Submitted:
04 October 2023
Posted:
05 October 2023
You are already at the latest version
Abstract
Keywords:
1. Introduction
2. Materials and Methods
2.1. Waterproofing formulations
- a)
- Hydrophobic formulations
- 1.-
- Tea tree oil mouthwash: glycerin (15%), sorbitol (4.5%), lauryl sulfate sodium (3%), ethanol (10%) (Merck, Darmstadt, Germany) and tea tree oil (1.5%) (Acofarma, Terrassa, Spain) in water.
- 2.-
- Semisolid anhydrous absorption base: lecithin (50%) in liquid Vaseline (50%).
- 3.-
- Lipophilic base MI: isopropyl myristate (10%) in Filant Vaseline.
- 4.-
- Lipophilic Base TGCM: Propylene glycol (10%) and medium chain triglycerides (10%) in Filant Vaseline.
- 5.-
- Fluid anhydrous absorption base: soy lecithin (50%) and isopropyl palmitate (50%).
- b)
- Hydrophilic formulations
- 6.-
- Sodium carboxymethyl cellulose gel 4%: sodium carboxymethylcellulose (4%) and glycerin (10%) in water
- 7.-
- Sodium hyaluronate gel 2%: sodium hyaluronate (2%) in water
- 8.-
- Chitosan gel 2%: Chitosan (2%) was dispersed in a lactic acid solution (1%) in water.
- 9.-
- Alginate gel 4%: Alginate sodium (4%) was dispersed in water, and CaCl2 solution (4%) was added.
- 10.-
- PLX-CBP Gel: Poloxamer in water (26%) was added to Carbopol 940 to reach a final concentration of 1%.
- c)
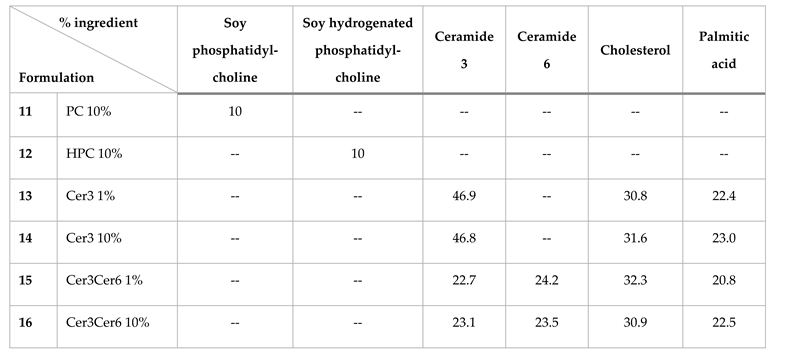
- Liposomal formulations
2.2. Water permeability study by determining transmucosal/transmembranal water loss (TMWL)
2.3. In vitro release of drugs and biocides
2.4. HPLC/DAD analytical measurements
2.5. Statistical analysis
3. Results and Discussion
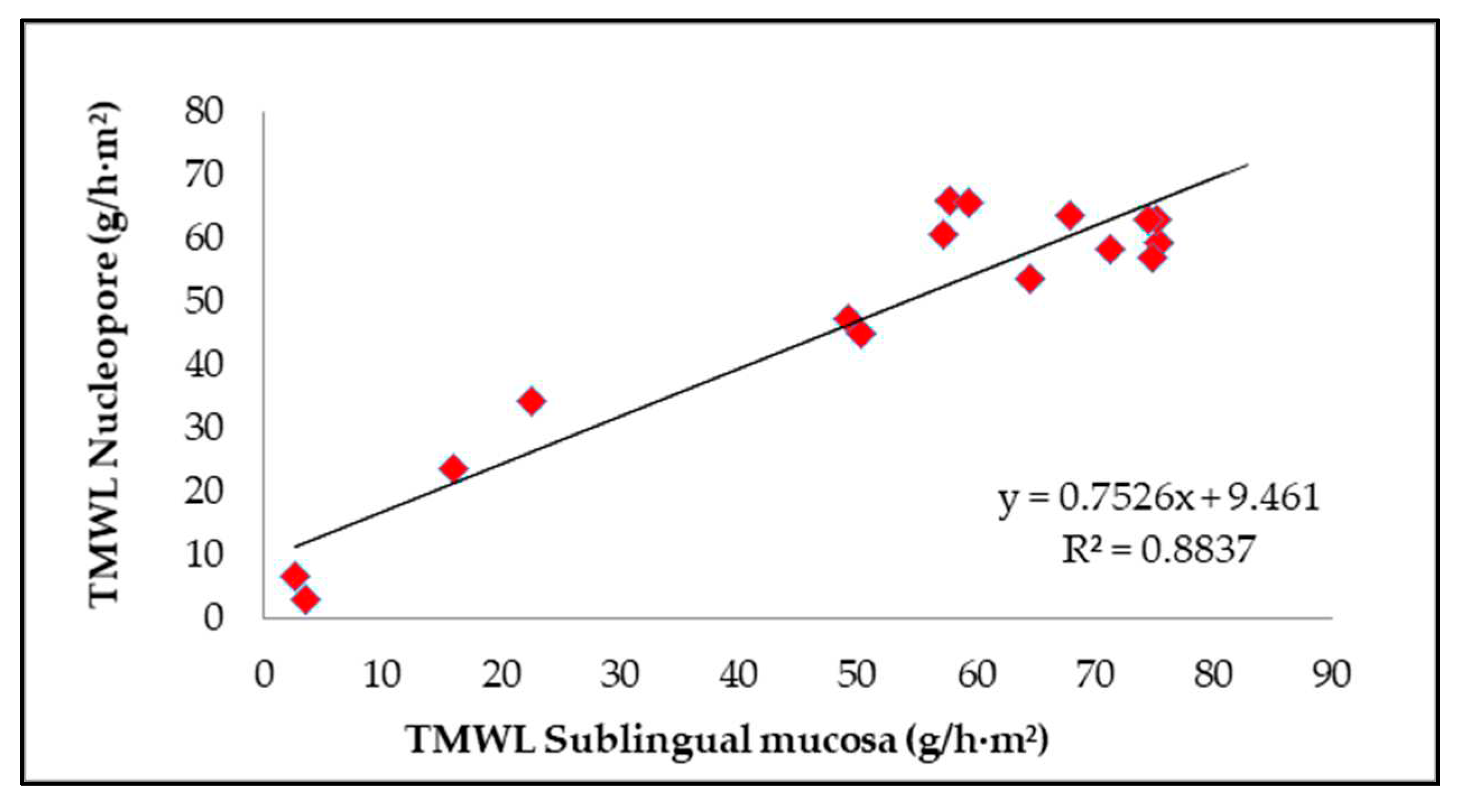
3.1. Water permeability of intact membranes and those with protective waterproofing formulations
3.2. In vitro release test of drugs and biocides and their permeation parameters
| Compounds | Log Ko/w (pH 7.4) | Molecular Weight (MW) | Skin Kp(10-3cm/h) |
Sublingual mucosa Kp(10-3cm/h) |
Nuclepore Kp(10-3cm/h) |
|---|---|---|---|---|---|
| Caffeine (CAF) | -0.1 | 194.2 | 5.1 ± 4.1 | 39.2 ± 6.6 | 54.7 ± 11.2 |
| Ketorolac Tromethamine (KET) | 2.3 | 376.4 | 2.2 ± 0.3 | 59.4 ± 0.6 | 67.5 ± 12.4 |
| Dexametasone (DEX) | 1.7 | 392.5 | 0.5 ± 0.3 | 25.7 ± 7.9 | 44.3 ± 7.0 |
| Ivermectine (IVE) | 5.8 | 875.1 | 0.3 ± 0.1 | 5.7 ± 3.1 | 41.1 ± 11.2 |
| Fungitrol (FUN) | 2.4 | 281.1 | 0.7 ± 0.4 | 4.0 ± 3.3 | 44.4 ± 11.9 |
| Propiconazole (PRO) | 3.7 | 342.2 | 0.2 ± 0.2 | 4.5 ± 3.6 | 40.8 ± 11.6 |
| Permethrin (PER) | 6.5 | 391.3 | 0.2 ± 0.1 | 2.7 ± 2.5 | 36.8 ± 17.0 |
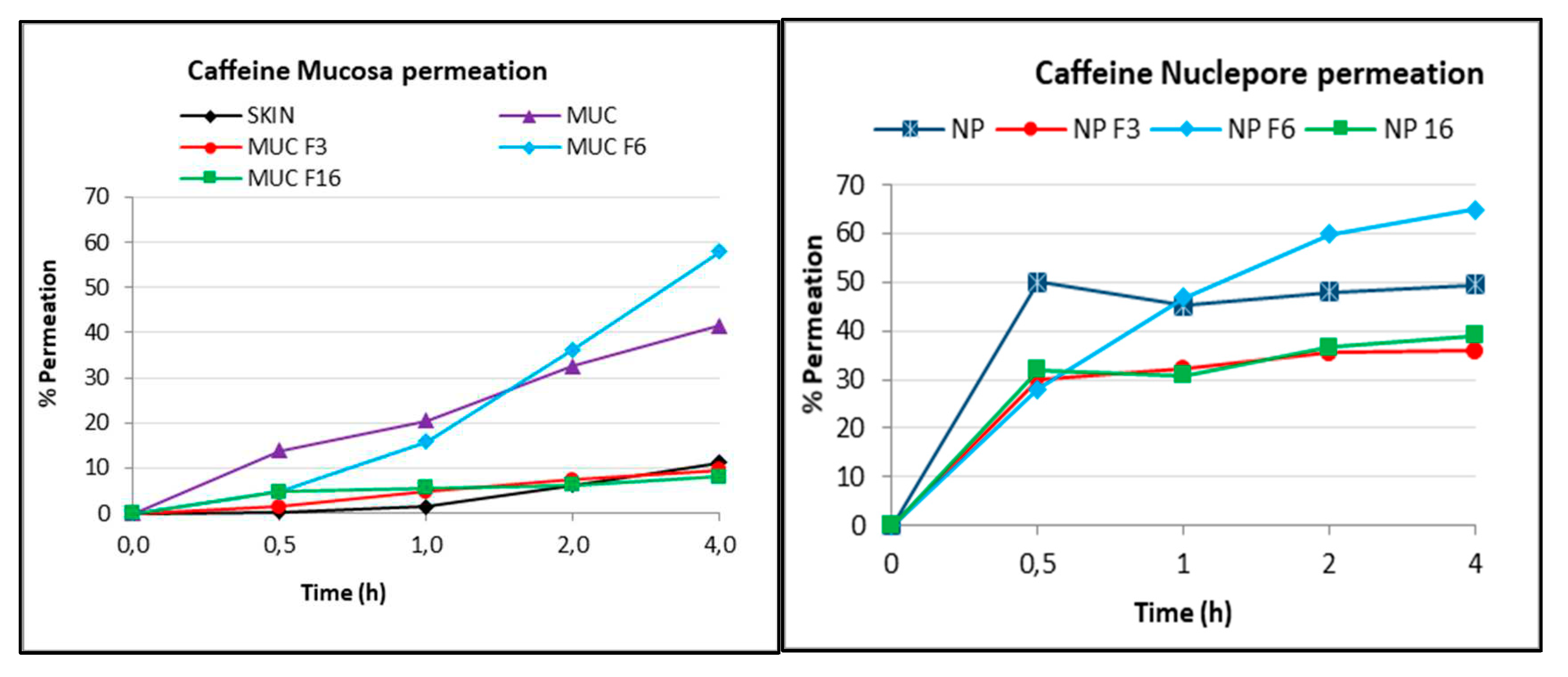
3.3. In vitro release test and permeation parameters of caffeine through the modified membranes
4. Conclusions
6. Patents
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Wertz, P.W. Roles of Lipids in the Permeability Barriers of Skin and Oral Mucosa. Int. J. Mol. Sci. 2021, 22, 5229. [Google Scholar] [CrossRef] [PubMed]
- White, S.H.; Mirejovsky, D.; King, G.I. Structure of lamellar lipid domains and corneocyte envelopes of murine stratum corneum. An x-ray diffraction study. Biochemistry 1988, 27, 3725–3732. [Google Scholar] [CrossRef]
- Bouwstra, J.; Pilgram, G.; Gooris, G.; Koerten, H.; Ponec, M. New Aspects of the Skin Barrier Organization. Ski. Pharmacol. Physiol. 2001, 14, 52–62. [Google Scholar] [CrossRef] [PubMed]
- Janůšová, B.; Zbytovská, J.; Lorenc, P.; Vavrysová, H.; Palát, K.; Hrabálek, A.; Vávrová, K. Effect of ceramide acyl chain length on skin permeability and thermotropic phase behavior of model stratum corneum lipid membranes, Biochim. Biophys. Acta - Mol. Cell Biol. Lipids. 2011, 1811, 129–137. [Google Scholar] [CrossRef]
- Kessner, D. ; Ruettinger, A.; Kiselev, M.A.; Wartewig, S.; Neubert,, R.H.H. Properties of ceramides and their impact on the stratum corneum structure: A review - Part 2: Stratum corneum lipid model systems, Skin Pharmacol. Physiol. 2008, 21, 58–74.
- Kinikoglu, B.; Damour, O.; Hasirci, V. Tissue engineering of oral mucosa: a shared concept with skin. J. Artif. Organs 2014, 18, 8–19. [Google Scholar] [CrossRef]
- Shojael, A. H. Buccal mucosa as a route for systemic drug delivery: A Review. J. Pharm. Pharmaceut Sci, 1988, 1, 15–30 1988. [Google Scholar]
- A Winning, T.; Townsend, G.C. Oral mucosal embryology and histology. Clin. Dermatol. 2000, 18, 499–511. [Google Scholar] [CrossRef] [PubMed]
- Wertz, P.W.; A Squier, C. Cellular and molecular basis of barrier function in oral epithelium. Crit. Rev. Therap. Drug Carr. Syst. 1991, 8, 237–69. [Google Scholar]
- Kontogiannidou, E.; Andreadis, D.A.; Zografos, A.L.; Nazar, H.; Klepetsanis, P.; van der Merwe, S.M.; Fatouros, D.G. Ex vivo buccal drug delivery of ropinirole hydrochloride in the presence of permeation enhancers: the effect of charge. Pharm. Dev. Technol. 2016, 22, 1017–1021. [Google Scholar] [CrossRef]
- Potts, R.O.; Guy, R.H. Predicting Skin Permeability. Pharm. Res. 1992, 9, 663–669. [Google Scholar] [CrossRef]
- Schmook, F.P.; Meingassner, J.G.; Billich, A. Comparison of human skin or epidermis models with human and animal skin in in-vitro percutaneous absorption. Int. J. Pharm. 2001, 215, 51–56. [Google Scholar] [CrossRef]
- Abd, E.; Yousuf, S.; Pastore, M.N.; Telaprolu, K.; Mohammed, Y.H.; Namjoshi, S.; Grice, J.; Roberts, M.S. Skin models for the testing of transdermal drugs. Clin. Pharmacol. Adv. Appl. 2016, ume 8, 163–176. [Google Scholar] [CrossRef]
- Neupane, R.; Boddu, S.H.; Renukuntla, J.; Babu, R.J.; Tiwari, A.K. Alternatives to Biological Skin in Permeation Studies: Current Trends and Possibilities. Pharmaceutics 2020, 12, 152. [Google Scholar] [CrossRef]
- Najib, O.N.; Martin, G.P.; Kirton, Botha, M.J.; Sallam, A.S.; Murnane, D. The Influence of Oily Composition and Vehicle-Membrane Interactions on the Diffusion of Model Permeants across barrier Membranes. Membranes. 2021, 11, 57.
- Madison, K.C.; Swartzendruber, D.C.; Wertz, P.W.; Downing, D.T. Presence of Intact Intercellular Lipid Lamellae in the Upper Layers of the Stratum Corneum. J. Investig. Dermatol. 1987, 88, 714–718. [Google Scholar] [CrossRef] [PubMed]
- Alonso, C.; Collini, I.; Martí, M.; Barba, C.; Coderch, L. Lanolin-Based Synthetic Membranes for Transdermal Permeation and Penetration Drug Delivery Assays. Membranes 2021, 11, 444. [Google Scholar] [CrossRef]
- Alonso, C.; Collini, I.; Carrer, V.; Barba, C.; Martí, M.; Coderch, L. Permeation kinetics of active drugs through lanolin-based artificial membranes. Colloids Surfaces B: Biointerfaces 2020, 192, 111024. [Google Scholar] [CrossRef]
- Sosnik, A. Tissue-based in vitro and ex vivo models for nasal permeability studies. In Concepts and models for drug permeability studies. Woodhead Publishing, 2016. pp. 237-254.
- Moharamzadeh, K.; Brook, I.; Van Noort, R.; Scutt, A.; Thornhill, M. Tissue-engineered Oral Mucosa: a Review of the Scientific Literature. J. Dent. Res. 2007, 86, 115–124. [Google Scholar] [CrossRef] [PubMed]
- Squier, C.A.; Kremer, M.; Wertz, P.W. Continuous Flow Mucosal Cells for Measuring the in-Vitro Permeability of Small Tissue Samples. J. Pharm. Sci. 1997, 86, 82–84. [Google Scholar] [CrossRef]
- Richter, T.; Keipert, S. In vitro permeation studies comparing bovine nasal mucosa, porcine cornea and artificial membrane: androstenedione in microemulsions and their components. Eur. J. Pharm. Biopharm. 2004, 58, 137–143. [Google Scholar] [CrossRef] [PubMed]
- Alonso, C.; Martí, M.; Coderch, L.; Calpena, A.C.; Mallandrich, M.; Pérez, L.; Clares, B.; Pérez, N. Lipophilic-based composition. N. Sol: EP23382737.7 (2023) N. Ref: ES1641.1822. CSIC, UB, UGR.
- Alonso, C.; Martí, M.; Coderch, L.; Calpena, A.C.; Pérez, L.; Clares, B. Liposomal-based composition N. de Sol: EP23382651.0 (2023) N. Ref: ES1641.1823. CSIC, UB, UGR.
- Thakker, K.D.; Chern, W.H. Development and Validation of In Vitro Release Tests for Semisolid Dosage Forms—Case Study. Dissolution Technol. 2003, 10, 10–15. [Google Scholar] [CrossRef]
- Mallandrich, M.; Fernández-Campos, F.; Clares, B.; Halbaut, L.; Alonso, C.; Coderch, L.; Garduño-Ramírez, M.L.; Andrade, B.; del Pozo, A.; Lane, M.E.; et al. Developing Transdermal Applications of Ketorolac Tromethamine Entrapped in Stimuli Sensitive Block Copolymer Hydrogels. Pharm. Res. 2017, 34, 1728–1740. [Google Scholar] [CrossRef] [PubMed]
- Ich, ICH Topic Q2 (R1) Validation of Analytical Procedures: Text and Methodology. Int. Conf. Harmon., 2005; 1994, (November 1996), 1-17.
- Babita, K.; Kumar, V.; Rana, V.; Jain, S.; Tiwary, A.K. Thermotropic and Spectroscopic Behavior of Skin: Relationship with Percutaneous Permeation Enhancement. Curr. Drug Deliv. 2006, 3, 95–113. [Google Scholar] [CrossRef] [PubMed]
- Lane, M. J. Skin penetration enhancers. Int. J. of Pharmaceutics 447 (2013) 12– 21.
| Ketorolac Tromethamine | Caffeine | Dexamethasone | Ivermectin | Fungitrol | Propiconazole | Permethrin | |
|---|---|---|---|---|---|---|---|
| Extractor solvent | PBS solution | Methanol LiChrosolv® | Methanol LiChrosolv® | Methanol LiChrosolv® |
Methanol LiChrosolv® | Methanol LiChrosolv® | Methanol LiChrosolv® |
| Column | Lichrosphere® 100RP-18, (250x4.6 mm, 5 µm) |
Lichrosphere® 100RP-18, (250x4.6 mm ,5 µm) |
Lichosphere® 100RP-18, (250x4.6 mm ,5 µm) |
Lichrosphere® 100RP-18, (250x4.6 mm, 5 µm) | Zorbax Eclipse XDB C18 (150x4.6 mm, 5 µm) | Zorbax Eclipse XDB C18 (150x4.6 mm, 5 µm) | Zorbax Eclipse XDB C18 (150x 4.6 mm, 5 µm) |
|
Wavelength (nm) |
314 | 280 | 240 | 240 | 200 | 210 | 210 |
| Injection volume (µL) | 20 | 40 | 40 | 40 | 20 | 20 | 20 |
| Movil phase | NaH2PO4 (0.75 g/L+0,5mL H3PO4 )/ CH3OH (340/660, v/v) |
Methanol/Water 60:40—90:10 (15’) —90:10(15’) —60:40 (10’) |
Methanol/Water 60:40—90:10 (15’) —90:10(15’) —60:40 (10’) |
Methanol/Water 60:40—90:10 (15’) —90:10(15’) —60:40 (10’) |
Acetonitrile/water 52/48 – 85/15 (10’) 85/15 (8’) 52/48 (5’) |
Acetonitrile/water 52/48 – 85/15 (10’) 85/15 (8’) 52/48 (5’) |
Acetonitrile/water52/48 – 85/15 (10’) 85/15 (8’)52/48 (5’) |
| Flux (mL/min) | 1 | 1 | 1 | 1 | 1 | 1 | 1 |
| Linear Reg. Eq. (R2) | A= 184224[KET]-70802 (0.9999) |
) |
A= |
|
A = 7.8218[FUN] + 0.6291 (0.9986) |
A = 41.302[PRO] – 0.7125 (0.9987) |
A = 34.733[PER] + 0.0206 (0.9986) |
| LoD / LoQ (µg/mL) | 0.10 / 0.28 | 0.82 / 2.49 | 0.23 / 0.70 | 0.55 /1.66 | 1.09 / 3.31 | 0.37 / 1.11 | 0.49 / 1.49 |
| Formulations | TMWL 1h Nuclepore (g/h·m2) |
TMWL 1h Sublingual Mucosa (g/h·m2) |
|---|---|---|
| Nuclepore control | 80,8 | -- |
| Sublingual mucosa control | -- | 72,4 |
| a) Hydrophobic formulations | ||
| 1 Tea tree mouthwash | 71,3 | 58,5 |
| 2 Semi-solid anhydrous absorption base | 15,98 | 23,6 |
| 3 Lipophilic base MI | 2,6 | 6,5 |
| 4 Lipophilic base TGCM | 3,6 | 3,0 |
| 5 Fluid anhydrous absorption base | 22,5 | 34,2 |
| b) Hydrophilic formulations | ||
| 6 Sodium carboxymethyl cellulose gel 4% | 64,6 | 53,8 |
| 7 Sodium hyaluronate gel 2% | 75,4 | 59,3 |
| 8 Chitosan gel 2% | 75,2 | 63,0 |
| 9 Alginate gel 4% | 74,5 | 62,9 |
| 10 Gel PLX-CBP | 74,8 | 57,0 |
| c) Liposomal formulations | ||
| 11 PC 10% | 57,8 | 66,1 |
| 12 HPC 10% | 68,0 | 63,7 |
| 13 Cer3 1% | 59,3 | 65,6 |
| 14 Cer3 10% | 57,2 | 60,8 |
| 15 Cer3Cer6 1% | 49,2 | 47,2 |
| 16 Cer3Cer6 10% | 50,4 | 45,1 |
| Parameter | SKIN | MUCOSA | MUCOSA F3 | MUCOSA F6 | MUCOSA F16 | NUCLEPORE | NUCLEPORE F3 | NUCLEPORE F6 | NUCLEPORE F16 |
|---|---|---|---|---|---|---|---|---|---|
| Permeability Coef.. Kp (10-3cm/h) | 5.1±4.1 | 39.2±6.6 | 8.3±2.5 | 49.3±0.1 | 6.4±0.9 | 54.7±11.2 | 36.5 | 57.1 | 32.1 |
| Maximal Conc. Cmax (µg/mL) | 107.2±57.7 | 514.5±120.0 | 118.3±34.8 | 719.7±93.0 | 99.3±8.5 | 507±136 | 364±247 | 673±55 | 407±131 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




