Submitted:
19 September 2023
Posted:
21 September 2023
Read the latest preprint version here
Abstract
Keywords:
1. Introduction
2. Clinical features
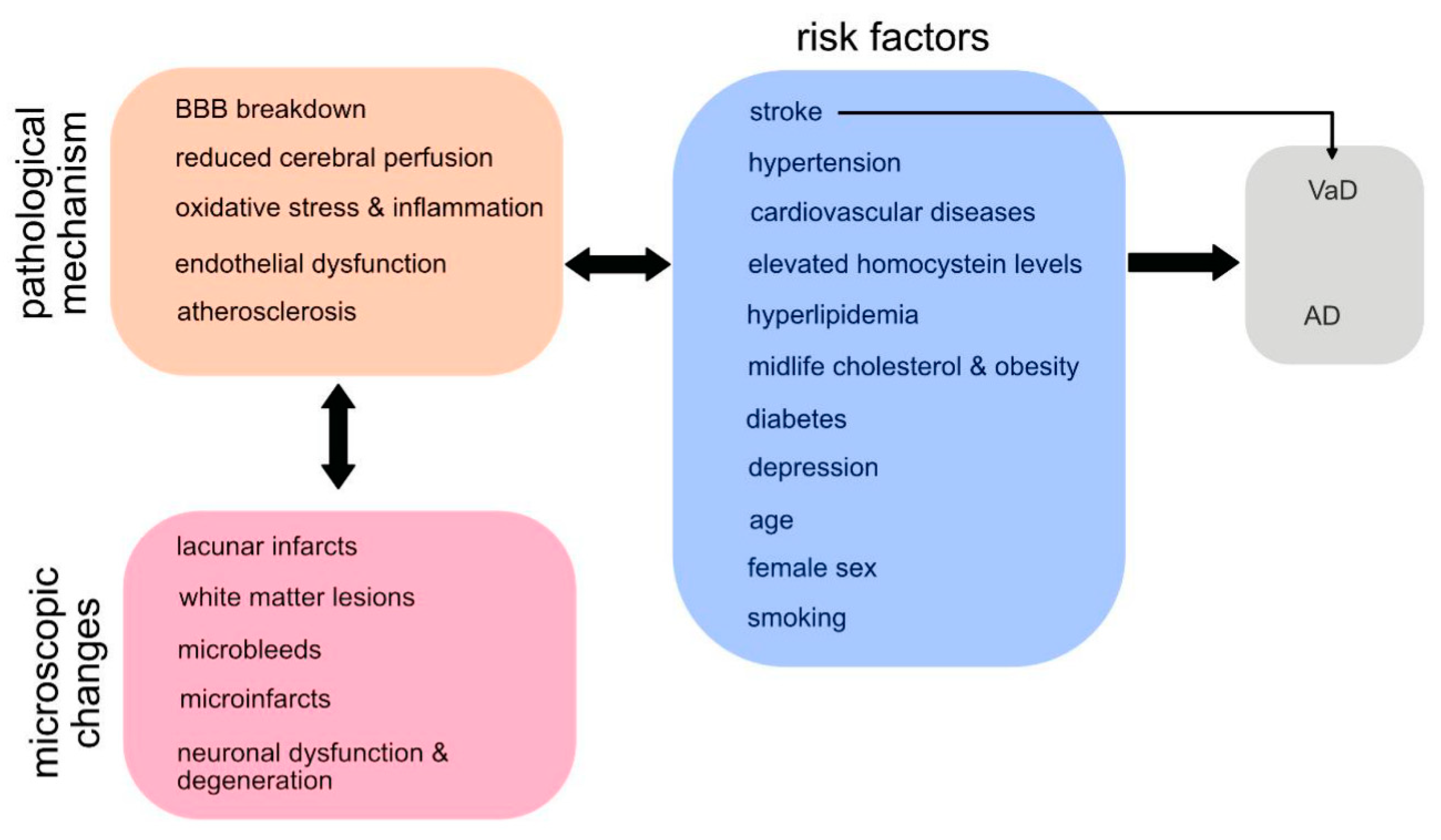
2.1. Prevalence and risk factors
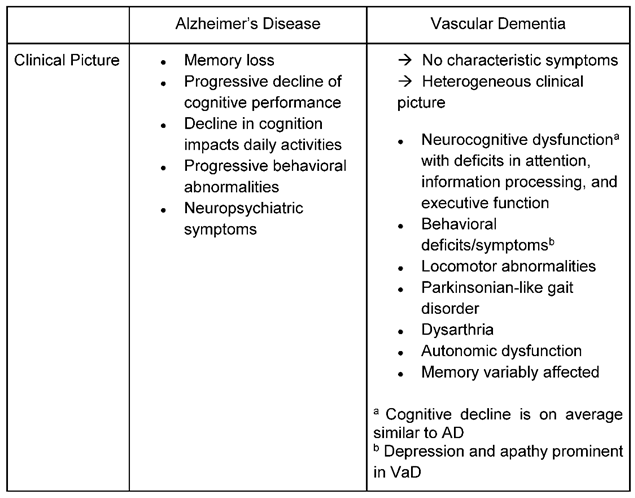
2.2. Clinical presentation
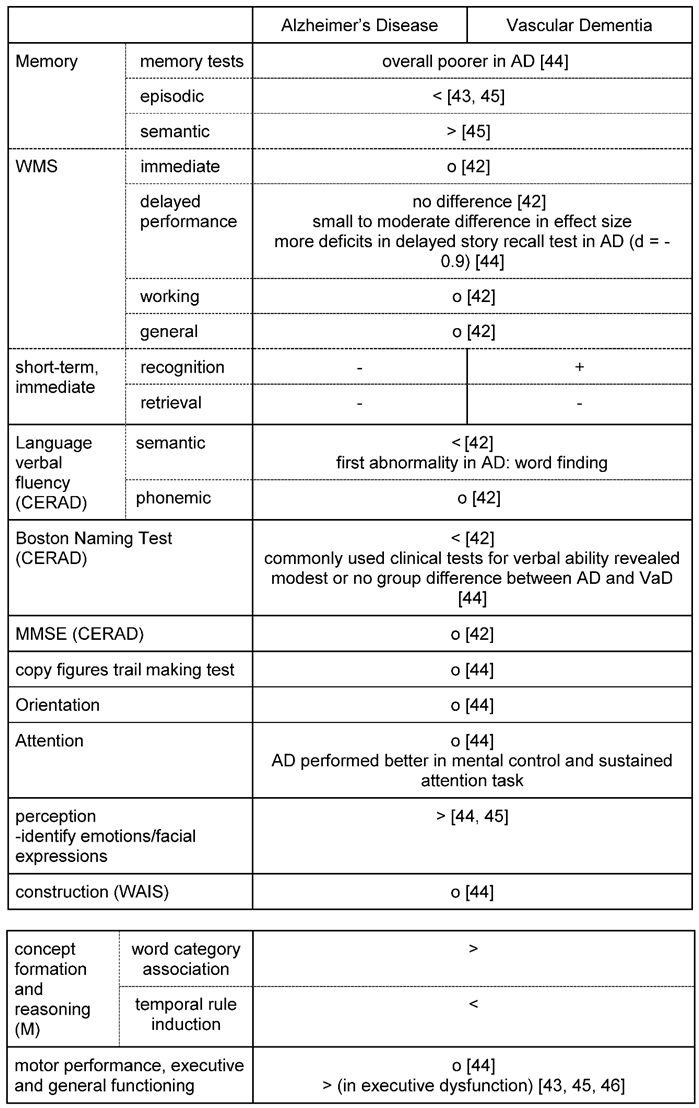
2.3. Typical clinical diagnosis parameters
3. Pathology
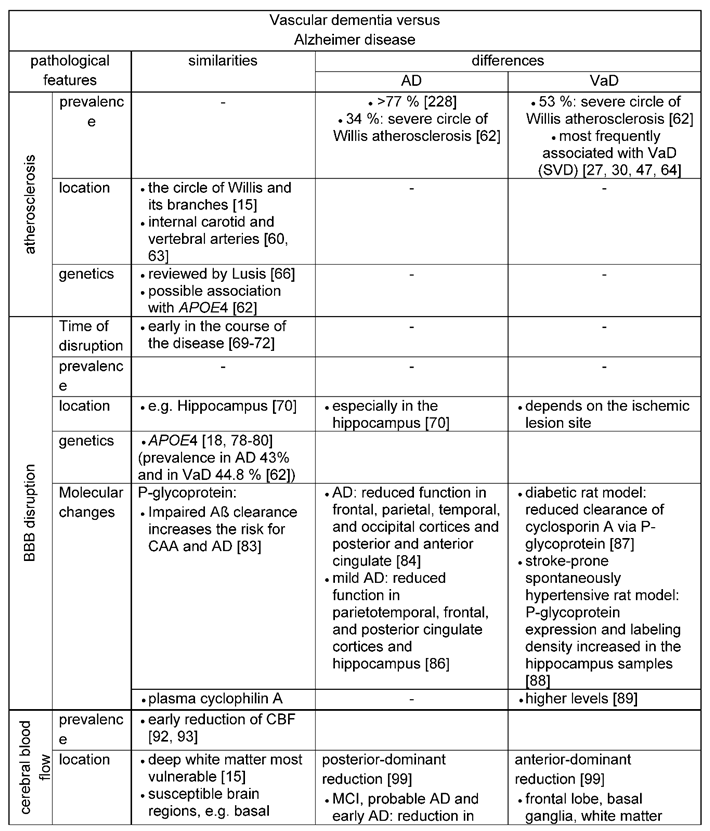
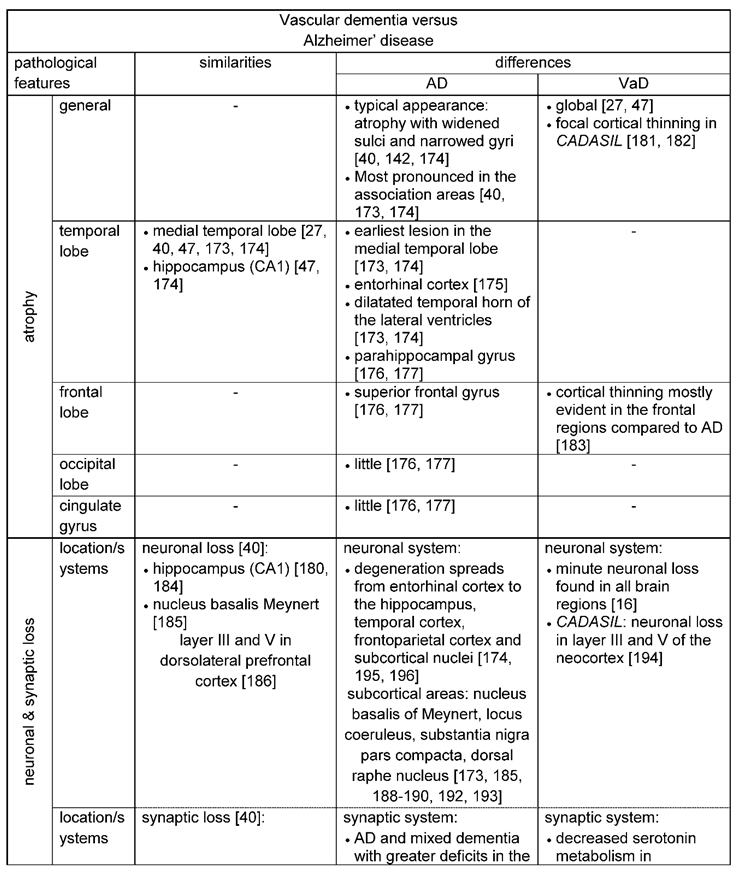
3.1. Similar and distinct vascular features of Alzheimer`s versus vascular dementia
3.1.1. Atherosclerosis
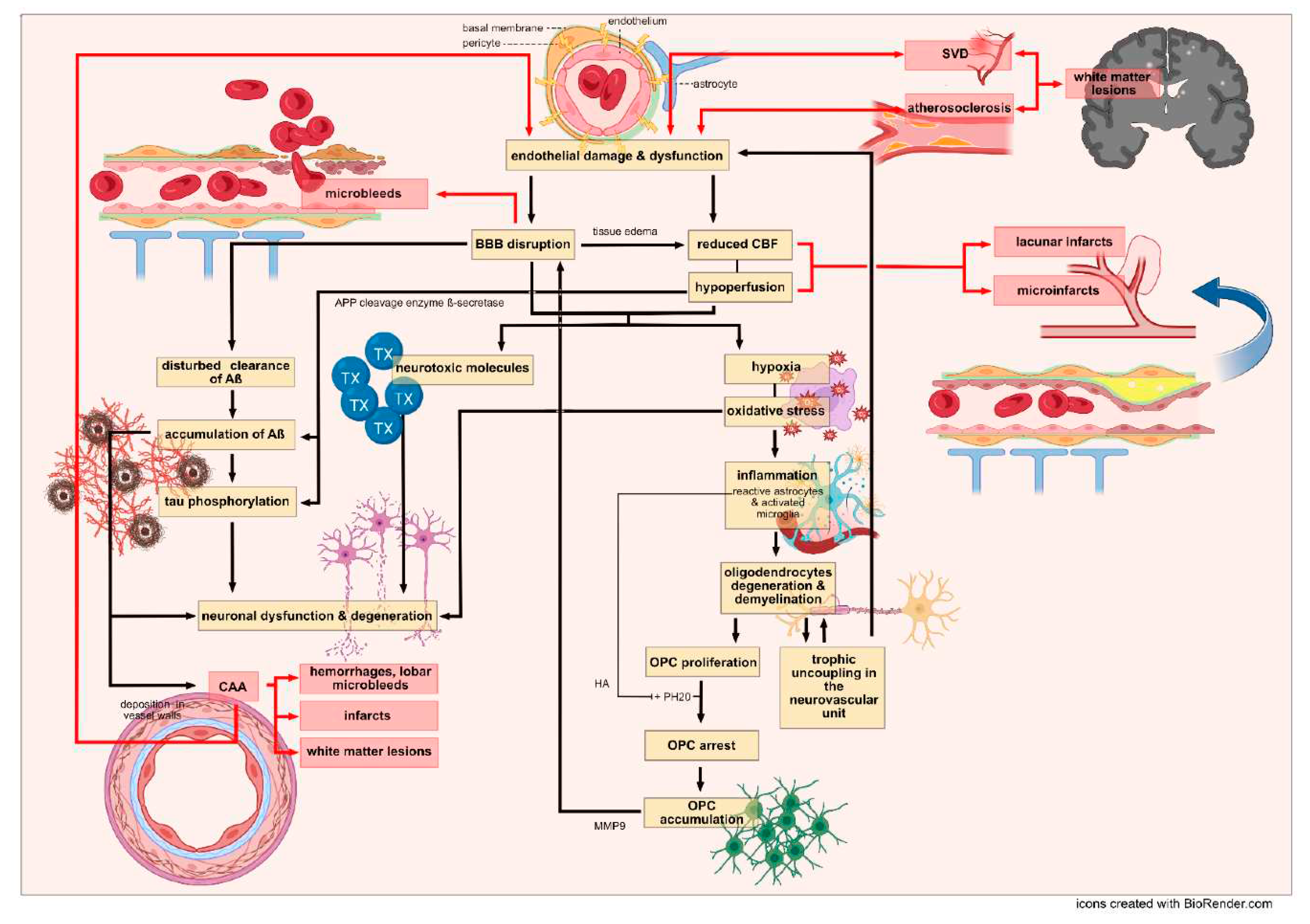
3.1.2. Mechanisms of endothelial dysfunction
3.1.3. Cerebral blood flow
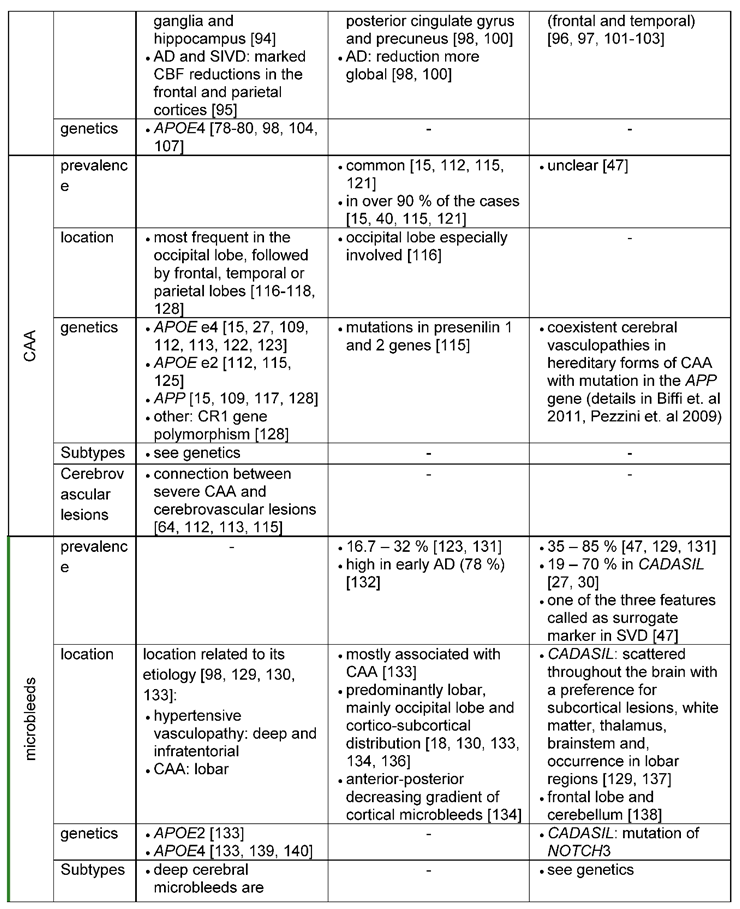
3.1.4. Cerebral amyloid angiopathy (CAA)
3.1.5. Hemorrhagic lesions
3.1.6. Ischemic lesions
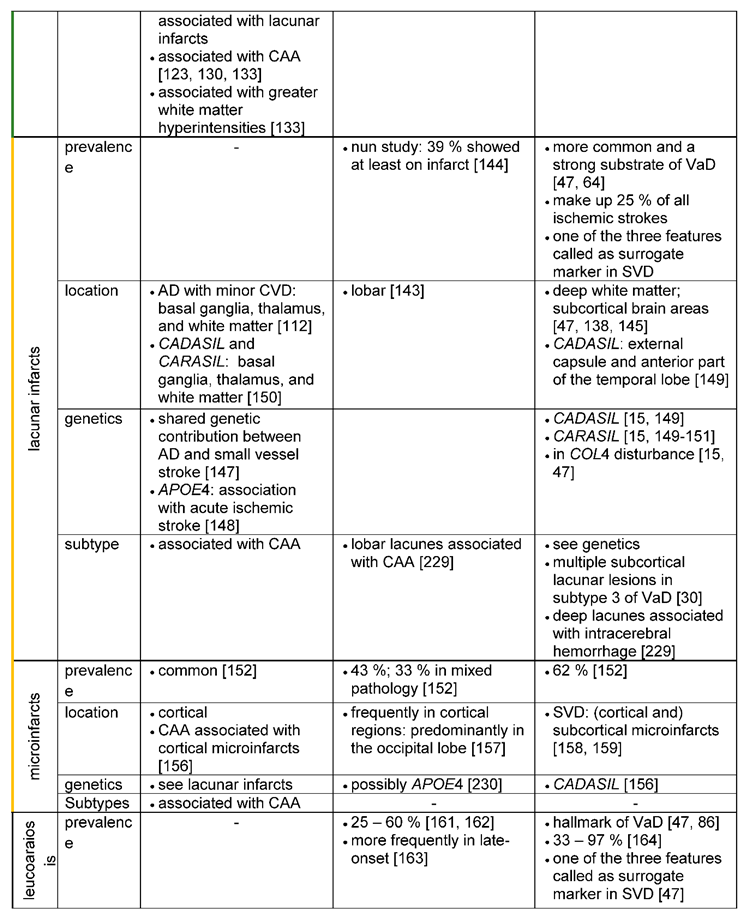
3.1.6.1. Lacunar infarcts
3.1.6.2. Microinfarcts
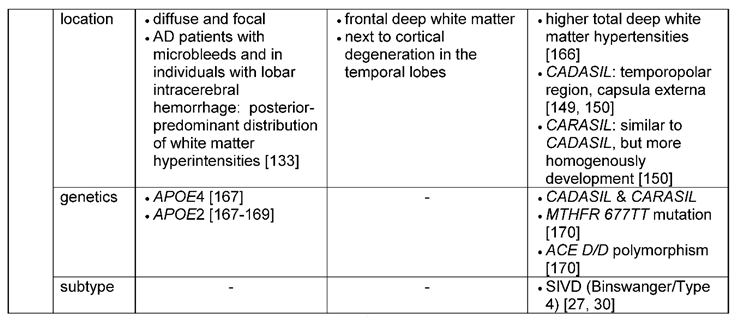
3.1.6.3. Leukoaraiosis
3.2. Similar and diverse neuronal features of Alzheimer`s versus vascular dementia
3.2.1. Brain areas differently affected in Alzheimer`s and vascular dementia
3.2.1.1. Macroscopical changes
3.2.1.2. Changes on cellular and subcellular structures
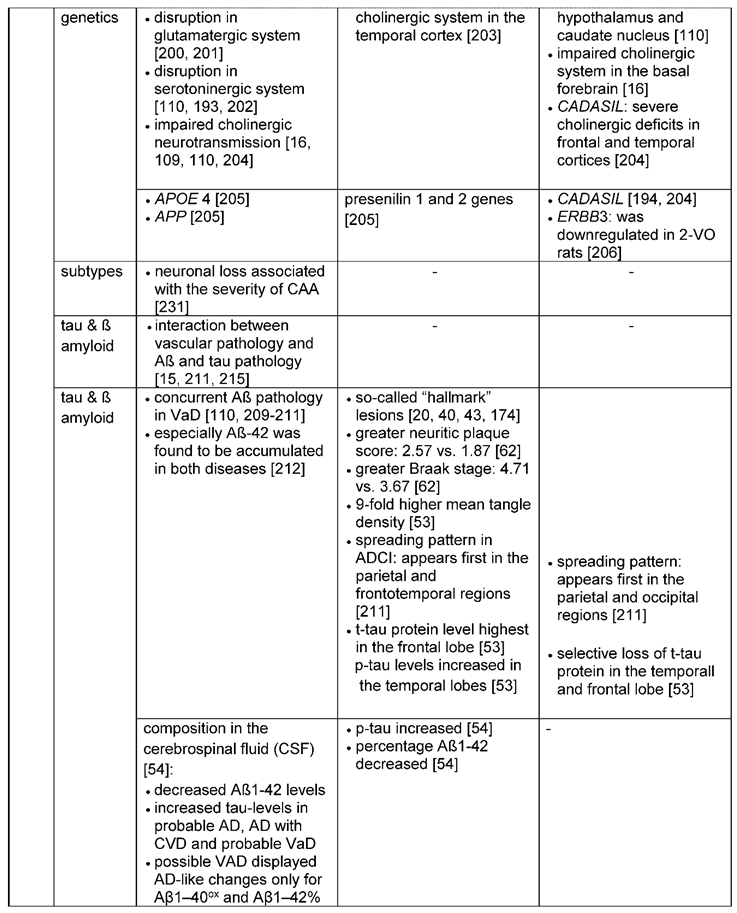
3.2.2. Genetic and cell-cycle related changes involved in neuronal loss and brain atrophy
3.2.3. Amyloid-ß and tau pathology
3.2.4. Brain metabolism
4. Conclusion
- 1)
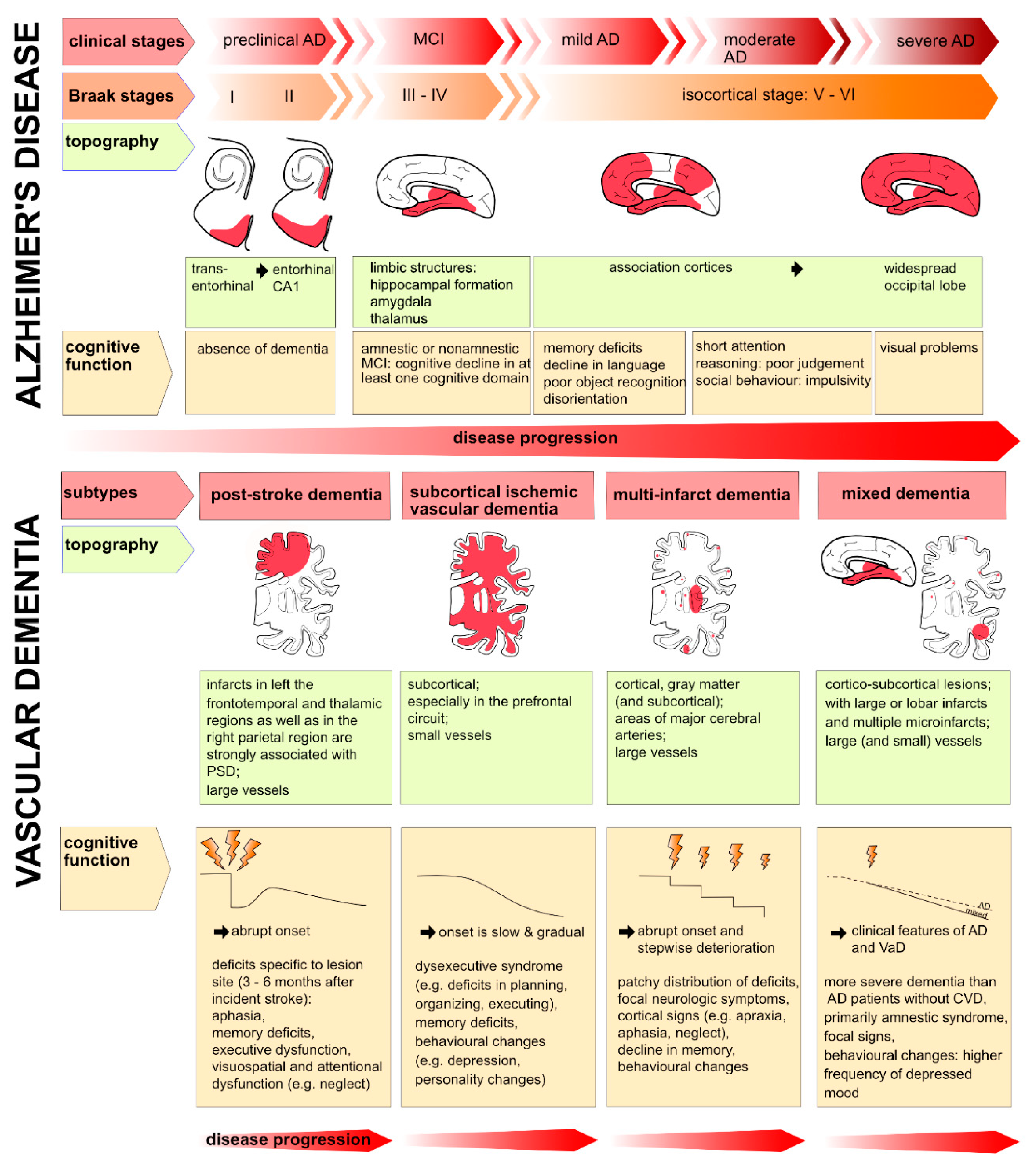
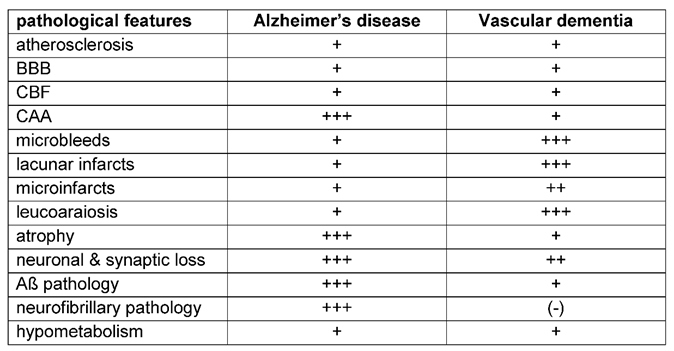
- MR Imaging can reveal medial temporal lobe atrophy or entorhinal cortex atrophy as early neuromorphological markers for AD [173,174,175]. While medial temporal lobe atrophy can also be detected in VaD, brain atrophy in VaD is more globally distributed [27,47]. Other imaging biomarkers are microbleeds, lacunar infarcts, microinfarcts and leukoaraiosis. Their location can provide additional diagnostic clues: E.g. Microbleeds are located preferentially lobar, mainly in the occipital lobe in AD [136]. In contrast, a scattered distribution throughout the brain (with described involvement of frontal lobe and cerebellum) would be typical for VaD [129,137].
- 2)
- CBF reduction and BBB breakdown are early pathological alterations in both diseases. Posterior-dominant reduction of CBF in AD and anterior-dominant reduction of CBF in VaD [99], as well as molecular factors related to the BBB, such as P-glycoprotein and plasma cyclophilin A levels can serve as early makers when distinguishing VaD and AD.
- 3)
- 4)
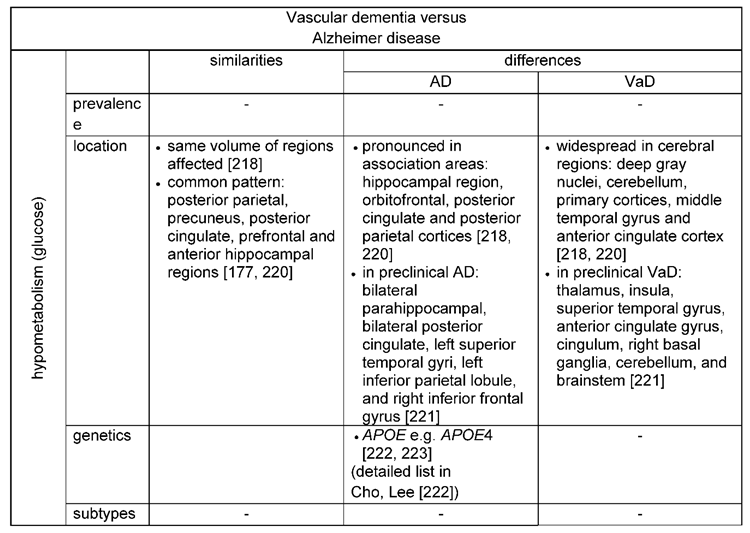
- Glucose metabolism measured by positron emission topography (PET) show a different pattern of hypometabolism in both forms of dementia.
- 5)
- Alzheimer’s presents with a typical disease progression with the hippocampal region being affected first, whereas in VaD the course of the disease depends on the subtype with an abrupt, stepwise or gradual development of cognitive decline (Figure 3).
Author Contributions
Funding
Acknowledgements
Conflict of interest
References
- World Health Organization. Dementia. 2023.
- Hurd, M.D.; Martorell, P.; Delavande, A.; Mullen, K.J.; Langa, K.M. Monetary costs of dementia in the United States. N Engl J Med. 2013, 368, 1326–1334. [Google Scholar] [CrossRef] [PubMed]
- Hardy, J.A.; Higgins, G.A. Alzheimer's disease: the amyloid cascade hypothesis. Science. 1992, 256, 184–185. [Google Scholar] [PubMed]
- Braak, H.; Braak, E. Neuropathological stageing of Alzheimer-related changes. Acta Neuropathol. 1991, 82, 239–259. [Google Scholar] [PubMed]
- Heneka MT, Carson MJ, El Khoury J, Landreth GE, Brosseron F, Feinstein DL, et al. Neuroinflammation in Alzheimer's disease. Lancet Neurol. 2015, 14, 388–405. [Google Scholar]
- Heneka, M.T.; Golenbock, D.T.; Latz, E. Innate immunity in Alzheimer's disease. Nat Immunol. 2015, 16, 229–236. [Google Scholar]
- Labzin, L.I.; Heneka, M.T.; Latz, E. Innate Immunity and Neurodegeneration. Annu Rev Med. 2018, 69, 437–449. [Google Scholar] [CrossRef]
- Le Page A, Dupuis G, Frost EH, Larbi A, Pawelec G, Witkowski JM, et al. Role of the peripheral innate immune system in the development of Alzheimer's disease. Exp Gerontol. 2018, 107, 59–66. [Google Scholar]
- Tejera, D.; Heneka, M.T. Microglia in Alzheimer's disease: the good, the bad and the ugly. Curr Alzheimer Res. 2016, 13, 370–380. [Google Scholar] [CrossRef]
- Jansen IE, Savage JE, Watanabe K, Bryois J, Williams DM, Steinberg S, et al. Genome-wide meta-analysis identifies new loci and functional pathways influencing Alzheimer's disease risk. Nat Genet. 2019, 51, 404–413. [Google Scholar]
- McQuade, A.; Blurton-Jones, M. Microglia in Alzheimer's Disease: Exploring How Genetics and Phenotype Influence Risk. J Mol Biol. 2019, 431, 1805–1817. [Google Scholar] [PubMed]
- de la Torre, JC. Alzheimer disease as a vascular disorder: nosological evidence. Stroke. 2002, 33, 1152–1162. [Google Scholar] [PubMed]
- Pąchalska, M.; Bidzan, L.; Bidzan, M.; Góral-Półrola, J. Vascular Factors and Cognitive Dysfunction in Alzheimer Disease. Med Sci Monit. 2015, 21, 3483–3489. [Google Scholar] [PubMed]
- Nucera A, Hachinski V. Cerebrovascular and Alzheimer disease: fellow travelers or partners in crime? J Neurochem. 2018, 144, 513–516. [Google Scholar] [CrossRef] [PubMed]
- Iadecola, C. The pathobiology of vascular dementia. Neuron. 2013, 80, 844–866. [Google Scholar]
- Jellinger, K.A. Pathology and pathogenesis of vascular cognitive impairment-a critical update. Front Aging Neurosci. 2013, 5, 17. [Google Scholar] [CrossRef]
- Toledo JB, Arnold SE, Raible K, Brettschneider J, Xie SX, Grossman M, et al. Contribution of cerebrovascular disease in autopsy confirmed neurodegenerative disease cases in the National Alzheimer's Coordinating Centre. Brain. 2013, 136, 2697–2706. [Google Scholar]
- Sweeney, M.D.; Sagare, A.P.; Zlokovic, B.V. Blood-brain barrier breakdown in Alzheimer disease and other neurodegenerative disorders. Nat Rev Neurol. 2018, 14, 133–150. [Google Scholar]
- Pugazhenthi, S. Metabolic Syndrome and the Cellular Phase of Alzheimer's Disease. Prog Mol Biol Transl Sci. 2017, 146, 243–258. [Google Scholar]
- DeTure, M.A.; Dickson, D.W. The neuropathological diagnosis of Alzheimer's disease. Mol Neurodegener. 2019, 14, 32. [Google Scholar]
- de la Torre, JC. Alzheimer's disease is a vasocognopathy: a new term to describe its nature. Neurol Res. 2004, 26, 517–524. [Google Scholar] [PubMed]
- Claudio, L. Ultrastructural features of the blood-brain barrier in biopsy tissue from Alzheimer's disease patients. Acta Neuropathol. 1996, 91, 6–14. [Google Scholar] [CrossRef] [PubMed]
- Yamashita, K.; Miyakawa, T.; Katsuragi, S. Vascular changes in the brains with Alzheimer's disease. Jpn J Psychiatry Neurol. 1991, 45, 79–84. [Google Scholar] [CrossRef] [PubMed]
- Kalaria, R.N.; Hedera, P. Differential degeneration of the cerebral microvasculature in Alzheimer's disease. Neuroreport. 1995, 6, 477–480. [Google Scholar] [CrossRef]
- Launer LJ, Petrovitch H, Ross GW, Markesbery W, White LR. AD brain pathology: vascular origins? Results from the HAAS autopsy study. Neurobiol Aging. 2008, 29, 1587–1590. [Google Scholar] [CrossRef]
- Schneider, J.A. High blood pressure and microinfarcts: a link between vascular risk factors, dementia, and clinical Alzheimer's disease. J Am Geriatr Soc. 2009, 57, 2146–2147. [Google Scholar] [CrossRef]
- O'Brien, J.T.; Thomas, A. Vascular dementia. Lancet. 2015, 386, 1698–1706. [Google Scholar] [CrossRef]
- Pendlebury, S.T.; Rothwell, P.M. Prevalence, incidence, and factors associated with pre-stroke and post-stroke dementia: a systematic review and meta-analysis. Lancet Neurol. 2009, 8, 1006–1018. [Google Scholar]
- Allan LM, Rowan EN, Firbank MJ, Thomas AJ, Parry SW, Polvikoski TM, et al. Long term incidence of dementia, predictors of mortality and pathological diagnosis in older stroke survivors. Brain. 2011, 134, 3716–3727. [Google Scholar] [CrossRef]
- Konno, S.; Meyer, J.S.; Terayama, Y.; Margishvili, G.M.; Mortel, K.F. Classification, diagnosis and treatment of vascular dementia. Drugs Aging. 1997, 11, 361–373. [Google Scholar] [CrossRef]
- Iadecola C, Duering M, Hachinski V, Joutel A, Pendlebury ST, Schneider JA, et al. Vascular Cognitive Impairment and Dementia: JACC Scientific Expert Panel. J Am Coll Cardiol. 2019, 73, 3326–3344. [Google Scholar]
- Scheyer O, Rahman A, Hristov H, Berkowitz C, Isaacson RS, Diaz Brinton R, et al. Female Sex and Alzheimer's Risk: The Menopause Connection. J Prev Alzheimers Dis. 2018, 5, 225–230. [Google Scholar]
- Podcasy, J.L.; Epperson, C.N. Considering sex and gender in Alzheimer disease and other dementias. Dialogues Clin Neurosci. 2016, 18, 437–446. [Google Scholar] [PubMed]
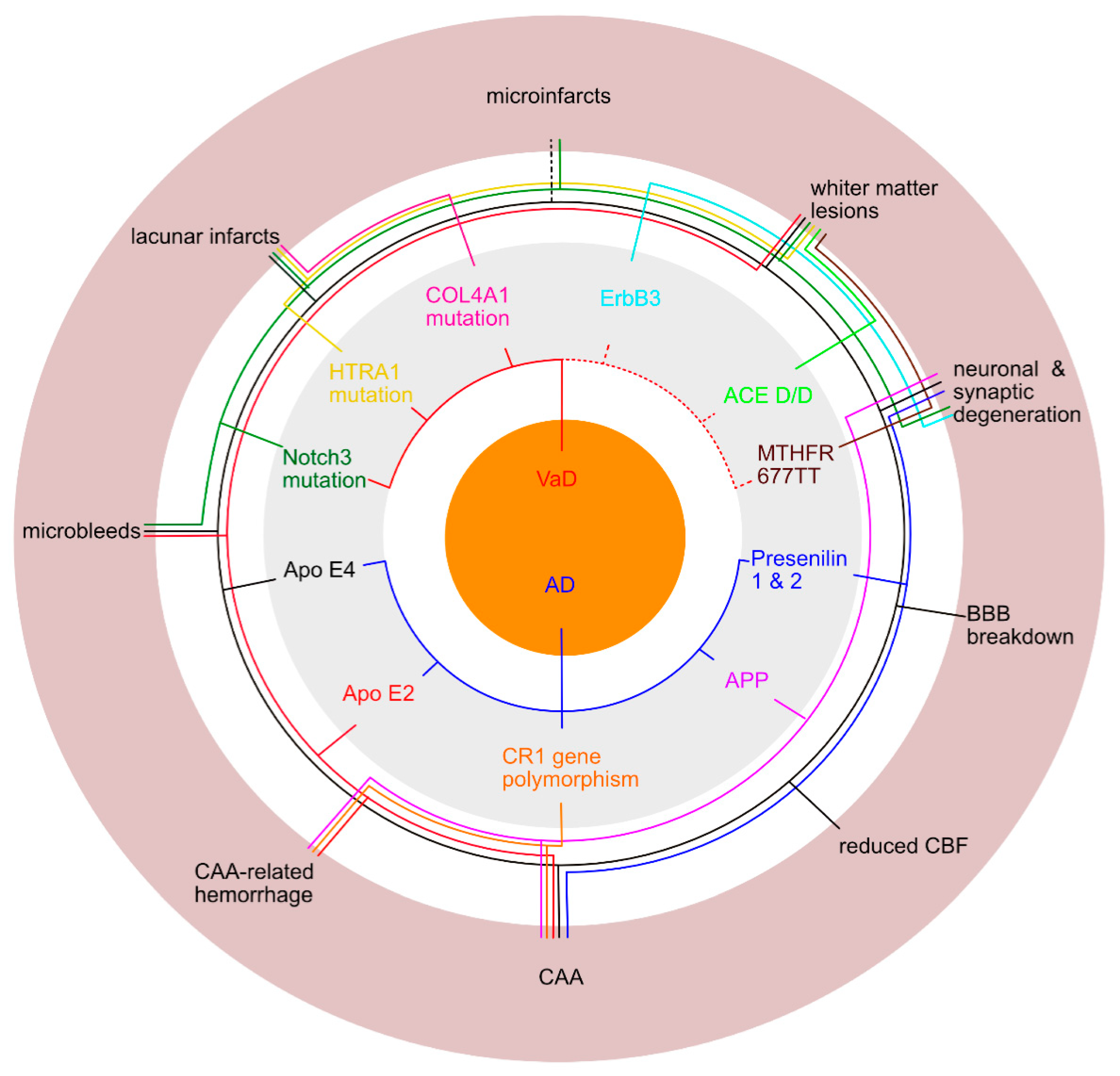
- Markus, H.S.; Schmidt, R. Genetics of Vascular Cognitive Impairment. Stroke. 2019, 50, 765–772. [Google Scholar]
- 35. National Institute on Aging. Alzheimer's Disease Genetics Fact Sheet.
- Dichgans, M.; Zietemann, V. Prevention of vascular cognitive impairment. Stroke. 2012, 43, 3137–3146. [Google Scholar]
- Barnes, D.E.; Yaffe, K. The projected effect of risk factor reduction on Alzheimer's disease prevalence. Lancet Neurol. 2011, 10, 819–828. [Google Scholar]
- Román, G.C. Vascular dementia prevention: a risk factor analysis. Cerebrovasc Dis. 2005, 20 Suppl. 2, 91–100. [Google Scholar]
- Diniz BS, Butters MA, Albert SM, Dew MA, Reynolds CF, 3rd. Late-life depression and risk of vascular dementia and Alzheimer's disease: systematic review and meta-analysis of community-based cohort studies. Br J Psychiatry. 2013, 202, 329–335. [Google Scholar]
- Castellani, R.J.; Rolston, R.K.; Smith, M.A. Alzheimer disease. Dis Mon. 2010, 56, 484–546. [Google Scholar]
- Lee, A.Y. Vascular dementia. Chonnam Med J. 2011, 47, 66–71. [Google Scholar]
- Braaten, A.J.; Parsons, T.D.; McCue, R.; Sellers, A.; Burns, W.J. Neurocognitive differential diagnosis of dementing diseases: Alzheimer's Dementia, Vascular Dementia, Frontotemporal Dementia, and Major Depressive Disorder. Int J Neurosci. 2006, 116, 1271–1293. [Google Scholar] [CrossRef] [PubMed]
- Weintraub, S.; Wicklund, A.H.; Salmon, D.P. The neuropsychological profile of Alzheimer disease. Cold Spring Harb Perspect Med. 2012, 2, a006171. [Google Scholar] [CrossRef] [PubMed]
- Mathias, J.L.; Burke, J. Cognitive functioning in Alzheimer's and vascular dementia: a meta-analysis. Neuropsychology. 2009, 23, 411–423. [Google Scholar] [CrossRef] [PubMed]
- Graham, N.L.; Emery, T.; Hodges, J.R. Distinctive cognitive profiles in Alzheimer's disease and subcortical vascular dementia. J Neurol Neurosurg Psychiatry. 2004, 75, 61–71. [Google Scholar]
- Murray, M.E.; Knopman, D.S.; Dickson, D.W. Vascular dementia: clinical, neuroradiologic and neuropathologic aspects. Panminerva Med. 2007, 49, 197–207. [Google Scholar]
- Kalaria, R.N. The pathology and pathophysiology of vascular dementia. Neuropharmacology. 2018, 134, 226–239. [Google Scholar]
- Jack CR, Jr. , Bennett DA, Blennow K, Carrillo MC, Dunn B, Haeberlein SB, et al. NIA-AA Research Framework: Toward a biological definition of Alzheimer's disease. Alzheimers Dement. 2018, 14, 535–562. [Google Scholar]
- Hachinski, V.C.; Bowler, J.V. Vascular dementia. Neurology. 1993, 43, 2159–2160. [Google Scholar] [CrossRef]
- Skrobot OA, Black SE, Chen C, DeCarli C, Erkinjuntti T, Ford GA, et al. Progress toward standardized diagnosis of vascular cognitive impairment: Guidelines from the Vascular Impairment of Cognition Classification Consensus Study. Alzheimers Dement. 2018, 14, 280–292. [Google Scholar]
- Scheltens P, Leys D, Barkhof F, Huglo D, Weinstein HC, Vermersch P, et al. Atrophy of medial temporal lobes on MRI in "probable" Alzheimer's disease and normal ageing: diagnostic value and neuropsychological correlates. J Neurol Neurosurg Psychiatry. 1992, 55, 967–972. [Google Scholar]
- Rau, A.; Urbach, H. The MTA score-simple and reliable, the best for now? Eur Radiol. 2021, 31, 9057–9059. [Google Scholar] [CrossRef] [PubMed]
- Mukaetova-Ladinska EB, Abdel-All Z, Mugica ES, Li M, Craggs LJ, Oakley AE, et al. Tau proteins in the temporal and frontal cortices in patients with vascular dementia. J Neuropathol Exp Neurol. 2015, 74, 148–157. [Google Scholar] [CrossRef] [PubMed]
- Bibl M, Mollenhauer B, Esselmann H, Schneider M, Lewczuk P, Welge V, et al. Cerebrospinal fluid neurochemical phenotypes in vascular dementias: original data and mini-review. Dement Geriatr Cogn Disord. 2008, 25, 256–265. [Google Scholar] [CrossRef] [PubMed]
- Thijssen EH, Verberk IMW, Kindermans J, Abramian A, Vanbrabant J, Ball AJ, et al. Differential diagnostic performance of a panel of plasma biomarkers for different types of dementia. Alzheimers Dement (Amst). 2022, 14, e12285. [Google Scholar]
- Leuzy, A.; Mattsson-Carlgren, N.; Palmqvist, S.; Janelidze, S.; Dage, J.L.; Hansson, O. Blood-based biomarkers for Alzheimer's disease. EMBO Mol Med. 2022, 14, e14408. [Google Scholar]
- Teunissen CE, Verberk IMW, Thijssen EH, Vermunt L, Hansson O, Zetterberg H, et al. Blood-based biomarkers for Alzheimer's disease: towards clinical implementation. Lancet Neurol. 2022, 21, 66–77. [Google Scholar]
- Arvanitakis, Z.; Capuano, A.W.; Leurgans, S.E.; Bennett, D.A.; Schneider, J.A. Relation of cerebral vessel disease to Alzheimer's disease dementia and cognitive function in elderly people: a cross-sectional study. Lancet Neurol. 2016, 15, 934–943. [Google Scholar]
- Lusis, AJ. Atherosclerosis. Nature. 2000, 407, 233–241. [Google Scholar]
- Shabir, O.; Berwick, J.; Francis, S.E. Neurovascular dysfunction in vascular dementia, Alzheimer's and atherosclerosis. BMC Neurosci. 2018, 19, 62. [Google Scholar]
- Fava, C.; Montagnana, M. Atherosclerosis Is an Inflammatory Disease which Lacks a Common Anti-inflammatory Therapy: How Human Genetics Can Help to This Issue. A Narrative Review. Front Pharmacol. 2018, 9, 55. [Google Scholar] [CrossRef]
- Beach TG, Wilson JR, Sue LI, Newell A, Poston M, Cisneros R, et al. Circle of Willis atherosclerosis: association with Alzheimer's disease, neuritic plaques and neurofibrillary tangles. Acta Neuropathol. 2007, 113, 13–21. [Google Scholar]
- van Oijen M, de Jong FJ, Witteman JC, Hofman A, Koudstaal PJ, Breteler MM. Atherosclerosis and risk for dementia. Ann Neurol. 2007, 61, 403–410. [Google Scholar] [CrossRef] [PubMed]
- Thal, D.R.; Grinberg, L.T.; Attems, J. Vascular dementia: different forms of vessel disorders contribute to the development of dementia in the elderly brain. Exp Gerontol. 2012, 47, 816–824. [Google Scholar] [CrossRef] [PubMed]
- Gustavsson, A.M.; van Westen, D.; Stomrud, E.; Engström, G.; Nägga, K.; Hansson, O. Midlife Atherosclerosis and Development of Alzheimer or Vascular Dementia. Ann Neurol. 2020, 87, 52–62. [Google Scholar] [CrossRef] [PubMed]
- Lusis, A.J. Genetics of atherosclerosis. Trends Genet. 2012, 28, 267–275. [Google Scholar] [CrossRef]
- Daneman, R.; Prat, A. The blood-brain barrier. Cold Spring Harb Perspect Biol. 2015, 7, a020412. [Google Scholar] [CrossRef]
- Skoog I, Wallin A, Fredman P, Hesse C, Aevarsson O, Karlsson I, et al. A population study on blood-brain barrier function in 85-year-olds: relation to Alzheimer's disease and vascular dementia. Neurology. 1998, 50, 966–971. [Google Scholar] [CrossRef]
- Montagne A, Barnes SR, Sweeney MD, Halliday MR, Sagare AP, Zhao Z, et al. Blood-brain barrier breakdown in the aging human hippocampus. Neuron. 2015, 85, 296–302. [Google Scholar] [CrossRef]
- Kurz, C.; Walker, L.; Rauchmann, B.S.; Perneczky, R. Dysfunction of the blood-brain barrier in Alzheimer's disease: Evidence from human studies. Neuropathol Appl Neurobiol. 2022, 48, e12782. [Google Scholar] [CrossRef]
- Montagne, A.; Nation, D.A.; Pa, J.; Sweeney, M.D.; Toga, A.W.; Zlokovic, B.V. Brain imaging of neurovascular dysfunction in Alzheimer's disease. Acta Neuropathol. 2016, 131, 687–707. [Google Scholar]
- van de Haar HJ, Burgmans S, Jansen JF, van Osch MJ, van Buchem MA, Muller M, et al. Blood-Brain Barrier Leakage in Patients with Early Alzheimer Disease. Radiology. 2016, 281, 527–535. [Google Scholar] [CrossRef]
- Muoio, V.; Persson, P.B.; Sendeski, M.M. The neurovascular unit - concept review. Acta Physiol (Oxf). 2014, 210, 790–798. [Google Scholar] [CrossRef]
- Bell, A.H.; Miller, S.L.; Castillo-Melendez, M.; Malhotra, A. The Neurovascular Unit: Effects of Brain Insults During the Perinatal Period. Front Neurosci. 2019, 13, 1452. [Google Scholar] [CrossRef]
- van de Haar HJ, Jansen JFA, van Osch MJP, van Buchem MA, Muller M, Wong SM, et al. Neurovascular unit impairment in early Alzheimer's disease measured with magnetic resonance imaging. Neurobiol Aging. 2016, 45, 190–196. [Google Scholar] [CrossRef] [PubMed]
- Bracko, O.; Cruz Hernández, J.C.; Park, L.; Nishimura, N.; Schaffer, C.B. Causes and consequences of baseline cerebral blood flow reductions in Alzheimer's disease. J Cereb Blood Flow Metab. 2021, 41, 1501–1516. [Google Scholar] [CrossRef] [PubMed]
- Ali M, Falkenhain K, Njiru BN, Murtaza-Ali M, Ruiz-Uribe NE, Haft-Javaherian M, et al. VEGF signalling causes stalls in brain capillaries and reduces cerebral blood flow in Alzheimer's mice. Brain. 2022, 145, 1449–1463. [Google Scholar] [CrossRef] [PubMed]
- Bell RD, Winkler EA, Singh I, Sagare AP, Deane R, Wu Z, et al. Apolipoprotein E controls cerebrovascular integrity via cyclophilin A. Nature. 2012, 485, 512–516. [Google Scholar] [CrossRef] [PubMed]
- Sagare, A.P.; Bell, R.D.; Zlokovic, B.V. Neurovascular defects and faulty amyloid-β vascular clearance in Alzheimer's disease. J Alzheimers Dis. 2013, 33 Suppl. 1, S87–S100. [Google Scholar] [CrossRef]
- Montagne A, Nation DA, Sagare AP, Barisano G, Sweeney MD, Chakhoyan A, et al. APOE4 leads to blood-brain barrier dysfunction predicting cognitive decline. Nature. 2020, 581, 71–76. [Google Scholar] [CrossRef]
- Zlokovic, B.V. Neurovascular pathways to neurodegeneration in Alzheimer's disease and other disorders. Nat Rev Neurosci. 2011, 12, 723–738. [Google Scholar] [CrossRef]
- Nelson, A.R.; Sweeney, M.D.; Sagare, A.P.; Zlokovic, B.V. Neurovascular dysfunction and neurodegeneration in dementia and Alzheimer's disease. Biochim Biophys Acta. 2016, 1862, 887–900. [Google Scholar] [PubMed]
- Vogelgesang S, Warzok RW, Cascorbi I, Kunert-Keil C, Schroeder E, Kroemer HK, et al. The role of P-glycoprotein in cerebral amyloid angiopathy; implications for the early pathogenesis of Alzheimer's disease. Curr Alzheimer Res. 2004, 1, 121–125. [Google Scholar] [CrossRef] [PubMed]
- van Assema DM, Lubberink M, Bauer M, van der Flier WM, Schuit RC, Windhorst AD, et al. Blood-brain barrier P-glycoprotein function in Alzheimer's disease. Brain. 2012, 135, 181–189. [Google Scholar] [CrossRef] [PubMed]
- Pathan, N.; Shende, P. Tailoring of P-glycoprotein for effective transportation of actives across blood-brain-barrier. J Control Release. 2021, 335, 398–407. [Google Scholar] [CrossRef]
- Deo AK, Borson S, Link JM, Domino K, Eary JF, Ke B, et al. Activity of P-Glycoprotein, a β-Amyloid Transporter at the Blood-Brain Barrier, Is Compromised in Patients with Mild Alzheimer Disease. J Nucl Med. 2014, 55, 1106–1111. [Google Scholar] [CrossRef]
- Maeng HJ, Kim MH, Jin HE, Shin SM, Tsuruo T, Kim SG, et al. Functional induction of P-glycoprotein in the blood-brain barrier of streptozotocin-induced diabetic rats: evidence for the involvement of nuclear factor-kappaB, a nitrosative stress-sensitive transcription factor, in the regulation. Drug Metab Dispos. 2007, 35, 1996–2005. [Google Scholar] [CrossRef]
- Ueno M, Nakagawa T, Huang CL, Ueki M, Kusaka T, Hosomi N, et al. The expression of P-glycoprotein is increased in vessels with blood-brain barrier impairment in a stroke-prone hypertensive model. Neuropathol Appl Neurobiol. 2009, 35, 147–155. [Google Scholar] [CrossRef]
- Gong, M.; Jia, J. Contribution of blood-brain barrier-related blood-borne factors for Alzheimer's disease vs. vascular dementia diagnosis: A pilot study. Front Neurosci. 2022, 16, 949129. [Google Scholar]
- Hachinski VC, Iliff LD, Zilhka E, Du Boulay GH, McAllister VL, Marshall J, et al. Cerebral blood flow in dementia. Arch Neurol. 1975, 32, 632–637. [Google Scholar] [CrossRef]
- Beason-Held, L.L.; Moghekar, A.; Zonderman, A.B.; Kraut, M.A.; Resnick, S.M. Longitudinal changes in cerebral blood flow in the older hypertensive brain. Stroke. 2007, 38, 1766–1773. [Google Scholar] [CrossRef]
- Korte, N.; Nortley, R.; Attwell, D. Cerebral blood flow decrease as an early pathological mechanism in Alzheimer's disease. Acta Neuropathol. 2020, 140, 793–810. [Google Scholar]
- Cohen, R.A. Hypertension and cerebral blood flow: implications for the development of vascular cognitive impairment in the elderly. Stroke. 2007, 38, 1715–1717. [Google Scholar] [PubMed]
- Román, G.C. Brain hypoperfusion: a critical factor in vascular dementia. Neurol Res. 2004, 26, 454–458. [Google Scholar] [CrossRef] [PubMed]
- Schuff N, Matsumoto S, Kmiecik J, Studholme C, Du A, Ezekiel F, et al. Cerebral blood flow in ischemic vascular dementia and Alzheimer's disease, measured by arterial spin-labeling magnetic resonance imaging. Alzheimers Dement. 2009, 5, 454–462. [Google Scholar]
- Yao, H.; Sadoshima, S.; Kuwabara, Y.; Ichiya, Y.; Fujishima, M. Cerebral blood flow and oxygen metabolism in patients with vascular dementia of the Binswanger type. Stroke. 1990, 21, 1694–1699. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.Z.; Zhang, J.J.; Liu, H.; Wu, G.Y.; Xiong, L.; Shu, M. Regional cerebral blood flow and cerebrovascular reactivity in Alzheimer's disease and vascular dementia assessed by arterial spinlabeling magnetic resonance imaging. Curr Neurovasc Res. 2013, 10, 49–53. [Google Scholar] [CrossRef] [PubMed]
- Sweeney, M.D.; Kisler, K.; Montagne, A.; Toga, A.W.; Zlokovic, B.V. The role of brain vasculature in neurodegenerative disorders. Nat Neurosci. 2018, 21, 1318–1331. [Google Scholar]
- Yoshikawa T, Murase K, Oku N, Imaizumi M, Takasawa M, Rishu P, et al. Heterogeneity of cerebral blood flow in Alzheimer disease and vascular dementia. AJNR Am J Neuroradiol. 2003, 24, 1341–1347. [Google Scholar]
- Kisler, K.; Nelson, A.R.; Montagne, A.; Zlokovic, B.V. Cerebral blood flow regulation and neurovascular dysfunction in Alzheimer disease. Nat Rev Neurosci. 2017, 18, 419–434. [Google Scholar]
- Enciu, A.M.; Constantinescu, S.N.; Popescu, L.M.; Mureşanu, D.F.; Popescu, B.O. Neurobiology of vascular dementia. J Aging Res. 2011, 2011, 401604. [Google Scholar] [CrossRef]
- Starkstein SE, Sabe L, Vazquez S, Teson A, Petracca G, Chemerinski E, et al. Neuropsychological, psychiatric, and cerebral blood flow findings in vascular dementia and Alzheimer's disease. Stroke. 1996, 27, 408–414. [Google Scholar] [CrossRef] [PubMed]
- Yang, D.W.; Kim, B.S.; Park, J.K.; Kim, S.Y.; Kim, E.N.; Sohn, H.S. Analysis of cerebral blood flow of subcortical vascular dementia with single photon emission computed tomography: adaptation of statistical parametric mapping. J Neurol Sci. 2002, 203-204, 199–205. [Google Scholar] [CrossRef] [PubMed]
- Thambisetty M, Beason-Held L, An Y, Kraut MA, Resnick SM. APOE epsilon4 genotype and longitudinal changes in cerebral blood flow in normal aging. Arch Neurol. 2010, 67, 93–98. [Google Scholar]
- Filippini N, Ebmeier KP, MacIntosh BJ, Trachtenberg AJ, Frisoni GB, Wilcock GK, et al. Differential effects of the APOE genotype on brain function across the lifespan. Neuroimage. 2011, 54, 602–610. [Google Scholar] [CrossRef] [PubMed]
- Michels L, Warnock G, Buck A, Macauda G, Leh SE, Kaelin AM, et al. Arterial spin labeling imaging reveals widespread and Aβ-independent reductions in cerebral blood flow in elderly apolipoprotein epsilon-4 carriers. J Cereb Blood Flow Metab. 2016, 36, 581–595. [Google Scholar] [CrossRef]
- McKiernan EF, Mak E, Dounavi ME, Wells K, Ritchie C, Williams G, et al. Regional hyperperfusion in cognitively normal APOE ε4 allele carriers in mid-life: analysis of ASL pilot data from the PREVENT-Dementia cohort. J Neurol Neurosurg Psychiatry. 2020, 91, 861–866. [Google Scholar] [CrossRef]
- Tomimoto, H.; Ohtani, R.; Shibata, M.; Nakamura, N.; Ihara, M. Loss of cholinergic pathways in vascular dementia of the Binswanger type. Dement Geriatr Cogn Disord. 2005, 19, 282–288. [Google Scholar] [CrossRef]
- Santos, C.Y.; Snyder, P.J.; Wu, W.C.; Zhang, M.; Echeverria, A.; Alber, J. Pathophysiologic relationship between Alzheimer's disease, cerebrovascular disease, and cardiovascular risk: A review and synthesis. Alzheimers Dement (Amst). 2017, 7, 69–87. [Google Scholar]
- Kalaria, R. Similarities between Alzheimer's disease and vascular dementia. J Neurol Sci. 2002, 203-204, 29–34. [Google Scholar] [CrossRef]
- Kozberg, M.G.; Perosa, V.; Gurol, M.E.; van Veluw, S.J. A practical approach to the management of cerebral amyloid angiopathy. Int J Stroke. 2021, 16, 356–369. [Google Scholar] [CrossRef]
- Jellinger, K.A. Alzheimer disease and cerebrovascular pathology: an update. J Neural Transm (Vienna). 2002, 109, 813–836. [Google Scholar] [CrossRef] [PubMed]
- Attems, J.; Jellinger, K.; Thal, D.R.; Van Nostrand, W. Review: sporadic cerebral amyloid angiopathy. Neuropathol Appl Neurobiol. 2011, 37, 75–93. [Google Scholar] [PubMed]
- Hecht, M.; Krämer, L.M.; von Arnim, C.A.F.; Otto, M.; Thal, D.R. Capillary cerebral amyloid angiopathy in Alzheimer's disease: association with allocortical/hippocampal microinfarcts and cognitive decline. Acta Neuropathol. 2018, 135, 681–694. [Google Scholar] [PubMed]
- Yamada, M.; Naiki, H. Cerebral amyloid angiopathy. Prog Mol Biol Transl Sci. 2012, 107, 41–78. [Google Scholar]
- Attems, J.; Jellinger, K.A.; Lintner, F. Alzheimer's disease pathology influences severity and topographical distribution of cerebral amyloid angiopathy. Acta Neuropathol. 2005, 110, 222–231. [Google Scholar] [PubMed]
- Pezzini, A.; Del Zotto, E.; Volonghi, I.; Giossi, A.; Costa, P.; Padovani, A. Cerebral amyloid angiopathy: a common cause of cerebral hemorrhage. Curr Med Chem. 2009, 16, 2498–2513. [Google Scholar] [CrossRef]
- Tian, J.; Shi, J.; Bailey, K.; Mann, D.M. Negative association between amyloid plaques and cerebral amyloid angiopathy in Alzheimer's disease. Neurosci Lett. 2003, 352, 137–140. [Google Scholar] [CrossRef]
- Masuda, J.; Tanaka, K.; Ueda, K.; Omae, T. Autopsy study of incidence and distribution of cerebral amyloid angiopathy in Hisayama, Japan. Stroke. 1988, 19, 205–210. [Google Scholar]
- Xu, D.; Yang, C.; Wang, L. Cerebral amyloid angiopathy in aged Chinese: a clinico-neuropathological study. Acta Neuropathol. 2003, 106, 89–91. [Google Scholar] [CrossRef]
- Haglund, M.; Sjöbeck, M.; Englund, E. Severe cerebral amyloid angiopathy characterizes an underestimated variant of vascular dementia. Dement Geriatr Cogn Disord. 2004, 18, 132–137. [Google Scholar] [CrossRef]
- Rannikmäe, K.; Samarasekera, N.; Martînez-Gonzâlez, N.A.; Al-Shahi Salman, R.; Sudlow, C.L. Genetics of cerebral amyloid angiopathy: systematic review and meta-analysis. J Neurol Neurosurg Psychiatry. 2013, 84, 901–908. [Google Scholar] [CrossRef] [PubMed]
- Yamada, M. Cerebral amyloid angiopathy: emerging concepts. J Stroke. 2015, 17, 17–30. [Google Scholar] [CrossRef] [PubMed]
- Verghese, P.B.; Castellano, J.M.; Holtzman, D.M. Apolipoprotein E in Alzheimer's disease and other neurological disorders. Lancet Neurol. 2011, 10, 241–252. [Google Scholar] [CrossRef] [PubMed]
- Nicoll JA, Burnett C, Love S, Graham DI, Dewar D, Ironside JW, et al. High frequency of apolipoprotein E epsilon 2 allele in hemorrhage due to cerebral amyloid angiopathy. Ann Neurol. 1997, 41, 716–721. [Google Scholar] [CrossRef]
- Biffi A, Shulman JM, Jagiella JM, Cortellini L, Ayres AM, Schwab K, et al. Genetic variation at CR1 increases risk of cerebral amyloid angiopathy. Neurology. 2012, 78, 334–341. [Google Scholar] [CrossRef]
- Pandey, P.; Pradhan, S.; Mittal, B. Presenilin gene predisposes to late-onset degenerative but not vascular dementia: a comparative study of PS1 and ApoE genes in a North Indian Cohort. Dement Geriatr Cogn Disord. 2007, 24, 151–161. [Google Scholar] [CrossRef]
- Biffi, A.; Greenberg, S.M. Cerebral amyloid angiopathy: a systematic review. J Clin Neurol. 2011, 7, 1–9. [Google Scholar] [CrossRef]
- Van der Flier, W.M.; Cordonnier, C. Microbleeds in vascular dementia: clinical aspects. Exp Gerontol. 2012, 47, 853–857. [Google Scholar] [CrossRef]
- Greenberg SM, Vernooij MW, Cordonnier C, Viswanathan A, Al-Shahi Salman R, Warach S, et al. Cerebral microbleeds: a guide to detection and interpretation. Lancet Neurol. 2009, 8, 165–174. [Google Scholar] [CrossRef]
- Cordonnier, C.; van der Flier, W.M.; Sluimer, J.D.; Leys, D.; Barkhof, F.; Scheltens, P. Prevalence and severity of microbleeds in a memory clinic setting. Neurology. 2006, 66, 1356–1360. [Google Scholar] [CrossRef]
- Brundel M, Heringa SM, de Bresser J, Koek HL, Zwanenburg JJ, Jaap Kappelle L, et al. High prevalence of cerebral microbleeds at 7Tesla MRI in patients with early Alzheimer's disease. J Alzheimers Dis. 2012, 31, 259–263. [Google Scholar] [CrossRef] [PubMed]
- Yates, P.A.; Villemagne, V.L.; Ellis, K.A.; Desmond, P.M.; Masters, C.L.; Rowe, C.C. Cerebral microbleeds: a review of clinical, genetic, and neuroimaging associations. Front Neurol. 2014, 4, 205. [Google Scholar] [CrossRef] [PubMed]
- De Reuck J, Auger F, Durieux N, Deramecourt V, Cordonnier C, Pasquier F, et al. Topography of Cortical Microbleeds in Alzheimer's Disease with and without Cerebral Amyloid Angiopathy: A Post-Mortem 7.0-Tesla MRI Study. Aging Dis. 2015, 6, 437–443. [Google Scholar] [CrossRef]
- Shams S, Martola J, Granberg T, Li X, Shams M, Fereshtehnejad SM, et al. Cerebral microbleeds: different prevalence, topography, and risk factors depending on dementia diagnosis—the Karolinska Imaging Dementia Study. AJNR Am J Neuroradiol. 2015, 36, 661–666. [Google Scholar] [CrossRef]
- Pettersen JA, Sathiyamoorthy G, Gao FQ, Szilagyi G, Nadkarni NK, St George-Hyslop P, et al. Microbleed topography, leukoaraiosis, and cognition in probable Alzheimer disease from the Sunnybrook dementia study. Arch Neurol. 2008, 65, 790–795. [Google Scholar]
- Dichgans, M.; Holtmannspötter, M.; Herzog, J.; Peters, N.; Bergmann, M.; Yousry, T.A. Cerebral microbleeds in CADASIL: a gradient-echo magnetic resonance imaging and autopsy study. Stroke. 2002, 33, 67–71. [Google Scholar] [CrossRef]
- De Reuck J, Auger F, Durieux N, Deramecourt V, Maurage CA, Cordonnier C, et al. Frequency and topography of small cerebrovascular lesions in vascular and in mixed dementia: a post-mortem 7-tesla magnetic resonance imaging study with neuropathological correlates. Folia Neuropathol. 2017, 55, 31–37. [Google Scholar]
- Maxwell SS, Jackson CA, Paternoster L, Cordonnier C, Thijs V, Al-Shahi Salman R, et al. Genetic associations with brain microbleeds: Systematic review and meta-analyses. Neurology. 2011, 77, 158–167. [Google Scholar] [CrossRef]
- Vernooij MW, van der Lugt A, Ikram MA, Wielopolski PA, Niessen WJ, Hofman A, et al. Prevalence and risk factors of cerebral microbleeds: the Rotterdam Scan Study. Neurology. 2008, 70, 1208–1214. [Google Scholar] [CrossRef]
- Caplan, L.R. Lacunar infarction and small vessel disease: pathology and pathophysiology. J Stroke. 2015, 17, 2–6. [Google Scholar] [CrossRef]
- Cai Z, Wang C, He W, Tu H, Tang Z, Xiao M, et al. Cerebral small vessel disease and Alzheimer's disease. Clin Interv Aging. 2015, 10, 1695–1704. [Google Scholar]
- Attems, J.; Jellinger, K.A. The overlap between vascular disease and Alzheimer's disease--lessons from pathology. BMC Med. 2014, 12, 206. [Google Scholar] [CrossRef]
- Snowdon, D.A.; Greiner, L.H.; Mortimer, J.A.; Riley, K.P.; Greiner, P.A.; Markesbery, W.R. Brain infarction and the clinical expression of Alzheimer disease. The Nun Study. Jama. 1997, 277, 813–817. [Google Scholar] [CrossRef] [PubMed]
- De Reuck J, Auger F, Durieux N, Cordonnier C, Deramecourt V, Pasquier F, et al. Topographic distribution of white matter changes and lacunar infarcts in neurodegenerative and vascular dementia syndromes: A post-mortem 7.0-tesla magnetic resonance imaging study. Eur Stroke J. 2016, 1, 122–129. [Google Scholar] [CrossRef]
- Uchino, M. [The pathomechanism and treatment of CADASIL]. Rinsho Shinkeigaku. 2011, 51, 945–948. [Google Scholar] [CrossRef]
- Traylor M, Adib-Samii P, Harold D, Dichgans M, Williams J, Lewis CM, et al. Shared genetic contribution to Ischaemic Stroke and Alzheimer's Disease. Ann Neurol. 2016, 79, 739–747. [Google Scholar] [CrossRef] [PubMed]
- Liu Y, Laakso MP, Karonen JO, Vanninen RL, Nuutinen J, Soimakallio S, et al. Apolipoprotein E polymorphism and acute ischemic stroke: a diffusion- and perfusion-weighted magnetic resonance imaging study. J Cereb Blood Flow Metab. 2002, 22, 1336–1342. [Google Scholar] [CrossRef]
- Chabriat H, Joutel A, Dichgans M, Tournier-Lasserve E, Bousser MG. Cadasil. Lancet Neurol. 2009, 8, 643–653.
- Tikka S, Baumann M, Siitonen M, Pasanen P, Pöyhönen M, Myllykangas L, et al. CADASIL and CARASIL. Brain Pathol. 2014, 24, 525–544. [Google Scholar] [CrossRef]
- Khandelwal D, Mathur V, Vyas A, Ghunawat J, Bagaria AK. CARASIL - A Review of Patients from India. Neurol India. 2021, 69, 1359–1362. [Google Scholar]
- Brundel, M.; de Bresser, J.; van Dillen, J.J.; Kappelle, L.J.; Biessels, G.J. Cerebral microinfarcts: a systematic review of neuropathological studies. J Cereb Blood Flow Metab. 2012, 32, 425–436. [Google Scholar] [CrossRef]
- Hartmann DA, Hyacinth HI, Liao FF, Shih AY. Does pathology of small venules contribute to cerebral microinfarcts and dementia? J Neurochem. 2018, 144, 517–526. [CrossRef]
- Arvanitakis, Z.; Leurgans, S.E.; Barnes, L.L.; Bennett, D.A.; Schneider, J.A. Microinfarct pathology, dementia, and cognitive systems. Stroke. 2011, 42, 722–727. [Google Scholar] [CrossRef]
- Corrada, M.M.; Sonnen, J.A.; Kim, R.C.; Kawas, C.H. Microinfarcts are common and strongly related to dementia in the oldest-old: The 90+ study. Alzheimers Dement. 2016, 12, 900–908. [Google Scholar] [CrossRef]
- Smith, E.E.; Schneider, J.A.; Wardlaw, J.M.; Greenberg, S.M. Cerebral microinfarcts: the invisible lesions. Lancet Neurol. 2012, 11, 272–282. [Google Scholar] [CrossRef] [PubMed]
- Okamoto, Y.; Ihara, M.; Fujita, Y.; Ito, H.; Takahashi, R.; Tomimoto, H. Cortical microinfarcts in Alzheimer's disease and subcortical vascular dementia. Neuroreport. 2009, 20, 990–996. [Google Scholar] [CrossRef] [PubMed]
- Rost, N.S.; Etherton, M. Cerebral Small Vessel Disease. Continuum (Minneap Minn). 2020, 26, 332–352. [Google Scholar] [CrossRef] [PubMed]
- Vinke EJ, Yilmaz P, van der Toorn JE, Fakhry R, Frenzen K, Dubost F, et al. Intracranial arteriosclerosis is related to cerebral small vessel disease: a prospective cohort study. Neurobiol Aging. 2021, 105, 16–24. [Google Scholar] [CrossRef]
- O'Sullivan, M. Leukoaraiosis. Pract Neurol. 2008, 8, 26–38. [Google Scholar] [CrossRef] [PubMed]
- van Gijn, J. Leukoaraiosis and vascular dementia. Neurology. 1998, 51, S3–S8. [Google Scholar] [CrossRef]
- Englund, E. Neuropathology of white matter changes in Alzheimer's disease and vascular dementia. Dement Geriatr Cogn Disord. 1998, 9 Suppl. 1, 6–12. [Google Scholar] [CrossRef] [PubMed]
- Blennow, K.; Wallin, A.; Uhlemann, C.; Gottfries, C.G. White-matter lesions on CT in Alzheimer patients: relation to clinical symptomatology and vascular factors. Acta Neurol Scand. 1991, 83, 187–193. [Google Scholar] [CrossRef]
- Erkinjuntti T, Ketonen L, Sulkava R, Sipponen J, Vuorialho M, Iivanainen M. Do white matter changes on MRI and CT differentiate vascular dementia from Alzheimer's disease? J Neurol Neurosurg Psychiatry. 1987, 50, 37–42.
- Bozzali M, Falini A, Franceschi M, Cercignani M, Zuffi M, Scotti G, et al. White matter damage in Alzheimer's disease assessed in vivo using diffusion tensor magnetic resonance imaging. J Neurol Neurosurg Psychiatry. 2002, 72, 742–746. [Google Scholar] [CrossRef] [PubMed]
- Barber R, Scheltens P, Gholkar A, Ballard C, McKeith I, Ince P, et al. White matter lesions on magnetic resonance imaging in dementia with Lewy bodies, Alzheimer's disease, vascular dementia, and normal aging. J Neurol Neurosurg Psychiatry. 1999, 67, 66–72. [Google Scholar] [CrossRef] [PubMed]
- Szolnoki, Z. Pathomechanism of leukoaraiosis: a molecular bridge between the genetic, biochemical, and clinical processes (a mitochondrial hypothesis). Neuromolecular Med. 2007, 9, 21–33. [Google Scholar] [CrossRef]
- Bronge L, Fernaeus SE, Blomberg M, Ingelson M, Lannfelt L, Isberg B, et al. White matter lesions in Alzheimer patients are influenced by apolipoprotein E genotype. Dement Geriatr Cogn Disord. 1999, 10, 89–96. [Google Scholar] [CrossRef]
- Schmidt R, Schmidt H, Fazekas F, Schumacher M, Niederkorn K, Kapeller P, et al. Apolipoprotein E polymorphism and silent microangiopathy-related cerebral damage. Results of the Austrian Stroke Prevention Study. Stroke. 1997, 28, 951–956. [Google Scholar] [CrossRef]
- Szolnoki, Z.; Somogyvári, F.; Kondacs, A.; Szabó, M.; Fodor, L. Evaluation of the roles of common genetic mutations in leukoaraiosis. Acta Neurol Scand. 2001, 104, 281–287. [Google Scholar] [CrossRef]
- Dichgans, M. CADASIL: a monogenic condition causing stroke and subcortical vascular dementia. Cerebrovasc Dis. 2002, 13 Suppl 2, 37–41. [Google Scholar] [CrossRef]
- Tariq, S.; Barber, P.A. Dementia risk and prevention by targeting modifiable vascular risk factors. J Neurochem. 2018, 144, 565–581. [Google Scholar] [CrossRef] [PubMed]
- Perl, D.P. Neuropathology of Alzheimer's disease. Mt Sinai J Med. 2010, 77, 32–42. [Google Scholar] [CrossRef] [PubMed]
- Serrano-Pozo, A.; Frosch, M.P.; Masliah, E.; Hyman, B.T. Neuropathological alterations in Alzheimer disease. Cold Spring Harb Perspect Med. 2011, 1, a006189. [Google Scholar] [CrossRef] [PubMed]
- Enkirch SJ, Traschütz A, Müller A, Widmann CN, Gielen GH, Heneka MT, et al. The ERICA Score: An MR Imaging-based Visual Scoring System for the Assessment of Entorhinal Cortex Atrophy in Alzheimer Disease. Radiology. 2018, 288, 226–333. [Google Scholar] [CrossRef] [PubMed]
- Koval I, Schiratti JB, Routier A, Bacci M, Colliot O, Allassonnière S, et al. Spatiotemporal Propagation of the Cortical Atrophy: Population and Individual Patterns. Front Neurol. 2018, 9, 235. [Google Scholar] [CrossRef]
- Koval I, Bône A, Louis M, Lartigue T, Bottani S, Marcoux A, et al. AD Course Map charts Alzheimer's disease progression. Sci Rep. 2021, 11, 8020. [Google Scholar] [CrossRef]
- O'Brien JT, Paling S, Barber R, Williams ED, Ballard C, McKeith IG, et al. Progressive brain atrophy on serial MRI in dementia with Lewy bodies, AD, and vascular dementia. Neurology. 2001, 56, 1386–1388. [Google Scholar] [CrossRef]
- van de Pol L, Gertz HJ, Scheltens P, Wolf H. Hippocampal atrophy in subcortical vascular dementia. Neurodegener Dis. 2011, 8, 465–469. [Google Scholar] [CrossRef]
- Kril, J.J.; Patel, S.; Harding, A.J.; Halliday, G.M. Patients with vascular dementia due to microvascular pathology have significant hippocampal neuronal loss. J Neurol Neurosurg Psychiatry. 2002, 72, 747–751. [Google Scholar] [CrossRef]
- Duering M, Righart R, Csanadi E, Jouvent E, Hervé D, Chabriat H, et al. Incident subcortical infarcts induce focal thinning in connected cortical regions. Neurology. 2012, 79, 2025–2028. [Google Scholar] [CrossRef]
- Seo SW, Lee JM, Im K, Park JS, Kim SH, Kim ST, et al. Cortical thinning related to periventricular and deep white matter hyperintensities. Neurobiol Aging. 2012, 33, 1156–1167. [Google Scholar] [CrossRef]
- Kim CH, Seo SW, Kim GH, Shin JS, Cho H, Noh Y, et al. Cortical thinning in subcortical vascular dementia with negative 11C-PiB PET. J Alzheimers Dis. 2012, 31, 315–323. [Google Scholar] [CrossRef] [PubMed]
- Vinters HV, Ellis WG, Zarow C, Zaias BW, Jagust WJ, Mack WJ, et al. Neuropathologic substrates of ischemic vascular dementia. J Neuropathol Exp Neurol. 2000, 59, 931–945. [Google Scholar] [CrossRef]
- Mann, D.M.; Yates, P.O.; Marcyniuk, B. The nucleus basalis of Meynert in multi-infarct (vascular) dementia. Acta Neuropathol. 1986, 71, 332–337. [Google Scholar] [CrossRef] [PubMed]
- Foster V, Oakley AE, Slade JY, Hall R, Polvikoski TM, Burke M, et al. Pyramidal neurons of the prefrontal cortex in post-stroke, vascular and other ageing-related dementias. Brain. 2014, 137, 2509–2521. [Google Scholar] [CrossRef] [PubMed]
- Bartsch, T.; Wulff, P. The hippocampus in aging and disease: From plasticity to vulnerability. Neuroscience. 2015, 309, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Liu, A.K.; Chang, R.C.; Pearce, R.K.; Gentleman, S.M. Nucleus basalis of Meynert revisited: anatomy, history and differential involvement in Alzheimer's and Parkinson's disease. Acta Neuropathol. 2015, 129, 527–540. [Google Scholar] [CrossRef]
- Lyness, S.A.; Zarow, C.; Chui, H.C. Neuron loss in key cholinergic and aminergic nuclei in Alzheimer disease: a meta-analysis. Neurobiol Aging. 2003, 24, 1–23. [Google Scholar] [CrossRef]
- Gannon, M.; Che, P.; Chen, Y.; Jiao, K.; Roberson, E.D.; Wang, Q. Noradrenergic dysfunction in Alzheimer's disease. Front Neurosci. 2015, 9, 220. [Google Scholar] [CrossRef]
- Braak, H.; Thal, D.R.; Ghebremedhin, E.; Del Tredici, K. Stages of the pathologic process in Alzheimer disease: age categories from 1 to 100 years. J Neuropathol Exp Neurol. 2011, 70, 960–969. [Google Scholar] [CrossRef]
- Slater, C.; Wang, Q. Alzheimer's disease: An evolving understanding of noradrenergic involvement and the promising future of electroceutical therapies. Clin Transl Med. 2021, 11, e397. [Google Scholar] [CrossRef]
- Aaldijk, E.; Vermeiren, Y. The role of serotonin within the microbiota-gut-brain axis in the development of Alzheimer's disease: A narrative review. Ageing Res Rev. 2022, 75, 101556. [Google Scholar] [CrossRef] [PubMed]
- Gray, F.; Polivka, M.; Viswanathan, A.; Baudrimont, M.; Bousser, M.G.; Chabriat, H. Apoptosis in cerebral autosomal-dominant arteriopathy with subcortical infarcts and leukoencephalopathy. J Neuropathol Exp Neurol. 2007, 66, 597–607. [Google Scholar] [CrossRef] [PubMed]
- Sheppard, O.; Coleman, M. Alzheimer’s Disease: Etiology, Neuropathology and Pathogenesis. In: Huang X, editor. Alzheimer’s Disease: Drug Discovery. Brisbane (AU): Exon Publications Copyright: The Authors.; 2020.
- Rao, Y.L.; Ganaraja, B.; Murlimanju, B.V.; Joy, T.; Krishnamurthy, A.; Agrawal, A. Hippocampus and its involvement in Alzheimer's disease: a review. 3 Biotech. 2022, 12, 55. [Google Scholar] [CrossRef] [PubMed]
- Asai H, Ikezu S, Tsunoda S, Medalla M, Luebke J, Haydar T, et al. Depletion of microglia and inhibition of exosome synthesis halt tau propagation. Nat Neurosci. 2015, 18, 1584–1593. [Google Scholar] [CrossRef] [PubMed]
- Rauchmann BS, Brendel M, Franzmeier N, Trappmann L, Zaganjori M, Ersoezlue E, et al. Microglial Activation and Connectivity in Alzheimer Disease and Aging. Ann Neurol. 2022, 92, 768–781. [Google Scholar] [CrossRef]
- Xiang X, Wind K, Wiedemann T, Blume T, Shi Y, Briel N, et al. Microglial activation states drive glucose uptake and FDG-PET alterations in neurodegenerative diseases. Sci Transl Med. 2021, 13, eabe5640. [CrossRef]
- Kirvell, S.L.; Elliott, M.S.; Kalaria, R.N.; Hortobágyi, T.; Ballard, C.G.; Francis, P.T. Vesicular glutamate transporter and cognition in stroke: a case-control autopsy study. Neurology. 2010, 75, 1803–1809. [Google Scholar] [CrossRef]
- Benarroch, E.E. Glutamatergic synaptic plasticity and dysfunction in Alzheimer disease: Emerging mechanisms. Neurology. 2018, 91, 125–132. [Google Scholar] [CrossRef]
- Prokšelj, T.; Jerin, A.; Muck-Seler, D.; Kogoj, A. Decreased platelet serotonin concentration in Alzheimer's disease with involuntary emotional expression disorder. Neurosci Lett. 2014, 578, 71–74. [Google Scholar] [CrossRef]
- Perry, E.; Ziabreva, I.; Perry, R.; Aarsland, D.; Ballard, C. Absence of cholinergic deficits in "pure" vascular dementia. Neurology. 2005, 64, 132–133. [Google Scholar] [CrossRef] [PubMed]
- Keverne, J.S.; Low, W.C.; Ziabreva, I.; Court, J.A.; Oakley, A.E.; Kalaria, R.N. Cholinergic neuronal deficits in CADASIL. Stroke. 2007, 38, 188–191. [Google Scholar] [CrossRef] [PubMed]
- Selkoe, D.J.; Podlisny, M.B. Deciphering the genetic basis of Alzheimer's disease. Annu Rev Genomics Hum Genet. 2002, 3, 67–99. [Google Scholar] [CrossRef] [PubMed]
- Hu Z, Hu K, Wang R, Gu Y, Ouyang W, Zhou J, et al. Differentially expressed genes accompanying neurobehavioral deficits in a modified rat model of vascular dementia. Neurosci Lett. 2021, 750, 135774. [Google Scholar] [CrossRef] [PubMed]
- Gooch, J.; Wilcock, D.M. Animal Models of Vascular Cognitive Impairment and Dementia (VCID). Cell Mol Neurobiol. 2016, 36, 233–239. [Google Scholar] [CrossRef]
- Smith, M.Z.; Nagy, Z.; Esiri, M.M. Cell cycle-related protein expression in vascular dementia and Alzheimer's disease. Neurosci Lett. 1999, 271, 45–48. [Google Scholar] [CrossRef]
- Kalaria, R.N.; Ballard, C. Overlap between pathology of Alzheimer disease and vascular dementia. Alzheimer Dis Assoc Disord. 1999, 13 Suppl 3, S115–S123. [Google Scholar] [CrossRef]
- van der Flier WM, Skoog I, Schneider JA, Pantoni L, Mok V, Chen CLH, et al. Vascular cognitive impairment. Nature Reviews Disease Primers. 2018, 4, 18003. [Google Scholar] [CrossRef]
- Kim SE, Kim HJ, Jang H, Weiner MW, DeCarli C, Na DL, et al. Interaction between Alzheimer's Disease and Cerebral Small Vessel Disease: A Review Focused on Neuroimaging Markers. Int J Mol Sci. 2022, 2022.
- Lewis H, Beher D, Cookson N, Oakley A, Piggott M, Morris CM, et al. Quantification of Alzheimer pathology in ageing and dementia: age-related accumulation of amyloid-beta(42) peptide in vascular dementia. Neuropathol Appl Neurobiol. 2006, 32, 103–118. [Google Scholar] [CrossRef]
- Noh Y, Seo SW, Jeon S, Lee JM, Kim JH, Kim GH, et al. White matter hyperintensities are associated with amyloid burden in APOE4 non-carriers. J Alzheimers Dis. 2014, 40, 877–886. [Google Scholar] [CrossRef]
- Frontzkowski L, Ewers M, Brendel M, Biel D, Ossenkoppele R, Hager P, et al. Earlier Alzheimer's disease onset is associated with tau pathology in brain hub regions and facilitated tau spreading. Nat Commun. 2022, 13, 4899. [Google Scholar] [CrossRef]
- van Norden AG, van Dijk EJ, de Laat KF, Scheltens P, Olderikkert MG, de Leeuw FE. Dementia: Alzheimer pathology and vascular factors: from mutually exclusive to interaction. Biochim Biophys Acta. 2012, 1822, 340–349. [Google Scholar] [CrossRef]
- Zhang X, Zhou K, Wang R, Cui J, Lipton SA, Liao FF, et al. Hypoxia-inducible factor 1alpha (HIF-1alpha)-mediated hypoxia increases BACE1 expression and beta-amyloid generation. J Biol Chem. 2007, 282, 10873–10880. [Google Scholar] [CrossRef]
- Pellerin, L. Food for thought: the importance of glucose and other energy substrates for sustaining brain function under varying levels of activity. Diabetes Metab. 2010, 2010 Suppl 3, S59–S63. [Google Scholar] [CrossRef]
- Heiss WD, Zimmermann-Meinzingen S. PET imaging in the differential diagnosis of vascular dementia. J Neurol Sci. 2012, 322, 268–273. [Google Scholar] [CrossRef]
- McKenna, M.C.; Schuck, P.F.; Ferreira, G.C. Fundamentals of CNS energy metabolism and alterations in lysosomal storage diseases. J Neurochem. 2019, 148, 590–599. [Google Scholar] [CrossRef]
- Kerrouche N, Herholz K, Mielke R, Holthoff V, Baron JC. 18FDG PET in vascular dementia: differentiation from Alzheimer's disease using voxel-based multivariate analysis. J Cereb Blood Flow Metab. 2006, 26, 1213–1221. [Google Scholar] [CrossRef]
- Seo, S.W.; Cho, S.S.; Park, A.; Chin, J.; Na, D.L. Subcortical vascular versus amnestic mild cognitive impairment: comparison of cerebral glucose metabolism. J Neuroimaging. 2009, 19, 213–219. [Google Scholar] [CrossRef]
- Cho, S.; Lee, H.; Seo, J. Impact of Genetic Risk Factors for Alzheimer's Disease on Brain Glucose Metabolism. Mol Neurobiol. 2021, 58, 2608–2619. [Google Scholar] [CrossRef]
- Jagust, W.J.; Landau, S.M. Apolipoprotein E, not fibrillar β-amyloid, reduces cerebral glucose metabolism in normal aging. J Neurosci. 2012, 32, 18227–18233. [Google Scholar] [CrossRef]
- Stefani A, Bernardini S, Panella M, Pierantozzi M, Nuccetelli M, Koch G, et al. AD with subcortical white matter lesions and vascular dementia: CSF markers for differential diagnosis. J Neurol Sci. 2005, 237, 83–88. [Google Scholar] [CrossRef]
- Congdon, E.E.; Sigurdsson, E.M. Tau-targeting therapies for Alzheimer disease. Nat Rev Neurol. 2018, 14, 399–415. [Google Scholar] [CrossRef]
- Weller, J.; Budson, A. Current understanding of Alzheimer's disease diagnosis and treatment. F1000Res. 2018, 7. [Google Scholar] [CrossRef]
- Pinheiro, L.; Faustino, C. Therapeutic Strategies Targeting Amyloid-β in Alzheimer's Disease. Curr Alzheimer Res. 2019, 16, 418–452. [Google Scholar] [CrossRef]
- Yarchoan M, Xie SX, Kling MA, Toledo JB, Wolk DA, Lee EB, et al. Cerebrovascular atherosclerosis correlates with Alzheimer pathology in neurodegenerative dementias. Brain. 2012, 135, 3749–3756. [Google Scholar] [CrossRef]
- Pasi M, Boulouis G, Fotiadis P, Auriel E, Charidimou A, Haley K, et al. Distribution of lacunes in cerebral amyloid angiopathy and hypertensive small vessel disease. Neurology. 2017, 88, 2162–2168. [Google Scholar] [CrossRef]
- Yip AG, McKee AC, Green RC, Wells J, Young H, Cupples LA, et al. APOE, vascular pathology, and the AD brain. Neurology. 2005, 65, 259–265. [Google Scholar] [CrossRef]
- Zarow, C.; Zaias, B.; Lyness, S.A.; Chui, H. Cerebral amyloid angiopathy in Alzheimer disease is associated with apolipoprotein E4 and cortical neuron loss. Alzheimer Dis Assoc Disord. 1999, 13, 1–8. [Google Scholar] [CrossRef]
 |
 |
 |
 |
 |
 |
 |
 |
 |
 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).




