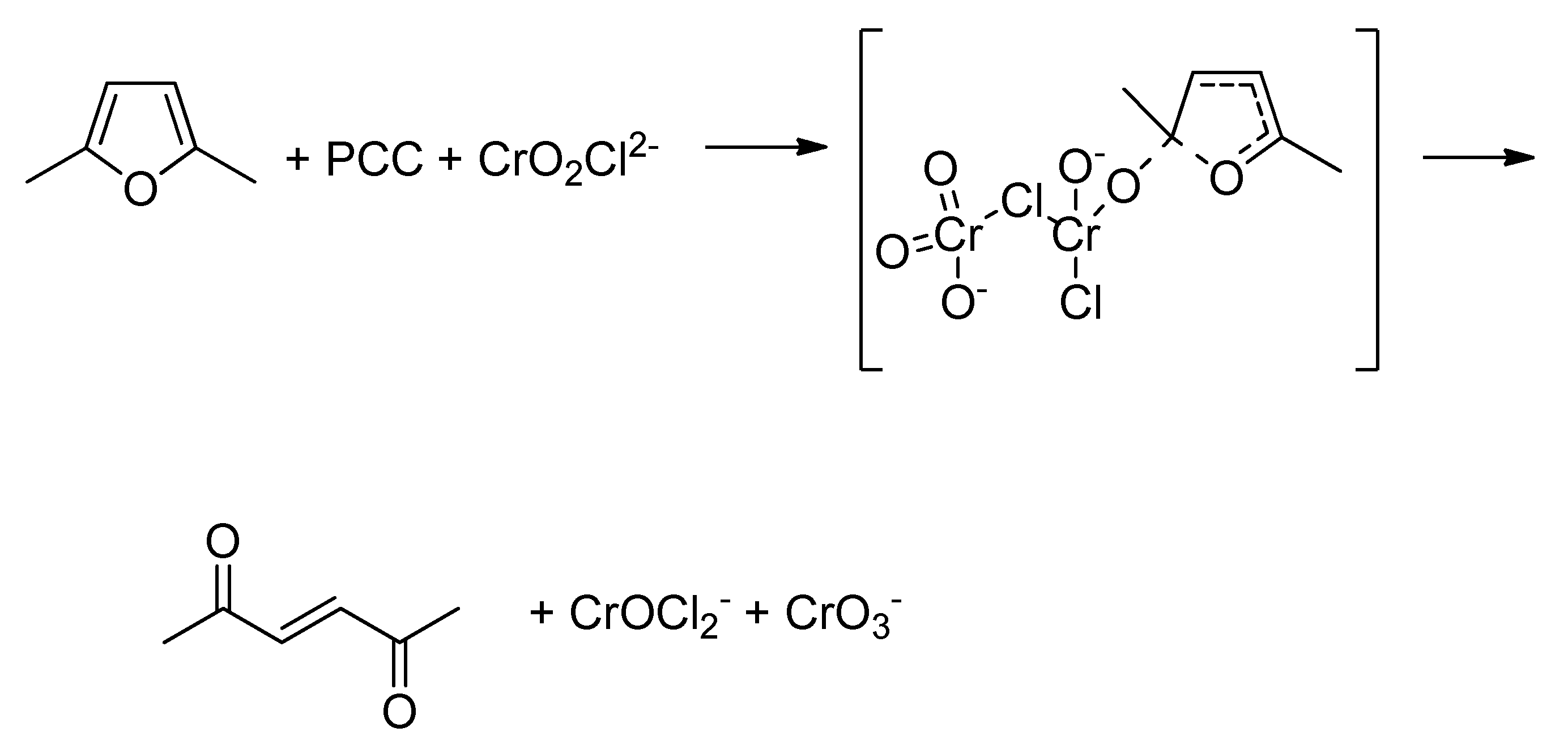
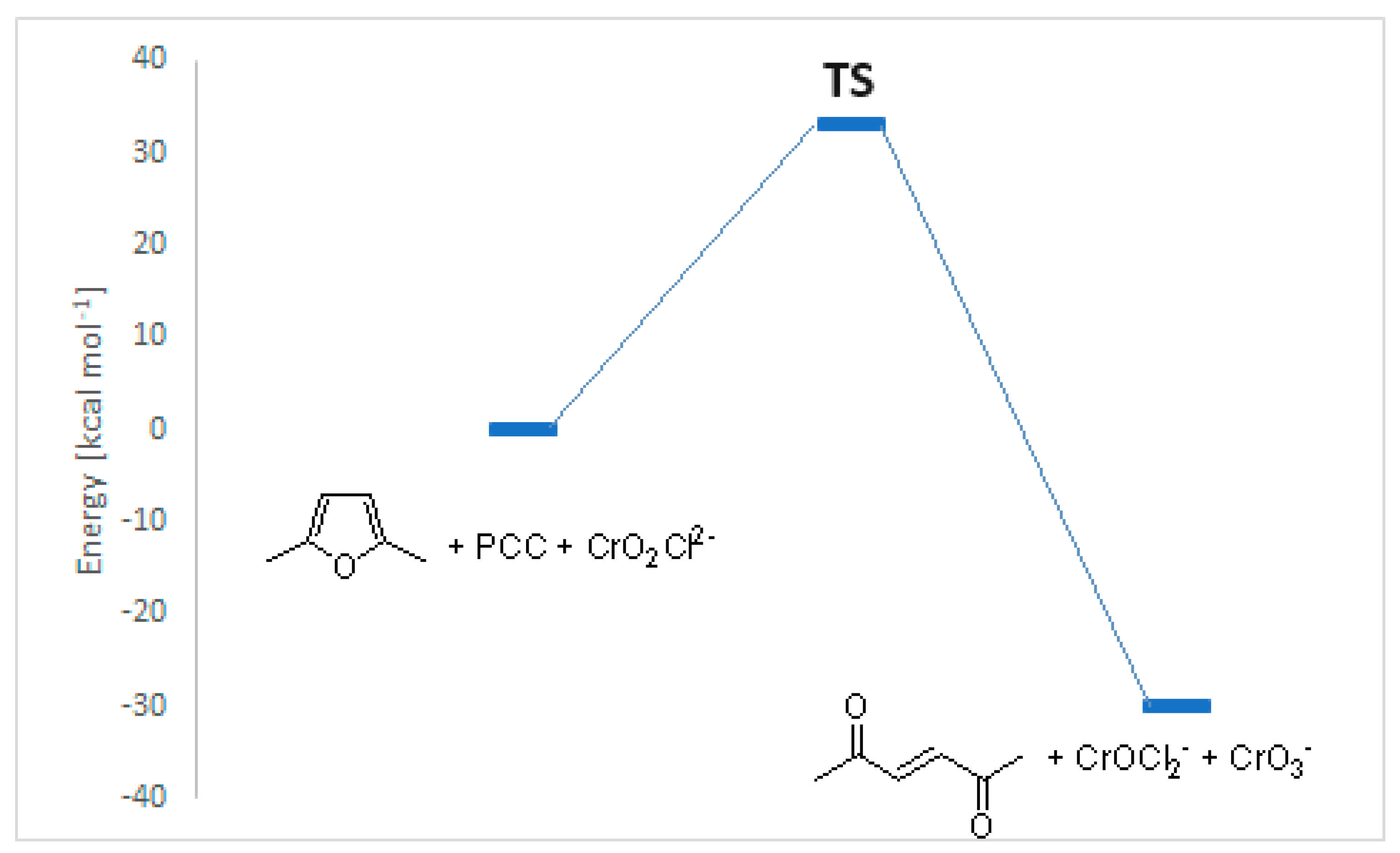
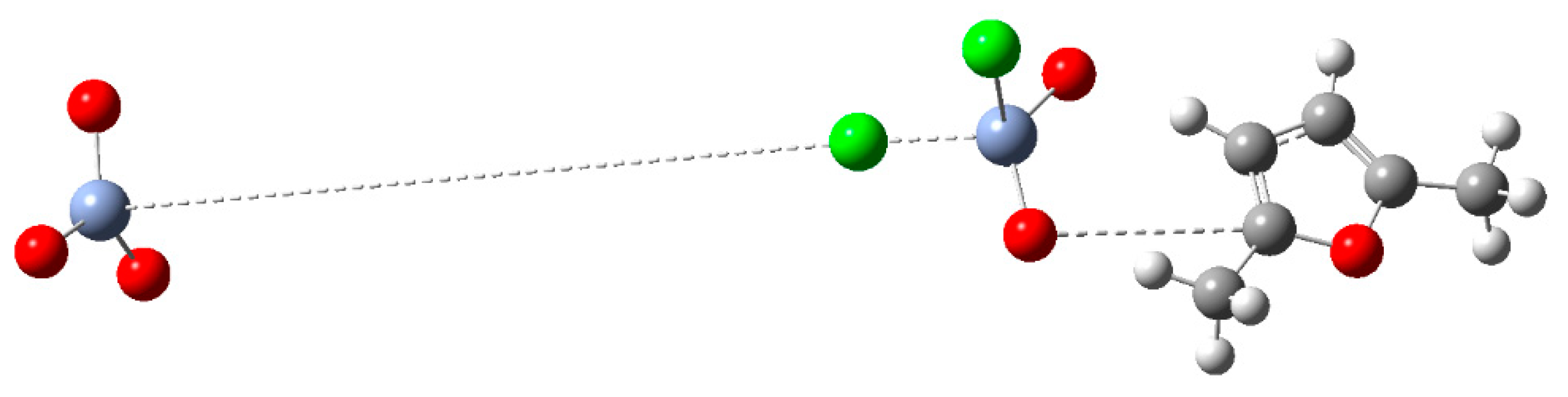
| Peak | Corrected BE (eV) | Normalized area | Assignement |
|---|---|---|---|
| 1 Cr2p3/2 | 577.3 | 641.6 | Cr(III) |
| 2 Cr2p3/2 | 579.6 | 2454.5 | Cr(VI) |
| 3 Cr2p!/2 | 586.1 | 320.8 | Cr(III) |
| 4 Satellite peak 1 (Cr2p3/2) | 588.3 | 89.8 | Sat 1 |
| 5 Cr2p1/2 | 588.7 | 1164.8 | Cr(VI) |
| 6 Satellite peak 3 (Cr2p1/2) | 597.1 | 44.9 | Sat 2 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




