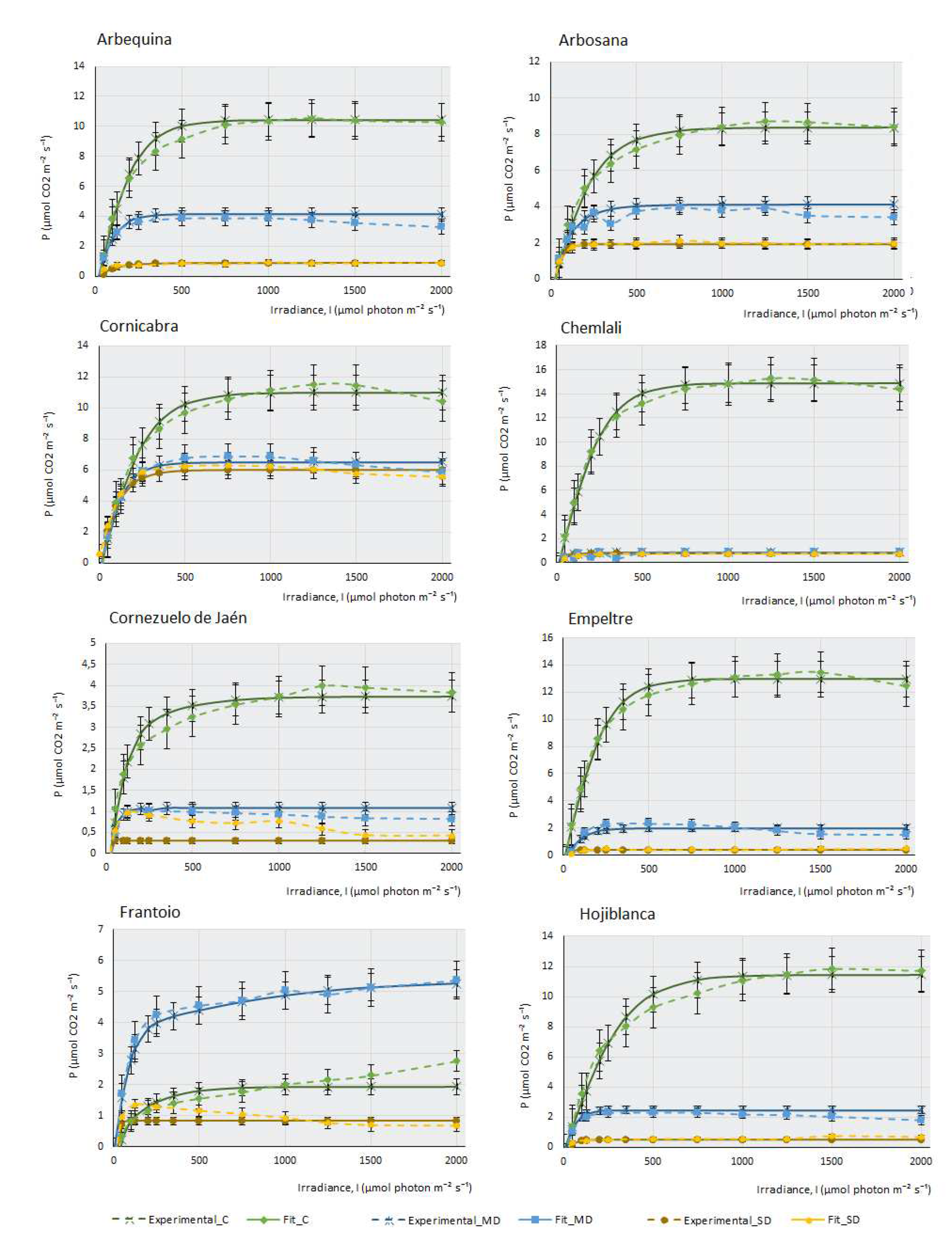
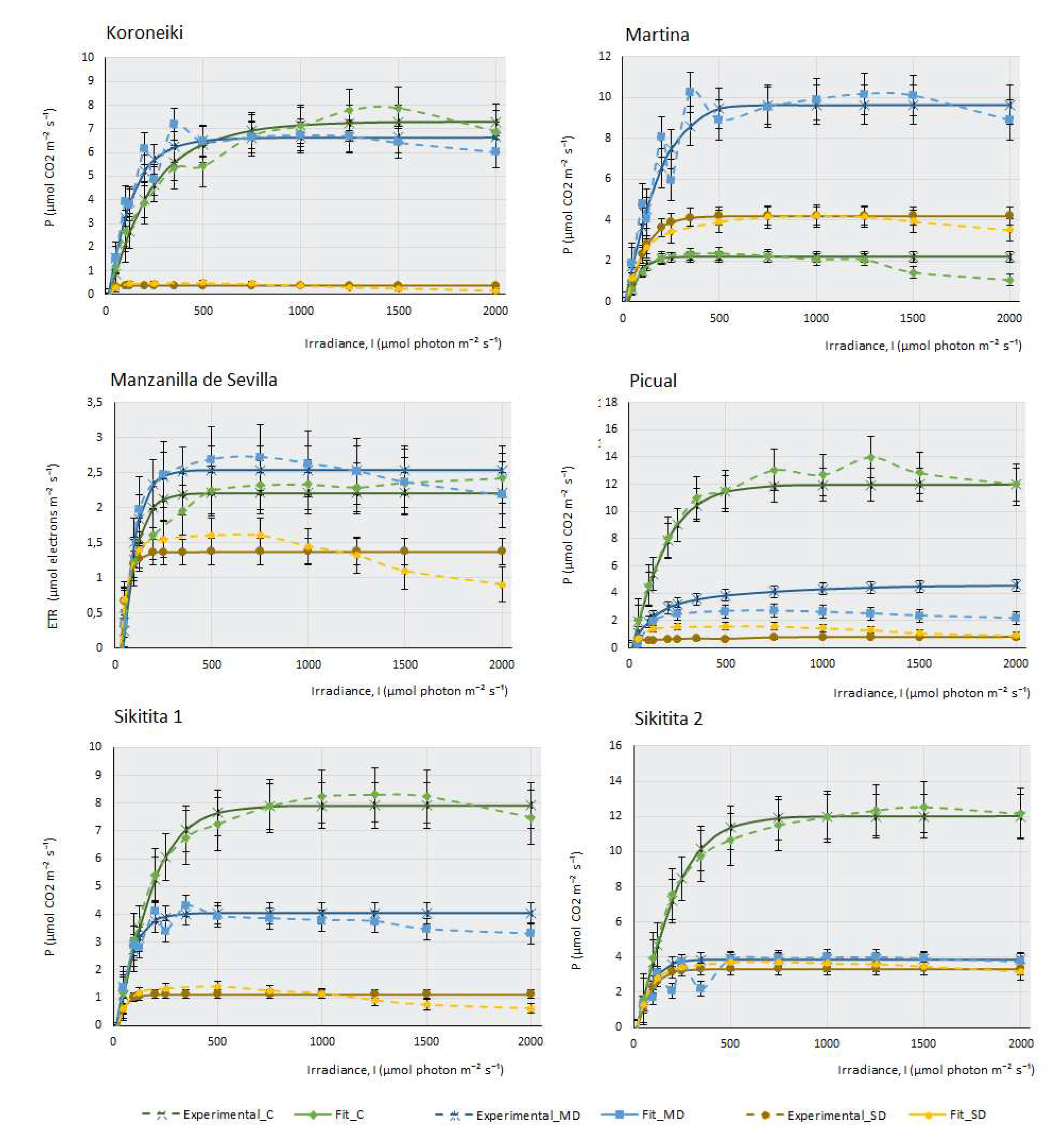
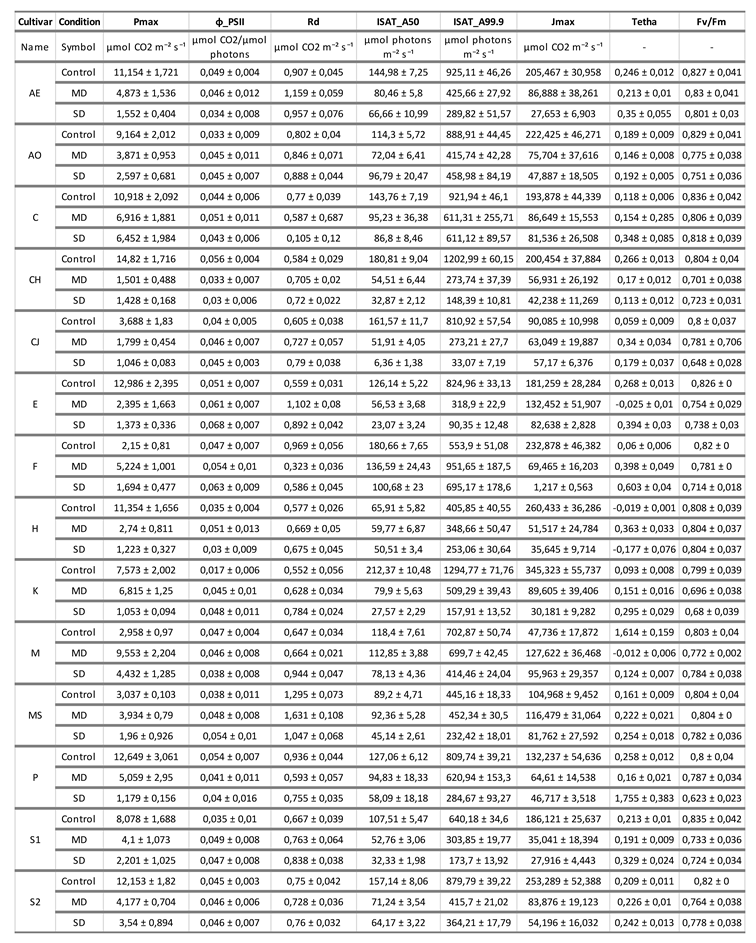
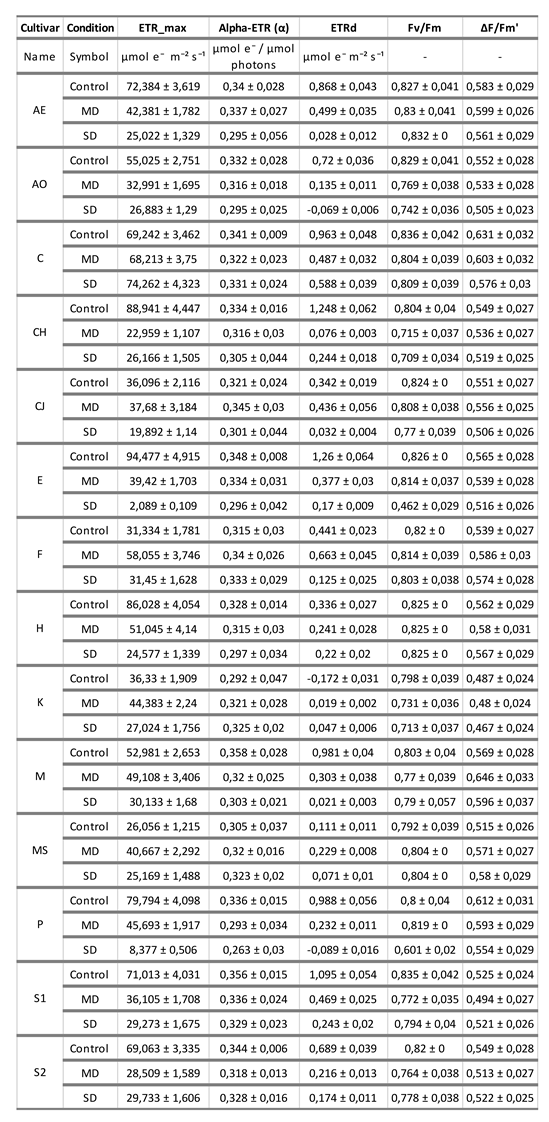
3. Photosynthesis-light response and ETR-light curves of the 14 olive cultivars
Figure 1 represents the three real curves (C, MD and SD) obtained for each cultivar and the theoretical curves that were mathematically adjusted to each of the real curves. In most cultivars, control samples present higher maximal photosynthesis (Pmax) and a higher saturation irradiance (Isat) than samples subjected to moderate drought (MD), and these, in turn, explain higher values of maximal photosynthesis and saturation irradiance than severe drought samples (SD). This behaviour confirms that plants with sufficient water availability, moderate temperature and adequate CO2 concentration have a greater capacity to photosynthesise efficiently, their stomata are open for the correct gas uptake, and chlorophylls, photosystems and chloroplast electron transport chains show good functionality.
Figure 1 indicates that, although all the cultivars studied belong to the same species, Olea europaea ssp europaea, their behaviour is very different. It can be observed that cultivars such as Chemlali (Ch), Empeltre (E) and Cornicabra (C) produce high assimilation of CO2 (above 12 µmols of CO2 m-2. s-1). Comparatively, Manzanilla de Sevilla (MS) does not exceed 2.5 µmols.m
-2.s
-1 and Hojiblanca (H) does not assimilate more than 6 µmols.m
-2.s
-1 under control conditions. It is striking that some of the cultivars photosynthesise more efficiently under MD conditions than under control conditions. This is the case of MS, Frantoio (F), Koroneiki (K) and Martina (M). Knowing the drought resistance of the olive tree, which is a typically Mediterranean species, it is not surprising that some olive cultivars may be more efficient when water availability is relatively reduced, as in the case of moderate drought conditions. This peculiarity is not directly related to drought tolerance: while K is one of the most sensitive cultivars, Martina is one of the most tolerant.
The behaviour of the cultivars follows several different patterns: there are some cultivars in which a small water deficit leads to a very sharp drop in photosynthesis. The most striking case occurs in Ch, which goes from being the most photosynthetically efficient plant under control conditions, assimilating more than 14 µmols of CO2 m-2·s-1, to producing almost no CO2 assimilation under MD and SD conditions. Although not to the extent of Chemlali, the same pattern occurs in Arbequina (A), E, F, H and Sikitita2 (S2). Other cultivars reduce their assimilation capacity sharply as the lack of water in the plant increases. This pattern of behaviour occurs in Arbosana (Ab), C, Cornezuelo de Jaén (CJ), K, M, MS, Picual (P) and Sikitita 1(S1). It should be noted that some cultivars, despite the extreme lack of water, continue to produce considerable CO2 assimilation in SD. This is the case of C, which produces a maximum photosynthesis of 6 µmols of CO2. m-2.s-1 in SD or M, which produces 4 µmols of CO2.m-2.s-1 under the same conditions. MS is the least photosynthetically efficient cultivar in control conditions, although the lack of water does not affect its assimilation capacity considerably in MD or SD.
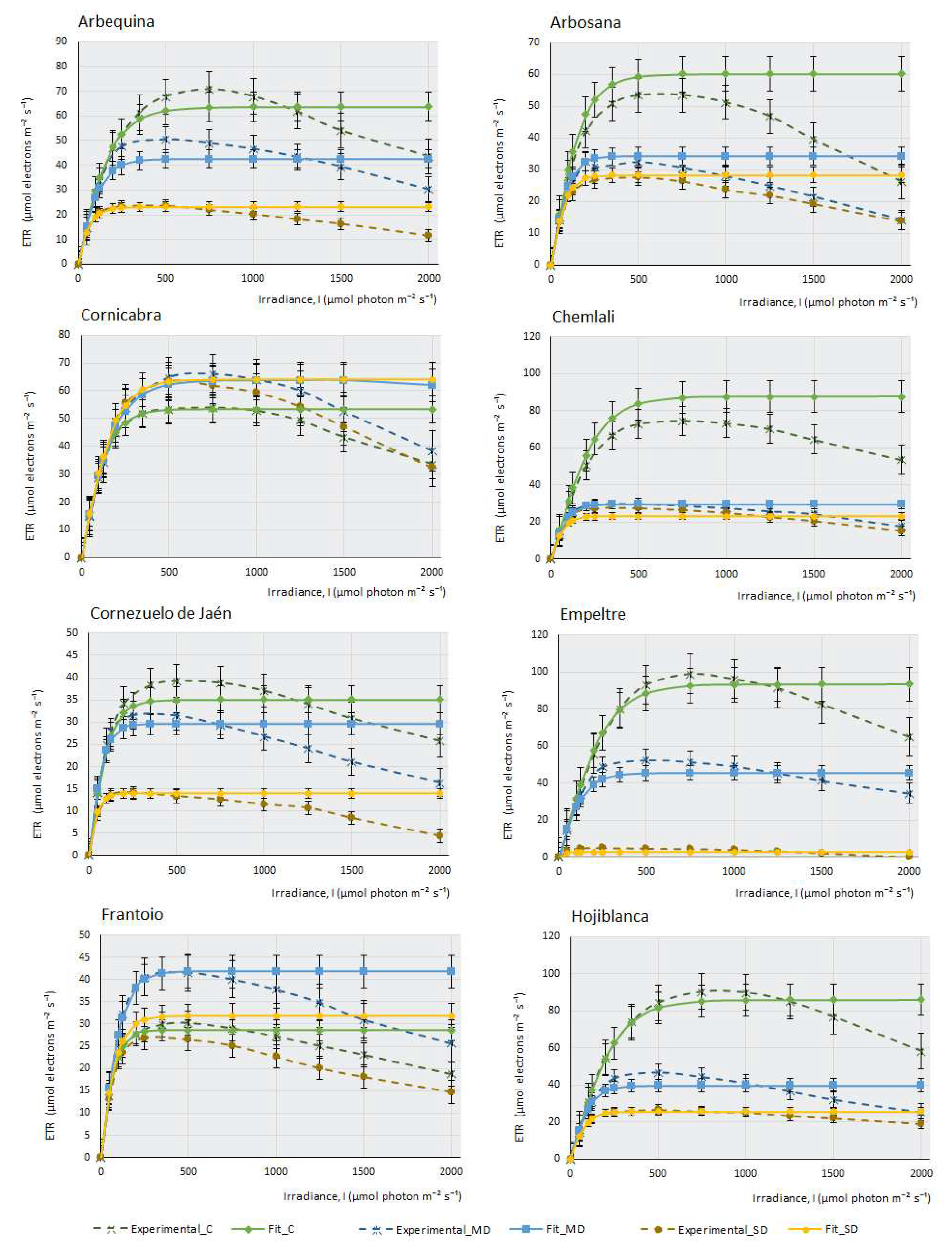
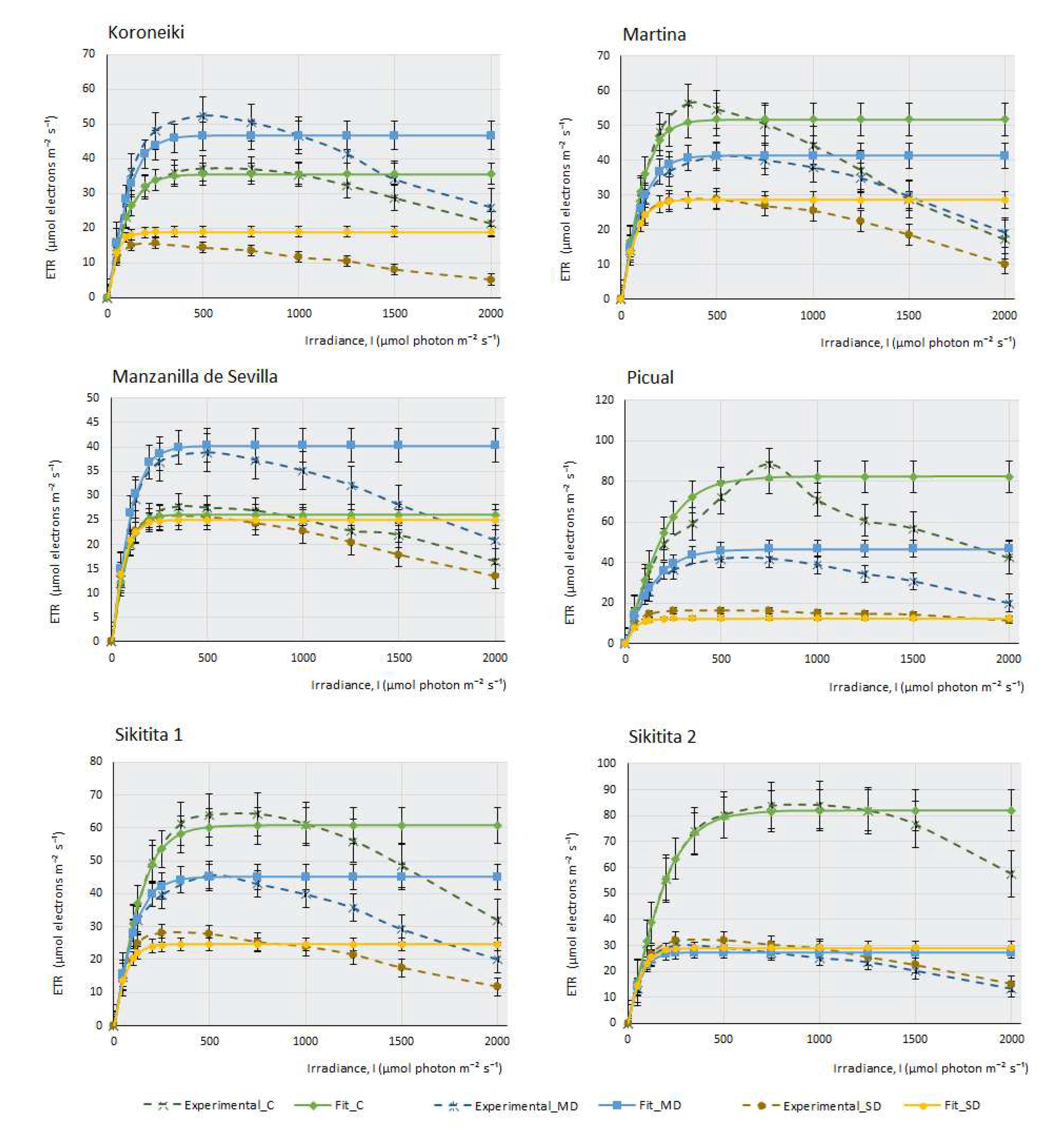
This work measured the electron transfer rate (ETR) that occurs along the transport chain during the luminous phase of photosynthesis in the thylakoids (
Figure 2), that is, the number of electrons that pass through PSII mainly, and also through PSI, as a consequence of the excitation produced by light on photosynthetic pigments that are able to produce energy in the form of ATP and NADPH in the plant.
Under drought conditions, ETR can be affected more severily or not, depending on the cultivar’s adaptation and tolerance to water stress. Water deprivation significantly impacts photosynthesis and electron transport along PSII, resulting in a reduction of the maximum electron transport speed (Jmax). In this case, PSII is less efficient (as an adaptive strategy to prevent damage from photo-oxidation under high light intensities during drought), affecting the activity and stability of these complexes. There is a decrease in the saturation irradiance (Isat) that produces Jmax. Another consequence of water stress is a decrease in the curve slope, indicating a minor transfer of electrons with increasing light intensity and, finally, water deficit produces an increased susceptibility to photo-inhibition.
The ETR curves at different light intensities (ETR/l) were obtained for each olive cultivar (
Figure 2). Under control conditions, with ad libitum irrigation of the samples, it can be observed that most cultivars produce a maximum ETR, except for some cultivars, such as F, K and MS, in which the efficiency of electron transfer is higher under MD conditions, although under ad libitum irrigation conditions, M cultivar suffers a considerable decrease of assimilation (
Figure 1) (proof that it is a very drought-tolerant variety); however, it is curious that the ETR value under control conditions is higher than in MD. Therefore, the amount of photosynthesis this plant produces does not depend so much on an increased electron transfer along the oxidation-reduction chain, but perhaps on enzymes related to the Calvin cycle, which may not be fully functional when the amount of water the plant receives is high.
Figure 2 indicates that, in general terms, the photosystems do not seem to be affected severely by the lack of water in the plant. Even so, although this electron transition occurs in the plant in all experimental conditions, including SD conditions, it does not translate into effective photosynthesis. The minimum amount of ETR produced in SD (except for E cultivar, where electron transfer seems to be very affected), occurs in Picual, with values of 10 µmols of electrons m-2. s-1. As an example, we can see that, in the case of F, the affectation produced in the capacity of the photosystems to transfer electrons is very small, (Emax in SD >25 µmols electrons.m-2.s-1); however, the assimilation of CO2 produced by this cultivar under MD conditions and, especially, in SD, is strongly affected by the lack of water. This observation suggests, once again, that it is not the macro-complexes associated with the thylakoids (PSII, PSI, Cit b6f) structures and their electron transfer which cause this sudden decrease in photosynthesis during drought stress. It should be noted that, in C, CJ and F, electron transfer is practically unaffected by drought, with very similar response curves in the three experimental conditions. In some cultivars (CH, H, and S2), there are barely noticeable differences between both drought conditions.
The purpose of adjusting these theoretical curves from the curves measured with the Li-Cor 6800 is to obtain and calculate parameters that will serve as indicators of the capacity of each cultivar to cope with the lack of water. These parameters are shown in
Table 1,
Table 2 and
Table 3. From the adjustment of the curves, provided that this adjustment has a correlation coefficient with a value greater than 0.95, very interesting parameters were calculated in the three experimental conditions in order to discern objectively the tolerance of each cultivar to drought.
Small discrepancies can be observed between the real values of Pmax, represented in
Figure 1 and
Figure 2, and the theoretical values of Pmax that these curves show after mathematical adjustment (which represents the maximum theoretical photosynthesis rate under ideal conditions provided by the ExpertCurve programme, necessary in eq 1 described in material and methods). This could be due to several reasons. Firstly, the equation being used to model the relationship between irradiance (I) and photosynthesis rate (P) may be a simplified representation of reality and may not capture all the complexities of the system. In some cases, a more complex model might be necessary to accurately describe the behaviour of photosynthesis. Secondly, the experimental data collected may have limitations or biases that affect the model’s ability to fit them. Experimental conditions, such as carbon dioxide (CO2) concentration, temperature, humidity, and other factors, can affect the ability of photosynthesis to reach its maximum rate under ideal conditions. Thirdly, photosynthesis shows a saturation response, which means that, as the irradiance increases, the rate of photosynthesis increases until it reaches a maximum value, after which the rate of photosynthesis no longer increases. This could explain why the calculated values of Theorical_P do not reach the maximum value of “P” in the range of irradiances. Lastly, respiration in darkness (Rd) is not being considered, which eventually reduces (depending on its value) the rate of photosynthesis in the graph.
In summary, although the “Pmax” value represents the theoretical maximum rate of photosynthesis under ideal conditions, in practice, several factors can limit the ability of plants to achieve this maximum rate under all irradiance conditions. Photosynthesis is a complex process influenced by multiple variables, and it is important to consider these limitations when interpreting the results and fitting the model.
Table 1 shows that the maximum photosynthesis (Pmax) progressively decreases in general with the lack of water and, in turn, the saturation irradiances (Isat50 and Isat99.9) decrease in a positive correlation with it. This decrease is logical, since drought stress directly affects the availability of CO₂ inside the cells of the leaf as a consequence of the stomatal closure induced by ABA. The efficiency of photosystem II, ф_PSII also decreases, indicating that, with the lack of water, a greater number of photons are necessary to fix a molecule of CO
2. The maximum electron transport speed (Jmax, or the ability to assimilate CO2) decreases in all cases, becoming almost zero for cultivars like F in SD. Rd and MS present strikingly high respiration in the dark compared to the rest of the cultivars. This parameter seems to remain constant in most cultivars for all experimental conditions, except for C and F, where it decreases with drought. The same occurs with the Fv/Fm values, which remain constant or decrease slightly with water deficit in all cultivars.
Table 2 shows the values of some parameters obtained from the fitted curves of ETR response versus irradiance (
Figure 2). Although the mathematical fit of these curves with the real curves is not as good as in the first case (photosynthesis-irradiance response), the calculation of the parameters indicated in
Table 2 were obtained from the zones of the curve in which there is a fit with a value of R> 0.95 with the real curves. ETRmax is the maximum electron transfer rate in the thylakoids during the luminous reactions. This parameter, as can be seen in
Table 2, decreases with drought, which indicates the importance of water in the light phase of photosynthesis. The plant, by not having good functionality, decreases its capacity to receive light and transform it into stable chemical energy. This occurs in all cultivars, except in F, K and MS, where the maximum radius of electron transfer occurs under MD conditions. Alpha-ETR is the slope of the curve, that is, the efficiency of photosynthesis in relation to incident light intensity (measured in micromoles electrons/micromols photons). This efficiency is quite stable with the lack of water, which indicates that a practically constant number of electrons jump in the protein complexes of the chloroplast with a photon of light, regardless of the water deficit. Of course, the result produced on the ETRmax is different, since the efficiency of transferring those electrons from one macro-complex to another is affected by the lack of water. For example, in the case of the CH cultivar, although alphaET () remains almost constant in the three experimental conditions, ETRmax goes from 88.9 under control conditions to 22.9 in MD and 26.1 in SD. The exception occurs in two cultivars, CJ and P, in which decreases with drought.
ETRd is the electron transport rate when the plant is adapted to the dark and, therefore, all the macrocomplexes inserted in the thylakoids are completely reduced. Logically, this parameter has a very low value, since the plant is in darkness and does not receive luminous energy. ISAT_ETR50 refers to the point at which the electron transport rate reaches 50% of its maximum value in response to light intensity. Therefore, ISAT_ETR75 and ISAT_ETR99.9 are the irradiances at which 75% and 99.9% of electron transfer occurs, respectively, between the protein macro complexes involved in the red-ox chain of the light phase.
With respect to the data reflected in
Table 3, it should be noted that ΔF/Fv’ represents the photosynthetic efficiency of the plant adapted to light. The value of this parameter decreases with the lack of water in general terms. There are some exceptions, such as M, where the maximum efficiency occurs in MD conditions, and S1, where its maximum efficiency occurs in SD. Both cultivars turn out to be very drought-tolerant. Moreover, Fv/Fm is always greater than ΔF/Fm’, since the first parameter is measured when the plant is adapted to darkness, where all macrocomplexes are in a reduced state. NPQ (Non-Photochemical Quenching) is the capacity of photosystems to dissipate excess energy as heat. To this end, the amount of carotenoids that are part of the antennas of the photosystems, which are responsible for obtaining this excess energy and dissipating it into the atmosphere in the form of heat, play a fundamental role. qP (Photochemical Quenching) represents the fraction of photosystems that are in the reduced state, i.e., available for the photochemical process, that is, the fraction of light that is, at those moments, used to produce photosynthesis, and qN (Non-Photochemical Quenching) represents the fraction of reaction centres of PSII that are in an oxidised state, or the quantity of dissipated light. The amount of photons necessary to make an electron jump increases with drought; this parameter, above all, is greatly increased in SD conditions. The number of photons required to fix a CO2 molecule is slightly increased between the control samples and the samples subjected to MD. In some cultivars, even a moderate lack of water in the plant favours the CO2 fixation process.
Finally, the amount of electrons that the plant needs to transfer along the redox chain to produce the fixation of a CO2 molecule is also increased with the lack of water.